1 Department of Pediatrics, Guang’anmen Hospital, China Academy of Chinese Medical Sciences, 100053 Beijing, China
Abstract
Childhood obesity poses a global threat to children’s health, impacting both physical and psychological well-being. This disease results from a complex interplay between genetic and environmental factors. The gut microbiota is an environmental factor that plays a crucial role in the progression of childhood obesity. Diet, as a key lifestyle factor, affect obesity; the gut microbiota may be at a crossroads between them. Several clinical studies have indicated that diet can modulate microbial dysbiosis and gut microbiota metabolic networks, effectively improving childhood obesity. A high-fiber diet and low-saturated fat diet can increase the abundance of beneficial bacteria, such as Akkermansia and Bifidobacterium, and reduce the presence of harmful bacteria, like Proteobacteria. This review elucidates the key mechanisms by which diet modulates the gut microbiota to influence obesity, examining the cascade from alterations in the gut environment and microbial composition to the production of microbial metabolites and their subsequent impact on the host physiological state. Additionally, we summarize clinical trials from the last two decades involving specific dietary patterns, which modulate the gut microbiota and impact the development of childhood obesity. This study provides theoretical and practical insights for comprehensive childhood obesity interventions.
Keywords
- pediatric obesity
- gastrointestinal microbiome
- diet
- dysbiosis
- metabolome
Obesity, characterized by excessive fat accumulation, has reached epidemic proportions, especially among young people [1, 2, 3]. With an estimated 254 million children aged 5–19 years projected to be overweight or obese by 2030, pediatric obesity represents a significant global health crisis [4]. It has detrimental effects on the physical and mental well-being of children and is a powerful precursor to adult chronic diseases such as type 2 diabetes, cardiovascular disease, and non-alcoholic fatty liver disease [5, 6]. In the quest to understand and combat this crisis, emerging evidence points to the gut microbiota—the complex ecosystem of microorganisms within the gastrointestinal tract—as a critical mediator in the pathogenesis of obesity.
The healthy human gut is home to more than 10 trillion microorganisms that form
symbiotic relationships with their host [7]. The gut microbiota and their
metabolites play an important role in maintaining human health. Numerous studies
have confirmed that disturbances in the gut microbiota affect immune, metabolic,
and cognitive functions, and are closely related to obesity, enteritis, and
irritable bowel syndrome [8, 9, 10]. The underlying mechanisms are multifaceted but
often involve impaired gut barrier integrity—which allows for the systemic
translocation of pro-inflammatory molecules like lipopolysaccharide (LPS)—and
an altered profile of critical microbial metabolites, such as the production of
short-chain fatty acids (SCFAs) and the transformation of bile acids. This is
particularly evident in children. A meta-analysis of 63 studies revealed that
pediatric obesity is linked to a state of dysbiosis, characterized by
significantly decreased microbial
The composition of this microbial community is not static; it is highly adaptive to gut environment, with diet being a primary modulating factor [16, 17, 18]. This provides a powerful avenue for intervention. In sharp contrast to the dysbiosis seen in obesity, healthy dietary patterns like the Mediterranean diet are associated with longevity and health benefits, partly by promoting the growth of beneficial microbes such as Clostridium, Bifidobacterium, and Bacteroides [19, 20, 21, 22]. Although the link between diet, the gut microbiota, and childhood obesity is established, the precise mechanisms underlying this interaction remain to be fully elucidated. Therefore, this article aims to review how different dietary patterns affect the gut microbiota and how this process influences the pathology of childhood obesity.
Studies published in the PubMed electronic database between January 1993 and December 2024 were retrieved. We used a comprehensive list of search terms related to obesity, childhood obesity, gut microbiota, and lifestyle (diet and exercise), focusing on their interactions and underlying mechanisms in human and animal models. Search terms used for childhood obesity included “obesity”, “childhood obesity”, “pediatric obesity”, “child obesity”, “childhood overweight”, “adolescent obesity”, “adolescent overweight”. Search terms used for gut microbiota included “microbiome”, “microbiota”, “gastrointestinal microbiome”, “gastrointestinal microbiota”, “gut microbiome”, “gut microflora”, “gut flora”, “gut microbiota”, “intestinal microbiome”, “intestinal flora”, “intestinal microbiota”, “intestinal microflora”, “enteric bacteria”. Search terms used for lifestyle included “diet”, “exercise”, “sedentary”, “healthy”, “high-fat”, “high-fiber”, “energy intake”, “Mediterranean”, “Western”, “dietary patterns”, “carbohydrate-restricted”, “physical exercise”, “physical activity”, “training”. Duplicate and irrelevant studies were excluded and full-text articles were assessed. The clinical trials conducted on diet and exercise, which modulate the gut microbiota and impact the development of childhood obesity, were selected to be discussed in-depth (Fig. 1).
 Fig. 1.
Fig. 1.
Research flowchart.
The human gut microbiota, a complex ecosystem comprising bacteria, archaea, fungi (mainly yeasts), viruses (mainly bacteriophages), and protozoa, is now recognized as a pivotal endocrine organ influencing host health. The two principal bacterial phyla are Bacteroidetes and Firmicutes, with approximately 90% of bacteria localized in the colon, an environment characterized by low oxygen levels and alkaline pH that facilitates the growth of anaerobic communities [23]. This microbial community and its derived metabolites are deeply involved in regulating processes central to obesity pathogenesis, including energy harvesting from the diet, fat storage, insulin resistance, and the modulation of immune and inflammatory responses [24]. Through intricate neuro-immune-endocrine networks, such as the gut-brain, gut-lung, gut-skin, and gut-muscle axes, the microbiota plays a critical role in systemic physiological balance [25, 26, 27]. Given its profound impact on metabolism and inflammation, understanding how this microbial ecosystem is established and how it dysfunctions is crucial for elucidating the mechanisms of childhood obesity [28, 29, 30].
The establishment of the gut microbiota during early life is a critical window that can significantly influence long-term health, including the risk of developing childhood obesity [31, 32]. Therefore, studying the factors that shape microbial composition during infancy is crucial for understanding the underlying mechanisms of this condition. While some studies suggest that microbial colonization may initiate in utero through exposure to microbes in the placenta and amniotic fluid [33, 34], this concept remains controversial [35]. Consequently, birth is widely considered the primary and most significant event for the initial colonization of the infant gut. The mode of delivery is a key determinant of this initial microbial profile. For instance, studies have reported that vaginally delivered neonates, who experience a greater overall mother-to-infant microbial transfer, have a microbiota dominated by Actinomycetes and Bacteroides in the first week of life. In contrast, infants born by cesarean section are primarily colonized by bacteria from the skin and hospital environment, often resulting in an early dominance of Firmicutes [36, 37].
Following the initial colonization at birth, postnatal factors—particularly feeding patterns—become the primary drivers shaping the infant’s gut microbiota. A clear dichotomy is observed between feeding methods: breastfed infants typically exhibit a microbiota rich in beneficial Lactobacillus and Bifidobacteria, whereas formula-fed infants often show a more diverse community dominated by genera such as Clostridium [38]. The introduction of solid foods marks the next major developmental shift, substantially increasing microbial diversity and altering the dominant organisms. This developmental trajectory can be significantly disrupted by external exposures, most notably the use of antibiotics. For instance, a randomized controlled trial demonstrated that antibiotic administration within the first week of life decreased the abundance of beneficial Bifidobacterium spp. while increasing Klebsiella spp. and Enterococcus spp. [39]. The subsequent recovery process often involves an accelerated, yet altered, maturation of the microbiota, characterized by an increase in Clostridium and the persistent suppression of Bifidobacteria dominance [40]. Ultimately, the gut microbiota’s development is a complex interplay of numerous influences. Beyond diet and medication, it is continuously shaped by a broader set of environmental factors, including geographical location, family lifestyle, and the administration of probiotics or prebiotics [41, 42, 43]. Therefore, while host genetics provide the foundational blueprint, these cumulative external exposures during early life are the predominant forces shaping the final composition and metabolic network of the gut microbiota [44].
A growing body of evidence links gut microbiota dysbiosis to the development of childhood obesity. This dysbiosis can originate from early-life events and be modified by later lifestyle factors. Certainly, genetics plays a leading role which has been extensively reported in previous studies [45, 46].
The previous text has detailed how early life experiences influence the gut microbiota. This highlights the importance of identifying specific microbial markers in infancy that may predict future weight gain. In a large birth cohort study (n = 2834), researchers examined the microbiome of 1-month-old infants and tracked their body mass index (BMI) for the first 10 years of life. The findings revealed that early colonization by Bacteroides fragilis was significantly associated with a higher BMI, a correlation that was even more pronounced in the low-fiber intake subgroup [47]. Compared with breastfeeding, formula feeding may increase the risk of being overweight in the first year of life, and the gut microbiota (such as higher microbiota diversity and enrichment of Lachnospiraceae) may contribute to this effect [48].
To elucidate the characteristics of the gut microbiota in obese children, researchers have compared overweight/obese and normal-weight children. A positive correlation exists between the abundance of Firmicutes and BMI [49], whereas the abundance of Bacteroidetes was negatively correlated with BMI [50]. The elevated Firmicutes/Bacteroidetes ratio is thought to be a characteristic alteration of gut microbiota in obese children [12, 51]. Moreover, the abundances of Verrucomicrobia and Bifidobacterium were significantly lower and the abundance of Proteobacteria was higher in obese children than in normal-weight children, as detailed in previous reviews [52, 53].
We have understood that characteristic changes in the gut microbiota are closely related to childhood obesity [54, 55]. Under the influence of various factors in early life, the development of the gut microbiota tends to stabilize but remains dynamic. Subsequently, diet emerge as the primary factors influencing its composition.
Diet can have a direct impact on health and may indirectly affect host health by altering the gut microbiota composition and metabolite production. The indirect effect is mediated through the gut environment, where diet-induced remodeling of the intestinal milieu—including its physical structure, chemical composition, and motility—imposes strong selective pressures on the resident microbial community (Fig. 2). The resulting microbial adaptation is characterized by a shift in metabolic output, favoring either the production of beneficial SCFAs or pro-inflammatory LPS. This differential metabolite profile is a critical determinant of host metabolic homeostasis, ultimately steering the trajectory towards either health or obesity. In this section, we discuss two primary dietary patterns and their distinct impacts.
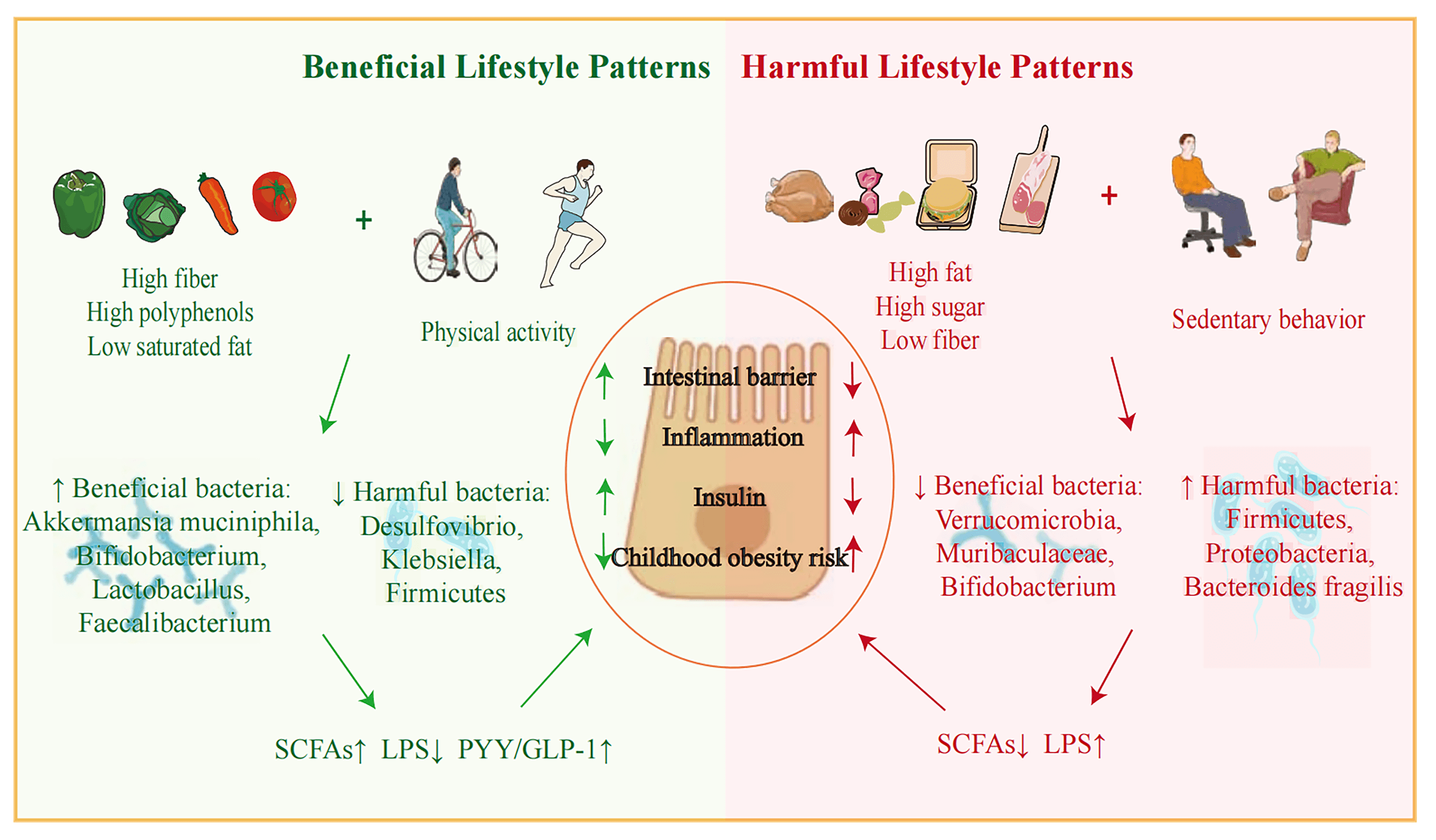 Fig. 2.
Fig. 2.
Diet and lifestyle have been shown to exert indirect effects on
host health by modulating the composition of the gut microbiota and the
production of metabolites. These effects are manifested through alterations in
the gut environment. For instance, dietary intake serves as a source of
substrates for microbial proliferation, influences gastrointestinal acid-base
balance, introduces antioxidants, and impacts the immune system. Diet and
lifestyle lead to modifications in the gut environment, affecting components such
as the gut barrier, mucus layer, gut motility, and the secretion of hormones and
myokines, all of which play pivotal roles in maintaining host-microbial
homeostasis. The figure was created using Adobe Illustrator 2020 (version 24.0; Adobe Inc., San Jose, CA, USA).
Beneficial dietary patterns, such as the Mediterranean and vegetarian diets, exert their positive health effects primarily through two key components that are mediated by the gut microbiota: dietary fibers and polyphenols [54, 55]. Dietary fiber, particularly fermentable polysaccharides, serves as a primary energy source for beneficial intestinal bacteria. Clinical trial have demonstrated that supplementation with soluble fibers like polydextrose increases the abundance of Bifidobacterium [56]. High-fiber diets promote the growth of Lactobacillus, Bifidobacterium, and Akkermansia while suppressing opportunistic pathogens like Desulfovibrio and Klebsiella [57]. The generation of microbial metabolites relies on dietary substrates [58]. Although fermentable polysaccharides cannot be broken down by digestive enzymes, they serve as important energy sources for beneficial bacteria residing in the large intestine. The fermentation of these fibers by the microbiota yields beneficial metabolites, most notably SCFAs. These SCFAs, along with the fermentation process itself, fortify the intestinal barrier by stimulating the production of a thick, protective mucus layer [59, 60]. Conversely, a diet lacking in fiber has been shown in animal models to compromise this barrier and decrease SCFA production [61]. Complementing the effects of fiber, polyphenols and their derivatives, such as phenolic acids, also contribute significantly to gut health. These compounds are known to remodel the gut microbiota, leading to a reduction in harmful metabolites like trimethylamine-n-oxide (TMAO) [62]. Furthermore, they can directly alleviate intestinal inflammation by modulating macrophage activation in a microbiota-dependent manner [63]. Therefore, it is the synergistic effect of dietary fiber and polyphenols—promoting beneficial microbes, enhancing SCFA production, fortifying the gut barrier, and attenuating inflammation—that largely explains the health benefits of these dietary patterns.
In stark contrast to beneficial dietary patterns, Western-style diets-characterized by a high intake of calorie-dense animal meats, processed grains, and sugars—are known to disrupt gut microbiota homeostasis and fuel inflammation [64, 65]. Animal studies consistently show that such diets induce gut dysbiosis, characterized by reduced microbial diversity, an elevated Firmicutes/Bacteroidetes ratio, and an expansion of pro-inflammatory phyla like Proteobacteria [66, 67, 68]. This dysbiosis also involves the depletion of Akkermansia muciniphila, a bacterium essential for gut barrier integrity, and the beneficial, butyrate-producing Muribaculaceae family, collectively contributing to metabolic dysregulation [69, 70]. Furthermore, the high content of animal protein in these diets can lead to a state of putrefactive fermentation in the colon. When undigested complex proteins are metabolized by gut bacteria, they produce a range of noxious byproducts, including branched-chain fatty acids, trimethylamine, phenols, amines, and indoles. These compounds are known to increase luminal pH, which can compromise the intestinal barrier, resulting in a “leaky gut” [71, 72]. Ultimately, the combination of a dysbiotic microbial community and the influx of these pro-inflammatory byproducts triggers the systemic low-grade inflammation and insulin resistance that are central to the pathogenesis of obesity.
While the link between the gut microbiota and adult obesity has been extensively explored, research into pediatric weight-loss interventions remains limited, with most studies being observational in nature. These cross-sectional studies, however, consistently highlight a complex interplay between diet, the gut microbiota, and the development of obesity in children. For instance, one study by Orbe-Orihuela et al. [73] demonstrated how specific diet-microbiota combinations correlate with anthropometric measures. They found that an abundance of Paraprevotella xylaniphila and Bilophila, when paired with high consumption of saturated fat and simple carbohydrates, was associated with an increased waist circumference. Similarly, a high prevalence of Coprococcus catus alongside low protein intake correlated with a larger hip circumference, and an abundance of Holdemania spp. and high protein intake decreased the BMI z-score. In contrast, another large survey of 1111 children revealed a potentially protective role for certain bacteria; a higher abundance of Lactobacillus was associated with a reduced risk of obesity, even in the context of a high-fat, high-carbohydrate diet [74]. This association extends beyond diet-dependent obesity to genetic forms as well. In a study of children and adolescents with Prader-Willi syndrome, the genus Alistipes was negatively correlated with body fat mass, cholesterol, and insulin resistance, and its abundance was lower in those with higher meat intake [75]. Moving beyond static cross-sectional data, a 4-year prospective study provided further insight, identifying individual gut microbiome configurations and long-term dietary habits as key predictors of future childhood obesity. The study concluded that a low-diversity microbiota combined with a high-carbohydrate, high-fat diet promotes an inflammatory state that increases obesity risk, whereas high-intensity physical activity may serve as a protective factor [76]. Collectively, while these observational studies strongly demonstrate an association between dietary habits, the gut microbiota, and childhood obesity, they cannot establish a causal relationship. Rigorously designed longitudinal and interventional trials are therefore essential to confirm these findings and inform future therapeutic strategies.
To explore the temporal effects of dietary interventions, several controlled clinical trials have been conducted in children with obesity. These studies consistently show that lifestyle interventions can lead to reductions in body weight or body fat, accompanied by significant changes in the composition, richness, and metabolic output of the gut microbiota (Table 1, Ref. [77, 78, 79, 80, 81, 82, 83, 84, 85]). A critical question arising from these human trials, however, is whether the observed changes in the gut microbiota are merely associated with weight loss or are causally responsible for the metabolic improvements. To address this question of causality, a landmark study by Zhang et al. [77] utilized germ-free mice. Mice were colonized with intestinal microbiota from children sampled both before and after a dietary intervention. The results were compelling: mice receiving the post-intervention microbiota exhibited lower levels of inflammation and smaller adipocytes, providing strong evidence that the diet-altered gut microbiota can directly confer metabolic benefits.
| Authors/Year/Country | Study design | Diagnosis (Sample size)/Age | Intervention | Outcomes | Conclusion | |
| Huang et al. [78] 2020 | China | Before-after | Obesity (n = 20), 9–16 years old | A calorie-restricted diet for 6 weeks | BMI |
Diet intervention not only reduced body weight but also improved central hemodynamic measures that were associated with altered gut microbiota in adolescents with obesity. |
| Li et al. [79] 2021 | China | Before-after | Prader-Willi Syndrome (n = 17), simple obese (n = 19), 3–16 years old | A high-fiber diet for 30 or 90 days | A significant shift was observed: Pathogen-specific virulence factors genes |
The shifted gut microbial virulence factors, mediated by a high-fiber diet, might lower the host inflammation response. |
| Zhang et al. [77] 2015 | China | Before-after | Prader-Willi Syndrome (n = 17), simple obese (n = 21), 3–16 years old | A diet of whole-grains, traditional Chinese medicinal food and prebiotics for 30 days | Bodyweight |
A diet rich in non-digestible carbohydrates significantly improved the gut microbiota dysbiosis and alleviated genetic and simple obesity. |
| Cho [80] 2021, Lee et al. [81] 2023 | Korea | Before-after | Obesity (n = 36), 7–18 years old | 2-month intervention of diet | The fat loss group (n = 17): Bacteroidetes phylum and Bacteroidia class |
The lifestyle interventions resulted in obesity-related microbial and metabolic alterations in the responder group. |
| Morán-Ramos et al. [82] 2022 | Mexico | Before-after | Obesity (n = 6), 11–14 years old | A 6-week hypoenergetic dietary | A significant shift was observed: Waist circumference |
Personalized gut microbiota profiles may lead to different response to interventions, but it needs to be validated in a wide range of people. |
| Santacruz et al. [83] 2009 | Spain | Before-after | Obesity (n = 36), 13–15 years old | A 10-week calorie-restricted diet | All participants: Bacteroides fragilis and Lactobacillus |
Lifestyle interventions have an impact on gut microbiota composition related to body weight loss, and individualized gut microbiota profiles may result in differential responses. |
| Morgado et al. [84] 2023 | Portugal | Before-after | Obesity and overweight (n = 15), 7–10 years old | A 12-week intervention of nutrition and football | All participants: BMI and BMI z-score |
Nutritional education and physical activity combination can induced limited but significant positive alteration in the gut microbiota composition in overweight and obese children. |
| Aqeel et al. [85] 2025 | USA | Randomized controlled trial | Obesity (n = 33), 6–11 years old | A 4-week high-fiber, low-dairy diet provided through weekly dietitian-guided grocery orders | A potential shift was observed (p = 0.088): Faecalibacterium |
Dietitian-guided food provisioning shows potential for improving diet quality in children with obesity; however, a longer and more sustained intervention is needed to impact weight, food security status, and the gut microbiome. |
Once reshaped by the dietary patterns detailed previously, the gut microbiota mediates its influence on obesity primarily through the production of bioactive metabolites and signaling molecules. As established, diet remodels the intestinal milieu, applying selective pressures that shape microbial composition and function (Fig. 2). The critical link in this diet-microbiota-host axis is the subsequent production of a distinct profile of bioactive microbial metabolites [86]. These compounds act as pivotal signaling molecules that orchestrate host physiology by modulating inflammatory pathways, intestinal barrier integrity, and gut-brain communication, thereby determining the overall metabolic trajectory toward either health or obesity. Studies of the mechanisms behind gut microbiota interactions with obesity rely primarily on animal experiments. This section will elucidate the mechanisms of several key classes of these microbially-derived or -regulated active compounds, including SCFAs, bile acids, LPS, tryptophan metabolites, N-acyl amides, neurotransmitters, and gut hormones (Fig. 3).
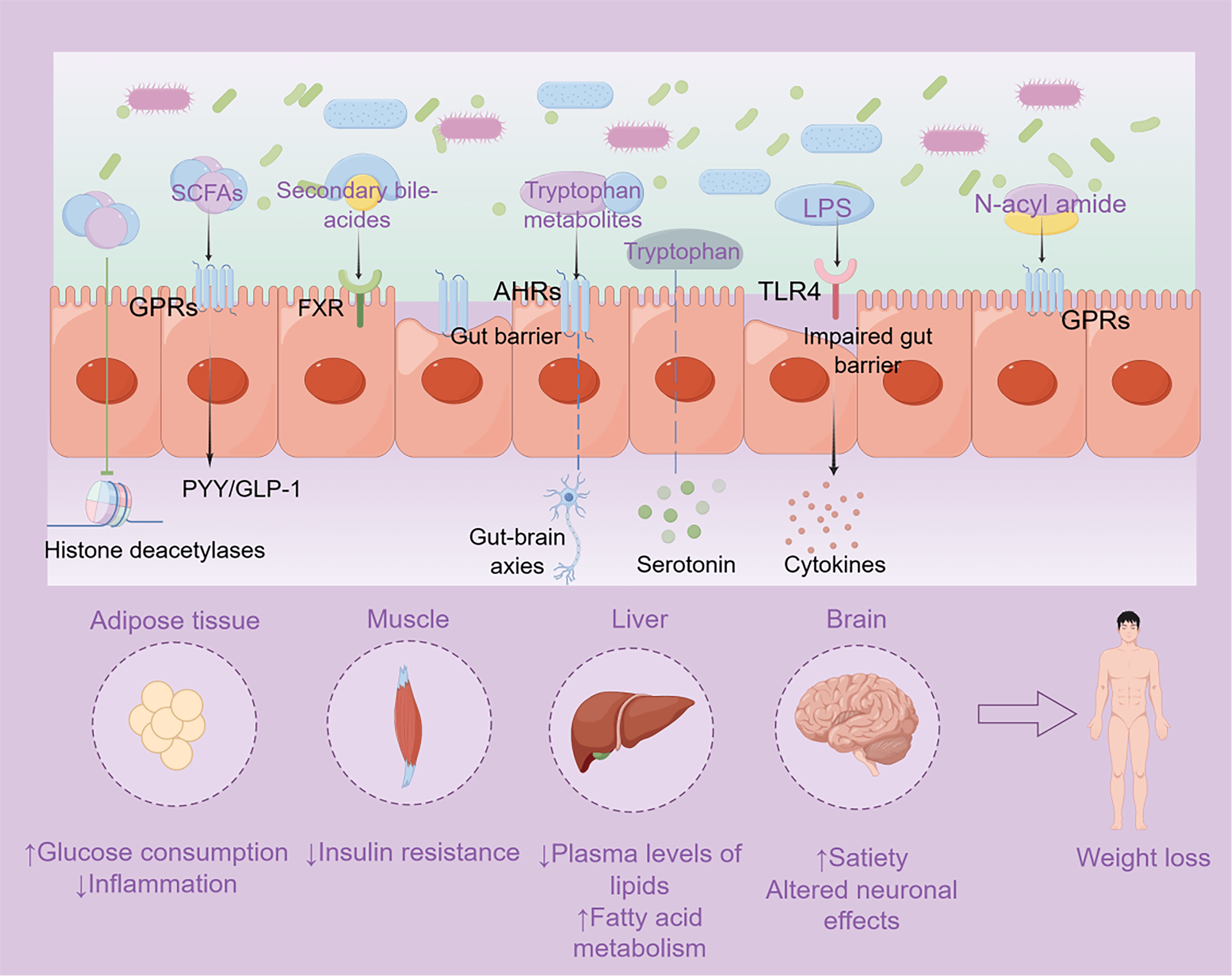 Fig. 3.
Fig. 3.
Microbial compounds regulate host metabolism. The
SCFAs activate G protein-coupled receptors (GPRs) and then boost the secretion of
peptide YY (PYY) and glucagon-like peptide 1 (GLP-1), which enhance satiety in
brain and promote glucose consumption in adipose tissue and muscles. SCFAs
regulate the expression of inflammatory cytokine-related genes by inhibiting
histone deacetylases. Secondary bile acids produced by the gut microbiota bond to
host receptors and regulate lipid and glucose homeostasis in liver. Tryptophan
metabolites (indole, 2-oxindole, and indole-3-acetic acid) act through the
aromatic hydrocarbon receptors (AHRs) to maintain the intestinal mucosal barrier
and regulate adipocyte metabolism and inflammation in adipose tissue and central
nervous system (CNS). Serotonin, synthesized from tryptophan, is an important
neurotransmitter in the CNS. The microbiota could influence the serotonergic
neurotransmission in brain through gut-brain axis. LPS derived from Gram-negative
bacteria acts via Toll-like receptor 4 (TLR-4) to activate immune response
increasing intestinal permeability and adipocyte inflammation. N-acyl amides,
acting as endocannabinoid-like compounds, which bond to GPRs and regulate energy
metabolism. The figure was created using Figdraw (https://www.figdraw.com).
SCFAs mainly acetate, propionate, and butyrate, are the primary metabolic products of fiber fermentation by the gut microbiota. They are not merely byproducts but act as pivotal signaling molecules that orchestrate host metabolism through several distinct, yet interconnected, mechanisms. One of the primary anti-obesity mechanisms of SCFAs is the modulation of appetite and energy intake through the gut-brain axis. Luminal acetate or propionate activates G protein-coupled receptors (GPRs) boosting the secretion of satiety hormones [87] (Fig. 3). By enhancing satiety, SCFAs directly counteract a key driver of obesity—excessive energy consumption. Furthermore, SCFAs contribute to this neuro-modulatory role by regulating the permeability of the blood-brain barrier and affecting neurotransmission [88]. At both the epigenetic and cellular levels, SCFAs play a crucial role in regulating inflammation and glucose metabolism. They exert epigenetic effects by inhibiting histone deacetylases, which in turn regulates inflammatory cytokine-related genes, helping to maintain intestinal homeostasis and reduce the low-grade inflammation characteristic of obesity [89]. Concurrently, butyrate directly affects glucose metabolism by upregulating AMPK-dependent gene expression, a key pathway for cellular energy sensing [90].
Given these potent biological effects, there is growing interest in using SCFAs as therapeutic agents. Indeed, studies have indicated that SCFAs interventions can increase fat oxidation and improve insulin sensitivity [91, 92, 93]. This suggests that strategies aimed at increasing SCFAs production are valuable for obesity prevention. However, a significant research gap remains, as it is unclear whether these short-term effects translate into long-term benefits. Evidence regarding the optimal mode of administration, specific ingredients, duration, and dosage is still limited, highlighting a critical area for future clinical investigation.
Beyond their classical role in facilitating dietary fat absorption, bile acids (BAs) are now recognized as critical signaling molecules that regulate glucose homeostasis and immune responses [94]. The gut microbiota is central to this signaling function, as it metabolizes liver-derived primary BAs into a diverse pool of secondary BAs. The composition of this secondary bile acid pool is therefore highly dependent on the structure of the microbial community, creating a crucial nexus between diet, the microbiota, and host metabolic homeostasis [95]. These microbially-produced secondary BAs act as ligands for a suite of host nuclear and GPRs, including the farnesoid X receptor [96, 97, 98] (Fig. 3). Activation of these receptors is integral to maintaining lipid and glucose homeostasis and modulating immunoregulatory pathways. These receptors are potential targets for obesity treatment, however, the complex interplay among host factors, bile acids, and the microbiota requires further investigation.
Dietary tryptophan serves as a critical precursor not only for protein synthesis but also for a diverse array of bioactive molecules generated by the gut microbiota. These metabolites create multi-faceted signaling pathways that form a crucial communication hub influencing gut integrity, energy metabolism, and neurobiology. One major pathway involves the microbial conversion of tryptophan into metabolites such as indole, 2-oxindole, and indole-3-acetic acid. These molecules act as key ligands for the aromatic hydrocarbon receptor (AHR), and this activation is essential for maintaining the intestinal mucosal barrier, regulating local immune function, and directly influencing adipocyte metabolism [99, 100, 101] (Fig. 3). Intriguingly, the production of these beneficial metabolites is more closely associated with high-fiber diets than with high-protein intake, highlighting a synergistic relationship between fiber fermentation by bacteria like Firmicutes and the regulation of protein metabolite signaling [102]. Beyond the gut, lifestyle factors like exercise can also activate tryptophan biotransformation in skeletal muscle, leading to the accumulation of kynurenate [103, 104]. This metabolite functions as a potent anti-obesity signal, inhibiting weight gain in animal models by activating GPR35 in adipose tissue to stimulate lipid metabolism, thermogenesis, and anti-inflammatory gene expression [105].
Furthermore, the gut microbiota plays an indispensable role in regulating the host’s serotonin system, a key neurotransmitter synthesized from tryptophan that is closely related to emotional control and food intake. The microbiota can influence serotonin production by enterochromaffin cells and alter its systemic availability [106]. Evidence from germ-free mice confirms that the presence of a gut microbiota significantly increases proteins involved in serotonin synthesis and transport [106]. While peripheral serotonin cannot cross the blood-brain barrier, the microbiota’s influence on the systemic serotonin pool can affect CNS serotonergic neurotransmission through the humoral pathway [107]. This regulation of a key neurotransmitter involved in both mood and appetite helps to explain the frequent co-occurrence of obesity and mood disorders [108], underscoring the importance of the gut-brain axis in metabolic health.
LPS, a major component of the outer membrane of Gram-negative bacteria, serves as a key pro-inflammatory trigger linking diet-induced gut dysbiosis to the systemic inflammation that drives obesity. In the context of obesity, high-fat diets alter the gut microbiome, causing elevated plasma LPS levels [109]. LPS activates TLR4, triggering an immune response that increases intestinal permeability [110] (Fig. 3). A compromised intestinal barrier increases the leakage of LPS into the circulation, which is associated with endotoxemia. Endotoxemia increases the permeability of the blood-brain barrier and induces a neuroinflammatory response and glial cell formation, altering appetite regulation circuits in the hypothalamic region [111]. Concurrently, LPS acts as a potent chemoattractant for macrophages, promoting their infiltration into adipose tissue and causing chronic adipocyte inflammation, a hallmark of obesity [112, 113]. Probiotics, GLP-1, GLP-2, cannabinoid receptors, and GPRs have been implicated as targets for the treatment of endotoxin-induced diseases [113].
The endocannabinoid system (ECS) is a crucial regulator of energy metabolism, immune responses, and host-microbiota communication. An emerging area of research indicates that this system is not exclusively host-regulated but is also directly influenced by the gut microbiota through a sophisticated mechanism of molecular mimicry [111, 114, 115]. Commensal bacteria have been found to possess synthase genes for N-acyl amides, which are endocannabinoid-like compounds [116] (Fig. 3). This represents a direct pathway for bacteria to “speak the host’s language” and tap into a powerful metabolic regulatory system. The significance of this mimicry is underscored by findings that these bacterially-produced lipids can bind to host GPRs with an efficacy equivalent to that of human ligands, directly regulating energy metabolism [116, 117]. In humans, the abundance of specific microbial families, such as Veillonellaceae, Peptostreptococcaceae and Akkermansiaceae, has been correlated with circulating levels of these N-acyl amides [118]. This microbiota-ECS axis appears to be directly implicated in the pathogenesis of obesity. For example, studies have demonstrated that a high-fat diet can alter the gut microbiota in a manner that dysregulates endocannabinoid receptor signaling, thereby promoting lipogenesis [119, 120].
The discovery that the gut microbiota can manipulate the widely-distributed ECS opens up novel therapeutic possibilities [121, 122]. Rather than only targeting host receptors, future interventions could aim to modulate the microbial production of these endocannabinoid-like compounds, offering a new strategy for treating obesity and other metabolic diseases.
This review consolidates the evidence that the gut microbiota is a pivotal mediator of the effects of diet on childhood obesity, influencing host metabolism, immunity, and neurological functions. Diet has long been recognized as a primary intervention for obesity, and its profound impact on the microbiota presents a powerful therapeutic avenue. However, the translation of these findings into effective, long-term clinical strategies remains a central challenge. Current interventions often yield inconsistent results, largely due to the high inter-individual variability of the gut microbiome and the complexity of host-microbe-diet interactions. Furthermore, moving beyond short-term observations to understand the long-term developmental impacts of these interventions is crucial. Therefore, the future of this field lies in a shift from broad dietary recommendations to “microbiome-aware” precision nutrition. This requires a deeper understanding of causality, transitioning from descriptive analyses to mechanistic studies. Key to this paradigm shift will be integrating multi-omics data, such as targeted metabolomics, to identify personalized biomarkers and leveraging individualized trial designs to tailor interventions. Ultimately, through interdisciplinary collaboration, harnessing the power of dietary modulation of the gut microbiota holds immense promise for developing safe and efficacious strategies for the prevention and treatment of childhood obesity.
FF and SZ designed the research study. SZ and ZC performed the research. FH, BW and JL designed the research study and provided help and advice on the manuscript. SZ and FF wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors gratefully acknowledge the China Academy of Chinese Medical Sciences, Beijing, China.
Beijing Traditional Chinese Medicine Science and Technology Development Fund Project (No. BJZYZD-2023-09), The Fundamental Research Funds for the Central Public Welfare Research Institutes (No. ZZ17-XRZ-043, No. ZZ15-XY-PT-03, HLCMHPP2023021).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



