1 Las Colinas Institutes, Irving, TX 75039, USA
2 R&D, Sirio Life Technology (Shanghai) Co., Ltd., 201700 Shanghai, China
Abstract
Magnesium is an essential mineral required for energy metabolism, glucose regulation, cardiovascular function, bone integrity, and neural activity. Despite the vital physiological roles of magnesium, , dietary magnesium deficiency remains a widespread and underrecognized global public health concern. The recommended dietary allowance (RDA) for adults in the United States is approximately 420 mg/day for men and 320 mg/day for women, yet large proportions of the population fail to meet these levels with national nutrition surveys consistently documenting inadequate intake. For instance, 64.4% of Chinese adults consume less than the estimated average requirement (EAR) of 270 mg/day for both males and females. Globally, an estimated 2.4 billion people, or roughly 31% of the global population, fail to meet the recommended magnesium intake levels. This deficiency reflects multiple converging factors, including modern dietary patterns low in whole grains and vegetables, soil nutrient depletion from intensive agriculture, food processing losses, aged populations, chronic diseases, and socioeconomic disparities. The health implications are substantial, as magnesium deficiency is associated with elevated risks of cardiovascular disease, metabolic disorders, bone loss, and neuropsychiatric conditions. This review synthesizes current evidence on the biological importance of magnesium, global intake patterns, and determinants of deficiency, and discusses strategic interventions (such as dietary diversification, food fortification, biofortification, supplementation, and public health policies) to enhance magnesium nutrition and reduce the burden of noncommunicable diseases worldwide.
Keywords
- mineral nutrient
- magnesium deficiency
- dietary intake
- magnesium supplementation
- public health
Magnesium is a vital mineral and essential for a wide range of biochemical and physiological processes, including energy metabolism, protein and nucleic acid synthesis, muscle contraction, neuronal activity, glucose regulation, and blood pressure control [1, 2, 3]. There are approximately 24–25 g of magnesium in an adult body [4], of which approximately 50–60% of the body’s magnesium is stored in the bones, with the remainder distributed in muscles and soft tissues; less than 1% of total magnesium is present in serum [5]. Around 24–76% of ingested magnesium is absorbed by the intestine, and the remaining is eliminated in the feces [6]. Magnesium is primarily excreted in the urine, at approximately 100 mg per day [4]. As serum magnesium homeosis including absorption and excretion is under a tight control, its serum levels are generally a poor indicator of total body magnesium status [7]. At the cellular and molecular level, magnesium is fundamental for proper gene expression and cell replication by participating in nucleic acid metabolism [8].
It also stabilizes adenosine triphosphate (ATP), the primary energy molecule of the cell, thereby playing a central role in energy metabolism. In addition, it serves as a structural component and cofactor for numerous enzymes.
In the brain and neural system, magnesium plays a critical role in modulating ion channels and neurotransmitter release, thereby influencing neuronal excitability and synaptic plasticity [9]. It supports synaptic function and protects neurons by regulating N-methyl-D-aspartate (NMDA) receptor activity [10], highlighting its importance in neuroprotection and cognitive processes. Several meta-analyses have reported significant associations between magnesium status and neurological or psychological health outcomes, including insomnia, depression, Alzheimer’s disease, and cognitive decline [11, 12, 13, 14].
Predominantly stored intracellularly, magnesium is essential for cardiovascular physiology, including the regulation of endothelial function/vascular tone, heart rate and rhythm, as well as modulation of myocardial excitability [15, 16, 17], in which magnesium facilitates calcium reuptake into the sarcoplasmic reticulum during muscle relaxation—an energy-dependent process that also requires magnesium as a cofactor for ATP [1, 2]. It contributes to blood pressure regulation through its effects on vascular smooth muscle where it acts as a natural calcium channel blocker that can antagonize vasoconstriction and inhibit norepinephrine release from sympathetic nerve terminal [18], its regulatory role in the renin-angiotensin-aldosterone system and improved endothelial function [19, 20]. Magnesium is linked to heart rate and arrhythmias [21, 22]. The elevated resting heart rate is an emerging risk factor related to cardiovascular aging, morbidity and (all-cause) mortality [23, 24, 25].
Like calcium, magnesium is fundamentally important for skeletal muscle and bone health and the pathogenesis of bone-related disorders [15, 26]. Beyond serving as a storage site, bone tissue is an active participant in magnesium homeostasis, and magnesium in turn exerts significant regulatory effects on bone mineralization, parathyroid hormone (PTH) secretion, and vitamin D metabolism [26]. It contributes to the structural integrity of hydroxyapatite crystals, the primary mineral component of the bone, and works synergistically with calcium and phosphorus to preserve skeletal strength [27, 28]. Magnesium deficiency has been linked to reduced bone mineral density (BMD), disrupted osteoblast and osteoclast activity, and an increased risk of osteoporosis and fractures, particularly in postmenopausal women [29, 30, 31]. Magnesium also participates in skeletal muscle contraction and relaxation, like in cardiomyocytes.
Magnesium is also involved in glucose homeostasis and insulin sensitivity [32, 33]. Its roles in glucose utilization and insulin signaling are particularly important in the context of metabolism and metabolic syndrome. Fundamentally, magnesium participates in multiple enzymatic steps of carbohydrate metabolism and supports insulin signaling pathways by activating insulin receptor tyrosine kinase, enhancing its ability to bind ATP and phosphorylate downstream proteins. These actions make magnesium indispensable for glucose transport across cell membranes in the liver and skeletal muscle.
Additionally, magnesium influences the immune system and may play a regulatory
role in inflammation. Chronic magnesium deficiency is associated with elevated
levels of pro-inflammatory cytokines such as TNF-
Magnesium deficiency can disrupt numerous physiological processes, leading to symptoms such as muscle cramps, fatigue, irregular heartbeat, and neurological disturbances. Chronically low magnesium levels have been linked to a higher risk of diverse disorders, including osteoporosis, cardiovascular disease, type 2 diabetes mellitus, migraine, and certain cancers. These associations underscore magnesium’s systemic importance and its role as a determinant of overall health and disease susceptibility.
Given the broad physiological significance of magnesium and its involvement in numerous cellular and systemic functions, assessment of magnesium status is crucial for both clinical and public health research.
Laboratory evaluation of magnesium status: Several laboratory methods are available to assess magnesium status [7, 37]. (1) Serum magnesium test: The most common and convenient clinical method, measuring magnesium concentration in the liquid portion of the blood. (2) 24-hour urinary magnesium test: Quantifies the total amount of magnesium excreted over 24 hours, providing a more comprehensive assessment, though less practical for routine use. (3) Magnesium loading (retention) test: Involves an intravenous infusion of magnesium followed by a 24-hour urine collection to determine the amount excreted. This method is particularly useful when serum magnesium levels appear normal but deficiency is still suspected. (4) Red blood cell (RBC) magnesium test: Measures magnesium within erythrocytes and is considered a more sensitive indicator of deficiency, as it reflects intracellular magnesium stores rather than extracellular concentrations.
Evaluation of dietary magnesium intake: For epidemiological and nutritional studies, laboratory-based methods used in clinical settings are often impractical due to high cost, logistical complexity, and limited scalability. As mentioned previously, the body’s homeostatic mechanisms tightly regulate serum magnesium concentrations within a narrow range (approximately 1.7–2.2 mg/dL). Consequently, serum levels or even clinical manifestations of hypomagnesemia or hypermagnesemia may not accurately reflect dietary magnesium status.
Aside from 24-hour urinary magnesium excretion, population-level assessments typically rely on dietary intake surveys [38], which estimate magnesium consumption using food frequency questionnaires or dietary recalls. These data are then compared with the Dietary Reference Intakes (DRIs) to evaluate how well populations meet recommended magnesium requirements, representing the most informative approach from a public nutrition and health perspective.
Despite its vital role in numerous physiological processes, magnesium deficiency is often overlooked and inconsistently reported, suggesting that it has yet to receive the level of clinical and public health attention warranted by its global prevalence and health implications.
North and South America: In both continents, the inadequacy of magnesium intake has been reported. In the US, data from the National Health and Nutrition Examination Survey (NHANES) indicate that nearly 50% of adults consume less magnesium than the Estimated Average Requirement (EAR) [39]. Among the sampled population, the highest prevalence of deficiency was observed in males aged 14–18 years, with intakes less than 78% of the EAR, and in those over 75 years old, with intakes less than 75% of the EAR. For females, magnesium intake was below 89% of the EAR in the 14–18 age group and below 63% in those over 75 years old [39]. The findings from the Canadian Community Health Survey (CCHS) 2015 revealed that the inadequate intake of magnesium affected more than 45% of the sampled Canadian adults, with older adults and females generally exhibiting higher rates of deficiency [40]. A National Health and Nutrition Survey (ENSANUT) conducted in Mexico showed that magnesium intake inadequacy was 34% for both sexes aged 12–19 years, and 35% in men and 25% in women aged 20 years or older [41]. In Brazil, the National Household Budget Survey/National Food Survey 2017–2018 (POF/INA 2017–2018) revealed that, the mean magnesium intake was 299.2 mg/day in users of dietary supplements, and 309 mg/day in non-users in adult men, in which EAR was 330 mg/day for 20–30 years old and 350 for 31–59; and the intake was 138.9 mg/day in dietary supplement users, and 231.6 mg/day in non-users in adult women, in which EAR was 255 for ages 20–50 years old and 265 for ages 51–59 [42].
Europe: Comparable trends are evident across European countries, with multiple studies revealing suboptimal magnesium intakes across various age groups [43]. For example, in females aged from 18–60 years, the intake less than EAR was 26.4% in France and 38.3% in UK; in males aged from 18–60 years, the intake less than EAR was 32.3% in France and 35.1% in UK. A later NutriNet-Santé cohort in French adults revealed that in 13,549 men who did not take dietary supplement the prevalence of magnesium inadequacy was 53.2%; in 30,197 women who did not take dietary supplement the prevalence of magnesium inadequacy was 51.6% [44]. In Spain, the inadequate intake for magnesium was 47% among 13,983 participants of the SUN cohort [45]. According to the nationwide survey on the nutrition of adolescents and adults (Ergebnisbericht Teil 2. Nationale Verzehrsstudie II. Die bundesweite Befragung zur Ernährung von Jugendlichen und Erwachsenen), 26% of men and 29% of women did not reach the recommended daily intake of magnesium in Germany [46].
Asia: Magnesium intake inadequacy or insufficiency is also prevalent in many Asian countries. In China, the dietary magnesium insufficiency rate (intake below the EAR that is 270 mg/day for both males and females) was 59.9% overall (53.9% for males and 65.4% for females) in a sample of 14,740 residents from 15 provinces in 2018 [47]. The median intake was 283.70 mg/day in 1991 and 238.89 mg/day in 2018, representing a remarkable decline. The China Development and Nutrition Health Impact Cohort Survey showed sampled from 10 provinces in 2022–2023, 64.4% of adults had magnesium intake below the EAR [48]. In Japan, data from the MINNADE—a nationwide survey study on dietary intake evaluation, demonstrated that the mean magnesium intake in male adults was 241 mg/day in the 18–29 age group, 257 in 30–49, 305 in 50–64, 326 in 65–74, and 334 in 75–79 years, representing inadequacy prevalence of 76.4%, 81.3%, 55.4%, 31.9% and 20% below respective EARs; in female adults, it was 205 mg/day in the age group of 18–29 years, 236 in 30–49 years, 276 in 50–64, 308 in 65–74, and 283 in 75–79, representing inadequacy prevalence of 74.6%, 57.8%, 26.8%, 14.0% and 13.6% below respective EARs [49]. In India, the percentage of geriatric subjects who did not consume the recommended daily intake for magnesium was 48% [50]. In Korea, the Korea National Health and Nutrition Examination Survey 2016–2019 revealed that the mean dietary magnesium intake among 28,418 participants who were 1 years or older was 300.4 mg/day. Although this was above the Korean EAR, nearly half of the population had inadequate magnesium intake [51].
Global perspective: A 2024 modeling study that estimated median intakes of 15 micronutrients for 34 age–sex groups across 185 countries found that 2.4 billion people or approximately 31% of the global population had inadequate magnesium intake [52]. This underscores that a substantial portion of individuals in both developed and developing countries fail to meet intake recommendations (either EAR or Recommended Dietary Allowance [RDA]) for magnesium (see Fig. 1, Ref. [52]).
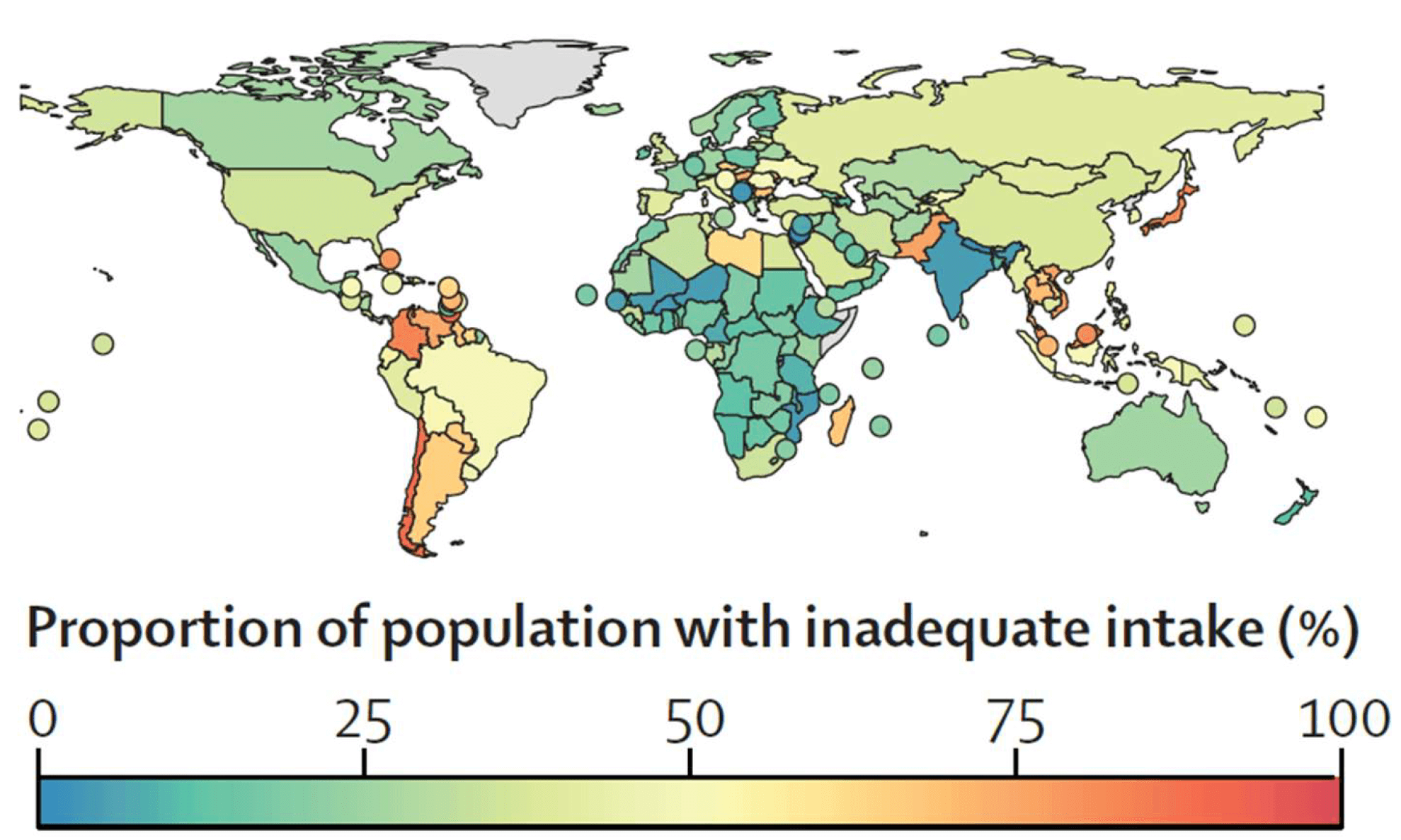 Fig. 1.
Fig. 1.
Estimated prevalence of inadequate magnesium intake by country in 2018. Globally, an estimated 2.4 billion people or approximately 31% of the population had insufficient magnesium intake. (Adapted with permission from Passarelli et al. [52] Lancet Global Health. 2024; 12: e1590–e1599).
In low- and middle-income countries, dietary magnesium deficiency is often compounded by food insecurity, poor dietary diversity, and limited access to magnesium-rich foods. Regions in Sub-Saharan Africa and South Asia report high prevalence of magnesium deficiency, though comprehensive data are limited. For example, studies conducted in India and Bangladesh indicate that a large portion of the population, especially in rural areas, consumes less than the daily recommended intake of magnesium due to reliance on nutrient-poor staple crops.
Certain demographic groups, including the elderly, individuals with gastrointestinal disorders, diabetics, pregnant women, and those consuming high levels of processed foods, are at greater risk. Magnesium intake is inversely related to socio-economic status, with low-income populations more likely to experience deficiency due to limited access to diverse and fresh food sources. In urban environments, dietary transitions toward energy-dense but micronutrient-poor foods have exacerbated the risk of micronutrient deficiencies, including magnesium.
Moreover, variations in food fortification policies, nutritional guidelines, and public health strategies across countries contribute to the global heterogeneity in magnesium intake. Standardization of assessment tools and inclusion of magnesium as a key nutrient in dietary surveillance programs are essential for better estimation and comparison of deficiency prevalence across regions.
Multiple factors, either acting independently or in combination, contribute to magnesium deficiency. These include dietary habits, lifestyle behaviors, physiological or pathological conditions, agricultural practices, and environmental influences such as climate change [53, 54].
Dietary Patterns: Magnesium is obtained from dietary sources (see Table 1 for magnesium content in selected foods). Modern diets rich in refined grains, added sugars, and processed foods are typically low in magnesium. Processing and boiling certain vegetables can result in 80% to 90% loss of magnesium content [53]. Refining whole grains can remove up to 80% of their magnesium content [54]. Furthermore, increased consumption of fast food and ultra-processed snacks displaces magnesium-rich foods (such as legumes, green leafy vegetables, nuts, and whole grains). Sugary beverages, including sodas and sweetened juices, are also linked to decreased magnesium status, partly due to increased urinary excretion.
| Dietary source | Serving size | Mg/Serving | DV (%) |
| Pumpkin seeds (roasted) | 1 ounce | 156 | 37 |
| Chia seeds | 1 ounce | 111 | 26 |
| Almonds (dry roasted) | 1 ounce | 80 | 19 |
| Spinach (boiled) | ½ cup | 78 | 19 |
| Cashews (dry roasted) | 1 ounce | 74 | 18 |
| Peanuts (oil roasted) | ¼ cup | 63 | 15 |
| Soymilk | 1 cup | 61 | 15 |
| Black beans (cooked) | ½ cup | 60 | 14 |
| Edamame (shelled, cooked) | ½ cup | 50 | 12 |
| Peanut butter | 2 tbs | 49 | 12 |
| Potato (baked with skin) | 3.5 ounces | 43 | 10 |
| Rice (brown, cooked) | ½ cup | 42 | 10 |
| Yogurt (plain, low fat) | 8 ounces | 42 | 10 |
| Oatmeal (instant) | 1 packet | 36 | 9 |
| Kidney beans (canned) | ½ cup | 35 | 8 |
| Banana | 1 medium | 32 | 8 |
| Salmon | 3 ounces | 26 | 6 |
| Milk | 1 cup | 24–27 | 6 |
| Halibut (cooked) | 3 ounces | 24 | 6 |
| Raisins | ½ cup | 23 | 5 |
| Bread (whole wheat) | 1 slice | 23 | 5 |
| Avocado (cubed) | ½ cup | 22 | 5 |
| Chicken breast (roasted) | 3 ounces | 22 | 5 |
| Beef, ground, 90% lean | 3 ounces | 20 | 5 |
| Broccoli (chopped, cooked) | ½ cup | 12 | 3 |
| Rice (white, cooked) | ½ cup | 10 | 2 |
| Apple | 1 medium | 9 | 2 |
| Carrot (raw) | 1 medium | 7 | 2 |
One cup = 8 ounces; One ounce = 28.3 grams; Adapted from https://ods.od.nih.gov/factsheets/Magnesium-HealthProfessional.
Abbreviation: Mg, magnesium; DV, daily value; tbs, tablespoon.
Soil Depletion and Agricultural Practices: Intensive farming and the excessive use of synthetic fertilizers have significantly depleted magnesium in agricultural soils. Unlike nitrogen, phosphorus, and potassium, magnesium is often omitted from conventional fertilization protocols [55, 56]. Because plant magnesium uptake depends directly on soil magnesium availability, soil depletion translates into lower magnesium content in food crops. Continuous cultivation without magnesium replenishment accelerates leaching losses, especially in acidic or sandy soils [54]. Consequently, declining magnesium concentrations in crops have contributed to dietary deficiencies across populations. Due to its neglect in soil management programs, magnesium has been described as the “forgotten element” of modern agriculture [57].
Socioeconomic Disparities and Food Insecurity: “Hidden hunger” - defined as micronutrient deficiency despite adequate caloric intake—may explain insufficient magnesium intake, particularly where diets rely heavily on refined carbohydrates and fats [1, 53]. In low- and middle-income countries, widespread food insecurity, limited dietary diversity, and restricted access to magnesium-rich foods exacerbate the problem. Many underserved populations depend on polished rice and other refined staples poor in magnesium [58], which may in part explain why some developing areas exhibit particularly high prevalence of magnesium deficiency, though comprehensive national data remain limited [52]. These findings underscore the nutritional vulnerability associated with low socioeconomic status and agricultural dependence.
Population-Based Risk Factors: Magnesium deficiency is more common among certain groups due to physiological, pathological, lifestyle, and socioeconomic influences.
Individuals with chronic health conditions face elevated risk. Gastrointestinal disorders (such as Crohn’s disease, celiac disease, and chronic diarrhea) reduce magnesium absorption, while conditions like uncontrolled diabetes promote excessive urinary magnesium excretion [59, 60]. Chronic alcoholism further reduces absorption and increases renal losses [61].
The use of medications plays a significant role in iatrogenic magnesium deficiency. Common culprits include loop and thiazide diuretics, certain antibiotics, proton pump inhibitors, and chemotherapeutic agents, which can impair intestinal absorption or enhance urinary excretion of magnesium [62, 63, 64, 65]. Patients on long-term polypharmacy may therefore require closer monitoring of magnesium status.
Psychological stress and intensive physical activity can increase magnesium requirements through neuroendocrine and metabolic pathways. Stress activates the hypothalamic-pituitary-adrenal (HPA) axis, raising cortisol levels and altering magnesium metabolism [66], which can perpetuate a cycle of deficiency. Physically active individuals, including athletes and manual laborers, may experience additional losses through sweat and increased cellular demand [67]. Without adequate dietary intake, these individuals are prone to depletion.
Older adults are particularly vulnerable due to age-related declines in intestinal magnesium absorption and changes in renal handling that promote urinary magnesium loss [68]. Furthermore, this population often has restricted dietary variety [69], particularly in institutionalized settings, and commonly uses medications (such as diuretics, proton pump inhibitors, and laxatives) that interfere with magnesium metabolism [63, 70, 71]. Consequently, magnesium deficiency in older adults is often more pronounced than in younger populations [68, 72].
Limited Recognition and Monitoring of Magnesium Deficiency: Magnesium deficiency remains underrecognized in many public health frameworks. A key barrier is the limited inclusion of magnesium in national dietary assessments, making it difficult to identify at-risk populations or guide effective nutrition planning.
Public awareness of magnesium’s importance to health is also low. Unlike sodium, iron, or calcium, magnesium receives minimal attention in dietary education, nutrition guidelines, or clinical practice. This invisibility contributes to poor recognition of deficiency and weak consumer demand for magnesium-rich foods. Although food fortification efforts exist in some regions, they remain limited in scale and public acceptance, often due to low awareness, cost, and perceived necessity.
To address these gaps, magnesium should be more consistently integrated into nutrition monitoring systems, food policy initiatives, and public health messaging. Increasing education and awareness could help promote healthier dietary choices and reduce the global burden of magnesium deficiency.
Water Treatment: Drinking water can provide a meaningful contribution to magnesium intake [73]. Populations consuming mineral-rich water from natural sources or springs may receive a non-negligible contribution to daily magnesium intake. Modern water treatment methods (e.g., water softening, reverse osmosis, and distillation) can significantly reduce the magnesium content of drinking water [74, 75]. In urban settings, the growing reliance on bottled or filtered water may further diminish this source, potentially contributing to the overall decline in dietary magnesium intake.
Magnesium deficiency has significant implications for public health due to its association with major chronic diseases (see Fig. 2). Low magnesium intake is linked to increased risks of hypertension, cardiovascular disease, type 2 diabetes, metabolic syndrome, osteoporosis, and even depression, which collectively constitute the prevailing noncommunicable diseases (NCDs), and magnesium supplementation helps meet dietary requirements and reduce related risks from clinical and public health perspectives.
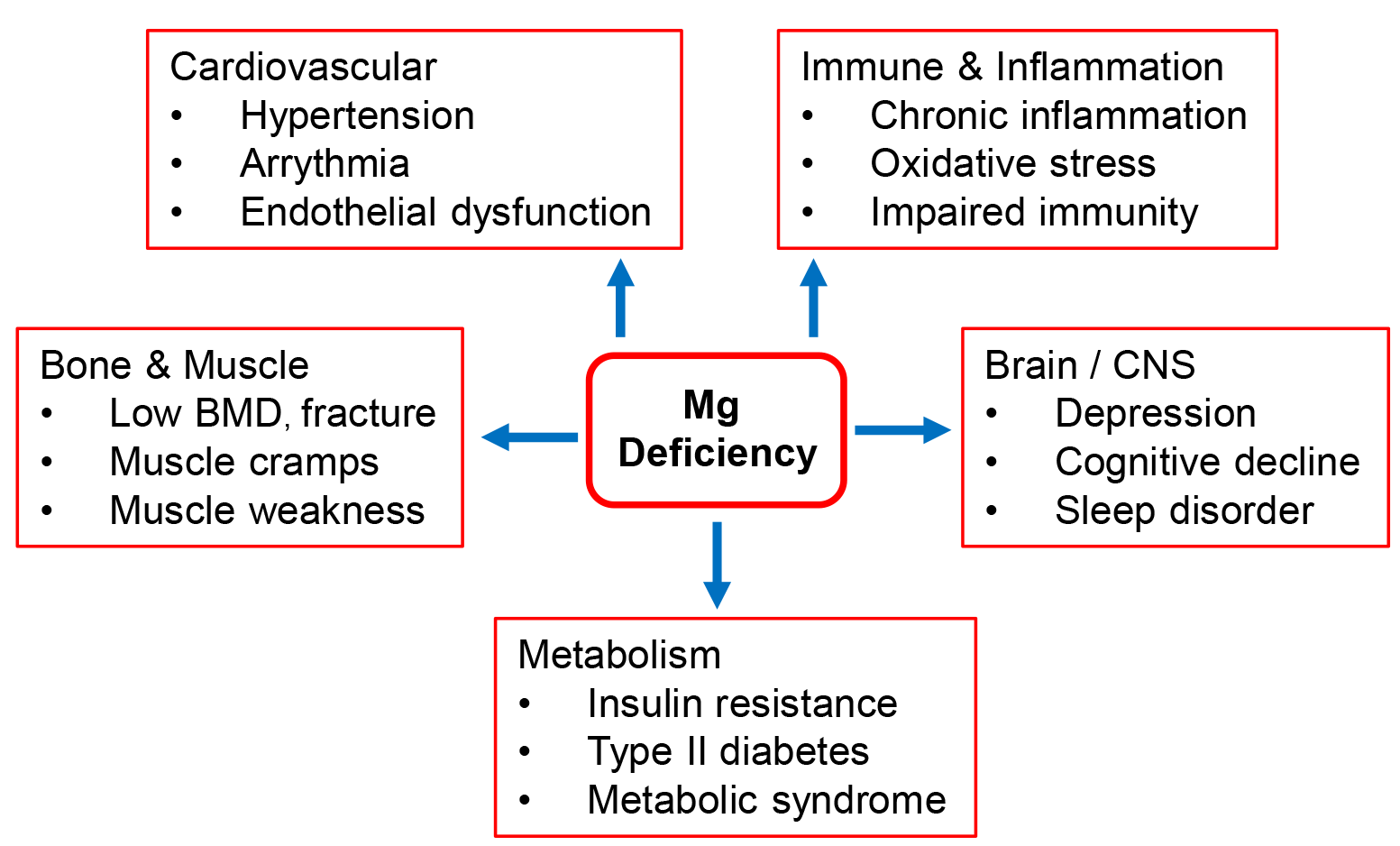 Fig. 2.
Fig. 2.
Major health consequences of magnesium deficiency. Magnesium serves as a cofactor for hundreds of enzymatic reactions critical to physiological function. Its deficiency is implicated in the development of type 2 diabetes, cardiovascular disease, hypertension, and osteoporosis. Although these disorders are multifactorial, magnesium deficiency–induced chronic low-grade inflammation and oxidative stress may further aggravate their underlying pathophysiology. BMD, bone mineral density; CNS, central nervous system.
Cardiovascular Health: Magnesium is a critical regulator of cardiovascular physiology, contributing to vascular tone, endothelial function, and the maintenance of normal heart rate and rhythm. Magnesium deficiency has been consistently associated with an increased risk of cardiovascular diseases (CVD), including hypertension, arterial stiffness, atherosclerosis, and arrhythmias [76].
A substantial body of epidemiological and clinical evidence supports a strong, inverse, dose-dependent association between magnesium status and major cardiovascular events. Meta-analyses encompassing more than half a million participants have demonstrated that individuals with higher (or relatively normal) serum and dietary magnesium levels exhibit significantly lower cardiovascular risk. The pooled relative risk (RR) for CVD was 0.64 (95% confidence interval [CI]: 0.51–0.80) for serum magnesium and 0.90 (95% CI: 0.84–0.96) for dietary magnesium, when comparing the highest with the lowest exposure categories. Correspondingly, for coronary heart disease (CHD), the pooled RR was 0.70 (95% CI: 0.57–0.85) and 0.86 (95% CI: 0.77–0.94), respectively. Each 0.1 mg/dL increment in serum magnesium concentration was associated with a reduced risk of CVD (RR: 0.93, 95% CI: 0.88–0.97) and CHD (RR: 0.90, 95% CI: 0.84–0.96), while every 100 mg/day increase in dietary magnesium intake corresponded to a 7–10% lower risk of both CVD and CHD [26]. The cardioprotective effect of magnesium appears particularly pronounced in reducing stroke risk [77, 78]. Pathophysiologically, magnesium deficiency promotes several mechanisms critical to the onset of hypertension and atherosclerosis, thereby accelerating the progression of CVD and CHD [79, 80].
Although serum magnesium concentrations are tightly regulated, numerous studies reveal a consistent inverse relationship between circulating magnesium levels and blood pressure [81, 82]. A meta-analysis of ten cohort studies reported that individuals with the highest dietary magnesium intake had a lower risk of hypertension (RR = 0.92; 95% CI: 0.86–0.98) compared with those withthe lowest intake, and that each 100 mg/day increment in magnesium intake was associated with a 5% reduction in hypertension risk (RR = 0.95; 95% CI: 0.90–1.00) [81]. Similarly, a 0.5 mg/dL increase in serum magnesium concentration was associated with a 7% lower risk of high blood pressure [82]. Interventional trials corroborate these findings: magnesium supplementation at a median dose of 368 mg/day for three months increased serum magnesium by approximately 0.1 mg/dL and lowered systolic and diastolic blood pressures by 2.00 and 1.78 mmHg, respectively [19]. In another trial, magnesium oxide supplementation at 450 mg/day for 24 weeks reduced resting heart rate by approximately 3 beats per minute (90% CI: 1–5 bpm) [83]. Long-term dietary evidence further supports the cardiovascular benefits of magnesium-rich diets. A 30-year prospective cohort study of 15,022 participants revealed that greater consumption of magnesium-rich foods (such as whole grains, nuts, legumes, vegetables, fruits, coffee, and tea) was associated with significantly lower risks of CVD and CHD [84].
Mechanistically, magnesium deficiency contributes to endothelial dysfunction, vascular calcification, inflammation, and oxidative stress, thereby promoting the initiation and progression of hypertension and atherosclerosis [79, 80]. These interrelated pathophysiological processes underscore magnesium’s essential role in cardiovascular homeostasis and highlight its potential as a modifiable dietary factor in the prevention of cardiovascular morbidity and mortality.
Diabetes and Metabolic Disorders: Magnesium insufficiency is commonly reported in individuals with insulin resistance and is frequently observed in people with type 2 diabetes. Hypomagnesemia has been consistently linked to poor glycemic control, insulin resistance/hyperinsulinemia, and increased risk of metabolic disorders [59, 85]. Longitudinal studies indicate that adequate magnesium intake is associated with a reduced risk of developing type 2 diabetes and metabolic syndrome [86, 87]. Moreover, magnesium supplementation has demonstrated beneficial effects in improving insulin sensitivity and lowering fasting blood glucose levels in individuals with prediabetes or metabolic syndrome [88, 89, 90]. Improvements in magnesium status have also been associated with favorable changes in HbA1c and a reduced incidence of diabetes-related complications [91, 92]. These findings highlight magnesium as a modifiable dietary factor with promising potential in the global fight against metabolic diseases.
Bone Health: Magnesium deficiency has been linked to impaired bone formation, disrupted osteoblast and osteoclast activity, and reduced bone mineral density, leading to an increased risk of osteoporosis and fractures, especially in older adults and postmenopausal women [29, 30, 31]. As magnesium plays a synergistic role with calcium and vitamin D, its insufficiency, including lower serum magnesium, can further compromise skeletal integrity, leading to a higher risk of incident fractures [93]. Research also suggests that higher magnesium intake is associated with greater improvement in bone health, including hip and femoral neck BMD, among these vulnerable populations [30, 94].
It is particularly noteworthy that magnesium is essential for the activation of
vitamin D, where magnesium functions as a cofactor for enzymes such as
25-hydroxylase and 1
Mental Health and Neurological Disorders: Magnesium plays a key role in neurotransmission and regulation of the hypothalamic-pituitary-adrenal (HPA) axis [98], which may affect stress responses, mood stability, and brain function. Epidemiological studies have shown evidence linking magnesium deficiency to a range of mental health problems, including depression, stress, anxiety, migraine, and cognitive impairment [99, 100]. Mechanistically, these associations are thought to involve neuroinflammation and oxidative stress driven by excess reactive oxygen species (ROS) [101, 102]. Emerging research also suggests that magnesium supplementation may alleviate inflammatory stress and improve sleep quality in older adults with low magnesium status [103].
Despite the biological plausibility and potential benefits of magnesium supplementation, findings remain inconsistent. Cross-sectional studies often show inverse associations between magnesium intake and depression, whereas prospective data fail to confirm clear causality [104]. Further longitudinal and interventional studies are therefore needed to elucidate the neurological and psychiatric consequences of magnesium deficiency and to determine whether supplementation can improve neurophysiological function and mental well-being.
Given its broad clinical relevance across multiple organ systems and its contribution to the global burden of NCDs, implementing scalable, evidence-based strategies to improve magnesium status represents an important public health priority.
Addressing magnesium deficiency requires a multi-level strategy that integrates dietary improvement, clinical management, agricultural reform, and public health policy. Effective interventions must be tailored to both individual risk profiles and population-wide nutritional contexts to achieve sustainable improvements in magnesium status.
Dietary Strategies: Dietary modification remains the foundation for correcting magnesium insufficiency in the general population. Emphasis should be placed on increasing the intake of magnesium-rich foods including dark leafy greens (e.g., spinach, Swiss chard), legumes (e.g., black beans, lentils), nuts and seeds (e.g., almonds, pumpkin seeds), whole grains (e.g., brown rice, oats), and selected fruits and seafood [84, 105, 106].
Public health initiatives should strengthen food literacy programs and community-based nutrition education to promote awareness of these food sources. Policy instruments (such as front-of-pack labeling, agricultural subsidies for nutrient-dense crops, and promotion of minimally processed foods) can help close the gap between recommended and actual magnesium intake. Alignment with global dietary patterns emphasizing plant-forward or Mediterranean-style diets may also enhance magnesium sufficiency alongside other essential micronutrients.
Food Fortification and Biofortification: In populations where dietary intake remains chronically low or where agricultural soils are magnesium-depleted, food fortification and biofortification represent cost-effective and sustainable solutions. Fortifying staple foods such as flour, rice, or even drinking water has demonstrated potential to elevate population magnesium status [106, 107].
Biofortification, through either genetic breeding or agronomic practices, enhances the intrinsic magnesium content of crops and helps address the long-term challenge of soil mineral depletion [107, 108, 109]. Techniques such as soil enrichment with magnesium fertilizers, foliar application, and microbial-assisted nutrient uptake can improve both yield and nutritional quality. Importantly, integrating biofortified crops into local agricultural systems also supports food security and resilience to climate change.
Supplementation and Clinical Interventions: For individuals with confirmed magnesium deficiency or elevated risk (such as those with malabsorption syndromes, type 2 diabetes, gastrointestinal disorders, or prolonged diuretic use) magnesium supplementation is an effective therapeutic option. In critically ill patients or cases of severe depletion, parenteral (intravenous) supplementation is indicated when oral absorption is compromised.
For the general population with suboptimal intake, oral magnesium supplements provide a practical means to maintain adequate magnesium levels. Consultation with healthcare professionals can help determine optimal dosage and monitor serum magnesium to avoid excess. The bioavailability and gastrointestinal tolerability of magnesium vary by formulation; organic salts (such as magnesium citrate, lactate, and glycinate) generally offer superior absorption compared to inorganic forms such as magnesium oxide [110].
Emerging research also suggests potential adjunctive benefits of magnesium supplementation in the prevention or management of chronic diseases including hypertension, type 2 diabetes, and metabolic syndrome, though optimal dosing regimens require further clinical validation.
Policy and Educational Frameworks: A coordinated policy framework is essential to elevate magnesium from a neglected nutrient to a public health priority. Recommended measures include:
Public Awareness Campaigns: Use of mass media and healthcare platforms to communicate the importance of magnesium for cardiovascular, metabolic, and bone health.
Integration into Existing Health Programs: Incorporating magnesium education and monitoring into maternal-child nutrition, chronic disease prevention (particularly NCDs), and healthy aging initiatives.
Health System Incentives: Encouraging primary care providers to assess magnesium status in the context of hypertension, diabetes, and osteoporosis management.
Cross-sector Collaboration: Engagement among departments of health, agriculture, and education to ensure consistent messaging, resource allocation, and implementation of nutrition-sensitive policies.
Research and Surveillance: Strengthening research capacity and surveillance systems is critical for sustained progress. Regular dietary intake assessments, biomarker monitoring, and population-level magnesium mapping can help identify at-risk groups and guide resource allocation. Investment in implementation science can also determine which interventions (e.g., fortification vs. supplementation) are most effective in specific cultural and socioeconomic contexts.
These multi-pronged strategies for addressing magnesium deficiency are summarized in Fig. 3.
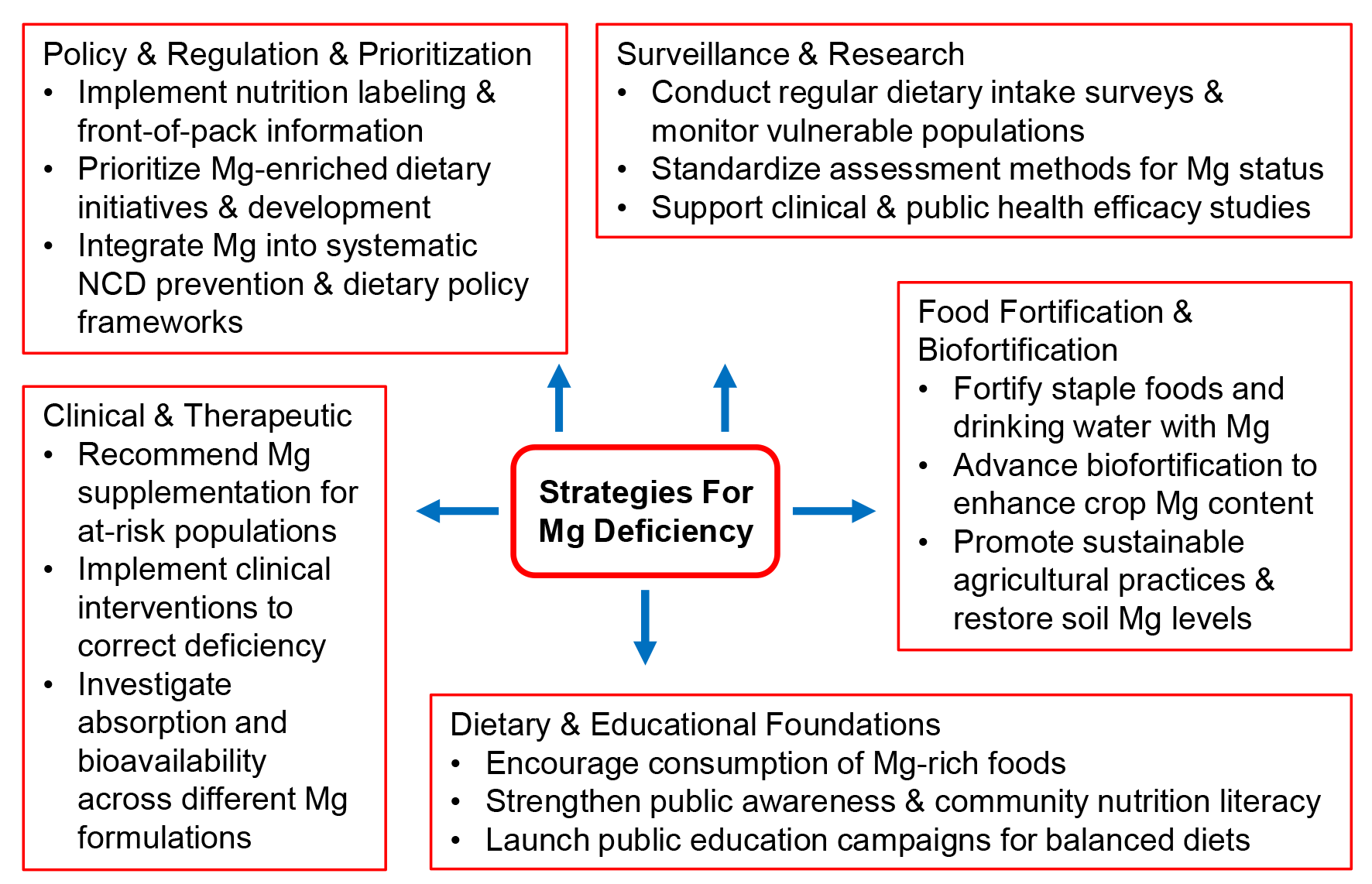 Fig. 3.
Fig. 3.
Addressing magnesium deficiency requires a multifaceted approach. While dietary awareness and public education form the foundation, coordinated efforts across academic research, food processing and industrial R&D, sustainable agricultural practices, and nutritional assessment at both individual and population levels are indispensable. Together, these stakeholders are encouraged to form an orchestrated consortium to implement effective and actionable strategies.
Magnesium is one of the cornerstones of human health, yet its deficiency remains a widespread and underrecognized global concern. The global burden of magnesium deficiency stems from a complex interplay of dietary inadequacy, environmental degradation, agricultural depletion, and socioeconomic disparities. Its impact spans physiological, clinical, and public health dimensions, affecting millions of individuals across all age groups and regions.
Actionable strategies are required to mitigate this deficiency, including enhancing dietary intake through diversification, food fortification, improved agricultural practice, expanding public awareness, and integrating magnesium screening into healthcare protocols for at-risk groups. A coordinated global effort involving authorities, health professionals, educators, and the agricultural sector will be essential to address this silent but significant public health issue.
Despite growing research interest, critical gaps persist in understanding the long-term health consequences of subclinical magnesium deficiency and in developing reliable biomarkers for early detection and monitoring. Future investigations should emphasize population-level interventions, longitudinal health outcomes, and the cost-effectiveness of magnesium-focused public health strategies.
From a broader perspective, advancing translational and preventive approaches that integrate nutrition science, precision medicine, and sustainable food systems will be critical for improving magnesium status globally. Emerging innovations such as digital health monitoring, agricultural biofortification, and personalized nutrition offer promising pathways for targeted intervention. Ultimately, strengthened international collaboration and policy alignment will be key to translating scientific evidence into tangible global action, ensuring that magnesium receives the recognition it deserves as a cornerstone of human and planetary health.
WZ and YZ conceptualized and designed the framework. WZ drafted the original manuscript. WZ and YZ critically reviewed and revised the content. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors express their sincere gratitude to Ms. Xiaomin Huang and Dr. Haotian Feng from Sirio Pharma for their constructive review and comments during the preparation of this manuscript and also thank the peer reviewers for their valuable opinions and suggestions.
This research received no external funding.
At the time of manuscript preparation and submission, YZ was employees of Sirio Pharma; no products or materials from Sirio Pharma were involved in the preparation of this review. The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



