1 Department of Cardiovascular Medicine, The First People’s Hospital of Changzhou, The Third Affiliated Hospital of Soochow University, 213000 Changzhou, Jiangsu, China
†These authors contributed equally.
Abstract
Currently, atherosclerotic cardiovascular disease (ASCVD) is the primary cause of mortality globally. Meanwhile, accumulating evidence continues to implicate gut microbiota and the associated metabolites in the pathogenesis and progression of atherosclerosis. Red yeast rice (RYR), a traditional functional food, contains monacolin K as the primary bioactive component, which exerts statin-like lipid-lowering effects. Thus, RYR plays a protective role in regulating lipid metabolism and preventing ASCVD through multiple pathways, including improving endothelial dysfunction, attenuating oxidative stress, and inhibiting inflammatory responses. Therefore, this review summarizes current evidence regarding the regulatory effects of RYR and the associated preparations on hyperlipidemia and ASCVD. Furthermore, we highlight the protective role of RYR in the management of hyperlipidemia, with particular emphasis on the capacity of RYR to modulate the gut microbiota and the associated metabolites, thereby exerting anti-atherosclerotic effects. By elucidating the intricate interactions between RYR and the gut microbiota, this review provides novel insights into the therapeutic potential of RYR and reveals possible roles for the gut microbiota in ASCVD prevention.
Keywords
- red yeast rice
- atherosclerosis
- gut microbiota
The fundamental pathophysiological mechanism underlying the development and progression of atherosclerotic cardiovascular diseases (ASCVD) is atherosclerosis, a chronic condition that poses a significant threat to human health [1]. The core mechanism of atherosclerosis begins with impaired endothelial function, leading to the accumulation of modified lipoproteins (e.g., oxidized low-density lipoprotein, ox-LDL) in the subendothelial space. This process triggers immune cell aggregation and chronic inflammation, ultimately resulting in the formation of fibrous plaques that elevate the risk of vascular stenosis or rupture. According to an examination of trends of the Global Burden of Diseases Study (GBD) 2019, the prevalence of each of the three clinical signs of atherosclerosis steadily increased globally from 1990 to 2019, despite notable improvements in prevention and treatment methods [2]. Therapies aimed at conventional atherosclerosis risk factors have notably lowered the mortality of ASCVD, but the cumulative benefit has plateaued, and identifying novel treatment targets for atherosclerosis continues to be a formidable task [3]. In addition to traditional risk factors such as elevated low-density lipoprotein cholesterol (LDL-C), hypertension, smoking and diabetes, other non-traditional factors contributing to atherosclerosis, including disrupted sleep patterns, gut microbiota, air contamination, and environmental stress, have also gained attention [4].
The gut microbiota axis metabolites directly influence the onset and progression of atherosclerosis [5]. Recent studies have shown that gut microbiota is a key predisposing factor for atherosclerosis, contributing to disease progression through multiple mechanisms. For example, intestinal microbial metabolites such as trimethylamine N-oxide (TMAO) have been shown to exert pro-atherogenic effects [6], and changes in microbial flora can affect lipid absorption, vascular inflammation and oxidative stress level [7]. A comprehensive study conducted by Shi et al. [8], combining clinical observations and animal experiments, has demonstrated that dysbiosis, or imbalance in gut microbial composition, has been linked to increased intestinal permeability, systemic inflammation, and oxidative stress, exacerbating atherosclerotic plaque formation. Modulation of the intestinal microbiota (such as probiotics, prebiotics or antibiotics) has demonstrated protective effects against atherosclerosis in preclinical models, highlighting intestinal microorganisms as an important target for disease intervention [9]. The potential of natural products (NPs) to reshape the gut microbiota ecosystem by promoting the growth of healthier microbial species and limiting proatherogenic metabolites [10], may provide a new strategy for ASCVD. Chinese red yeast rice (RYR), as traditional Chinese fermented food, is produced by fermenting rice with Monascus, has gained attention for its lipid-lowering properties, primarily attributed to monacolin K, a bioactive compound chemically identical to lovastatin [11]. A randomized controlled trial has shown that RYR lowers LDL-C by 15–25% within 6–8 weeks, an effect comparable to that of low-dose statins, thereby establishing RYR as a widely adopted nutraceutical for cholesterol management [12]. RYR could regulate the lipid metabolism and be a functional food in the prevention of ASCVD by reducing endothelial apoptosis, improving the bioactivities of endothelial progenitor cells, inhibiting oxidative stress and inflammation [13]. Popularly used RYR-containing commercial Chinese polyherbal preparations (CCPPs) include the Zhibitai capsule, Zhibituo capsule, and Xuezhikang capsule. The regulatory effect of RYR and RYR preparations on the gut microbiota is not yet clear. Therefore, this review details how RYR and its preparations effectively mitigate atherosclerosis by focusing on the gut microbiota. These findings may pave the way for novel therapeutic strategies leveraging RYR and gut microbiota interactions to combat cardiovascular disease.
RYR exerts its lipid-modulating effects through a diverse array of bioactive
compounds, the composition and variability of which are critical to its efficacy
and safety (Table 1) [14, 15]. These compounds include polyketides, unsaturated
fatty acids, phytosterols, pigments, and condensed tannins [16]. Additionally, a
review has shown that Monascus-fermented items contain dimerumic acid and
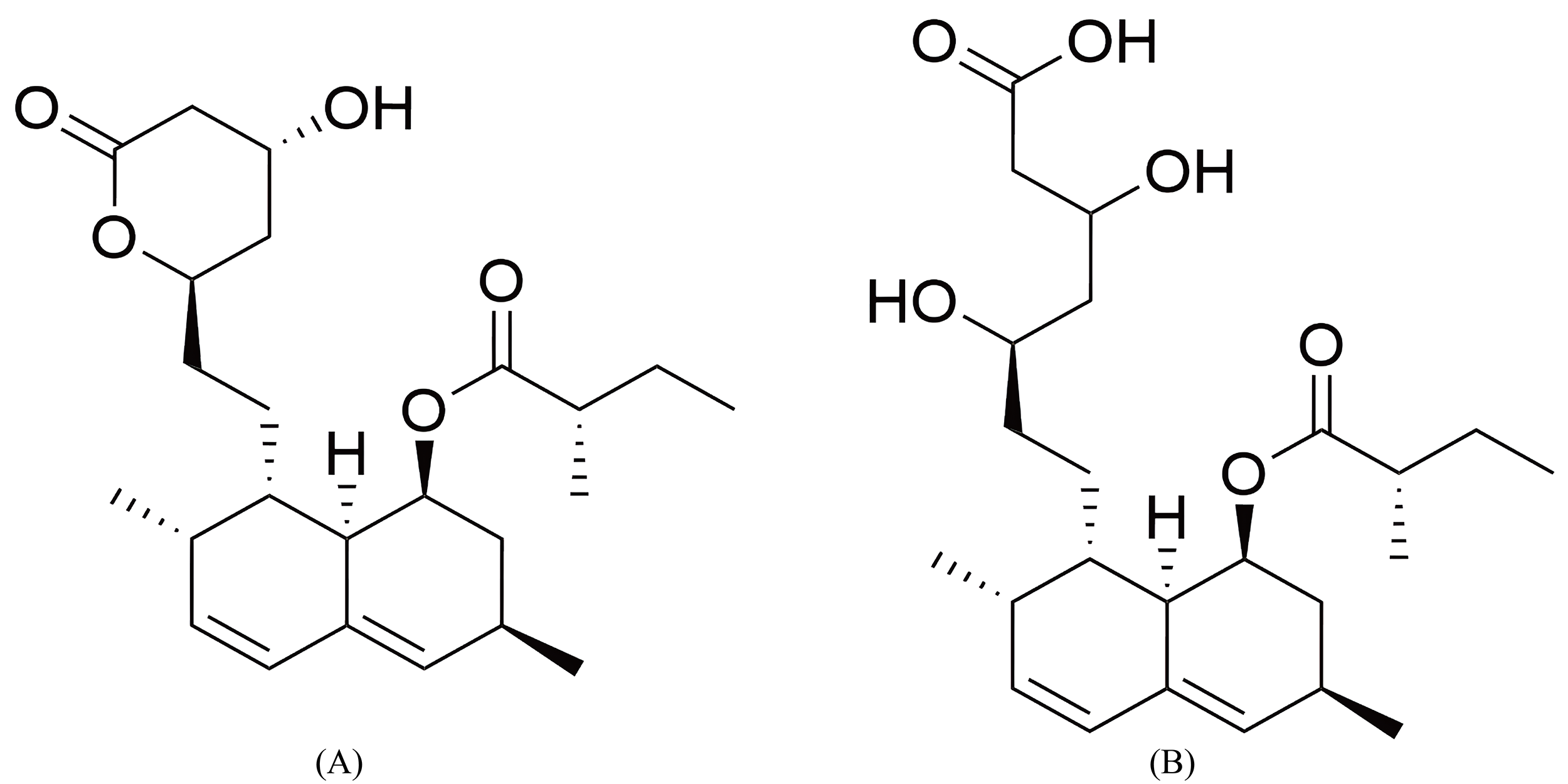 Fig. 1.
Fig. 1.
Comparative structures of monacolin K and lovastatin. (A) Monacolin K’s lactone form (which shares the same structure as lovastatin). (B) Monacolin K’s acid form. The figure was created with ChemDraw 2022.
| Composition | Including | Content range |
| Carbohydrate | About 73.4% | |
| Protein | About 5.8% | |
| Fiber | About 0.8% | |
| Mineral | Phosphorus, sodium, calcium, iron, magnesium, etc. | Trace amount |
| Phytosterol | Mainly for ergosterol | Trace amount |
| Polyketides | Mainly for Monacolin | 0.05%–7.48% |
| Fatty acids | Saturated fatty acids (palmitic acid, stearic acid, peanut acid) and unsaturated fatty acids (oleic acid, linoleic acid, linolenic acid) | About 3% |
| Pigments | Ankaflavin, monascin, rubropunctatin, monascorubrin, rubropunctamine and monascorubramine | About 0.3% |
| Condensed tannins | Trace amount | |
| Dimerumic acid | Trace amount | |
| Trace amount |
As observed in clinical trials, LDL-C levels decreased by approximately 15% to 34% from baseline after at least 4 weeks of RYR therapy [9]. Substantial reductions in total cholesterol (TC) were also observed. However, the effects of RYR on high-density lipoprotein cholesterol (HDL-C) and triglycerides (TG) were generally modest and inconsistent [21, 22]. A few studies documented notable declines in apolipoprotein B levels [23], as well as small, mostly non-significant rises in apo-A1 when using RYR [24]. In a long-term study, the safety and efficacy of a dietary supplement containing RYR along with policosanols, coenzyme Q10, astaxanthin, and folic acid were evaluated in 84 adolescents with moderate-to-severe primary hypercholesterolaemia [25]. The consumption of the nutraceuticals resulted in a remarkable decrease in TC, LDL-C, non-HDL-C, and apolipoprotein B levels 6 months later, and this effect was sustained with long-term use (16 months).
RYR extracts are among the most effective nutraceuticals for cholesterol reduction currently available [26]. A double-blind, placebo-controlled, randomized clinical trial was conducted to determine whether a short-term treatment with 10 mg monacolin derived from RYR could improve high-sensitivity C-reactive protein (hs-CRP), and matrix metalloproteinases 2 and 9 (MMP-2 and MMP-9) levels [27]. Patients who received monacolin treatment demonstrated significant reductions in TC, LDL-C, non-HDL-C, hs-CRP, MMP-2, and MMP-9. In young patients with a low-to-moderate CV risk profile, a one-year treatment regimen of RYR combined with coenzyme Q10 resulted in lipid-lowering effects, a reduction in hs-CRP, and improvements in mean carotid intima-media thickness (IMT) and carotid elasticity [28]. A meta-analysis of four trials, comprising 220 patients in the “RYR plus conventional therapy group” and 204 patients in the conventional therapy group, showed that the “RYR plus conventional therapy” significantly reduced their CRP levels [29]. Existing data have demonstrated the impact of RYR and its related products on enhancing endothelial function, particularly in improving endothelial reactivity, preventing endothelial progenitor cells apoptosis, reducing oxidative stress and inhibiting inflammation [13].
Although RYR has been proposed as a possible option for lowering cholesterol and preventing CVD, its efficacy remains debated. This is mainly due to the significant differences in the amount of monacolin K (which can range from 0.09 to 5.48 mg per 1200 mg in some studies) [30]. In terms of safety, a meta-analysis of 53 randomized controlled trials showed that RYR did not increase the risk of musculoskeletal disorders [31]. In 2018 the European Food Safety Authority (EFSA) evaluated the safety of monacolin in RYR supplements and concluded that 10 mg/day of RYR-derived monacolins in supplements raises major safety concerns, and individual cases of severe adverse reactions have been reported at an intake of RYR-derived monacolins as low as 3 mg/day [14]. Rhabdomyolysis and hepatotoxicity are the two predominant adverse effects. This assessment underscores the potential toxicological risks associated with monacolin K and has prompted calls for stricter regulation of RYR supplements [15].
As monacolin K and lovastatin work by inhibiting the rate-limiting enzyme in the
cholesterol biosynthesis pathway, they may cause mitochondrial malfunction in
muscle cells, which can result in rhabdomyolysis and injury to the muscle cells
[32]. Meanwhile, Monacolin K needs to be metabolized by the liver, and may also
cause liver damage by affecting oxidative stress and inflammatory responses
within liver cells (such as the nuclear factor kappa-B (NF-
Growing evidence suggests that gut microbiota (GM) and its byproducts can directly influence on the onset and course of atherosclerosis; a phenomenon now termed the GM-artery axis [37]. The gut microbiome compositions in ASCVD were found to differ significantly between healthy control in a case-control study involving 218 patients with ASCVD and 187 healthy participants [38]. The abundance of Enterobacteriaceae and Streptococcus in ASCVD was higher, as well as an increase in the copy numbers of bacterial genes that encode enzymes involved in TMAO production and a rise in TMAO levels, which contributed to the deviation from a healthy state in individuals with ASCVD. The microbial metabolites, especially the two crucial and compelling GM-derived secondary products, TMAO and short-chain fatty acids (SCFAs), have gained significant recognition on atherosclerosis pathophysiology [39, 40]. Traditional Chinese medicine (TCM) and its active ingredients have shown great therapeutic potential in ASCVD [41]. TCM can exert an anti-atherosclerosis effect by influencing GM-derived metabolites, such as decreasing the generation of TMAO and facilitating bile acid (BA) metabolism [42]. For example, berberine can inhibit the activities of bacterial enzymes or coenzymes in choline-TMA-TMAO axis, lowering the quantity of TMA or TMAO in use [43]. The pro-atherogenic metabolite TMAO is generated through a specific host-microbial co-metabolic pathway. Dietary nutrients rich in phosphatidylcholine and L-carnitine are first metabolized by gut microbial enzymes into TMA in the intestine. Upon absorption, TMA is transported to the liver where it is oxidized by hepatic flavin monooxygenases (FMOs), primarily FMO3, to form TMAO. Elevated systemic TMAO levels promote atherosclerosis through mechanisms including impaired cholesterol metabolism, enhanced foam cell formation, and platelet hyperreactivity [44].
Given that RYR exerts statin-like effects on LDL-C production, it has some non-lipid lowering effects such as direct vascular protection, anti-inflammatory, and anti-atherosclerotic effects. Few studies have investigated the interaction between RYR and gut microbiota. One research investigated the impact of RYR on blood lipid levels in rats fed with high-fat diet (HFD) and the mechanisms underlying its lipid-lowering effects [45]. Compared with the HFD group, the RYR group showed a decrease in the relative abundance of Firmicutes by 18.91%, while Bacteroidetes and Verrucomicrobia rose by 75.69% and 37.03%, and the Firmicutes/Bacteroidetes ratios declined. Following RYR intervention, the abundance of the Bacteroidales_S24-7_group in rat intestine increased by 61.85% compared with the HFD group. Conversely, the abundance of Lactobacillaceae decreased by 23.12%. The increase in Bacteroidales_S24-7_group can mitigate obesity brought on by HFD [46], which may be a key mechanism underlying the ability of RYR to reduce obesity caused by HFD. Meanwhile, the abundance of Lachnospiraceae decreased by 23.78% in RYR groups compared to the HFD. The abundance of Prevotellaceae increased by 18.37% in the RYR group. Prevotellaceae is widely associated with high dietary fiber intake, and the increase in Prevotellaceae abundance may be attributed to the high content of dietary fiber in the RYR-supplemented diet.
In one atherosclerotic mouse model fed with HFD, it was observed that administration of RYR markedly reduced plaque formation as well as TC and LDL-C levels [47]. RYR treatment also increased intestinal villus height, suggesting its potential to ameliorate HFD-induced intestinal barrier disruption. Western blot analysis targeting the tight junction proteins also revealed that the intestinal tissue of RYR group exhibited significantly higher expression levels of junctional adhesion molecule 1 (JAM-1) and occludin proteins. Furthermore, RYR treatment caused notable changes in the organization of the gut microbiota and reduced the relative abundance of Alistipes and Flavonifractor, which were positively correlated with LDL-C and plasma cholesterol levels. Previous research has indicated that a higher presence of Alistipes is linked to type-2 diabetes in heart failure patients [48]. In contrast, RYR treatment increased the abundance of Anaeroplasma, a genus inversely associated with plasma LDL levels and recognized for its atheroprotective role [49]. Collectively, these findings suggest that RYR may protect against HFD-induced atherosclerosis by modulating the composition and diversity of the gut microbiota. The changes in gut microbiota composition caused by RYR and its metabolites are summarized in Table 2 (Ref. [45, 47, 50, 51, 52, 53, 54]).
| Animal models | Dosage | Key findings | Refs. |
| Hyperlipidemia rats | 6 mg/kg·d RYR (contains 3% lovastatin), i.g. for 8 weeks | • Decreased the Firmicutes/Bacteroidetes ratio. | Yang et al. [45] |
| • Increased the abundance of Bacteroidales_S24-7_group (by 61.85%). | |||
| • Modulated the abundances of Lachnospiraceae and Prevotellaceae. | |||
| D-galactose-induced aging mouse model | RYR diet [containing 5% (w/w)] for 8 weeks | • Restored beneficial genera downregulated by D-galactose (e.g., Lactobacillus, Rikenellaceae). | Huang et al. [50] |
| • Reduced genera enriched by D-galactose (e.g., Akkermansia, Helicobacter). | |||
| Atherosclerotic mice model fed with HFD | 0.34 g/kg·d RYR, oral gavage for 12 weeks | • Lowered abundance of pro-atherogenic genera: Alistipes, Barnesiella, and Flavonifractor. | Dong et al. [47] |
| • Increased abundance of beneficial genera: Bacteroides, Anaeroplasma. | |||
| ApoE-/- mice fed with HFD | 0.3 g/kg·d solution made from natto red yeast rice (NR) capsules, oral gavage for 12 weeks | • Reduced the relative abundance of the phylum Actinobacteria. | Zhou et al. [53] |
| • Reduced genera including Coriobacteriaceae_UCG-002, Bacteroides and Lactococcus. | |||
| Rats fed with HFD | Daily gavage of 20 mg/kg·d MYPs, MRPs or MOPs for 8 weeks | • Increased the abundance of Ruminococcus and Clostridium. | Zhou et al. [51] |
| C57BL/6J mice fed with HFD | Red yeast rice polysaccharide for 8 weeks, low-dose group: 10 mg/day; high-dose group: 20 mg/day | • High-dose RYR inhibited HFD-induced increase in Firmicutes/Bacteroidetes ratio. | Qi et al. [52] |
| • Increased probiotics: Bifidobacterium, Akkermansia. | |||
| Salmonella enterica-induced intestinal inflammation mice model | RYR diet [containing 5% (w/w)] for 4 weeks | • Increased diversity and restored homeostasis of intestinal flora. | Huang et al. [54] |
| • Increased the abundance of Lactobacillaceae, Lachnospiraceae. | |||
| • Lowered abundance of Ruminococcaceae and Helicobacteraceae. |
Abbreviations: RYR, red yeast rice; HFD, high fat diet; i.g., intragastric; w/w, weight/weight; MYPs, Monascus yellow pigments; MRPs, Monascus red pigments; MOPs, Monascus orange pigments.
Microbiota composition of the RYR diet aging group and the basal chow aging
group was compared to determine the effect of RYR dietary intervention on the GM
of aging mice induced by D-galactose [50]. Analysis of
According to reports, the fermented RYR contains a variety of secondary metabolites with distinct physiological and therapeutic roles, including monascus pigments (MPs) and GABA. Different MPs induced significant changes in the overall structure of the intestinal microbiota, which partially ameliorated the gut dysbiosis of gut microbiota induced by HFD [51]. One of the main phylotypes, the abundance of Ruminococcus and Clostridium was significantly reduced by HFD but was notably increased by Monascus yellow pigments (MYPs, mainly monascin and ankaflavin) supplementation. Moreover, the abundance of Clostridium and Oscillibacter was significantly elevated by Monascus red pigments (MRPs) administration. The genus Enterococcus sp. was increased by HFD but down-regulated in the MYPs and MRPs-treated group. As naturally occurring components of RYR, polysaccharides also show significant anti-tumor, blood pressure-lowering, and antioxidant effects, which may contribute to their lipid-regulating effects [55]. A purified highly abundant polysaccharide of RYR, named PRY1-1, can improve HFD-induced intestinal barrier dysfunction via promoting tight junction protein production [52]. The increase in the Firmicutes/Bacteroidetes ratio induced by HFD was greatly prevented by a high-dose PRY1-1 intervention, and the abundance of certain probiotics, including Bifidobacterium and Akkermansia, rose dramatically. Following a high-dose PRY1-1 treatment, the levels of all identified metabolites involved in the butyrate metabolic pathway, such as Maleic Acid, Acetoacetic Acid and L-Glutamic Acid, increased significantly.
Beyond the compositional shifts in gut microbiota, the anti-atherosclerotic
effects of RYR and its preparations are mediated by the modulation of specific
signaling pathways. Monacolin K has been shown to inhibit the NF-
Beyond its primary lipid-lowering component monacolin K, RYR contains a spectrum of non-monacolin bioactive compounds—such as pigments, isoflavones, and polysaccharides. Although present in varying or relatively low quantities, these components may act synergistically with monacolin K to enhance its biological effects [26]. The ability of RYR to alter gut microbiota and intestinal barrier function provides a multifaceted therapeutic strategy for ameliorating dyslipidemia, reducing inflammation and oxidative stress, and preventing age-related and diet-induced metabolic disorders. The active ingredients in RYR, including monacolin K, Monascus pigments, and polysaccharides, may play a synergistic lipid-lowering role in improving lipid metabolism disorders, inhibiting inflammation and oxidative stress by regulating intestinal microbiota structure and enhancing intestinal barrier function by upregulating the expression of tight junction proteins Occludin, Claudin-5 and zonula occludens-1 (ZO-1) (Fig. 2). However, the trends in the change of gut microbiota species and abundances after RYR intervention differ across various studies. For example, the abundance of Firmicutes/Bacteroidetes, Lachnospiraceae, and Verrucomicrobia, as well as their respective implications varies. Moreover, one in vitro study showed that gut microbiota may have hampered the effects of monacolin K on lipids because they catabolized it rather of converting it into the b-hydroxy acid form [56]. The functional metabolites generated from the gut microbiota following RYR and its secondary metabolites intervention should be further characterized using targeted metabolomics and validated in additional in vivo studies. A comprehensive investigation into the synergistic effects of its diverse bioactive components could better illuminate RYR’s potential as a functional food or therapeutic agent for lipid management, gut health, and broader metabolic disorders. It is important to acknowledge the heterogeneity in the reported impacts of RYR on specific microbial taxa such as Firmicutes/Bacteroidetes ratio and Lachnospiraceae. These apparent inconsistencies likely stem from critical variations in experimental conditions, including the composition of RYR preparations such as monacolin content, presence of pigments/polysaccharides, the animal model and its baseline microbiota, and the dosage and duration of the intervention. These inconsistencies likely reflect variations in experimental conditions (e.g., RYR composition, animal model, dosage) rather than negating RYR’s prebiotic potential. Rather than negating RYR’s prebiotic potential, they underscore the context-dependent nature of its action and highlight the need for standardized human clinical trials to establish definitive conclusions.
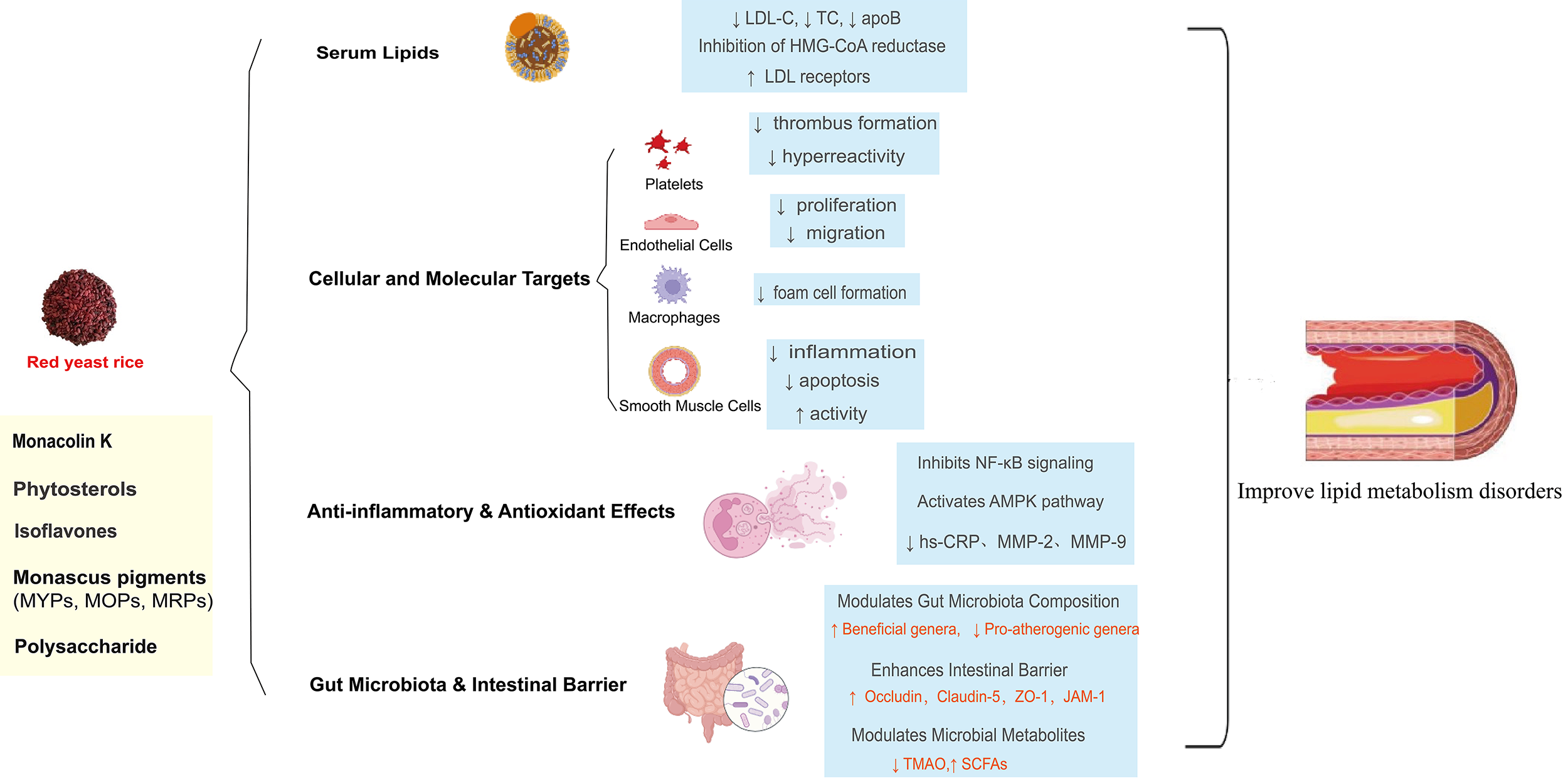 Fig. 2.
Fig. 2.
Multitarget mechanism of RYR in improving lipid metabolism
disorders. MOPs, Monascus orange pigments; MYPs, Monascus yellow pigments; MRPs,
Monascus red pigments; LDL-C, low-density lipoprotein cholesterol; TC, total
cholesterol; apoB, apolipoprotein B; NF-
RYR-containing CCPPs, authorized by the Chinese State Food and Drug Administration, include the Zhibitai, Zhibituo, and Xuezhikang capsules [57]. RYR-containing CCPPs alone or combined with statins appear to improve lipid profiles and have clinical efficacy in patients with dyslipidemia. The composition, dosing, clinical efficacy, and safety profiles of these preparations are summarized in Table 3 (Ref. [30, 36, 58, 59, 60]).
| Preparation | Key components | Daily dose | Lipid-lowering efficacy | Safety notes |
| Zhibitai | RYR, CPB, AR and AMR | 480 mg | Vs. conventional statin therapy: Achieved comparable reductions in TC, TG, LDL‑C, and comparable increases in HDL‑C; Combined use of statins can reduce LDL-C by an additional 20.9% [59] | Vs. conventional therapy group: Showed fewer late adverse events and differences in liver function markers [58] |
| Xuezhikang | RYR extract + amino acids, fatty acids, flavonoids | 1200 mg | Vs. placebo: LDL‑C decreased by 27%, non‑HDL‑C decreased by 24% [60] | Vs. pure lovastatin: Carries a lower risk of muscle toxicity [30]; its daidzein content may ameliorate statin-induced myopathy [36] |
| Zhibituo | RYR extract (Lovastatin + acid form) | 480–3150 mg [59] | Vs. other RYR preparations: Ranked as the most effective for modulating TG and HDL‑C levels [59] | Vs. other RYR preparations: Exhibited a similar safety profile with no significant increase in adverse events [59]. |
Abbreviations: TC, total cholesterol; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; CPB, Crataegus pinnatifida Bunge; AR, Alisma plantago-aquatica subsp. orientale (Sam.) Sam.; AMR, Atractylodes macrocephala Koidz; RYR, red yeast rice.
Except of RYR, Zhibitai contains three other medicinal herbs known for their lipid-modulating properties: Crataegus pinnatifida Bunge (CPB) (Rosaceae; crataegi fructus) (hypolipidemic effects, anti-inflammatory), Alisma plantago-aquatica subsp. orientale (Sam.) Sam. (AR) (Alismataceae; alismatis rhizoma) (anti-hyperlipidemia, antioxidative, anti-inflammatory), Atractylodes macrocephala Koidz (AMR) (Asteraceae; atractylodis macrocephalae rhizoma) (neuroprotective effect, anti-inflammatory activity) [58]. A meta-analysis indicated that Zhibitai achieved comparable efficacy to conventional therapy in terms of TG, TC, LDL-C, HDL-C, and overall effectiveness rate [58]. Results from a multi-center clinical trial in patients with coronary heart disease (CHD) or at high risk of CHD have shown that the combination of Zhibitai and statins is equally effective in lowering LDL-C levels compared to high-dose statins alone [61]. In terms of safety, Zhibitai group exhibited fewer late adverse events compared to the control group, with a notable difference in liver function metrics [58].
Both Xuezhikang and Zhibituo are made from RYR. Xuezhikang primarily lowers blood cholesterol levels through monacolins that inhibit HMG-CoA reductase. Each capsule contains 6 mg of total monacolins, along with 25.7 mg of amino acids, 24 mg of unsaturated fatty acids, 0.9 mg of ergosterol, and 0.14 mg of flavonoids [62]. Monacolin K lactone (lovastatin lactone), K hydroxy acid (lovastatin hydroxy acid), L, J, M, and X are among the 13 varieties of monacolins found [45]. A dose of 1200 mg per day of Xuezhikang is equivalent to 10 mg of lovastatin because each tablet of Xuezhikang contains 2.5 mg of lovastatin [63]. The lovastatin in Xuezhikang has a lower crystallinity and a higher dissolution rate in the body compared with chemically synthesized lovastatin, and other components in Xuezhikang act synergistically to reduce hepatic first-pass elimination of its active ingredient lovastatin acid, thereby increasing the bioavailability of lovastatin acid in the liver [64, 65].
Clinical evidence has shown that Xuezhikang could effectively lower LDL-C and reduce the potential for severe coronary artery events and mortality. A study conducted in the United States and China evaluated the effects of Xuezhikang on lipids in patients with dyslipidemia but no CHD (Xuezhikang group, n = 78; placebo, n = 38). In this randomized, double‑blind, placebo‑controlled trial, with percent change in LDL‑C as the primary endpoint, Xuezhikang dramatically decreased LDL‑C levels by 27% (95% CI: 31% to 23%) and non‑HDL‑C levels by 24% (95% CI: 28% to 20%) within 12 weeks of treatment compared to placebo [60]. In addition to reducing LDL-C, Xuezhikang was demonstrated to reduce the level of plasma TG [66]. Xuezhikang may change lipoprotein subfractions, which include a drop in small LDL particles and a rise in big LDL particles, along with a decrease in ox-LDL, small LDL-C, and apolipoprotein B and an increase in HDL-C [63, 67]. In the CCSPS trial—a large-scale, randomized, double‑blind, placebo‑controlled secondary prevention study—4.5 years of Xuezhikang treatment significantly reduced the composite primary endpoint of major coronary events (hazard ratio [HR] 0.55, 95% CI: 0.43–0.70), corresponding to a 45% relative risk reduction. It also reduced all‑cause mortality (HR 0.67, 95% CI: 0.53–0.84) and cardiovascular mortality (HR 0.70, 95% CI: 0.53–0.93) compared to placebo [68].
Zhibituo primarily contains lovastatin and lovastatin acid as its main bioactive
components, with a reported lovastatin content of approximately 2.7 g/kg [59]. A
meta-analysis assessing the efficacy and safety of RYR formulations in primary
hyperlipidemia found no significant differences in lipid-modifying effects
between Xuezhikang and Zhibituo [69]. No discernible variations in the lipid
profiles were observed between Xuezhikang and Zhibituo. Another network
meta-analysis, involving 47 RCTs and 4824 participants, used a Bayesian framework
to compare the lipid-lowering efficacy of different RYR preparations, while
accounting for variations in monacolin K content. To enable fair cross‑product
comparisons, it accounted for variations in monacolin K content across
preparations—for example, Xuezhikang contains approximately 11.1 g
Currently, limited research has examined the influence of RYR preparation on the
GM and its relative metabolites. The potential mechanism of Xuezhikang and Rosa
roxburghii Tratt fruit vinegar (RFV) in addressing obesity and hyperlipidemia was
examined in an animal experiment. In such preclinical studies, the impact on the
GM-artery axis is typically evaluated using a combination of microbiome profiling
and host metabolic analyses. Microbiome alterations are assessed via 16S rRNA
gene sequencing, with metrics including alpha diversity (e.g., Shannon index) and
beta diversity (e.g., PCoA based on Bray–Curtis distances), alongside reporting
of specific taxa shifts at phylum, family, and genus levels. To link microbial
changes to host physiology, bile acid profiling is performed using liquid
chromatography–tandem mass spectrometry (LC-MS/MS) to quantify individual bile
acid species in feces and liver. Concurrently, the expression of key bile acid
signaling receptors—farnesoid X receptor (FXR) and Takeda G-protein coupled
receptor 5 (TGR5)—and their downstream target genes (e.g., SHP,
BSEP, CYP7A1) is evaluated via qRT-PCR and Western blot to
elucidate pathway-level mechanisms [70, 71]. Applying these methods, the study
found that in HFD-induced obese mice, Xuezhikang intervention reversed the marked
rise in Firmicutes and decline in Bacteroidetes, thereby
correcting the severe imbalance in the Firmicutes/Bacteroidetes ratio
[59]. Furthermore, Xuezhikang (at 1200 mg
The gut microbiota constitutes a highly complex ecosystem of interacting microorganisms. There is growing evidence that GM and its metabolites directly influence the pathogenesis and progression of atherosclerosis. Gaining a more in-depth comprehension of the molecular mechanisms underlying the GM-artery axis will facilitate the exploration of GM’s therapeutic potential and provide a novel entry for the mechanism study of the TCM in the prevention and treatment of atherosclerosis-related diseases. The extracts of RYR are one of the most effective cholesterol-lowering nutraceuticals currently. RYR’s ability to modulate gut microbiota and its metabolites offers a multifaceted approach to improving dyslipidemia, lowering oxidative stress and inflammation. Currently, there is limited data on the effects of RYR preparations on the gut microbiota and related metabolites. More in vivo studies and targeted metabolomics approach should be applied to investigate the functional metabolites derived from the gut microbiota after RYR and its preparations intervention. Fully investigating the synergistic effects of RYR and its various bioactive components may highlight its potential as a potent therapeutic agent for lipid management and ASCVD. Despite the compelling preclinical evidence summarized in this review, it is important to note that direct evidence demonstrating the modulation of gut microbiota by RYR and its preparations in human clinical studies remains limited. Most current findings are derived from animal models, and their translational relevance to humans warrants further validation. Therefore, future well-designed human intervention trials, incorporating metagenomic sequencing and targeted metabolomic analyses, are imperative to unequivocally establish the causal relationships between RYR consumption, specific microbial shifts, and consequent improvements in host metabolic health. Such studies will bridge the current knowledge gap and solidify the role of RYR in cardiovascular health through the gut microbiota-axis. Regarding the safety profile of monacolin K, we recognize the concerns raised by regulatory bodies such as the European Food Safety Authority, particularly at higher intake levels. While standardized extracts like Xuezhikang appear to have a distinct pharmacokinetic and safety profile potentially due to synergistic components, the variability in commercial RYR supplements necessitates caution. Future studies should prioritize well-designed human trials to clarify the dose-response relationship, identify risk factors for adverse effects, and establish clear safety guidelines for long-term use.
FJ conceived and drafted the initial manuscript. YM and JX collected relevant literature and revised the manuscript. MZ prepared the figures. All authors contributed to the conception and editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
Chinese Cardiovascular Association-Natural lipid-lowering drugs fund (2023-CCA-NLD-431). Changzhou Health Talents Domestic Training Program (GN2023006). Changzhou Science and Technology Plan (Application-Fundamental Research Special Project, CJ20253080). Changzhou Key Medical Discipline (CZXK202202).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


