1 Department of Food Science and Technology, Uva Wellassa University, 90000 Badulla, Sri Lanka
2 Graduate School of Education, Dong-A University, 49315 Busan, Republic of Korea
3 Nutrinomics Lab. Co., Ltd., 49315 Busan, Republic of Korea
Abstract
Gluten-free diets (GFDs) are gaining popularity worldwide. Originally, GFDs were designed for individuals with gluten intolerance, including conditions such as celiac disease, non-celiac gluten sensitivity, gluten ataxia, or dermatitis herpetiformis. However, many individuals without gluten sensitivity have subsequently adopted this diet, leading to a growing demand for gluten-free food products. Meanwhile, the health risks associated with a long-term gluten-free diet (LTGFD) and the inherent related issues, such as nutrient imbalances, remain uncertain. Scientific studies have indicated that obesity, non-alcoholic fatty liver disease (NAFLD), dysbiosis, and poor mental health represent the most probable health risks associated with a LTGFD. Moreover, high fat and simple carbohydrate content, increased calorie content, and vitamin deficiencies, including vitamin B derivatives (B1, B2, B3, B5, B6, B7, B9, and B12), vitamin D, and minerals (Fe, Mg, Se, and Zn), are key components in a GFD that are at the helm of health issues due to a LTGFD. Anthocyanins, a class of flavonoids found predominantly in red, purple, and blue fruits and vegetables, may provide a promising solution to alleviate health risks associated with a LTGFD due to their inherent antioxidant, anti-inflammatory, neuroprotective, anti-diabetic, and anti-obesity properties. Therefore, this review critically examines in vivo, in vitro, clinical, and mechanistic studies to explore the potential of anthocyanin-enriched GFDs in alleviating the health risks associated with a LTGFD, highlighting their significant advantages over regular GFDs.
Graphical Abstract
Keywords
- anthocyanin
- dysbiosis
- gluten-free diet
- mental health
- non-alcoholic fatty liver disease
- obesity
A gluten-free diet (GFD) is a popular global diet ranked among the top five most prominent diets worldwide [1]. Although a GFD was typically used to diagnose gluten-related disorders, it has recently become a leading dietary trend based on contentious beliefs. The promotion of weight loss through a GFD by celebrities, the endorsement of popular media and books, and the recommendations provided by health and fitness communities have been identified as the root causes of this fashionista [2, 3]. Arslain et al. [4] have investigated the main motivations of non-gluten-sensitive GFD adherents. They found that the most prevalent beliefs were that a GFD promotes weight loss, is healthier, gluten-free (GF) products are more nutritious, gluten can cause diseases in non-sensitive individuals, and can alleviate acne. The idea was supported by book publications [3] and research that found accumulation of fat and stimulation of insulin hormone which triggers fat accumulation on long-term wheat consumption [5]. Furthermore, going GF made consumers omit processed high-caloric wheat products with high glycemic index such as bread, pasta, cookies, and snacks which promoted weight loss, reduced metabolic risks, and alleviated acne [6]. Moreover, a “gluten-free” label on packed food products was a key determinant of the purchase decision [7].
Gluten is a group of prolamins found in wheat and some other grains such as rye, barley, their cross-linked grains, and possibly in oats. Prolamins are the key components responsible for celiac disease, non-celiac gluten sensitivity, gluten ataxia, and dermatitis herpetiformis. To date, a GFD followed by all patients with gluten disorders has been identified as the most successful treatment [8]. Furthermore, GFD has continued to appeal to the regular community as a lifestyle choice in the absence of any gluten sensitivity. Based on the previous study by Kamiński et al. [1], the compliance rate of GFD was 11.11% of the relative study volume.
Just as if, several studies have identified issues of long-term gluten-free diet (LTGFD).
Considering the nutritional status of a GFD, several nutrient deficiencies and excesses have been identified through research. Siminiuc and Țurcanu [9] found that communities following a GFD commonly experience deficiencies in protein, fiber, vitamin A, vitamin D, vitamin E, vitamin B, vitamin B1, vitamin B2, vitamin B3, vitamin B5, vitamin B6, vitamin B12, folic acid, Ca, Fe, Mg, Zn, Mn, and Na in all age groups. Concurrently, adhering to a GFD is associated with high amounts of fat, sugar, and salt, consistent with the high glycemic index of GF processed food [10, 11, 12]. Long-term intake of GF food products can lead to various health defects such as hyperlipidemia, overweight and obesity, hepatic steatosis, increased risk of cardiovascular diseases, and deprived mental health, especially depression [13]. However, the dilemma of the beneficial health impact of a GFD on communities sensitive to gluten or with celiac disease and the detrimental health impacts on community adhering to a GFD without any diagnosed symptoms of gluten sensitivity (asymptomatic community) does not clearly address some health issues such as osteoporosis, infertility, growth failure, and delayed puberty in children.
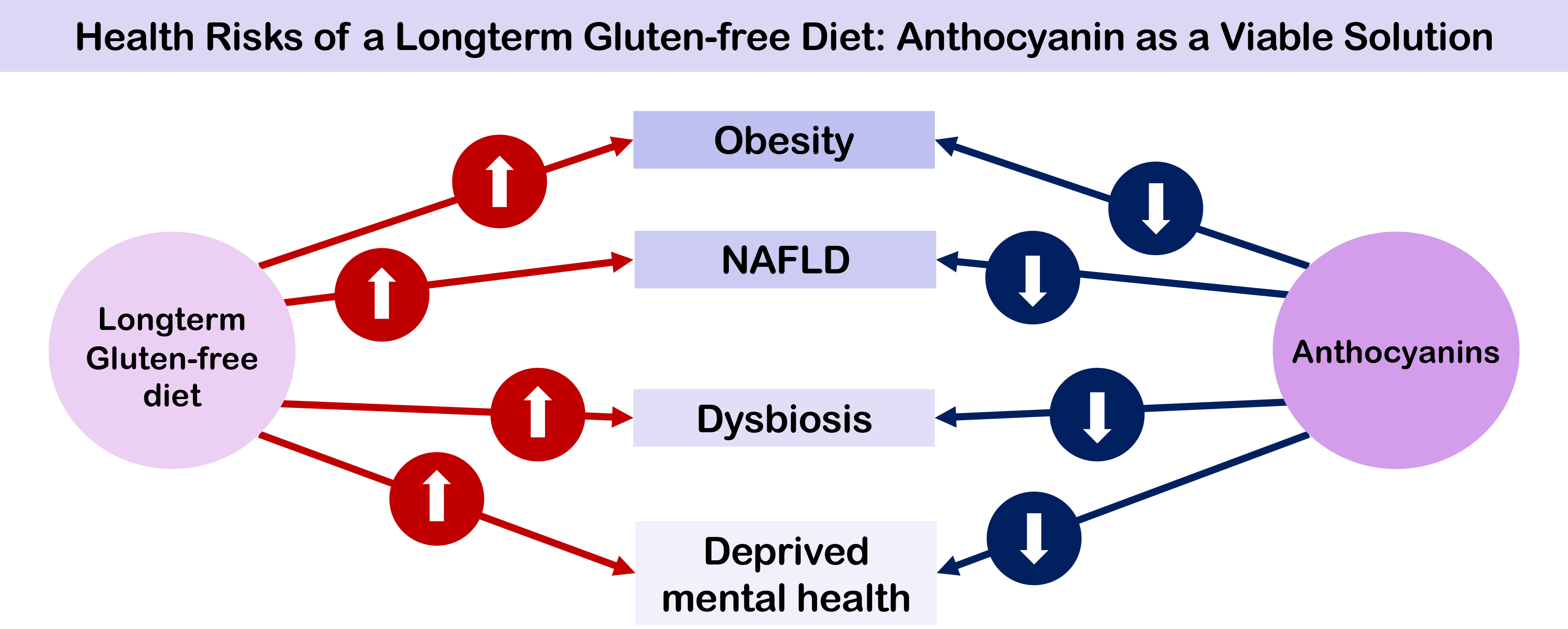
Anthocyanins are a class of flavonoids that function as plant pigments [14]. Flowers, dark-colored fruits, vegetables, and grains could be identified as rich sources of anthocyanins [15]. Usually, anthocyanins are predominant in red, purple, and blue fruits and vegetables such as mulberries, chokeberries, black elderberries, black currants, blackberries, blueberries, pomegranate, purple corn, purple sweet potato, red cabbage, black carrots, eggplants. Anthocyanins are a widespread group of compounds that promote a wide range of health benefits with their antioxidant [16], anticancer [17], anti-inflammatory activities [16], neuroprotective activities [18], anti-obesity [19], antidiabetic activities [20], antiatherosclerosis activity [21] and ability to prevent cardiovascular diseases [17]. Thus, although anthocyanins may not serve as a direct solution to address nutritional imbalances of a GFD, they may offer a potential solution for preventing or reducing the health complications associated with LTGFD. This review assesses the potential use of anthocyanins in alleviating the clinical effects of LTGFD. Fig. 1 demonstrates the potential of anthocyanins to address the health risks of a LTGFD. In this context, a GFD enriched with anthocyanins is always more beneficial than one lacking these bioactive compounds due to their proven efficacy.
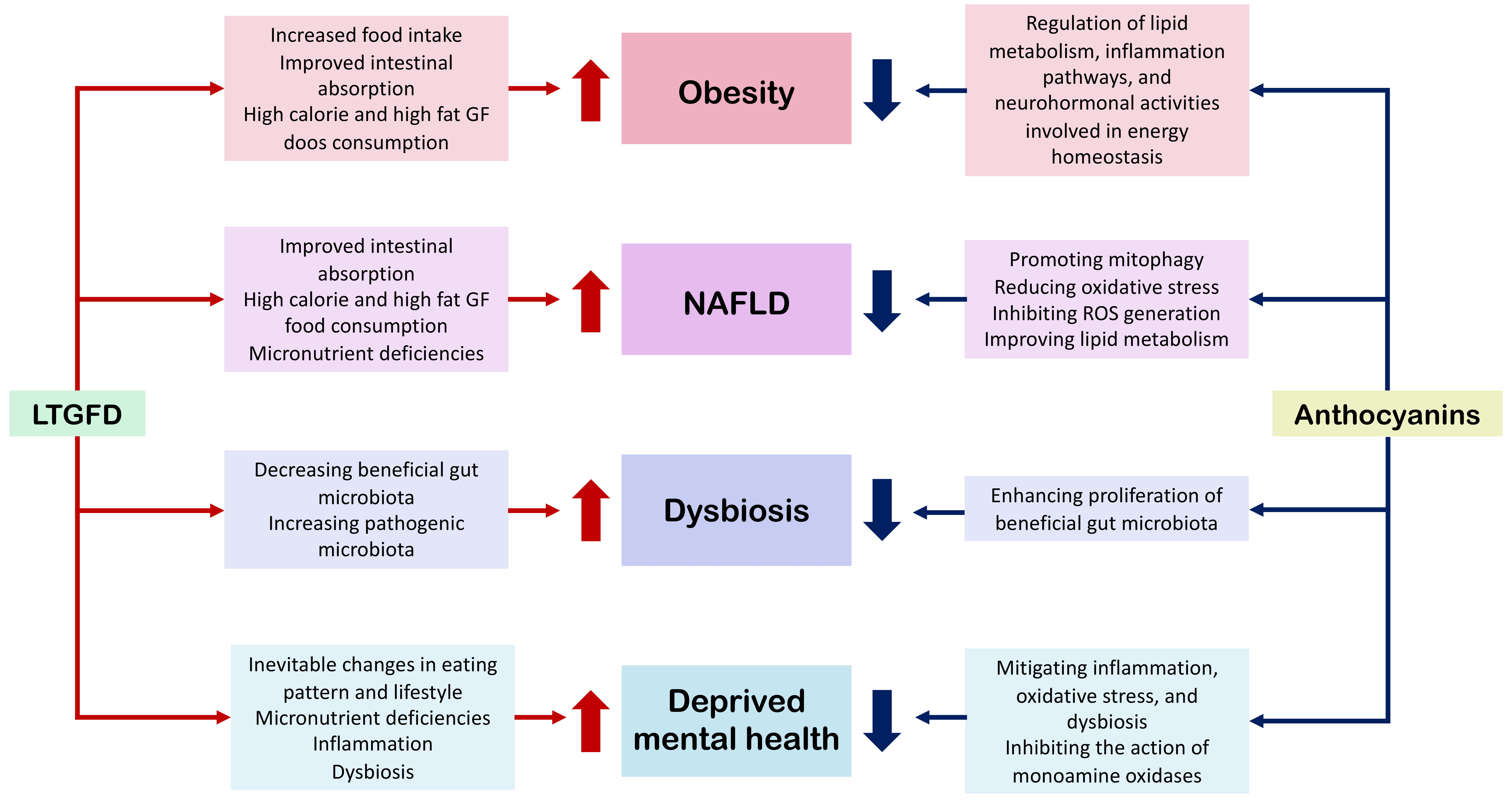 Fig. 1.
Fig. 1.
The possible health risks of a LTGFD and potential functions of anthocyanins to mitigate those health risks of LTGFD. LTGFD, long-term gluten-free diet; NAFLD, non-alcoholic fatty liver disease.
Several studies have highlighted various sources of anthocyanins and their beneficial effects on human health. While few review studies have highlighted the downside of the LTGFD intake [2, 22], there is a lack of solid evidence supporting the role of anthocyanins in addressing these issues. Hence, this review aims to explore the potential of anthocyanins to alleviate health risks due to LTGFD, thereby opening new avenues for future research. Recent literature linking GFD with obesity, overweight, non-alcoholic fatty liver disease, dysbiosis, mental health, and therapeutic effects of anthocyanins were comprehensively reviewed. To the best of our knowledge, this study is the first to provide an exclusive account of the potential use of anthocyanins to alleviate LTGFD-associated health risks.
Adhering to GFD is the most efficacious therapy for treating celiac disease, non-celiac gluten sensitivity, gluten ataxia, and dermatitis herpetiformis [23]. Nonetheless, it remains uncertain whether adopting GFD confers advantages among individuals who abstain from gluten without celiac disease or non-coeliac gluten sensitivity. Despite this uncertainty, the number of GFD devotees is constantly increasing contributing to the growing demand for GF products [3]. Nutrient imbalance is a core dispute that has been identified as related to a GFD through a study that can cause several health defects inadvertently. Salazar Quero et al. [24] reported a higher intake of lipids especially monounsaturated fatty acids, in a group of 69 celiac-diseased patients of 1–13 years. Barone et al. [25] also report a significantly higher intake of lipids by celiac-diseased adults representing both sexes. Melini and Melini [12] identified various nutritional deficiencies and excessive intake through a review done using 105 research articles. These include iron, calcium, selenium, zinc, and magnesium deficiencies, as well as low intake of vitamin B12, folate, vitamin C, and vitamin D deficiency [12]. Furthermore, there is evidence of low intake of dietary fiber and controversial protein intake as well as excess intake of fat and sugar. Similar conclusions were drawn in previous studies by Cardo et al. [26] and Perrin et al. [27]. The nutritional quality of GFD has always been questioned by the scientific community due to the higher fat composition, higher glycemic index, and high salt content of GF food on the market [28]. These imbalances can cause intricate health risks associated with LTGFD. This could be more intricate with the community adapted to processed GF food rather than for a natural GFD. So, it is important to understand that having a properly planned, nutritious, and equilibrated GFD with naturally whole foods and fortified products, can offer adequate nutrients and therapeutic benefits beyond celiac disease.
Overweight, non-alcoholic fatty liver disease (NAFLD), dysbiosis, and psychological defects are some identified possible health risks of a LTGFD. Though LTGFD could play a crucial role in these health risks, other factors such as genetic factors, biological determinants, behavioral factors, environmental factors, and sociodemographic factors interplay collectively for the cause of these health defects. Scientific research is scarce on the prolonged impact of following a GFD due to the majority of studies being conducted for brief intervals ranging from 6 months to 2–3 years. Additionally, the available literature predominantly concentrates on celiac disease gluten-sensitive groups with few studies focusing on the asymptomatic community of GFD adherents. It is important to distinguish between GFD adherents with gluten sensitivity and those who voluntarily follow a GFD as the long-term health impacts may differ significantly.
Overweight and obesity are global health crises that have led to a rise in the number of obese people worldwide and an increased risk of other non-communicable diseases [29]. A study has identified possible correlations between the GFD and overweight and obesity among individuals who adhere to a strict GFD. These interactions may be attributed to increased food intake, improved intestinal absorption due to intestinal mucosal injuries, and high calories and fat content in most processed GF food products [30].
A study conducted with 39 followers of a GFD revealed a general trend toward increased body mass index (BMI) after following the GFD. Specifically, followers with a normal BMI experienced a significant weight gain, and did not switch to the overweight or obese category within the approximately two-year study period [25]. Kabbani et al. [31] examined BMI and risk of obesity in communities with celiac disease and found a significant increase in BMI over an average of 39.5 years of study. Specifically, 21.2% of the cohort adhering to a strict GFD experienced an increase in BMI by more than two units, and 17% of the cohort became overweight or obese [31]. A study by Perrin et al. [27] on asymptomatic French adults found that individuals who partially avoided gluten or non-strict adherents of a GFD exhibited a higher energy intake than non-avoiders (regular community) and total avoiders. Ciccone et al. [32] and Kozioł-Kozakowska et al. [33] also observed a significant increase in BMI after a GFD.
Asri et al. [34] conducted a study to investigate the impact of a GFD on the body weight and BMI of Iranian patients across three time periods: after less than 6 months, after 6 months to 2 years, and after more than 3 years of GFD. The results showed that within the first six months of starting a GFD, 50% of the underweight participants had achieved normal weight. After 6 months to 2 years, 33.3% of the underweight individuals and 5.17% of those with normal weight had achieved normal and overweight status respectively. Of the participants who adhered to a GFD for over 2 years, 64.7% of the underweight, 6.25% of the normal weight, and 25% of the overweight transitioned to the normal weight, overweight, and obese BMI categories respectively. Nevertheless, a few cases experienced a reduction in body weight and BMI, whereas a significant number of subjects maintained their initial BMI level throughout the study. However, this study observed a positive correlation between a GFD, and an increase in body weight [34].
When studying children, Więch et al. [35] discovered a notable rise in body weight and BMI during their evaluation of the effects of a GFD on the body composition of children with celiac disease. Lionetti et al. [36] did not detect any substantial differences in energy intake, weight, height, BMI, physical activity, or sedentary behavior between children adhering to a GFD and non-adherents. Amirikian et al. [37] found no significant effect on the BMI of children, but a trend toward higher BMI after GFD adherence was observed in adolescents. The study suggested that the increased amenability of adolescents towards more convenient processed food over the health aspects was the most possible reason.
It is contentious whether a GFD contributes to overweight, and obesity as certain studies have concluded that such a diet has no detrimental or even favorable impact on BMI or body weight [38]. These effects can be influenced by other factors such as gender, age, occupation, type of residence, income, marital status, and educational level [39]. In addition, other contributors including lack of physical activity, sedentarism, genetic predisposition, pathophysiological conditions (such as hormonal imbalances, and physical disabilities), and medications (such as diabetic treatments, steroids, and antidepressants) and psychological health are strongly correlated with overweight and obesity. While it is a proven fact that GF processed food products contain excess fat and sugar, following a GFD poses a clear vulnerability to develop or succor overweight and obesity over a lifetime, either independently or in conjunction with other contributing factors [34]. Importantly, maintaining a proper GFD rich with GF whole food over processed GF alternatives combined with adequate physical activities could significantly reduce the risk of obesity.
Non-alcoholic fatty liver disease (NAFLD) is a set of conditions linked to metabolic dysfunction of the liver and is recognized as the most widespread chronic liver disease affecting about 25% of the adult population worldwide [40]. NAFLD is closely associated with metabolic syndrome in a bidirectional way [41]. Therefore, NAFLD has been identified as another risk factor of LTGFD exposure. This was confirmed by Tovoli et al. [42], who conducted a human clinical study in individuals with coeliac disease after GFD and controls. The demographic and clinical factors such as age, gender, BMI, diabetes, cholesterol, and triglycerides were matched across the study groups. The study found a positive correlation between the risk of NAFLD and communities with celiac disease especially among overweight patients with celiac disease. Therefore, Tovoli et al. [42] reported a three-fold higher risk of NAFLD among patients with celiac disease who followed a GFD compared to the control group. Reilly et al. [43] also reported an increased risk of NAFLD among patients with celiac disease compared with the regular population in a study conducted over 15 years. Age, sex, calendar year, and county of residence at the time of biopsy were matched. According to the study, the risk of NAFLD is the highest within the first year after diagnosis, and then reduces in magnitude, but persists beyond 15 years of diagnosis [43]. Although the study revealed patients with celiac disease following a GFD, no relation between GFD and NAFLD was made. As for the study conducted by Imperatore et al. [44], 23.9% of metabolic syndrome and 37.2% of hepatic steatosis occurrences were found in a cohort of 301 patients with celiac disease after one year of GFD treatment. During the study, the dietary quality was maintained through a professional dietitian with a patient-tailored approach based on the daily calorie requirements [44].
The elevated prevalence of NAFLD following the adoption of a GFD among those
with gluten sensitivity has been attributed to the absorption of more nutrients
due to mucosal recovery post-GFD as well as consumption of high-calorie foodstuff
that are rich in fats and simple carbohydrates [45]. These suggestions may be
more accurate with long-term exposure to a GFD. Excessive fatty acids consumption
through a GFD can lead to adipocyte exhaustion and liver damage progress through
inhibition of adiponectin and stimulation of production of proinflammatory
adipokines, leptin, resistin, TNF-
Vitamin and micronutrient deficiencies resulting from a GFD in communities with celiac disease have an impact on liver health. Vitamin A, D, E, B12, B6, folate (vitamin B9), and Zn deficiencies are especially significant with notable effects on the liver. However, these nutrients insufficiencies have been identified in celiac disease communities after adopting a GFD [47]. Vitamin A, an antioxidant in the liver, induces oxidative stress at insufficient levels. Vitamin A deficiency is associated with the progression of cirrhosis and the appearance of hepatic encephalopathy, a brain dysfunction condition caused by toxins produced due to liver malfunctioning [48]. Vitamin D deficiency is also associated with cirrhosis despite causing chronic liver disease. These effects can lead to fibrosis, resulting in infections, hepatocellular carcinoma, and mortality. Vitamin D deficiency is also associated with metabolic syndrome and NAFLD is considered as the hepatic expression of metabolic syndrome. However, research has demonstrated that vitamin D can enhance liver health through its anti-proliferative, anti-inflammatory, and antifibrotic properties. In contrast, vitamin D deficiency is associated with the progression of chronic liver diseases such as NAFLD [49]. Vitamin E is a vital nutrient with antioxidant and immunomodulating properties. Vitamin E deficiency can induce oxidative stress in the liver leading to the progress of liver inflammations [50]. Vitamin B12 is another important vitamin that plays a vital role in the maintenance of liver health. Vitamin B12 deficiency concomitant with pernicious anemia, an autoimmune gastritis condition can lead to liver damage and cirrhosis [51]. On the contrary, vitamin B12 deficiency inhibits the synthesis of S-adenosylmethionine, the main cellular antioxidant in hepatocytes where liver damage and differentiation can occur in the absence [52]. Furthermore, vitamin B12 deficiency can lead to reduced liver detoxification and restoration of damaged hepatocytes, and aid in the progression of chronic liver disorders, including cirrhosis and NAFLD [51]. Folate has a significant effect on liver health, and low serum folate levels can lead to NAFLD [53]. Vitamin B6 is a vital antioxidant that can cause oxidative stress in the liver during deficiency by limiting glutathione synthesis [54]. Vitamin B6 deficiency also leads to endoplasmic reticulum stress and de novo lipid synthesis resulting in increased lipid deposition in the liver and NAFLD [46]. Besides vitamin deficiencies, the scarcity of minerals such as Zn affects liver health. Zn deficiency also causes endoplasmic reticulum stress and hepatocyte apoptosis. In contrast, Zn deficiency induces insulin resistance, enhancing metabolic syndrome and NAFLD [55]. However, studies have revealed that restoring recommended levels of vitamins A, D, E, B6, B9, B12, and Zn can effectively combat NAFLD [46, 48, 52, 53, 56].
Accordingly, a GFD can expose consumers to a higher risk of liver-related disorders including NAFLD during long-term consumption. Focusing on the intake of gluten-free whole food over gluten-free processed options may help to minimize these risks. However, it is difficult to establish that adherence to a LTGFD is the sole cause of NAFLD following the adoption of a GFD.
Symbiotic bacteria colonize the human body, with the skin, mouth, gut, and vagina being the primary sites of colonization. Numerous bacteria, archaea, microeukaryotes, and viruses inhabit the human microbiome. The highest proportion of this microbiota inhabits the gut, which is estimated to have ten trillion symbionts. The microbiome, which is 150 times larger than the human genome, has evolved from collective microbial genes. The microbiome can be affected by the genotype of the host, the environment, and the diet [57].
The term ‘eubiosis’ is used to describe a well-balanced intestinal microbial ecosystem whereas per contra is described as ‘dysbiosis’. The balance of the intestinal microbiota or eubiotic state is maintained by potentially beneficial species of microbiota, whereas potentially pathogenic species are also present in small numbers. Potentially beneficial bacteria were primarily from two bacterial phyla, Firmicutes and Bacteroidetes (more than 90%) and potentially pathogenic species, such as those from the phylum Proteobacteria (Enterobacteriaceae), were present at low levels [58].
The gut microbiome is a symbiotic, ubiquitous organ that performs different important functions in the human body. A few key functions of the gut microbiome include, but are not limited to production of bioactive metabolites that aid in the regulation of immunity and defense against pathogens [59], the production of bioactive compounds for energy homeostasis [60], the metabolism of nutrients and other food components [61], regulation of gut epithelial development [62], maintenance and protection of normal gut physiology and gut health [62], influence on neural development, cognition, behavior, and mental health through interactions with the gut-brain axis [63], regulation of gut-brain communication in immune-related neurological disorders [63], and contribution to the formation of fecal bulk, and dilution and elimination of toxins through fecal bulk [64]. Dysbiosis influences these functions and can cause other conditions including inflammatory bowel disorders and metabolic disorders like obesity and type II diabetes. Dysbiosis may also precede conditions such as Parkinson’s disease and facilitate the development of various other immune-mediated, metabolic, neurological, and psychiatric disorders [65].
Diet is a major cause of dysbiosis. The composition of a diet such as amounts of fat, sugar, proteins, and fiber affects the intestinal gut microbiota composition. Unbalanced diets, specifically rich in animal protein, saturated fats, and low dietary fiber, have been shown to decrease beneficial bacteria while increasing potentially harmful bacteria [66]. The rapid absorption of saturated fats and sugars in the small intestine, leaving no substrate for microbiota to ferment, and alteration of gut ecosystem promotes dysbiosis [67, 68]. As for the research, GFD also causes dysbiosis in GFD adherents. De Palma et al. [69] analyzed the effects of GFD on gut microbiota in 10 healthy individuals over one month. Results showed a decrease in beneficial gut microbiota, such as Bifidobacterium, Bifidobacterium longum, and Lactobacillus, and an increase in pathogens such as Escherichia coli and total Enterobacteriaceae [69]. The decreased availability of polysaccharides in the GFD, which act as a habitat for the gut microbiota is believed to be the primary factor behind the decline in Bifidobacterium and Lactobacillus levels [69]. In a study conducted by Bonder et al. [70], the gut microbiome of 21 healthy individuals were tested after undergoing a GFD for four weeks, followed by a habitual diet for an additional four weeks. Age, gender, BMI, nationality, and pre-antibiotic treatments of the study group were considered. The results showed a significant variation in bacterial diversity, including a significant reduction of Veillonellaceae, Ruminococcus bromii, and Roseburia faecis and a significant increase in Victivallaceae, Clostridiaceae, Coriobacteriaceae, and Slackia when comparing bacterial counts in different intervals [70]. Hansen et al. [71] conducted a crossover study by introducing a low-gluten diet (2 g gluten per day) for 8 weeks, and a high-gluten diet (18 g gluten per day) for another 8 weeks, with a regular diet (12 g gluten per day) for 6 weeks as a washout period in 60 adults with no identified diseases. Consequently, the low-gluten diet was more proficient for dysbiosis than the high-gluten diet, with a diminished relative and absolute availability of Bifidobacterium spp. [71].
Morrison et al. [72] conducted an animal-based study introducing a GFD into wild-type mice. A significant deviation in the gut microbiome of mice was observed compared to wild-type control mice fed a regular diet. A decrease in the Shannon diversity index with a significantly lower number of bacterial species was observed compared to the control group. Furthermore, Atopobiacae, Streptococcaceae, and Erysipelotrichaceae were prominent in GFD mice, whereas the Lactobacillaceae family and unidentified Lactobacillales were characterized in mice fed a normal diet. Despite this, the Actinobacteria count was significantly increased by introducing a GFD to wild-type mice [72].
Di Cagno et al. [73] conducted a study to investigate the microbiome of 19 children with celiac disease who had followed a GFD for 2 years compared to children without celiac disease. Duodenal biopsies and fecal samples revealed that cell counts of Lactobacillus, Enterococcus, and Bifidobacterium were significantly lower, whereas levels of pathogenic microbes such as Bacteroides, Staphylococcus, Salmonella, Shigella, and Klebsiella like were significantly higher in children with celiac disease after a GFD than in healthy controls. Furthermore, Di Cagno et al. [73] highlight the inability of a GFD to rehabilitate eubiosis even after two years of adherence in patients with celiac disease. Dysbiosis due to a GFD is also proved by Zopf et al. [74].
While diet is a major intrinsic factor affecting the gut microbial composition, various other factors also play a significant role, including physiological, genetic, mental, environmental, and lifestyle factors. The causative dietary factors include alcohol consumption, food additives, and dietary changes [75]. Physiological influences comprise the use of antibiotics and other medications [76], infections [77], gene mutations, chronic diseases such as obesity [78] and diabetes [79], and aging [80]. Mental health factors such as psychological stress and anxiety also lead to dysbiosis [81]. Environmental influences include hygiene practices [82], chemical exposure [83], and early-life factors such as cesarean delivery, formula feeding instead of breastfeeding, and early exposure to antibiotics [84]. Additionally, lifestyle factors such as lack of sleep [85] and nicotine use [86] contribute to dysbiosis.
GFD is the only available treatment for coeliac disease and non-coeliac gluten sensitivity. However, adherence to a strict GFD is challenging due to inevitable changes in eating patterns, lifestyle, and daily routine activities. Consequently, subjects may encounter stressful and difficult-to-accept conditions, followed by psycho-social-behavioral disorders [2].
Celiac disease is correlated with behavioral and psychiatric disorders such as anxiety, depression, dysthymia, attention-deficit hyperactive disorders, social adjustment disorder, sleep disorders, mood disorders, addictive and neurocognitive disorders, feeding and eating disorders, autistic spectrum disorders, schizophrenia, learning disabilities, and suicide attempts, among which depression found to be strongly correlated [87, 88]. A strict GFD may worsen these mental health issues and a study shows that community with celiac disease has a higher suicide rate compared to the general population [89]. Furthermore, celiac disease is associated with a higher intake of psychiatric drugs [88]. Despite the nutrient and vitamin deficiency associated with LTGFD, potential causes of mental health deprivation from adherence to the diet, including immune dysregulation, cerebral hypo perfusion, and stress have been proposed but not yet scientifically proven [2].
A poor diet is associated with poor psychiatric health. In contrast, LTGFD is associated with nutrient deficiencies, such as vitamin and mineral deficiencies which play vital roles in maintaining physical and mental health. Vitamin B derivatives are essential nutrients that improve brain health and mood and increase the risk of depression in their dearth [90]. Vitamin B deficiency can also affect memory, and cause cognitive impairment, and dementia [91]. Vitamin B1 deficiency is also directly associated with anxiety, mood disorders, and depression [92]. Vitamin B2 or riboflavin deficiency can affect fetal development causing congenital impairments [93] and poor cognitive outcomes, depression, and adverse personality changes due to changes in the central nervous system in adults [94]. Vitamin B3 (niacin) deficiency is strongly associated with depression and niacin supplementation has reduced depression symptoms. Furthermore, schizophrenia, manic depression, benign depression, anxiety, and pellagra are associated with niacin shortage. Vitamin B5 or pantothenic acid, also called ‘anti-stress factor’ causes depression and fatigue when deficient [95]. Vitamin B5 is widely used to treat anxiety, depression, and mood disorders [96]. Vitamin B6 or pyridoxine is vital for the regulation of anxiety and depression, and its shortage results in high serum homocysteine levels. Higher levels of homocysteine are associated with migraine, anxiety, and depression [97]. Vitamin B7 (biotin) is also used to treat severe depression; however, its mechanisms of action have not yet been scientifically proven [98]. Vitamin B9 (folate) deficiency is directly related to depression and behavioral disorders [99] where sufficient folate intake leads to antidepressant activities, such as the regulation of serotonin and noradrenalin receptors [100]. Vitamin B12 or cobalamin deficiency can cause acute depression, suicidal behavior, mood disorders, poor cognition, mental fatigue, mania, psychosis, and severe agitation. Vitamin B12 is widely used in the treatment of psychiatric disorders [101]. Besides vitamin B, vitamin D is also crucial for mental health. Vitamin D plays an indispensable role in neurocognitive function and functions as an antidepressant via gene transcription in the brain, in which serotonin synthesis induces and increases serotonin in the central nervous system [102]. Vitamin D deficiency is associated with a higher risk of depression, anxiety, autism, and dementia [103]. Vitamin D deficiency has also been associated with sleep and mood disorders [104].
Besides vitamins, minerals, such as iron, magnesium, selenium, and zinc play an important role in maintaining psychiatric health. Iron deficiency is responsible for mental, social, behavioral, emotional, neurophysiological, and neurocognitive impairments in all age groups [105]. Depression, anxiety, sleep disorders, and psychotic disorders are associated with iron deficiency among adults and affect the hippocampus, corpus striatum, and precise neurotransmitters such as serotonin, noradrenaline, and dopamine [106]. Magnesium deficiency causes anxiety, depression, and attention deficit hyperactivity disorder [107]. Dysregulation of the hypothalamic-pituitary-adrenal axis, increased calcium ions in the brain, and increased inflammatory responses have been suggested as possible mechanisms [108]. Selenium deficiency is directly correlated with depression, mood disorders, and autism and selenium supplementation improves symptoms of depressive disorders [107, 109]. Proposed explanations include the ability of selenium to induce thyroid function and modulate hormonal activity and neurotransmitters in the brain (serotonin, dopamine, noradrenalin) [108, 109]. Zinc deficiency leads to neuropsychological disorders, anxiety, and depression in severe deficit conditions. Increased cortisol levels, decreased neurogenesis and neural plasticity, and downregulated glutamate homeostasis are believed to contribute to zinc deficiency-associated depression [108]. Because all the micronutrient deficiencies are held by GF food products, a LTGFD can easily cause severe micronutrient deficiencies and their consequences such as an increased risk of mental disorders.
Inflammation can cause anxiety, stress, and depression [110]. Linking depression
with inflammation, proinflammatory cytokines such as IL-6 and TNF-
The gut microbiota regulates the gut-brain axis, through which they play functional roles in the regulation of brain functions, including mood, behavior, and emotional processing [112]. Research has found that dysbiosis is strongly related to anxiety and depression [113] where dysbiosis occurs due to a GFD [71]. Dysbiosis is also associated with mood disorders and autism [114].
However, it is important to understand that a wide range of other causative factors may affect mental health beyond following a GFD. These include genetic predispositions [115], hormonal imbalances [116], neurochemical changes [117], chronic stress, social isolation, traumatic experiences, and pre-existing psychological disorders [118]. Other determinants such as environmental stress factors, socioeconomic status, and lifestyle factors such as poor sleep quality and lack of physical activity can affect the mental well-being of a person [115, 118]. As mental health depends upon a complex interplay of numerous biological, psychological, and social factors, a GFD does not fully account for the mental well-being of a GFD adherent. Regardless, it is proven that adhering to a GFD can significantly affect mental well-being.
To date, more than 700 anthocyanins have been identified as they have garnered scientific interest due to their therapeutic significance beyond their roles as plant pigments. Extensive research including in vitro, in vivo, and clinical studies recognize anthocyanins as bioactive compounds with potent antioxidant and anti-inflammatory properties that foster a wide range of biological effects. In regards, anthocyanins can be identified as a potential solution that can alleviate the above-discussed health risks due to a LTGFD. Although not a direct remedy for nutrient imbalances associated with a GFD, anthocyanins may serve as a promising strategy to reduce the risk of discussed health risks due to a LTGFD.
Anthocyanins are a highly effective bioactive compound in combating several health defects including overweight and obesity [119]. Studies have recently shown the potential benefits of anthocyanins in mitigating obesity risk. Three potential protective effects of anthocyanins against obesity were identified: regulation of lipid metabolism, regulation of inflammatory pathways, and regulation of neurohormonal activities involved in energy homeostasis.
Adipose tissue is functional for energy storage as lipids for future consumption
[120]. With continuous storage of lipids in adipose tissue, they expand and
generate anorexic signals that inhibit further energy intake [121]. However, when
the adipose tissues reach the limit of lipid storage, hyperlipidemia occurs.
Furthermore, they trigger the production of proinflammatory cytokines and the
activation of leukocytes [122]. With their potential to reduce plasma cholesterol
levels and to improve adipocyte function, anthocyanins are functional compounds
that play a role in lipid metabolism regulation [21]. Anthocyanins’ primary
function in the regulation of lipid metabolism can be attributed to decreasing
accumulation of fat and serum lipids, activation of 5′ adenosine
monophosphate-activated protein kinase (AMPK) regulator [123], suppression of
fatty acid synthase (FAS), triggered expression of lipid-metabolism related genes
such as peroxisome proliferator-activated receptors
AMPK is a mammalian energy sensor that is activated when the cellular ATP levels
decrease. Its responsibilities include lipid synthesis, lipolysis, and fatty acid
oxidation [126]. Upregulation and activation of AMPK regulators induce the
oxidation of fatty acids to generate ATP [127]. PPAR
| Source of anthocyanin | Prominent parent anthocyanins | Model | Dose of Anthocyanin | Treated period | Effects | Reference |
| Aronia melanocarpa | Cyanidin | Male C57BL/6J mice | 50 (min.)–200 (max.) mg/kg body weight per day | 8 weeks | Dose dependent inhibited body weight gain | [131] |
| Reduced weights of white fat tissue | ||||||
| Suppressed adipogenesis | ||||||
| Decreased levels of enhancer binding protein |
||||||
| Berry rich anthocyanin supplement | - | Human clinical study | 320 mg/day | 4 weeks | Decreased blood cholesterol, triglycerides, and LDL | [124] |
| Enhanced expression of PPAR |
||||||
| Blackberry | - | Male C57BL/6J mice | 200 mg/kg food | 12 weeks | Inhibited body weight gain | [132] |
| Blueberry | Reduced serum and hepatic lipid levels | |||||
| Black chokeberry | - | 3T3-L1 preadipocytes | 10–50 µg/mL chokeberry and cranberry, 5–25 µg/mL of promegranate | 14 days | Inhibited lipid accumulation | [133] |
| Cranberry | Increased lipolysis | |||||
| Pomegranate | ||||||
| Blackcurrant | - | Male SPF C57BL/6J mice | 150 mg/kg body weight per day | 14 weeks | Reduced body weight | [134] |
| Reduced levels of hyperlipidemia | ||||||
| Black soybean testa | Cyanidin | Human clinical study | 2.5 g/day | 4 weeks | Decreased abdominal fat | [126] |
| Delphinidin | Decreased triacylglycerols, LDL, and non-high density lipoprotein cholesterol | |||||
| Petunidin | ||||||
| Blueberry (Vaccinium ashei) | Malvidin | HepG2 cell line | 5 µg/mL | 24 hours | Decreased fatty acid and cholesterol synthesis | [135] |
| Male C57BL/6J mouse | 100–400 mg/kg body weight per day | 5 weeks | Reduced free fatty acid circulation in the blood | |||
| Amelioration of hyperglycemia and hyperlipidemia | ||||||
| Activation of AMPK regulator | ||||||
| Cherry | Cyanidin | C57BL/6J mice | 200 mg/kg food | 16 weeks | Attenuated weight gain | [136] |
| Pelargonidin | Decreased total cholesterol and triglycerides level | |||||
| Mulberry | Cyanidin | 200 mg/kg food | ||||
| Pelargonidin | ||||||
| “Moro” Blood orange (Citrus sinesis) | - | Human clinical study | 400 mg/day | 6 months | Reduced body weight (4.2% mean overall weight loss after 6 months of study) | [137] |
| Reduced hip and waist circumference | ||||||
| Reduced BMI | ||||||
| Reduced total fat mass (android fat, gynoid fat, visceral fat, subcutaneous fat) | ||||||
| Purple maize pericarp | Pelargonidin | Male C57BL/6J mice | 200–500 mg/kg body weight per day | 12 weeks | Regulated AMPK pathway | [138] |
| Cyanidin | Reduced adipogenesis | |||||
| Raspberry | Malvidin | C57BL/6J mice | 200 mg/kg food | 12 weeks | Reduced body weight gain | [139] |
| Cyanidin | Reduced serum lipid levels | |||||
| Pelargonidin | ||||||
| Peonidin | ||||||
| Vitis vinifera L. grape skin | Peonidin | Male C57BL/6J mice | 200 mg/kg body weight per day | 12 weeks | Lowered total cholesterol, LDL, and VLDL | [140] |
| Petunidin | Increased expression of AMPK | |||||
| Malvidin | ||||||
| Vitis coignetiae Pulliat fruit | - | Murine 3T3-L1 adipocytes | 50–200 µg/mL per day | 72 hours | Activated AMPK signaling pathway | [125] |
| Suppressed PPAR |
||||||
| Inhibited expression of adipocyte fatty acid-binding proteins, leptin, and FAS |
PPAR
Obesity is associated with chronic low-grade inflammation, called
“meta-inflammation”. In response, proinflammatory cytokines are produced by
innate immune cells in adipose tissues. Interleukin-6 (IL-6),
interleukin-1
Anthocyanin as an anti-inflammatory agent, can interfere with meta-inflammation. Therefore, anthocyanins decrease the expression of proinflammatory markers mentioned above [119]. Table 2 (Ref. [122, 124, 132, 136, 138, 139, 146, 147, 148, 149, 150, 151, 152]) summarizes the various sources of anthocyanins researched for their effectiveness in reducing proinflammatory markers and presenting positive outcomes.
| Source of anthocyanin | Prominent parent anthocyanin | Model | Dose of Anthocyanin | Treated period | Effects | Reference |
| Aronia berry | - | Male BALB/c mice | 10–100 mg/kg body weight per day | 10 days | Decreased levels of IL-1 |
[146] |
| Berry rich anthocyanin supplement | - | Human clinical study | 320 mg/day | 4 weeks | Downregulation of TNF- |
[124] |
| Bilberry | - | Adult male Wistar rats | 200 mg/kg body weight per day | 7 days | Reduced proinflammatory markers (IL-6, TNF- |
[122] |
| Blackberry Blueberry | - | Male C57BL/6J mice | 0.48 g/kg food | 12 weeks | Reduced expression levels of IL-6 and TNF- |
[132] |
| Bilberry (Vaccinium myrtillus) | - | Human clinical study (diabetic patients) | 320 mg/day | 4 weeks | Reduced proinflammatory molecules IL-6, IL-8, and TNF- |
[147] |
| Blackcurrant (Ribes nigrum) | ||||||
| Blueberry | - | Wistar rats | 10% dry blueberry power in diet | 8 weeks | Normalized TNF- |
[148] |
| Improved markers of insulin sensitivity | ||||||
| Cooked glutinous purple rice | - | Male Wistar rats | 500 mg/kg body weight per day | 15 weeks | Decreased levels of IL-6 and IL-1 |
[149] |
| Elderberry (Sambucus ebulus) | - | Human clinical study | 200 mL infusion per day | 4 weeks | Decreased levels of IL-6, IL-1 |
[150] |
| Passovia ovata | - | BALB/c mice | 125–500 mg/kg body weight per day | - | Decreased IL-6, IL-1 |
[151] |
| Purple maize pericarp | Pelargonidin | Male C57BL/6J mice | 200–500 mg/kg body weight per day | 12 weeks | Reduced levels of TNF- |
[138] |
| Reduced adipose inflammation | ||||||
| Raspberry | Malvidin | C57BL/6J mice | 200 mg/kg food | 12 weeks | Downregulated proinflammatory markers IL-6 and TNF- |
[139] |
| Cyanidin | ||||||
| Pelargonidin | ||||||
| Peonidin | ||||||
| Mulberry | Cyanidin | C57BL/6J mouse (4 weeks older) | 200 mg/kg food | 16 weeks | Reduced expression levels of IL-6 and TNF- |
[136] |
| Pelargonodin | ||||||
| Cherry | Cyanidin | |||||
| Pelargonidin | ||||||
| Tart cherry (Hungarian sour cherry VN1 variety) | Cyanidin | Male C57BL/6J mice | 60 mg/kg body weight per day | 6 weeks | Reduced levels of IL-6 in plasma | [152] |
| Decreased plasma leptin level compared to high fat diet induced obese mice control |
IL, interleukin; TNF-
Insulin resistance and hyperinsulinemia are implicated in triggering obesity
[153]. Insulin, a vital hormone, plays a crucial role in the onset of obesity.
Anthocyanins have the potential to modulate the central nervous system by
regulating insulin thereby alleviating insulin-resistant conditions and enhancing
insulin sensitivity. The mechanisms through which anthocyanins activate the AMPK
regulatory pathway, decrease phosphorylation of insulin receptor substrate 1
(IRS-1), and reduce expression of retinol-binding protein 4 (RBP4) and sterol
regulatory element-binding protein 1 (SREBP-1) are primarily responsible for
regulating energy homeostasis. The fatty acid and triacylglycerol synthesizing
enzymes are downregulated and are the lipogenic factors. Furthermore, the
upregulation of lipolytic enzymes, reducing the concentration of high sensitivity
C-reactive protein (hs-CRP), and upregulation of PPAR
Leptin is a hormone secreted by white adipocytes that plays a vital role in
lipolysis. It also regulates the food intake by reducing the appetite [155].
Although leptin helps prevent body weight gain, Różańska and
Regulska-Ilow [154] found it ineffective in obese individuals. However, leptin is
positively correlated with proinflammatory cytokines increasing the production of
IL-6, IL-12, and TNF-
Several researchers have reported the influence of anthocyanins on the secretion of adiponectin. Adiponectin is another hormone that regulates glucose homeostasis through various mechanisms mainly in the liver and skeletal muscles. In vitro, adiponectin has been shown to improve insulin secretion and insulin sensitivity, suppress gluconeogenesis in the liver, lower blood glucose levels, and suppress fatty acid oxidation and lipogenesis [157]. Table 3 (Ref. [136, 138, 140, 152, 158, 159, 160, 161, 162]) summarizes the different sources of anthocyanin together and their effects on neurohormones involved in energy homeostasis.
| Source of anthocyanin | Prominent parent anthocyanins | Model | Dose of anthocyanin | Treated period | Effects | Reference |
| Bilberry | Delphinidin | Male Sprague Dawley rats | 0.2 mg/mL of drinking water | 14 weeks | Altered expression of insulin signaling genes | [158] |
| Luteolinidin | ||||||
| Cyanidin | ||||||
| Petunidin | ||||||
| Pelargonidin | ||||||
| Peonidin | ||||||
| Malvidin | ||||||
| Bilberry, Blackcurrant | - | Human clinical study | 320 mg per day | 4 weeks | Decreased fasting blood glucose level | [159] |
| Decreased insulin resistant index | ||||||
| Elevated serum adiponectin | ||||||
| Mulberry | Cyanidin Pelargonidin | Male C57BL/6J mice | 50–125 mg/kg body weight per day | 8 weeks | Decreased uptake and consumption of glucose in HepG2 cells | [160] |
| Suppressed hepatic gluconeogenesis | ||||||
| Improved glucose homeostasis in plasma, liver, and skeletal muscle in mice | ||||||
| Mulberry | Cyanidin | C57BL/6J mice | 200 mg/kg food per day | 16 weeks | Attenuated weight gain | [136] |
| Pelargonidin | Decreased total cholesterol and triglycerides level | |||||
| Cherry | Cyanidin | Increased levels of adiponectin | ||||
| Pelargonidin | ||||||
| Purple maize pericarp | Pelargonidin | Male C57BL/6J mice | 200–500 mg/kg body weight per day | 12 weeks | Attenuated body weight gain | [138] |
| Cyanidin | Reduced blood glucose, and blood triglycerides | |||||
| Induced energy expenditure | ||||||
| Tart cherry (Hungarian sour cherry VN1 variety) | Cyanidin | Male C57BL/6J mice | 60 mg/kg body weight per day | 6 weeks | Decreased leptin levels compared to control | [152] |
| Vaccinium meridionale Sw. fruit | Cyanidin | C57BL/6J mice | 160–280 µg/mL of oral dose per day | 4 weeks | Ameliorated insulin resistance and glucose intolerance | [161] |
| Delphinidin | Reduced body weight, plasma insulin, glucose, leptin, and total cholesterol level | |||||
| Malvidin | ||||||
| Vitis vinifera L. grape skin | Peonidin | Male C57BL/6J mice | 200 mg/kg body weight per day | 12 weeks | Suppressed hyperglycemia | [140] |
| Petunidin | Lowered total cholesterol, LDL, and VLDL | |||||
| Malvidin | Increased expression of AMPK and GLUT4 signaling pathways | |||||
| Prevented insulin resistance | ||||||
| Prevented increase of leptin and resistin | ||||||
| Prevented reduction of adiponectin | ||||||
| - | Cyanidin | Male C57BL/6J mice | 1.6 mg/mL of drinking water | 3 weeks | Attenuated body weight gain | [162] |
| Improved glucose tolerance |
LDL, low density lipoprotein; VLDL, very low-density lipoprotein.
NAFLD involves a range of liver disorders that result from fat buildup in the absence of excessive alcohol consumption. The spectrum ranges from simple steatosis to non-alcoholic steatohepatitis, fibrosis, cirrhosis, or hepatocellular carcinoma [163]. Disturbances in liver homeostasis (glucose and lipid metabolism), mitochondrial and endoplasmic reticulum dysfunction, and subsequent oxidative stress due to the production of reactive oxygen species (ROS) can explain the mechanisms underlying NAFLD and its progression [46].
Several studies have found an accumulation of dysfunctional mitochondria in the
liver which release excessive amounts of ROS. ROS mediate signaling in metabolic
pathways, leading to changes in those pathways that impact the development and
progression of liver disorders. Furthermore, oxidative stress in the liver
results in cell death, fibrogenesis, inflammation, and heightened susceptibility
to infections [164]. In contrast, excessive ROS activates the NLR family pyrin
domain containing 3 (NLRP3) inflammasome. NLRP3 activates Caspase 1 which induces
the production of proinflammatory cytokines IL-1
Superoxide dismutase and glutathione peroxidase are first-line antioxidants capable of scavenging ROS and free radicals [169]. Anthocyanins increase the activities of these enzymes and reduce the oxidative stress in the liver [169, 170, 171]. Furthermore, anthocyanins can inhibit lipid peroxidation, and the activity of enzymes associated with free radicals which inhibit ROS generation [169, 172, 173, 174, 175]. Moreover, anthocyanins mitigate NAFLD by improving lipid oxidation in the liver, inhibiting de nova synthesis of lipids in the liver, inhibiting fat deposition in hepatocytes, activating the AMPK pathway, and inhibiting proinflammatory cytokine expression (Tables 1,2). Table 4 (Ref. [122, 168, 170, 171, 172, 173, 174, 176, 177, 178, 179, 180, 181, 182, 183, 184]) summarizes different sources of anthocyanins and their functions in alleviating NAFLD.
| Sources of anthocyanin | Prominent parent anthocyanins | Model | Dose of Anthocyanin | Treated period | Effects | Reference |
| Bilberry (Vaccinim myrtillus) | Delphinidin | Wistar rats | 200 mg/kg body weight per day | 7 days | Decreased liver damage markers (glutamate dehydrogenase, sorbitol dehydrogenase, malate dehydrogenase) | [122] |
| Cyanidin | ||||||
| Petunidin | Inhibited pro-oxidative mediators | |||||
| Peonidin | Induced hepatic phase II antioxidant enzymes (glutathione S-transferase, quinone reductase) | |||||
| Malvidin | ||||||
| Decreased polyamine catabolism | ||||||
| Bilberry (Vaccinium myrtillus) | - | Human clinical study | 200 mg/kg body weight per day | 7 days | Decreased insulin resistance | [176] |
| Blackcurrant (Ribesnigrum) | Decreased plasma biomarkers of liver injury | |||||
| Blackberry | Cyanidin | HepG2 cells | 20–100 µg/mL | 4 hours | Suppressed generation of reactive oxygen species | [172] |
| Chokeberry | Delphinidin | Suppressed intracellular triglyceride overaccumulation | ||||
| Strawberry | Pelargonodin | |||||
| Wild blueberry | Malvidin | |||||
| Blackberry leaf + fruit | - | Sprague-Dawley rats | 150 mg/kg body weight | 12 weeks | Suppressed oxidative stress through Impaired accumulation of triglycerides and lipid peroxides | [173] |
| Improved insulin sensitivity and dyslipidemia | ||||||
| Increased antioxidant enzymes | ||||||
| Blueberry | Mavidin | Human hepatocyte cell line L02 | 10–400 µM | 24 hours | Suppressed excessive production of ROS and superoxide anion and attenuated oxidative stress | [171] |
| Enhanced antioxidant enzyme activity | ||||||
| Upregulated transcription factor EB (TFEB) mediated lysosomal function | ||||||
| Activated Nrf2/ARE signaling pathway | ||||||
| Cranberry (Vaccinium macrocarpon) | Petunidin | Male C57BL/6J mice | 0.5–7 g/day | 21 weeks | Decreased histological severity of NAFLD | [177] |
| Peonidin | Reduced inflammatory cytokines | |||||
| Suppressed NLRP3 inflammasome activation | ||||||
| Grape skin (polymerized anthocyanins) | - | C57BL/6 mice | 400 mg/kg body weight | 4 weeks | Decreased degree of hepatic steatosis | [178] |
| Inhibited inflammation pathway activation | ||||||
| Increased antioxidant levels and reduced oxidative stress | ||||||
| Inhibited mitochondrial dysfunction through increased |
||||||
| Hibiscus sabdariffa calyces | Delphinidin | Male Sprague Dawley rats | 100 mg/kg body weight per day | 4 weeks | Decreased serum levels of liver damage markers (alanine aminotransferase, aspartate aminotransferase, hepatic malondialdehyde) | [179] |
| Cyanidin | Reduced levels of hepatic inflammatory markers (TNF- |
|||||
| Restoration of altered hepatic architecture | ||||||
| Increased activity of superoxide dismutase and glutathione | ||||||
| Honey berry (Lonicera caerulea) | Cyanidin | HepG2 cells | 20 mg/mL | 24 hours | Suppressed expression of adipogenic genes involved in hepatic lipid metabolism (SREBP-1c, CCAAT/enhancer binder protein alpha, PPAR |
[180] |
| Obese mice | 0.5–1% of diet | 6 weeks | Increased mRNA and proteins involved in fatty acid oxidation (carnitine palmitoyltransferase, PPAR |
|||
| Reduced accumulation of triglycerides | ||||||
| Lingonberry (Vaccinium vitis-idaea L.) | - | Male C57BL/6J mice | 1–5% (freeze dried Lingoberry powder) of diet (w/w) | 12 weeks | Impaired lipid accumulation in liver | [181] |
| Suppressed expression of inflammatory cytokines | ||||||
| Inhibited expression of adipogenic genes | ||||||
| Activated AMPK pathway in liver | ||||||
| Reduced hepatic oxidative stress and inflammation | ||||||
| Mirtoselect (Bilberry extract, 36% anthocyanin) | - | ApoE*3 Leiden mice | 0.1% of diet (w/w) | 20 weeks | Suppressed development of hepatic steatosis and fibrosis | [182] |
| Decreased lipid accumulation in hepatocytes | ||||||
| Decreased expression of pro-inflammatory genes | ||||||
| Myrtaceae fruit peel powder | - | Female C3H/HeJ mice | 2% of diet (w/w) | 10 weeks | Reduced serum levels of liver injury marker alanine aminotransferase | [174] |
| Reduced levels of pro-inflammatory cytokine TNF- |
||||||
| Reduced liver lipid peroxidation | ||||||
| Increased total glutathione levels | ||||||
| Decreased oxidative stress | ||||||
| Purple carrot | Cyanidin | Male Sprague Dawley rats | 100 mg/kg body weight per day | 42 days | Decreased lipid peroxidation | [183] |
| Pelargonidin | ||||||
| Purple corncob | Peonidin | L02 cells | 100–300 µM | 24 hours | Decreased lipid accumulation | [184] |
| Attenuated oxidative stress through inhibited overproduction of reactive oxygen species and superoxide anions | ||||||
| Increased glutathione levels | ||||||
| Alleviated inflammation | ||||||
| Upregulated lipid oxidation | ||||||
| Purple sweet potato (Ipomea batats) | Cyanidin | Kunming mice | 100–400 mg/kg body weight per day | 10 days | Decreased levels of liver damage markers (alanine aminotransferase, aspartate aminotransferase, hepatic malondialdehyde) | [170] |
| Peonidin | Normalized activity of superoxide dismutase and glutathione | |||||
| - | Cyanidin | AML-12 cells | 100 µM | 12 hours | Suppressed hepatic oxidative stress | [168] |
| HepG2 cells | 100 µM | 12 hours | Suppressed NLRP3 inflammasome activation | |||
| Mice | 0.2% of diet (w/w) | 4 weeks | Suppressed hepatic steatosis | |||
| Improved systemic glucose metabolism | ||||||
| Increased PINK1 pathway expression |
NAFLD, Non-alcoholic fatty liver disease; IL, interleukin; TNF-
Maintaining the balance of the gut microbiota is vital for human health. Anthocyanins are recognized as beneficial for maintaining eubiosis by regulating microorganisms. Anthocyanins not only ameliorate the beneficial gut bacteria but also reduce harmful bacteria. In addition, the gut microbiota participates in the metabolism of anthocyanin, the synthesis of vitamins, and carbohydrate catabolism as reported by Tian et al. [185]. Anthocyanins may enhance the proliferation of beneficial microorganisms in vitro and in human subjects including species of Bifidobacterium and Lactobacillus-Enterococcus [186]. Table 5 (Ref. [148, 187, 188, 189, 190, 191, 192, 193, 194]) summarizes the impact of anthocyanins from different sources on the gut microbiota.
| Source of anthocyanin | Prominent parent anthocyanins | Model | Dose of Anthocyanin | Treated period | Effects | References |
| Billberry | Delphinidin | Female C57BL/6 mice | 156 µg per day | 27 days | Improved species diversity | [187] |
| Cyanidin | Increased Lactobacillus johnsonii, Staphylococcus sciuri, Adlercreutzia, and Firmicutes counts with lower relative abundance | |||||
| Petunidin | ||||||
| Peonidin | ||||||
| Malvidin | ||||||
| Black rice | Cyanidin | Male C57BL/6J mice | 0.48 g/kg food | 12 weeks | Improved Shannon index and Simpson index | [188] |
| Petunidin | Significantly improved Bifidobacterium and Lactobacillus counts | |||||
| Increased relative abundance of Allobaculum, Bacteroides, Parabacteroides, Oscillospira, Akkermansia, Ruminococcus, and Butyricimonas | ||||||
| Decreased relative abundance of Clostridium and Desulfovibrio | ||||||
| Blueberry | - | Male Wistar rats | 10% of diet (w/w) | 8 weeks | Decreased abundance of Firmicutes and Bacteriodetes | [148] |
| Increased abundance of Proteobacteria (Actinobacillus, Aggregatibacter), Fusobacteria, and Bacilli (Lactobacialles) | ||||||
| Blueberry and Cranberry | Cyanidin | Male C57BL/6J mice | 1–2% of diet | 24 weeks | Increased relative abundance of Bacteroidetes with high doses of blueberry anthocyanin | [189] |
| Decreased abundance of Rikenellaceae and Streptococcaceae with both blueberry and cranberry | ||||||
| Opuntia ficus-indica | - | Male Kunming mice | 50–150 mg/kg body weight per day | 20 days | Increased Shannon index and Simpson index | [190] |
| Increased abundance of Bacteroides and Firmicutes | ||||||
| Decreased abundance of Proteobacteria | ||||||
| Mulberry (Morus alba L.) | Cyanidin | Male C57BL/6J mice | 100–200 mg/kg body weight per day | 14 days | Inhibited decrease of the bacterial community richness index | [191] |
| Quercetin | Improved Firmicutes/Bacteriodetes ratio by high doses of anthocyanin | |||||
| Increased population of Proteobacteria | ||||||
| Increased abundance of Allobaculum, Muribaculaceae, and Akkermansia | ||||||
| Decreased abundance of Escherichia-shigella | ||||||
| Strawberry (Fragaria ananassa) | Delphinidin | Mice | 50 mg/kg body weight per day | 14 days | Increased relative abundance of Ruminococcus | [192] |
| Cyanidin | Decreased relative abundance of Escherichia, Klebsiella, Proteus, Enterococcus, Staphylococcus, Mucispirillum, and Acinetobacter | |||||
| Red goji berry (Lycium barbarum) | Petunidin | Increased relative abundance of Prevotella, Odoribacter, Acetanaerobacterium, Intestinimonas, Ruminococcus, and Parabacteroides | ||||
| Pelargonidin | Decreased relative abundance of Escherichia, Klebsiella, Proteus, Blautia, Enterococcus, Staphylococcus, Mucispirillum, and Acinetobacter | |||||
| Black goji berry (Lycium ruthenicum) | Peonidin | Increased relative abundance of Intestinimonas and Desulfovibrio | ||||
| Malvidin | Decreased relative abundance of Staphylococcus, Mucispirillum, and Acinetobacter | |||||
| Red Chinese bayberry (Morella rubra) | Increased relative abundance of Prevotella, Allobaculum, Ruminococcus, and Saccharibacteria | |||||
| Decreased relative abundance of Staphylococcus, Roseburia, Mucispirillum, Acinetobacter and Alloprevotella | ||||||
| White Chinese bayberry (Morella rubra) | Increased relative abundance of Rikenella, Helicobacter, Allobaculum, Butyricimonas, and Intestimonas | |||||
| Decreased relative abundance of Klebsiella, Blautia, Mucispirillum, and Acinetobacter | ||||||
| Mulberry (Morus alba) | Increased relative abundance of Intestimonas and Ruminococcus | |||||
| Decreased relative abundance of Vampirovibrio, Escherichia, Klebsiella, Proteus, Enterococcus, Staphylococcus, Mucispirillum, and Acinetobacter | ||||||
| Raspberry (Rubus idaeus) | Increased relative abundance of Aerococcus, Butyricicoccus, and Ruminococcus | |||||
| Decreased relative abundance of Escherichia, Klebsiella, Proteus, Enterococcus, Mucispirillum, and Acinetobacter | ||||||
| Blackberry (Rubus spp.) | Increased abundance of Streptococcus, Helicobacter, Aerococcus, Faecalicoccus, and Saccharibacteria | |||||
| Decreased relative abundance of Klebsiella, Proteus, Acetatifactor, Mucispirillum, and Acinetobacter | ||||||
| Blueberry (Vaccinium spp.) | Increased relative abundance of Prevotella, Ruminococcus, and Barnesiella | |||||
| Decreased relative abundance of Staphylococcus, Clostridium, Mucispirillum, and Acinetobacter | ||||||
| Wild raspberry | Pelargonidin | Male db/db mice with C57BL/6J origin | 150 mg/kg body weight per day | 8 weeks | Increased Bacteroides/Firmicutes ratio | [193] |
| Increased abundance of Bacteroidetes, Bacteroidia, Bacteroidales, Prevotella, and Prevotellaceae | ||||||
| - | Cyanidin | Human clinical study | Self administration of anthocynin rich sources (112 items) | 1 year | Increased microbial diversity with increasing intake of anthocyanins | [194] |
| Delphinidin | Increased abundance of Clostridiales and Ruminococcaceae | |||||
| Malvidin | Decreased abundance of Clostridium XIVa | |||||
| Pelargonidin | ||||||
| Petunidin | ||||||
| Peonidin |
Anthocyanins play an important role in preserving mental health and may alleviate numerous health issues. These compounds demonstrate efficacy against major depressive disorders, anxiety, attention-deficit hyperactivity disorder, and schizophrenia. Proposed mechanisms underlying the efficacy of anthocyanins against these psychiatric disorders include mitigating inflammation, oxidative stress, and dysbiosis [195]. Although anthocyanins cannot solve nutrient deficiency issues associated with LTGFD, they will probably reduce the impact of nutrient deficiencies on mental health through their mechanisms of action.
Depression and anxiety, the most abundant and expected to be the most prevalent psychiatric problems, present with various symptoms, including feelings of distress, a persistently low mood, impaired cognitive function, and a loss of interest [196]. Monoamines, such as serotonin, dopamine, and adrenaline play a crucial role in maintaining mental well-being. Reduced synthesis of monoamine and impaired monoamine functioning are two primary contributors to depression and anxiety. Monoamine oxidase is responsible for this phenomenon [197]. Anthocyanins can inhibit the action of monoamine oxidases, specifically monoamine oxidase A and monoamine oxidase B [198, 199, 200]. Table 6 (Ref. [198, 199, 200, 201, 202, 203, 204, 205]) provides evidence for anthocyanins as a therapy for psychiatric disorders, especially depression and anxiety.
| Sources of anthocyanin | Prominent parent anthocyanins | Model | Dose of Anthocyanin | Treated period | Effects | Reference |
| Black rice (Oryza sativa L.) | Cyanidin | Adult male Kunming mice | 15–60 mg/kg body weight per day | 8 weeks | Inhibited activity of monoamine oxidases | [200] |
| Peonidin | ||||||
| Blueberry (Vaccinium myrtillus) | Cyanidin | Male Swiss mice | 200–400 mg/kg body weight | - | Antidepressant-like effects in mice | [201] |
| Delphinidin | ||||||
| Malvidin | ||||||
| Peonidin | ||||||
| China rose flower (Hibiscus rosa-sinesis Linn) | - | Adult male Swiss albino mice | 30–100 mg/kg body weight | - | Increased activity of serotonin, dopamine, and noradrenaline | [202] |
| Grevia asiatica L. berry fruit | - | Male Sprague Dawley rats | 20–30% in drinking water (v/v) | 28 days | Reduced anxiety and depression like behavior | [203] |
| Mulberry (Morus alba) | Cyanidin | Human clinical study | 180 mL serving that contain 34.3 |
6 weeks | Suppressed cortisol activity | [198] |
| Inhibited activity of monoamine oxidases (Monoamine oxidase A and Monoamine oxidase B) | ||||||
| Positive effects on depression, anxiety, and insomnia | ||||||
| Saffron (Crocus sativus) | - | Human clinical study | 28 mg per day | 4–8 weeks | Decreased depressive symptoms | [204] |
| Sour cherry (Prunus cerasus) | Cyanidin | HeLa cells | 0.001–1 mg/mL | 24 hours | Inhibited activity of monoamine oxidase A | [199] |
| Wild blueberry | - | Human clinical study | 253 mg anthocyanin | 14 days | Decreased depressive symptoms and anxiety symptoms | [205] |
Since anthocyanins are considered as non-essential nutrients, none of the major health authorities have established universal dietary recommendations. Researchers have found different dietary intake limits that are beneficial over discussed pathological issues. Sources of anthocyanins and their effective dosages found during clinical studies are included in Tables 1,2,3,4,5,6.
Once anthocyanins are consumed, they undergo an extremely complex in vivo metabolism due to their extreme sensitivity to the outside environment and susceptibility to degradation. Their metabolism is further influenced by factors such as food matrix, food processing methods, enzyme concentrations, gut microbiota, and genetic factors [206].
Absorption of anthocyanins starts from the oral cavity where saliva, oral epithelial cells, and oral microbiome contribute to their metabolism. Saliva partially degrades anthocyanins, with the resulting metabolites primarily influenced by oral microbiota. Once ingested, anthocyanins are rapidly absorbed in the stomach without undergoing hydrolysis as the acidic gastric environment (pH 1.5–4) provides optimum conditions for their absorption and stability of most anthocyanins other than highly active anthocyanins such as pelargonidin. In the small intestine, the alkaline pH (7.5–8) breaks down the stability of anthocyanins converting them to hemiketal and chalcone. Most anthocyanins are hydrolyzed by the hydrolytic enzymes in the jejunum, while limited absorption takes place in the duodenum and none in the ileum or colon. Two major mechanisms facilitating the absorption of anthocyanin glycosides in the intestine are sodium-glucose co-transporter 1 (SGLT1) and glucose transporter 2 (GLUT2). Unabsorbed anthocyanins are metabolized by the gut microbiota which possess enzymatic capabilities for de-glycosylation and hydrolysis. In the liver, anthocyanins are subjected to metabolic transformation through two phases where Phase I involves demethylation and hydrolysis, whereas Phase II includes glucuronidation, sulfation, and methylation occurring primarily in the liver or kidney [185, 207].
Although anthocyanins play an array of health properties in the human body, their effectiveness can be limited by low bioavailability and their extensive metabolic processing within the body. The low bioavailability of anthocyanins has notoriously been claimed by research. However, Lila et al. [208] state that anthocyanins are far more available than originally envisioned, as anthocyanins undergo enterohepatic recycling where the metabolites last longer performing functional properties in different physiological systems. Gui et al. [207] identify key challenges in fully understanding anthocyanin metabolism and bioavailability, including shortage of anthocyanin monomers, complex metabolism affected by many internal and external factors, and limitation of detection technology. Nonetheless, the dose of intake remains crucial for rewarding results. Although different gluten-free foods enriched with anthocyanins have been produced, a sparse of studies address their functional activity within conventional gluten-free products.
The review comprehensively explores the health risks associated with a LTGFD, particularly those that arise due to nutrient imbalances. It emphasizes the importance of adapting to a well-planned, balanced GFD that embodies natural, whole GF foods over processed GF alternatives. Furthermore, it highlights the potential of anthocyanins in alleviating the health risks concomitant with a LTGFD, offering a detailed account of their mechanisms of action. It summarizes the evidence from in vivo, in vitro, and human clinical studies that support the efficacy of anthocyanins in addressing the discussed health risks.
However, based on current research, it is challenging to directly claim that the GFD alone can be responsible for the discussed health risks, as this review does not delve into other contributory factors such as demographic, physiological, economic, and social influences. Moreover, though anthocyanins remained promising, evidence supporting the use of anthocyanin-enriched GFD or its efficacy is limited, and further studies are essential to validate their functional efficacy along with a LTGFD. Therefore, this review not only identifies this gap but also gives insights for further research, highlighting the need for scientific investigations to prove the role of anthocyanins in alleviating the potential health risks of a LTGFD. It also highlights the need for the development of functional GF products in the market rather than general alternatives for conventional processed food.
Consuming a GFD over a prolonged period may increase various health risks, particularly when the diet is mainly composed of processed gluten-free foods high in fat, simple carbs, salt, and calories. Such foods may exacerbate micronutrient deficiencies, leading to conditions such as obesity, NAFLD, dysbiosis, and psychiatric disorders including depression. However, it is also important to note that not all GF processed foods are inherently unhealthy and a well-planned GFD can provide a well-balanced nutritional diet that brings functional benefits beyond the nutrition. However, research has shown that anthocyanins can address the potential health risks mentioned above. Therefore, anthocyanins can be researched as a dietary supplement for all adherents to GFD, regardless of their symptomatic or asymptomatic background. Furthermore, anthocyanins can help alleviate potential health risks from LTGFD. However, anthocyanins are not to mitigate the causative factors of a GFD such as nutrient imbalances, but to minimize the effect of a LTGFD on the cause of discussed health risks. Consequently, more scientific studies are required to claim the clinical efficacy of anthocyanins in addressing the potential health risks of a LTGFD.
Conceptualization, E-KK; methodology, SDNK; investigation, WAJPW; writing—original draft preparation, SDNK and WAJPW; writing—review and editing, E-KK, SDNK and WAJPW; supervision, E-KK; project administration, E-KK; funding acquisition, E-KK. All authors have contributed to the editorial changes made to the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (RS-2025-00562716) and the BB21plus funded by Busan Metropolitan City and Busan Techno Park. Further, the study was also supported by the Rural Development Administration, Republic of Korea (Project No. RS-2022-RD010228), “A study on the expansion of the functional materials of locally specialized medicinal crops using cultivar.”.
The authors declare no conflict of interest. Although Nutrinomics Lab Co. Ltd. is listed in the author affiliations, it had no involvement in the conceptualization, preparation, writing of this review or decision to publish.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

