1 Department of Nutrition Faculty of Pharmacy and Medical Sciences, University of Petra, 11196 Amman, Jordan
Abstract
Unfiltered Turkish coffee (UTC) is a traditional drink with high levels of bioactive compounds, but evidence of the associated specific physiological effects is inconclusive, and few studies have examined coffee in general. This pilot study aimed to investigate the short-term effects of daily UTC consumption on cardiovascular parameters, lipid profile, appetite-regulating hormones (leptin, ghrelin), glucose metabolism, inflammatory markers, and sleep quality in healthy young women.
This is a pilot randomized controlled trial that randomly assigned 40 healthy young women (aged 18–25 years) to intervention and control groups at a 1:1 ratio after 3 weeks of caffeine washout. The intervention group consumed three 40 mL cups of traditional-brewed UTC daily for 4 weeks, whereas the controls maintained abstinence from caffeine. The primary outcomes were cardiovascular (blood pressure, heart rate), lipid parameters, and the secondary ones were appetite hormones (leptin, ghrelin), glucose metabolism (markers), inflammatory biomarkers, and sleep quality, which were evaluated at baseline and at week 4.
UTC consumption produced significant between-group differences (time × group interactions) compared to controls: systolic blood pressure (+3.0 mmHg; p = 0.025), heart rate (+10.6 bpm; p = 0.007), and insomnia severity scores (+4.05 points intervention vs. –1.00 points control; p ≤ 0.001), while significantly decreasing leptin levels (–0.04 ng/mL; p = 0.014). Significant changes in low-density lipoprotein (LDL) cholesterol were found (p = 0.002), although high-density lipoprotein (HDL) changes were no longer found significant on baseline correction (p = 0.385). Body composition parameters (body mass index (BMI), body fat mass, fat-free mass, skeletal muscle mass) remained unchanged throughout the intervention (all p > 0.05). No significant effects were observed for fasting blood glucose, glycated hemoglobin (HbA1c), inflammatory markers (C-reactive protein (CRP), tumor necrosis factor-alpha (TNF-α)), or ghrelin (all p > 0.05).
Four weeks of UTC intake in this pilot trial were associated with variations in several cardiometabolic variables: interventions in systolic blood pressure (SBP) (+3.0 mmHg) and heart rate (HR) (+11.9 bpm) resulted in higher LDL levels, reduced leptin levels, and poor sleep quality, independent of body composition alterations. However, since the p-values are nominal and not multiplied by a correction, hypothesis-generating results require verification through properly powered studies. These exploratory findings should be considered by individuals with prior cardiovascular risk factors or sleep disorders when considering the intake of unfiltered coffee.
This trial was registered at ClinicalTrials.gov (NCT07133373, https://clinicaltrials.gov/study/NCT07133373). Retrospectively registered on 13 August 2025.
Keywords
- unfiltered Turkish coffee (UTC)
- female
- caffeine
- cardiovascular health
- inflammation
- insomnia
- appetite
Coffee is also among the most popular beverages in the world that has approximately 2.25 billion cups of coffee being taken by people of various cultures like nationalities [1, 2]. Besides the social worth, coffee is an important source of food bioactive molecules like caffeine, chlorogenic acids, diterpenes and polyphenols with metabolic, cardiovascular and neurological effects [3]. Unfiltered Turkish coffee (UTC) has recently gained a new following, especially with younger generations in Middle Eastern, Mediterranean, and urban academia, among the classic techniques of brewing coffee [4, 5, 6]. In regions such as Turkey, Cyprus, and much of the Middle East, UTC remains deeply embedded in cultural traditions: finely ground coffee is boiled in a cezve without filtration, creating a beverage consumed with suspended grounds [7]. This unfiltered method retains substantially higher concentrations of certain bioactive compounds, especially the diterpenes and kahweol that are mostly eliminated by filtration using paper in other brewing processes [8]. The quantitative variations in the bioactive compounds lead to different physiological responses of cardiovascular and metabolic health to filtered coffee [8, 9]. Coffee has bioactive compounds that are present in all the brewing processes and therefore have complicated and even contradictory health effects [10]. Having been found in all types of coffee, chlorogenic acids slow down the absorption of glucose in the intestine and improve insulin sensitivity by regulating the glucose-6-phosphatase activity [11], which can help people with glycemic regulation [10, 12]. These polyphenolic compounds have comprehensive antioxidant and metabolic advantages irrespective of the means of preparation [10, 12]. The primary distinction between unfiltered and filtered coffee is the existence of diterpenes: cafestol and kahweol are found 4–6 times more in UTC than in the filtered ones due to the absence of paper filtration [8, 13]. Although these diterpenes have antioxidant and hepatoprotective effects [13] they are also linked with high levels of low-density lipoprotein (LDL) cholesterol as a result of hepatic cholesterol metabolism alteration which are possible cardiovascular risk factors at the higher concentrations obtained in unfiltered coffee [14, 15]. It has been proved in human trials that consumption of unfiltered coffee can increase total cholesterol and homocysteine levels, which are cardiovascular disease risk factors [16] but the actual effect of Turkish coffee (TC) has been inconsistently demonstrated [17]. Certain reports have found an increase in homocysteine and poor lipid modifications, whereas others have found a decrease in endothelial functioning and decrease in arterial rigidity, with results varying depending on the mode of preparation, dosage, timing, and inter-individual variation [17]. Extensive literature indicates moderate levels of coffee are linked to reduced risk of cardiovascular disease, but these effects seem to be nonlinear in nature and depend upon timing, amount, and genetic polymorphisms to process caffeine, specifically the Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) polymorphs [18, 19]. Besides the cardiovascular effect, the caffeine in TC can control leptin and ghrelin hormones, which form the centre of the counter of appetite regulation. Caffeine suppresses the appetite and increases the energy expenditure by activation of the sympathetic nervous system [18, 19]. However, most of the investigations have involved overall coffee preparation or individual caffeine, and little is known regarding TC, particularly among women [19]. Additionally, caffeine as an adenosine receptor antagonist, may disrupt sleep architecture by delaying the onset and reducing total sleep duration [20, 21]. Timing seems to be the key: caffeine in the evening has a significant negative effect on the quality of sleep [21], but in the morning, it might produce the least disruptive effect and some cardiovascular effect due to circadian regulation [22].
Special attention is justified by these effects in young women, as they have different caffeine pharmacokinetics, in which the enzymatic activity of CYP1A2 is slower, with the result that caffeine half-life is prolonged, and there is prolonged physiological exposure [23, 24]. The menstrual cycle hormonal changes further modify the caffeine metabolism and recent evidence suggests that women may have a greater response to the identical dose of caffeine in terms of the cardiovascular effect [23, 24]. In spite of the fact that women constitute the majority of UTC consumers in Middle Eastern societies [25], in the intervention trials, it is mostly the male or mixed-gender cohort that is enrolled without sex-stratified analyses, which is a knowledge gap. The newer consumer trends in Jordan suggest that cultural biases were high towards medium dark roasts blends that are made in the traditional Turkish fashion with the average 40 mL portions having around 6–7 g of coffee [25], which underscores the need to determine the health outcomes of the practice using culturally representative preparation techniques in populations where UTC is regularly used.
Nevertheless, the absence of strict scientific studies examining its specific physiological effect on the human body despite the cultural significance and the fact that it has popularized among people is rather surprising. Most coffee research extrapolates throughout the brewing procedure without taking into account the fact that bioactive substances, particularly diterpenes, are of high concentration and represent the characteristics of unfiltered preparations. Chlorogenic acids and caffeine are common in any kind of coffee [10, 12], UTC’s unfiltered method results in diterpene concentrations (cafestol, kahweol) 4–6 times higher than filtered coffee [8, 14], fundamentally altering the balance of cardiovascular and metabolic effects [8, 9]. With the increased attention to conventional preparation methods among young adults who care about their health, specific studies are needed to provide evidence-based advice. Although comparative studies on brewing techniques would provide useful mechanistic data, developing a baseline understanding of the physiological action of UTC versus caffeine withdrawal is an essential preliminary measure, as there are virtually no controlled studies directly examining UTC. It is here that coffee research methodology has established a precedent [5, 7].
Therefore, this pilot randomized controlled trial investigated the short-term
effects of four consecutive weeks of standardized daily UTC consumption on
health-related parameters in healthy young women. Cardiovascular parameters
systolic blood pressure (SBP), diastolic blood pressure (DBP), resting heart rate
(HR), and lipid profile [high-density lipoprotein (HDL), LDL cholesterol, total cholesterol, triglycerides]) were
assessed as primary outcomes. Secondary outcomes included appetite-regulating
hormones (leptin, ghrelin), glucose metabolism markers (fasting blood glucose,
glycated hemoglobin (HbA1c)), inflammatory biomarkers (C-reactive protein (CRP),
tumor necrosis factor-alpha (TNF-
This pilot randomized controlled trial examined the physiological effects of
four-week UTC consumption in healthy young women aged 18–25 years. The study
received ethical approval from the Institutional Review Board (IRB) at the
University of Petra (UOP) under protocol number E/H/5/10/2024. The trial was
registered post factum by the Open Science Framework (OSF, DOI:
10.17605/OSF.IO/2V96N; registered 13 May 2025) and ClinicalTrials.gov
(NCT07133373; registered 13 August 2025) when the data collection had been
finalized but the final manuscript was being prepared. Transparency statement:
The registration was done retrospectively to comply with the existing reporting
requirements, but the study protocol, primary outcomes (cardiovascular parameters
and lipid profile), secondary outcomes, and the plan of statistical analysis had
been pre-determined before the participants were recruited, and no changes were
made after data inspection. This assertion agrees with the retrospective
registration time and protocol integrity [26]. All study procedures were
conducted in accordance with the Declaration of Helsinki ethical principles and
adhered to CONSORT 2010 guidelines for transparent reporting of randomized
controlled trials [27]. Primary outcomes were pre-specified as cardiovascular
parameters: SBP, DBP, HR, and lipid profile components (HDL, LDL, total
cholesterol, triglycerides). Appetite- regulating hormones (leptin, ghrelin),
glucose metabolism (fasting blood glucose, HbA1c), inflammatory (C-reactive
protein (CRP), TNF-
Fifty healthy young women were recruited from the University of Petra and
surrounding Amman community (Jordan) via digital advertisements and social media.
All participants were of Middle Eastern ethnicity, representing the primary UTC
consumer demographic in this cultural region. Eligibility was assessed through
structured face-to-face interviews collecting demographic and health data.
Inclusion criteria: age 18–25 years, Body Mass Index (BMI) 18.5–24.9
kg/m2, non-smokers, minimal caffeine intake (
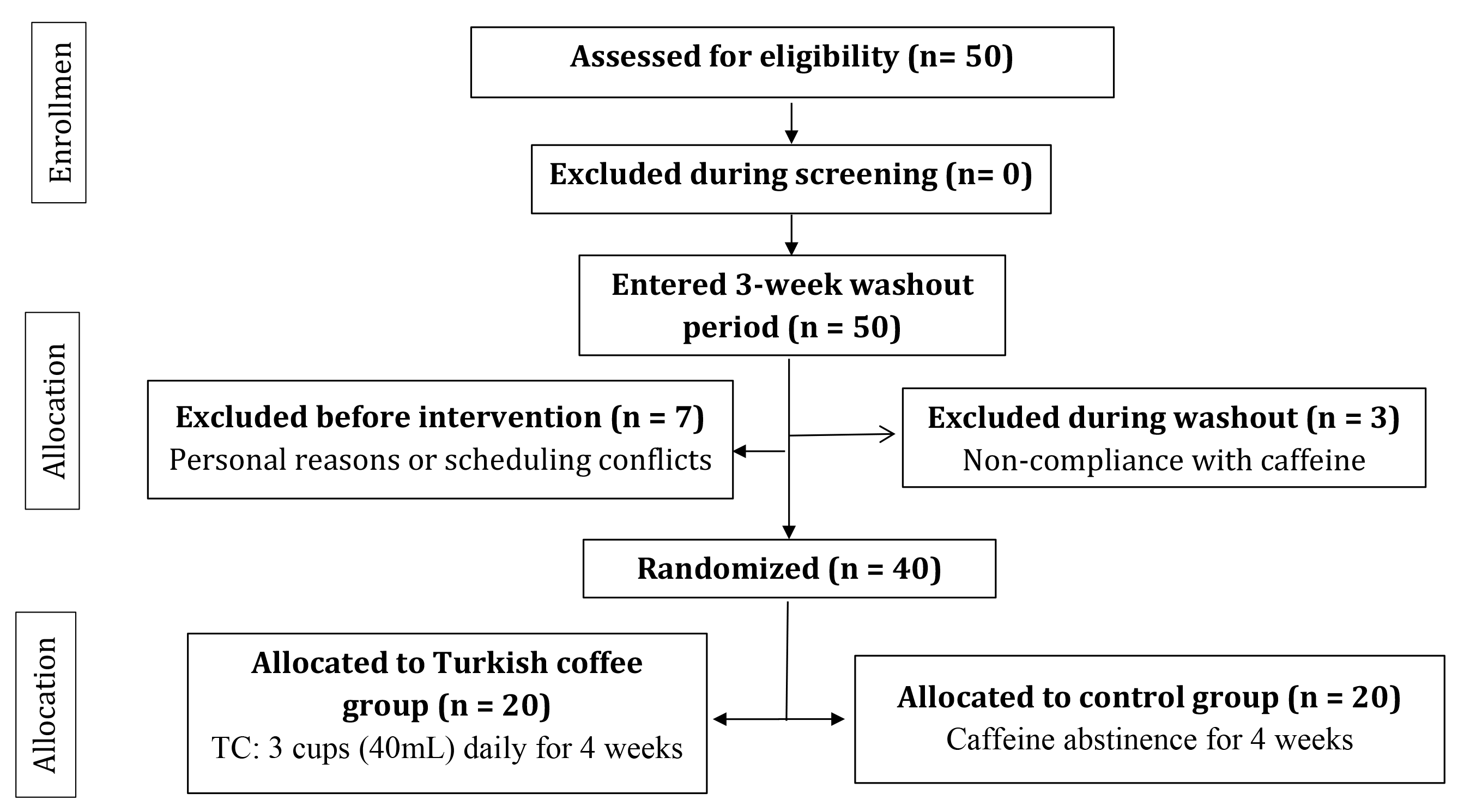 Fig. 1.
Fig. 1.
CONSORT flow diagram showing participant recruitment, screening, randomization, and allocation. The diagram illustrates the complete participant flow through the study from initial recruitment (n = 50) through final analysis (n = 40). Following initial screening, 47 participants completed a mandatory three-week caffeine washout period. Three participants were excluded during washout due to non-compliance with caffeine abstinence requirements. The remaining 47 eligible participants underwent stratified block randomization (stratified by BMI) into intervention (Turkish coffee consumption) or control (caffeine abstinence) groups. Seven participants withdrew voluntarily before intervention commencement due to personal reasons or scheduling conflicts. The final analyzed cohort comprised 40 participants: 20 in the intervention group (receiving three 40 mL cups of standardized Turkish coffee daily for four weeks) and 20 in the control group (maintaining caffeine abstinence for four weeks). All 40 participants who commenced the intervention completed the four-week protocol and were included in the final analysis with no dropouts or loss to follow-up. Abbreviations: TC, Turkish coffee; BMI, body mass index.
Following randomization, intervention participants consumed UTC daily for four weeks while control participants-maintained caffeine abstinence and usual dietary habits. Due to TC’s distinct sensory characteristics (foam, suspended grounds, aroma, flavor), participant blinding was not feasible; this was an open-label trial. Biochemical analysis outcome assessors were also blinded to the group assignment and anthropometric measurements were automated to reduce group recognition at this point. All the participants were given the uniform guidelines in order to maintain constant dietary patterns during the research. A standardized batch of finely ground Brazilian Arabica coffee (medium-dark roast blend: 50% medium + 50% dark roast) was purchased from a single supplier. Each 10-gram serving (5 g medium + 5 g dark roast) represents typical Jordanian UTC preparation [25]. This roast combination balances chlorogenic acid preservation (medium roast) with diterpene availability (dark roast) [31, 32, 33]. Coffee was distributed to participants with standardized storage, preparation, and consumption instructions. UTC was prepared traditionally by steeping 5 grams finely ground coffee in 55 mL cold water in a Rakwa (traditional Turkish coffee pot), heated slowly over low heat to form surface foam. Final serving volume was 40 mL/cup per Jordanian consumption standards. This preparation method preserved bioactive compounds including chlorogenic acids and diterpenes. Participants consumed three 40-mL cups daily at standardized times: 7–9 AM, 12–2 PM, and 3–4 PM, with evening consumption prohibited to minimize sleep disruption [21]. Participants were monitored with regards to adherence to the protocols by daily logs and phone calls. UTC was consumed without any additives, except that habitual sugar users did not reduce their sugar amount. Coffee was standardized with pre-weighed utensils and preparation guidelines to follow. Caffeine content was quantified via high-performance liquid chromatography with diode array detection and mass spectrometry HPLC-DAD-MS (LCMS-2020 system with photodiode array detector, Shimadzu Corporation, Kyoto, Japan (at 1.77 mg/mL (71 mg/40-mL cup). Diterpenes (cafestol, kahweol) and chlorogenic acids were not directly measured in our UTC samples. Mechanistic discussions referencing these compounds rely on literature-derived estimates from chemically similar UTC preparations (Brazilian Arabica, medium-dark roast, unfiltered brewing) suggesting typical concentrations of cafestol 4.4–6.2 mg/cup, kahweol 3.2–4.8 mg/cup [31, 32], and chlorogenic acids 70–115 mg/150 mL [33]. These estimates provide context for hypothesis-generating mechanistic interpretations but do not establish causation, and substantial batch-to-batch variability may exist. The lack of direct bioactive compound quantification represents a major limitation discussed below. Single 24-hour dietary recalls at baseline and post-intervention assessed dietary intake, with total macronutrient intake included as a covariate in statistical analyses to control for dietary confounding.
Blood samples were collected at baseline and week 4 after
Fasting blood glucose was measured using the TRUE METRIX® Meter (TRUE METRIX, Trividia Health, Inc., Fort Lauderdale, FL, USA) (electrochemical biosensor technology; 0.5 µL sample volume). HbA1c was assessed via automated immunoassay analyzer (HP-AFS/1-PLUS, Shijiazhuang Hipro Biotechnology Co., Ltd., Shijiazhuang, Hebei, China) using integrated nephelometry, turbidimetry, and immunofluorescence methods.
Total cholesterol, triglycerides, LDL, and HDL were analyzed using an automated
immunoassay analyzer using nephelometry, turbidimetric immunoassay, and
fluorescence immunoassay according to the manufacturer’s instructions. The
analyzer was calibrated to international reference standards with output in
mmol/L, which is reported throughout this manuscript. Lipid measurements were
performed by certified laboratory technicians following standardized protocols.
Resting SBP, DBP, and HR were measured at baseline and week 4 using an automated
upper-arm oscillometric monitor, M7 Intelli IT (HEM-7361T-EBK; Omron Healthcare
Co., Ltd., Kyoto, Japan). Measurements were conducted between 8:00–10:00 AM in a
quiet environment after
Appetite regulatory mechanisms were studied by measuring serum levels of leptin and ghrelin at intervention baseline and postintervention. Quantification of these hormones was performed with a commercially available enzyme-linked immunosorbent assay (ELISA) kit (EH0216, EH0544, Wuhan Fine Biotech Co., Ltd. Wuhan, Hubei, China) that was specific to humans. The two assays were conducted as per the instructions of the manufacturer in the standardized procedures. The absorbance was measured by a BioTek Epoch™ 2 Microplate Spectrophotometer (EPOCH2NSC, Agilent Technologies, Santa Clara, CA, USA) in Gen 5™ software (version 3,Agilent Technologies, Santa Clara, CA, USA). Each sample was subjected to a duplicate analysis, and a statistical analysis was done considering the mean values.
Single 24-hour dietary recalls were used to evaluate dietary intake at the time of intervention. The rationale of the choice of this approach was feasibility and minimizing possible confounding by energy and macronutrient differences that could affect the cardiovascular, metabolic, and hormonal outcomes [29, 30, 35]. All the food and beverage products that had been consumed during the last 24 hours were described by a trained researcher including the quantity of the consumed food, the method of preparation, and the time taken. ESHA Food Processor software (version 8.6, ESHA Research, Salem, OR, USA) was used to analyze the intake of macronutrients and micronutrients. All statistical analyses had covariates on total energy and macronutrient intake (carbohydrates, proteins, fats) as a potential dietary factor confounding cardiovascular, metabolic and hormonal results [29, 30, 35]. Although single 24-hour recalls lacked day-to-day dietary variability, such a method is the usual methodology of short-term coffee intervention trials in terms of feasibility to statistical control [29, 30].
The Arabic version of the Insomnia Severity Index (ISI) was subjected to
validation and included the evaluation of insomnia symptoms at baseline and week
4 [36]. This version demonstrated acceptable internal consistency (Cronbach’s
Baseline anthropometric parameters were assessed during initial screening to
determine eligibility. Body composition parameters (fat mass, fat-free mass,
total body water, skeletal muscle mass) were measured using InBody 970 Body
Composition Analyzer (MC-780 MA, Tanita Co., Seoul, Korea) via multifrequency
bioelectrical impedance analysis. The device was regularly calibrated according
to the requirements of the manufacturer. All testing took place in the morning (a
single and uniform time slot for all participants) at least 10 hours after
overnight fasting before any study procedures. The participants were advised not
to drink alcohol and engage in vigorous activity in the 24 hours before the
experiment, to keep themselves well-hydrated, remove any metal objects, and the
clothes were standardized. Where feasible, measurements were done at the
follicular phase to reduce the effects of hormones on bioelectrical impedance
[23, 24]. Parameters of body composition were used to affirm baseline homogeneity
and ensure high BMI eligibility (18.5–24.9 kg/m2), as well as to check how
the parameters might change with the 4-week intervention [29, 37]. The 40
participants (20 assayed in thereof) went through the 4-week protocol with 100%
retention. Protocol adherence (verified via daily logs and random phone calls)
was
Sample size was calculated a priori using G*Power (version 3.1.9.4,
Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany) for
repeated-measures ANOVA with two groups and two time points (baseline, week 4).
Assumptions: medium effect size (f = 0.25),
As a pilot hypothesis-generating trial [27, 38], no formal corrections for multiple testing were applied. All p-values are nominal and findings should be interpreted exploratorily, prioritizing primary outcomes (cardiovascular parameters, lipid profile) over secondary outcomes.
All 40 randomized participants (20 per group) completed the 4-week intervention
with 100% retention (Fig. 1). Mean coffee consumption in the intervention group
was 20.8
The baseline characteristics were fairly balanced between the groups (Table 1),
and no significant differences could be found in most demographic,
anthropometric, and body composition variables (all p
| Characteristic | Intervention group (n = 20) | Control group (n = 20) | p-value |
| Age (years) | 20.55 |
20.43 |
0.83 |
| Baseline caffeine intake (cups/week) | 0.65 |
0.70 |
0.62 |
| BMI (kg/m2) | 22.71 |
21.75 |
0.23 |
| Weight (kg) | 57.98 |
57.72 |
0.92 |
| TBW (L) | 28.00 |
29.82 |
0.19 |
| ICW (L) | 17.35 |
18.51 |
0.19 |
| ECW (L) | 10.65 |
11.31 |
0.18 |
| BFM (kg) | 19.67 |
17.00 |
0.07 |
| FFM (kg) | 38.31 |
40.72 |
0.22 |
| SMM (kg) | 20.64 |
22.12 |
0.21 |
| PBF (%) | 33.60 |
29.43 |
0.02 |
| BMR (kcal) | 1197.3 |
1249.6 |
0.21 |
Data are mean
Body composition parameters were assessed at baseline and week 4 to monitor
potential changes during the intervention period (Table 2). Body weight showed
minimal changes in both groups, with the intervention group increasing by 0.52 kg
(57.98
| Measure | Intervention group (n = 20) | Control group (n = 20) | F (Time Effect) | p value (Time Effect) | F (Time × Group) | p value (Time × Group) | Partial Eta Squared | ||
| Baseline | Post-Intervention | Baseline | Post-Intervention | ||||||
| Weight (kg) | 57.98 |
58.50 |
57.72 |
57.92 |
1.19 | 0.28 | 0.23 | 0.64 | 0.03 |
| BMI (kg/m2) | 22.71 |
22.80 |
21.75 |
21.83 |
0.33 | 0.57 | 0.96 | 0.01 | |
| TBW (L) | 28.00 |
27.93 |
29.82 |
29.81 |
0.08 | 0.79 | 0.04 | 0.84 | 0.00 |
| ICW (L) | 17.35 |
17.30 |
18.51 |
18.51 |
0.08 | 0.78 | 0.08 | 0.77 | 0.00 |
| ECW (L) | 10.65 |
10.63 |
11.31 |
11.30 |
0.06 | 0.81 | 0.01 | 0.94 | 0.00 |
| BFM (kg) | 19.67 |
20.30 |
17.00 |
17.20 |
1.95 | 0.17 | 0.50 | 0.48 | 0.05 |
| FFM (kg) | 38.31 |
38.20 |
40.72 |
40.72 |
0.07 | 0.80 | 0.07 | 0.80 | 0.00 |
| SMM (kg) | 20.64 |
20.57 |
22.12 |
22.12 |
0.07 | 0.80 | 0.07 | 0.80 | 0.00 |
| PBF (%) | 33.60 |
34.44 |
29.43 |
29.70 |
2.18 | 0.15 | 0.59 | 0.45 | 0.05 |
| BMR (kcal) | 1197.3 |
1194.8 |
1249.6 |
1249.4 |
0.09 | 0.77 | 0.06 | 0.80 | 0.00 |
Data are mean
Baseline Comparison and Adjusted Analysis for HDL: Independent samples
t-tests comparing baseline values between groups revealed that HDL was
the only outcome with significant baseline imbalance (intervention: 2.45
| Measure | Intervention group (n = 20) | Control group (n = 20) | F (Time Effect) | p-value (Time Effect) | F (Time × Group) | p-value (Time × Group) | Partial Eta Squared | ||
| Baseline | Post-Intervention | Baseline | Post-Intervention | ||||||
| SBP (mmHg) | 108.30 |
112.3 |
110.95 |
111.95 |
1.37 | 0.25 | 5.459 | 0.025 | 0.126 |
| DBP (mmHg) | 81.90 |
81.85 |
78.55 |
79.20 |
0.21 | 0.65 | 0.245 | 0.624 | 0.006 |
| HR (bpm) | 82.45 |
93.00 |
82.70 |
81.45 |
5.02 | 0.03 | 8.08 | 0.007 | 0.175 |
| HDL (mmol/L) | 2.45 |
1.26 |
1.81 |
1.43 |
10.19 | 0.772 | 0.385 | 0.019 | |
| Cholesterol (mmol/L) | 4.36 |
5.14 |
4.05 |
4.84 |
14.63 | 0.001 | 0.978 | 0.000 | |
| TG (mmol/L) | 2.86 |
2.91 |
2.97 |
3.10 |
0.47 | 0.49 | 0.103 | 0.75 | 0.003 |
| LDL (mmol/L) | 0.60 |
2.56 |
1.37 |
1.77 |
26.43 | 11.492 | 0.002 | 0.232 | |
| FBS (mg/dL) | 92.40 |
91.45 |
90.80 |
89.25 |
2.20 | 0.15 | 0.127 | 0.724 | 0.003 |
| HbA1c | 4.60 |
4.16 |
3.78 |
4.56 |
0.23 | 0.64 | 2.905 | 0.096 | 0.071 |
Data are mean
Triglycerides showed minimal non-significant changes (intervention: 2.86 to 2.91
mmol/L; control: 2.97 to 3.10 mmol/L; p = 0.495). LDL increased in both
groups (intervention: 0.60 to 2.56 mmol/L; control: 1.37 to 1.77 mmol/L) with
significant time (p
Fasting blood glucose showed minimal non-significant changes (intervention:
92.40
Leptin decreased in the intervention group (0.218 to 0.180 ng/mL) while
remaining stable in controls (0.302 to 0.304 ng/mL), with significant time
(p = 0.014, F = 6.673, partial
| Measure | Intervention group (n = 20) | Control group (n = 20) | F (Time Effect) | p value (Time Effect) | F (Time × Group) | p value (Time × Group) | Partial Eta Squared | ||
| Baseline | Post-Intervention | Baseline | Post-Intervention | ||||||
| Leptin (ng/mL) | 0.218 |
0.180 |
0.302 |
0.304 |
6.67 | 0.01 | 8.65 | 0.01 | 0.19 |
| Ghrelin (pg/mL) | 1237.22 |
1314.15 |
1628.83 |
1615.38 |
0.18 | 0.68 | 0.36 | 0.56 | 0.01 |
| CRP (mg/L) | 2.61 |
1.85 |
2.83 |
2.53 |
0.49 | 0.49 | 0.09 | 0.77 | 0.00 |
| TNF- |
160.76 |
147.30 |
103.0 |
132.16 |
0.14 | 0.71 | 1.03 | 0.32 | 0.03 |
Data are mean
Insomnia severity increased significantly in the intervention group (8.95
| Measure | Intervention group (n = 20) | Control group (n = 20) | F (Time Effect) | p value (Time Effect) | F (Time × Group) | p value (Time × Group) | Partial Eta Squared | ||
| Baseline | Post-Intervention | Baseline | Post-Intervention | ||||||
| Insomnia | 8.95 |
13.00 |
11.00 |
10.00 |
5.25 | 0.03 | 14.40 | 0.28 | |
Data are mean
Despite TC’s widespread consumption in Middle Eastern cultures, rigorous research examining its specific physiological effects remains limited, with most coffee studies generalizing across brewing methods without accounting for unfiltered preparations’ distinct biochemical properties [8, 9]. The present study aimed to evaluate the effects of four weeks of UTC consumption on key health parameters among young females. The effects of four-week of UTC consumption on cardiovascular, metabolic, hormonal, and sleep parameters among healthy young women were explored in this pilot trial. Novel contributions include: (1) first controlled trial examining UTC in young women from the Middle Eastern region—the primary consumer demographic—using culturally authentic preparation methods reflecting local consumption practices [41]; (2) simultaneous multi-system assessment (cardiovascular, metabolic, hormonal, sleep) providing integrated physiological insight; (3) first documentation of significant leptin reduction without ghrelin changes in coffee trials, suggesting selective appetite-regulatory mechanisms; (4) validated insomnia assessment (ISI) rather than self-reported sleep duration [20, 21]; and (5) rapid lipid profile changes (HDL decrease, LDL increase) within four weeks, faster than previously recognized [41, 42].
The significant methodological limitation needs to be mentioned first. Although we directly determined caffeine content (71 mg/cup), diterpenes (cafestol, kahweol) and chlorogenic acids were not measured in our UTC samples. Mechanistic interpretations using these compounds are hypothesis-generating inferences using literature estimates of chemically related preparations [31, 32, 33], not definitive causal attributions. The observed physiological changes (cardiovascular, lipid, hormonal, sleep) are empirical facts; the biochemical mechanisms underlying these changes remain speculative pending direct bioactive compound analysis. We prioritize discussion of measured outcomes while contextualizing findings within established coffee pharmacology literature.
Our findings confirmed the hypothesized effects of UTC consumption on cardiovascular function, lipid metabolism, leptin regulation, and sleep quality. Body composition parameters (weight, BMI, body fat, fat-free mass, skeletal muscle mass) remained unchanged over four weeks, consistent with prior studies showing that short-term coffee consumption does not alter body composition without concurrent exercise or dietary modification [29, 35, 43]. The 4-week duration was insufficient for body composition remodeling, which typically requires 8–12 weeks [37]. The stability of body composition despite major cardiovascular and metabolic changes (reduction in HDL, rise in LDL, hypertension, fall in leptin) is plausibly due to direct effects of pharmacological actions and not the effect of adipose. This association reinforces the fact that bioactive compounds of TC, especially diterpenes and caffeine, have direct hepatic and cardiovascular effects even without considering the changes in energy balance or fat mass [5, 29, 37].
UTC consumption significantly increased SBP (+4.0 mmHg intervention vs. +1.0
mmHg control; Time
HR increased significantly in the intervention group (82.45 to 93.00 bpm;
approximately 13% increase) with significant time (p = 0.031) and Time
CRP and TNF-
This pilot trial offers several important contributions to coffee intervention
research. We provide the first controlled investigation of the unfiltered Turkish
coffee’s physiological effects specifically in young women—the primary consumer
demographic—using culturally authentic preparation methods and addressing
systematic underrepresentation of women in coffee research [8, 23, 24]. The rigor
of the study is well established (pre-registered protocol OSF DOI:
10.17605/OSF.IO/2V96N), three-week caffeine washout control of baseline
standardization, coffee preparation which was required to be precisely controlled
(quantified caffeine content by HPLC-DAD-MS), high retention of all study
participants (100 percent completion rate), and validated measurement measures of
all results. The simultaneous recording of cardiovascular, metabolic, hormonal
and sleep variables provide composite physiological data, which is rarely
elucidated in the study of the effects of coffee intervention. Our findings
reveal novel mechanistic observations, such as the leptin loss without body
composition changes and high rates of lipid changes in four weeks, to add
significant preliminary information to be used in future confirmatory studies.
Several limitations warrant acknowledgment. The caffeine abstinence control
design, while appropriate for our primary research question and consistent with
established coffee intervention methodology [29, 30, 68], limits conclusions about
Turkish coffee’s specificity compared to other brewing methods. Female-only
enrollment enhances internal validity through reduced inter-individual
variability but restricts generalizability to males and older populations, though
this approach directly addresses sex-specific caffeine pharmacokinetics
documented in the literature [23, 24]. The small sample size (n = 20 each group),
is an indication that this research was an exploratory pilot study and limits
that research to population level representativeness, where a need to replicate
these studies with larger and demographically varied populations is necessary
before making generalizations. Only caffeine content was directly quantified (71
mg/cup via HPLC-DAD-MS); diterpenes (cafestol, kahweol) and chlorogenic acids
were not measured in our UTC samples. All mechanistic discussions attributing
effects to these compounds represent hypothesis-generating inferences based on
literature estimates from chemically similar preparations [31, 32, 33], not
empirically established causation. Substantial batch-to-batch variability in
bioactive compound concentrations may exist depending on bean origin, roasting
degree, grinding fineness, and brewing time [31]. Without direct measurement and
dose-response analysis, we cannot definitively attribute observed physiological
changes to specific bioactive compounds. Future trials must incorporate
comprehensive phytochemical profiling (HPLC-MS quantification of diterpenes,
chlorogenic acids, trigonelline, melanoidins) coupled with pharmacokinetic
assessments to establish causal mechanisms underlying cardiovascular, lipid, and
hormonal responses. Menstrual cycle phase was not biochemically verified or
controlled as a stratification variable. While participants self-reported regular
cycles and randomization should distribute cycle-phase variability equally
between groups, hormonal fluctuations may have contributed to individual response
variability. The 4-week intervention duration spans 1–2 complete menstrual
cycles, potentially averaging hormonal effects across time points. Future trials
would benefit from cycle-phase stratification to reduce unexplained variance
[23, 24]. These uncontrolled hormonal variations are inherent limitations to the
study as a pilot study due to the small sample size (n = 40), though
randomization will evenly allocate this variability across the groups. Genetic
polymorphisms affecting caffeine metabolism (CYP1A2) and cardiovascular responses
(ADORA2A adenosine receptors) were not assessed. The *1F and *1A alleles of
CYP1A2 produce up to 40% differences in caffeine clearance rates between fast
and slow metabolizers [66, 67], while ADORA2A variants modulate blood pressure and
anxiety responses to caffeine [64]. Our ethnically homogeneous Middle Eastern
cohort may reduce inter-ethnic genetic variability compared to multi-ethnic
samples, as allele frequencies differ substantially across populations [67].
While randomization should distribute genotypes equally between groups, genetic
heterogeneity likely contributed to individual response variability (reflected in
standard deviations). The robust group-level effects observed for cardiovascular,
lipid, and sleep outcomes suggest treatment signals detectable despite genetic
variability. Future trials would incorporate genotyping to enable stratified
analyses and identify individuals at higher risk for adverse responses
[64, 66, 67]. Single 24-hour dietary recalls, while feasible for pilot studies and
statistically controlled through covariate adjustment, cannot capture day-to-day
dietary variability. Dietary assessment using single 24-hour recalls at baseline
and post-intervention, while consistent with established coffee trial methodology
[29, 30], does not capture day-to-day dietary variability or long-term nutritional
patterns. Total macronutrient intake was statistically controlled as a covariate
in all analyses, and the complete stability of body composition parameters
(weight, BMI, body fat mass; all p
These research design limitations provide important future UTC research priorities. To begin with, it would increase the intervention time to 8–12 weeks to evaluate adenosine receptor tolerance formation, CYP1A2 metabolic accommodation [57], and long-term cardiovascular effects reported in long-term coffee studies [35, 45, 46]. Second, head-to-head comparative trials directly quantifying diterpene content across brewing methods (UTC vs. filtered preparations) with parallel cardiometabolic monitoring would definitively isolate cafestol/kahweol effects [8, 13, 42] from caffeine pharmacology [48, 49], addressing our limitation of inferring mechanisms from literature-derived bioactive estimates [32, 33, 34]. Third, sex-stratified (male) cohort replication using CYP1A2 genotype would be used to ascertain whether our exaggerated response (SBP +4.0 mmHg, HR +10.6 bpm) is indicative of female-specific slower caffeine clearance (20–25% longer half-life [24, 25] or are more generalizable UTC effects. Fourth, the trials that involve populations with cardiometabolic risk (dyslipidemia, prehypertension, metabolic syndrome) baseline would help determine clinical thresholds of contraindications versus acceptable risk [5, 35]. Fifth, objective polysomnography coupled with systematic consumption timing manipulation would quantify circadian disruption beyond self-reported ISI scores [22, 80], revealing optimal windows preserving alertness while minimizing sleep architecture impairment [21, 81]. Lastly, genetic stratification (CYP1A2 -1A-1F alleles dictating metabolizer phenotype; ADORA2A variants dictating adenosine sensitivity [64, 66, 67]) should be included, which would allow precise definition of individuals at high risk, and change UTC guidance (population generalizations) to genetically sensitive, baseline health status-specific and consumption patterns change. All of these would help overcome existing limitations and progress mechanistic insight into complex physiological effects of UTC.
The current results are relevant to the practice of UTC consumers and healthcare providers. The four weeks of moderate daily UTC (three 40-mL cups) resulted in statistically significant changes in a variety of parameters: SBP elevation (+4 mmHg), the increase in HR (+10.6 bpm), reduction of HDL, and increase of LDL levels, leptin decrease, and clinically significant sleep impairment (1.5-point rise in the ISI score). Although the changes were observed in healthy young women, their combination and speed are worth being considered among people with risk factors in the cardiovascular system, dyslipidemia, or sleeping disorders. Counselors who discuss the use of coffee with a patient must separate the filtered and unfiltered coffee because the lipid effects of UTC and filtered coffee vary significantly [8, 41, 63]. It can be recommended that individuals who have pre-existing dyslipidemia, hypertension, or insomnia should limit the intake of UTC or use filtered preparations. The considerable sleep disruption observed even when the consumption ceased by 3–4 PM implies the need to be very mindful of the timing of consumption especially among the caffeine-sensitive individuals. On the other hand, no negative effects on glycemic or inflammatory in our healthy group imply that moderate UTC intake can be permissible among metabolically healthy people with no cardiovascular or sleep issues, however, it is better to consume it earlier in the day [21, 22]. The findings offer evidence-based information on the population in which UTC is culturally relevant and recognize that different people would respond differently depending on genetic influences (CYP1A2 polymorphisms), the current health status, and patterns of consumption [64, 66, 67].
This pilot randomized controlled trial is the first to have conducted a full assessment of the short-term effects of UTC consumption on young women who used culturally sensitive preparation procedures. The nature of the pilot study design such as small sample size (n = 40), no control over the menstrual cycle, and 4 weeks of intervention limits the generalizability and accuracy of effect estimates. This nominal statistically significant (no multiplicity adjustment) hypothesis-generating study showed that four weeks of standardized daily UTC intake was associated with physiological alterations in various systems: cardiovascular (SBP +3.0 mmHg, HR +11.9 bpm), lipid profile (LDL increased; HDL none after baseline adjustment), hormonal (leptin reduced with no effect on ghrelin) and sleep quality (worsened ) impairments, which all happened without a change in body composition. The 4-week intervention period provides initial mechanistic information but is not able to deal with longer period adaptations, tolerance formation, and chronic health results, and all the outcomes of the study are nominal without multiple testing correction. These results are to be construed as coming up with certain hypotheses to be validated in sufficiently powered trials, as opposed to conclusive evidence of causal associations. The rapidity and magnitude of these effects, especially lipid changes and sleeping disturbance, should be given clinical attention in giving advice to UTC consumers. Notably, neither body composition, glycemic, nor inflammatory changes were present despite the severe impact of the cardiovascular and lipid effects, which indicates direct pharmacological action of the bioactive compounds of UTC (diterpenes, caffeine, chlorogenic acids) rather than indirect metabolic pathways. These findings address critical knowledge gaps by: (1) establishing baseline physiological responses in the primary consumer demographic (young women), (2) demonstrating that unfiltered coffee’s effects differ from general coffee literature, and (3) providing evidence for sex-specific research in coffee interventions. Although these preliminary results are pending validation, healthcare professionals can take them into account when advising patients who have already acquired a cardiovascular risk or dyslipidemia or sleep disorders, about UTC drinking habits. Much-power adequately studies to overcome these pilot study limitations such as stratification of the menstrual cycle, larger samples with greater population diversity, and longer intervention periods are necessary to determine clinical applicability and population-wide guidelines. Future research should examine dose-response relationships, conduct comparative trials between brewing methods, investigate sex differences, and evaluate longer-term adaptations to inform evidence-based public health guidance for populations where UTC holds cultural significance.
BFM, body fat mass; BMI, body mass index; BMR, basal metabolic rate; CRP, C-reactive protein; DBP, diastolic blood pressure; ECW, extracellular water; ELISA, enzyme-linked immunosorbent assay; FBS, fasting blood glucose; FFM, fat-free mass; HDL, high-density lipoprotein cholesterol; HbA1c, glycated hemoglobin; HPLC, high-performance liquid chromatography; HR, heart rate; ICW, intracellular water; IRB, institutional review board; ISI, Insomnia Severity Index; LDL, low-density lipoprotein cholesterol; MS, mass spectrometry; PBF, percent body fat; SBP, systolic blood pressure; SMM, skeletal muscle mass; TBW, total body water; UTC, unfiltered Turkish coffee; TG, triglycerides; TNF-
The data presented in this study are available upon request from the corresponding author for privacy, legal or ethical reasons.
Conceptualization, NAE; Methodology, NAE and OAA; Formal analysis, NAE; Investigation, NAE and OAA; Writing—review & editing, NAE & OAA. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This research was conducted based on the ethical principles outlined in the Declaration of Helsinki guidelines. in addition, the Research Ethics Committee of the Faculty of Pharmacy and Medical Sciences at the University of Petra, Amman, Jordan, granted approval before embarking on the research, with an ethical approval number of (Grand number: E/H/5/10/2024, August 2024). All participants gave their informed consent before joining the study. They were fully briefed on the research details, and we ensured that they met the study criteria before moving forward. Each participant signed a written consent form before we began.
The authors would like to express their sincere gratitude to the University of Petra for granting ethical approval to conduct this study
This study was funded (2023/4/27) by the University of Petra, Amman, Jordan.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

