1 Nutrition and Foods Program, School of Family and Consumer Sciences, College of Applied Arts, Texas State University, San Marcos, TX 78666, USA
Abstract
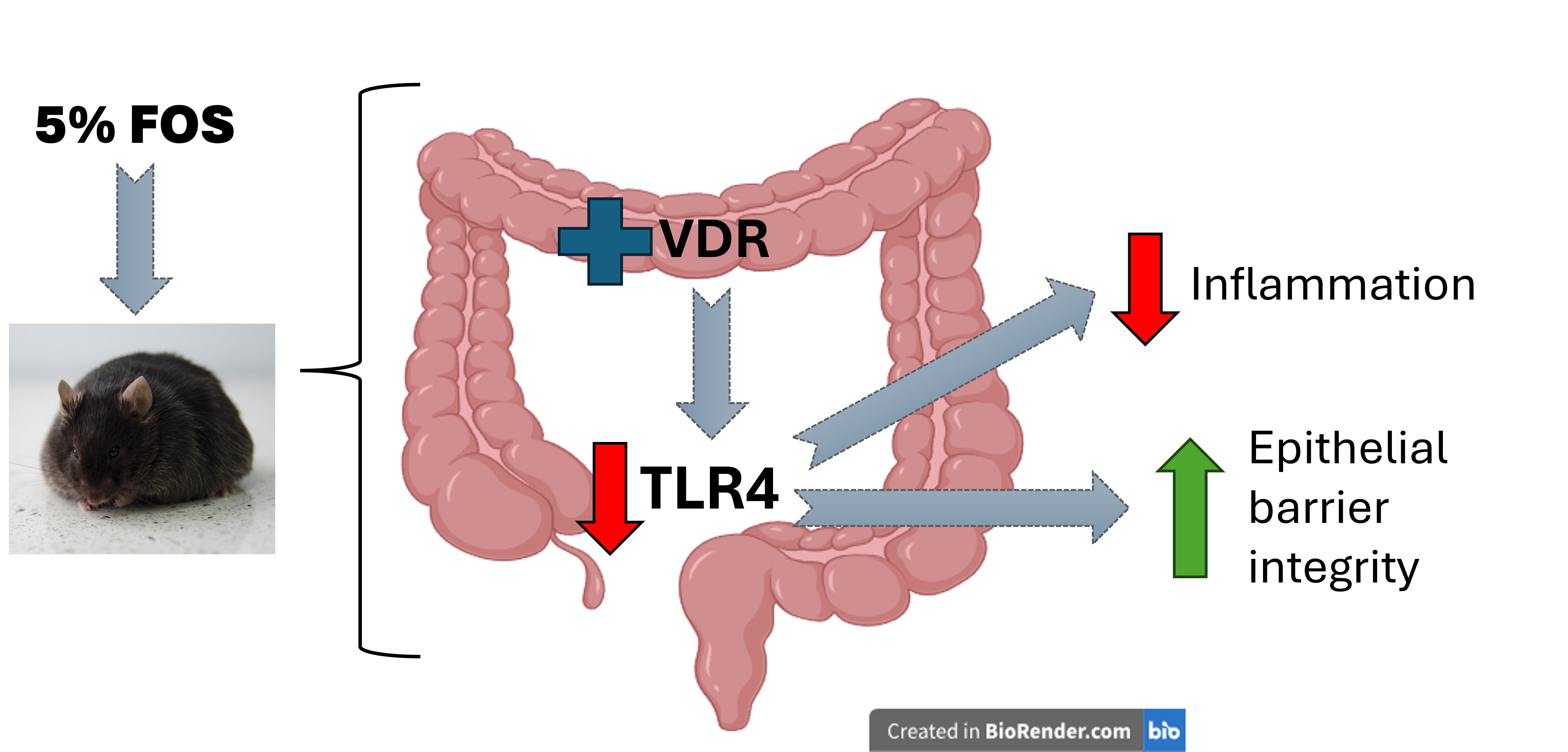
Vitamin D (VD) deficiency is commonly observed in obesity, which may increase morbidity risk. This study explores the effect of fructooligosaccharide (FOS) on VD signaling and inflammatory status in diet-induced obese mice.
Therefore, 5-week-old male C57BL/6J mice were randomly assigned (n = 10/group) to groups that received either a (1) standard purified maintenance AIN-93G control diet (CON), (2) CON + 5% FOS (CON-FOS), (3) high-fat diet (HFD) of which 60% kcal was from fat, or (4) HFD + 5% FOS (HFD-FOS) for 10 weeks.
Mice fed an HFD exhibited reduced serum 25-hydroxycholecalciferol (25D) levels (p < 0.01) and a 70% decrease in the expression of colonic vitamin D receptor (Vdr) mRNA (p = 0.018) compared to the mice fed the CON. A 2-fold increase in colonic Vdr mRNA expression was observed in the mice fed the HFD-FOS compared to the HFD (p < 0.01), although the increased FOS did not alter the serum 25D levels in the HFD group. The mRNA and protein expression of colonic Toll-like receptor 4 (Tlr4) was downregulated in the HFD-FOS group compared to the HFD group, which was negatively correlated to colonic Vdr expression (r = –0.747; p < 0.001). Additionally, the addition of FOS resulted in a 44% reduction in circulating proinflammatory cytokine Interleukin-6 (IL-6) in the HFD group (p < 0.01). The observed upregulation of β-defensin 1 (p = 0.017) and zona-occluden 1 (Zo-1) mRNA expression in mice fed FOS and an HFD compared with the HFD group further suggests that FOS supplementation can improve epithelial barrier integrity in HFD-induced obese mice.
Our data suggest that FOS may be a potential dietary strategy for preventing obesity-induced complications.
Graphical Abstract
Keywords
- fructooligosaccharide
- vitamin D
- obesity
- high-fat diet
- toll-like receptor 4
- colonic inflammation
To date, approximately 42% of American adults (aged 20 or older) are obese [1],
highlighting the need of dietary strategies to prevent obesity and/or
obesity-associated complications. Among the obese population, the prevalence of
vitamin D (VD) deficiency was 35% greater compared to non-obese individuals [2].
VD is a fat-soluble vitamin that plays a critical role in calcium homeostasis. VD
enters the liver where it is converted into 25-hydroxycholecalciferol (25D), also
known as calcidiol, by the enzyme 25-hydroxylase. From the liver, 25D is
transported to the kidney via VD binding proteins for further hydroxylation by
1-hydroxylase, which is converted into 1,25-dihydroxycholecalciferol (1,25D).
1,25D is a ligand of the VD receptor (VDR) to initiate gene transcriptions for
regulation of various physiological functions [3, 4]. In recent years, it was
suggested that the health benefits of VD could extend beyond bone health. Studies
have shown that low level of VD (serum 25D
Obesity is often characterized by low-grade inflammation, which is usually associated with gut dysbiosis. Studies have shown an increase in Firmicutes and a decrease in Bacteroidetes in an obese intestinal environment [9, 10, 11]. In addition, the Western diet and obesity have been associated with a decrease in gut microbiota diversity [12, 13] and a high-fat diet (HFD) has been linked to decreased Bifidobacterium and increased gram-negative phylum that may lead to the production of lipopolysaccharides (LPS) [14, 15]. LPS is a part of the membrane of the gram-negative bacteria and functions as a ligand for Toll-like receptor 4 (TLR4) to enhance pro-inflammatory reaction. Consumption of a HFD or typical Western diet have been linked to increased levels of circulating LPS, likely due to compromised intestinal barrier integrity resulting from epithelial mucosa damage that allows the translocation of endotoxin. This could further promote local and systemic inflammation leading to a condition that is known as metabolic endotoxemia [16, 17, 18]. It is also important to note that gut microbiota can serve as a regulator of antimicrobial peptides productions (AMPs), such as alpha and beta defensins, to regulate the innate immune response within the gastrointestinal tract [19, 20, 21, 22]. Probiotics and microbial metabolites, such as short chain fatty acids, have been shown to promote the expressions of tight junction proteins and AMP productions in vivo [23, 24], suggesting a vital role of gut microbiota in maintaining the integrity and innate response of the gut epithelium.
Fructooligosaccharide (FOS), a soluble fiber also known as oligofructose, contains approximately 2–60 units of fructose molecules linked by beta (2,1) glycosidic bonds [25, 26], which allows FOS to be fermented by gut bacteria and resist enzymatic digestion. FOS can be found in multiple plant sources such as onions, wheat, and chicory and has been shown to exert bifidogenic potential [27, 28, 29]. Administration of FOS has been linked to improve metabolic health, such as improvement of glucose and lipid metabolism [30, 31, 32]. Our previous work demonstrated that intake of resistant starch, a prebiotic fiber, prevented renal VD loss and morbidities in diabetic rats [33, 34]. These findings suggest that restoration of VD balance could be beneficial in preventing secondary complications resulting from metabolic diseases. Because resistant starch is a fermentable fiber, the gut microbiota is thought to constitute one of the underlying mechanisms. In support of this concept, we have further demonstrated that supplementation of FOS and VD upregulated the colonic VDR expression in lean mice compared to those fed on VD alone [35, 36]. Furthermore, the regimen suppressed the abundance of a pathobiont, Romboutsia ilealis, altered the beta diversity of gut microbiome, and modulated the expressions of intestinal defensins, a class of AMPs [36]. Taken together, this may suggest that FOS could potentially activate colonic vitamin D signaling to regulate innate immune response, possibly via modulation of the gut microbiota.
Though we have demonstrated that the combination of FOS and VD can regulate intestinal permeability and innate response through intestinal VDR activations [36], the mechanism by which FOS and/or its fermented by-products can regulate VD homeostasis under an obesogenic environment remains unclear. Because activation of vitamin D signaling has shown to modulate the gut microbial profile and protect against gastrointestinal diseases [37, 38], it is speculated that FOS can serve as an effective dietary intervention for prevention obesity-associated inflammation by modulating VD signaling pathway. This study aims to investigate the effect of FOS on colonic VD signaling in relation to their metabolic changes and inflammatory status utilizing a HFD-induced animal model. We expect that the information generated from this study can further elucidate the role of gut microbiota on bioavailability of essential nutrients and serve as a novel strategy to improve quality of life among populations who are susceptible to VD deficiency.
In this experiment, five-week-old male C57BL/6J mice were obtained from Jackson Laboratory (Bar Harbor, ME, USA) and all mice were individually caged and acclimated for one week. Diets were purchased from Research Diets, Inc (New Brunswick, NJ, USA) and FOS was supplied by Beneo (Orafti-P95, Mannheim, Germany), composed of ~95% of oligofructose derived from chicory root, to Research Diets for customization. All diets were irradiated. At 6-week of age, mice were randomized into 4 groups (n = 10/group) to receive either a control AIN-93G diet (CON), high-fat diet of which 60% kcal was from fat (HFD; D12492), CON + 5% w/w FOS (CON + FOS), and HFD + 5% w/w FOS (HFD + FOS) for 10 weeks. Both the CON and HFD diets contain a base concentration of 1000 IU/kg of VD. The selected FOS dosage has been shown to upregulate the expression of colonic VDR in our previous study [35, 36], alter metabolic phenotypes, as well as induce the abundance of Bifidobacterium [39, 40]. All mice had unlimited access to water and their assigned diet throughout the experimental period. Body weight and food intake were monitored and recorded weekly. By the end of the 10th week of intervention, mice were subjected to overnight fasting and anesthesized with 3% isoflurane via inhalation. Blood was collected via cardiac puncture, followed by euthanasia by cervical dislocation. Colon and ileum sections were collected, and mucosa was scraped for subsequent analyses. All samples were stored at –80 °C until analysis.
The serum was obtained via centrifugation of whole blood at 1200
Mouse serum was prepared from whole blood via centrifugation at 1200
Colonic and ileal mucosa were scraped and homogenized prior to total RNA extraction using TRIzol® reagent (#15-596-026, Thermo Fisher Scientific, Waltham, MA, USA). RNA concentrations were determined using a Nanodrop Lite (Thermo Fisher Scientific, Waltham, MA, USA) and cDNA was synthesized using the Superscript® IV First-Strand Synthesis System (#18-090-050, Invitrogen, Waltham, MA, USA). Amplification of target transcripts was obtained via Power up SYBR® Green Master Mix and Quant Studio 3® real-time PCR system (A25776, Thermo Fisher Scientific, Waltham, MA, USA). Primers for all target genes used in this study are listed in Supplementary Table 1. The expression of each target gene was normalized against glyceraldehyde-3-phosphate dehydrogenase (Gapdh) and relative expression compared with the CON group was calculated.
Colonic mucosa was homogenized in 1X RIPA buffer (PI89900, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with protease and phosphatase inhibitors (PIA32961, Thermo Fisher Scientific, Waltham, MA, USA) and total protein was extracted. Protein quantification was obtained using the BCA assay (#23227, Thermo Fisher Scientific, Waltham, MA, USA). A total of 50 µg of protein sample was loaded on a 4–12% gradient sodium dodecyl sulfate–polyacrylamide gel and transferred onto a nitrocellulose membrane (PI88014, Invitrogen, Waltham, MA, USA). Membranes were probed with primary TLR4 (NB100-56580SS, Novus Biologicals, Centennial, CO, USA) and GAPDH (SC32233, Santa Cruz Biotechnology, Dallas, TX, USA) antibodies, followed by incubation with anti-rabbit (AP187MI, MilliporeSigma, Burlington, MA, USA) or anti-mouse (#45-000-692 Cytiva, Marlborough, MA, USA) secondary antibodies. All antibodies were diluted to 1:1000, unless otherwise specified. Proteins were visualized with enhanced chemiluminescence reagent (PI34094, Thermo Fisher Scientific, Waltham, MA, USA) using a Gel-Doc Image Analysis System (Fotodyne Incorporated, Hartland, WI, USA). Densitometry of protein bands were quantified using ImageJ software (version 1.54p, National Institutes of Health, Bethesda, MD, USA) and data are expressed as ratio relative to CON.
Sigma Plot v. 14.5 (Inpixon, Palo Alto, CA, USA) was used for all statistical
analyses. All endpoints were analyzed using two-way analysis of variance followed
by Tukey’s Honestly Significant Difference (HSD) post hoc test, which adjusts for
multiple comparisons and controls the family-wise error rate. Data for colonic
TLR4 protein expression were not normally distributed; hence, data were
log-transformed for analysis to achieve normality (p = 0.765).
Statistical analyses of mRNA expression data were determined with cycle threshold
and expressed in relative to CON as indicated. Correlation between colonic
Vdr and colonic Tlr4 mRNA expressions were determined by
Pearson’s correlation coefficient. Statistical significance was indicated at
p
HFD mice exhibited lower VD status, as measured by serum
25D, compared to CON mice (p
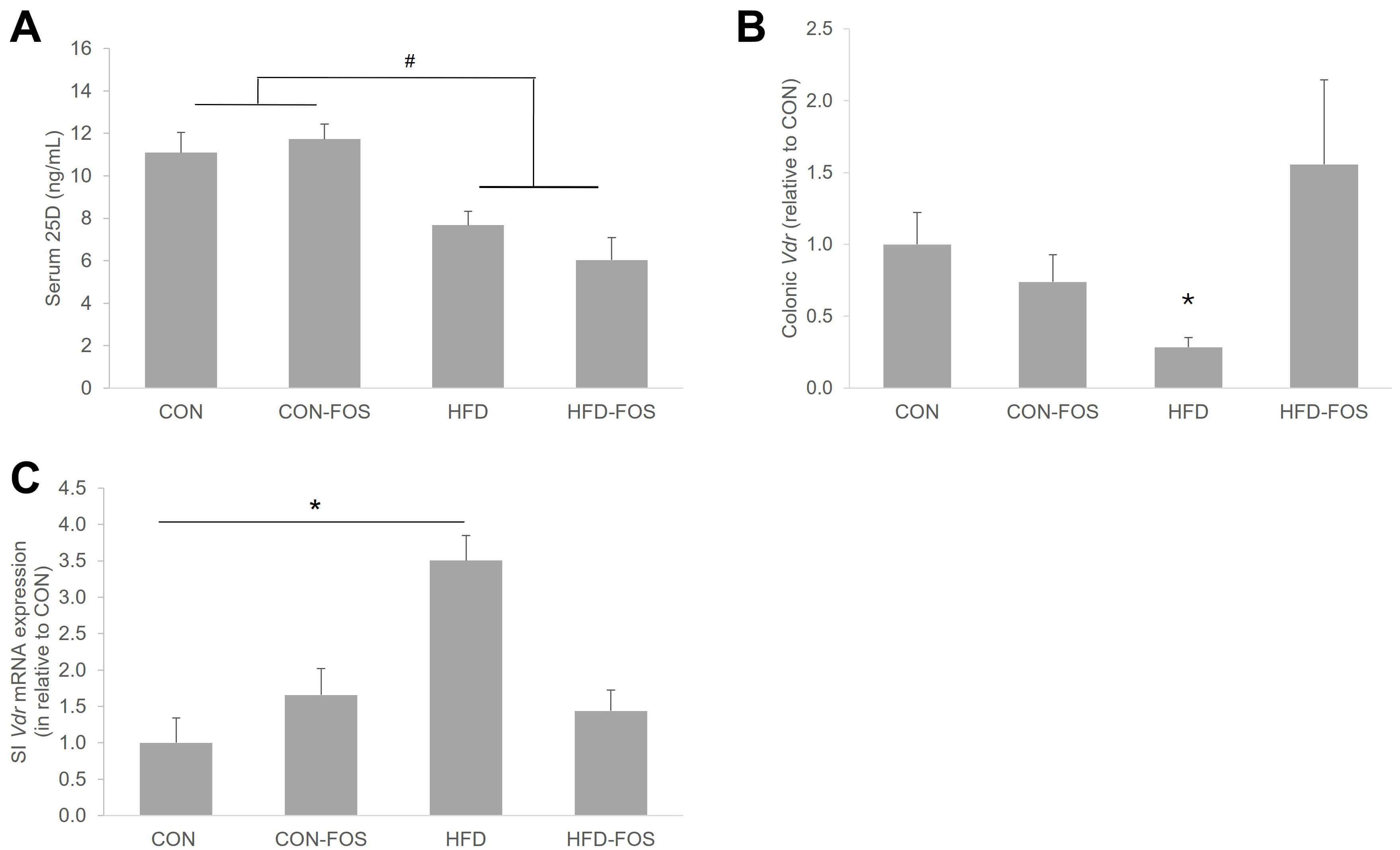 Fig. 1.
Fig. 1.
Circulating 25D in the HFD mice was not impacted by FOS
intervention (A), though differential effects of FOS were observed with
Vdr mRNA expressions in colon (B) and ileum (C) in the HFD mice.
Statistical differences between dietary interventions are expressed as
*p
We further measured the circulating levels of pro-inflammatory cytokines, IL-6
and IL-1
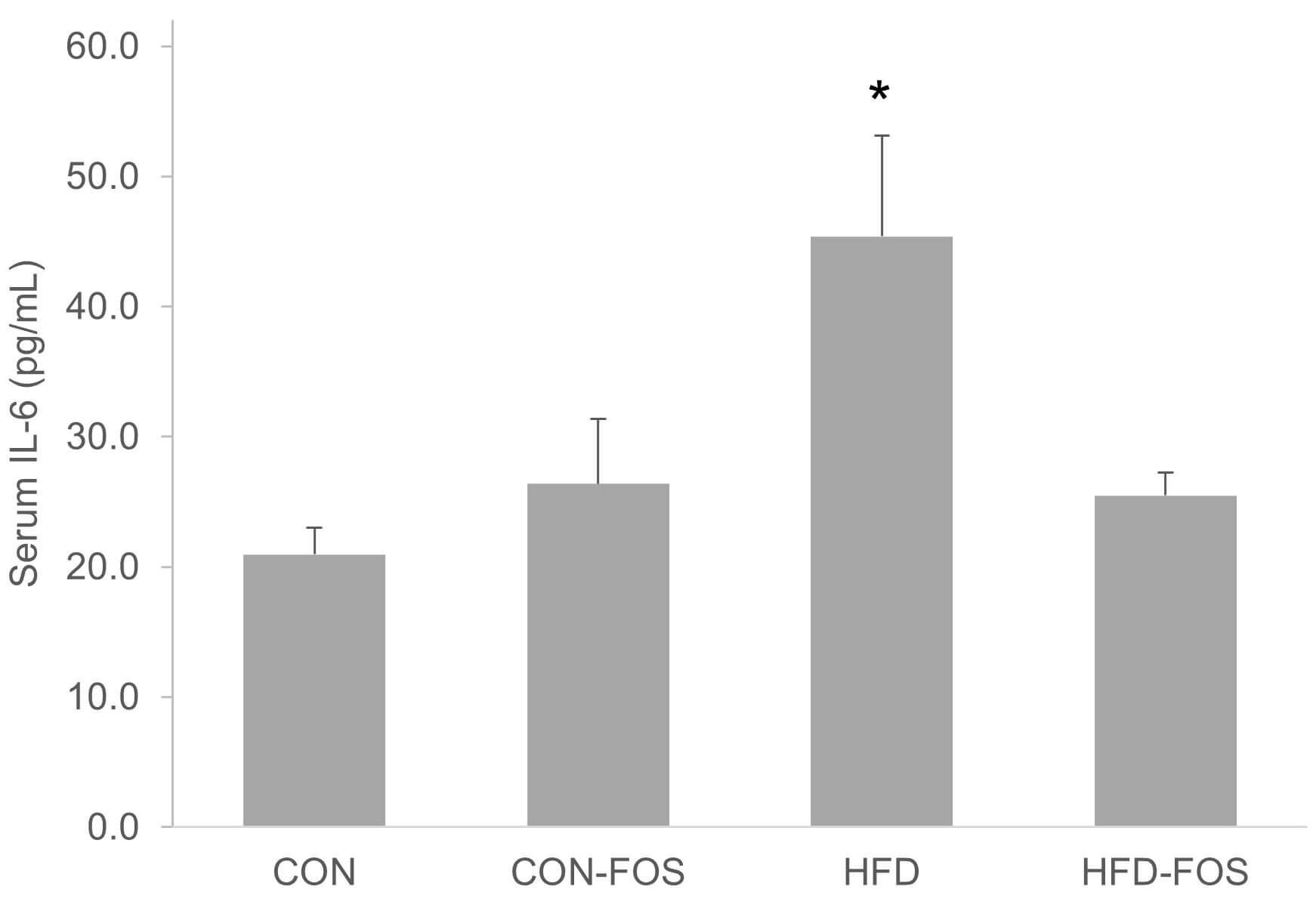 Fig. 2.
Fig. 2.
FOS suppressed the elevation of circulating IL-6 in HFD mice.
Statistical differences between dietary interventions are expressed as
*p
To further evaluate the local inflammation within the colon, mRNA expressions of
selective pro-inflammatory cytokines were measured. In contrast to circulating
IL-6 concentration, colonic mRNA expression of Il6 in HFD mice was lower
compared to CON mice (p = 0.066), and that HFD-FOS mice was 4-fold
greater compared to HFD-fed mice, though it was not statistically different
(p = 0.079; Fig. 3A). A similar trend was observed with colonic
Il1
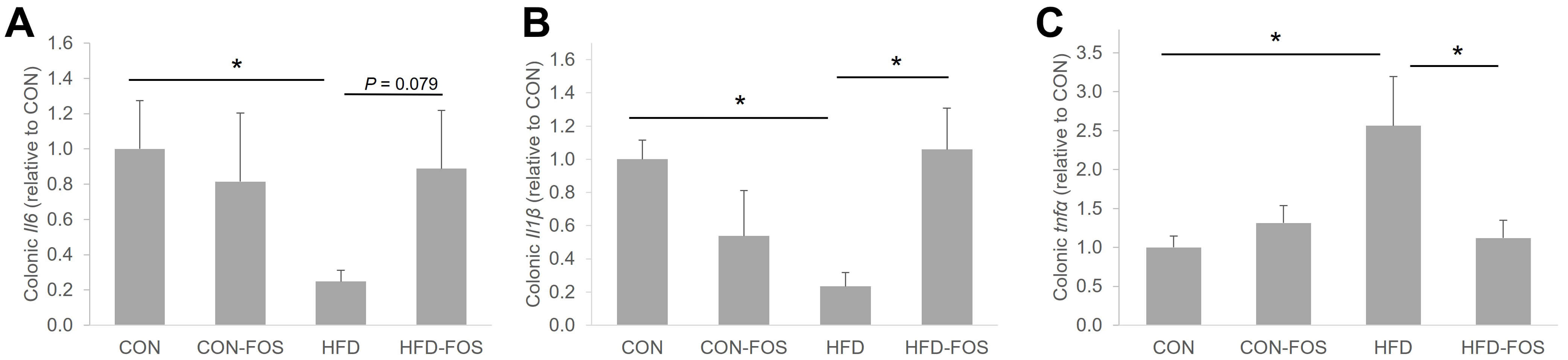 Fig. 3.
Fig. 3.
Colonic Il6 (A) and Il1
Compared to the CON mice, the endotoxin level, as indicated by circulating LPS
concentration, in the HFD mice was 33% higher (p
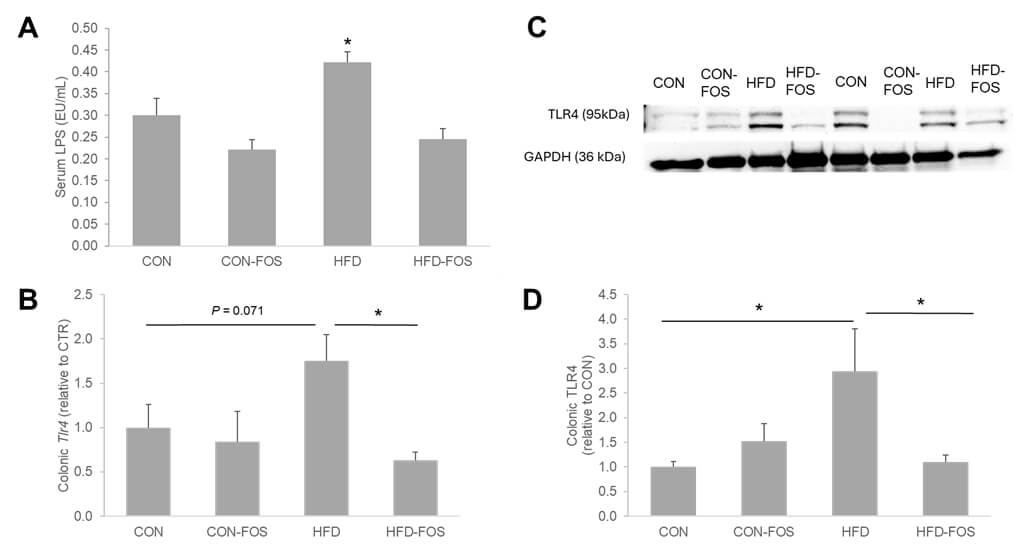 Fig. 4.
Fig. 4.
FOS attenuated the elevation of circulation LPS in HFD mice (A),
along with a suppression of TLR4 mRNA (B) and protein (C,D) expressions. (C)
Representative Western blot analysis of TLR4. Statistical differences between
dietary interventions are expressed as *p
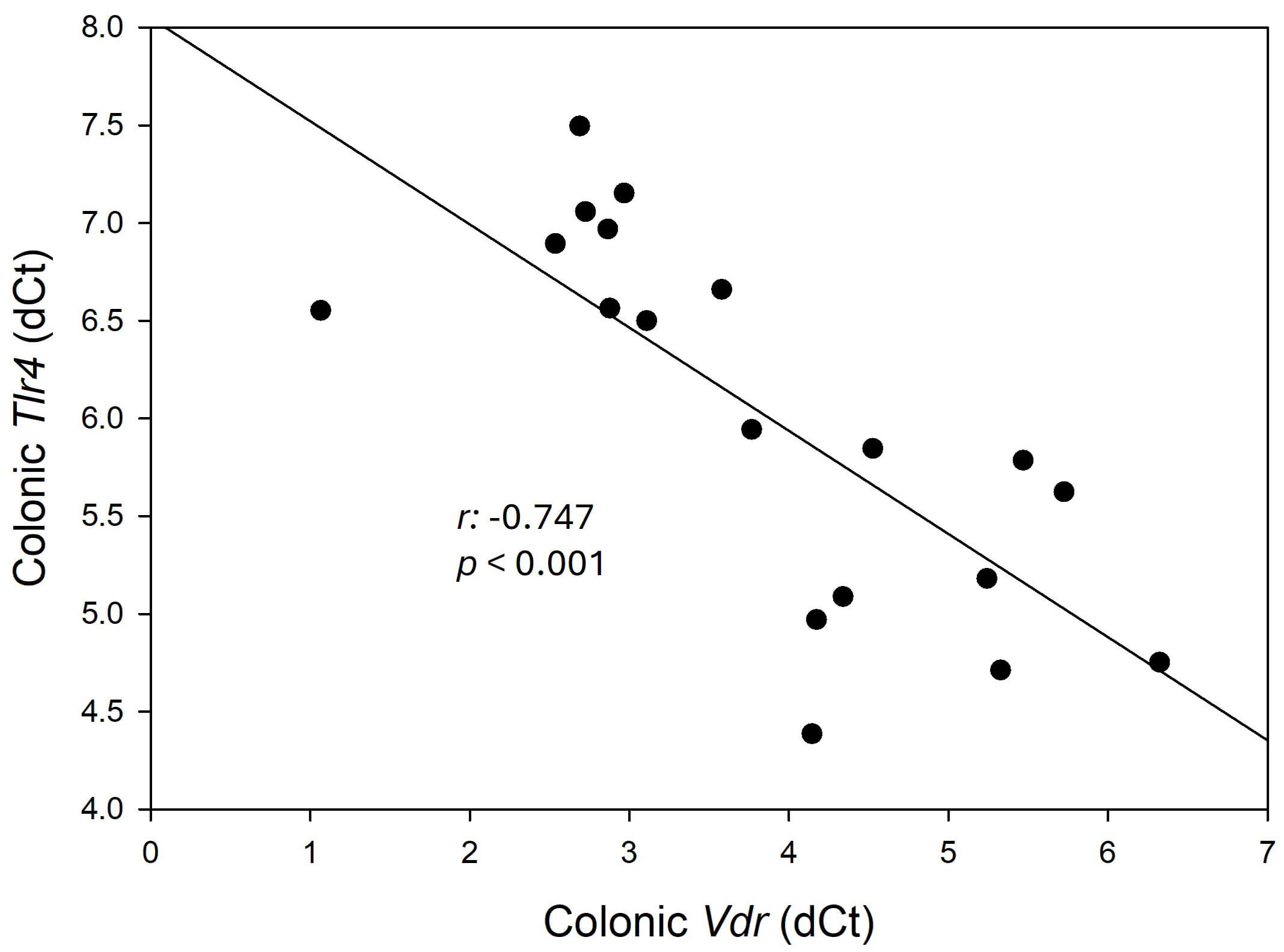 Fig. 5.
Fig. 5.
Colonic Vdr mRNA expression is inversely correlation
with colonic Tlr4 mRNA expression. Correlational analysis was conducted
using Pearson’s correlation. Each dot represents individual data. Data are
expressed as mean
In our previous study [36], it was demonstrated that treatment with VD in combination with FOS downregulated selected defensins in the ileum of mice fed a control diet suggesting a potential role of FOS in regulating the secretion of these antimicrobial peptides. Here, utilizing the HFD-induced obese model, we demonstrated a diet effect (CON vs. HFD) in both mRNA expressions of Dfa1 (p = 0.01) and Dfa5 (p = 0.02). However, FOS did not affect Dfa1 nor DFa5 mRNA expressions in the ileum (Fig. 6A,B), though a trend of Dfb1 upregulation by almost 10-fold was observed in the FOS-treated HFD mice compared to the HFD mice (p = 0.017; Fig. 6C).
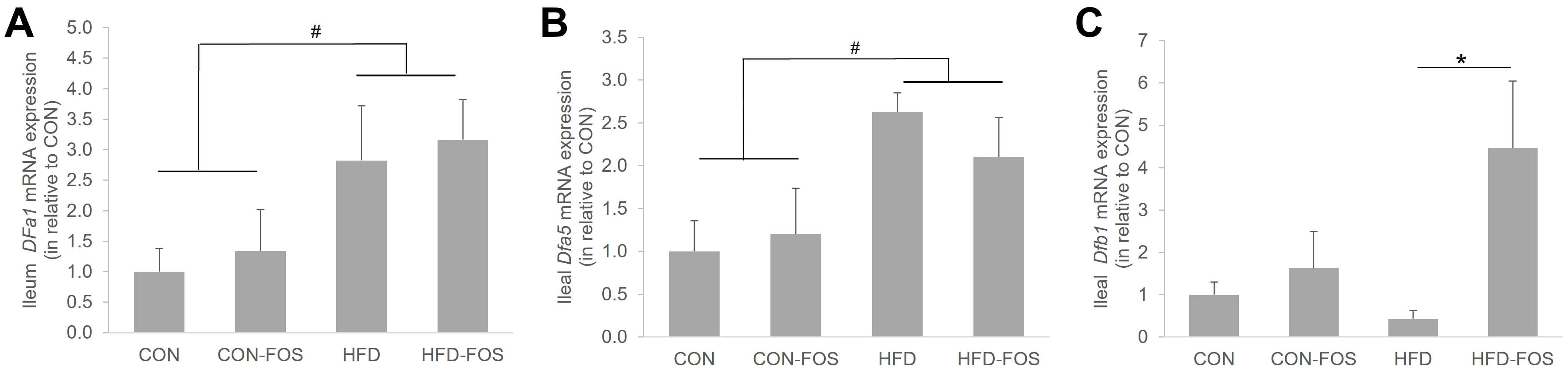 Fig. 6.
Fig. 6.
FOS did not affect the mRNA expressions of ileal Dfa1
(A) and Dfa5 (B) but upregulated ileal Dfb1 mRNA expression (C)
in HFD mice. Data are expressed as mean
VDR has been shown to regulate the secretions of defensins. Our current study confirmed a positive correlation between the mRNA expressions of Vdr and Dfa1 (r = 0.48; p = 0.03) and Dfa5 (r = 0.47; p = 0.03) in the ileum, respectively (Table 1). However, no correlation was detected between ileal Vdr and Dfb1 mRNA expressions (Table 1).
| Target genes | Pearson’s correlation coefficient (r) with ileal Vdr | p-value |
| Dfa1 | 0.487 | 0.02 |
| Dfa5 | 0.477 | 0.03 |
| Dfb1 | –0.13 | 0.57 |
The integrity of the gut barrier was indicated by the mRNA expressions of
Zona-occluden-1 (Zo1) and occludin (Ocln) in
the colon. As expected, HFD downregulated the mRNA expression of colonic
Zo1 by 80% compared to mice on a CON diet (p
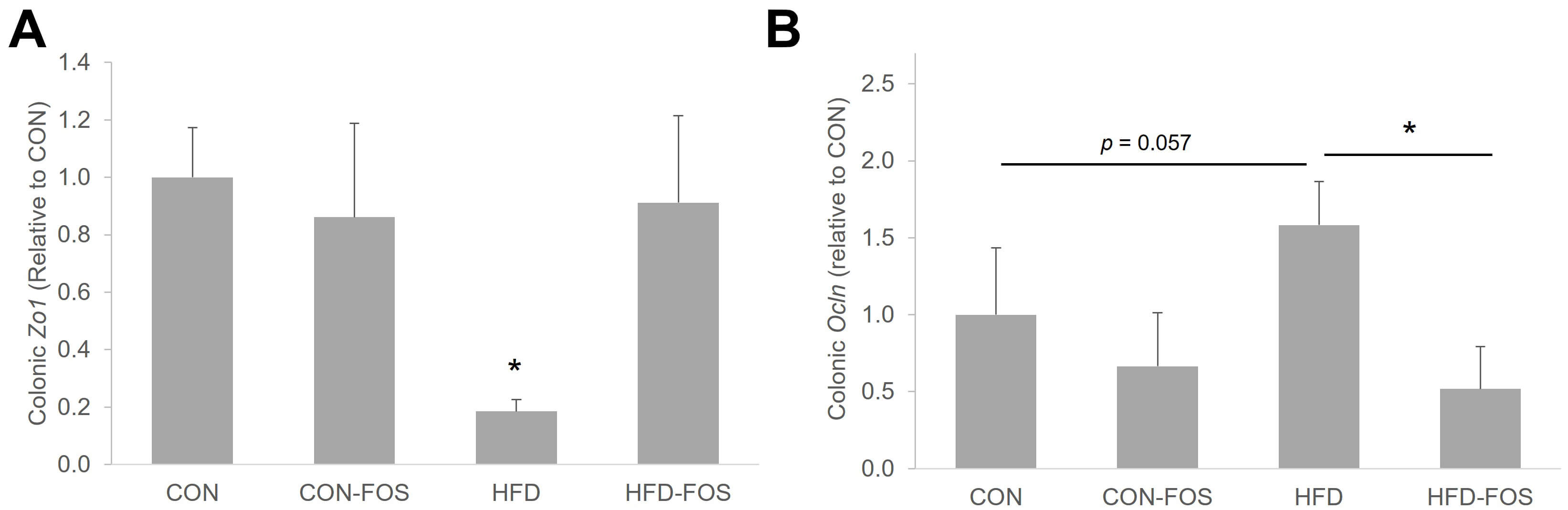 Fig. 7.
Fig. 7.
FOS exerted differential effect on mRNA expressions of
colonic Zo1 (A) and Ocln (B). Statistical differences between
dietary interventions are expressed as *p
Our current study suggests a potential role of FOS in suppressing LPS-induced
TLR4 activation in HFD-induced obese mice, likely through a VDR-dependent
pathway. Though it is expected to observe low vitamin D status in obesity due to
it being sequestered within the adipose tissues [2], our results indicated that
5% FOS was not effective in restoring vitamin D levels within the HFD mice,
regardless of body weight changes. However, the FOS administration upregulated
colonic Vdr among the HFD mice. This is in alignment with our previous
study, in which we demonstrated that FOS, in combination with vitamin D
supplementation, enhanced the mRNA expression of colonic Vdr, though
serum 25D was unaltered [35]. Because food intake did not differ among groups,
this may suggest that the effect of FOS or gut microbial metabolites derived from
FOS fermentation could be targeting the downstream pathway of vitamin D
metabolism, possibly the conversion of 25D to 1,25D locally in the colon. Unlike
in the colon, we observed a down-regulation of ileal Vdr in the
FOS-treated HFD mice. This coincides with our previous studies that mRNA
expressions of Vdr in the colon and ileum are independently expressed
despite the status of vitamin D [35, 36]. Because FOS fermentation mainly occurs
in the colon, this led us to speculate that the differential expressions of
Vdr along the GI tract could be relevant to bacterial activity and
concentrations of microbial metabolites, such as short chain fatty acids, which
have been shown to directly regulate VDR activity in vivo [41]. A recent
study further demonstrated that Carnobacterium maltaromaticum can induce
the production of intestinal vitamin D to activate VDR for prevention of
colorectal cancer [42]. This may partially explain the changes of colonic VDR
independent of circulating 25D level and body weight changes in our study.
Characterization on relevant vitamin D markers, such as 1,25D concentrations and
1-
It is well characterized in obesity models that elevation of LPS, or metabolic
endotoxemia, leads to the activation of TLR4, which could subsequently promote
the secretion of pro-inflammatory cytokines [14, 15, 43]. While our results are
in line with the literature, we further demonstrate that the inhibition of
colonic TLR4 could be attributed to VDR activation due to the strong inverse
correlation between colonic Tlr4 and Vdr mRNA expressions. It
is well known that TLR4, when bound to LPS, recruits either TRIF or MYD88 adaptor
proteins that ultimately leads to nuclear factor kappa-B (NF-kB) translocation
and production of cytokines [44]. The relationship between TLRs and the VDR
remains understudied. TLR interplay with VD is primarily understood in the
context of immunology in which a co-receptor of TLR4, CD14, is known to be
regulated by 1,25D [45]. In addition, ex vivo treatment of 1,25D to
healthy human monocytes downregulated both mRNA and protein levels of TLR4 and
TLR2 [46]. Taken together, these studies may partially explain the inverse
relationship we observed in the present study between the mRNA expressions
of Vdr and Tlr4. Another key aspect of TLR-VDR interactions may
be their shared modulation of NF-kB transcription. Typically, NF-kB is downstream
to TLRs signaling pathway, while VDR modulates NF-kB post- and
pre-transcriptionally [47]. VDR can inhibit LPS or TNF
Chronic inflammation is often characterized by an increased level of
pro-inflammatory cytokines, along with a decreased level of anti-inflammatory
cytokines, which is commonly observed during the progression of obesity. At the
circulating level, though we were unable to detect IL-1
Compromised tight junctions can result in bacterial translocation leading to inflammation as previously discussed. Studies have shown that prebiotics or dietary fiber can normalize the intestinal permeability and immune health in vivo [57, 58, 59]. In the present study, we showed that the protective effects of FOS against obesity-induced low-grade inflammation may be partially mediated through enhanced expressions of beta defensin and tight junction protein, ZO-1. It should be noted that the expressions of colonic Zo1 and Ocln in the HFD mice were differentially impacted by FOS, leading us to speculate that the effect of FOS was specific to ZO-1. This is likely due to the short duration of our study in which the initial destabilization of tight junctions may be primarily involved with the cytoplasmic scaffolding proteins than transmembrane proteins like Ocln. Hence, the observed differences may reflect selective remodeling of tight junction components by FOS rather than restoration of disrupted tight junction.
The role of defensins in promoting epithelial barrier damage in addition to their antibacterial activities has been extensively reviewed and researched [19, 20, 60]. Compromised expressions of alpha and beta defensins have been reported in patients with Crohn’s disease, which further elevates colonic inflammation among these patients [61, 62]. Gut microbial metabolites, such as short chain fatty acids, can upregulate the expressions of alpha and beta defensins in GI tract [21, 23, 63] and hence may promote intestinal barrier integrity. However, in our current study, specifically in the HFD mice, alpha defensins were not affected by FOS intervention, yet the expression of beta defensin was 4-fold greater in FOS-treated HFD mice. The secretions of these defensins can be differentially regulated. For example, specific live microbiota, such as Enterococcus faecium, has been shown to promote beta defensin productions [64], while VDR has also been shown to regulate the synthesis of alpha defensin 5 [60]. However, utilizing a HFD-induced obese model in this present study, though we confirmed the role of ileal VDR in regulating the secretions of alpha defensins as reported previously [36], the mRNA expression of Dfb1 remained unchanged by FOS. While it is beyond our scope of investigation, it is possible that specific gut microbiota resulting from FOS fermentation may cause the enhanced production of Dfb1, a pathway that could be independent from VDR activation under obesogenic conditions. However, the impact of the gut microbiota and AMP secretions on intestinal permeability and to what extent remain unclear.
This is the first study demonstrating the protective effect of FOS on obesity-associated colonic inflammation and gut health, possibly via activation of VDR. Limitations in this study are the short duration of interventions and exclusion of female mice in this study. Previously, we reported a significant difference with colonic VDR utilizing a healthy model, hence justified the current study timeline. However, because obesity is a chronic disease, and low-grade inflammation often develops gradually over time, our interpretation of these measured outcomes could be limited. Furthermore, while both male and female mice fed a HFD diet exhibited greater body weight gain compared to those fed a CON diet, FOS attenuated body weight gain only in male mice by ~10%, but not in female (data not shown). Hence, only male mice were included in the study to further our investigations. We further acknowledge the importance of quantifying the proteins levels of critical markers in our study, specifically Vdr, to validate the function impact of VDR on obesity-associated inflammation. Due to sample limitations, we were unable to quantify the protein level of VDR, though our previous studies, mRNA expression of Vdr has shown strong correlation with our measured outcomes [35, 36, 49]. Additionally, since FOS is a prebiotic commonly metabolized by gut microbiota, profiling of the gut microbiome and microbial metabolites may provide insight into the mechanisms underlying the protective effect of FOS. However, this is currently beyond our scope of investigations.
Collectively, we demonstrated that FOS suppressed HFD-induced TLR4 upregulation in our mouse model, improved intestinal barrier integrity, and potentially stimulated the production of beta defensin. These positive outcomes observed in the colonic microenvironment that is associated with FOS intake could be attributed to the activation of VDR, which lead to subsequent attenuation of TLR4. However, interactions between VDR and TLR4 in regulating colonic inflammation will require further investigation. Due to the immunomodulatory role of VDR, the use of FOS to target VD signaling may serve as a crucial and novel intervention for prevention of metabolic complications in populations who are more susceptible to vitamin D deficiency, such as obesity and Type 2 diabetes. Future research is required to elucidate the interactions between the gut microbiota, vitamin D, and innate immunity following FOS intervention.
VD, vitamin D; 25D, 25-hydroxycholcalciferol; 1,25D, 1,25-dihydroxycholecalciferol; FOS, fructooligosaccharide; HFD, high-fat diet; CON, control diet-fed; LPS, lipopolysaccharide; TLR4, toll-like receptor 4; IL-6, interleukin-6; IL-1
All data generated or analyzed during this study are indicated in this article. The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Conceptualization, GYK; methodology, GYK; formal analysis, KB, EB, SH; investigation, KB, EB, ZN, LL; data curation, GYK; writing—original draft preparation, GYK, KB, SH; writing—review and editing, KB, EB, ZN, LL, SH, GYK; visualization, GYK; supervision, GYK; funding acquisition, GYK. All authors have read and agreed to the published version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animals were handled according to the Guide for the Care and Use of Laboratory Animal (National Research Concils, USA). Animal protocol was approved by the Institute Animal Care and Use Committee at Texas State University, San Marcos, TX 78666, USA with an assigned protocol number 9201.
Not applicable.
This research is sponsored by Research Enhancement Program 2024, an internal fund awarded to Dr. Gar Yee Koh provided by Texas State University.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/IJVNR45457.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







