1 Key Laboratory of Environmental Medicine and Engineering of Ministry of Education, Department of Nutrition and Food Hygiene, School of Public Health, Southeast University, 210009 Nanjing, Jiangsu, China
2 Jiangsu Engineering Technology Research Center for Modernization of Traditional Chinese Medicine, 210028 Nanjing, Jiangsu, China
†These authors contributed equally.
Abstract
Cardiovascular disease (CVD) remains a significant global health burden, while carotenoids are considered to have beneficial protective effects against CVD. Thus this study aimed to synthesize available relevant findings regarding the potential correlation between carotenoid consumption (including α/β-carotene, β-cryptoxanthin, lutein, lycopene, zeaxanthin, and astaxanthin) and CVD risk through an umbrella meta-analysis and systematic reviews.
Literature searches were conducted in four online academic databases, including PubMed, Web of Science, Embase, and the Cochrane Library, as well as in the literature reference lists of existing meta-analyses and systematic reviews. The search was completed with a cutoff date of July 2023.
The association between carotenoids, including blood, carotenoid intake, and carotenoid supplements, and CVD risk was estimated as an odds ratio (OR) of 0.889 (95% confidence interval (95% CI): 0.857–0.922; p < 0.001), indicating a notable protective association. Furthermore, our analysis demonstrated that the protective impact of carotenoids on total CVD was the most significant for coronary heart disease (OR = 0.875; 95% CI: 0.822–0.932) and stroke (OR = 0.815; 95% CI: 0.734–0.906).
By controlling for the confounding effects of heterogeneity, including clinical heterogeneity, methodological heterogeneity, and statistical heterogeneity, and publication bias, including positive result enrichment bias, duplicate publication bias, selective study inclusion bias, and selective bias in data extraction and analysis, etc., this study conducted a methodical assessment of the correlation between carotenoids and clinical outcomes of CVD, demonstrating significant inverse relationships between multiple carotenoids and both CVD incidence and mortality of CVD.
Keywords
- carotenoids
- cardiovascular disease
- stroke
- coronary heart disease
- umbrella meta-analysis
Cardiovascular disease (CVD) is defined as disorders of the cardiac and circulatory system caused by multiple factors that evolve gradually throughout life [1], which represent the primary global mortality cause with profound socioeconomic implications [2]. Diet-related risks are now recognized as the primary adjustable risk determinant contributing to the elevation of the global CVD-related burden [3] and plant-based dietary patterns endorsed by major clinical guidelines for CVD prevention [4]. Approximately 12 major carotenoid species, lipophilic bioactive metabolites primarily concentrated in yellow, orange, and red fruits and vegetables, are key contributors to dietary micronutrient consumption [5]. Extensive epidemiological findings indicate that carotenoid-rich fruit and vegetable-dense diets exert protective effects against CVD [6] and several chronic diseases [7].
Risk factors for the development of CVD primarily include oxidative stress [8], chronic inflammation, and metabolic disorders [9], with oxidative stress as a key pathological mechanism underpinning CVD initiation and progression [10, 11, 12]. Carotenoid intake mitigates CVD risk through these multifaceted pathways. Notably, carotenoids exhibit robust antioxidant activity, enabling them to scavenge free radicals and attenuate oxidative stress [10, 11, 12]. Inflammation represents a major driver of CVD pathogenesis, and carotenoid intake alleviates cardiovascular injury by modulating inflammatory signaling pathways [13]. Furthermore, carotenoid consumption effectively regulates blood pressure, blood lipids, and blood glucose levels, thereby directly targeting CVD risk pathways [14].
Based on their chemical structure, carotenoids can be divided into two classes:
xanthophylls, which encompass
Preclinical investigations demonstrate that carotenoids exhibit antioxidant capacity and immunomodulatory activities [5, 17, 18], regulating cellular proliferation, gene expression profiles, and immune signaling cascades [19]. However, the evidence regarding benefits of carotenoids to cardiovascular remains inconclusive, primarily due to the structural diversity of these compounds and the heterogeneity in study designs and conflicting findings [20, 21]. Therefore, a systematic umbrella review synthesizing epidemiological and mechanistic evidence is warranted to resolve inconsistencies in the evidence base and clarify the protective potential of carotenoids against cardiovascular outcomes.
Despite advancements in evidence synthesis methodologies, the translational utility of meta-analyses and systematic reviews with respect to human health-related outcomes remains constrained through persistent inconsistencies arising from heterogeneous study designs, outcome measurement disparities, and incomplete data reporting. To address these constraints, Ioannidis et al. [22] originally conceptualized the umbrella review methodology in 2009 as a framework for synthesizing evidence from multiple systematic reviews. Within nutritional epidemiology, umbrella reviews have been particularly impactful for reconciling conflicting evidence on carotenoid bioavailability and CVD risk, where study designs often diverge in population demographics, intervention durations, and biomarker measurement techniques [23, 24]. Up to the present, no umbrella review has conducted a systematic assessment of the strength of evidence and potential sources of bias across meta-analyses that investigate associations between carotenoids and CVD. To achieve a more comprehensive and evaluative understanding of the available evidence, we performed the umbrella review that systematically synthesized all published meta-analyses and systematic reviews. This approach was employed to reappraise and clarify the possible roles of various nutritional interventions in the prevention of CVD.
This umbrella review adheres to PRISMA reporting guidelines and was prospectively registered with PROSPERO under identifier CRD42023426292, ensuring transparency and reproducibility in evidence synthesis.
This umbrella review systematically evaluated the assessment of evidentiary data
from existing systematic reviews and meta-analyses to assess correlations between
carotenoid and CVD outcomes, adhering to rigorous methodological standards for
evidence synthesis. Two investigators independently and systematically performed
a literature search in the PubMed, Web of Science, Embase, and Cochrane Library
databases. This review covered the period was set from the inception of each
database to July 2023, with the restriction that only English-language
articles were included. The search included the following Medical Subject
Headings (MeSH) terms: “(carotenoids OR
In this study, systematic reviews or meta-analyses that estimate the correlation
between carotenoid and CVD risk were deemed eligible for inclusion: (1)
encompassed cohort studies, cross-sectional studies, and case-control studies, as
well as randomized controlled trials (RCTs); (2) assessed
Criteria for exclusion were defined as follows: (1) meta-analyses focusing on non-observational research or non-randomized controlled trials; (2) studies lacking original data for analyzing pooled risk estimates and 95% confidence intervals (95% CI); (3) systematic reviews that did not incorporate a meta-analytic component; (4) non-research articles (e.g., letters, editorials, and conference abstracts); (5) duplicated publications.
The PRISMA-compliant systematic screening workflow, shown in Fig. 1, details database searches for carotenoid-CVD meta-analyses.
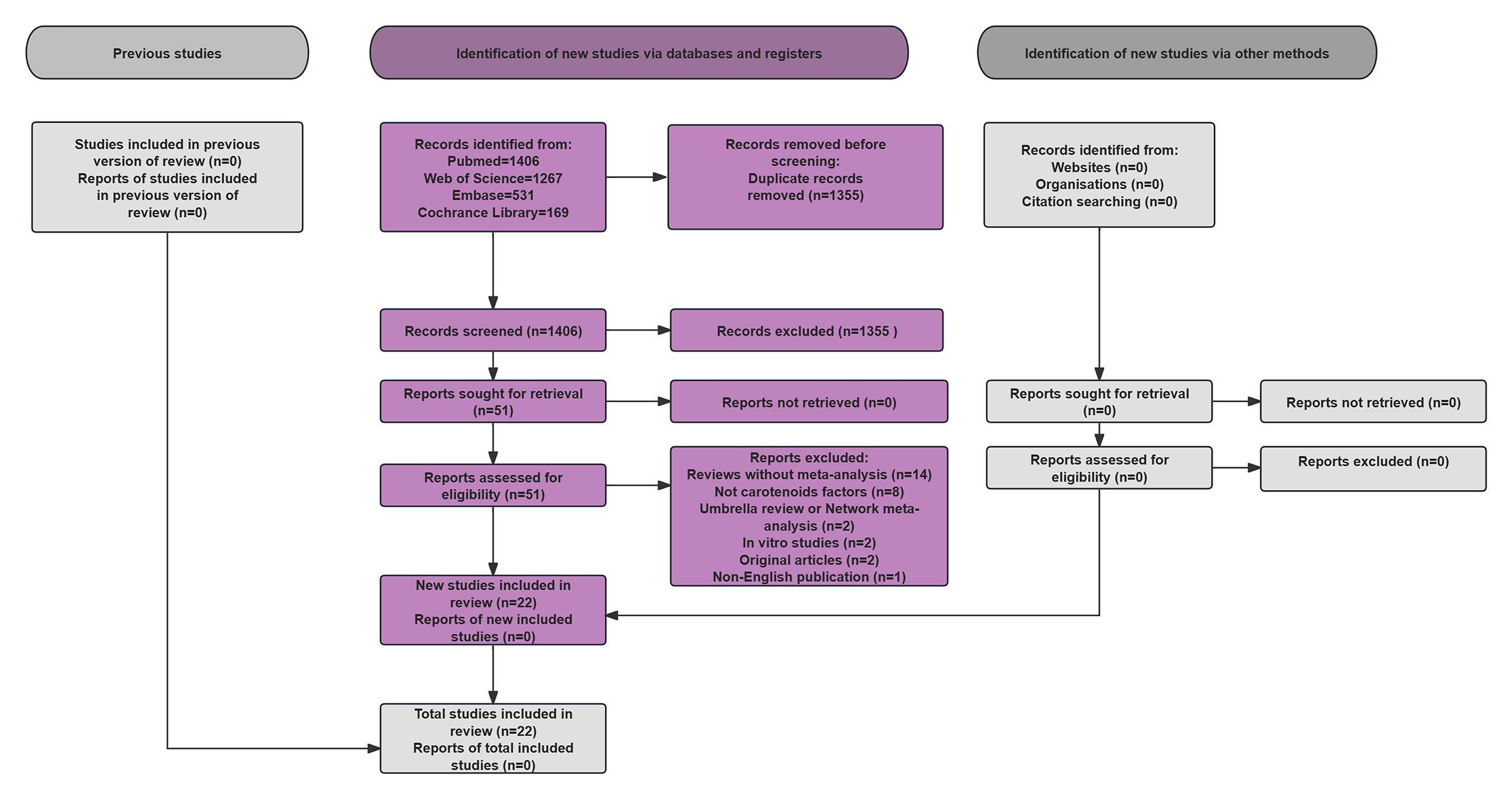 Fig. 1.
Fig. 1.
Flow chart of the literature search.
Extraction of Data was independently carried out by reviewers using a standardized protocol, capturing study characteristics, cardiovascular endpoints, carotenoid types, study designs, summary effect metrics, meta-analytic parameters, and risk of bias assessment tools.
Methodological evaluation of quality of eligible meta-analyses was systematically estimated using the A MeaSurement Tool to Assess the methodological Quality of systematic reviews (AMSTAR-2) [25], which evaluates 16 domains across 7 critical and 9 non-critical items. Each meta-analysis was categorized into High, Moderate, Low, or Critically Low quality based on item-level assessments.
Effect sizes (OR/RR/HR with 95% CI) from qualified meta-analyses were
systematically evaluated to quantify carotenoid-CVD associations, with subgroup
analyses stratified by carotenoid type and CVD outcome. The presence and
magnitude of heterogeneity were determined through using Cochran’s Q test and
the I2 statistic [26], with random-effects models applied when
I2
Following systematic database searches (PubMed, Web of Science, Cochrane Library, Embase), 3373 records were identified, yielding 22 eligible articles containing 64 meta-analyses evaluating carotenoid-CVD associations (Table 1, Ref. [23, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48]). Published between 1998 and 2023, these studies were systematically categorized into four CVD risk categories based on carotenoid type and outcome specificity. Eligible meta-analyses were categorized into four CVD outcome categories: total CVD, coronary heart disease, stroke and myocardial infarction and ischemic heart disease [28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49]. Given the limited number of available meta-analyses, myocardial infarction and ischemic heart disease were combined into a single category.
| Author & year | Type of CVD | N | Type of studies | Type of carotenoids | Type of metrics | Summary effect size (95% CI) | Model | I2 | Egger’s p value | Statistically significant |
| Li et al. 2021 a [23] | coronary heart disease | 10 | cohort | lutein intake | RR | 0.88 (0.80–0.98) | random | 0.242 | NR | Yes |
| Li et al. 2021 b [23] | stroke | 3 | cohort | lutein intake | RR | 0.82 (0.72–0.93) | random | 0 | NR | Yes |
| Farashi et al. 2023 [28] | stroke | 6 | CC, cohort, RCT | OR | 0.66 (0.47–0.92) | random | 0.69 | 0.3 | Yes | |
| Corbi et al. 2022 [29] | CVD mortality | 12 | RCT | RR | 1.04 (0.98–1.11) | random | 0 | NR | No | |
| O’Connor et al. 2022 a [30] | CVD mortality | 5 | RCT | OR | 1.10 (1.02–1.19) | random | 0 | NR | Yes | |
| O’Connor et al. 2022 b [30] | CVD incidence | 2 | RCT | OR | 1.01 (0.92–1.10) | random | 0 | NR | No | |
| An et al. 2022 a [31] | CVD mortality | 12 | RCT | RR | 1.12 (1.06–1.18) | random | 0.4 | 0.38 | Yes | |
| An et al. 2022 b [31] | stroke mortality | 6 | RCT | RR | 1.09 (1.01–1.17) | random | 0.73 | 0.065 | Yes | |
| An et al. 2022 c [31] | Myocardial Infarction | 7 | RCT | RR | 0.99 (0.93–1.05) | random | 0.02 | 0.428 | No | |
| Yang et al. 2022 a [32] | CVD incidence | 15 | RCT | RR | 1.04 (1.00–1.08) | random | 0 | 0.051 | Yes | |
| Yang et al. 2022 b [32] | CVD mortality | 12 | RCT | RR | 1.12 (1.04–1.19) | random | 0.24 | 0.5 | Yes | |
| Cheng et al. 2019 a [33] | stroke | 4 | CC, cohort | lycopene Intake | HR | 0.79 (0.64–0.97) | random | 0.44 | NR | Yes |
| Cheng et al. 2019 b [33] | stroke | 4 | CC, cohort | lycopene blood | HR | 0.61 (0.40–0.92) | random | 0.157 | NR | Yes |
| Cheng et al. 2019 c [33] | CVD incidence | 3 | CC, cohort | lycopene Intake | HR | 0.88 (0.78–0.99) | random | 0 | NR | Yes |
| Cheng et al. 2019 d [33] | CVD incidence | 4 | CC, cohort | lycopene blood | HR | 0.78 (0.63–0.98) | random | 0 | NR | Yes |
| Jayedi et al. 2019 a [34] | CVD mortality | 4 | CC, cohort, RCT | RR | 0.89 (0.73–1.05) | random | 0.35 | NR | No | |
| Jayedi et al. 2019 b [34] | CVD mortality | 6 | CC, cohort, RCT | RR | 0.68 (0.52–0.83) | random | 0.5 | NR | Yes | |
| Aune et al. 2018 a [35] | coronary heart disease | 4 | cohort | total carotenoids intake | RR | 0.82 (0.69–0.99) | random | NR | NR | Yes |
| Aune et al. 2018 b [35] | coronary heart disease | 3 | cohort | total carotenoids blood | RR | 0.68 (0.50–0.93) | random | NR | NR | Yes |
| Aune et al. 2018 c [35] | coronary heart disease | 4 | cohort | RR | 0.73 (0.63–0.85) | random | NR | NR | Yes | |
| Aune et al. 2018 d [35] | coronary heart disease | 4 | cohort | RR | 0.69 (0.53–0.90) | random | NR | NR | Yes | |
| Aune et al. 2018 e [35] | coronary heart disease | 4 | cohort | RR | 0.88 (0.71–1.10) | random | NR | NR | No | |
| Aune et al. 2018 f [35] | coronary heart disease | 2 | cohort | RR | 1.01 (0.43–2.37) | random | NR | NR | No | |
| Aune et al. 2018 g [35] | coronary heart disease | 2 | cohort | lycopene blood | RR | 0.88 (0.71–1.10) | random | NR | NR | No |
| Aune et al. 2018 h [35] | stroke | 3 | cohort | lycopene Intake | RR | 0.80 (0.63–1.01) | random | NR | NR | No |
| Aune et al. 2018 I [35] | cardiovascular disease | 2 | cohort | total carotenoids intake | RR | 0.87 (0.74–1.01) | random | NR | NR | No |
| Aune et al. 2018 j [35] | cardiovascular disease | 2 | cohort | total carotenoids blood | RR | 0.81 (0.64–1.03) | random | NR | NR | No |
| Aune et al. 2018 k [35] | stroke | 7 | cohort | RR | 0.84 (0.75–0.94) | random | NR | NR | Yes | |
| Aune et al. 2018 l [35] | cardiovascular disease | 4 | cohort | RR | 0.98 (0.84–1.15) | random | NR | NR | No | |
| Aune et al. 2018 m [35] | stroke | 3 | cohort | RR | 0.85 (0.71–1.01) | random | NR | NR | No | |
| Aune et al. 2018 n [35] | cardiovascular disease | 6 | cohort | RR | 0.73 (0.57–0.92) | random | NR | NR | Yes | |
| Aune et al. 2018 o [35] | stroke | 3 | cohort | RR | 0.74 (0.48–1.14) | random | NR | NR | No | |
| Aune et al. 2018 p [35] | cardiovascular disease | 3 | cohort | RR | 0.91 (0.64–1.29) | random | NR | NR | No | |
| Aune et al. 2018 q [35] | cardiovascular disease | 3 | cohort | RR | 0.83 (0.67–1.03) | random | NR | NR | No | |
| Aune et al. 2018 r [35] | cardiovascular disease | 2 | cohort | lycopene Intake | RR | 0.94 (0.79–1.12) | random | NR | NR | No |
| Aune et al. 2018 s [35] | stroke | 2 | cohort | lycopene blood | RR | 0.59 (0.36–0.96) | random | NR | NR | Yes |
| Aune et al. 2018 t [35] | cardiovascular disease | 4 | cohort | lycopene blood | RR | 0.88 (0.70–1.10) | random | NR | NR | No |
| Schwingshackl et al. 2017 a [36] | CVD mortality | 8 | RCT | RR | 1.06 (0.93–1.21) | random | 0 | NR | No | |
| Schwingshackl et al. 2017 b [36] | CVD incidence | 5 | RCT | RR | 1.14 (0.87–1.48) | random | 0 | NR | No | |
| Song et al. 2017 a [37] | CVD incidence | 7 | CC, cohort | lycopene Intake | RR | 0.87 (0.79–0.96) | random | 0.063 | 0.236 | Yes |
| Song et al. 2017 b [37] | CVD incidence | 7 | CC, cohort | lycopene blood | RR | 0.74 (0.62–0.87) | random | 0.45 | 0.753 | Yes |
| Leermakers et al. 2016 a [38] | coronary heart disease | 10 | CC, cohort | lutein intake | RR | 0.89 (0.83–0.97) | fixed | 0.242 | 0.221 | Yes |
| Leermakers et al. 2016 b [38] | stroke | 5 | CC, cohort | lutein intake | RR | 0.82 (0.72–0.93) | fixed | 0 | 0.47 | Yes |
| Li et al. 2014 [39] | stroke | 7 | CC, cohort | total lycopene | RR | 0.81 (0.68–0.96) | random | 0.255 | 0.277 | Yes |
| Fortmann et al. 2013 [40] | CVD incidence | 2 | RCT | RR | 1.01 (0.93–1.09) | random | NR | NR | No | |
| Myung et al. 2013 [41] | CVD | 17 | RCT | RR | 1.04 (0.96–1.12) | random | 0.55 | NR | No | |
| Mente et al. 2009 a [42] | coronary heart disease | 10 | cohort | RR | 0.73 (0.65–0.82) | random | NR | NR | Yes | |
| Mente et al. 2009 b [42] | coronary heart disease | 14 | RCT | RR | 1.01 (0.92–1.09) | random | NR | NR | No | |
| Ye et al. 2008 [43] | coronary heart disease | 3 | cohort | RR | 0.78 (0.53–1.04) | random | NR | NR | No | |
| Knekt et al. 2004 a [44] | coronary heart disease incidence | 9 | cohort | RR | 0.90 (0.77–1.04) | NR | NR | NR | No | |
| Knekt et al. 2004 b [44] | coronary heart disease incidence | 9 | cohort | RR | 0.92 (0.79–1.06) | NR | NR | NR | No | |
| Knekt et al. 2004 c [44] | coronary heart disease incidence | 9 | cohort | lycopene Intake | RR | 0.99 (0.85–1.14) | NR | NR | NR | No |
| Knekt et al. 2004 d [44] | coronary heart disease incidence | 9 | cohort | RR | 0.94 (0.79–1.12) | NR | NR | NR | No | |
| Knekt et al. 2004 e [44] | coronary heart disease incidence | 9 | cohort | lutein intake | RR | 0.89 (0.75–1.04) | NR | NR | NR | No |
| Vivekananthan et al. 2003 [45] | CVD mortality | 6 | RCT | OR | 1.10 (1.03–1.17) | random | NR | NR | Yes | |
| Asplund et al. 2002 a [46] | CVD | 8 | cohort | OR | 0.88 (0.77–1.01) | fixed | NR | NR | No | |
| Asplund et al. 2002 b [46] | CVD | 4 | cohort | OR | 0.46 (0.37–0.58) | fixed | NR | NR | Yes | |
| Asplund et al. 2002 c [46] | CVD | 6 | RCT | OR | 1.02 (0.96–1.08) | fixed | NR | NR | No | |
| Marchioli et al. 1999 a [47] | CVD | 7 | cohort | OR | 0.66 (0.57–0.78) | random | NR | NR | Yes | |
| Marchioli et al. 1999 b [47] | CVD | 3 | CC | OR | 0.61 (0.40–0.93) | random | NR | NR | Yes | |
| Marchioli et al. 1999 c [47] | coronary heart disease | 5 | RCT | OR | 1.02 (0.98–1.07) | random | NR | NR | No | |
| Law et al. 1998 a [48] | ischaemic heart disease | 4 | CC, cohort | carotenoids intake | RR | 0.85 (0.77–0.93) | NR | NR | NR | Yes |
| Law et al. 1998 b [48] | ischaemic heart disease | 3 | CC, cohort | carotenoids blood | RR | 0.57 (0.47–0.69) | NR | NR | NR | Yes |
| Law et al. 1998 c [48] | ischaemic heart disease | 5 | RCT | RR | 1.07 (0.98–1.16) | NR | NR | NR | No |
CVD, cardiovascular disease; N, number of meta-analyses; RCT, randomized controlled trial; CC, case control; CI, confidence interval; OR, odds ratio; RR, relative risk; NR, not reported; a-t, different letters present Meta-analyses selected from an article.
Methodological quality assessment using the AMSTAR 2 tool revealed that included
meta-analyses (n = 64) comprised 10 high-quality, 6 moderate-quality, and 24 low
and 24 critically low-quality meta-analyses (Supplementary Table 1). And
the association linking carotenoids and CVD risk was OR = 0.889; 95% CI:
0.857–0.922; p
Using the leave-one-out deletion method, we found the study that Asplund
et al. [46] exerted a relatively large influence. After its removal, the
risk was OR = 0.899; 95% CI: 0.868–0.931; p
Across all qualified meta-analyses investigating the association between dietary
carotenoid intake, carotenoid supplementation, or blood concentrations and CVD
outcomes, and a total number of 64 effect meta-analyses were identified. These
included studies focused on total CVD (n = 30) as well as three other specific
subtypes of CVD (n = 34), with the latter collectively accounting for the
remaining 34 effect meta-analyses in the dataset. A notable inverse correlation
was identified between carotenoid and CVD risk (OR = 0.889; 95% CI:
0.857–0.922; p
| Type of CVD | Type of carotenoids | Number of meta-analyses | OR (95% CI) | I2 (p value) |
| CVD | total carotenoids | 2 | 0.852 (0.748–0.970) | 0 (0.622) |
| 1 | 0.830 (0.670–1.030) | NA (NA) | ||
| 20 | 0.959 (0.906–1.015) | 0.863 ( | ||
| lycopene | 6 | 0.857 (0.807–0.909) | 0 (0.417) | |
| 1 | 0.910 (0.640–1.290) | NA (NA) | ||
| Coronary heart disease | total carotenoids | 2 | 0.781 (0.664–0.917) | 0.004 (0.307) |
| 7 | 0.849 (0.743–0.970) | 0.884 ( | ||
| 2 | 0.943 (0.795–1.119) | 0 (0.872) | ||
| lutein | 3 | 0.887 (0.841–0.935) | 0 (0.982) | |
| lycopene | 2 | 0.954 (0.845–1.078) | 0 (0.381) | |
| 2 | 0.894 (0.789–1.011) | 0 (0.868) | ||
| Stroke | 4 | 0.877 (0.722–1.064) | 0.870 ( | |
| lycopene | 5 | 0.774 (0.695–0.862) | 0 (0.596) | |
| lutein | 2 | 0.820 (0.749–0.898) | 0 (0.950) | |
| 1 | 0.740 (0.480–1.140) | NA (NA) | ||
| Myocardial infarction/ischaemic heart disease | total carotenoids | 2 | 0.702 (0.475–1.039) | 0.925 ( |
| 2 | 1.023 (0.949–1.103) | 0.535 (0.143) |
CI, confidence interval; OR, odds ratio; NA, not available.
A systematic literature search yielded 18 meta-analyses investigating
carotenoids and coronary heart disease. This umbrella meta-analysis revealed a
statistically notable protective role of carotenoids against coronary heart
disease (OR = 0.875; 95% CI: 0.822–0.932; p
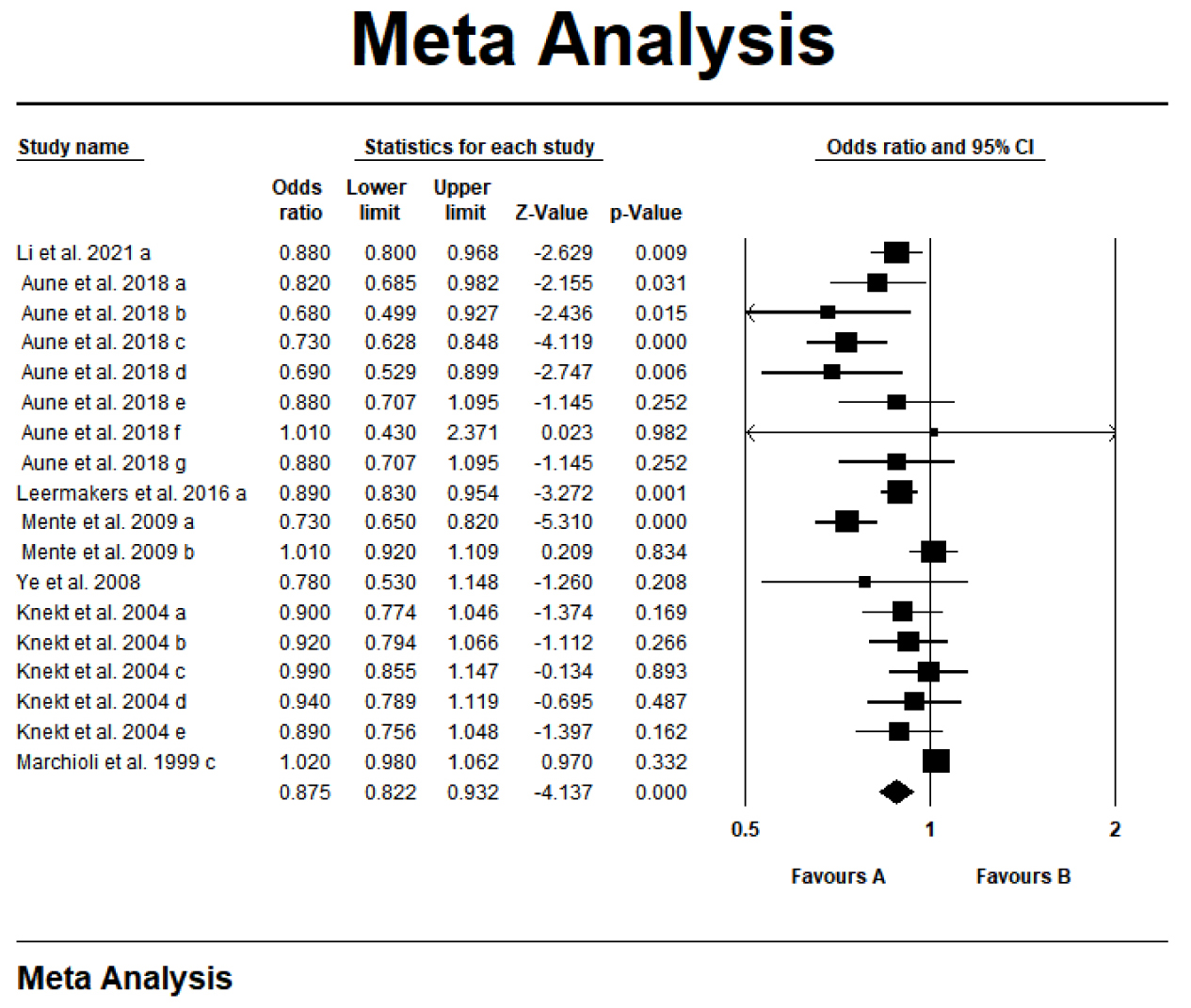 Fig. 2.
Fig. 2.
Forest plot of the effect of carotenoids on coronary heart disease. a-g, different letters represent different Meta-analyses selected from an article.
Of the 64 meta-analyses reviewed, 12 demonstrated a statistically significant
finding regarding the lowering of stroke risk with carotenoids. Fig. 3 presented
a significant negative correlation between carotenoid intake/blood levels and
stroke risk (OR = 0.815; 95% CI: 0.734–0.906; p
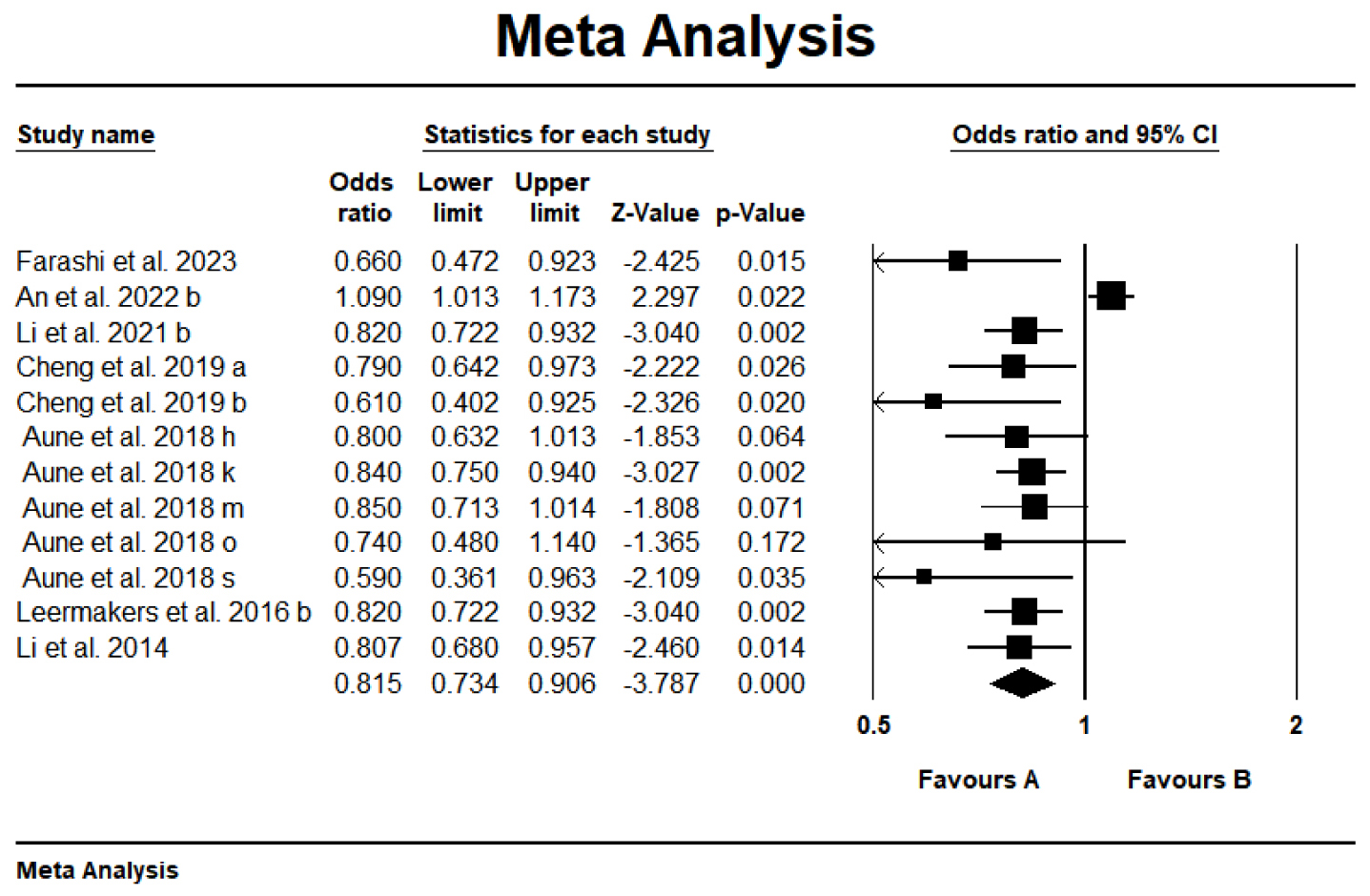 Fig. 3.
Fig. 3.
Forest plot of the effect of carotenoids on stroke. a-s, different letters represent different Meta-analyses selected from an article.
The methodologically rigorous systematic review was performed on the limited
available meta-analyses evaluating carotenoid consumption and myocardial
infarction/ischemic heart disease, with the objective of deriving pooled effect
estimates. This umbrella review identified four meta-analyses reporting
non-significant associations between carotenoids and two key cardiovascular
conditions: myocardial infarction and ischemic heart disease (OR = 0.866; 95%
CI: 0.727–1.031; p = 0.105, Fig. 4), characterized by substantial
heterogeneity (I2 = 0.928, p
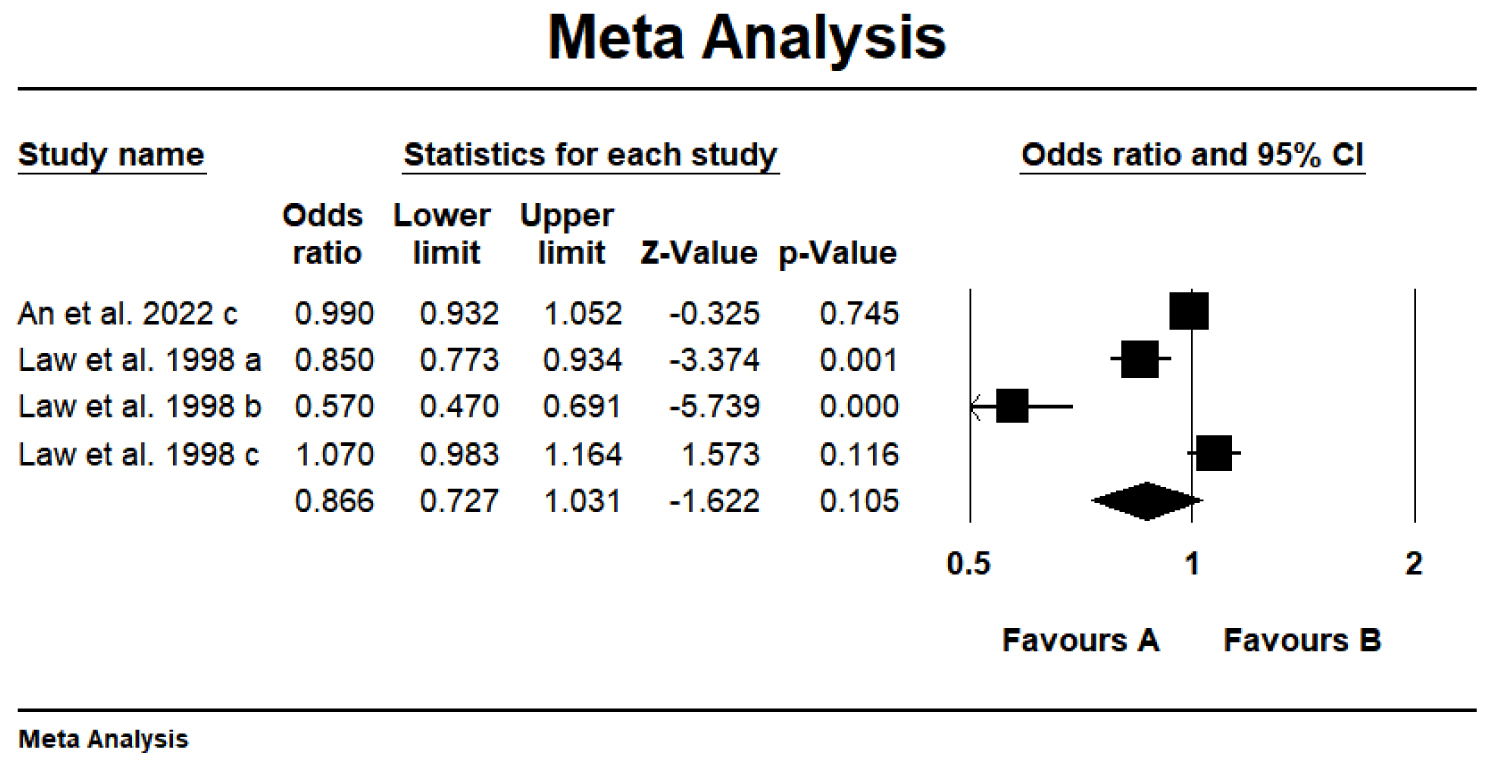 Fig. 4.
Fig. 4.
Forest plot of the effect of carotenoids on myocardial infarction/ischemic heart disease. a-c, different letters represent different Meta-analyses selected from an article.
Additional subgroup analyses were implemented to investigate source-specific effects of carotenoids on CVD outcomes. No significant effect modifications were observed by dietary intake (OR = 0.859; 95% CI: 0.820–0.900) and serum concentrations (OR = 0.738; 95% CI: 0.676–0.806) of carotenoids. However, supplementation was correlated with significantly increased risks for total CVD (OR = 1.061; 95% CI: 1.037–1.086), coronary heart disease (OR = 1.018; 95% CI: 0.982–1.057), stroke (OR = 1.090; 95% CI: 1.010–1.117), and myocardial infarction/ischemic heart disease (OR = 1.023; 95% CI: 0.949–1.103) (Table 3).
| Type of CVD | Source of carotenoids | Number of meta-analyses | OR (95% CI) | I2 (p value) |
| CVD | Blood | 9 | 0.741 (0.650–0.845) | 0.653 (0.003) |
| Carotenoids intake | 9 | 0.855 (0.789–0.926) | 0.583 (0.014) | |
| Carotenoids supplement | 12 | 1.061 (1.037–1.086) | 0.234 (0.213) | |
| Coronary heart disease | Blood | 5 | 0.806 (0.714–0.910) | 0 (0.408) |
| Carotenoids intake | 11 | 0.862 (0.813–0.914) | 0.492 (0.032) | |
| Carotenoids supplement | 2 | 1.018 (0.982–1.057) | 0 (0.849) | |
| Stroke | Blood | 4 | 0.757 (0.634–0.903) | 0.156 (0.314) |
| Carotenoids intake | 5 | 0.822 (0.771–0.877) | 0 (0.987) | |
| Carotenoids supplement | 1 | 1.090 (1.010–1.117) | NA (NA) | |
| Myocardial infarction/ischaemic heart disease | Blood | 1 | 0.57 (0.47–0.69) | NA (NA) |
| Carotenoids intake | 1 | 0.85 (0.77–0.93) | NA (NA) | |
| Carotenoids supplement | 2 | 1.023 (0.949–1.103) | 0.535 (0.143) |
CI, confidence interval; OR, odds ratio; NA, not available.
This umbrella meta-analysis aimed to synthesize current evidence and deliver a comprehensive review of carotenoid effects on CVD risk, building on previous systematic reviews and meta-analyses. Total 64 eligible meta-analyses from 22 primary studies were incorporated to evaluate the impact of carotenoids on CVD. Total carotenoids demonstrated significant inverse associations with incidence and CVD mortality, coronary heart disease, stroke, myocardial infarction and ischemic heart disease. These findings indicate potential protective effects of carotenoid consumption on CVD despite methodological variations across included studies.
The present umbrella review provided sufficient proof that reinforces evidence for the carotenoids to play a protective impact against the development of CVD. Aune et al. [35] reported negative correlations between carotenoid consumption and the hazard of several health outcomes, including coronary heart disease, stroke, and mortality. Bahonar et al. [50] observed in a 165-patient cohort study that plasma total carotenoid concentrations above median levels was related to a 71% decrease in the hazard of CVD mortality (HR = 0.29, 95% CI: 0.12–0.71). Leermakers et al. [38] demonstrated protective effects of dietary lutein on coronary heart disease (RR = 0.88; 95% CI: 0.80–0.98) and stroke (RR = 0.82; 95% CI: 0.72–0.93). Song et al. (2017) [37] demonstrated a statistically notable negative correlation between lycopene exposure and CVD risk (RR = 0.83, 95% CI: 0.76–0.90), with consistent results in dietary (RR = 0.87; 95% CI: 0.79–0.96) and biomarker-based (RR = 0.74; 95% CI: 0.62–0.87) analyses. Moreover, diet-related lycopene consumption showed protective effects for mitigating coronary heart disease risk (RR = 0.87; 95% CI: 0.76–0.98) and stroke (RR = 0.83; 95% CI: 0.69–0.96) [37]. Further study confirmed these findings in a pooled data analysis of 116,127 study participants, demonstrating a 19.3% decrease in the hazard of stroke with lycopene intake (RR = 0.807; 95% CI: 0.680–0.957) [39].
Despite the protective evidence, conflicting findings exist regarding
carotenoid-CVD associations. Yang et al. [32] reported
Dietary carotenoids possess a physiologically balanced combination of moderate bioavailability, complex synergistic components, and natural dosage, which collectively underpin their protective effects against CVD and chronic illnesses [54]. By contrast, carotenoid supplements—characterized by high bioavailability, simplified composition, and supraphysiological doses—may disrupt redox balance and metabolic regulation, with particularly pronounced impacts in high-risk populations [13]. This discrepancy stems from the inherent difference between natural occurrence and artificial formulation, and the food matrix plays a central role in mediating these variations. Synergistic nutrients in natural foods can enhance the bioactivity of carotenoids while mitigating potential pro-oxidant effects; furthermore, the sustained and moderate plasma concentrations resulting from dietary intake are compatible with the body’s antioxidant defense system. Although supplements offer advantages such as high bioavailability and targeted delivery, they lack this protective mechanism, rendering them prone to inducing oxidative stress or interfering with physiological processes at high doses [55, 56].
Carotenoids may have the potential to reduce CVD risk through a range of
biological mechanisms, specifically by lowering blood pressure, dampening the
levels of pro-inflammatory cytokines, lessening inflammatory markers (e.g.,
C-reactive protein) and enhancing insulin sensitivity within critical metabolic
tissues including the liver, skeletal muscles, and adipose tissue [19].
Furthermore, carotenoids can have the capacity to influence gene expression
levels linked to key cellular metabolic pathways [57]. The protective actions of
carotenoids against chronic illnesses stem from their versatile characteristics,
which include anti-inflammatory, anticoagulant, antiviral, and antioxidant
capacities, as well as directly modulate immune responses [58]. Linnewiel-Hermoni
et al. (2014) [59] documented that carotenoid interventions
significantly reduced C-reactive protein (Weighted Mean Difference (WMD) = –0.54
mg/L, 95% CI: –0.71 to –0.37, p
The antioxidant effect of carotenoids, considered health-promoting agents against atherosclerotic CVD, is one of the potential mechanisms [63]. Milani et al. [19] proposed that lycopene may have a preventive effect against CVD in humans, given that it exhibits notable antioxidant activity in vitro, suppresses cholesterol synthesis, and enhances the degradation of low-density lipoprotein (LDL). Carotenoids quench molecular singlet oxygen and scavenge reactive oxygen species, particularly peroxyl radicals [64]. Moreover, carotenoids promote the localization of Nuclear factor erythroid 2-related factor 2 (Nrf-2) in the cell nucleus and the activation of phase II enzymes to decrease oxidative stress [65]. Through the capacity to neutralize free radicals, carotenoids promote the clearance of cells under oxidative stress, thereby mitigating their deleterious effects. Collectively, carotenoids mediate protective effects against CVD through modulation of lipid profiles, inhibition of lipid peroxidation, and enhancement of antioxidant defense systems, thereby counteracting vascular inflammation, stabilizing membrane integrity during atherosclerotic processes, and reducing CVD risk [66].
The US Preventive Services Task Force emphasized that single carotenoid supplementation may exert complex physiological effects in smokers, with potential dose-dependent deleterious outcomes on CVD risk due to pro-oxidant activity [30]. Concurrently, exceeding tolerable upper intake levels poses universal health risks for the general population [67]. Future studies should systematically examine co-administration of concomitant substances and dose-response relationships associated with adverse health effects, while determining safe carotenoid consumption thresholds through strict human trials.
Our present umbrella review comprehensively collects and evaluates all previously published meta-analyses and systematic reviews to reach a conclusion on the effectiveness of carotenoids in CVD prevention with the highest quality of evidence and minimized bias. However, our current investigation has several limitations that require further consideration. Firstly, the interaction between modifiers such as smoking may bias the calculated effect sizes, which we should take into consideration. Secondly, a systematic selection of studies included in previously published meta-analyses was performed, representing a potential limitation due to possible exclusion of non-identified studies. Thirdly, we were unable to evaluate whether carotenoids supplementation would be beneficial against CVD for populations who are deficient in carotenoids at baseline. Fourthly, in our present umbrella review, multiple meta-analyses incorporated the uniform primary observation-focused research. Fifthly, while total included studies and research are population-based that encompassing cohort studies, case-control studies, and randomized controlled trials (RCTs), they employ distinct research designs and data processing strategies, which could introduce variability into the findings. Sixth, a major limitation involves the inability to establish dose-response relationships between carotenoid exposure and CVD outcomes due to insufficient data availability and substantial inter-study dose variations. Additionally, limited evidence exists regarding individual carotenoid subtypes in specific CVD contexts, thereby introducing potential bias in pooled effect estimates.
This umbrella meta-analysis represents the most comprehensive assessment to date of carotenoid-CVD associations, revealing significant inverse relationships between multiple carotenoids and reduced incidence and mortality of CVD. Despite these findings, current evidence remains inconclusive regarding definitive protective effects, highlighting the need for future studies to mitigate potential biases through stratified subgroup analyses and improve evidence consistency via standardized outcome reporting.
Data will be made available on request.
DG: Writing—original draft, Writing—review & editing, Software, Methodology, Formal analysis, Conceptualization, Data curation. HX: Writing—review & editing, Methodology, Formal analysis, Project administration, Funding acquisition. BH: Software, Data curation. PT: Software, Methodology. YY: Data curation. YZ: Software, Methodology. DP: Methodology, Data curation. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/IJVNR46996.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




