1 Translational Research Unit, Medica Sur Clinic & Foundation, 14050 Mexico City, Mexico
2 Escuela Nacional de Medicina y Homeopatía, Instituto Politécnico Nacional, 07320 Mexico City, Mexico
3 Gastrobariatric Santa Fe, ABC Medical Center, 05348 Mexico City, Mexico
4 Digestive Diseases Unit, Medica Sur Clinic & Foundation, 14050 Mexico City, Mexico
Abstract
The prevalence of liver disease is steadily increasing worldwide. Meanwhile, metabolic dysfunction-associated steatotic liver disease (MASLD) has become the most prevalent chronic liver disease globally, characterized by the extensive involvement of multiple organs. Currently, the recommended treatment for MASLD is adopting a healthy lifestyle, which includes regular exercise and a balanced diet that incorporates plenty of vegetables. In this context, black radish is a cruciferous vegetable rich in glucosinolates, which represent the most beneficial active components. Glucosinolates act by preventing the induction of reactive oxygen species (ROS) and factors related to lipid metabolism, such as fibroblast growth factor 21 (FGF21) and nuclear factor erythroid 2-related factor 2 (Nrf2). There are several other components, such as precursors for glutathione (GSH) synthesis, which regulate liver enzymes and lipid peroxidation, as well as flavonoids, which help ameliorate lipid accumulation and possess antioxidant properties. These preventive health effects contribute to lipid metabolism and cellular energy balance, which, in turn, aid in liver detoxification and preventing diseases. This review aims to assess the biological detoxification mechanisms and effects of black radish in MASLD and related chronic diseases.
Keywords
- fatty liver
- steatohepatitis
- Raphanus sativus
- glucosinolates
- flavonoids
- polyphenols
- anthocyanins
- oxidative stress
Non-alcoholic fatty liver disease (NAFLD) has recently been renamed metabolic dysfunction-associated steatotic liver disease (MASLD), emphasizing the independence of this disease from exclusion criteria. This change recognizes the fact that MASLD can coexist with other chronic liver diseases [1]. Moreover, MASLD provides a more specific term that offers a positive, non-stigmatizing description of the condition, rather than one based on exclusion [2]. MASLD has metabolic dysfunction as its foundation, highlighting the significant impact and reduced heterogeneity this disease has. Key criteria include being overweight or obese, as well as having type 2 diabetes mellitus [3]. There are important risk factors that promote the development of MASLD, such as an increase in waist and/or abdominal circumference, obesity, hyperglycemia, dyslipidemia, and high blood pressure. Steatohepatitis can lead to the development of organic failure, resulting in liver cirrhosis, which requires liver transplant, or could lead to hepatocellular carcinoma due to inappropriate treatment, for example, if pharmacological or non-pharmacological treatments are not carried out [4].
MASLD is becoming a more frequent chronic disease (particularly in the Western world), with a worldwide prevalence of 25%, and is a main cause of cirrhosis and hepatocellular carcinoma. Metabolic alterations in this disease mainly occur in the liver, with other characteristics including alterations in cell and liver function indicators. One-quarter of the global population has been reported to have developed this disease [5]. Moreover, diseases such as obesity (with a prevalence of 82%), dyslipidemia (70%), and hypertension (68%) represent diseases that are most frequently related to fatty liver-associated metabolic syndrome [6]. Approximately 30% of the adult population is considered to have developed MASLD [7].
The human body is constantly exposed to toxins from different sources, such as foods or medicines, and the liver is one of the main organs involved in their detoxification [8]. The liver is an organ in which physiological functions such as the metabolism of macronutrients; production of bile for the absorption of fats; homeostasis of cholesterol, triglycerides, and drugs; and glucose storage (in the form of glycogen) are carried out [9]. It also processes primary metabolites, such as fatty acids, when they are supplied in large quantities or when their proper elimination cannot be carried out, promoting the production of toxic lipid species and inducing stress, injury, and death in liver cells [10].
Previous study has shown that consumption of supplements or vegetables naturally
promotes functions that have beneficial effects on the liver [8]. Specifically,
Raphanus sativus var. Sativus, also known as black
radish or Spanish black radish, belongs to the Brassicaceae or cruciferous family
[8, 11] and can be eaten naturally (i.e., as fresh food) or used as a food
supplement [12]. This vegetable has European origins, and in Mexico, it is
cultivated for medicinal and culinary purposes [13]. Interestingly, it has been
demonstrated that black radish facilitates the release of detoxification enzymes,
such as quinone reductase, cytochrome P450 (CYP) family, heme oxygenase 1, and
glutathione S-transferase
It is important to mention some of the core pathways of certain principal mediators involved in lipids and glucose regulation. The nuclear factor erythroid 2-related factor 2 (Nrf2) is a basic leucine zipper transcription factor that promotes the induction of genes involved in glucose metabolism and lipid oxidation. It has been demonstrated that its activation via Keap1 gene hypomorphic knockdown (Keap1flox/-) markedly suppresses the onset of diabetes; furthermore, liver triglyceride levels are reduced via the activation of Nrf2 signaling in a murine model [16]. This pathway reduces the accumulation of hepatic triglyceride, improves insulin sensitivity, and alleviates fibroblast growth factor 21 (FGF21) resistance [17]; according to this, FGF21 functions as an endocrine agent and is considered an energy metabolic regulator [18].
Cysteine-rich proteins and sulfates are precursors for glutathione (GSH) synthesis, when GSH and catalase activity increased, they conferee protection from oxidative damage reducing lipid peroxidation [19]. Furthermore, it has been described the ability of quercetin to neutralize free radicals such as superoxide anions, nitric oxide, and peroxynitrites apparently due to its chemical structure, which in turn also increase endogenous antioxidant levels and prevent cell death by inhibiting several enzymes such as xanthine, lipoxygenase, and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase [20]. Xanthine oxidase is an iron–molybdenum flavoprotein that plays a crucial role in catabolism, while lipoxygenase belongs to a large monomeric protein family that comprises iron cofactor-containing dioxygenases that catalyze the oxidation of lipids, producing leukotrienes and lipoxins as a result [21]. Finally, NADPH oxidase has an important role in several inflammatory diseases; for instance, quercetin can modulate NADPH oxidase activity in a macrophage-cell model via heme oxygenase-1 induction [22].
Glucosinolates are sulfur-containing phytochemicals abundant in cruciferous vegetables, and their biological effects are mainly attributed to their metabolites. Glucosinolates are found in different types of plant families; the family richest in them is Brassica, which includes black radish and other vegetables such as cabbage, brussels sprouts, broccoli, cauliflower, collards, kale, and kohlrabi [23]. The chemical properties of glucosinolates’ metabolites determine their biological activity and, thus, their effects on human health [14]. They are composed mainly of sulfate and thioglucose, which are precursors of isothiocyanates, to which they are hydrolyzed by myrosinase [24]. Their metabolism is characterized into phases: Phase I consists of their biotransformation, from which free radicals and reactive oxygen species are generated; in phase II, the free radicals and reactive oxygen species generated in phase I are deactivated. Phase II is known as the detoxification phase and has anticancer, antibacterial, anti-inflammatory, and antioxidant characteristics [8].
The two principal glucosinolates in black radish are glucoraphasatin and glucoraphanin:
Glucoraphasatin accounts for
Glucoraphanin, a sulforaphane precursor, has demonstrated metabolic benefits in murine models of obesity. Sulforaphane is capable of activates Nrf2 signaling, which has been explained before. On the other hand, sulforaphane is a hydrolysate of glucosinolates found in large quantities in cruciferous vegetables [14] capable of regulating lipid metabolism by deactivating c-Jun N-terminal kinase and glucose metabolism and alleviating FGF21 resistance in an obese mouse model [26]. Indeed, FGF21 has been related to the improvement of hepatic steatosis [18]. Moreover, sulforaphane has been shown to confer bactericidal properties, mainly against Helicobacter pylori and Staphyloccocus aureus; consequently, these properties exert a beneficial effect on diabetes [27].
Sinigrin and gluconapin are two other glucosinolate extracts from
Brassica juncea that have demonstrated hepatoprotective effects in a
murine model of MASLD, including the inhibition of lipid accumulation and a
reduction in the activities of inflammatory molecules, such as tumor necrosis factor-alpha (TNF
Mechanistic relevance: By enhancing phase II detoxification, promoting antioxidant gene expression, and modulating lipid/glucose pathways, black radish glucosinolates provide a strong molecular basis for their hepatoprotective role in MASLD.
Black radish contains high concentrations of sulfates and cysteine-rich proteins. It was observed that a methanol extract of Raphanus sativus root reduced the levels of thiobarbituric acid substances, as well as serum glutamate oxaloacetate transaminase (SGOT) and serum glutamate aspartate transaminase (SGPT): Enzymes that are specific indicators of liver cell injury. Therefore, this extract can reduce damage induced by paracetamol and return serum SGOT and SGPT to normal levels, which may translate to liver recovery [19]. Furthermore, an in vitro study has demonstrated that radish seeds contain two homologous 5 kD cysteine-rich proteins designated Raphanus sativus-antifungal protein 1 (RsAFP1) and RsAFP2, which exhibit potent antifungal activity; they are important components of the defense system of radish seeds [29, 30]. This evidence needs to be applied to studies in animal and human models.
Flavonoids are abundant phytochemicals in black radish, with quercetin being one of the most studied components. Quercetin exerts antioxidant, anti-inflammatory, and antifibrotic effects relevant to MASLD. For example, in an in vitro steatosis model, flavonoids have been demonstrated to ameliorate lipid accumulation and mitigate the enhancement of de novo lipid synthesis [31]. Quercetin is an interesting plant pigment and a flavonoid which is well known for its antioxidant properties [20] as described before.
In MASLD murine and cell models, quercetin has been demonstrated
to regulate liver lipid metabolites through the glycerophospholipid
metabolic pathway, thereby improving liver lipid accumulation and injury, as well
as inhibiting ferroptosis through the p38 mitogen-activated protein
kinases (MAPK)/extracellular signal-regulated kinase (ERK) signaling pathway
and alleviating the progression of MASLD [32]. Furthermore, it has been found
that quercetin, either alone or in combination with phosphodiesterase inhibitors,
improves liver biochemical markers, such as TNF
Moreover, another benefit of fermented black radish is its inhibition of transforming growth factor beta-1 and the collagen type I alpha 1 chain, which are involved in liver fibrosis mitigation [34]. This appears to explain quercetin’s antiproliferative function [35].
Clinical relevance: Quercetin supplementation, either alone or in combination with phosphodiesterase inhibitors, has been shown to improve biochemical markers of fibrosis, and inflammation, highlighting its therapeutic potential for MASLD.
Anthocyanins, a subclass of flavonoids, provide additional metabolic benefits.
They regulate carbohydrate metabolism, increase adiponectin and leptin secretion,
and protect pancreatic
A key metabolite, protocatechuic acid, reduces lipid accumulation in hepatocytes by inhibiting the cluster of differentiation 36 (CD36) palmitoylation, apparently by lowering zinc finger Asp-His-His-Cys (DHHC)-type palmitoyltransferase 5 palmitoylation [36]. Palmitoylation is a reversible post-translational lipid modification of proteins, catalyzed by the zinc finger DHHC domain-containing (ZDHHC) family of palmitoyltransferases; its dysregulation has been implicated in metabolic disorders [37, 38], which could impact MASLD inhibition. Importantly, a study indicated that anthocyanins account for 31.7% of flavonoid intake in reducing the risk of MASLD [39].
Polyphenols are secondary plant metabolites widely recognized for their antioxidant and hepatoprotective properties. In black radish and related plants, polyphenols contribute to liver detoxification by donating hydrogen atoms, chelating metal ions, and neutralizing free radicals [11, 40].
A previous study evaluated an extracted polyphenolic fraction (EPF) from bergamot (citrus bergamia Risso and poiteau) and administered it in a murine MASLD model. While no effect on body weight was observed, a significant decrease was evident in circulating leptin, insulin, lipid levels, and proinflammatory cytokine and chemokine levels in a Western diet murine model [41]. It is important to mention that leptin is considered an anorexigenic hormone and an important mediator of energy homeostasis; serum leptin levels are directly proportional to adipose tissue [42]. Thus, EPF is capable of restoring leptin levels and improving glucose and lipid metabolism.
Another product of the Rutaceae family, named after citrus lumia Risso, plays an important role in reducing the risk of metabolic diseases due to its polyphenol content. In an experimental study, a concentrated extract of citrus lumia, principally enriched with eriocitrin and hesperidin (rutinosidic flavanones), was administered to co-cultures of hepatic cell lines (HepG2/LX2), where a decrease in intracellular lipid content was observed [43]. Furthermore, curcumin, a principal active compound in turmeric and a plant-derived polyphenol, has substantial therapeutic potential; however, its low bioavailability and rapid metabolic degradation have limited its use [44].
Mechanistic relevance: Polyphenols exert hepatoprotection by regulating adipokine signaling, reducing oxidative stress, and downregulating inflammatory pathways. Collectively, these actions support black radish as a dietary adjunct in MASLD management.
Obesity represents the main risk factor for developing various diseases that
affect different organs, such as the heart, pancreas, and liver. In this
scenario, a diet rich in cholesterol can lead to fatty liver complications, such
as steatohepatitis, liver fibrosis, cirrhosis, and, ultimately, liver cancer
[6, 45, 46]. The hepatoprotective effects and reduction in lipid levels following
the ingestion of black radish juice have been studied in a mouse model of
hyperlipidemia, demonstrating its antioxidant properties [47]. In this context, a
Korean group has carried out a murine study with pretreated fermented black
radish, demonstrating that its hepatoprotective factor decreases the deposition
of cholesterol and triglycerides in hepatocytes and liver inflammation through
the regulation of adipogenic transcription factors and genes related to lipid
metabolism. These include CCATT/enhancer binding protein
A study conducted by Evans et al. [8] aimed to investigate the effects of black radish on liver function in a human model by measuring acetaminophen metabolites after administrating a 1000 mg dose both before and after 28 days of supplementation with black radish (370 mg). It was shown that dietary supplementation with black radish promoted detoxification, reduced estradiol in the blood, and helped the liver profile to reach normal ranges more rapidly, thus promoting liver cell protection [8].
Moreover, there is evidence that black radish also has a beneficial effect on diabetes mellitus, as it reduces glucose absorption. The above evidence demonstrates that this antioxidant activity reduces oxidative stress and lipid peroxidation. It has been observed that various parts of black radish have different effects on glucose. For example, black radish seeds reduce insulin resistance and glucose absorption; meanwhile, its leaves decrease the intestinal absorption of glucose, and leaf juice has strong antioxidant behavior, protecting against oxidative activities and preventing H2O2-induced hemolysis by more than 50% in red blood cells of a murine model [48, 49].
It is well known that a high-cholesterol diet can lead to the formation of gallstones, as cholesterol is their main component. In a murine study, it was speculated that glucoraphasatin would reduce serum levels of cholesterol and triglycerides [50]. According to this assumption, in another murine model, after gallstones were induced with a lithogenic diet, the efficacy of black radish root juice was evaluated. The eradication of gallstones was observed alongside decreases in triglycerides, cholesterol, and high-density protein levels in the groups that received concentrated black radish root juice at a concentration of 1:10, in comparison with the group that received root juice at a ratio of 1:100, urodeoxycholic acid (0.05%), or no treatment [51]. Elevated cholesterol levels are an important factor affecting abdominal inflammation and poor food intake, consequently decreasing intestinal absorption. There are other risk factors related to the formation of gallstones and the development of fatty liver disease, including an increase in body mass index in patients with obesity and patients losing weight too quickly because of low-calorie diets or bariatric surgery [52].
Importantly, the consumption of fast food, meat, refined sugars, and
carbohydrates may also lead to the formation of gallstones. The carbohydrate
sucrose has been related to a higher sediment concentration, thus promoting the
formation of gallstones; this event can be attributed to a reduction in hepatic
fatty acid oxidation, which leads to the incomplete activation of leptin’s signal
transduction pathway, specifically through the fatty acid catabolism regulators
AMPK and the nuclear receptor peroxisome proliferator-activated receptor alpha
(PPAR
Black radish has long been used as a homeopathic medicine for indigestion and abdominal inflammation, as well as stimulating bile juice production, thus improving digestion [47]. In mouse models, black radish favorably impacts the kidneys as it prevents the formation of stones and possesses diuretic properties [51]. In Mexican states, such as Guerrero and Oaxaca, this plant is well known for the treatment of urolithiasis, as well as a wide variety of urinary disorders [13]. Moreover, previous findings have indicated that this tuber has antifungal functions due to its rich cysteine content [13]. In the context of traditional medicine, its ethanolic and aqueous extracts have been shown to have antibiotic properties [55].
The pharmacological treatment of liver disease recommended by clinical practice guidelines includes vitamin E and pioglitazone. As black radish seems to have powerful antioxidant effects, it has the potential to ameliorate liver disease, as supported by experimental evidence [56]. According to an epidemiological study, there is a close association between an unhealthy lifestyle and fatty liver; indeed, weight loss can lead to the reversal of fatty liver disease, as well as a decrease in insulin resistance, when combined with personalized aerobic exercise. Successful treatment is reflected in the patient’s clinical condition by a reduced risk of progressive disease; however, there is currently no specific medication for this disease [57].
At present, non-pharmacological treatment does not lead to entirely favorable results for the control of MASLD [2]. Treatment with medicinal plants is accessible for patients living in developing countries, and traditional medicine has become the first option in such regions for ethical, practical, and economical treatments as opposed to conventional treatments, such as surgery, which are sophisticated and expensive [58].
In this regard, Mexico represents a country that has a great variety of medicinal plant species (4500 species). As a result, 90% of the population employs medicinal plants to treat different diseases, representing a very frequent practice among this population. However, it is still necessary to continue research on liver damage and mechanisms that impact the liver, as well as the effectiveness and safety of these plants and their active components when consumed [56, 58].
Liver disease is becoming increasingly important due to the growing number of people worldwide who suffer from it and the increasing prevalence of sedentary lifestyles and diets rich in high-carbohydrate foods. The recently renamed MASLD encompasses diseases that can coexist with other chronic liver diseases. At present, it is known that patients who suffer from insulin resistance and type 2 diabetes mellitus are more likely to be diagnosed with this disease. While a healthy lifestyle—including a balanced diet and regular exercise—is a major part of treatment and disease prevention, it has not been possible, to date, to propose a global treatment that mitigates all the complications of metabolic disease. This scenario has led to the search for alternative adjuvant solutions.
Black radish has been used for many years to treat certain discomforts and diseases, such as digestive problems and prevent gallstones and kidney stones, due to its ability to reduce blood levels of cholesterol and triglycerides. Even though the exact mechanisms associated with the metabolites present in black radish are unclear, there is evidence that the juice of black radish roots can eradicate gallstones. In current medical practice, one of the chemicals used to disintegrate bile stones is ursodeoxycholic acid; however, research has indicated that black radish root has a more powerful effect. In addition, it seems that black radish confers a liver-protective effect due to the presence of one of its main compounds—namely, glucosinolates—in addition to having multiple antioxidant functions. However, interestingly, there are other components such as anthocyanins, which at least partly account for the reduction in liver disease risk through their detoxifying abilities. Indeed, it seems that various parts of black radish have different effects on specific metabolic pathways (Table 1, Ref. [8, 11, 12, 14, 15, 17, 18, 19, 21, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 59, 60]). Therefore, specific extracts of bioactive compounds from plants could have beneficial effects on lipid metabolism and cellular energy balance, providing potential key strategies for biology and clinical applications (Fig. 1).
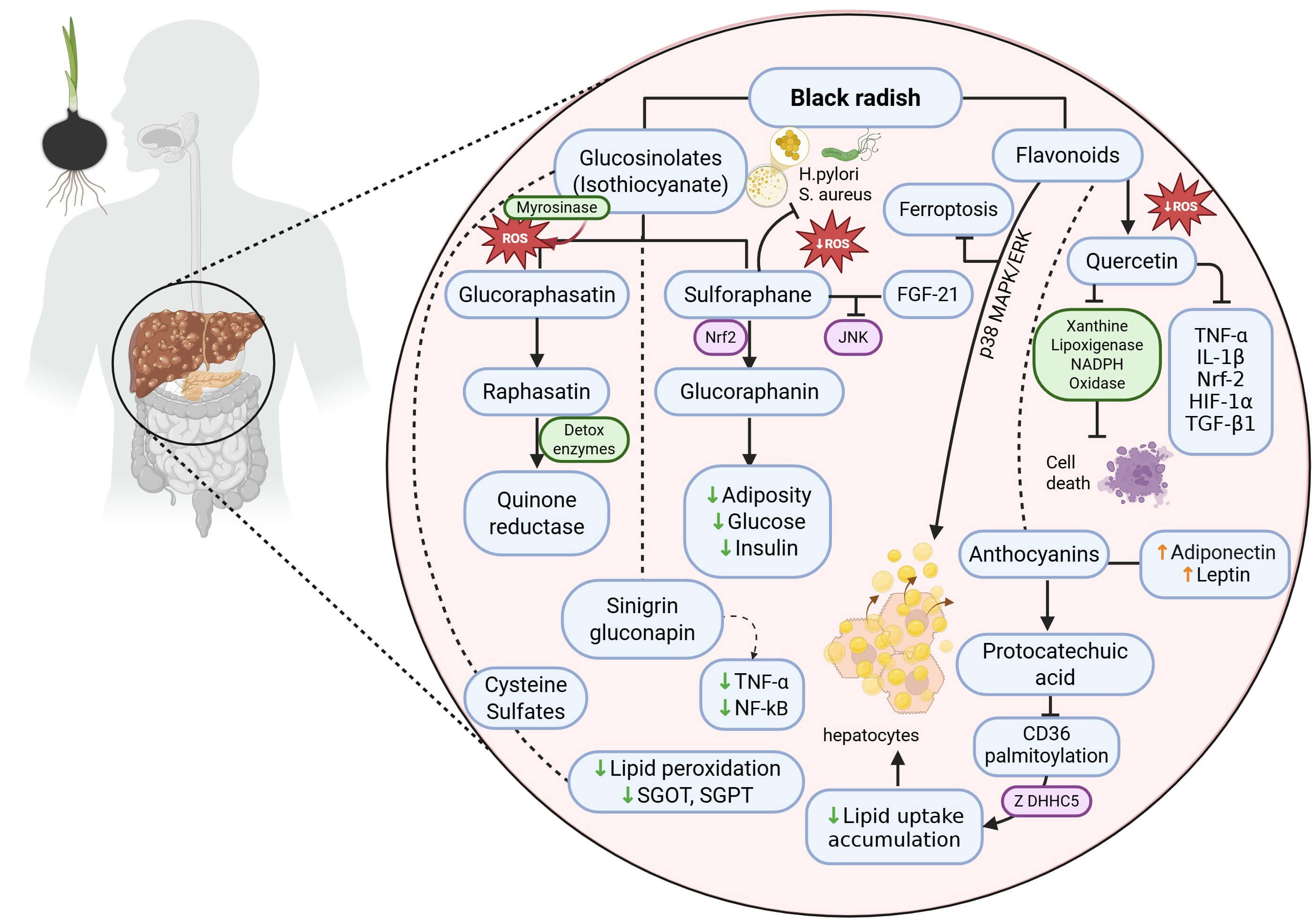 Fig. 1.
Fig. 1.
Molecular mechanisms of the hepato-metabolic effects of black
radish (Raphanus sativus L. var. niger) in MASLD.
Glucosinolates (glucoraphasatin, sulforaphane, and sinigrin) are metabolized
into active isothiocyanates (raphasatin and glucoraphanin), which upregulate
detoxification enzymes (e.g., quinone reductase) and regulate pathways such as
Nrf2 and c-Jun N-terminal kinase (JNK), thereby decreasing lipid peroxidation,
the serum levels of hepatic transaminases such as glutamic oxaloacetic
transaminase (SGOT) and serum glutamic pyruvic transaminase (SGPT), and
proinflammatory cytokines (TNF-
| Family compound | Metabolites | Principal chemical compounds | Biological effects | Systemic effects | Structure | References |
| Glucosinolates | Isothiocyanates: | Sulfate | Cancer prevention | 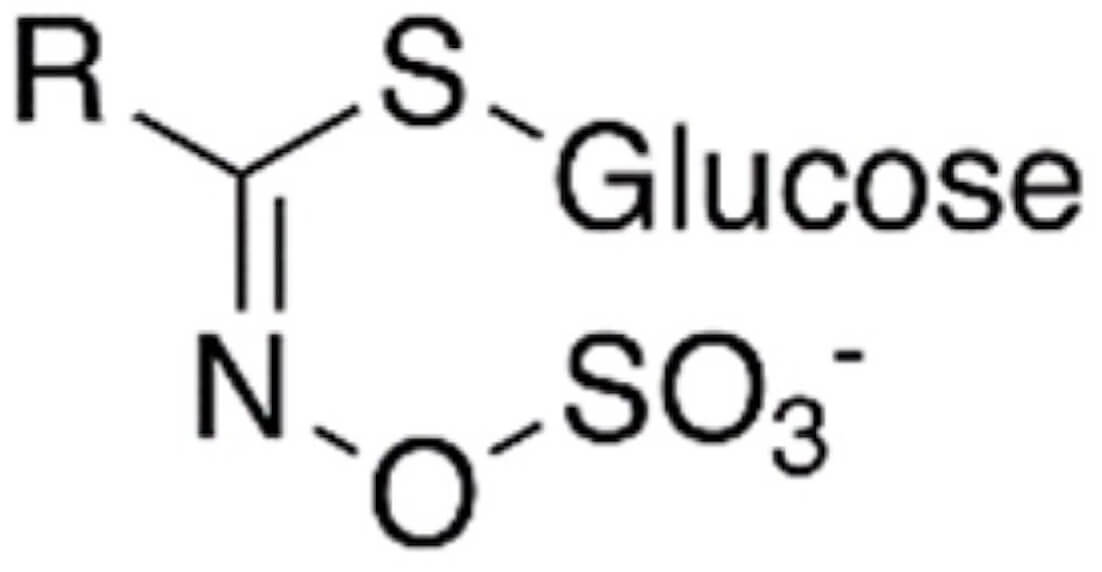 |
[8, 14, 15, 17, 18, 19, 21, 24] | |
| ∙Glucoraphasatin | Thioglucose | Hepatoprotective effects | ||||
| Raphastin | (0.3–560 mg/kg) | Lipid and glucose metabolism regulation | ||||
| ∙Glucoraphanin | Antioxidant and bactericidal properties | |||||
| Sulforaphane | c-Jun N-terminal kinase, | |||||
| ∙Sinigrin | TNF |
|||||
| ∙Gluconapin | ||||||
| Cysteine/Sulfates | GSH precursor | Sulfates and Cysteine-rich proteins | 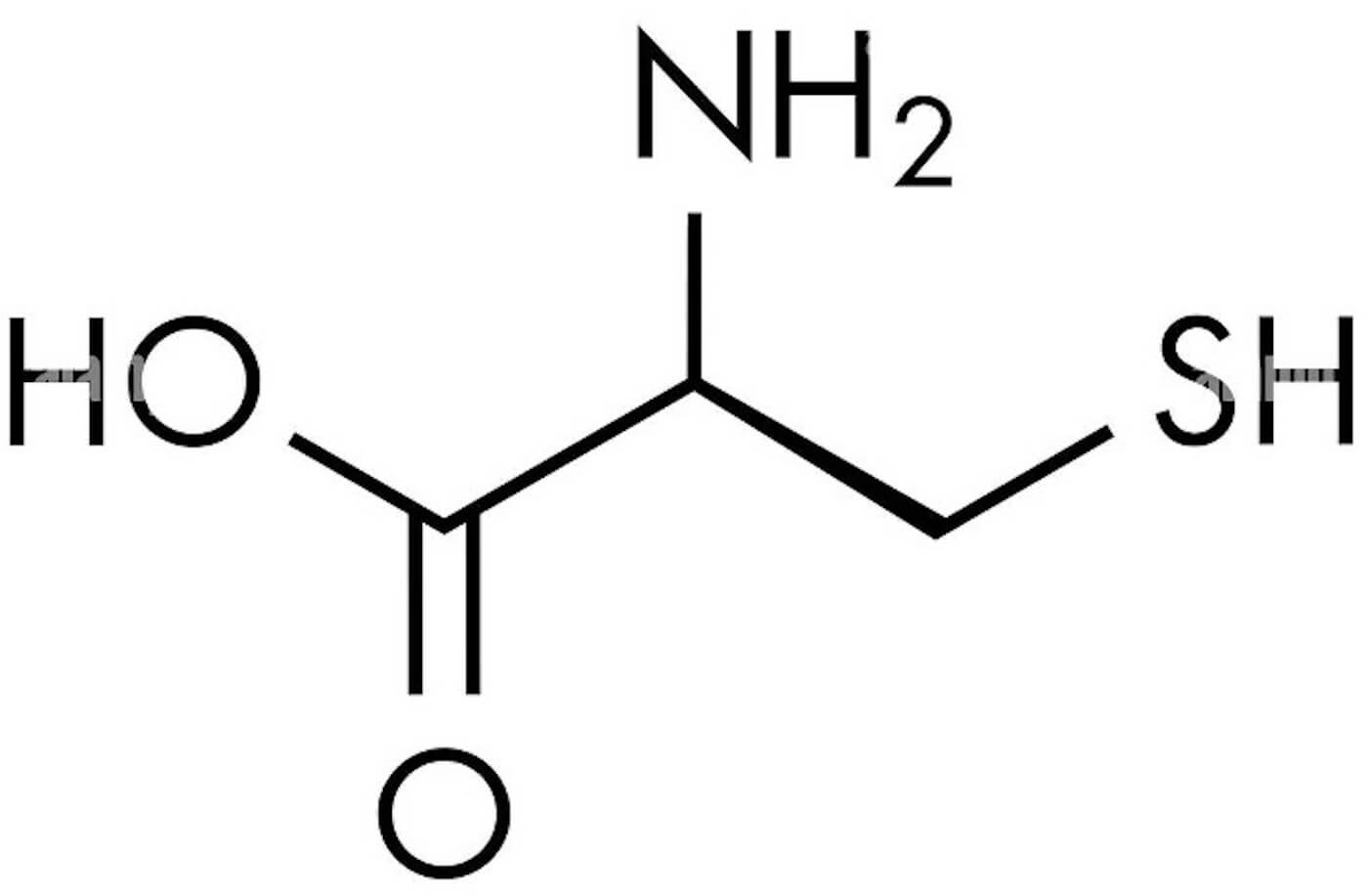 |
[25, 26, 27] | ||
| (10 mg/kg) | Liver recovery | |||||
| Flavonoids | Quercetin | Phenolic structures | Hepatoprotection | 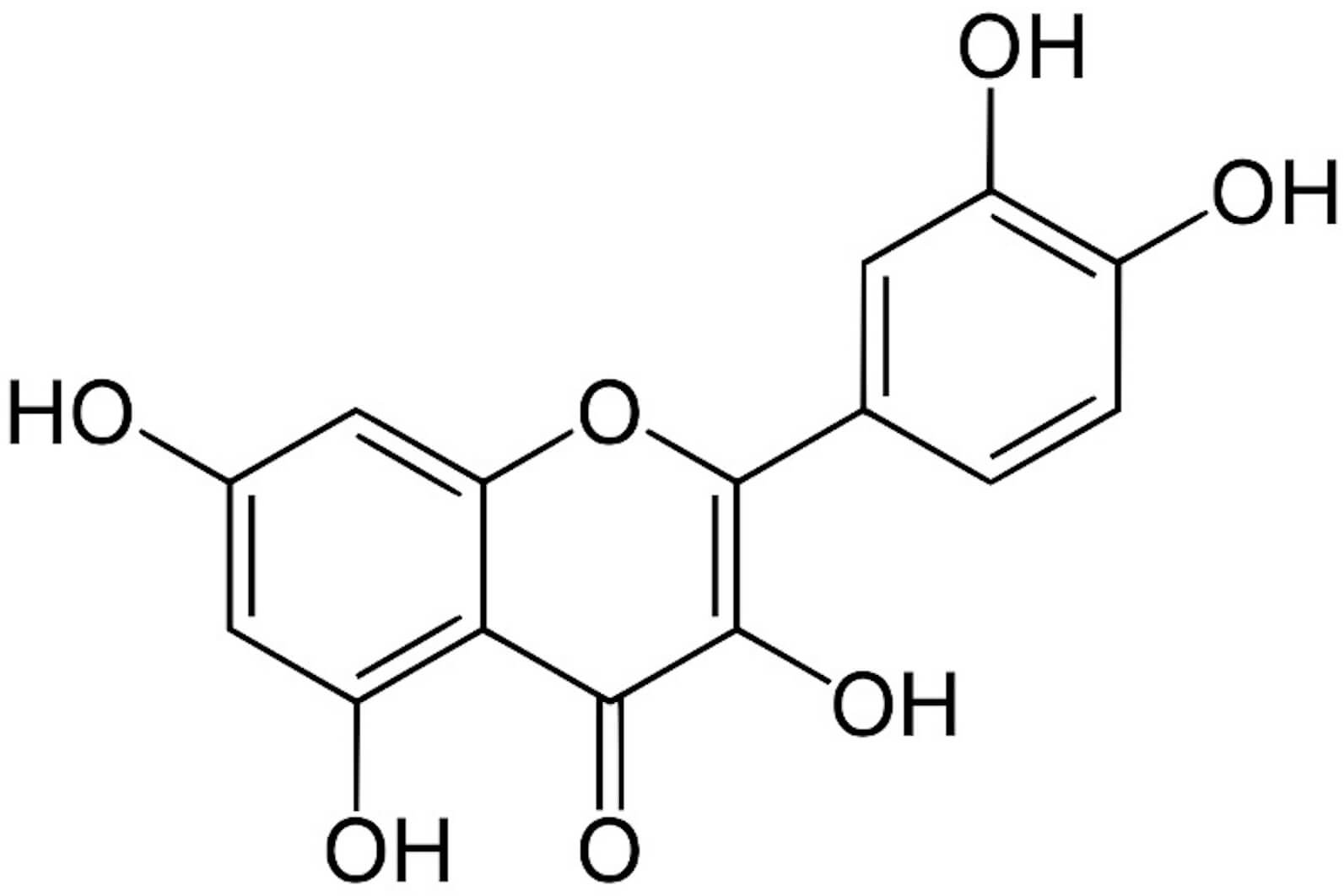 |
[28, 29, 30, 31, 32, 33, 34, 35] | |
| Aglycones, Glycosides, and Methylated derivatives | Antifibrotic activity | |||||
| (0.0015–150 mg/kg) | ||||||
| Anthocyanins | Protocatechuic acid | Aglycones | Prevention of T2DM | 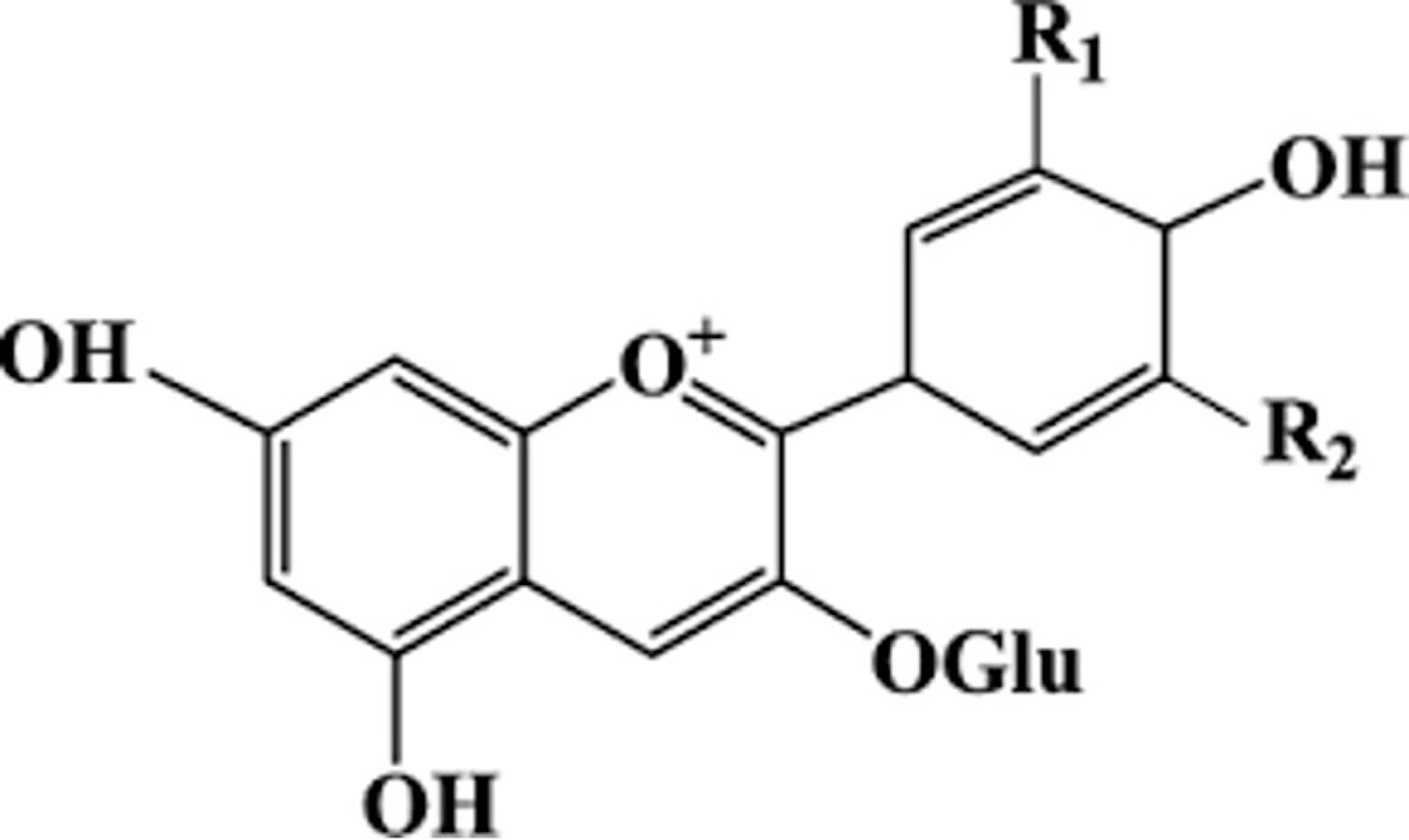 |
[12, 36, 37, 38, 39, 59, 60] | |
| (20–320 mg/kg) | Carbohydrate regulation | |||||
| Hepatic and pancreatic protective effects | ||||||
| Polyphenols | Bergamotin | Broad categories such as Glycosides, Aglycones | Liver detoxification |  |
[11, 40, 41, 42, 43, 44] | |
| Citrus lumia | Metabolic regulation | |||||
| Curcumin | (25 µg–100 mg/mL) | Improved glucose and lipid metabolism | ||||
| Antioxidant properties |
Although research has shown the beneficial effects of black radish, optimal concentrations and dose–response relationships have yet to be established. Determining these factors could help define a therapeutic dose that is effective without being harmful. Additionally, many of the abovementioned studies mentioned do not consider the female gender in their methodologies. Given that women are four times more likely than men to develop gallstones, inter-gender comparisons are an important aspect to address. Moreover, the methods of administering black radish and other drugs varied among the study groups. The detoxifying properties of black radish suggest that similar effects could potentially be observed with other drug treatments, underscoring the need for further research into the beneficial detoxifying effects of black radish in different contexts (Table 2, Ref. [8, 11, 15, 17, 19, 26, 28, 31, 32, 33, 34, 35, 41, 43, 47, 51]).
| Author/Year | Country | Population | Black Radish Compound | Study Design/Method | Outcomes/Observation |
| Evans et al., 2014 [8] | London | n = 20 | Spanish black radish | Pre-acetaminophen administration (1 g) | |
| healthy men | Spanish Black Radish (370 mg/wk) 3wk | ||||
| (Phase I and II enzymes) | |||||
| Jeon et al., 2020 [11] | Korea | Cell line RAW264.7 | Black radish extract | [25/50/100 and 200 µg/mL] | |
| (6–24 hrs) | |||||
| Hanlon et al., 2007 [15] | USA | Hepatocellular carcinoma cell line | SBR | SBR dry (0.3–3.0 mg) | |
| MIBITC | MIBITC [10 µM] | ||||
| Nagata et al., 2017 [17] | Japan | High-fat-diet Nrf2-KO murine model | Glucoraphanin | 135 mg/gr [0.31 mmol/g] | |
| 14 wk | |||||
| Tian et al., 2021 [26] | China | Obese mouse model | Sulforaphane | 10 mg/kg body weight | Regulation of glucose and lipid metabolism |
| 8 wk | |||||
| Sheu et al., 2023 [28] | Taiwan | High-fat-diet rat model and HepG2 cell line | WBJ | Murine: WBJ (0.5–2.0%) 8 wk | Promote fatty acid |
| BGE | Cell: BGE (2–6 mg/mL) 24 hrs | ||||
| Sinigrin (97.3%) | [50 µM glucosinolates] | ||||
| Gluconapin (2.67%) | |||||
| Chaturvedi and Machacha, 2007 [19] | Africa | Murine albino rat model | RSME | 80 and 120 mg/kg | |
| Paracetamol (100 mg/kg) | |||||
| 30 days | |||||
| Ma et al., 2025 [31] | EUA-China | Steatosis model HepG2 cells | Flavonoids | Naringenin, Morin and Silibinin [10 µM] | |
| Topiramate [44 µM] | |||||
| 72 hrs | |||||
| Chen et al., 2025 [32] | China | MASLD murine and HepG2 cell line models | Quercetin | Murine model: [35/70/140 mg/kg] 28 days | |
| Cell model: [5–80 µM] 48 hrs | |||||
| Nasr et al., 2025 [33] | Egypt | Fibrosis murine model | Quercetin | 50 mg/kg/day | |
| Sild 50 mg/kg/day | |||||
| PTX 20 mg/kg/day | |||||
| 4 wk | |||||
| Asghari et al., 2015 [35] | Iran | Pulmonary fibrosis rat model | Raphanus sativus L. var niger extract | 75–150 mg/kg | |
| 3 wk | |||||
| Cardamone et al., 2024 [41] | Italy | MASLD murine model | Polyphenolic fraction of bergamot | 50 mg/kg/day | Improve leptin, insulin and lipid metabolism |
| 14 wk | |||||
| Musolino et al., 2025 [43] | Italy | Hepatic cell lines (HepG2/LX2) | Citrus Lumia Risso extract (polyphenols) | 25/50/100 mg/mL | |
| 48 hrs | |||||
| Kocsis et al., 2002 [47] | Hungary | Male Wistar rats | Black radish root juice | 150 mL/kg | |
| [1:10] | 9 days | ||||
| Improved liver enzyme values | |||||
| Ahn et al., 2019 [34] | Korea | NAFLD murine models | Fermented black radish | Murine: 50/100/200 and 400 mg/kg (2 wk) | Hepatoprotective effect |
| Adipocyte cell line (3T3L1) | Silymarin (100 mg/kg) | Cells: 0.7–1.0% (6 d) | |||
| Castro-Torres et al., 2012 [51] | Mexico | Mouse model with lithogenic diet | Black radish juice | 1:100, 1:10 | |
| Ursodeoxycholic acid 0.5% | [0.1 mL/10 g] juice |
However, several limitations of the current evidence must be acknowledged. Most studies were conducted in preclinical settings, either in vitro or using animal models, which may not accurately reflect the complexities of MASLD in humans. Clinical trials are scarce, often involve small sample sizes, and exhibit heterogeneity in the types of black radish and preparations used (fresh root, fermented extracts, or isolated metabolites), as well as in the doses and methods of administration. These methodological inconsistencies restrict the comparability of results and the establishment of standardized therapeutic protocols. Additionally, outcome measures differ across studies, making it challenging to determine consistent clinical endpoints.
Overall, these constraints emphasize the need for well-designed, large-scale clinical studies to confirm efficacy, define optimal dosing, and ensure safety across diverse patient populations. Importantly, some findings remain inconsistent across studies, particularly regarding lipid metabolism outcomes and antifibrotic activity. These discrepancies underscore the importance of replication, the harmonization of study protocols, and a critical comparison of experimental designs to better establish the clinical potential of black radish in MASLD.
MASLD is the most common chronic liver disease worldwide, affecting more than 30% of the global population. At present, its treatment includes healthy lifestyle modifications, such as exercise and diet. Black radish possesses certain components—such as glucosinolates, anthocyanins, flavonoids, and polyphenols—which exert anti-inflammatory and antioxidant biological activities (Fig. 2). As such, this vegetable could be promising as an adjuvant treatment for liver detoxification and the prevention of disease.
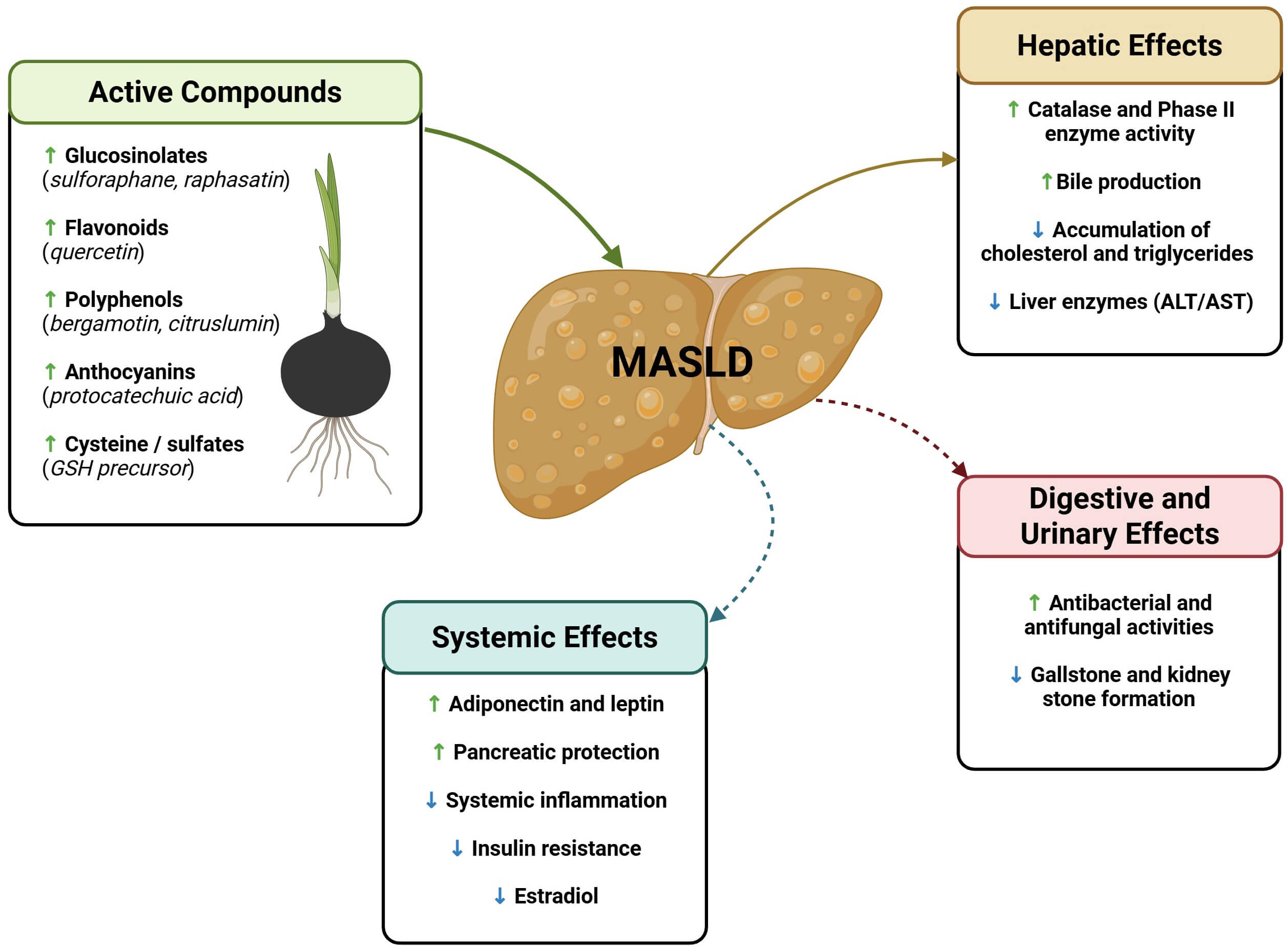 Fig. 2.
Fig. 2.
Hepatic, digestive, and systemic effects of black radish
(Raphanus sativus L. var. niger) in MASLD pathophysiology. Key
phytochemicals—including glucosinolates, flavonoids, polyphenols, anthocyanins,
and sulfur-containing compounds—exert multiple beneficial effects on hepatic
and metabolic functions. In the liver, black radish increases catalase and phase
II detoxification enzyme activity, promotes bile production, and reduces
cholesterol and triglyceride accumulation. In the digestive and urinary systems,
its antibacterial, antifungal, and antilithiasic effects support its overall
detoxifying profile. Systemically, it increases adiponectin and leptin levels,
protects pancreatic function, and reduces inflammation, insulin resistance, and
estradiol levels.
SAT: design concept, acquisition of information, and writing of the first draft manuscript; PSCF: figure elaboration and edition, review and correction of final manuscript version. MU: conceptualization, funding acquisition and critical revision; VJBB: guidance in the design and conceptualization, supervision, critical revision, writing—review & editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was funded by the Medica Sur Clinic & Foundation.
The authors declare no conflict of interest. This research was funded by the Medica Sur Clinic & Foundation, the institution where several authors are affiliated. The authors declare that the funder had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


