1 Department of Dairy Technology and Functional Foods, Faculty of Food Sciences and Biotechnology, University of Life Sciences in Lublin, 20-704 Lublin, Poland
2 Department of Food Technology and Assessment, Institute of Food Sciences, Warsaw University of Life Sciences—SGGW, 02-776 Warsaw, Poland
3 Department of Food Biotechnology and Microbiology, Institute of Food Sciences, Warsaw University of Life Sciences—SGGW, 02-776 Warsaw, Poland
Abstract
Selenium, a trace element with antioxidant properties, plays a vital role in the metabolism of microorganisms. Meanwhile, supplementation with selenium may also modify the activity of probiotics. Thus, this study aimed to analyze the effect of selenium supplementation on the growth, tolerance, and selenium binding capacity of two probiotic strains: Lactiplantibacillus plantarum DSM24730 and 299v. In particular, this study aimed to determine whether introducing this microelement into the culture environment would affect the growth capacity and detoxification mechanisms of these bacteria. Additionally, the ability of the selected strains to absorb and store selenium was analyzed, which could have potential benefits for both human health and the quality of probiotic preparations.
Bacterial cultures of Lactiplantibacillus plantarum DSM24730 and 299v were grown in specially prepared media supplemented with different concentrations of selenium (0–100 mg/L). This study assessed several key physiological parameters of microorganisms in real-time, including biomass production, growth dynamics, and the ability to survive in conditions that simulate the gastric and intestinal environments.
Growth curves and biomass analyses revealed that moderate selenium concentrations (5–10 mg/L) supported the growth of both strains, whereas higher concentrations (50–100 mg/L) inhibited biomass production and delayed the onset of growth, especially in DSM24730. Intensive growth of bacterial biomass (0.23 g/L; p < 0.05) in the experimental medium supplemented with 5 mg Se4+/L was observed for L. plantarum 299v after 24 h of cultivation. In the case of the DSM24730 strain, the lag phase (Δtlag) was prolonged at higher selenium concentrations, reaching 12 h at 100 mg/L, while the logarithmic phase (Δtlog) was shortened from 12 h in the control medium to only 2 h at 100 mg/L. The 299v strain demonstrated faster growth, higher biomass yield, and more rapid selenium uptake at moderate concentrations, while DSM24730 accumulated higher final levels of selenium after prolonged incubation. The highest selenium content (0.45 mg Se4+/g; p < 0.05) after 72 hours of cultivation was accumulated by strain L. plantarum 299v. Meanwhile, after the same culture time, the second bacterial strain (DSM24730) accumulated only 0.29 mg Se4+/g; p < 0.05). Tolerance assays using simulated gastric and intestinal fluids demonstrated that both strains survived under acidic gastric conditions; however, the viability of these strains significantly declined in intestinal juice at selenium concentrations of ≥10 mg/L, indicating an apparent dose-dependent inhibitory effect.
These observations suggest that 299v is more efficient in rapid selenium assimilation and biomass formation, while DSM24730 may be more suitable for high-capacity selenium loading over time. These findings are consistent with other studies on selenium-enriched probiotics, highlighting strain-specific responses to selenium supplementation. Strain selection and selenium dose optimization are essential for developing safe and effective selenium-enriched probiotic products.
Keywords
- Lactiplantibacillus plantarum
- selenium supplementation
- bacterial growth kinetics
- bioaccumulation
- selenium tolerance
Selenium (Se) is a trace mineral widely found in the environment and crucial for human health. Although initially considered toxic, its significance as a vital element in the body was recognized years after its discovery [1]. Se exists in organic and inorganic forms, playing a crucial role in human nutrition. Organic selenium appears as analogs of sulfur amino acids, such as selenomethionine (SeMet), selenocysteine (SeCys), and their methylated derivatives. SeMet is commonly found in products of both plant and animal origin and in certain dietary supplements [2, 3]. Conversely, SeCys is primarily present in animal-based foods, while selenium-methyl selenocysteine (SeMeCys) naturally occurs in vegetables like garlic, leeks, onions, and broccoli [2, 3, 4]. Inorganic selenium includes salts like selenate (SeO42-) and selenite (SeO32-) [5]. They are typically found in food supplements, whereas selenate is present in fish and plant sources. These selenium forms are additionally utilized to biofortify various vegetables [3, 6].
The trace mineral selenium has unique biological properties essential for maintaining health. Despite its presence in the body at very low concentrations, a selenium deficiency can lead to dysfunction in multiple systems. Since the human body cannot synthesize selenium, it must be obtained primarily through the diet [1]. Selenium’s physiological roles are largely carried out by selenoproteins, which are highly effective in combating oxidative stress [7, 8, 9, 10] and inflammation [11, 12]. Proper selenium supplementation can boost the immune system and influence brain function [13, 14, 15], cardiovascular diseases (CVDs) [15, 16], cancer [17, 18], and illnesses caused by heavy metals [19]. While some studies have indicated that high selenium levels may negatively impact certain conditions like type 2 diabetes mellitus (T2DM) [20], further research remains valuable, particularly in the context of novel delivery methods.
A promising approach to combat selenium deficiency is the delivery of bioavailable selenium via fermented products enriched with lactic acid bacteria (LAB). LAB such as Lactiplantibacillus plantarum are nonpathogenic, food-grade microorganisms widely used in the food industry as starter cultures and probiotics. They are renowned for their technological and functional benefits and can accumulate selenium in their biomass [21]. Previous studies have demonstrated the biotransformation of inorganic selenium (e.g., sodium selenite) into organic selenium compounds primarily selenocysteine and SeMet by LAB strains, enhancing its bioavailability and reducing toxicity [22]. Building on this, research has increasingly explored the potential of selenium-enriched microorganisms as functional food ingredients [23, 24, 25], with multiple studies investigating LAB-mediated selenium accumulation under varying fermentation and culture conditions [26]. L. plantarum, in particular, has demonstrated the ability to convert inorganic Se into nutritionally adequate organic forms [23]. Its use in developing selenium-enriched dairy products has shown promise for enhancing dietary selenium intake [27].
L. plantarum 299v is a probiotic strain that naturally occurs in the human gut and can modulate the immune system. Various studies have demonstrated its undeniable beneficial impact on the human microbiome and overall body functions, even in individuals with severe health issues [28]. For example, L. plantarum 299v has been consistently demonstrated to alleviate symptoms of Irritable Bowel Syndrome (IBS), including bloating, irregular bowel movements, and abdominal discomfort. Furthermore, it enhances gut microbial diversity, supports individuals with IBD (Inflammatory Bowel Disease), and helps lower the risk of Clostridium difficile infections in patients on antibiotics. Additionally, it has been shown to enhance iron absorption significantly, likely by improving iron bioavailability and improving long-term iron status in healthy female athletes and non-anemic pregnant women [29]. On the other hand, L. plantarum DSM24730, as a part of a multi-strain probiotic, improves motor function [30]. Additionally, when combined with different probiotic strains like Bifidobacterium longum and Lactobacillus acidophilus in the VSL#3 probiotic formulation, it was shown to prevent diet-induced memory deficits and improve hippocampal-dependent memory tasks, highlighting its positive impact on cognitive function [31]. It is recognized as an effective maintenance treatment for patients with mild ulcerative colitis, either as an adjuvant therapy or for those who are intolerant to 5-ASA [32]. L. plantarum DSM24730, as part of a multi-strain probiotic mix, was shown to reduce the severity of autism spectrum disorder (ASD) symptoms in children, according to improvements on the ADOS-2 scale, highlighting its potential role in managing ASD symptoms [33].
Therefore, this study aimed to evaluate the growth capacity of L. plantarum 299v and DSM24730 strains at different selenium concentrations. Bacterial biomass was obtained after cultivation under conditions with varying concentrations of selenium. The study also assessed the survival of lactic acid bacteria in simulated gastrointestinal (GI) conditions. This study aims to expand our understanding of how specific L. plantarum strains can serve as effective selenium carriers. Ultimately, this study aims to investigate the potential of these selenium-enriched strains as functional ingredients in fermented food products targeted for dietary selenium supplementation.
The biological materials used for the experiments included two probiotic strains: Lactiplantibacillus plantarum 299v and DSM24730 obtained from the collection of pure cultures of the Department of Food Biotechnology and Microbiology, Warsaw University of Life Sciences-SGGW. The microorganisms were cultivated in a liquid de Man, Rogosa, and Sharpe (MRS broth, Merck, Darmstadt, Germany) medium for the time necessary to increase the biomass required.
Liquid MRS medium enriched in sodium selenite (Na2SeO3) was used as an experimental medium for submerged bacterial cultures. The active acidity level of the medium was found to be 5.0 (pH-meter, pH/OXYGEN METER CPO-505, Elmetron, Poznań, Poland). Media and aqueous sodium selenite solution were sterilized at a temperature of 121 °C for 20 min (Systec DE-45, Systec GmbH, Kornwestheim, Germany). For submerged bacterial cultures, sterile liquid MRS media were supplemented with a sterile Na2SeO3 solution (1000 mg Se4+/L) to obtain a final selenium content in the experimental media of 5 to 100 mg/L.
Inoculation cultures of Lactiplantibacillus plantarum 299v and DSM24730 strains were carried out in a flat flask containing 500 mL of liquid MRS medium. The inoculum was prepared by inoculating liquid MRS medium with the 24-h culture of the bacterial strain collected from a slant with the inoculation loop. Inoculation cultures were grown until termination of the logarithmic growth phase (24 h, the late exponential growth phase) until the largest number of bacterial cells was obtained. The resulting inoculum was used to inoculate liquid control and experimental media in a test series.
Bacterial cultures were initiated by inoculating growth media supplemented with
selenium (5, 10, 20, 50, and 100 mg Se4+/L) with a 1% (v/v) inoculum.
Cultures were grown for 72 h under the same parameters as were used for the
inoculation culture. The bacterial biomass was recovered in the pellet after
centrifugation (10,000
To determine the effect of different selenium concentrations on bacterial growth, optical density (OD) changes were monitored using an automated Bioscreen C instrument (Oy Growth Curves Ab Ltd., Turku, Finland), allowing for high-throughput, real-time monitoring of microbial growth. To each well of a 100-well microplate, 270 µL of appropriately prepared MRS medium was added: a control (without selenium added) and an experimental medium (concentrations ranging from 5 to 100 mg Se4+/L). Subsequently, 30 µL of bacterial inoculum suspension was added, prepared from a previous logarithmic-phase culture. Control samples, devoid of biological material, were prepared in parallel to subtract the optical background of the medium and exclude false signals. Microcultures were maintained for 34 h at 34 °C. OD measurements were performed automatically at 30 min intervals using a broadband filter in the 420–580 nm wavelength range. This range corresponds to the maximum absorption of bacteria. It allows for accurate monitoring of changes in cell density in the suspension, providing an indirect indicator of the bacterial population’s growth rate and metabolic activity under the conditions studied.
Growth parameters were calculated as follows:
All experiments were performed in triplicate. Selenium concentrations are reported in mg/L.
To examine the selenium tolerance of L. plantarum strains 299v and DSM24730, we followed the methodology of Herigstad et al. [34]. The bacteria were first incubated at 37 °C for 24 h, followed by droplet plating in serial dilutions (3–7). After incubation in MRS, the medium was centrifuged, and the bacterial pellets were resuspended in simulated gastric and intestinal juices containing varying selenium concentrations (5, 10, 20, 50, and 100 mg/L), as well as control conditions without selenium. The bacterial suspensions were incubated in artificial gastric juice for 3 h and in intestinal juice at 37 °C for 5 h. Following these incubation periods, viable cell counts were determined using the droplet plating method. Specifically, 10 µL of each serial dilution was spotted in triplicate onto MRS agar plates. Plates were left at room temperature for 10–15 min to allow droplets to absorb, and then incubated anaerobically at 37 °C for 48 h. Colonies in each droplet were counted individually and averaged to calculate CFU/mL. This method allows for accurate quantification from small volumes and minimizes media usage while maintaining reproducibility [34].
The gastric juice base composition was adopted from the publication [35]. The
preparation involved dissolving 4.8 g of NaCl, 1.56 g of NaHCO3, 2.2 g of
KCl, and 0.22 g of CaCl2 in 1000 mL of distilled water. The solution was
adjusted to a pH of 2.40 using HCl, in accordance with previously described
in vitro gastrointestinal simulation protocols [35]. It was then
sterilized in an autoclave at 121 °C for 15 min. Immediately before the
experiment, the complete gastric juice was prepared by adding crystalline pepsin
(Sigma-Aldrich, Warsaw, Poland, Cat. P.6887) at a concentration of 1 mg per 6 mL
of the gastric juice base. This corresponds to an estimated final pepsin activity
of approximately 166.7 U/mL, based on the manufacturer’s activity range
(
The intestinal juice model was adopted from publication [36]. In a 1 M NaHCO3 solution, 5 g of NaCl, 0.6 g of KCl, 0.25 g of CaCl2, and 8.5 g of ox bile were dissolved. The 1 M NaHCO3 solution was prepared by dissolving 84.01 g of NaHCO3 in 1000 mL of distilled water. The pH was adjusted to 7.0 using 1 M HCl before sterilization. The intestinal juice was sterilized in an autoclave at 121 °C for 15 min (Systec DE-45, Systec GmbH, Kornwestheim, Germany). Immediately before the experiment, the contents of 2 capsules of the pharmaceutical product Kreon® 10,000 (Solvay Pharmaceutical, Brussels, Belgium) were added to 25 mL of the intestinal juice base. Each capsule contains 150 mg of pancreatin with enzymatic activities of approximately 10,000 Ph. Eur. units of lipase, 8000 units of amylase, and 600 units of protease. This resulted in final activities in the solution of approximately 800 U/mL lipase, 640 U/mL amylase, and 48 U/mL protease.
A calibration curve for determining selenium concentration in samples was prepared according to the methodology described in the literature [37]. Selenium standards with various concentrations ranging from 0 to 2 mg/L were prepared in 100 mL volumetric flasks. To each flask, 10 mL of 2% potassium iodide (KI) solution was added, which acts as a reducing agent, enabling the reduction of selenium ions (Se4+) to a soluble and reactive form. Then, 10 mL of 2 M hydrochloric acid (HCl) was added to acidify the reaction medium. The mixture was thoroughly mixed until a distinct yellow color was obtained, which was evidence of the initiation of the selenium reduction reaction. After a stable color was obtained, 5 mL of 0.05% Variamine Blue solution (Variamine Blue, Sigma-Aldrich, Warsaw, Poland) was added to each flask. The next step was to add 20 mL of 1 M sodium acetate solution, acting as a buffer, which stabilized the pH of the reaction medium and ensured optimal conditions for complexation. After preparing solutions of appropriate concentrations, the final volume was adjusted to 100 mL with distilled water and mixed thoroughly. Absorbance was measured at a wavelength of 546 nm (Bio-Rad SmartSpec 3000 spectrophotometer (Bio-Rad Polska Sp. z o.o., Warsaw, Poland), as this is the wavelength at which the formed variamine blue selenium complex exhibits maximum absorption. A free solution without this element (selenium) was used as a blank sample.
The assay aimed to determine changes in selenium concentration in the culture medium, which allows for an indirect estimation of its accumulation by bacterial cells. It was assumed that selenium loss from the medium after cultivation was primarily due to its absorption and binding in the microbial biomass. A spectrophotometric method based on selenium complexation with the color reagent Variamine Blue (VB) was used for analysis [37]. The analytical procedure began with collecting 1 mL of supernatant from each culture sample after determining the dry weight of the biomass. The supernatants were transferred to 50 mL volumetric flasks, and 5 mL of 2% potassium iodide (KI) solution and 5 mL of 2 M hydrochloric acid (HCl) were added. After thoroughly mixing and developing a characteristic yellow color (indicating reaction initiation), 2.5 mL of 0.05% Variamine Blue reagent solution (Sigma-Aldrich, Warsaw, Poland) and 10 mL of 1 M sodium acetate were added to each flask. The solutions were made up to a final volume of 50 mL with distilled water, mixed thoroughly, and allowed to stabilize. The absorbance of the resulting color complex was measured against a control sample (without selenium) at 546 nm using a Bio-Rad SmartSpec 3000 spectrophotometer (Bio-Rad, Warsaw, Poland). After averaging the measurements, the obtained absorbance values were converted to selenium concentration in the sample based on a previously prepared standard curve. To estimate the amount of selenium absorbed by the cells, the calculated selenium content in the supernatant was compared to its initial concentration in freshly prepared culture medium. The difference corresponded to the amount of selenium bound to the cellular fraction (biomass). Subsequently, the resulting value was converted to a unit of bacterial biomass, allowing for the expression of selenium bioaccumulation efficiency per gram of dry cell mass.
The obtained experimental results were statistically analyzed using multivariate
analysis of variance (ANOVA). The analysis was performed using Statistica 13.3
software (StatSoft Inc., Tulsa, OK, USA). After ANOVA, Tukey’s Honest Significant
Difference (HSD) test was used to understand further which groups differed in
terms of mean values. This test allows for comparisons of all possible pairs of
groups in a way that controls false positives and minimizes the risk of bias
resulting from multiple comparisons. The level of statistical significance was
set at
Fig. 1A,B presents the biomass measurement results (gd.w./L) for Lactiplantibacillus plantarum strains DSM24730 (A) and 299v (B) over time (0–72 h), depending on the selenium concentration in the medium (mg/L).
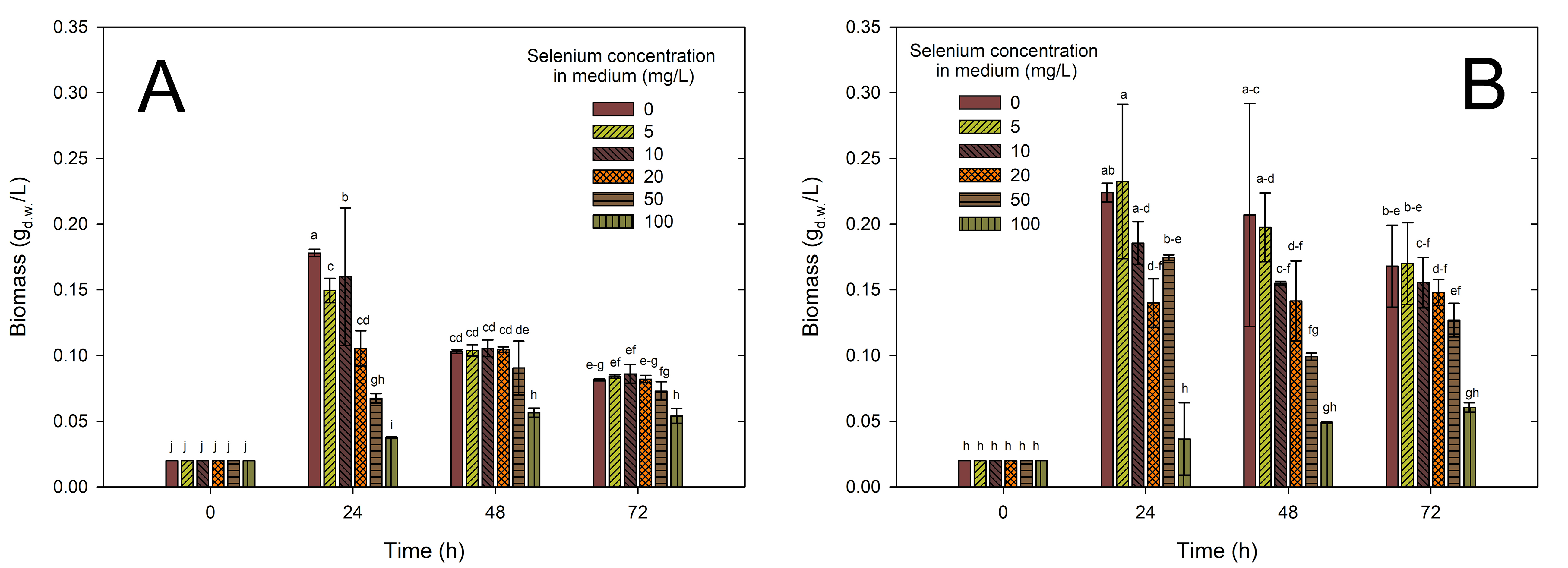 Fig. 1.
Fig. 1.
Effect of selenium on the growth of
Lactiplantibacillus plantarum strains DSM24730 (A) and 299v (B) cells.
Bacterial biomass (Y-axis) was measured at different time points (X-axis) under
experimental conditions with or without selenium supplementation. Data were
presented as mean
In both diagrams, it can be observed that after 24 h, the initial biomass value increase is most evident for the sample without selenium (0 mg/L and 5 mg/L), reaching a value near 0.17 gd.w./L (A) or above 0.20 gd.w./L (B). The biomass growth is lower in the medium with higher selenium concentrations (50 and 100 mg/L in A or 20 and 100 mg/L in B), where the value remains much lower than the control (0 mg/L). In both cases, an increase in biomass was observed in each substrate with the addition of Se (5–100 mg/L). However, only for strain Lactiplantibacillus plantarum 299v did this increase persist, or the biomass amount remained at a similar level after another 24 h incubation. On the other hand, for strain Lactiplantibacillus plantarum DSM24730, biomass value was decreased in the following days of incubation. The highest value was observed for Lactiplantibacillus plantarum DSM24730 (A) in the medium with the addition of 10 mg Se4+/L (0.16 g/L) and for strain Lactiplantibacillus plantarum 299v (B) in the medium with 5 mg Se4+/L (0.23 g/L). Based on this observation, it can be noted that the addition of an appropriate amount of Se supported the growth of the studied bacteria’s strains. In the case of strain Lactiplantibacillus plantarum 299v, better bacterial growth (a higher amount of biomass recorded) was additionally observed in the medium with the addition of 5 mg Se4+/L compared to the control sample (0 mg Se4+/L).
Higher concentration of Se can cause disturbances in the normal physiological condition of bacteria. For example, in Lactobacillus bulgaricus, this type of problem occurred in a medium with 80 mg/L Na2SeO3. In particular, cultures turned red during the stationary phase due to forming Se(0) under elevated selenite levels. The intensity of the red color increased with rising selenite concentrations, indicating that L. bulgaricus can withstand higher levels of selenite by detoxifying it through the reduction of Se(IV) to insoluble Se(0) [23]. The same colour appearance was observed in our research. Both the mentioned strain and the strains tested by us have a similar tolerance to the amount of selenium added to the medium. Functional LAB can generally modify raw materials to enhance their bioavailability while reducing toxic components and antinutritional factors [38]. Pescuma et al. [39] examined two strains: Lactobacillus acidophilus CRL 636 and Limosilactobacillus reuteri CRL 1101 to investigate how these bacteria grow in a medium with the addition of selenium. They observed, similar to us, that the two strains behaved differently with just 5 mg/L of selenium supplementation. L. acidophilus CRL636 exhibited a low growth rate in the presence of Se, whereas the differences were less pronounced for L. reuteri CRL1101. The author also noticed that L. reuteri CRL1101 exhibited higher levels of intracellular SeCys and SeMet compared to the CRL636 strain [39]. The incorporation of selenium through SeCys and SeMet helps bacteria resist oxidative stress. These enzymes play a crucial role in protecting bacterial cells from oxidative stress by neutralizing reactive oxygen species [40, 41]. This could also explain some of the results observed in our study, where the bacterial strains demonstrated improved survival under conditions that might induce oxidative stress. Gonzalez-Olivares et al. [42] in their research tested different LAB strains (Lactobacillus delbrueckii subsp. bulgaricus NCFB-2772, Lacticaseibacillus rhamnosus GG, Lactobacillus helveticus IUAMI-70129 and Lactobacillus johnsonii). The study aimed to assess and measure the ability of lactobacilli to incorporate selenium (0–200 mg/L) into its metabolic processes. They described the relationship between selenium absorption and biomass generation. They observed that the amount of absorbed selenium and the resulting biomass values varied significantly depending on the tested strain. For example, for L. rhamnosus GG, it was 196 mg Se4+/L and 8.83 mg of biomass, while for L. helveticus IUAMI-70129, it was 43 mg/L Se and 14.06 mg of biomass [42]. In this context, studies have indicated that when inorganic selenium is present in the medium, lactobacilli and bifidobacteria can suppress cysteine production in favor of synthesizing SeC. The greater the need for cysteine in microbial development, the higher the proportion of selenium absorbed during growth [43]. Jingjing et al. [44] proved that supplementation with 0.05 g/L L-cysteine significantly improved the lyophilization survival of Lactiplantibacillus plantarum strains, likely due to its antioxidant properties. Similarly, selenium, when added in controlled amounts, supports bacterial growth by providing protection against oxidative stress, suggesting that both cysteine and selenium enhance the bacteria’s resilience under stressful conditions, but only within optimal concentrations.
Fig. 2 shows differences in the growth of the tested bacterial strains. The optical density (OD) of Lactiplantibacillus plantarum DSM24730 (A) increases with time, showing a significant difference that depended on selenium concentration. At 0 mg/L (control), the OD is higher than that of strains developed in MRS with selenium addition. The optical density reaches around 0.9. As selenium concentration in the medium increases (5, 10, 25, 50, and 100 mg/L), there is a noticeable reduction in growth compared to the control, especially at 100 mg/L. On the other hand, the growth pattern of Lactiplantibacillus plantarum 299v is more similar to that of Lactiplantibacillus plantarum DSM24730 during the first 8 h of incubation, but then it diverges significantly. Fig. 2B shows that in the medium without selenium addition, the growth is moderate, reaching an optical density of around 0.7 after 14 h. The growth is slightly lower at 5, 10, and 25 mg/L compared to the control. Additionally, after 16 h, the OD value in medium with 20 mg Se4+/L is higher than that of the control strain. On the other hand, strains in medium with 50 and 100 mg/L Se addition show noticeable inhibition of growth, with a lower optical density compared to lower concentrations. Still, after 17 h (50 mg/L) and 18 h (100 mg/L), an increase in OD value can be observed, compared to the control and other tested concentrations. Both strains (DSM24730 and 299v) exhibit a decrease in growth in the first hour of inhibition as selenium concentration increases. Lactiplantibacillus plantarum DSM24730 shows more pronounced sensitivity to higher selenium concentrations (especially 100 mg/L) than 299v. Lactiplantibacillus plantarum 299v tolerates selenium slightly better than DSM24730, as evidenced by its higher optical density at intermediate selenium concentrations (10 and 25 mg/L). Overall, 100 mg/L selenium is inhibitory for both strains, but Lactiplantibacillus plantarum DSM24730 is more affected by selenium supplementation than Lactiplantibacillus plantarum 299v. Tolerance to the presence of selenium in the medium varies depending on the bacterial type and strain. Different microorganisms exhibit varying sensitivity; even strains within the same species can show distinct responses [45]. Additionally, red color of the deposit indicates that it is amorphous Se(0) rather than crystalline Se(0), which appears gray. This clearly shows that the strains tested can reduce selenite to insoluble elemental selenium, forming electron-dense Se(0) granules, which are subsequently deposited in both the extracellular environment and the cytoplasm [23]. Zan et al. [46] tested six industrial LAB strains (L. plantarum 21,805, L. paracasei 20,241, L. fermentum 21,828, L. casei 23,185, L. acidophilus 6064, and L. plantarum 6076). They noticed that they were capable of normal growth at Se concentrations of 5 and 10 µg/mL, but higher Se concentrations inhibited their growth. Comparable patterns of selenium tolerance have been noted in research on L. reuteri, L. casei and L. animalis [26, 47, 48]. The reduction of Se(IV) is a common characteristic observed in various microorganisms. For instance, in L. bulgaricus, the Se oxyanions are likely reduced by membrane-associated reductase(s) located on the inner membrane surface, which is then followed by the expulsion of Se deposits from the cell [23]. Additionally, Zan et al. [46] prepared an evaluation of the similarity in 16S rRNA sequences between Se-enriched L. plantarum 6076 and the original L. plantarum 6076 strain. The results showed no significant genetic alterations, indicating that the adaptation of L. plantarum 6076 to selenium-enriched conditions did not result in notable genetic variation.
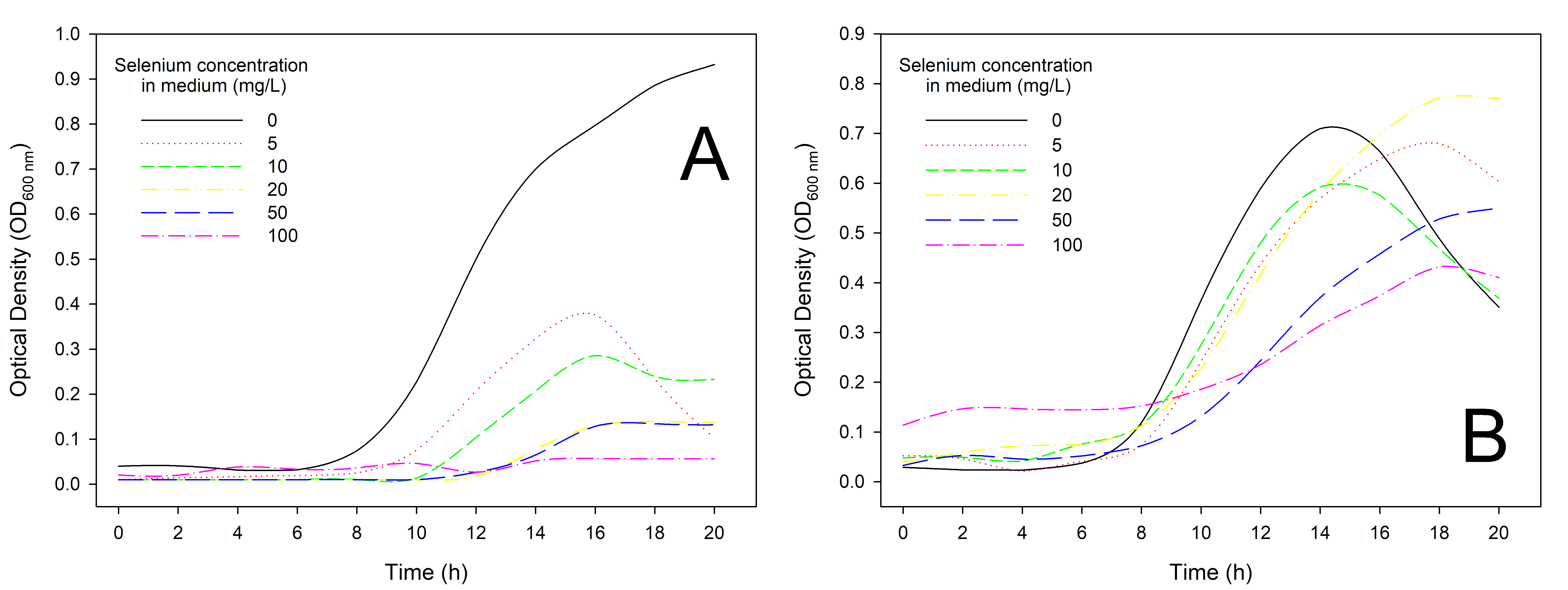 Fig. 2.
Fig. 2.
Effect of selenium concentrations on the growth of different lactic acid bacteria strains: Lactiplantibacillus plantarum DSM24730 (A) and 299v (B) cells. Bacterial growth was assessed by measuring optical density (OD) over time under varying selenium concentrations (0, 5, 10, 20, 50, and 100 mg/L). The X-axis represented incubation time (in hours), and the Y-axis showed biomass expressed as OD values.
This suggests that the ability of this bacteria to thrive in selenium-rich environments relies on its pre-existing metabolic pathways and genomic features. Authors indicated that in high-selenium environments, microorganisms may convert inorganic selenium into Se nanoparticles, potentially altering their morphology and biosynthesis by binding selenium to extracellular polysaccharides, proteins, nucleic acids, and polysaccharides within the microbial cell [46]. When selenium is present in the medium, lactobacilli can effectively convert inorganic selenium into selenocysteine (SeC) through a biochemical process in the cytoplasm. Specific nutrients, such as those found in fermented milk, enhance this bioconversion, allowing up to 73% of inorganic selenium to be transformed during fermentation. However, as the demand for cysteine increases for microbial growth, the ability of these bacteria to absorb selenium and produce SeC becomes more pronounced, highlighting their adaptability to selenium-rich environments [42]. Conversely, when selenium concentrations become excessively high, it can be toxic to these microorganisms, leading to inhibited growth and cell division. This toxicity is primarily due to the disruption of essential cellular processes and the accumulation of harmful selenium species, ultimately limiting their proliferation.
Table 1 presents data characterizing the growth of Lactiplantibacillus
plantarum DSM24730 and Lactiplantibacillus plantarum 299v under varying
selenium concentrations. As selenium content in the medium increases, distinct
effects on bacterial growth can be observed. For Lactiplantibacillus
plantarum DSM24730, the lag phase duration (
| Strains | Selenium content in medium (mg/L) | Lag phase duration | Exponential phase duration | Initial OD | Maximum OD | Maximum specific growth rate | Generation time | ΔOD |
| Δtlag (h) | Δtlog (h) | ODmin log | ODmax log | µmax (h−1) | G (h) | |||
| Lactiplantibacillus plantarum DSM24730 | 0 | 8 | 12 | 0.06 | 0.932 | 0.228 | 3.032 | 0.892 |
| 5 | 9 | 6.3 | 0.06 | 0.932 | 0.228 | 3.032 | 0.366 | |
| 10 | 10 | 5.7 | 0.04 | 0.376 | 0.355 | 1.947 | 0.275 | |
| 20 | 10 | 5.5 | 0.01 | 0.285 | 0.484 | 1.429 | 0.129 | |
| 50 | 12 | 4 | 0.01 | 0.139 | 0.544 | 1.271 | 0.124 | |
| 100 | 12 | 2 | 0.02 | 0.134 | 0.409 | 1.690 | 0.037 | |
| Lactiplantibacillus plantarum 299v | 0 | 6 | 10 | 0.03 | 0.709 | 0.293 | 2.359 | 0.680 |
| 5 | 7.6 | 10 | 0.06 | 0.679 | 0.235 | 2.946 | 0.627 | |
| 10 | 5.2 | 8.7 | 0.06 | 0.592 | 0.261 | 2.652 | 0.544 | |
| 20 | 7.5 | 10 | 0.09 | 0.771 | 0.204 | 3.386 | 0.732 | |
| 50 | 7.7 | 12 | 0.06 | 0.528 | 0.174 | 3.983 | 0.517 | |
| 100 | 8 | 10 | 0.15 | 0.432 | 0.104 | 6.644 | 0.318 |
Selenium-enriched strains were inoculated into the simulated gastrointestinal
fluids, and the viable counts were calculated to investigate the tolerance of the
strains to survive in this type of conditions. The related results are shown in
Table 2. Without selenium, both strains demonstrate high survival rates in
gastric juice, with minimal changes in their CFU/mL after incubation, indicating
good tolerance to acidic conditions. Both strains exhibit a relatively stable
survival rate in gastric juice, as indicated by only minor changes in their
CFU/mL after incubation, suggesting that they are resilient to acidic conditions.
However, in intestinal juice, there is a significant decrease in bacterial counts
after incubation, especially at higher selenium concentrations. This suggests
that while both strains can tolerate gastric conditions reasonably well, their
survival decreases notably in the intestinal environment, particularly for
Lactiplantibacillus plantarum 299v, which shows a more pronounced
decline than Lactiplantibacillus plantarum DSM24730. The presence of
selenium negatively affects the survival of both strains, with higher
concentrations correlating with lower bacterial counts, especially in intestinal
juice. This indicates that the intestinal environment and selenium concentration
play crucial roles in determining the viability of these probiotic strains.
Importantly, our data demonstrate that selenium concentrations
| Condition | Bacterial strain | Selenium (mg/L) | In gastric juice (log CFU/mL) | In intestinal juice (log CFU/mL) |
| Before 24 h incubation | Lactiplantibacillus plantarum DSM24730 | 0 | 9.02d |
9.01ef |
| After incubation | 0 | 9.02d |
6.48b | |
| 5 | 9.06d |
7.02bc | ||
| 10 | 6.85b |
7.35b–d | ||
| 20 | - | 7.09b–d | ||
| 50 | - | - | ||
| 100 | - | - | ||
| Before 24 h incubation | Lactiplantibacillus plantarum 299v | 0 | 11.00e |
10.00f |
| After incubation | 0 | 7.72c |
8.29de | |
| 5 | 6.94b |
8.09c–e | ||
| 10 | 5.70a |
6.84bc | ||
| 20 | 5.70a |
3.90a | ||
| 50 | - | - | ||
| 100 | - | - |
Results were expressed as log CFU/mL: logarithmic values of colony-forming units
per milliliter. “Gastric juice” and “intestinal juice” refer to simulated
fluids mimicking the conditions of the stomach and small intestine, respectively.
Data were presented as mean
The survival ability of lactobacilli bacteria has already been tested. Shu et al. [52] examined 11 strains that were grown on a medium supplemented with 1 mg Se4+/mL. Their results indicated that the survival rate of the bacteria varied depending on the strain tested. Lactobacilli strain L20 showed the highest tolerance to simulated gastric and intestinal fluids compared to other strains. Its survival rate remained relatively high at 63.19% after 240 min in the intestinal fluid, whereas other strains like L22 and L69 exhibited significantly lower survival rates. Strains 8014 and L49 displayed moderate resistance, but overall, L20 demonstrated superior resilience, making it more effective at reaching the intestinal tract intact [52]. On the other hand, researchers examining the survival of L. plantarum 6076 observed the opposite trend. Its viability in the digestive system was found to be influenced by selenium concentration. While higher concentrations of Se (10 µg/mL) enhance the strain’s survival in both gastric and intestinal fluids, lower concentrations result in a gradual decline in viability, with the strain showing better tolerance in gastric conditions compared to the intestinal environment [26]. In our study, we observed reduction in the bacterial count at higher selenium concentrations could imply that selenium is being absorbed or interacting with bacterial cells, leading to toxic effects that reduce growth and biomass. It is known that selenium plays a dual role in bacteria. While it is an essential trace element that can enhance bacterial growth and stress resistance at low levels, higher concentrations may generate oxidative stress and impair the cell’s internal mechanisms. Selenium absorption could increase bacterial sensitivity to acidic environments and bile salts, leading to higher mortality when exposed to gastric and intestinal conditions [53, 54]. The reduction in bacterial count at high selenium concentrations could indicate oxidative stress, potentially linked to the need for increased antioxidant enzyme activity to counteract selenium toxicity. Literature suggests that selenium at certain levels can induce oxidative stress, requiring bacterial antioxidant systems to be activated [55].
These strain-specific responses may reflect differences in intracellular selenium metabolism, including the activity of selenium reductase enzymes and stress-response systems. For instance, DSM24730 may express more efficient antioxidant or detoxification pathways—such as glutathione reductase and thioredoxin systems while 299v may exhibit greater selenium uptake but less capacity to buffer oxidative damage [26, 56]. The data indicates that selenium at increasing concentrations reduces bacterial biomass during simulated digestion, likely due to selenium toxicity. Selenium might bind to the bacterial cells, and the reduction in growth could be due to oxidative stress or other toxic effects caused by selenium. Therefore, the hypothesis related to biomass production under varying selenium concentrations is supported by the observation that biomass decreases at higher Se levels.
In this study, under simulated gastrointestinal conditions, we evaluated the selenium-binding capacities of two bacterial strains, Lactiplantibacillus plantarum DSM24730 (A) and 299v (B). Both strains were exposed to varying initial concentrations of selenium in the growth medium and subsequently subjected to simulated gastric and intestinal fluids to mimic the human digestive process. The amount of selenium retained in the bacterial biomass was measured over time, providing insights into each strain’s ability to assimilate and retain selenium under these conditions. The results indicated that Lactiplantibacillus plantarum DSM24730 strain exhibited a delayed selenium uptake, with negligible accumulation observed at the initial time points. However, over time, particularly after 24 h, the selenium content within the strain increased significantly, especially at elevated selenium concentrations in the medium (Fig. 3). Finally, after 72 h of submerged cultivation, bacteria accumulated 0.14, 0.19 and 0.29 mg Se4+/g, at 5, 10, and 20 mg Se4+/L, respectively. This suggests that while strain Lactiplantibacillus plantarum DSM24730 does not immediately accumulate selenium, its retention capacity improves over time, even under gastrointestinal conditions. In contrast, strain Lactiplantibacillus plantarum 299v demonstrated a more rapid selenium uptake, with significant accumulation observed earlier. Moreover, this strain maintained consistent selenium levels in its biomass over the incubation period. Between the second and the third day of cultivation, selenium accumulation in L. plantarum 299v biomass increased, reaching a concentration of 0.45 mg Se4+/g in the cultures with the highest concentration of the supplemental element (20 mg Se4+/L). This stability indicates a robust selenium assimilation and retention mechanism, making strain Lactiplantibacillus plantarum 299v a promising candidate for selenium delivery in probiotic applications [53].
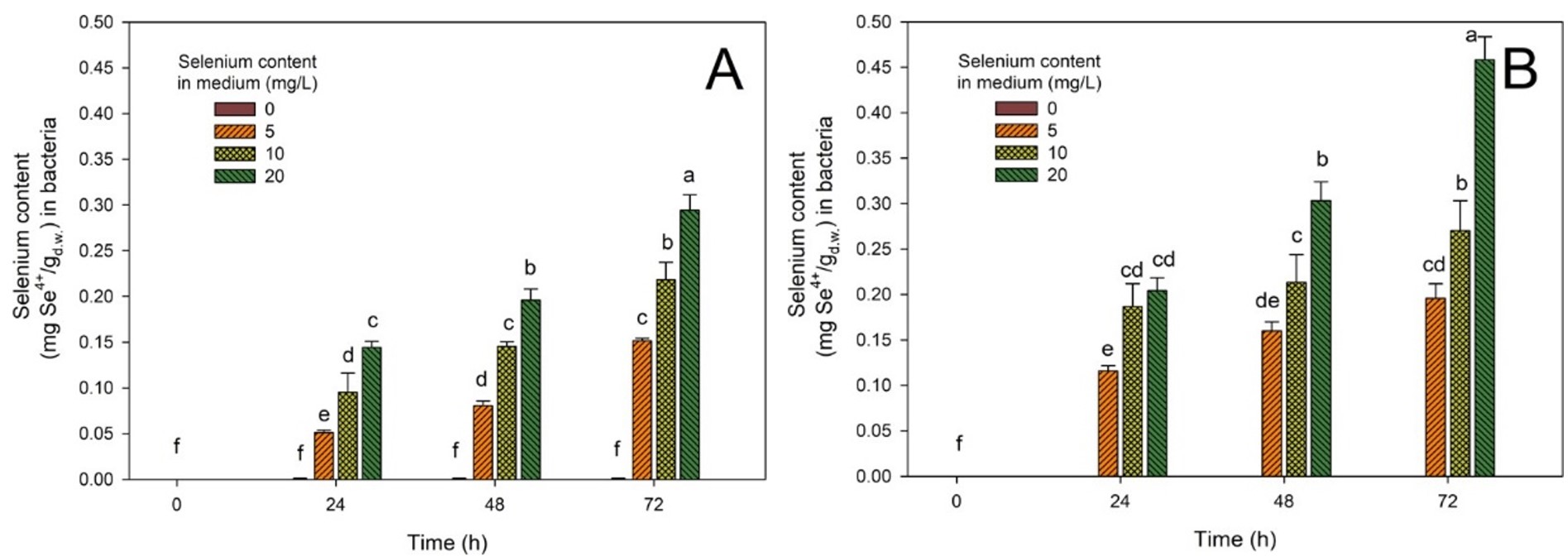 Fig. 3.
Fig. 3.
Effect of selenium binding capacity of different lactic
acid bacteria strains: Lactiplantibacillus plantarum DSM24730 (A) and
299v (B) cells after cultivation in selenium-supplemented media and subsequent
exposure to simulated gastric and intestinal conditions. The X-axis represented
incubation time (in hours), and the Y axis showed the amount of selenium
(Se4+) bound by bacterial biomass, expressed as mg Se4+ per gram of dry
weight (mg Se4+/gd.w.). Data were presented as mean
Moreover, variations in antioxidant capacity and efflux mechanisms may explain these strain-specific differences. The SeZi‑enriched L. plantarum strain exhibited enhanced oxidative stress resistance and higher selenium bioaccumulation, tied to elevated antioxidant protein synthesis [57]. Furthermore, differential expression of membrane efflux pumps, which actively export toxic metal ions and reduce intracellular accumulation, could underlie DSM24730’s superior selenium tolerance compared to the faster Se uptake but less regulated 299v (ABC and RND heavy‑metal efflux systems) [58]. These findings align with recent studies highlighting the importance of selenium-binding stability in probiotic strains. For instance, research by Martínez et al. [26] demonstrated that certain lactic acid bacteria could accumulate and biotransform selenite into selenium nanoparticles and seleno-amino acids, enhancing their probiotic properties and potential for selenium supplementation. Similarly, Norouzi et al. [59] evaluated the probiotic properties and selenium bioaccumulation of lactic acid bacteria isolated from poultry gastrointestinal tracts, finding that strains like Lactobacillus acidophilus and L. animalis exhibited significant selenium accumulation capacities and resilience under acidic and bile conditions. Furthermore, a study by Zhao et al. [60] on selenium-enriched Bifidobacterium longum DD98 showed that selenium enrichment enhanced the strain’s probiotic effects, including improved gut barrier integrity and modulation of gut microbiota composition [26, 59, 61].
It is worth emphasizing that our studies considered both growth capacity and selenium accumulation mechanisms and bacterial survival. The presented information provides valuable insights into the functional properties of probiotic L. plantarum strains. Furthermore, significant differences between strains were demonstrated in their ability to tolerate and accumulate selenium, which may have practical implications for selecting appropriate strains for enriching probiotic products or producing selenium-enriched foods.
In conclusion, the differential selenium retention capacities observed between both strains underscore the significance of selecting probiotic strains with stable selenium-binding properties for effective selenium supplementation. Strain 299v with its consistent selenium retention under simulated gastrointestinal conditions, emerges as a potential candidate for developing selenium-enriched probiotic formulations aimed at enhancing selenium bioavailability in the human body.
The main strength of this study is its comprehensive experimental design, which simultaneously assessed growth kinectics, selenium bioaccumulation capacity, and gastrointestinal survival in two well characterized probiotic strains under controlled laboratory conditions. Furthemore, evaluating both immediate and long-term selenium uptake provides valuable insight for practical application in functional food development. However, the study has some limitations. All experiments were conducted in vitro, which may not fully reflect the complex physiological conditions in the human gastrointestinal tract. The work focused on only two strains of Lactiplantibacillus plantarum, limiting the generalizability of the findings to other lactic acid bacteria. Additionally, only inorganic selenium was tested, and potential differences in the assimilation of organic forms were not explored. Future in vitro studies and the inclusion of additional selenium forms and bacterial strains are warranted to confirm and expand these findings.
This study comprehensively evaluated the effects of sodium selenite on two
probiotic strains of Lactiplantibacillus plantarum DSM24730 and 299v
concerning growth kinetics, selenium accumulation capacity, and survivability
under simulated gastrointestinal (GI) tract conditions. The results show sodium
selenite’s dose dependent inhibitory effect on both strains’ growth and
viability, with strain 299v showing faster growth, shorter lag phase, and greater
biomass yield at moderate selenium concentrations (5–10 mg Se4+/L).
DSM24730, in contrast, exhibited delayed but ultimately higher selenium
accumulation per dry biomass unit, especially after prolonged incubation,
indicating greater total Se retention. Under gastrointestinal simulation, both
strains maintained viability in gastric juice. Still, significant reductions in
CFU were observed in intestinal juice at selenium concentrations
Future research should focus on the in vivo bioavailability of selenium from enriched strains and the long-term safety and efficacy of selenium-enriched probiotic products in human populations. Future research should focus on broadening the scope of selenium probiotic interaction studies by including a wider range of lactic acid bacteria (LAB) strains with diverse ecological origins. This would help identify strains with superior selenium tolerance, bioaccumulation capacity, or gastrointestinal stability. Additionally, investigating the effects of varying culture parameters such as oxygen availability, carbon source, pH, or redox potential on selenium uptake dynamics may reveal environmental factors that modulate Se assimilation. Most importantly, in vivo studies in animal models or human clinical trials are needed to validate the bioavailability, safety, and probiotic efficacy of selenium-enriched LAB strains under physiological conditions. Such studies could clarify whether selenium bound in bacterial biomass is effectively released and absorbed in the human digestive tract, and whether strain-specific differences observed in vitro translate into health-relevant outcomes.
The original contributions presented in the study are included in the article; further inquiries can be directed to the first author.
JS performed the investigation, provided resources, conducted formal analysis, and wrote the original draft. MZ contributed to the investigation and provided resources. MK contributed to the conceptualization, methodology, investigation, resources, formal analysis, writing—review and editing, and supervised the study. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was funded by the European Union from the European Regional Development Fund under the Regional Operational Programme of the Mazowieckie Voivodeship for 2014–2020, grant number Project No. RPMA.01.01.00-14-8276/17 and utilized equipment from the “Food and Nutrition Centre—modernization of the WULS campus to create a Food and Nutrition Research and Development Centre (CŻiŻ)” project.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



