1 Genetics and Bioengineering Program, Faculty of Engineering and Natural Sciences, International University of Sarajevo, Hrasnička cesta 15, Ilidža 71210 Sarajevo, Bosnia and Herzegovina
2 Gottfried Schatz Research Center for Cell Signaling, Metabolism and Aging, Medical University of Graz, Neue Stiftingtalstraße 6/6 8010 Graz Austria
3 Department of Biological Sciences, Middle East Technical University, Üniversiteler Mahallesi, Dumlupınar Bulvarı No:1 06800 Çankaya, Ankara, Turkey
Abstract
The γ-Aminobutyric acid type A receptors (GABAARs) are heteropentameric chloride channels responsible for primary inhibition in the mammalian brain. Studies have shown the expression of recombinant GABAAR subunits tagged with the green fluorescent protein (GFP), a 26.9 kDa protein that exhibits bright green fluorescence when exposed to light in the blue to ultraviolet range. This allows the formation of recombinant proteins essential for the development of relevant in-vitro and in-vivo methodologies. Among the GABAAR subunits, the δ subunit was never tagged in its cytoplasmic domain, an evolutionary conserved domain found in between the third and the fourth transmembrane domains. In this study, first, we have cloned the mouse cDNAs encoding for the δ, α1, β2 subunits of GABAARs, and then developed two fusion proteins of δ-subunit each tagged with the GFP variant, EGFP (enhanced GFP) at unique sites in the cytoplasmic domain. The recombinant proteins were expressed alone or in combination with α1 and/or β2 subunits in neuroblastoma 2a cells. Live cell confocal microscopy indicated that the cytoplasmically tagged δ-subunits were targeted to the cell membrane when expressed in the presence of α1 and β2 subunits in neuroblastoma 2a cells. However, this was not observed when they were expressed alone or only with α1 or β2 subunits in the same cell line. These results confirm the general oligomerization and targeting pattern of GABAAR subtypes described in the other in-vitro studies in the literature. Thus, our results suggest that the EGFP tagging in the ctoplasmic domain did not interfere with the oligomerization and cell surface expression of recombinant δ-subunits. To our knowledge, this is the first study showing the generation, expression and preliminary analysis of the δ-GABAARs tagged in the cytoplasmic domain of the δ-subunit which can be further elaborated to probe intracellular protein interactions of GABAARs via the δ-subunit.
Keywords
- GABAA receptor
- Cys-loop receptors
- ion channel
- delta subunit
- extrasynaptic
- recombinant protein expression
- EGFP
- oligomerization
- protein tagging
- fusion protein
- cDNA cloning
- TA cloning
- confocal microscopy
- live cell imaging
- fluorescence imaging
- neuroblastoma
The γ-Aminobutyric acid type A receptors (GABAARs) are GABA gated heteropentameric chloride channels and responsible for major inhibition in the brain. They belong to the superfamily of ligand-gated ion channels (“Cys-loop receptors”) including the nicotinic acetylcholine receptors (nAChR), the 5-hydroxytryptamine type 3 (5-HT3) receptors, and the glycine receptors (Sigel and Steinmann, 2012). Assembled from a large subunit pool (α1-α6, β1-β3, γ1-γ3, δ, ε, π, ρ1-ρ3), the GABAAR subunit composition determines the receptor’s distribution both in the cellular and sub-cellular level (Fritschy and Mohler, 1995; Sieghart and Sperk, 2002; Sun et al., 2004). Each subunit is about 450 amino acid residues in length and share a common topological structure, characterized by a long extracellular N-terminus and a short C- terminus, four transmembrane domains and one large intracellular or cytoplasmic domain located in between the 3rd and the 4th transmembrane domain. The topological organization is confirmed by structural studies of Cys-loop receptors, including the subunits of acetyl choline receptors and the recently published β3 subunit of the GABAAR (Miller and Aricescu, 2014; Unwin, 2013). On the other hand, structures of assembled heteropentameric GABAAR had been hard to pin down until recently, when several cryo-electron microscopy studies (Laverty et al., 2019; Masiulis et al., 2019; Phulera et al., 2018; Zhu et al., 2018), have led to the structural resolution of heteropentameric receptors.
Assembly, trafficking, clustering and stabilization of neurotransmitter receptors are essential for neural function and plasticity during health and disease (Anggono and Huganir, 2012; Mele et al., 2019). This phenomenon is especially critical for GABAARs: The inhibitory control of a massive excitatory tune is provided by only a relatively low number of GABAergic cells, which is in part compensated by the GABAARs, as these receptors are characterized by an unusual molecular and cellular diversity (Mody and Pearce 2004). However, that also comes with a cost: the analysis of this sophisticated molecular machinery for assembly, trafficking, clustering and stabilization of GABAARs is very complex.
Accumulating evidences suggest that the subunit domains including cytoplasmic domain of GABAAR subunit are the site of interaction with other proteins causing an impact on the state of the receptor for diverse functions (Arslan et al., 2014; Arslan, 2015a; Comenencia-Ortiz et al., 2014; Kittler and Moss, 2003; Krishek et al., 1994; Mukherjee et al., 2011). However, there is no direct evidence if all GABAARs show this pattern. For example, one GABAAR specific subtype, the δ-subunit containing GABAAR (δ-GABAAR) is not located in the synapses: Electron microscopy studies clearly show that these receptors are non-synaptic (Nusser et al., 1998; Wei et al., 2003). So called extra-synaptic receptors, these receptors specifically contain δ-subunit (Shivers et al., 1989; Sommer et al., 1990) together with 2 α and 2 β subunits (Patel et al., 2014). The in-situ hybridization analysis (Wisden et al., 1992) shows that, in the adult rat brain, δ-subunit is expressed weakly or moderately in the regions of the olfactory bulb (granule cells and periglomerular), neocortex (layers IV, V/VI and pyriform cortex), hippocampus (dentate gyrus granule cells, str. pyramidalis CA1 and str. pyramidalis CA3), basal ganglia (caudate, putamen, nucleous accumbens, claustrum), thalamus (medio dorsal, ventral posterior nucleous, medial-, dorso- and ventrolateral geniculate nucleous). In the cerebellum’s granule cells, this subunit shows very strong expression (Wisden et al., 1992). The δ-GABAARs have higher affinity for GABA, neurosteroids as well as for alcohol (Arslan, 2016; Joshi and Kapur, 2019) and mediate slow, tonic inhibition (Farrant and Nusser, 2005; Hausser and Clark, 1997; Semyanov et al., 2004). Tonic inhibition is a special form of inhibition, which is important for the threshold of action potential generation and thus critical for the neural excitability (reviewed in Arslan 2015a). This unique form of GABAergic inhibition mediated by δ-GABAARs are thought to be involved in the pathophysiology of several disorders such as epilepsy, schizophrenia, major depression, childhood onset mood disorders, alcohol use disorders, sleep disorders, premenstrual dysphoric disorder (Brickley and Mody, 2012).
The sequence alignment of δ-subunit revealed that the cytoplasmic domain is highly conserved across the whole span of vertebrate evolution (Arslan et al., 2014). This suggests a significant role of the cytoplasmic domain in the function of δ-GABAARs. However, direct experimental evidences are required to fulfill this hypothesis, which then requires the establishment of integrative methodologies. For example, the subunit of interest and/or (candidate) interacting protein may be identified by co-immunoprecipitation, yeast two hybrid or domain specific mutations. Then, the regulated subunit assembly, targeting, clustering and trafficking can be further studied within the specific context. Indeed, in one study, the co-immunoprecipotation analysis showed the interaction of δ-subunit with the ATP-gated ionotropic p2X2 receptors in human and rodent sperm (Xu et al., 2017). Besides, a very recent yeast two hybrid study, screening the mouse cerebellum cDNA library, reported a novel interacting protein (transthyretin (TTR)) for the δ-subunit (Zhou et al., 2019). The TTR protein, (a transport agent that carries the thyroid hormone thyroxine (T4) and retinol), co-immunoprecipitated with the δ-subunit. The TTR knock-out mice showed a significant decrease of the surface expression of the δ-GABAAR in the cerebellar granule neurons (Zhou et al., 2019). These suggest that the TTR, interacting with the δ-subunit, is a regulatory partner οf the δ-GABAAR. Thus, as such novel protein interactors of the δ-subunit are being decoded, the molecular dissection of these interactions with the δ-subunit and δ-GABAARs, a relatively virgin territory of the GABAAR research, is required. Regarding this, fluorescent fusion proteins are valuable tools for various molecular and cellular biology applications including the microscopic studies of receptor localization in the fixed cell preparations as well as receptor subunit assembly, interaction, targeting and trafficking during live cell imaging. For example, methods such as Fluorescence Resonance Energy Transfer (FRET) will help identify molecular interaction domains. The FRET would require the establishment of fluorescent fusion proteins, generated by the recombinant DNA technology by which an in-frame insertion of a fluorescent tag, such as Enhanced Green Fluorescent Protein (EGFP) -athirty five times brighter variant of the wild type green fluorescent protein-GFP (Zhang et al., 1996) in to the mature peptide of the protein of interest is performed (Arslan et al., 2014; Bueno et al., 1998; Connor et al., 1998; Shrivastava et al., 2011).
On the other hand, EGFP is relatively a long peptide, and thus, its fusion may interfere with the protein structure, stability and function, making the validation of the specific fluorescent fusion proteins crucial. Despite this, tagging the subunits of GABAARs with EGFP is particularly a robust strategy. Accumulating literature reports the studies, in which subunits of GABAARs are tagged with the GFP are utilized, reflecting the robustness of this strategy. Majority of these GABAAR studies have a focus on the abundantly expressed α, β or γ subunit isoforms which are usually tagged with the GFP or EGFP in their N- terminus or C- terminus (Arslan et al., 2014; Bueno et al., 1998; Connor et al., 1998). However, to our knowledge, there is no δ-subunit tagged in the evolutionary conserved cytoplasmic domain, which is found in between the TM3-TM4 regions of the subunit.
In this study, two fusion proteins have been generated by which the δ-subunit cDNA was tagged with the EGFP in its cytoplasmic domain, i.e., either between the residues A398 and K399 (δ398EGFP399) in the mature peptide of δ-subunit or between the residues K in the position of 346 and V in the position of 347 (δ346EGFP347) in the mature peptide of δ-subunit. Each of these recombinant subunits were then transfected into the Neuroblastoma 2a cells under four different conditions: (i) alone, (ii) with α1 subunit, (iii) with β2 subunit, (iv) with α1 and β2 subunit. The confocal microscopy analysis showed that, only co-transfection of δ398EGFP399 with α and β subunit isoforms (condition (iv)) led to the membrane targeting of the recombinant receptors. Similarly, for the δ346EGFP347, the confocal microscopy analysis showed that, only co-transfection of δ346EGFP347 with α1 and β2 subunits (condition (iv)) led to the membrane targeting of the receptors. These results are in line with general assembly and membrane targeting patterns of GABAARs (Comenencia-Ortiz et al., 2014). Thus, our data provide the evidence that EGFP fused at the cytoplasmic domain, in the specified locations, does not interfere with the membrane targeting of recombinant δ-GABAARs in the neuroblastoma 2a cells. To our knowledge, this is the first study showing the oligomerization and cell surface expression of the δ-subunit tagged with the EGFP at its cytoplasmic domain.
This study is approved by International University of Sarajevo. All experiments were performed in accordance with relevant guidelines and regulations.
Mouse C57 Brain cDNA (#MD-201-C57) was obtained from Zyagen (USA). pTZ57R vector was obtained from InsTAclone PCR Cloning Kit (#K1213) Thermo Scientific (USA). pcDNA3.1(-) vector and EGFP plasmid were gifted by Çağdaş Devrim Son, Middle East Technical University, Turkey. Primers were obtained by Micropolo (Sarajevo/Bosnia and Herzegovina) and Oligamer (Ankara/Turkey). All constructs were verified by sequencing.
Phire Hot Start II PCR Master Mix (#F125S), Taq DNA polymerase (#EP0402), dNTP Mix (#R1121), GeneRuler 1kb DNA ladder (#SM0313) and Loading Dye (#R0611) were all obtained from Thermo Scientific (USA). T4 DNA Ligase (#NE-M0202S) and all restriction enzymes (BamHI, DpnI, EcoRI and HindIII) were purchased from New England Biolabs (USA). InsTAclone PCR Cloning Kit (#K1213) purchased from Thermo Scientific (USA) used for post PCR sub-cloning experiments. For DNA extraction from gel and for the post-PCR purifications, GeneJET Gel Extraction Kit (#K0691) and GeneJET PCR Purification Kit (#K0701) were used respectively (Thermo Scientific, USA). For transfection experiments, Lipofectamine® LTX and Plus™ Reagent (#15338-100) by Invitrogen (USA) were used. Additionally, glass bottom dishes were supplied from In-Vitro Scientific (USA).
The mouse GABAARs α1, β2 and δ-subunits were amplified from whole-brain Mouse C57 cDNA (#MD-201-C57, Zyagen USA) by PCR and were cloned using InsTAclone PCR Cloning Kit (#K1213, Thermo Scientific). The PCR reactions for α1, β2 and δ-subunits used the primer pairs which were flanked by EcoRI sites (underlined): (5′-TGC CAT $\underline{GAA}$ $\underline{TTC}$ CGC GCC CGC GCT CGC AGA CTC GCG -3′), (5′-TGC CAT $\underline{GAA}$ $\underline{TTC}$ TGA AAT ATG GAA ATA AGG AAA CCT-3′), (5′-TGC CAT $\underline{GAA}$ $\underline{TTC}$ ATC CCT CAA TCC CAC CAG AAC CTA-3′), (5'-TGC CAT $\underline{GAA}$ $\underline{TTC}$ TTG CTT CTC ACG GAA GGC TGT AGT-3′), (5′-TGC CAT $\underline{GAA}$ $\underline{TTC}$ GCA ACT TTG CTT GCG CTG GGG CTA-3′) and (5′-TGC CTA $\underline{GAA}$ $\underline{TTC}$ GAG GGC CTC CAG GAG TTT GTG GCA-3′), respectively. The PCR reactions were performed in 20 µL reaction volumes containing 3 ng cDNA, 1 µM of each of the primers, 10 µL of 2x Phire Hot Start II PCR Master Mix (Thermo-fisher) and initially incubated at 98 °C for 2 minutes and cycled at 98 °C for 30 seconds, 58 °C for 45 seconds, 72 °C for 1 minute followed by a final incubation at 72 °C for 20 minutes in the thermal cycler (T100™ Thermal Cycler, BIO RAD). The generated amplicons were subcloned by TA cloning. The blunt-end amplicons generated by Phire Hot Start II polymerase was used as a template (200-250 ng) which were A-tailed by 1 µL of Taq DNA polymerase (5 U/µL) in the presence of 0.4 µL of 10 mM dATP, 1.2 µL of 2.5 mM MgCl2, 2 µL of 10X Taq Buffer in total of 20 µL reaction by incubation at 70°C for 30 minutes. Then the reaction product was isolated with PCR purification kit (Qiagen). The A tailed subunit cDNAs (α, β, δ, δ398EGFP399 and δ346EGFP347) were cloned into mammalian expression vector (pcDNA 3.1 (-)) ligation with T4 DNA ligase from NEB (#NE-M0202S) at 4 °C for 16 hours before transformation in to chemically competent E. coli DH5α. The transformed bacteria were plated on agar plates containing ampicillin (100 μg/ml) and incubated overnight at 37 °C. The following day, the colonies were picked and grown as miniprep in LB medium containing ampicillin (100 μg/ml). After overnight incubation of minipreps, the plasmids which were purified by alkaline lysis method, were screened and selected by restriction enzyme digestion and agarose gel electrophoresis. The final verification of positive clones was done by DNA sequencing.
In this study two different constructs (δ398EGFP399 and δ346EGFP347) were generated by which the each of the δ subunit was tagged by in frame insertion of EGFP in the cytoplasmic domain, i.e. either between the lysine (K) in the position of 346 and valine (V) in the position of 347 in the mature peptide or between the alanine (A) in the position of 398 and lysine in the position of 399 in the mature peptide of the subunit cDNA. The nested PCR method which consists of two consecutive PCR reactions was used for this procedure. Primers were designed in a way that 20-25 base pairs of primers are complementary to the fluorophore (EGFP) sequence and the remaining 30-35 base pairs were complementary to the sequence of the cDNA which is going to be integrated. For the δ398EGFP399 PCR reactions, the following primer pairs were used (5′-GGG TTC CTA TAG GTC TGT GGA AGT GGA GGC AAT GGT GAG CAA GGG CGA GGA G-3′) and (5′-GGC CCC CTG GGC GGG ACC CCC CCT CCT TCT TCT TGT ACA GCT CGT CCA TGC CG-3′). For the δ346EGFP347 PCR reactions the primer pairs (5′-GAC TAC AGG AAG AAA CGG AAA GCC AAG GTC AAG ATG GTG AGC AAG GGC GAG GAG-3′) and (5′-CCT CAC ATC CAT CTC TGC CCT TGG CTT GGT GAC CTT GTA CAG CTC GTC CAT GCC G-3′) were used. In the first PCR reaction, EGFP cDNA was used as a template and amplified (stop codon excluded) with 30 base pairs overhangs complementary to the cDNA sequence of the δ-subunit at the insertion point. Generated PCR amplicons were gel purified. The purified amplicons were used as primers for the second PCR reaction, during which the molar ratio between the template (δ-subunit cDNA) and the primers was 1 : 5. After the termination of the second PCR reaction, the DpnI restriction enzyme digestion was applied to PCR amplicons in order to remove untagged constructs. Then, the PCR products were transformed in to chemically competent DH5α cells and plated on ampicillin (100 μg/ml) containing agar plates. The details of the PCR reactions are described in Table 1.
| PCR Conditions | Duration | Temperature | Cycle | |
|---|---|---|---|---|
| Initial Denaturation | 3 Minutes | 95° C | ||
| Denaturation | 30 seconds | 95° C |  |
|
| Annealing | 1 minute | 51° C | 18 cycles | |
| Extension | 15 Minutes | 68° C | ||
| Final Extension | 5 Minutes | 68° C | ||
Transfection of recombinant cDNAs to Neuroblastoma 2a (N2a) cell line was performed by Lipofectamine LTX with PlusTM reagents (Invitrogen). 90.000 cells were seeded on a 35 mm diameter glass bottom dish (In Vitro Scientific) and were grown for 24 hours in the growth media (EMEM, Thermoscientific (670086) with 10% FBS and penicillin-streptomycin). After overnight incubation at 37 °C in the 5% CO2 incubator, 500 ng of each construct was diluted in 100 µL of OptiMEM® (Thermoscentific) and 4 µL of PlusTM (Thermoscentific) reagent was added to each tubes. Then, the tubes were incubated at room temperature for 15 minutes. Following this, OptiMEM~Lipofectamine LTX mixture (containing 4 µL of Lipofectamine LTX and 100 μl of OptiMEM), was added to tubes and the tubes were left for incubation at room temperature for 15 minutes. During incubation time, the cells were prepared for transfection: medium on the cells were removed and the cells were washed with 1 mLLive cell imaging with spinning disc confocal microscopy of 37 °C sterile PBS solution. After washing step, 800 µL of OptiMEM was added to each plate. As soon as incubation time was completed, 800 µL OptiMEM was added to the cells, followed by incubation for 3 hours in the 5% CO2 incubator at 37 °C. After the incubation, 2 ml of the growth medium was added to the cells which were incubated for 48 hours in the 5% CO2 incubator at 37 °C.
Live cell imaging studies were done by Leica Microsystems CMS GmBH's DMI4000B confocal microscope with Andor DSD2 spinning differential disc confocal device. ImageJ program was used for analysis of images that is obtained by confocal microscopy. After neuroblastoma 2a cell lines were transfected with fusion proteins (δ346EGFP347 and δ398EGFP399) alone or co-transfected in combination with α1, β2 or α1 and β2 subunits together. After 48 hours incubation, the cells were imaged by Leica DMI 4000 equipped with Andor DSD2 spinning disk confocal microscope using oil immersion objective lens with 63X magnification and 1.4 numerical aperture. The filter set was 485 nm-530 nm.
A total of ten GABAAR cDNA constructs, namely, α1, β2, δ, δ398EGFP399, and δ346EGFP347 were cloned. Each subunits of the GABAAR cDNAs were cloned in two different plasmids including pTZ57R and pcDNA3.1(-). These constructs are shown in the diagram below (Fig. 1). Firstly, subunits were cloned under T7 promoter in pTZ57R vector by the use of TA cloning method (Yao, et al., 2016). Following this, δ-subunits were further modified and tagged with EGFP flourophore in the cytoplasmic domain at two different positions. Generated constructs were then sub-cloned in to the pcDNA3.1(-) for transfection experiments in the mammalian cells.
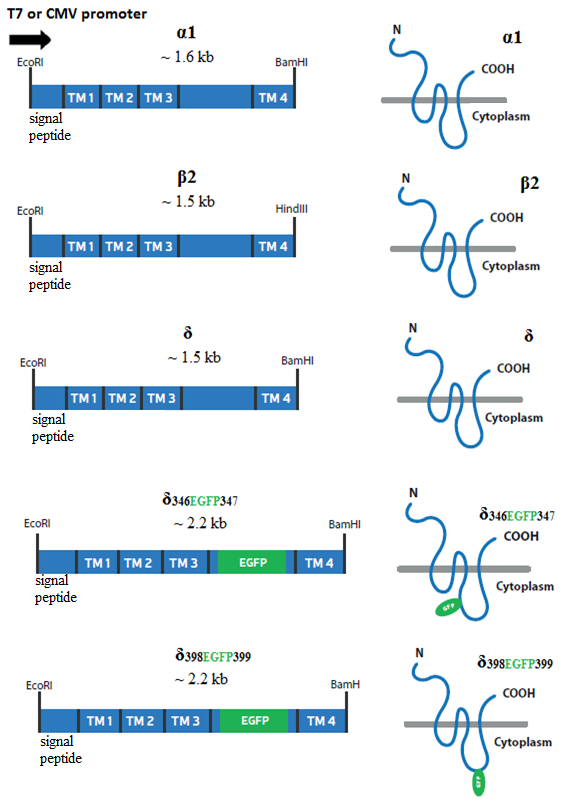 Figure 1.
Figure 1.Schematic representation of the generated constructs of GABAARs. The cDNAs of α1, β2, δ, δ398EGFP399, and δ346EGFP347 are shown on the left column. The cDNAs were initially cloned into pTZ57R/T vector with T7 promoter and then cloned in to the mammalian expression vector under the control of CMV promoter. On the right, schematic representation of the α1, β2, δ, δ346EGFP347 and δ398EGFP399 subunits and the location of their extracellular N- and C- terminus as well as cytoplasmic domains in the absence or presence of EGFP tag are shown.
Next we conducted the transfection experiments for the δ398EGFP399. In order to test the pattern of subunit assembly and membrane targeting, we co-transfected the δ398EGFP399 with or without partner subunits to asses oligomerization. As shown in the Fig. 2, the expression of δ398EGFP399 was tested in four different conditions in which the following transfections of respective cDNAs were conducted: (A) δ398EGFP399 alone, (B) δ398EGFP399 with α1 subunit, (C) δ398EGFP399 with β2 subunit, (D) δ398EGFP399 with α1 and β2 subunit. As shown in the Fig. 2,the δ398EGFP399 when expressed alone or with α1 or β2 subunits (Fig. 2A, B, C respectively) fails to target cell membrane as the transfected cells gave a strong green fluorescence signal throughout the cytoplasm (purple arrow) excluding the nucleous (yellow arrow). On the other hand co-transfection of δ398EGFP399 with α and β subunits (2D) led to the membrane targeting of the recombinant receptors (red arrows). This shows localization of ternary subunit co-expression. Moreover, since subunit oligomerization occurs in the ER (Comenencia-Ortiz et al., 2014), the EGFP signals were also visible in the cytoplasm (purple arrow) which may suggest the idea about the trafficking of the ternary composition of the receptor to the cell surface.
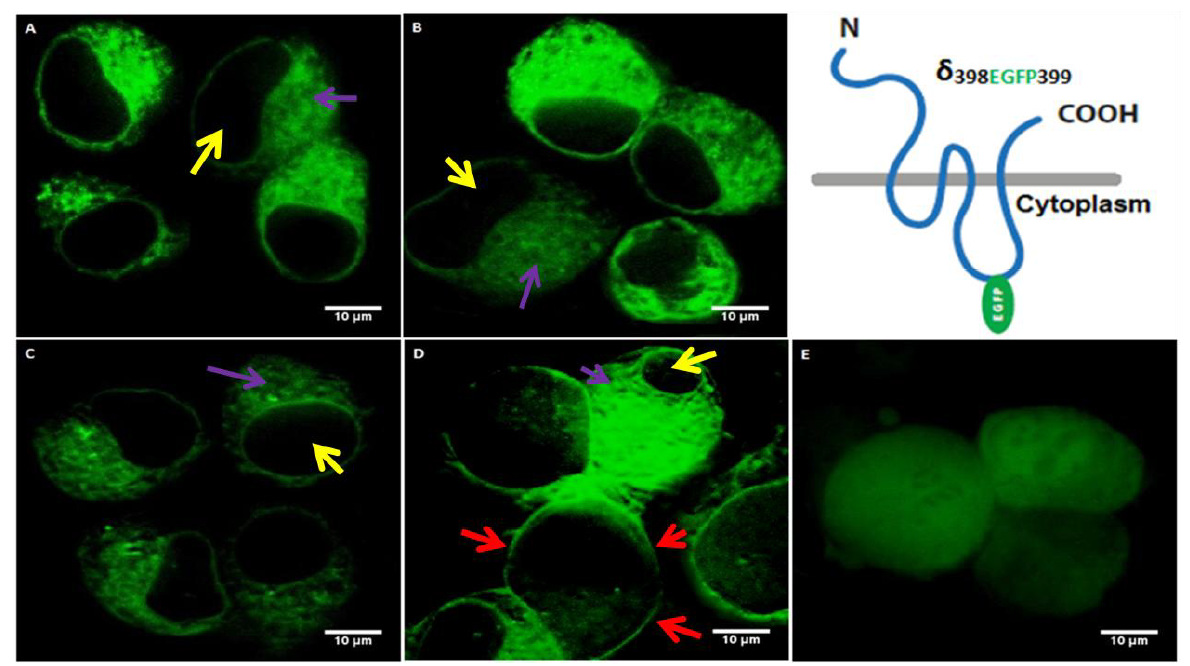 Figure 2.
Figure 2.Neuroblastoma 2a cells transfected with δ398EGFP399 construct in the absence or presence of α and/or β subunits. Yellow arrows show nucleous, red arrows show cell membrane, purple arrows show cytoplasmic/ER expression. (A): Transfection of δ398EGFP399 shows cytoplasmic expression as observed by the green signal (purple arrow) which is not observed in the nucleous (yellow arrow) (B): Co-transfection of δ398EGFP399 with α1 shows cytoplasmic expression as observed by the green signal (purple arrow) which is not observed in the nucleous (yellow arrow). (C): Co-transfection of δ398EGFP399 with β2 shows cytoplasmic expression as observed by the green signal (purple arrow) which is not observed in the nucleous (yellow arrow).(D): Co-transfection of δ398EGFP399 with α1and β2 in neuroblastoma 2a cells show membrane targeting (red arrows) as well as cytoplasmic expression excluding the nucleous (purple and yellow arrows, respectively). (E): Transfection of EGFP plasmid as control. The various EGFP intensity between the figures is due to the co-transfections which may lead to differential efficiency and thus variations in the signal intensity. Transfection of EGFP plasmid alone (E) showed a distribution of the EGFP signal not only in the cytoplasm but in the whole cell as EGFP can translocate to the nucleous.
In contrast, the expression of EGFP plasmid alone (2E) showed a whole cell expression including EGFP signal in the nucleus. This is expected as GFP peptide does not have any localization signal and it is known to translocate to nucleous on its own (Seibel et al., 2007).
In order to evaluate the subunit assembly and localization of the δ346EGFP347, the expression of δ346EGFP347 was tested in the four different conditions in which the following transfections of respective cDNAs were conducted (Fig. 3): (A) δ346EGFP347 alone, (B) δ346EGFP347 with α1 subunit, (C) δ346EGFP347 with β2 subunit, (D) δ346EGFP347 with α1 and β2 subunit. As shown on the Fig. 3, the expression of δ398EGFP399 alone or with α1 subunit or β2 subunit (Fig. 3A, B, C respectively) caused cells to give a strong green fluorescence signal throughout the cytoplasm (purple arrow) excluding the nucleous (yellow arrow) and the subunit fails to target cell membrane. On the other hand, the co-transfection of δ346EGFP347 with α and β subunits (3D) led to the membrane targeting of the recombinant receptors (red arrows). Moreover, since the subunit oligomerization occurs in the ER (Comenencia-Ortiz et al., 2014), the EGFP signals were also visible in the cytoplasm (purple arrow). The control experiment, which is the expression of EGFP plasmid alone (3E) showed a whole cell expression including EGFP signal in the nucleus. This is expected as GFP peptide does not have any localization signal and it is known to translocate to nucleous on its own (Seibel et al., 2007).
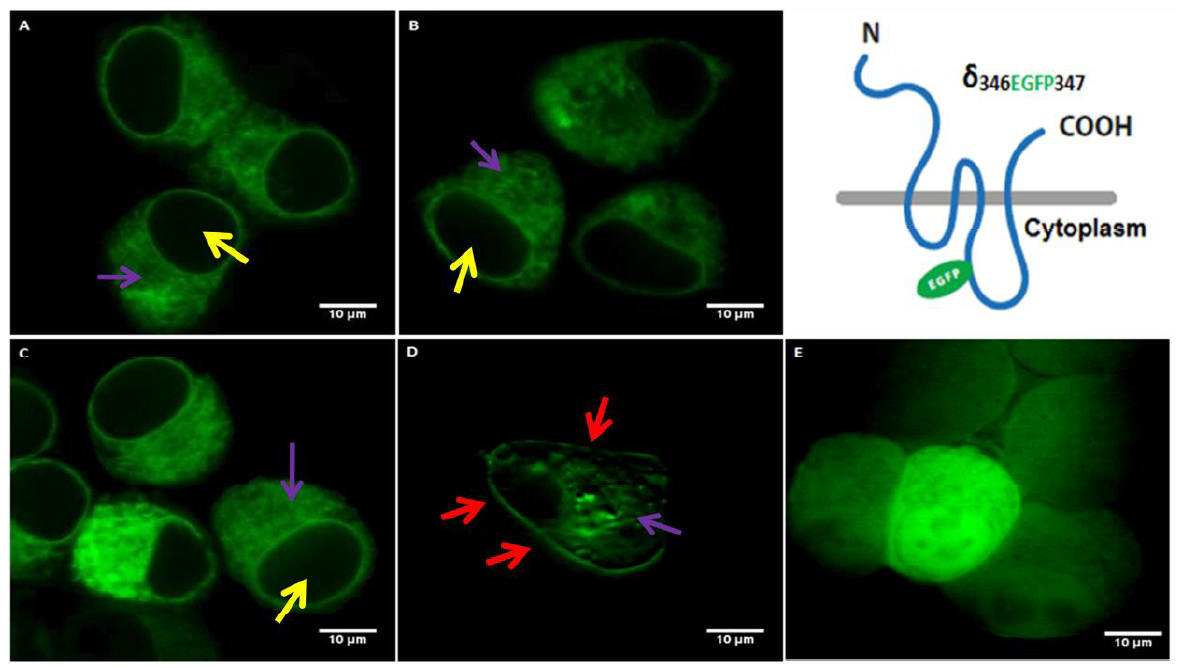 Figure 3.
Figure 3.Neuroblastoma 2a cells transfected with δ346EGFP347 construct in the absence or presence of α and/or β subunits. Yellow arrows show nucleous, red arrows show cell membrane, purple arrows show cytoplasmic/ER expression. (A): Transfection of δ346EGFP347 shows the cytoplasmic expression as observed by the green signal (purple arrow) which is not observed in the nucleous (yellow arrow). (B): Co-transfection of δ346EGFP347 with α1 shows cytoplasmic expression as observed by the green signal (purple arrow) which is not observed in the nucleous (yellow arrow). (C): Co-transfection of δ346EGFP347 with β2 shows the cytoplasmic expression as observed by the green signal (purple arrow) which is not observed in the nucleous (yellow arrow). (D): Co-transfection of δ346EGFP347 with α1 and β2 which shows expression in the membrane (red arrows). Also some intracellular expression (purple) isseen. (E): Transfection of the EGFP plasmid as the control. The various EGFP intensity between the cells is due to the co-transfections which may lead to differential efficiency and thus variations in the signal intensity. Transfection of the EGFP plasmid alone (E) showed a strong EGFP signal in the whole cells as EGFP can translocate to the nucleous.
The subunit assembly rules determine the cellular and sub-cellular localization and functionality of GABAARs suggesting that specific subunit domains are involved in the determination of specific function in precise localization (Arslan, 2015b; Goetz et al., 2007). Regarding this, much of the studies focus on the synaptic GABAARs, showing the domain specific functionalities of the respective receptor subunits (Alldred et al., 2005) but the role of δ-subunit domains on the functionality of the the extrasynaptic δ-GABAARs are mostly unknown. Especially the functional role of the cytoplasmic domain of the δ-subunit, an evolutionary conserved domain (Arslan et al., 2014), is not understood. In this context, EGFP tagging of the the cytoplasmic domain offers a great potential to design specific molecular experiments. Thus, in our study the two novel recombinant proteins were generated by EGFP tagging of the δ-subunit at two different locations in the cytoplasmic domain between TM3 and TM4. In general, the cytoplasmic domain between TM3 and TM4 subunits of GABAARs is a site for protein interactions and post-translational modifications, which are important for intracellular transport and localization (Kittler and Moss, 2003). Based on these, we expect that some protein interactions could be mediated by the cytoplasmic domain δ-subunit as well, probably in recruiting the oligomerization and/or cell surface expression of the δ-GABAARs. Thus, in order to show that EGFP tagging in the cytoplasmic domain does not interfere with any possible interaction involved in the subunit assembly and localization, we tested the pattern of oligomerization and cell surface expression of recombinant receptors to see if they are oligomerized and localized in the cell membrane, and they were, indeed. To our knowledge, our study is the first study showing the tagging of the δ-subunit in the cytoplasmic domain, its oligomerization and cell surface expression. This suggests the potential of our tagging strategy to stimulate the future studies for identification of novel interactions via cytoplasmic domain of the subunit with the utility of the EGFP.
Previous studies analyzed the cell surface expression, oligomerization and functionality of most abundant GABAARs subunits. Majority of these studies showed that αβ and αβγ subunit combinations had the ability to localize on the cell membrane and form functional ion channels (Angelotti and Macdonald, 1993; Connolly et al., 1996; Taylor et al., 2000). On the other hand, homomeric subunit expressions may also result in functional channels sensitive to GABA agonists or antagonists but not to GABA (Connolly et al., 1996; Krishek et al., 1996; Wooltorton et al., 1997). Nevertheless, single subunit expression of δ-subunit showed no GABA-gated chloride channels (Connolly et al., 1996). Additionally, ER retention of the single subunit expression occurs in neurons as well (Gorrie et al., 1997), which indicates the importance of the ternary subunit composition.
Consequently, we tested the oligomerization and cell surface expression pattern for our recombinant subunits (fusion proteins). In our study, we confirmed that the cell surface expression of our fusion proteins occur only when the recombinant subunits were co-expressed with α and the β subunits (Figs. 2D and F3D), but, when expressed alone, or either with α or β subunits, we observed the ER retention of fusion proteins in Neuroblastoma N2a cell lines. This was characterized by the green fluorescence EGFP signal that showed the distribution throughout the cytoplasm (Fig. 2A, B & C and Fig. 3A, B & C) suggesting that specific regulatory mechanism should ensure that only appropriately assembled GABAARs have the ability to reach to the cell surface. In line with this, multiple lines of evidence have shown that GABAARs subunit assembly is facilitated by the interaction with chaperone molecules in the ER (Bradley et al., 2008; Connolly et al., 1996; Sarto-Jackson et al., 2008).
On the other hand, among the cells transfected with δ398EGFP399, α1 and β2 subunits, the membrane targeting of the recombinant receptor as shown by red arrows (Fig. 2D), is not observed in other cells in the Fig. 2D, where the green EGFP signals were distributed throughout the cytoplasm but not necessarily in the cell membrane. Thus, as seen in one of the cells in 2D (yellow arrow), this may reflect the fact that the assembly of δ398EGFP399 fusion protein with α1 and β2 subunits may not be efficient in that specific cell. Indeed, one study suggested that the process of receptor oligomerization is slow and inefficient and nearly 70% of subunits are degraded in the ER without being incorporated into functional heteropentameric receptor (Green and Millar, 1995). Driving from this study, we speculate that degradation of α1 subunit alone or together with β2 subunits in the ER led to failure of ternary assembly of the subunits which resulted in failure of cell membrane localization. Possibly, the expression vector (pcDNA3.1(-)) that is used in our study has a powerful promoter (CMV), which leads to over-expression of the subunits and thus may generate some inefficiency as well.
The co-transfection of δ346EGFP347 with α and β subunits (3D) led to the membrane targeting of the recombinant receptors (red arrows). But there is also clear cytoplasmic signal (purple arrow). There may be at least two reasons for this cytoplasmic signal: (i) It may suggest the idea about the trafficking of the ternary composition of the receptor to the cell surface. Since subunit oligomerization occurs in the ER (Comenencia-Ortiz et al., 2014), EGFP signals were also visible in the cytoplasm (purple arrow). (ii) Alternatively, the intracellular signal could be an artifact of co-transfection experiments. We transfected the N2a cell lines with three constructs but it is possible that some cells were not co-transfected with all three subunits. Among such cells, the EGFP positive ones seem to represent the excessive expression of EGFP tagged subunit which could not be saturated by the expression of partner subunits, i.e., α and/or β, for proper oligomerization. Thus, the intracellular signals (purple arrow) may represent subunits failed to oligomerize and retained in the ER. On the other hand, the cell does not seem to be optimally healthy (Fig. 3D) and this may support the second possibility that the excessive expression of the recombinant subunit might have stressed the cell. This may suggest a major drawback for its usefulness for in-vitro assays and thus a replication of our experiments by independent labs are required. Nevertheless, the membrane targeting is clearly seen.
Since we tagged the δ-subunit in the cytoplasmic domain, it is also important to discuss the ultimate functionality of the recombinant receptors and if their function would be degenerated as a result of EGFP tagging in the cytoplasmic domain of the δ-subunit. The function of GABAARs are provided by multiple subunit domains with specific functional properties including channel electrophysiological profile and membrane localization. For example, as reviewed by Goetz et al.(2007), the selectivity filter and gating properties are attributed to the regions in the transmembrane domains which were not mutated/tagged in our study. On the other hand, the cytoplasmic domain is expected to be involved in interacting with other cytoplasmic proteins and thus having a role in attaching an anchor and/or regulatory proteins for membrane oligomerization, trafficking and/or localization (Comenencia-Ortiz et al., 2014; Kittler and Moss, 2003). Therefore, we focused on the latter as a primary test (i.e., oligomerization and cell surface expression) which was conducted by co-transfection experiments and confirmed with the confocal imaging data described in the sections of Results 3.2 and 3.3.
On the other hand, due to the diversity of the receptor subunits, it is hard to generalize the functional roles of the GABAAR subunit domains. For example, tyrosine phosphorylation found in the cytoplasmic domain of the γ2 subunit amplified the whole-cell current induced by GABA (Moss et al., 1995). Whether a similar pattern would be observed in the cytoplasmic domain of the δ-subunit is not known. Interestingly, δ-subunit or γ2 subunit co-assemble with α and β subunits but they never co-assemble with each other (Araujo et al., 1998; Jechlinger et al., 1998; Quirk et al., 1995), although one study reported that δ and γ2 subunits co‐assemble (Mertens et al. 1993). Indeed, these subunits are thought to belong to two functionally distinct GABAAR subtypes mediating distinct GABAergic inhibitions, i.e., tonic and phasic, respectively (Brickley and Moddy, 2012, Whissell et al., 2015) (despite one recent study (Sun et al., 2018) suggesting that δ-GABAARs contribute to synaptic (phasic) inhibition). Therefore, δ-subunit is generally considered as a substitute of the γ2 subunit. This substutition in the receptor composition may reflect the two functionally distinct GABAergic inhibition. Thus, domain functionalities of these two subunits would also be expected to be different not similar. Nevertheless, due to conflicting results as mentioned above, it is hard to speculate on this matter without further experimental data. As a result, it is important to conduct the whole-cell patch clamp recordings in the EGFP-fluorescence-positive cells expressingthe recombinant δ-subunits (see also “4.3. Limitations of the study” below).
Our results need to be further verified due to some limitations. First, in our study we have used CMV promoter for expression of recombinant subunits. It is known that, CMV promoter leads to high level of gene expression (Khan, 2013) and thus over-expression of subunits may give us some erroneous results (Sigel et al., 2009). Second, we transfected the N2a cell lines with three plasmid constructs. There is a possibility for some subunits to be not transfected. If this was the case, it could have an effect on the localization of the receptors since the localization of the receptor depends on receptor oligomerization (Sarto-Jackson et al., 2008; Tretter et al., 1997). To solve this, dual promoter vector (Sinah, et al., 2012) could be used to increase transfection efficiency. Fourth, by the use of immunoblotting, it would be elegant to show that fusion proteins, when expressed in the cell membrane are intact but not truncated. On the other hand, based on the other GABAAR subunits tagged in the cytoplasmic domain (Shrivastava et al., 2011), a truncation of the δ-subunit due to EGFP insertion is not expected.
Finally, in our study, the recombinant constructs were tested only for oligomerization and cell surface expression, but not electrophysiologically, primarily due to the reasons discussed in the section called “4.2. Functionality of recombinant GABAARs”. Nevertheless, without direct experimental evidence, it is hard to formulate distinct, one-to-one, domain and function relationship in general and specifically for the GABAARs due to the diversity of subunits with unexpected functions of their domains (Alldred et al., 2005). Thus, a possibility of functional relation of cytoplasmic domain with the tonic currents cannot be ignored and this implies that our analysis requires a detailed electrophysiological study of recombinant receptors in different cell lines.
The molecular dissection of GABAAR subunit domains are essential to resolve the subunit specific functionality of GABAAR subtypes. For the δ-GABAAR subtype, the domain specific EGFP tagging of the δ-subunit will provide the relevant molecular tool for the development of the cellular assays to probe molecular interactions of this subunit. Our data provide the first study focusing on the EGFP tagging of the cytoplasmic domain of the δ-subunit, in which the tagging was performed in two distinct locations. The expression of each of the δ398EGFP399 or δ346EGFP347 recombinant δ-subunits in Neuroblastoma 2a cells confirm their oligomerization with α1 and β2 subunits and cell surface expression of the assembled recombinant receptors. This suggests that the EGFP tagging did not interfere with the oligomerization and cell surface expression of recombinant subunits. Our results show the potential of our cytoplasmic tagging strategy to stimulate future studies such as electrophysiology and FRET, which will eventually contribute to our -so far very limited- understanding of the molecular factors involved in the functions of extrasynaptic δ-GABAARs.
FEO performed the experiments and collected the data. AA conceived and designed the experiments, supervised the project and acquired the project funding. AA and ÇDS analyzed the data. AA and ÇDS contributed the reagents and materials. AA edited and revised the paper.
C-(terminus): Carboxyl (-COOH) terminus of the peptide; EGFP: Enhanced Green Fluorescence Protein; ER: Endoplasmic Reticulum; FRET: Fluorescence resonance energy transfer; GABA: γ-Aminobutyric acid;GABAARs: γ-Aminobutyric acid type A receptors; GFP: Green Fluorescence Protein;N- (terminus): Aminoterminus of the peptide ending with the amine group (-NH2); PCR: Polymerase Chain Reaction;TM3 3rd transmembrane domain;TM4 4th transmembrane domain; TTR Transthyretin.
The authors acknowledge the institutional and funding support from the International University of Sarajevo and The Middle East Technical University. The authors thank to reviewers and editor for valuable insights and critisisim during the evaluation and revision of this work.
The authors declare no conflict of interest.



