1 Noncommunicable Diseases Research Center, Neyshabur University of Medical Sciences, Neyshabur, Iran
2 Healthy Ageing Research Centre, Neyshabur University of Medical Sciences, Neyshabur, Iran
3 School of Health, Arak University of Medical Sciences, Arak, Iran
4 Ageing Clinical and Experimental Research Team, Institute of Applied Health Sciences, School of Medicine, Medical Sciences and Nutrition, University of Aberdeen, AB24 3FX Aberdeen, UK
5 Department of Epidemiology, School of Public Health, Iran University of Medical Sciences, Tehran, Iran
6 Dave C. Swalm School of Chemical Engineering, Mississippi State University, Mississippi State, MS 39762, USA
7 Department of Epidemiology and Biostatistics, School of Public Health, Neyshabur University of Medical Sciences, Neyshabur, Iran
Abstract
Emerging evidence suggests that Spirulina may reduce inflammation by modulating key cytokines, including C-reactive protein (CRP), tumor necrosis factor-α (TNF-α), and interleukin-6 (IL-6). Accordingly, this study conducted a systematic review and meta-analysis to evaluate the effects of Spirulina supplementation on serum levels of inflammatory mediators in adults aged 18 years and older.
Relevant randomized clinical trials (RCTs) were identified through searches of several databases, including the Cochrane Library, ClinicalTrials.gov, ISI Web of Science, Scopus, and PubMed up to August 2025. The pooled effects were calculated using the DerSimonian and Laird random-effects model. Statistical heterogeneity was assessed using I-squared statistics and Cochran’s Q test.
Eight studies met the inclusion criteria. The administered dosages of Spirulina varied from 1 g/day to 8 g/day, with intervention durations spanning 3 to 16 weeks. The pooled analysis demonstrated that Spirulina supplementation significantly reduced serum CRP concentrations compared with placebo group (weighted mean difference (WMD): –0.09 mg/L; 95% confidence interval (CI): –0.16 to –0.02). In contrast, Spirulina supplementation caused non-significant reduction in TNF-α concentration (WMD: –0.43 pg/mL; 95% CI: –1.44 to 0.59) and IL-6 (WMD: –0.44 pg/mL; 95% CI: –0.98 to 0.1). However, heterogeneity concerning all inflammatory mediators was significant.
The findings of this study indicated that Spirulina supplementation significantly reduced serum CRP levels, whereas its effects on IL-6 and TNF-α were not statistically significant. Owing to the significant heterogeneity, further high-quality RCTs are needed to confirm these beneficial effects.
The study was registered in PROSPERO (CRD42024606496).
Keywords
- Spirulina
- meta-analysis
- systematic review
- CRP
- TNF-α
- IL-6
Spirulina, a blue-green microalgae, naturally thrives in alkaline lakes. Its composition varies according to the source of algae, cultivation conditions, and season. It contains proteins (55–70%), carbohydrates (15–25%), fats (6–8%), minerals (7–13%), moisture (3–7%), and dietary fibers (8–10%). Spirulina is also rich in polyunsaturated fatty acids (PUFAs), vitamins (B1, B2, B3, B6, B9, B12, C, D, E), and minerals (K, Ca, Cr, Cu, Fe, Mg, Mn, P, Se, Na, Zn). In addition, it contains pigments and enzymes, including lipase, which further enhance its nutritional profile and health-promoting potential [1].
Spirulina refers to the commercial form of dried biomass obtained from green microalgae of the genus Arthrospira (family Microcoleaceae, order Oscillatoriales, class Cyanophyceae, phylum Cyanophyta) [2, 3]. In 1989, Castenholz distinguished the genera Spirulina and Arthrospira by defining their specific morphological characteristics. Although the term Spirulina is widely used for commercial products and in scientific literature, current taxonomic classifications recognize Arthrospira and Spirulina as closely related but distinct genera [4, 5].
The main Spirulina species utilized in nutrition and scientific research are Arthrospira platensis, Arthrospira maxima, and Arthrospira fusiformis [6]. Arthrospira platensis is among the most commonly cultivated microalgal species. Although no toxic effects have been documented, it is generally recommended that Spirulina intake as a dietary supplement should not exceed 15 grams per day [7]. Spirulina has been associated with a range of health benefits, including antioxidant properties, immune system modulation, and anti-inflammatory, anti-cancer, antiviral, and antibacterial properties [8]. Its bioactive compounds may play an important role in preventing or managing conditions such as obesity, hyperglycemia, hypercholesterolemia, cardiovascular diseases (CVDs), hypertension, neoplasms, and inflammatory disorders [9]. Spirulina plays a critical role in enhancing immune function through various biological mechanisms [10].
Spirulina’s immunological and antioxidant benefits are largely attributed to its mineral content and carbohydrate-rich composition, including unique polysaccharides such as “Immulina” [11]. Moreover, Spirulina produces phycobiliproteins with potential applications as natural colorants and with demonstrated promise in clinical and immunological research [12].
Evidence suggests that Spirulina modulates immune responses and reduces
inflammation by influencing key cytokines such as C-reactive protein (CRP), tumor
necrosis factor-
Anti-inflammatory properties of Spirulina has been investigated in randomized
clinical trials (RCTs), although results remain inconsistent [17, 18, 19, 20, 21, 22, 23, 24]. A
meta-analysis published in 2021 assessed the effects of Spirulina supplementation
on serum levels of IL-6 and TNF-
This systematic review followed the Preferred Reporting Items for Systematic
Review and Meta-analysis (PRISMA) guidelines [30] and was registered in PROSPERO
(protocol ID CRD42024606496). A comprehensive literature search was conducted in
PubMed, Scopus, Web of Science, Cochrane Library, ClinicalTrials.gov up to August
2025 to identify English-language RCTs evaluating the effectiveness of Spirulina
intake on serum concentration of inflammatory mediators. The search strategy
combined Medical Subject Headings (MeSH) and non-MeSH terms, including
“C-Reactive Protein”, “Protein-C Reactive”, “CRP”, “Tumor Necrosis
Factor-alpha”, “Tumor Necrosis Factor”, “Tumor Necrosis Factor
The Population, Intervention, Control, Outcomes (PICO) framework for this
systematic review and meta-analysis was defined as follows: Population/Patients
(P:
RCTs were excluded if they met any of the following criteria: (A) Use of other
nutrients in combination with Spirulina; (B) Absence of a comparison group; (C)
Lack of before or after intervention data on serum levels of mentioned
inflammatory data for both study groups or missing any essential data for effect
size calculations; (D) Reporting serum levels of CRP, TNF-
Three reviewers (MH, AGh, and MHe) independently performed screening, validation, selection, and methodological quality evaluation. We had a group discussion to resolve all discrepancies.
Data extraction was performed independently by two investigators (MH and FA),
who collected the following information: first author’s last name, year of
publication, the country where the research was conducted, sample size, trial
design (parallel or crossover, with relevant details), participants’ age (range
and/or mean), Spirulina supplementation dosage and intervention duration, type
and dosage of placebo, participants’ health status, and changes in the mean serum
concentration of mentioned inflammatory mediators and corresponding standard
deviations (SDs). Discrepancies during data extraction were resolved through
discussions with a third reviewer until consensus was achieved. Additionally, all
measurements of CRP, TNF-
Two reviewers (MH and AGh) independently assessed the overall methodological quality of the included studies using the Cochrane risk-of-bias tool (Rob2) [31]. The assessment considered several factors, including deviations from intended interventions, bias due to period and carryover effect (only for cross-over trials), randomization process, outcome measurement, handling of missing data, and selection of reported results. Based on these evaluations, each RCT was classified as having low risk, some concerns, or high risk of bias.
This meta-analysis was conducted by calculating the mean differences (MDs) and
their SDs for inflammatory mediators using data extracted from the included
studies. Effect sizes were determined based on the mean change in serum
concentrations of mentioned inflammatory mediators from baseline, along with
corresponding SDs for both intervention and comparison groups, following the
Cochrane Handbook guidelines [32]. Furthermore, when studies reported medians or
ranges instead of means, estimates of the mean of CRP, TNF-
Sensitivity analyses were performed to evaluate the influence of individual
studies on overall estimates, while publication bias was assessed using Begg’s
rank correlation test and visual inspection of Begg’s funnel plot [36]. All
statistical analyses were conducted using STATA version 15 software (StataCorp,
College Station, TX, USA). Statistical significance was set at p
The initial database search identified 713 articles. After removing duplicates, 483 articles remained for title and abstract screening. Of these, 462 were excluded, leaving 21 articles for full-text evaluation. Following detailed assessment, 13 more studies were excluded for the following reasons: participants younger than 18 years (n = 3), measurement of inflammatory markers other than mentioned inflammatory mediators (n = 6), inclusion of participants with acute inflammatory diseases (COVID-19) (n = 1), lack of pre- or post-intervention data on serum levels of IL-6 (n = 1), and duplicate populations reported in multiple publications (n = 2) (Fig. 1). Ultimately, eight RCTs with 337 participants met the eligibility criteria and were included in the systematic review and meta-analysis (Table 1, Ref. [17, 18, 19, 20, 21, 22, 23, 24]). In one study, the intervention results were reported separately for male and female participants [21], and as such, they were treated as two independent datasets in this analysis.
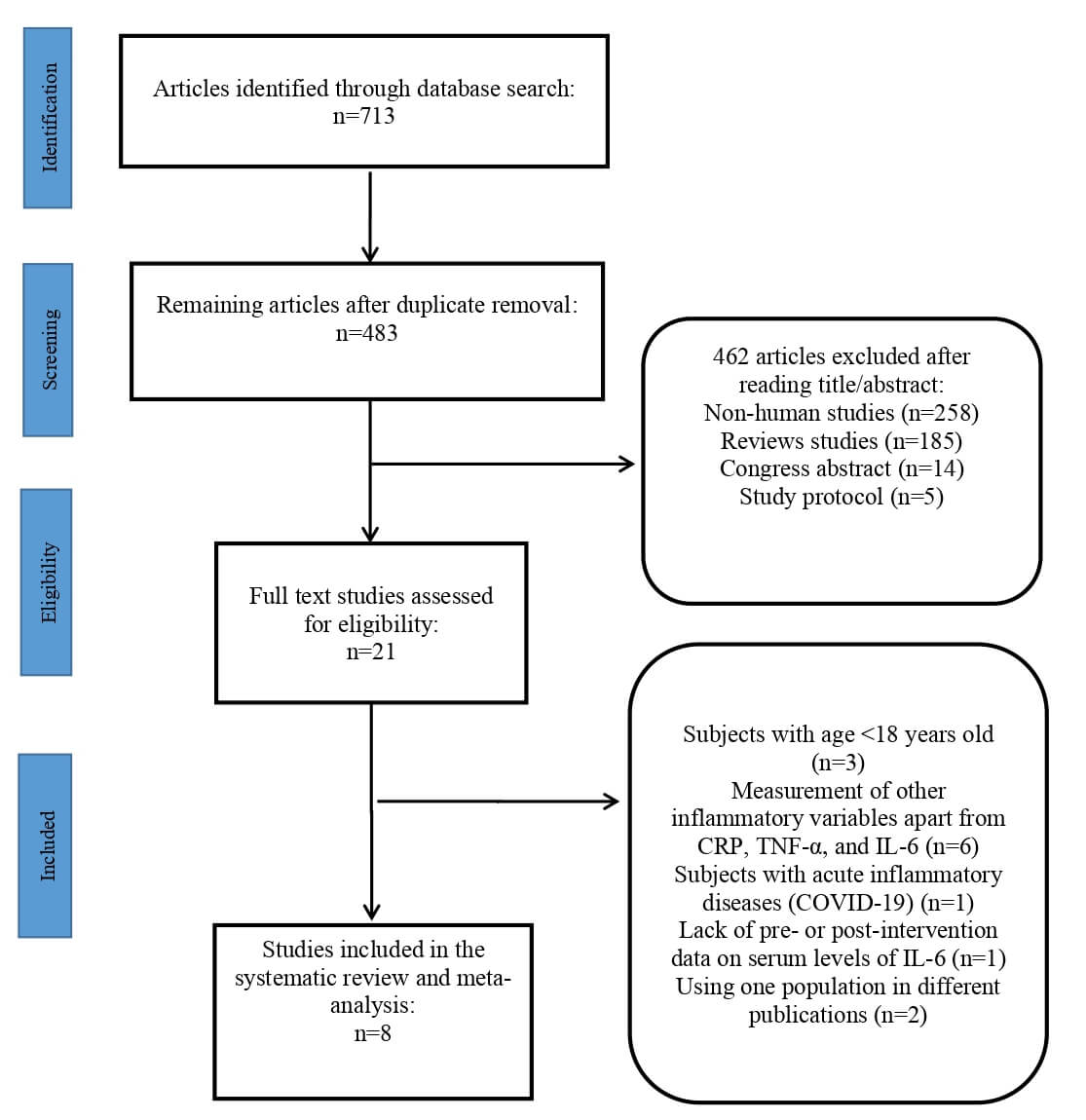 Fig. 1.
Fig. 1.
Flowchart of study selection process.
| Code | Study population (sample size) | Age (mean |
Study design | Intervention duration (week) | Spirulina dose | Placebo | Outcome | Results |
| author (year) (country) | ||||||||
1 Ghaem Far (2021) (Iran) [24] |
Hypertensive patients (41) | Spirulina group: 51.27 Placebo group: 50.21 M: 19, F: 22 |
Triple-blind randomized clinical trial | 8 | Salad dressing with 2 g Spirulina (Arthrospira platensis) | Salad dressing without spirulina | CRP | CRP did not change significantly |
2 Kashani (2022) (Iran) [23] |
Taekwondo Athletes (18) | Spirulina group: 26.6 Placebo group: 26.7 M: 18, F: 0 |
Double-blind, placebo-controlled crossover trial | 3 | 8 g (Spirulina platensis) | Starch | IL-6 | Spirulina supplementation resulted in a significant reduction in IL-6 level |
3 Lee (2008) (Korea) [22] |
type 2 diabetic patients (37) | Spirulina group: 52.1 Control group: 54.5 M: 20, F: 17 |
Randomized clinical trial | 12 | 8 g (Arthrospira platensis) | No placebo | TNF- |
TNF- |
4.1 Park (2008) (Korea) [21] |
Elderly male people (43) | Spirulina group: 66.18 Placebo group: 66.68 |
Double-blind, placebo-controlled trial | 16 | 8 g | Starch | TNF- |
Spirulina supplementation resulted in a significant reduction in IL-6, without any changes in TNF- |
4.2 Park (2008) (Korea) [21] |
Elderly female people (35) | Spirulina group: 65.68 Placebo group: 65.48 |
Double-blind, placebo-controlled trial | 16 | 8 g | Starch | TNF- |
TNF- |
5 Supriya (2023) (Iran) [20] |
Men with Obesity (22) | Spirulina group: N/M Placebo group: N/M | Randomized controlled trial | 12 | 6 g | Corn starch | CRP, TNF- |
Spirulina supplementation resulted in a significant reduction in CRP, IL-6, and TNF- |
6 Szulinska (2017) (Poland) [19] |
Patients with obesity and well-treated hypertension (50) | Spirulina group: 49.3 Placebo group: 50.2 M: 25, F: 25 |
Double-blind, placebo-controlled trial | 12 | 2 g (Spirulina maxima) | Microcrystalline cellulose | IL-6 | Spirulina supplementation resulted in a significant reduction in IL-6 level |
7 Tamtaji (2023) (Iran) [18] |
Patients with Alzheimer’s disease (53) | Spirulina group: 73.8 Placebo group: 76.9 M: 5, F: 48 |
Double-blind, placebo-controlled trial | 12 | 1 g (Arthrospira platensis) | Xylooligosaccharide, isomaltulose, hydroxypropyl methyl cellulose, polyethylene glycol, and carboxymethyl starch | CRP | Spirulina supplementation resulted in a significant reduction in CRP level |
8 Yousefi (2018) (Iran) [17] |
Healthy individuals with obesity or overweight (38) | Spirulina group: 40.16 Placebo group: 39.79 M: 7, F: 31 |
Randomized controlled trial | 12 | 2 g (Spirulina Platensis) | starch and lactose monohydrate | CRP | Spirulina supplementation resulted in a significant reduction in CRP level |
*: Mean
SD, Standard deviation; CRP, C-reactive protein; IL-6, Interleukin-6;
TNF-
Based on the findings of this systematic review, the effects of Spirulina on
serum levels of CRP, TNF-
Regarding trial design, one study was a triple-blind randomized clinical trial [24], one was a double-blind crossover randomized clinical trial [23], three were double-blind parallel randomized clinical trials [18, 19, 21], and the remaining studies were randomized controlled trials without blinding details [17, 20, 22]. Participants health status varied across studies and included the following subgroups: hypertensive patients [24], taekwondo athletes [23], type 2 diabetic patients [22], elderly individuals [21], men with obesity [20], patients with obesity and well-controlled hypertension [19], patients with Alzheimer’s disease [18], and healthy individuals with obesity or overweight [17]. More details are presented in Table 1.
Fig. 2 presents the quality assessment of each study. Among the eight included trials, three studies were rated as having some concerns [17, 23, 24], primarily due to selective outcome reporting. The remaining five studies were classified as high risk of bias [18, 19, 20, 21, 22], attributed to issues in the randomization process [19, 20, 21, 22], deviations from planned interventions [18, 19, 20, 21], issues with outcome measurement [19, 20, 21, 22], and selective reporting [18, 19, 20, 21, 22].
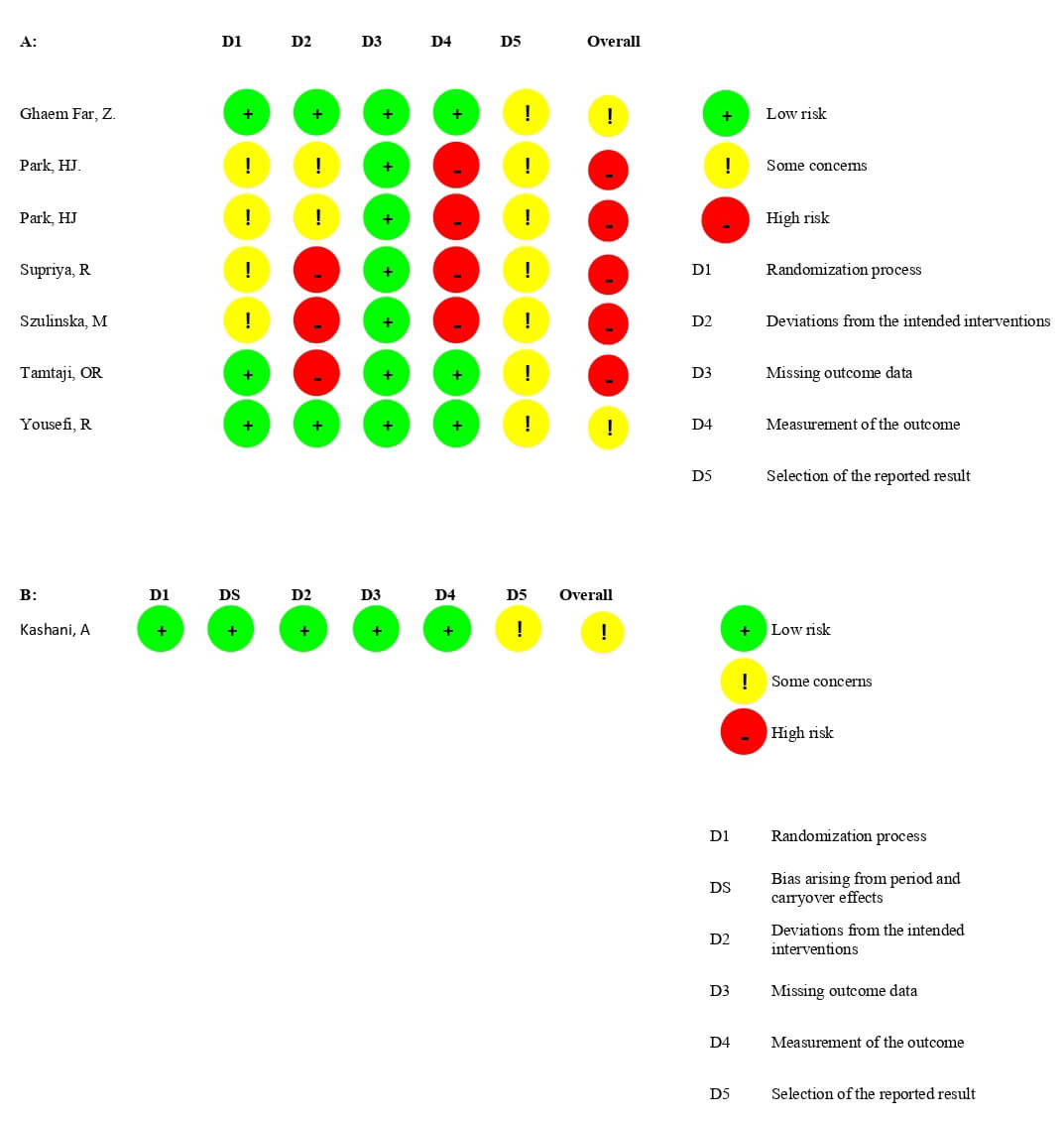 Fig. 2.
Fig. 2.
Quality assessment. (A) Parallel studies; (B) Crossover studies.
The meta-analysis of Spirulina’s effect on serum CRP levels included four
studies (four datasets), showing a significant reduction in CRP concentrations
compared with placebo group (weighted mean difference (WMD): –0.09 mg/L; 95%
CI: –0.16 to –0.02, p: 0.014) (Fig. 3). However, heterogeneity was
high (Cochrane’s Q test, p
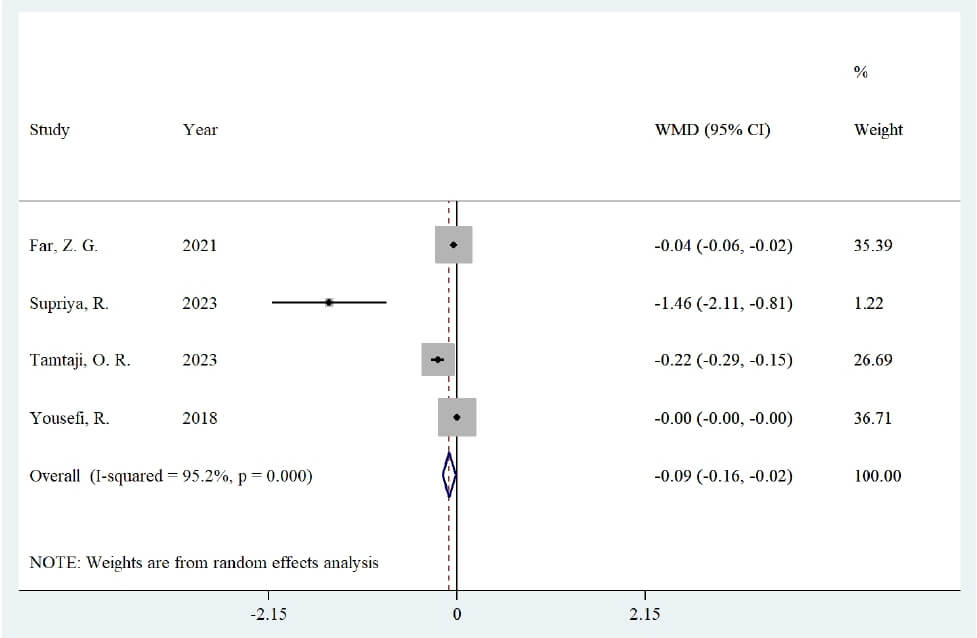 Fig. 3.
Fig. 3.
Forest plot of the effect of Spirulina supplementation on serum concentrations of C-reactive protein.
For serum level of TNF-
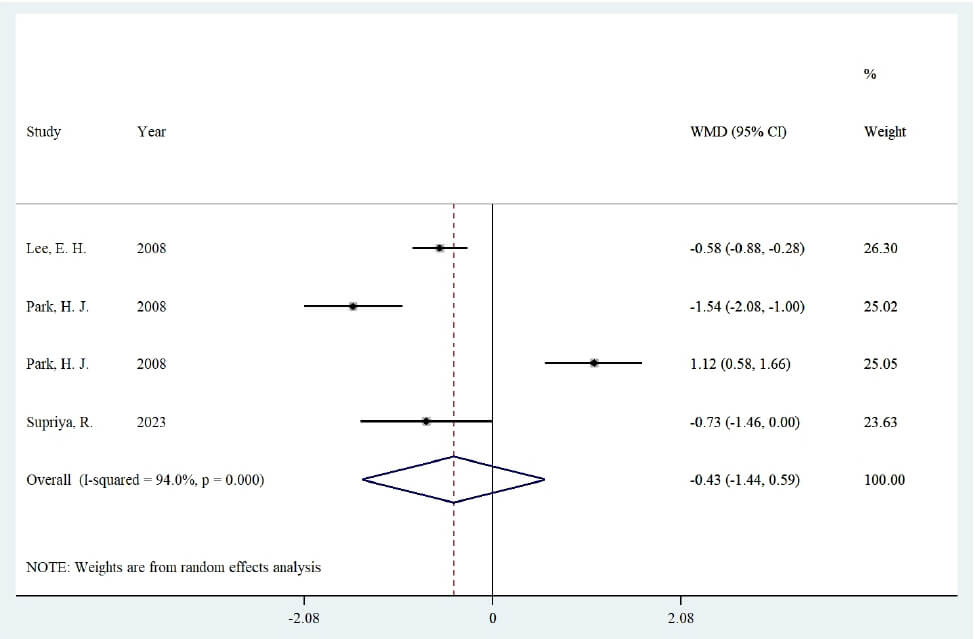 Fig. 4.
Fig. 4.
Forest plot of the effect of Spirulina supplementation on serum concentrations of tumor necrosis factor alpha.
The effect on serum IL-6 levels was assessed in five studies (six datasets) (Fig. 5). Using a random-effects model, no significant difference was observed in serum IL-6 levels between Spirulina and comparison groups (WMD: –0.44 pg/mL, 95% CI: –0.98 to 0.1, p: 0.111), with high heterogeneity (Cochrane Q test, p ˂ 0.001, I2: 98.2%).
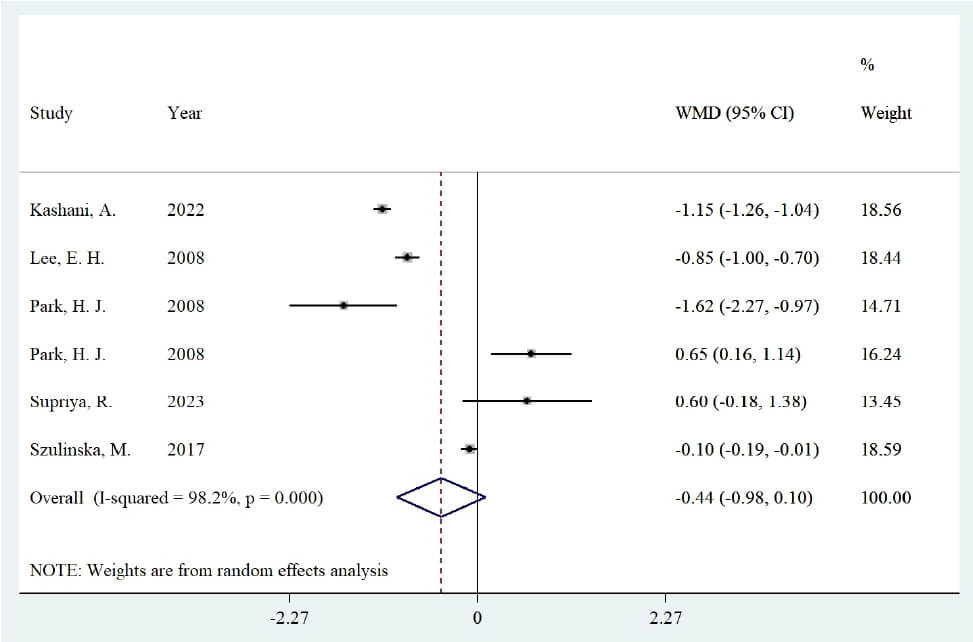 Fig. 5.
Fig. 5.
Forest plot of the effect of Spirulina supplementation on serum concentrations of interleukin-6.
Subgroup analyses were not conducted due to the limited number of included studies.
Sensitivity analysis indicated that excluding any single RCT did not alter the
overall effect for any of the studied variables. Although funnel plots for all
inflammatory mediators suggested some asymmetry (Fig. 6A–C), Begg’s test did not
indicate significant publication bias (p-values: CRP: 0.174,
TNF-
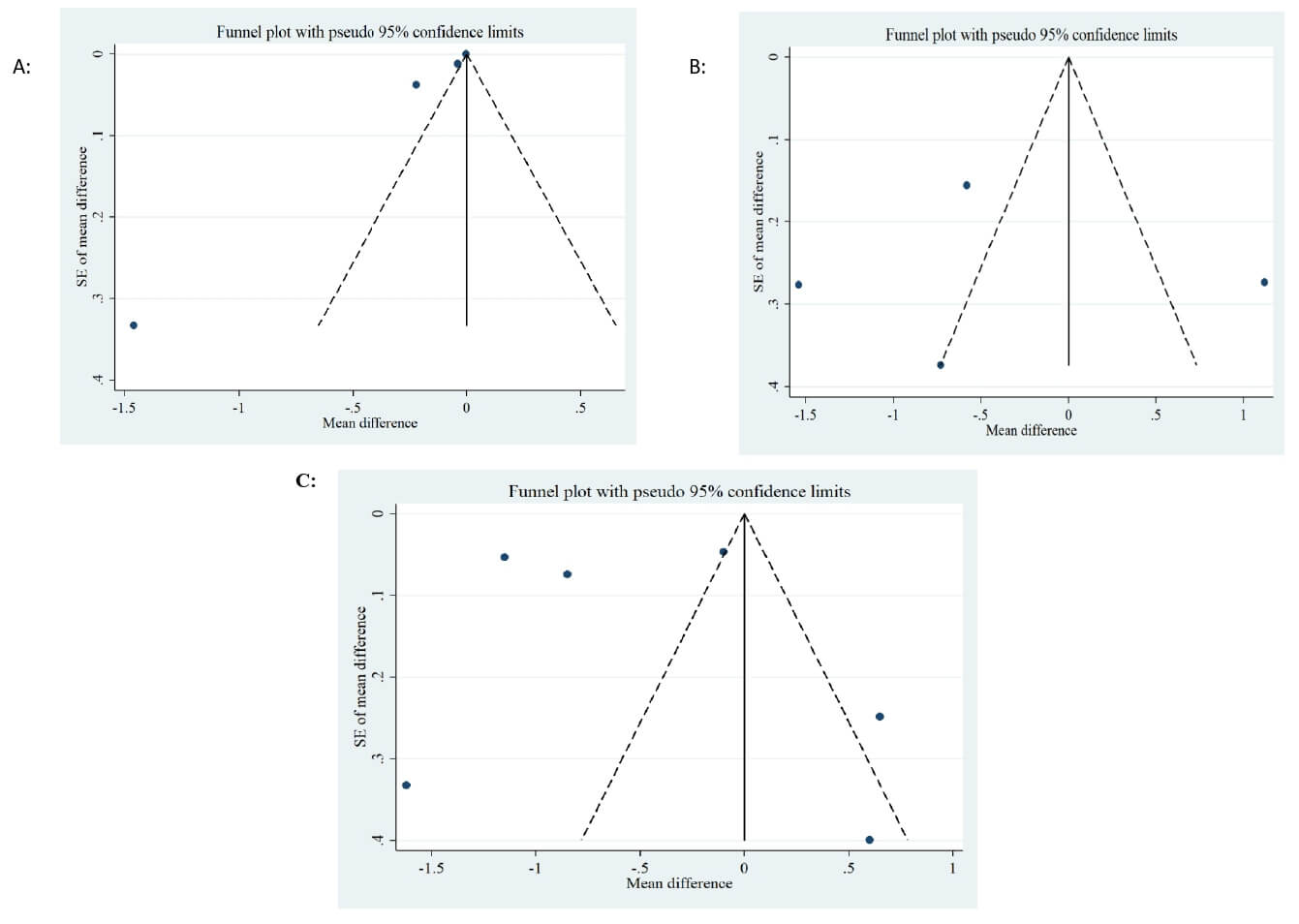 Fig. 6.
Fig. 6.
Funnel plots. (A) Funnel plots for the studies of the effects of Spirulina supplementation on serum levels of C-reactive protein; (B) Funnel plots for the studies of the effects of Spirulina supplementation on serum levels of Tumor necrosis factor-alpha; (C) Funnel plots for the studies of the effects of Spirulina supplementation on serum levels of Interleukin-6.
Spirulina, a microalga widely used as a nutritional supplement, exhibits a range of clinical and physiological benefits. Accumulating evidence suggests that it may help mitigate inflammatory processes associated with several chronic conditions, including dementia, vascular diseases, cancer, arthritis, asthma, diabetes, atherosclerosis, and inflammatory bowel disease [37, 38, 39, 40, 41, 42].
This systematic review and meta-analysis provides evidence that Spirulina supplementation in adults significantly reduces serum CRP levels. CRP is an acute-phase protein produced by the liver in response to inflammation [43], primarily driven by IL-6, which activates genes responsible for CRP transcription during the acute phase of the inflammatory response [44]. Elevated CRP levels are associated with increased risk of chronic conditions such as cardiovascular disease. Therefore, the observed reduction in CRP suggests that Spirulina may offer protective effects against inflammation-related chronic diseases.
Although reductions in serum TNF-
Beyond its antioxidant properties, Spirulina may exert anti-inflammatory effects through other mechanisms. It has been associated with weight loss, which can reduce obesity-related inflammation and oxidative stress, providing an additional anti-inflammatory pathway [51]. Moreover, Immulina, which is a high molecular weight polysaccharide extract, and gamma-linolenic acid (GLA), the predominant fatty acid in Spirulina, may enhance its anti-inflammatory effects. Immulina is known for its anti-inflammatory properties [52], while GLA may reduce the production of arachidonic acid metabolites when its ratio to arachidonic acid is increased [53].
Despite these promising findings, significant heterogeneity was observed among the included studies. This heterogeneity may stem from differences in genetic backgrounds, dietary habits, and lifestyle factors, which influence nutrient absorption and gut microbiota composition [54]. Previous research has suggested that the combination of Spirulina and probiotics may represent a novel synbiotic product [55]; therefore, the host’s diet and probiotic profile could be important determinants of Spirulina’s effect and a significant source of heterogeneity. Additionally, variations in Spirulina supplement formulations may contribute to this heterogeneity, as the concentration of bioactive compounds may differ across products. The geographical origin of Spirulina may further influence its bioactive composition [54]. Dosage (ranging from 1–8 g/day) and intervention duration (3–16 weeks) differed across studies, further contributing to variability. Emerging evidence suggests that age, gender, and body weight may also modulate Spirulina’s effects [54].
The findings of the present study align with those reported by Mohiti et al. [25] in their systematic review and meta-analysis. However, the study by Mohiti et al. [25] included two RCTs [56, 57] with populations comparable to those in the current study, which were published several years earlier [21]. In contrast, this study incorporated two additional more recent eligible studies [20, 23] that were not considered in the previous analysis. Furthermore, Mohiti et al. [25] did not evaluate the effect of Spirulina supplementation on serum CRP levels.
Several limitations should be considered when interpreting the findings of this study. First, subgroup analyses were not feasible due to the limited number of RCTs. Second, two studies did not report the Spirulina genera [20, 21], preventing genus-specific comparisons. Third, significant heterogeneity was observed across all studied variables, which may have influenced the pooled estimates and reduced the precision of the results. Fourth, the diversity in Spirulina types used may have introduced bias into the meta-analysis. Fifth, lifestyle and dietary data were unavailable and could not be accounted for in the analysis. Sixth, most RCTs were conducted in Asia, limiting generalizability due to regional differences in Spirulina composition. Seventh, no data were available on participants’ gut microbiota. Considering that Spirulina may act synergistically with probiotics as a potential novel synbiotic product [58], the baseline microbiota composition could be an important determinant of Spirulina’s effectiveness.
Nonetheless, this study has notable strengths. To the best of current knowledge, this is the first comprehensive systematic review and meta-analysis to evaluate the effects of Spirulina supplementation on a broad range of inflammatory mediators. Studies utilizing data similar to previously published research were excluded to avoid redundancy [56, 57]. Additionally, RCTs involving participants with acute inflammatory diseases were excluded to minimize confounding effects [59]. Finally, no restrictions were placed on publication date, allowing for a thorough inclusion of relevant studies.
The findings of this study suggest that Spirulina supplementation significantly
reduces serum CRP levels, whereas its effects on IL-6 and TNF-
The data presented in this study are available on request from the corresponding author.
The design of search strategy was done by AGh. Searching data bases was done by MHa and AGh, they also choose relevant RCTs based on inclusion and exclusion criteria. Reading articles full text, and data extraction was done by MHe and FA. AGh and HB performed statistical analysis. All authors contributed to the interpretation of data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We are extremely grateful to the data collection team at the Neyshabur University of Medical Sciences.
The research leading to these results has received funding from Neyshabur University of Medical Sciences (Grant Code: 140304111).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/IJVNR44330.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






