1 College of Biotechnology and Bioengineering, Zhejiang University of Technology, 310032 Hangzhou, Zhejiang, China
2 Research and Development Department, Suntory Beverage and Food Asia Pte. Ltd, 189773 Singapore, Singapore
Abstract
Neuroinflammation is a chronic disease of the nervous system, and timely prevention and treatment could effectively prevent its progression to severe diseases. Essence of chicken (EC) is a mixture of peptides and amino acids extracted from whole chickens which has been proven to alleviate neuroinflammation and improve cognition. The aim of the present study was to screen for potential peptides possessing excellent effects in alleviating neuroinflammation and to explore their synergistic effects.
BV2 cells were treated with 0.1, 1, or 10 mM dietary peptides found in EC in the presence of 1 μg/mL lipopolysaccharide (LPS) for 16 h to screen for potent compounds. LPS-treated BV2 cells were then cultured with 0.03 or 0.1 mM potent bioactive compounds alone or in combination with 0.1 mM carnosine (CAR) for 16 h to determine their synergistic effects. These synergistically treated BV2 cells were further cocultured with HT-22 cells for 24 h to clarify their neuroprotective effects.
Among the eight bioactives of EC, CAR, pyroglutamic acid (PA), and cyclo(Gly-Pro) exerted notable anti-inflammatory and antioxidative effects on BV2 cells, as evidenced by the downregulation of inflammatory cytokine and chemokine expression (10 mM treatment group vs. LPS: p < 0.05), attenuation of the inflammatory pathway (all treatment group vs. LPS: p < 0.05), and increase in activity of superoxide dismutase (SOD) (p < 0.01), catalase (CAT) (p < 0.05), and the content of glutathione (GSH) (p < 0.01). The experiment was conducted in triplicate. Interestingly, PA and cyclo(Gly-Pro) showed a marked synergistic effect with CAR on neuroinflammation. For example, the content of inflammation markers, tumor necrosis factor-α (TNF-α) (CAR + PA vs. CAR: p = 0.030, CAR + cyclo(Gly-Pro) vs. CAR: p = 0.008) and monocyte chemoattractant protein-1 (MCP-1) (CAR + PA vs. CAR: p = 0.039, CAR + cyclo(Gly-Pro) vs. CAR: p = 0.020), were decreased markedly. The coculture of BV2 microglia with HT-22 neurons revealed that CAR, PA, and cyclo(Gly-Pro) also worked synergistically to reduce LPS-induced cytotoxicity in BV2 cells to protect the growth of HT-22 cells (all treated groups vs. LPS: p < 0.05). The protective effect of these dietary peptides might be regulated by the Phosphatidylinositol 3-kinase/Protein Kinase B (PI3K/AKT) pathway-mediated inhibition of apoptosis.
CAR, PA, and cyclo(Gly-Pro) exhibited potent anti-inflammatory and antioxidant effects, suggesting that EC dietary peptides are neuroprotective candidates and that the intake of a mixture rather than individual peptides confers more beneficial effects in maintaining healthy neuron function.
Keywords
- essence of chicken
- carnosine
- cyclopeptides
- neuroinflammation
- antioxidant
- neuroprotection
Neuroinflammation refers to the inflammatory response that occurs within the nervous system and involves a series of changes in neurons and neuroglial cells. Typically, neuroinflammation is triggered by microbial infections [1], mechanical injuries [2], toxic substances in the environment [3], and chronic psychological factors such as anxiety [4] and aging [5]. Moreover, neuroinflammation also exerts detrimental effects, including impairment of consciousness, attention, and memory [6, 7], resulting in neurodegenerative disorders and autoimmune neurological diseases, which can significantly impair quality of life [8, 9, 10]. To mitigate preventable pathogenic factors in daily life, neuroinflammation can be effectively managed through the adoption of a healthy lifestyle and dietary interventions, thereby minimizing the adverse effects associated with pharmacological treatments. Hence, the intake of nutritional supplements and specific nutrients has become an important alternative for maintaining cognitive health and neurofunction.
Essence of chicken (EC) has a long history of consumption among Asian populations and is widely accepted as a dietary supplement [11]. BRAND’S Essence of Chicken is the extract of whole chicken through high-temperature and high-pressure cooking technology to retain most of the nutritional essence of the chicken. Studies have demonstrated that EC has a variety of biological activities, such as relieving stress and fatigue, improving learning and memory, promoting metabolism and postpartum lactation, and improving hyperglycemia and hypertension [12, 13, 14]. Our previous studies also revealed that EC supplementation has a significant protective effect on neuroinflammation and cognitive decline in elderly mice [15]. Therefore, EC is a potent supplement for protection against neuroinflammation.
Numerous studies have shown that food-derived bioactives markedly modulated human physiological processes. For example, licorice-derived licochalcone A prevents cognitive decline in a lipopolysaccharide(LPS)-induced neuroinflammation mouse model [16]. Dietary peptides are protein fragments bioactives derived from natural protein through digestion and decomposition that have improved effects on human neurofunction and metabolic homeostasis [17, 18, 19]. EC comprises approximately 95% proteinaceous material by dry weight and is free of fat and cholesterol [12], involving some key peptides and essential amino acids, such as carnosine (CAR), pyroglutamic acid (PA), cyclodipeptide, threonine, and valine [12, 20]. CAR, as the major dietary peptide of EC, has been demonstrated to possess anti-inflammatory and nitric oxide-scavenging abilities [21]. PA has been shown to reduce neuronal cell death induced by glutamate toxicity [22], enhance memory and cognitive function, and demonstrate a certain degree of antidepressant efficacy [23, 24]. However, few studies have reported the anti-neuroinflammatory effects of other small-molecule cyclic dietary peptides. Additionally, cyclopeptides are 2,5-diketopiperazine (DKP) bioactives that account for a significant proportion of EC. DKP is reported to exhibit high chemical stability and is resistant to digestion into amino acids [25]. Furthermore, their N-methylated derivatives readily cross the blood–brain barrier (BBB), which can increase their viability in the nervous system [26] and thereby protect neurofunction. For example, our previous study revealed that cyclo(Val-Pro) notably attenuated inflammation in HT-22 neurons [27]. With respect to the complex constituents of EC, the specific bioactives responsible for ameliorating neuroinflammation remain unidentified, and a research gap concerning the synergistic effects of multiple bioactives exists.
Thus, the present study aimed to screen the potential bioactive compounds of EC with anti-inflammatory and antioxidative effects and clarify their synergistic effects on neuroinflammation. The effects of eight bioactive compounds from EC were first investigated in BV2 microglia treated with LPS by assessing inflammatory gene expression and antioxidant enzyme activity. The screened bioactive compounds were further used to clarify the synergistic effect. Finally, the neuroprotective effect of these bioactive compounds was determined by coculture of BV2 cells and HT-22 neurons. The results of our study may establish a theoretical foundation for the neuroprotective effects of dietary peptides.
The bioactives in the EC and their concentrations were analyzed and determined by Suntory Beverage and Food Asia Pte. Ltd. The eight main bioactive compounds used for further investigation were CAR (C9625, Sigma Aldrich, St. Louis, MO, USA), PA (83160, Sigma Aldrich, St. Louis, MO, USA), cyclo(Gly-Pro) (GA21330, Glpbio, Montclair, CA, USA), cyclo(Ser-His), cyclo(Pro-Pro), cyclo(Arg-Pro), cyclo(Lys-Pro), and cyclo(Glu-Pro) (P240510-M2, P240510-M3, P240511-M1, P240510-M4, all customized by Apeptide Co., Ltd., Shanghai, China).
Mouse microglial BV2 cells (ZQ0397, Zhongqiao Xinzhou Biotechnology Co., Ltd., Shanghai, China) and mouse hippocampal neuronal HT-22 cells (ChuanQiu Biotechnology Co., Ltd., Shanghai, China) were cultured in Dulbecco’s modified Eagle’s medium (01-057-1A, DMEM, Boehringer-Ingelheim, Haemek, Israel) supplemented with 10% (vol/vol) fetal bovine serum (HN-FBS-500, Hakata, ChuanQiu Biotechnology Co., Ltd., Shanghai, China) and 1% penicillin-streptomycin solution (10378016, Gibco, Grand Island, NY, USA) in a humidified atmosphere (5% CO2, 95% air, 37 °C). The cell lines used in this study have been tested for mycoplasma contamination, and the results were negative. Cell identity of BV2 and HT-22 cells was verified by Short Tandem Repeat (STR) analysis.
To first screen for bioactive compounds with anti-inflammatory and antioxidative properties, BV2 cells were treated with 0.1, 1, and 10 mM bioactive compounds in the presence of 1 µg/mL LPS (L2630; Sigma Aldrich, St. Louis, MO, USA) for 16 h. The concentration of bioactive compounds and duration were determined on the basis of previous studies on CAR and EC [28, 29]. To evaluate the synergistic effects of these dietary peptides with CAR, BV2 cells were treated with 1 µg/mL LPS and 0.03 or 0.1 mM potent bioactive compounds alone or in combination with 0.1 mM CAR for 16 h. To determine the synergistic effects of CAR and PA and cyclo(Gly-Pro) on neuroprotection, BV2 cells were cocultured with HT-22 cells, as described previously [30]. In brief, BV2 cells were first pretreated with 0.1 mM CAR, PA, cyclo(Gly-Pro), or a combination of CAR with either bioactive agent, or in the presence of LPS for 16 h. HT-22 cells were seeded in a 6-well plate in DMEM. Next, the LPS- and bioactive-treated BV2 cells were seeded in Transwell inserts (0.4 µm pore polyester membrane, CLS3450, Corning, Corning, NY, USA) and cultured above the HT-22 cells for 24 h.
BV2 cells were treated with 1 µg/mL LPS and 0.1, 1, and 10 mM peptides for
16 h. Total RNA was extracted from BV2 cells using Total RNA Extraction Reagent
(R401-01, Vazyme, Nanjing, Jiangsu, China), and cDNA was synthesized using an Evo M-MLV RT
Kit with gDNA Clean for qPCR (R333-01, Vazyme, Nanjing, Jiangsu, China). Subsequent
quantitative real-time PCR was performed with SYBR Green Real-time PCR Master Mix
(Q312-02, Vazyme, Nanjing, Jiangsu, China) on a CFX Connect Optics Module (788BR09833,
Bio-Rad, Hercules, CA, USA).
BV2 cells were treated with 10 mM bioactive compounds from EC or 0.1 mM CAR, 0.03 mM and 0.1 mM PA, and cyclo(Gly-Pro) and their combinations along with LPS for 16 h. After treatment with the bioactive peptides, the BV2 cell samples were homogenized with a grinder. The samples were then centrifuged, and protein concentrations were determined with an enhanced BCA protein assay kit (P0009, Beyotime, Shanghai, China). The activities of catalase (CAT) and superoxide dismutase (SOD) and the content of glutathione (GSH) in BV2 cells were measured by a corresponding kit (A001-1-1, A001-3-2, A0060201, Jiancheng Institute of Biotechnology Co., Ltd., Nanjing, Jiangsu, China).
After treatment with 0.1 mM CAR, PA, cyclo(Gly-Pro), and their combinations and
LPS for 16 h, BV2 cells and cocultured HT-22 cells were lysed in RIPA buffer
(20-188, Millipore, Billerica, MA, USA), and the protein concentrations were determined
using an enhanced BCA protein assay kit (P0009, Beyotime, Shanghai, China). The proteins
were separated by sodium dodecyl sulfate‒polyacrylamide gel electrophoresis and
transferred to a PVDF membrane (0301004001, Millipore, Billerica, MA, USA). The membranes
were blocked and then incubated at 4 °C overnight with primary
antibodies including anti-Ras (ET1601-16, 1:1000 dilution), anti-B-Raf
(ET1608-36, 1:1000 dilution), anti-p-B-Raf (ET1701-20, 1:2000 dilution),
anti-mitogen-activated protein kinase (MEK) (ET1603-20, 1:1000 dilution),
anti-p-MEK (ET1612-40, 1:1000 dilution), anti-AKT (ET1609-47, 1:5000 dilution),
anti-PI3K (ET1608-70, 1:3000 dilution), anti-Bax (ET1603-34, 1:20000 dilution),
anti-Bcl2 (ET1702-53, 1:10000 dilution), anti-caspase-3 (ET1608-64, 1:1000
dilution) from HUABIO (Hangzhou, Zhejiang, China), anti-ERK1/2 (#4695, 1:1000
dilution), anti-p-ERK1/2 (#4370, 1:2000 dilution), anti-p-AKT (#4060, 1:2000
dilution), anti-p-PI3K (#4228, 1:1000 dilution) from Cell Signaling Technology
(Danvers, MA, USA), and anti-
BV2 cells were first treated with 0.1 mM CAR, PA, cyclo(Gly-Pro), and their combinations, and LPS for 16 h. The HT-22 cells were then cocultured with pretreated BV2 cells for another 24 h, and a TUNEL assay and an Annexin V-FITC assay were performed to detect apoptotic neural cells with a TUNEL staining kit (C1086; Beyotime, Shanghai, China) and an Annexin V-FITC apoptosis detection kit (C1062; Beyotime, Shanghai, China) according to the manufacturer’s instructions. The fluorescence data were further analyzed and quantified by ImageJ. The flow cytometry data were analyzed using a FACSMelody (BD Bioscience, San Jose, CA, USA). The fluorescence data were further analyzed by FlowJo software (Tree Star, Ashland, OR, USA).
BV2 cells were treated with 1 µg/mL LPS and 0.1 mM CAR, PA,
cyclo(Gly-Pro), and their combination for 16 h. The supernatant of the medium was
collected. The expression of tumor necrosis factor (TNF-
All bar charts presented in this study were generated using Prism 8 software
(Version 8.0.2(263), GraphPad Software, La Jolla, CA, USA). All the data are
presented as the mean
The major peptides extracted from EC and their concentrations in EC are listed
in Table 1. To evaluate the anti-inflammatory and antioxidative effects of these
bioactive cyclic peptides, qPCR analysis was conducted in LPS-treated BV2 cells.
The results revealed that the upregulated mRNA expression of proinflammatory
genes and oxidative stress-related markers, including TNF-
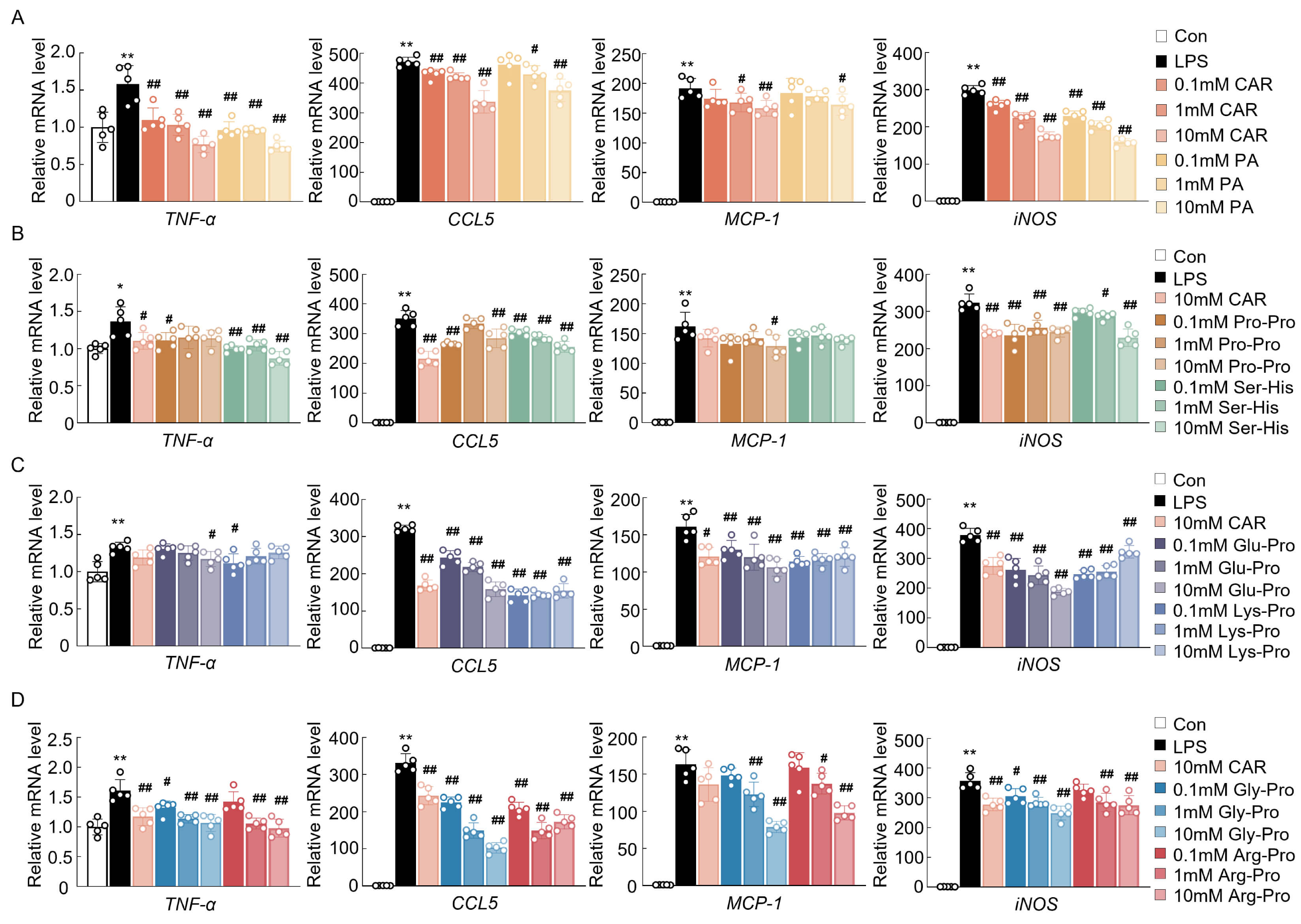 Fig. 1.
Fig. 1.
The effects of cyclopeptides on the mRNA expression of oxidative
stress and inflammatory markers in BV2 cells. (A–D) mRNA expression of
neuroinflammation- and oxidative stress-related genes (n = 5). Data are expressed
as means
| Compound name | Concentration in EC (mg/g DW) | % RSD | No. of replicates |
| Carnosine | 10.49 | 10.16 | 6 |
| Pyroglutamic acid | 2.83 | 3.52 | 6 |
| Cyclo(Glu-Pro) | 0.18 | 3.52 | 6 |
| Cyclo(Gly-Pro) | 1.51 | 5.73 | 6 |
| Cyclo(Arg-Pro) | 0.19 | 6.45 | 6 |
| Cyclo(Pro-Pro) | 0.6 | 6.10 | 6 |
| Cyclo(Ser-His) | N.A. | 6 | |
| Cyclo(Lys-Pro) | 0.21 | 2.62 | 6 |
DW, dry weight; RSD, relative standard deviation; N.A., Not Applicable; EC, essence of chicken.
To further confirm the effects of these bioactives and screen out potential bioactive compounds, their antioxidant enzyme activities were determined. Owing to the qPCR results showing no obvious changes in the relevant markers at 0.1 and 1 mM, the highest concentration of 10 mM was chosen for the antioxidant enzyme activity assay to identify potentially active cyclic peptides. In fact, the reduced activity of CAT was increased by CAR, PA, and cyclo(Gly-Pro), and the activity of SOD was increased by all of these bioactive compounds (Fig. 2A). In addition, the GSH content was also reduced after LPS treatment and was enhanced by CAR, PA, cyclo(Gly-Pro), and cyclo(Arg-Pro) (Fig. 2A). The overall antioxidative effects tended to be stronger in the CAR-, PA-, and cyclo(Gly-Pro)-treated groups. Therefore, on the basis of the integrated results of the qPCR and antioxidant enzyme activity assays, CAR, PA, and cyclo(Gly-Pro) were selected for the subsequent assessment of anti-inflammatory effects. Western blot analysis revealed that LPS treatment markedly increased the levels of Ras, phosphorylated B-Raf, MEK, and ERK1/2, which were attenuated by treatment with 10 mM CAR, PA, and cyclo(Gly-Pro) (Fig. 2B). Therefore, CAR, PA, and cyclo(Gly-Pro) exerted excellent antioxidative and anti-inflammatory effects and were used in subsequent experiments to investigate the synergistic effects. Since these bioactive compounds exerted significant effects at concentrations of 0.1 mM and 0.1 mM, a lower concentration was used in the synergistic effect experiments.
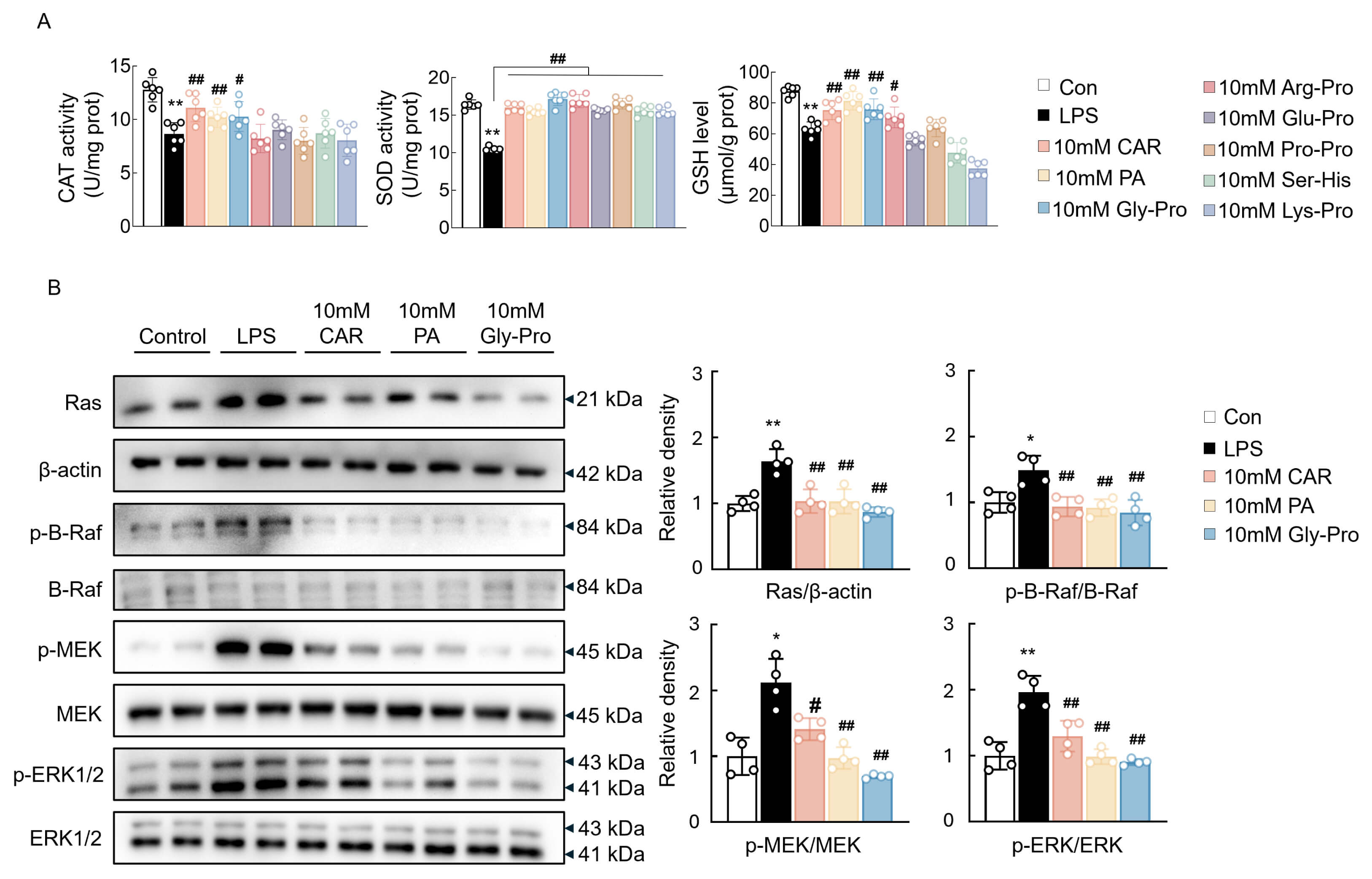 Fig. 2.
Fig. 2.
The effects of EC bioactives on antioxidant enzyme activity and
inflammatory signaling pathway in BV2 cells. (A) The activity of CAT and SOD and
the content of GSH in BV2 cells (n = 6). (B) Western blot analysis and
quantification data of Ras, p-B-Raf, p-MEK, p-ERK1/2, and their total proteins in
BV2 cells (n = 4). Data are expressed as means
Next, the synergistic effect of CAR with PA or cyclo(Gly-Pro) was determined.
qPCR analysis of inflammatory genes and oxidative stress markers revealed that
combined treatment with CAR and PA significantly reduced the expression of these
genes, with a more potent effect than 0.1 mM CAR or PA alone (Fig. 3A). The
synergistic effect of CAR and cyclo(Gly-Pro) was also stronger than that of CAR
or cyclo(Gly-Pro) alone (Fig. 3B). However, these synergistic effects occurred
only at a concentration of 0.1 mM (Fig. 3A,B). Similarly, CAR, PA, and
cyclo(Gly-Pro) increased the expression of M2 microglial markers, including
arginase 1 (Arg1) and cluster of differentiation 206 (CD206), in LPS-treated BV2
cells, and the synergistic effects were the same (Fig. 3C,D). The results of the
activities of antioxidant enzymes, such as CAT, SOD, and GSH, further verified
the synergistic effects of CAR with PA or cyclo(Gly-Pro), with a more prominent
effect in the CAR + PA and CAR + cyclo(Gly-Pro) groups than in the CAR, PA, or
cyclo(Gly-Pro) alone groups (Fig. 3E,F). Moreover, ELISA results revealed that
the elevated levels of inflammatory cytokines and chemokines in the supernatant
caused by LPS—including TNF-
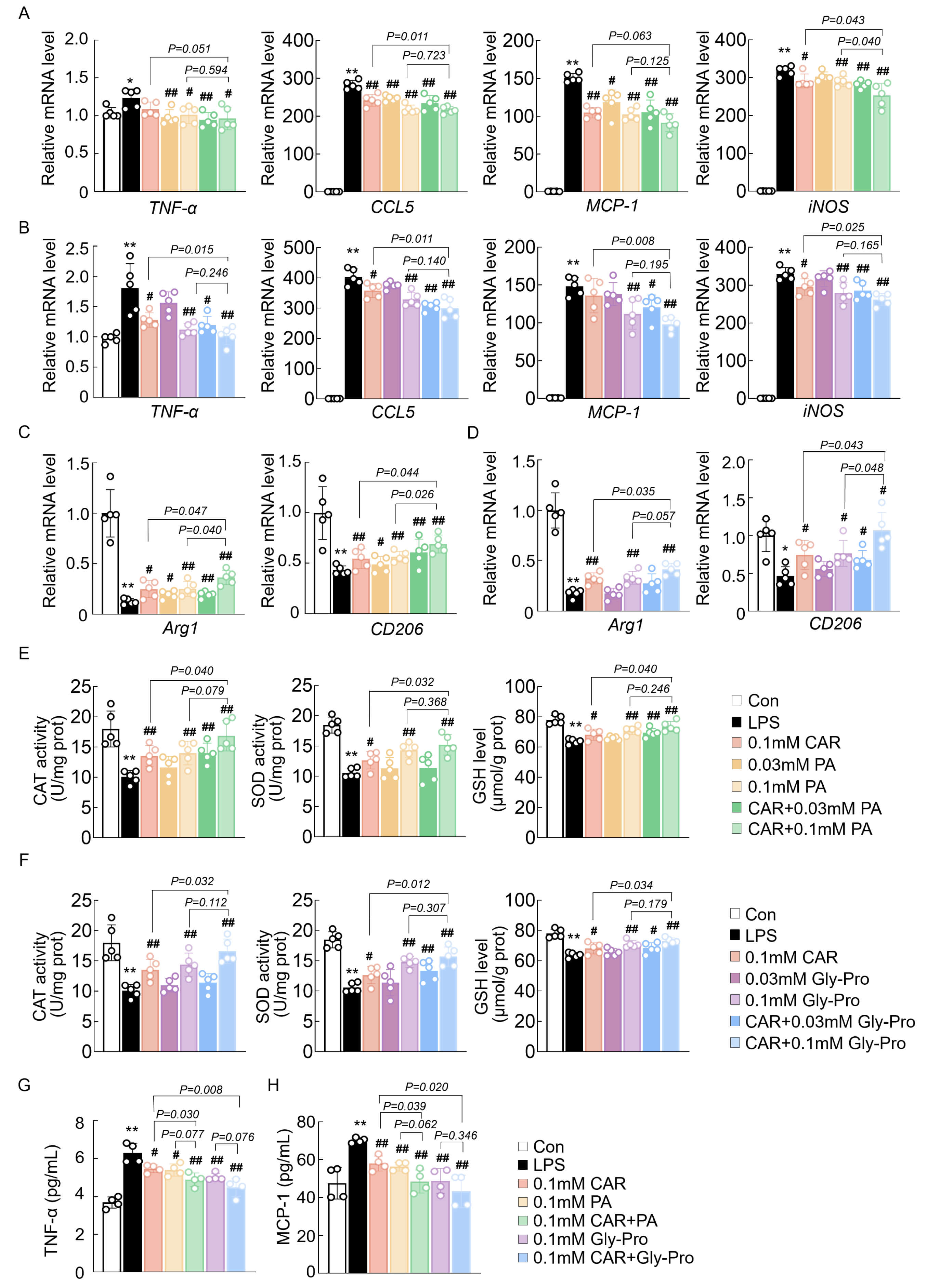 Fig. 3.
Fig. 3.
The synergistic effect of CAR with PA and cyclo(Gly-Pro) on
inflammation and oxidative stress in BV2 cells. (A,B) mRNA expression of
neuroinflammation-related and oxidative stress-related genes in the BV2 cells (n
= 5). (C,D) mRNA expression of M2-polarized microglia markers genes in the BV2
cells (n = 5). (E,F) The activity of CAT and SOD and the content of GSH in BV2
cells (n = 5). (G,H) ELISA analysis of the inflammatory cytokine levels in the
medium (n = 4). Data are expressed as means
Western blot analysis of the inflammatory pathway was performed to clarify involvement in the synergistic effect of CAR with PA and cyclo(Gly-Pro). Consistent with the previous findings, CAR, PA, and cyclo(Gly-Pro) significantly attenuated the activation of the Ras/Raf cascade and its downstream targets, such as MEK and ERK1/2, which was further notably reduced when CAR was combined with PA or cyclo(Gly-Pro) (Fig. 4A–D). These results suggested that the ability of the combination of CAR and PA or cyclo(Gly-Pro) to alleviate inflammation might be partly related to the Ras/MAPK pathway.
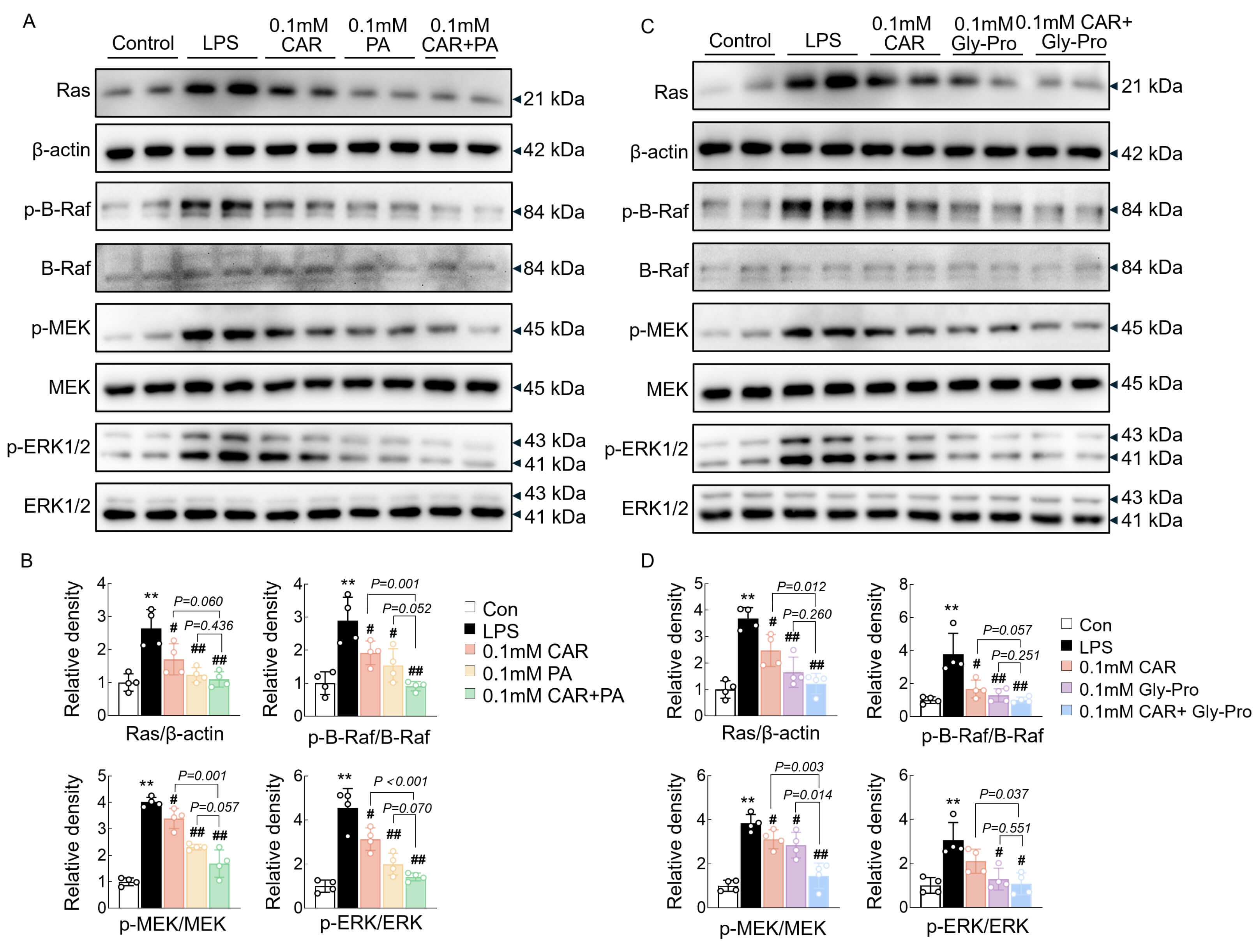 Fig. 4.
Fig. 4.
The synergistic effect of CAR with PA and cyclo(Gly-Pro) on
Ras/MAPK pathways. (A,B) Western blot analysis and quantification of Ras,
Since CAR, PA, and cyclo(Gly-Pro) exerted anti-inflammatory and antioxidative stress effects with marked synergistic effects on BV2 cells, a Transwell coculture system was used to investigate the neuroprotective effects of these three bioactive compounds. BV2 cells were pretreated with CAR, PA, cyclo(Gly-Pro), and LPS for 16 h and then seeded in Transwell inserts and cocultured with HT-22 cells for another 24 h (Fig. 5A). TUNEL assays were conducted to evaluate the apoptosis of HT-22 cells, and the results indicated that the number of TUNEL-positive cells significantly increased in the group of HT-22 cells cocultured with LPS-treated BV2 cells compared with that in the control group (Fig. 5B,C). Treatment with CAR, PA, or cyclo(Gly-Pro) resulted in a significant reduction in the number of apoptotic neurons, and the combination of CAR with PA or cyclo(Gly-Pro) showed more prominent antiapoptotic effects (Fig. 5B,C). Flow cytometry analysis of Annexin V-FITC-stained cells also revealed that treatment with CAR, PA, or cyclo(Gly-Pro) alone inhibited LPS-induced cell death, and the combination treatments showed stronger effects (Fig. 5D,E).
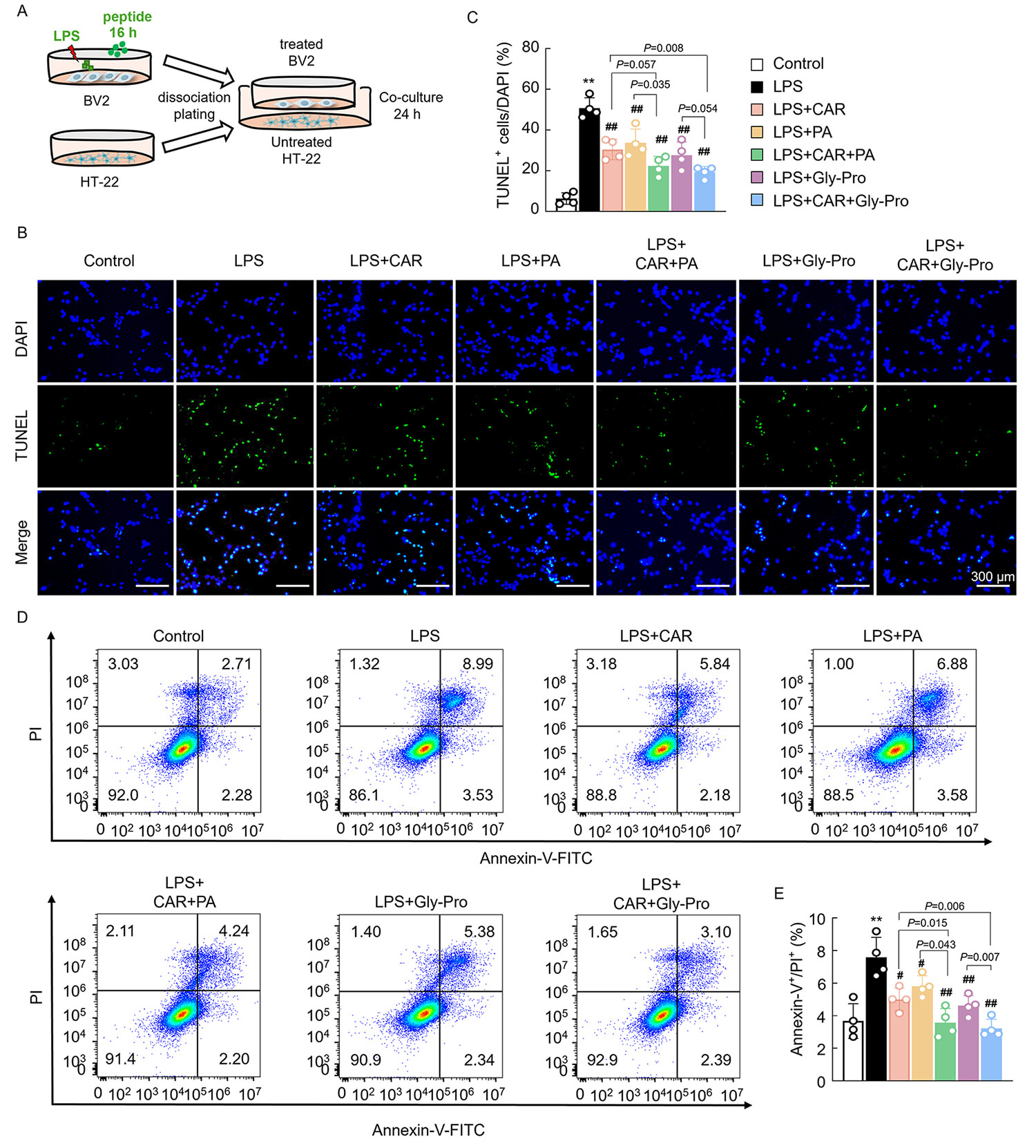 Fig. 5.
Fig. 5.
The effects of EC bioactives on BV2 cell-induced cytotoxicity to
HT-22 cells. (A) Experimental design of the coculture systems. (B,C) TUNEL
staining of HT-22 cells and the quantification data, scale bars = 300 µm (n
= 4). (D,E) Flow cytometry analysis of Annexin-V+/PI+ HT-22 cells and the
quantification data (n = 4). Data are expressed as means
Next, the involvement of the PI3K/AKT pathway in the protective effects of CAR, PA, and cyclo(Gly-Pro) against apoptosis was further investigated. CAR, PA, and cyclo(Gly-Pro) treatment notably increased the phosphorylation of PI3K and AKT in HT-22 cells cultured with BV2 cells (Fig. 6A,B). These results were associated with a reduction in the expression of the proapoptotic Bcl family proteins Bax and cleaved-Caspase3 and an increase in the expression of the antiapoptotic protein Bcl2 (Fig. 6A,B). Consistent with the previous results, CAR combined with PA or cyclo(Gly-Pro) had a synergistic effect on apoptosis and the PI3K/AKT pathway (Fig. 6A,B). Therefore, CAR, PA, cyclo(Gly-Pro), and their combination may attenuate LPS-induced BV2 cytotoxicity by partly regulating the PI3K/AKT pathway in HT-22 cells.
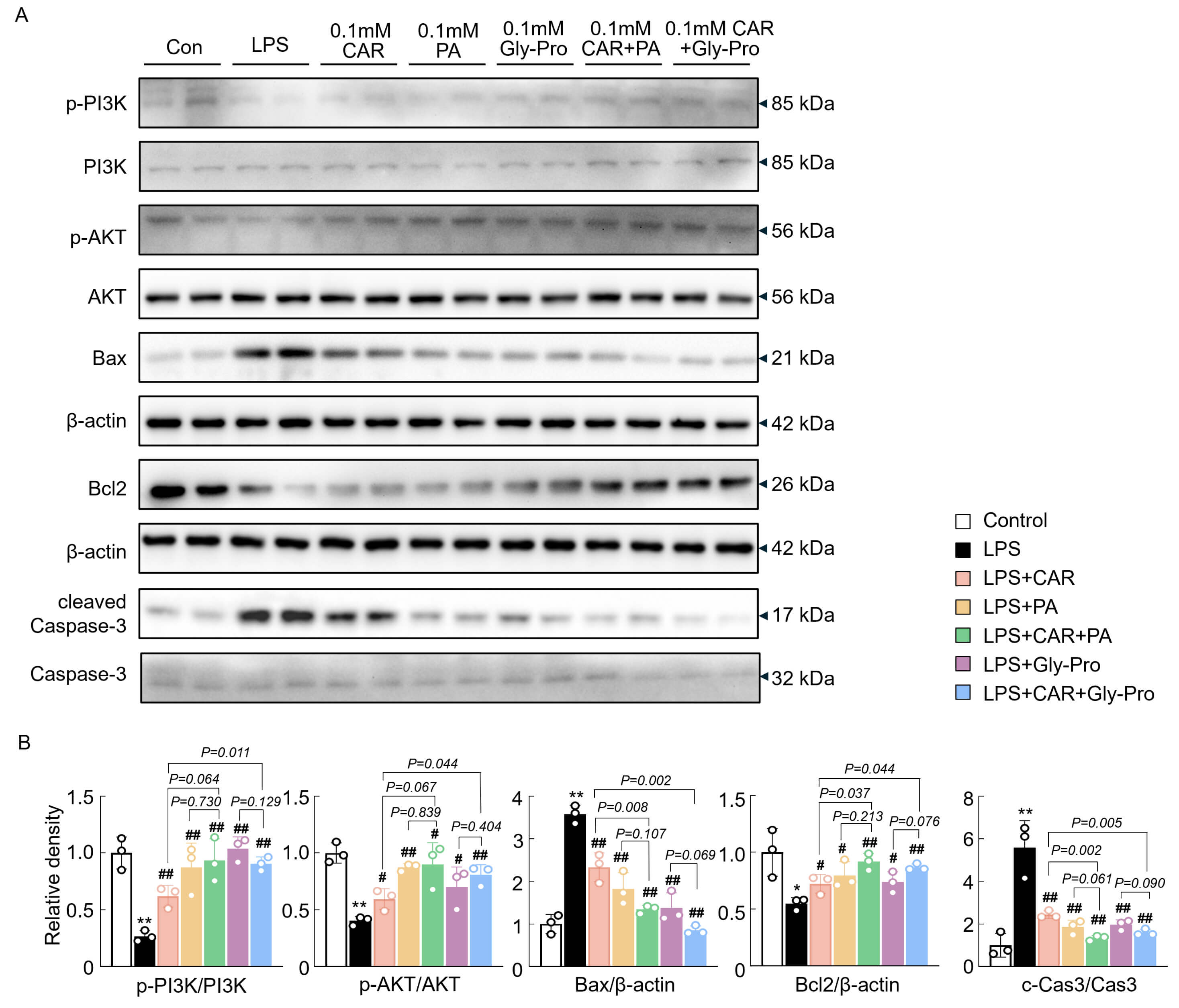 Fig. 6.
Fig. 6.
The synergistic effect of CAR with PA and cyclo(Gly-Pro) on
apoptosis and the PI3K/AKT pathways. (A,B) Western blot analysis and
quantification data of p-PI3K, PI3K, p-AKT, AKT, Bax, Bcl2,
Studies on the bioactivity of cyclopeptides and noncanonical amino acids have become popular because of their high permeability, high specificity, and high stability [32, 33]. Some food-derived peptides have demonstrated various bioactivities. For example, soy and dairy dietary peptides improved oxidative stress [34] and immunomodulation [35] and enhanced intestinal barrier integrity [36]. Glutamate, a vital neurotransmitter in the central nervous system, promotes signal transmission between neurons and is crucial for synaptic plasticity [37]. As an intramolecular cyclization product of glutamate, PA may exert similar protective effects [38]. In our previous study, we revealed the potent bioactivities of hydrolyzed chicken meat extract and its bioactive cyclopeptides, such as cyclo(Phe-Phe) [29] and cyclo(Val-Pro) [39]. On the basis of clinical evidence, EC supplementation resulted in quicker decision times, quicker reaction times, better memory [13], and improved mood conditions. Here, we also revealed that all bioactive compounds from EC have certain neuroprotective effects. CAR, PA, and cyclo(Gly-Pro) were selected as potential bioactive compounds for further study because of their notable anti-neuroinflammatory and antioxidative stress effects. We speculate that CAR, PA, and cyclo(Gly-Pro) might maintain the healthy function of cognition and mood by protecting microglia and neurons from inflammation and apoptosis.
Oxidative stress and neuroinflammation affect all classes of macromolecules
within cells, such as sugars, lipids, proteins, and DNA, inevitably leading to
dysfunction of the nervous system [40]. However, oxidative stress is often a
causative factor of neuroinflammation and frequently occurs simultaneously [41].
CAR prevents oxidative stress and inflammation in BV2 cells through the activity
of the transforming growth factor-
The Ras/MAPK pathway plays a vital regulatory role in the inflammatory process.
In neuroinflammatory cells, surface receptors bind to extracellular signaling
molecules and then become activated and interact with an adaptor, leading to the
activation of Ras [47]. Ras recruits and activates Raf kinases, which then
phosphorylate and activate MEK, ultimately activating MAPK and promoting the
transcription and production of inflammation-related cytokines [47, 48]. In a
previous study, CAR was reported to protect neurons against oxidative stress and
modulate MAPK cascade signaling [49]. Moreover, CAR was reported to inhibit the
activation of
In the nervous system, injured microglia are activated and accumulate, releasing
inflammatory mediators that exacerbate neuronal damage and apoptosis [54]. LPS
treatment induces the secretion of large amounts of proinflammatory cytokines and
chemokines, such as IL-1
Many food-derived peptides have been reported to exhibit similar neuroprotective
activities. Black soybean peptide affects AMP-activated protein kinase
(AMPK)/sirtuin 1 (SIRT1)/NF-
On the other hand, the neuroprotective effects of CAR, PA, and cyclo(Gly-Pro) in the coculture system may extend beyond cytotoxicity, potentially influencing neuronal function through the dynamic phenotypic switching of BV2 cells [62]. When BV2 cells transform from the classically activated M1 type to the alternatively activated M2 type, they secrete anti-inflammatory factors [63], such as brain-derived growth factor (BDNF) and insulin-like growth factor-1 (IGF-1), to regulate the growth status of neuronal cells [64]. Previous studies revealed that CAR might alter the M1/M2 macrophage ratio to decrease oxidative stress-associated cell damage [65]. Hydrolyzed chicken meat extract significantly decreased M1-polarized microglia activation while enhancing M2-polarized microglia, leading to anti-inflammatory and antioxidative stress effects, and its bioactive cyclo(Phe-Phe) exhibited excellent bioactivity in this response [39]. In fact, our results revealed that CAR, PA, and cyclo(Gly-Pro) decreased M1-polarized BV2 cells and increased M2-polarized BV2 cells. Therefore, CAR, PA, and cyclo(Gly-Pro) might reduce the apoptosis of HT-22 cells by regulating M1/M2 microglial polarization, thereby protecting neural function. Coupled with past clinical findings, CAR, PA, and cyclo(Gly-Pro) might be potential anti-neuroinflammatory agents contributing to the health benefits of EC in preserving cognitive function, and further studies are needed to verify this hypothesis.
In this study, we systematically compared the anti-inflammatory and antioxidant activities of 8 dietary peptides in EC, screened potential dietary peptides, and revealed their synergistic effects and underlying mechanisms (Fig. 7). This is the first study to show the synergism between CAR and small peptides. Although there had been mixed preclinical findings on effects and mechanisms of PA for cognitive function, in this study, we showed the beneficial effects of PA in neuroprotection. We are also the first to show that PA may partly modulates neuroinflammation and apoptosis via the ERK1/2 and PI3K/Akt pathways (Fig. 7). It may be worth investigating further whether CAR or other components in EC modulate the action of PA to give the synergistic effect. Furthermore, coculture experiments confirmed that CAR, PA, and cyclo(Gly-Pro) protect the growth of HT-22 cells by modulating BV2 cells, exerting an improved effect on the nervous system dysfunction.
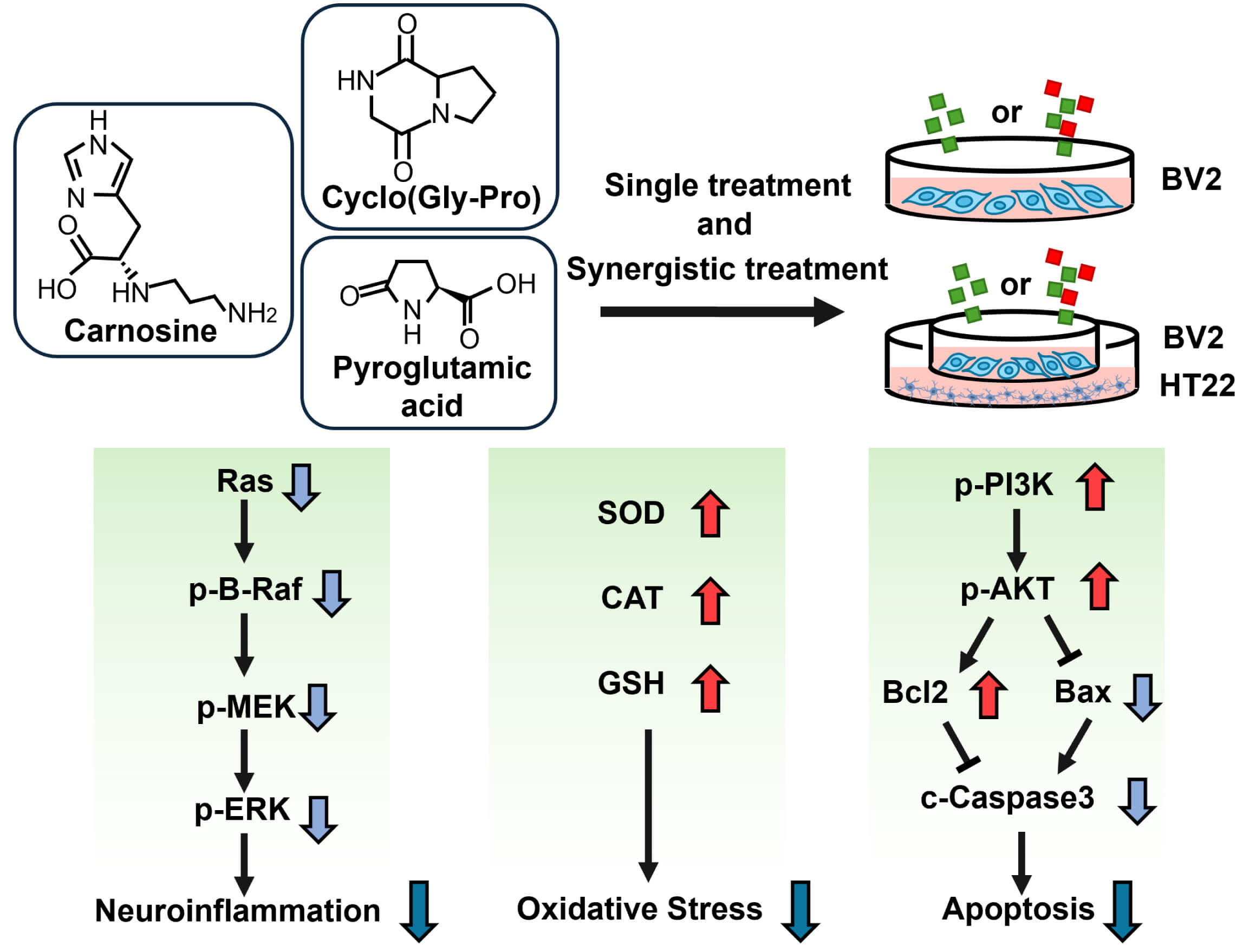 Fig. 7.
Fig. 7.
The diagram of the mechanisms of CAR, PA, and cyclo(Gly-Pro) on neuroinflammation. Among the eight bioactives of the essence of chicken, carnosine (CAR), pyroglutamic acid (PA), and cyclo(Gly-Pro) exerted a notable anti-inflammatory and antioxidative effect in BV2 cells, as evidenced by the attenuation of the inflammatory pathway and the increase of antioxidant enzyme activities. PA and cyclo(Gly-Pro) showed a marked synergistic effect with CAR on neuroinflammation. The coculture of BV2 microglia with HT-22 neurons revealed that CAR, PA, and cyclo(Gly-Pro) also worked synergistically to reduce LPS-induced cytotoxicity in BV2 cells to protect the growth of HT-22 cells. The protective effect of these dietary peptides might be regulated by the PI3K/AKT pathway-mediated inhibition of apoptosis.
Despite the valuable finding, the study had some limitations. Although in vitro inflammatory models and coculture experiments provide mechanistic insights into neurotoxicity, they do not fully recapitulate the systemic inflammatory responses or blood–brain barrier dynamics observed in vivo. More in vivo experiments should be conducted to verify whether PA and cyclo(Gly-Pro) and their synergistic effects improve cognitive ability and alleviate neuron dysfunction, and experiments in mice would provide more compelling evidence. Importantly, inhibitor-related studies may provide key data needed to elucidate the underlying molecular mechanisms of dietary peptides. Molecular docking or competitive binding assays of effective peptides and target proteins may also present protein interactions in the future. In future studies, we plan to address these limitations by performing inhibitor-related studies and molecular docking to investigate underlying mechanisms, employing LPS-induced inflammation mouse models to verify the regulatory effects of CAR, PA, and cyclo(Gly-Pro).
In summary, CAR, PA, and cyclo(Gly-Pro) may strongly affect neuroinflammation through inhibiting the Ras/MAPK pathway and protecting HT-22 cells against inflammatory BV2 cell-induced cytotoxicity by regulating the PI3K/AKT pathway. Moreover, CAR combined with PA or cyclo(Gly-Pro) showed marked synergistic effects on neuroinflammation and oxidative stress. Our research revealed that the intake of an essential mixture rather than individual dietary peptides yields more beneficial effects for maintaining healthy neuron function and may contribute to the maintenance of brain health by counteracting neuroinflammation. This warrants further in vivo investigation.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
XH contributed to the investigation, writing the original draft, and data curation. YZ and HJ contributed to the investigation and the validation. CCY, SMY, GKYC, and ZJL contributed to formal analysis, work designing, supervision, writing review, and draft editing. ZF contributed to the designing the research study, analyzing the data, writing review, and draft editing. YN contributed to conceptualization, data curation, funding acquisition, writing review, and draft editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Thank all the researchers participating in this study. We also appreciate the assistance and financial support provided by the Suntory Beverage and Food Asia Pte. Ltd.
This study was supported by the funds from Suntory Beverage and Food Asia Pte. Ltd (KYY-HX-20180429) and Shanghai Polaris Health Co., Ltd (KYY-HX-20210238).
Chin Chin Yau, Shan May Yong, Grace Kar Yarn Chan, and Zhen Jie Low are employees of Suntory Beverage and Food Asia Pte. Ltd. The authors declare no conflict of interest.
All the raw, uncroped, and unedited images for Western blot bands in Fig. 2B, Fig. 4A,B and Fig. 6A can be found in the Supplementary Material in the online version, at https://doi.org/10.31083/IJVNR45261.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







