1 Department of Plastic and Cosmetic Center, The First Affiliated Hospital, Zhejiang University, 310003 Hangzhou, Zhejiang, China
2 Department of Orthopedics, The First Affiliated Hospital, Zhejiang University, 310003 Hangzhou, Zhejiang, China
3 Department of Orthopedics, Zhejiang Chinese Medical University, The Second Affiliated School of Zhejiang Chinese Medical University, 310053 Hangzhou, Zhejiang, China
4 Department of Orthopedics, No. 903 Hospital of PLA Joint Logistic Support Force, 310003 Hangzhou, Zhejiang, China
†These authors contributed equally.
Abstract
The vitamin B complex, a group of water-soluble vitamins, is essential for various metabolic and cellular processes and critical for achieving optimal surgical outcomes in plastic and cosmetic procedures. This review examines the mechanistic contributions of this complex at the cellular level, including any roles in mitochondrial bioenergetics, redox balance, gene regulation, and cellular repair mechanisms. Niacinamide, as a precursor to NAD⁺, enhances mitochondrial efficiency and facilitates energy production, supporting tissue regeneration. Pyridoxine functions as a cofactor in neurotransmitter biosynthesis and amino acid metabolism, contributing to nerve recovery post-surgery. Furthermore, cobalamin is crucial for maintaining the myelin sheath and facilitating axonal repair, thereby ensuring neuronal integrity and minimizing nerve damage. Additionally, pantothenic acid, through its role in coenzyme A synthesis, regulates fatty acid metabolism and accelerates cellular repair, aiding in wound healing. Biotin is fundamental for keratin synthesis and follicular cell proliferation, promoting skin integrity and hair regeneration, which are vital in aesthetic and reconstructive treatments. Meanwhile, thiamine ensures a sufficient energy supply for neuronal recovery and cellular resilience through modulating carbohydrate metabolism. Thus, by integrating these cellular mechanisms, the vitamin B complex enhances tissue repair, minimizes inflammation, and improves both aesthetic and functional outcomes. Advances in precision supplementation, innovative drug delivery methods, and regenerative medicine continue to expand the therapeutic potential of B vitamins in plastic and reconstructive surgery. This comprehensive overview underscores the clinical significance of these vitamins in optimizing surgical recovery and promoting long-term tissue health.
Keywords
- vitamin B complex
- skin
- antiaging
- wound healing
- cellular metabolism
Plastic and cosmetic surgery is a multifaceted and rapidly advancing discipline
dedicated to the repair, reconstruction, and enhancement of the human body [1].
With the growing demand for procedures aimed at improving aesthetic appearance
and functional outcomes, there is an increasing focus on optimizing perioperative
care and outcomes. In recent years, the integration of nutritional science into
surgical practice has opened new avenues for enhancing recovery, minimizing
complications, and improving long-term results. Among various nutrients, the
vitamin B complex has emerged as a metabolic and therapeutic agent, particularly
in fields such as wound healing, scar modulation, nerve protection, and skin
rejuvenation. Its biochemical versatility and systemic effects offer potential
benefits that align with the physiological demands of plastic surgery [2]. The
vitamin B complex comprises water-soluble vitamins, such as niacinamide (B3),
pantothenic acid (B5), biotin (B7), thiamine (B1), pyridoxine (B6), and cobalamin
(B12). Each of these vitamins has a unique and critical role in energy
production, cellular metabolism, and neurological function [3, 4]. Thiamine is
crucial for carbohydrate metabolism, acting as a coenzyme in the Krebs cycle and
the pentose phosphate pathway [5, 6]. It facilitates the activity of key enzymes
such as pyruvate dehydrogenase and
These B vitamins work synergistically to support mitochondrial function and energy production, playing vital roles in the electron transport chain, oxidative phosphorylation, and ATP synthesis [27]. Deficiencies in multiple B vitamins can severely impair mitochondrial function, leading to chronic fatigue and an increased risk of neurodegenerative diseases like Alzheimer’s and Parkinson’s disease [28]. Beyond metabolism, the vitamin B complex exhibits protective effects against oxidative stress, inflammation, and neurodegeneration, which are associated with plastic and cosmetic surgeries [29]. In our article, we explore the emerging significance of the vitamin B complex in plastic and cosmetic surgery, with a focus on its mechanistic roles in wound healing, nerve regeneration, skin restoration, and postoperative recovery. The review highlights how each B vitamin contributes to enhancing surgical outcomes and emphasizes their potential as supportive agents in improving both functional recovery and aesthetic results.
The skin barrier serves as a protective layer against environmental stressors,
pathogens, and moisture loss [30]. Niacinamide plays a crucial role in enhancing
the integrity and function of the skin barrier by stimulating the production of
ceramides, free fatty acids, and cholesterol, all of which are essential for
maintaining skin hydration and preventing trans-epidermal water loss (Fig. 1)
[31]. A clinical study found that topical niacinamide significantly improved skin
hydration and barrier function within 12 to 24 weeks after application, leading
to enhanced resilience against environmental damage and reduced sensitivity to
irritants [26]. Furthermore, Gueniche et al. [32] investigated a
dermocosmetic formulation (M89PF) containing niacinamide, hyaluronic acid,
vitamin E, volcanic mineral water, and probiotic fractions. The study
demonstrated that M89PF significantly improved skin hydration, accelerated
barrier recovery, enhanced skin renewal, and reduced oxidative stress in both
in vivo and ex vivo models. Clinical trials in women with
stressed skin revealed that M89PF also rebalanced the microbiome, diminished fine
lines, improved elasticity and radiance, and reduced pigmentation, especially in
Asian populations [32]. Additionally, Lee et al. [33] found that topical
application of niacinamide (2%) combined with human adipocyte-derived stem
cell-conditioned media (ADSC-CM) significantly enhanced post-laser skin
rejuvenation. Over a 3-week period following ablative fractional CO2 laser
treatment, participants showed marked reductions in wrinkles and pigmentation on
the treated side, with greater patient satisfaction and improved global aesthetic
scores compared to the control [33]. In vitro assays confirmed the
combination’s anti-inflammatory, antioxidant, and collagen-promoting effects,
highlighting its synergistic potential in accelerating skin repair and remodeling
after resurfacing procedures [33]. Additionally, a randomized, double-blind
clinical study examined the synergistic effect of niacinamide and
4-hexylresorcinol, demonstrating superior skin tone correction and anti-aging
benefits by enhancing tyrosinase inhibition, reducing fine lines, and improving
skin firmness compared to niacinamide alone [34]. In addition, niacinamide is
widely recognized for its depigmenting effects, particularly in reducing
post-inflammatory hyperpigmentation (PIH) and melasma. It achieves this by
inhibiting the transfer of melanosomes from melanocytes to keratinocytes, thereby
decreasing visible skin pigmentation. This mechanism makes niacinamide an
effective and safer alternative to hydroquinone for managing hyperpigmentation
disorders [35, 36]. A study comparing niacinamide to 4% hydroquinone found that
it was equally effective in reducing melasma while exhibiting fewer side effects
such as irritation and rebound pigmentation [37]. An emerging research has
explored the role of niacinamide in combination with other depigmenting agents
such as alpha-hydroxy acids (AHAs) and ascorbic acid. The synergistic effects of
these compounds enhance skin brightening, reduce blotchiness, and even out skin
tone, making niacinamide a critical component in comprehensive anti-pigmentation
skincare regimens [38]. Hakozaki et al. [39] examined niacinamide and
found that it does not inhibit tyrosinase or melanogenesis but significantly
reduces melanosome transfer (35–68%) from melanocytes to keratinocytes.
Clinical trials in Japanese women showed that a 5% niacinamide moisturizer
decreased hyperpigmentation and increased skin lightness after four weeks, while
2% niacinamide with sunscreen further enhanced skin lightening [39]. A 12-week
clinical study assessed the efficacy of a serum containing 5% niacinamide, 3%
tranexamic acid (TXA), and 1% kojic acid in treating melasma and PIH in 55
Brazilian women with Fitzpatrick skin types I–IV, demonstrating significant
improvements in pigmentation, skin tone homogeneity, and texture as early as week
2 with continued progress through week 12, highlighting niacinamide’s role in
inhibiting melanosome transfer and improving skin hydration [40]. Kimball
et al. [41] reported that a topical formulation containing 4%
niacinamide and 2% N-acetyl glucosamine significantly improved facial
hyperpigmentation, enhanced skin tone uniformity, and supported epidermal
turnover over 10 weeks. The observed effects were attributed to the inhibition of
melanosome transfer and improved barrier function, offering a well-tolerated,
non-hydroquinone-based strategy for addressing uneven pigmentation [41].
Furmanczyk et al. [42] evaluated a depigmenting gel serum containing
niacinamide, tranexamic acid, 4-butylresorcinol, phytic acid, and hydroxy acids,
showing a 67% reduction in melanin production and a 31% decrease in melanin
index after 84 days, reinforcing niacinamide’s depigmenting effects. Mi
et al. [43] further confirmed niacinamide’s environmental protection
benefits, showing that 5 mmol/L niacinamide inhibited pollution-induced
melanogenesis, preventing benzo(a)pyrene-induced pigmentation in a reconstructed
human epidermis model, indicating its efficacy against environmental stressors.
Chronic inflammation and oxidative stress contribute to skin aging, leading to
fine lines, wrinkles, and loss of elasticity. In addition, niacinamide has been
shown to counteract these effects by downregulating the production of
pro-inflammatory cytokines, such as interleukin (IL)-6 and tumor necrosis
factor-alpha (TNF-
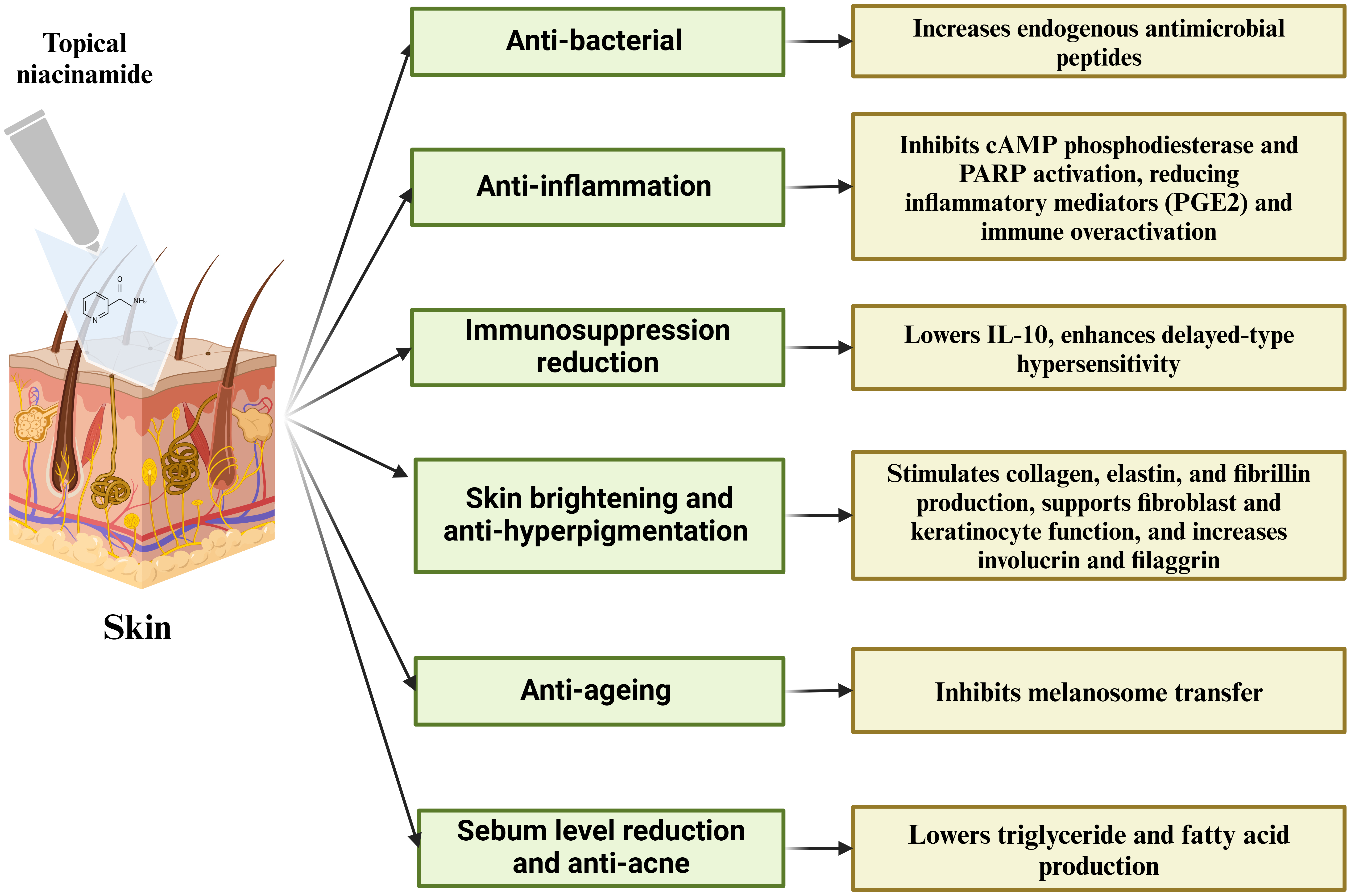 Fig. 1.
Fig. 1.
Therapeutic benefits and biological effects of topical niacinamide in skin health.
Pantothenic acid and its derivatives, including dexpanthenol, have an essential role in wound healing through their involvement in cellular regeneration, inflammation, modulation, and collagen synthesis. Recent studies have demonstrated the efficacy of these compounds in promoting faster wound closure and improving healing outcomes. A 5% dexpanthenol ointment significantly accelerates mucosal wound healing in a 3D nonkeratinized model with CO2 laser-induced lesions. It enhances wound closure and upregulates key wound-healing genes (CXCL10, MUC4, MUC16, MUC20, RARRES1), promoting epithelialization and tissue repair, confirming its therapeutic potential [51]. Yildizhan et al. [52] and Küba et al. [53] found that dexpanthenol accelerates wound healing in animal models, with dexpanthenol showing comparable effects to sucralfate and superior results to other compounds in certain contexts. Heise et al. [54] further confirmed that a dexpanthenol-containing ointment significantly accelerated wound healing and improved cosmetic outcomes following fractional CO2 laser resurfacing of photo-damaged skin. Compared to petroleum jelly, dexpanthenol-treated areas showed faster re-epithelialization, smaller lesion diameters, and higher patient and investigator satisfaction within the first 5 days post-treatment. These effects are attributed to dexpanthenol’s ability to enhance hydration, stimulate epithelial repair, and support collagen remodeling [54]. Pantothenic acid has been shown to promote wound healing by enhancing the migration and proliferation of keratinocytes and fibroblasts, which are essential for epithelialization and collagen production [51, 55]. Furthermore, the impact of pantothenic acid on the immune system, specifically its ability to modulate cytokine production. Pantothenic acid triggers immune cells to produce a range of cytokines, influencing both pro-inflammatory and anti-inflammatory responses, which is particularly beneficial in controlling excessive inflammation during wound healing [56]. The paradoxical nature of pantothenic acid’s effects can shift the balance between proinflammatory and anti-inflammatory cytokines depending on the physiological context, thus enhancing its therapeutic potential in various inflammatory and immune-related conditions [57]. Etensel et al. [58] showed dexpanthenol significantly mitigated oxidative stress and tissue injury in a rat model of testicular ischemia-reperfusion injury. Administering dexpanthenol at 500 mg/kg reduced serum malondialdehyde levels and improved histopathological scores, indicating lower lipid peroxidation and structural damage. These protective effects are attributed to its role in boosting reduced glutathione, Coenzyme A, and ATP synthesis, thereby enhancing cellular resilience against ROS-induced injury [58]. Similarly, Heise et al. [59] revealed that dexpanthenol modulates gene expression to promote skin regeneration and improve wound healing at the molecular level. This regenerative effect is further supported by Proksch et al. [60], who reviewed dexpanthenol’s long-established use in skin care, emphasizing its ability to restore the skin barrier and hydrate the skin, while Dell’Acqua and Schweikert [61] found that its derivative, panthenyl triacetate, activates metabolic pathways that stimulate skin healing. Furthermore, Ulger et al. [62] compared the wound healing efficacy of dexpanthenol and nebivolol in a rat model and found that both agents significantly enhanced healing compared to untreated controls. While wound closure rates were similar between the two treatment groups, dexpanthenol-treated wounds showed significantly lower inflammation and comparable levels of fibrosis and epithelialization [62]. This ability to improve healing was also seen in corneal wounds, as reported by Egger et al. [63], who found dexpanthenol effective in reducing pain and promoting recovery. Moreover, Li et al. [64] reported that a topical formulation containing 5% panthenol, madecassoside, and essential trace elements (copper, zinc, manganese) significantly improved skin recovery after fractional CO2 laser resurfacing. The treatment reduced erythema, trans-epidermal water loss, pain, and postoperative edema, while promoting faster crust removal and delivering superior cosmetic outcomes compared to a standard hospital-prepared emollient [64]. Finally, Celebi et al. [65] showed that dexpanthenol significantly improved post-tonsillectomy recovery by alleviating pain and accelerating mucosal healing. Taken together, these studies emphasize the significant therapeutic potential of pantothenic acid in facilitating tissue repair, reducing inflammation, and promoting skin regeneration, making it a highly effective and versatile agent in medical, dermatological, and cosmetic surgery applications.
Biotin has gained widespread use as a supplement for the management of hair
loss, particularly in cases of biotin deficiency, which can manifest as alopecia,
brittle nails, and other dermatologic issues [66]. Several studies have examined
the efficacy of biotin in both clinical and experimental settings. El-Esawy
et al. [67] demonstrated that male patients with androgenetic alopecia
(AGA) had significantly lower serum levels of zinc and suboptimal levels of
biotin, suggesting that biotin supplementation may improve hair quality and
texture, although no correlation with disease severity was observed. Duchi
et al. [68] introduced a novel encapsulated form of biotin, WS biotin,
which significantly enhanced the water solubility of biotin and was shown to
promote hair follicle keratin expression and the activation of hair
growth-related genes in vitro. In a randomized controlled trial by
Samadi et al. [69], the combination of biotin and dexpanthenol
injections significantly improved hair density and reduced hair loss in patients
with diffuse hair loss. Chavan [70] reported the successful
reversal of premature graying in a 25-year-old female using a topical solution
containing
Postoperative nerve injuries represent a significant complication following various surgical procedures, potentially resulting in prolonged recovery, sensory deficits, and motor dysfunction. Optimal nerve recovery and regeneration post-surgery require effective neurometabolic support to maintain neuronal integrity and facilitate repair processes [84]. Recent scientific research highlights the importance of thiamine and cobalamin in enhancing neuronal metabolic efficiency and supporting myelin sheath regeneration, making them essential components of postoperative neurometabolic strategies (Fig. 2) [85, 86]. Thiamine serves as a coenzyme for several enzymatic reactions essential for neuronal energy metabolism, particularly in the Krebs cycle and pentose phosphate pathway. These metabolic pathways support ATP production, essential for axonal transport, synaptic function, and neuronal repair [87, 88]. Thiamine deficiency has been associated with demyelinating disorders and impaired nerve function. Studies suggest that adequate thiamine levels contribute to Schwann cell function and myelin maintenance, reducing the risk of postoperative neuropathy [89]. In addition, thiamine’s role in modulating oxidative stress and inflammatory responses has been highlighted. It supports glutathione production, a key antioxidant, and reduces neuroinflammatory cytokine release, potentially mitigating neuropathic pain following surgery [90]. Experimental studies have demonstrated that thiamine supplementation enhances axonal regeneration following peripheral nerve injury. Research suggests that thiamine promotes the expression of nerve growth factors and accelerates functional recovery in nerve crush injury models. In clinical settings, patients receiving thiamine supplementation have shown improved nerve function and reduced neuropathic pain after surgery, supporting its potential role in postoperative nerve protection [91]. Alemanno et al. [92] reported that perineural administration of thiamine with levobupivacaine prolonged postoperative analgesia in patients undergoing interscalene block for shoulder surgery, extending pain relief without significant side effects. Thiamine enhances analgesia by increasing acetylcholine synthesis, which improves nociceptive inhibition at the spinal level, making it a potential adjunct for prolonged postoperative pain management [92]. In a chronic compression dorsal root ganglion model, thiamine administration leads to a dose-dependent reduction in pain sensitivity, stabilization of nerve excitability, and restoration of tetrodotoxin-resistant (TTX-R) sodium currents [93]. Moreover, Benfotiamine (lipid-soluble thiamine) significantly improved nerve conduction velocity (NCV) and reduced advanced glycation end-products, including carboxymethyl-lysine (CML) and 3-deoxyglucosone (3DG)-type AGEs. Preventive administration of benfotiamine nearly normalized NCV and completely inhibited glycoxidation in nerve tissues [94].
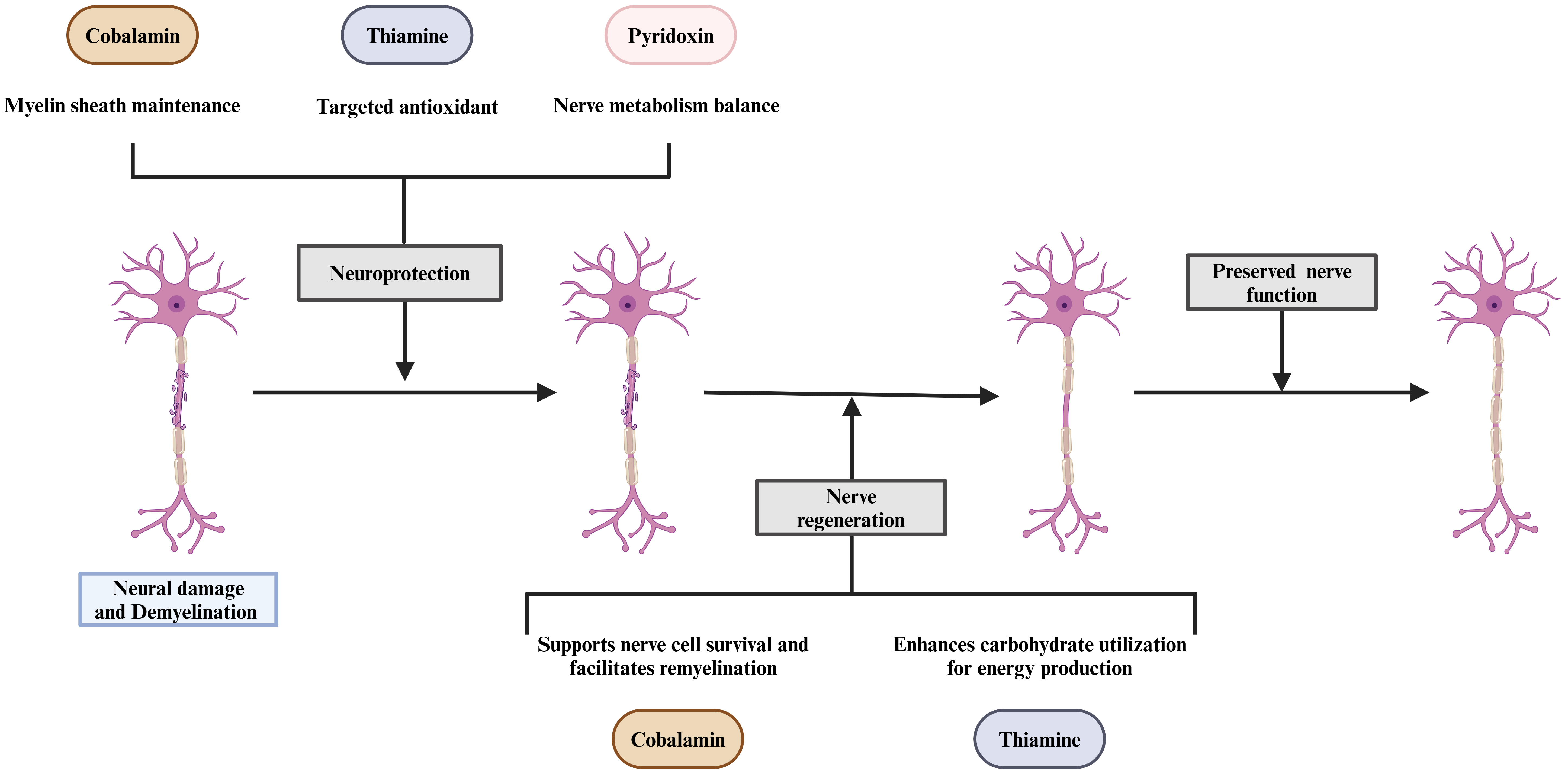 Fig. 2.
Fig. 2.
The role of Cobalamin, Thiamine, and Pyridoxine in neuroprotection, nerve regeneration, and function preservation.
Meanwhile, cobalamin is essential for myelin sheath maintenance and repair. It
functions as a coenzyme in methylation reactions crucial for myelin production,
ensuring nerve fibers’ structural integrity and conductivity. Mekaj and Mekaj [95] reviewed the role of pharmacological agents in peripheral nerve
regeneration and highlighted methylcobalamin as a key neuroregenerative compound.
They explained that methylcobalamin supports nerve repair by promoting myelin
sheath formation, increasing the number of Schwann cells and myelinated fibers,
and enhancing axon diameter. It also helps restore axoplasmic flow and
intracellular protein transport essential for nerve function. In addition, its
antioxidant activity and brain-derived neurotrophic factor (BDNF) upregulation
further strengthen its potential as a neuroprotective therapy in both systemic
and local delivery strategies for peripheral nerve injury [95]. This is
particularly relevant for postoperative patients with nerve damage, as myelin
regeneration is key to restoring nerve function. High-dose methylcobalamin
supplementation has been shown to significantly reduce neuropathic pain by
suppressing pro-inflammatory cytokines, such as TNF-
Cellular aging is characterized by a progressive loss of cellular function driven by key molecular changes, including mitochondrial dysfunction, oxidative stress, DNA damage, and impaired repair mechanisms. These alterations lead to reduced energy production, accumulation of ROS, genomic instability, and eventual cellular senescence, contributing to tissue degeneration and diminished regenerative capacity. The vitamin B complex is deeply integrated into these processes through its role in supporting mitochondrial metabolism, maintaining redox balance, and facilitating DNA and RNA synthesis. By acting as cofactors in enzymatic reactions central to ATP production, antioxidant defense, and cellular repair, B vitamins help preserve cellular integrity and delay the onset of senescence. Their involvement in epigenetic regulation and modulation of pro-inflammatory pathways further strengthens their role in maintaining homeostasis during aging (Table 1, Ref. [5, 9, 86, 103, 104, 105, 106, 107, 108, 109, 110, 111]) [112, 113].
| Compound | Primary Function | Molecular Targets | Key Pathways Involved | Anti-Aging Mechanism | Clinical/Experimental Evidence |
| Thiamine (B1) | Coenzyme in carbohydrate metabolism | Pyruvate dehydrogenase, |
Krebs cycle, ATP synthesis | Maintains mitochondrial energy output, reduces ROS accumulation | Deficiency linked to neurodegeneration and mitochondrial dysfunction [5, 9] |
| Niacin (B3) | NAD⁺ precursor | SIRT1, PARP1 | NAD⁺/Sirtuin signaling, DNA repair, mTOR modulation | Promotes autophagy, genomic stability, and oxidative stress resistance | NAD⁺ restoration reverses senescence in fibroblasts, protective against depression [103, 104] |
| Pantothenic acid (B5) | Component of Coenzyme A | Fatty acid synthase, Acetyl-CoA synthase | Lipid metabolism, mitochondrial |
Enhances mitochondrial dynamics and wound healing | Improves connective tissue strength in postoperative models [86] |
| Pyridoxine (B6) | Amino acid and neurotransmitter metabolism | PLP-dependent enzymes | One-carbon cycle, glutathione synthesis | Reduces inflammation, supports redox balance, collagen synthesis | Prevents LEV-induced behavioral symptoms; high doses linked to neuropathy [105, 106] |
| Biotin (B7) | Coenzyme in carboxylation reactions | Pyruvate carboxylase, Acetyl-CoA carboxylase | Gluconeogenesis, lipid biosynthesis | Supports skin/hair renewal, epithelial turnover | Limited direct aging data; supports keratin infrastructure [107] |
| Cobalamin (B12) | One-carbon metabolism, DNA synthesis | Methionine synthase, Methylmalonyl-CoA mutase | DNA methylation, Krebs cycle | Regulates epigenetic stability, prevents MMA-induced mitochondrial damage | Increases Klotho expression, improves histone methylation in stem cells [108, 109] |
| NMN | Direct NAD⁺ precursor | SIRT1, SIRT3, PGC-1 |
Mitochondrial biogenesis, autophagy, and neurovascular signaling | Restores mitochondrial function, improves cognitive and physical performance | Clinically shown to enhance NAD⁺ levels and physical endurance [110, 111] |
NAD, nicotinamide adenine dinucleotide; CoA, coenzyme A; PLP, pyridoxal phosphate; MMA, methylmalonic acid; NMN, nicotinamide mononucleotide.
Cobalamin supports metabolic processes that regulate one-carbon metabolism and
energy production, with its deficiency leading to systemic metabolic dysfunction
and contributing to cellular aging. Studies have shown that cobalamin is
essential for cellular reprogramming, with its depletion impairing cellular
plasticity and tissue repair, while supplementation enhances regeneration through
improved histone methylation and transcriptional fidelity [108, 114]. Under
normal conditions, cobalamin serves as a coenzyme for the enzyme
methylmalonyl-CoA mutase, which catalyzes the conversion of methylmalonyl-CoA to
succinyl-CoA. This reaction is essential for properly metabolizing certain fatty
acids and amino acids. The resulting succinyl-CoA enters the Krebs cycle, which
contributes to energy production and has a role in heme synthesis [115, 116].
Additionally, cobalamin deficiency leads to the accumulation of methylmalonic
acid (MMA), which disrupts mitochondrial function, increases oxidative stress,
and triggers inflammation, accelerating the aging process (Fig. 3) [117].
Research further indicates that cobalamin contributes to longevity by increasing
the expression of the anti-aging protein Klotho, potentially mitigating metabolic
decline and age-related diseases [109]. Its role in modulating immune responses
and maintaining gut microbiota balance has also been linked to reduced
inflammaging, highlighting its systemic benefits in aging prevention [113].
Furthermore, studies on metabolic demands during cellular reprogramming reveal
that B12 depletion impairs epigenetic modifications, leading to reduced
reprogramming efficiency and limiting regenerative capacity, while
supplementation restores these processes, reinforcing its potential in anti-aging
therapies [108]. Song et al. [104] study demonstrates that the Nampt
inhibitor FK866 mimics vitamin B3 deficiency by depleting intracellular NAD⁺
levels, which in turn reduces SIRT1 activity and induces cellular senescence in
human fibroblastic Hs68 cells. The senescence effect was reversed by nicotinic
acid, nicotinamide, or NAD⁺ supplementation, confirming that attenuated
NAD⁺-SIRT1 signaling is the primary mechanism [104]. The mTOR is a central
regulator of cellular growth, metabolism, and survival. It functions through two
distinct complexes, mTORC1 and mTORC2, with mTORC1 being particularly sensitive
to nutrient availability and playing a critical role in aging by inhibiting
autophagy and promoting anabolic processes. Chronic mTORC1 activation is
associated with cellular senescence, stem cell exhaustion, and age-related tissue
dysfunction [118]. Niacin and folate indirectly modulate mTOR signaling through
their roles in NAD⁺ metabolism, methylation cycles, and redox homeostasis. For
instance, Tripathi et al. [119] demonstrated that vitamin B
supplementation restores autophagic activity and suppresses aberrant mTOR
signaling in hyperhomocysteinemia-induced cellular stress. Abdullah et
al. [120] emphasize the interconnected roles of mTOR and sirtuins, particularly
SIRT1, in regulating cellular aging, neurodegeneration, and proteostasis. They
explain that mTOR inhibits autophagy, leading to the accumulation of misfolded
proteins, while SIRT1 promotes autophagy and mitochondrial function via
deacetylation of FOXO transcription factors and activation of PGC1
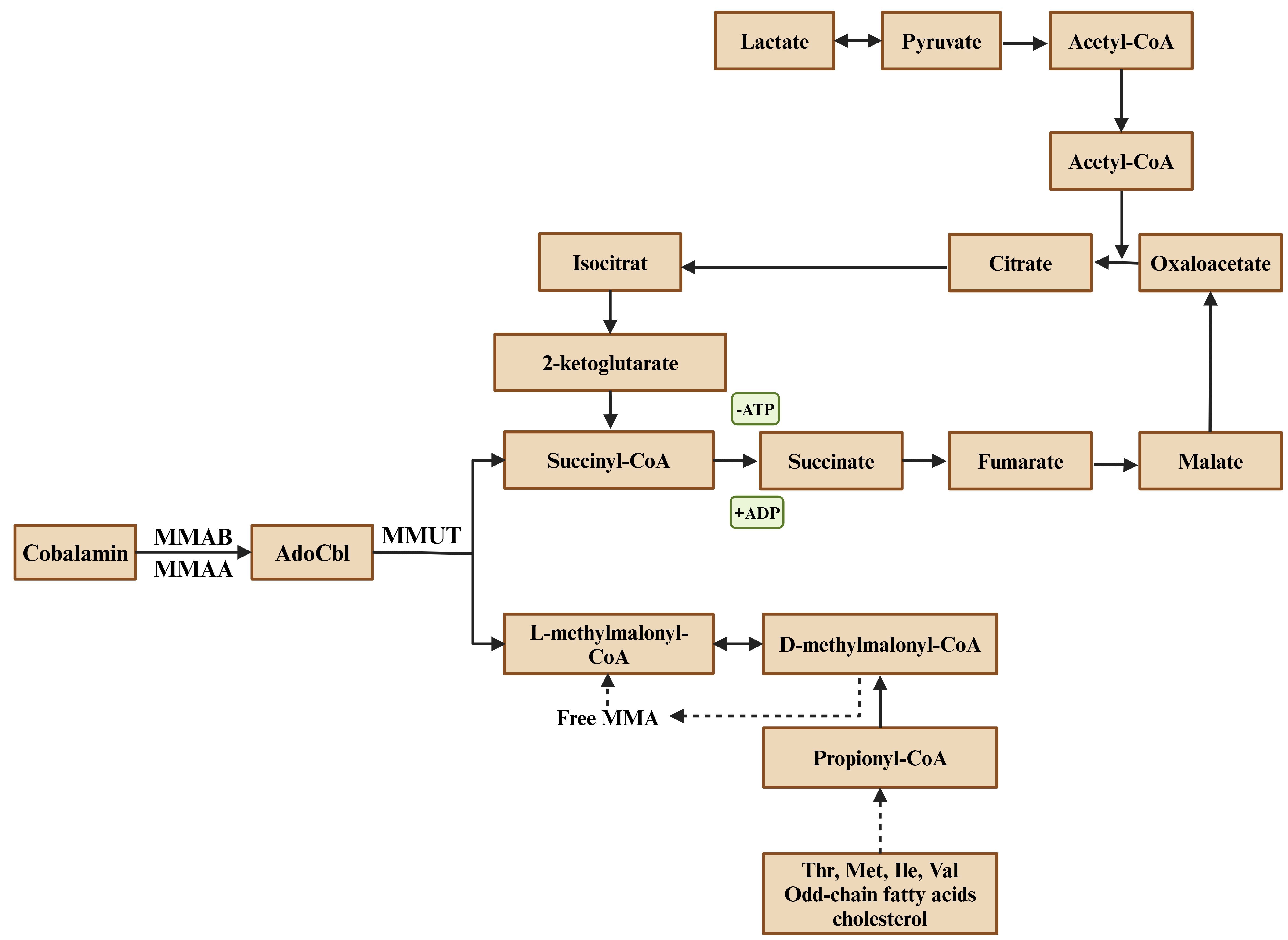 Fig. 3.
Fig. 3.
The role of cobalamin (vitamin B12) in the conversion of propionyl-CoA to succinyl-CoA via methylmalonyl-CoA mutase (MMUT), linking odd-chain fatty acid and amino acid metabolism to the TCA cycle, with deficiencies leading to methylmalonic acid (MMA) accumulation. CoA, coenzyme A; MMAB, methylmalonic aciduria type B; MMAA, methylmalonic aciduria type A.
Nicotinamide mononucleotide (NMN), a derivative of niacinamide, plays an
important role in mitochondrial energy metabolism by serving as a precursor to
NAD+, a coenzyme essential for ATP production [121]. NMN supplementation has been
shown to enhance mitochondrial function, improve oxidative phosphorylation, and
boost cellular energy levels, all of which are crucial for maintaining skin
vitality and delaying cellular senescence. By restoring NAD⁺ levels, NMN
counteracts age-related mitochondrial decline, which is a key factor in skin
aging and reduced metabolic efficiency [122]. Wang et al. [123] explored
NMN’s role in rescuing mitochondrial dysfunction and delaying cellular senescence
in mesenchymal stem cells (MSCs), emphasizing how its ability to restore NAD⁺
levels and activate Sirt3 contributes to longevity and regenerative medicine.
Similarly, Xu et al. [124] demonstrated NMN’s ability to reverse hair
follicle atrophy, enhance cell proliferation, and reduce oxidative stress,
highlighting its therapeutic potential in androgenetic alopecia through the
inhibition of the NF-
While the preceding sections have detailed the diverse physiological and therapeutic benefits of individual B vitamins in surgical and aesthetic contexts, it is equally important to contextualize these benefits within the broader framework of clinical safety. Understanding the potential adverse effects associated with high-dose or long-term supplementation provides essential guidance for integrating vitamin B complex into patient care. The following section addresses these safety considerations, ensuring a balanced approach that aligns therapeutic potential with evidence-based risk management.
Under normal physiological conditions, B vitamins are required for enzymatic functions that regulate metabolism, red blood cell production, and nervous system activity. Their widespread presence in food sources, including meats, dairy products, leafy greens, and fortified cereals, ensures adequate intake for most individuals [131]. Based on their water-soluble nature, excess amounts are typically excreted in urine, reducing the likelihood of toxicity [132]. Vitamin B supplementation is commonly used to prevent and manage deficiencies in high-risk groups, such as pregnant women, the elderly, vegetarians, and individuals with malabsorption disorders [4, 133]. In elderly individuals, age-associated gastric atrophy, hypochlorhydria, and polypharmacy contribute to diminished absorption of cobalamin and folate, increasing the risk of neuropathy and cognitive decline [26]. Li et al. [134] concluded that vitamin B supplementation, particularly in individuals with mild cognitive impairment (MCI), may be a valuable preventative intervention against cognitive decline. They suggest that routine B vitamin supplementation could benefit elderly adults at risk of developing dementia. Furthermore, Vitagliano et al. [135] highlights that optimal preoperative nutritional status, including B vitamin adequacy, is crucial for favorable surgical and aesthetic outcomes. This is particularly important for elderly individuals undergoing cosmetic procedures, who are more prone to micronutrient deficiencies and impaired wound healing. Importantly, elderly individuals undergoing plastic surgery often present complex health profiles [135]. Broughton et al. [136] caution that vitamin supplements, including B complex, may interact with anesthesia and medications. Thus, surgeons should review all supplements during preoperative assessments [136]. Individuals with malabsorption disorders, such as celiac disease or Crohn’s disease, often require higher doses of cobalamin and folic acid due to impaired intestinal absorption. Similarly, vegetarians and vegans are at higher risk of B12 deficiency since this vitamin is predominantly found in animal-based foods, making supplementation necessary to maintain optimal neurological and hematological function [137, 138]. Pregnant and lactating women require increased levels of certain B vitamins to support fetal development and maternal health, where vitamin pyridoxine has been found to alleviate pregnancy-related nausea and vomiting, further emphasizing the importance of individualized nutrient planning during this period [139, 140]. Athletes and individuals with high metabolic demands may require higher doses of B vitamins to support energy metabolism, muscle recovery, and endurance [141, 142].
Furthermore, diabetes patients may benefit from additional thiamine, as diabetes-related complications are associated with thiamine depletion [143, 144]. Toninello et al. [145] reported that glutathione and vitamins, including B-complex constituents, were reduced in diabetic wound tissue. Nutritional supplementation aimed at restoring these levels enhanced both tissue regeneration and cosmetic outcomes following surgery. Additionally, pyridoxine and cobalamin are critical in cardiovascular health, particularly in individuals with elevated homocysteine levels, which are linked to an increased risk of heart disease. Addressing these deficiencies through targeted supplementation can help mitigate disease progression and improve overall health outcomes [146, 147]. The safe dosage of vitamin B complex varies based on physiological needs, dietary intake, and individual health conditions. The recommended daily allowances (RDAs) and tolerable upper intake levels (UL) for each B vitamin are established to ensure optimal health while preventing toxicity (Table 2, Ref. [4, 148, 149]) [150]. In burn patients, daily supplementation of B vitamins (B1 100 mg, B6 200 mg, cobalamin 2000 mcg) significantly reduced infection and sepsis rates, and shortened hospital stays. The high-dose cobalamin likely overcame issues of bioavailability, particularly in acute stress and catabolic states common in severe burns [151]. Gardiner and Hartzell [152] highlight the effect of thiamine in plastic surgery, presenting a case where postoperative hypotension and hypothermia were reversed only after intravenous thiamine. Furthermore, Sebastian et al. [153] reported that unrecognized thiamine deficiency in a post-bariatric patient caused acute neurological compromise following aesthetic procedures, reinforcing the necessity of vitamin B assessment in plastic surgery settings. In such high-risk cases, thiamine supplementation of 100–200 mg/day intravenously for 3–5 days, followed by oral maintenance doses of 10–20 mg/day, is typically recommended to prevent or treat deficiency-related complications [153]. The excessive pyridoxine supplementation, particularly in bariatric surgery patients, led to a significant rise in cases of pyridoxine toxicity, including sensory neuropathy, highlighting the need for stricter guidelines on pyridoxine dosing in clinical nutrition [109]. In addition, patients with a history of bariatric procedures, screening for cobalamin deficiency is warranted. In cases of confirmed deficiency, treatment typically involves intramuscular injections of 1000 micrograms weekly for four weeks, transitioning to monthly doses, or alternatively, daily oral supplementation of 1000 micrograms may be effective. Importantly, prophylactic supplementation in individuals with normal serum levels is not recommended, as it offers no added clinical benefit [154, 155].
| Vitamin | RDA (Adults 19–50 years) | Pregnant female | Tolerable UL |
| Thiamine (B1) | 1.1–1.2 mg | 1.4 mg | No established UL |
| Niacin (B3) | 14–16 mg | 18 mg | 35 mg (nicotinic acid form) |
| Pantothenic Acid (B5) | 5 mg | 6 mg | No established UL |
| Pyridoxine (B6) | 1.3–1.7 mg | 1.9 mg | 100 mg |
| Biotin (B7) | 30 µg | 30 µg | No established UL |
| DCobalamin (B12) | 2.4 µg | 2.6 µg | No established UL |
RDAs, recommended daily allowance; UL, upper intake level.
Most individuals tolerate B vitamins well when consumed through a balanced diet or in moderate supplement doses. They are frequently prescribed for deficiency-related conditions, including anemia, neuropathy, and cognitive decline [156, 157]. However, certain health conditions, medication interactions, and long-term high-dose supplementation can lead to unintended effects. For instance, pyridoxine toxicity is well-documented, with prolonged intake of doses exceeding 200 mg per day leading to sensory neuropathy, causing symptoms such as numbness, tingling, and loss of coordination [158]. In extreme cases, these neurological impairments may become irreversible [158, 159]. An investigation by Alsaadi et al. [106] explored the use of pyridoxine to manage behavioral side effects, such as agitation and irritability, in adult patients treated with levetiracetam. Among 51 patients, 66.6% showed significant improvement in symptoms with an average dose of 54.5 mg/day, suggesting pyridoxine as a potentially effective and well-tolerated adjunct for levetiracetam-induced behavioral disturbances.
Similarly, excessive niacin intake can cause flushing, gastrointestinal distress, and, at very high doses, hepatotoxicity. Sustained-release niacin formulations have been particularly associated with liver damage and metabolic disturbances, including insulin resistance and hyperglycemia [160, 161]. Zhou and Han [162] identified a U-shaped relationship between dietary niacin intake and the risk of metabolic dysfunction-associated steatotic liver disease (MASLD), with the lowest risk observed at an intake of approximately 23.6 mg/day. The findings suggest that while moderate niacin consumption may be protective, excessive intake could increase MASLD risk. In addition, Li et al. [163] reported that excessive niacin intake was strongly associated with the rising prevalence of childhood obesity in the United States, with a 10-year lag correlation. Their study also showed that a 300 mg nicotinamide load induced insulin resistance, followed by reactive hypoglycemia in healthy individuals. Tian et al. [103] analyzed data from over 16,000 U.S. adults and found a U-shaped relationship between dietary niacin intake and depression risk, with the lowest risk observed at approximately 36 mg/day. Below this level, increased niacin intake was associated with reduced depression, while higher intakes were linked to elevated risk.
Other B vitamins, such as pantothenic acid, have an excellent safety profile, with minimal reported adverse effects. Scott et al. [107] found that panthenol, pantothenic acid, and related derivatives are safe for use in cosmetic products when formulated within currently accepted concentrations. The safety assessment, based on toxicological evidence and usage data, concluded that these ingredients do not pose significant health risks under intended conditions of use. Pantothenic acid toxicity is rare but may cause mild gastrointestinal symptoms such as diarrhea when taken in excessive amounts [164, 165]. Biotin is generally safe; however, recent studies have shown that high doses may interfere with laboratory tests, particularly those assessing thyroid function and cardiac biomarkers, leading to potential misdiagnoses [166, 167]. Furthermore, cobalamin is considered safe, even at high doses, due to its efficient storage and excretion mechanisms. However, hypersensitivity reactions and acne-like eruptions have been reported in some individuals taking high-dose cobalamin supplements [168, 169]. Additionally, some studies have suggested that excessive cobalamin intake in individuals with kidney disease may be associated with adverse vascular effects [170, 171]. The risk of vitamin B complex toxicity is further influenced by underlying health conditions and medication interactions. Individuals with liver disease should avoid excessive niacin intake due to its hepatotoxic effects, while those with kidney disease may be at risk of accumulating excess water-soluble vitamins, leading to unintended side effects [169, 171]. Furthermore, certain medications can interact with B vitamins, altering their metabolism and effectiveness. For example, pyridoxine can reduce the efficacy of levodopa, a medication used for Parkinson’s disease [172]. In addition, proton pump inhibitors and metformin can reduce cobalamin absorption, increasing the risk of deficiency in long-term users [173, 174].
Despite the established role of the vitamin B complex in plastic and cosmetic surgery, several critical areas require further exploration to optimize its therapeutic applications (Table 3, Ref. [5, 27, 30, 31, 51, 52, 53, 66, 68, 70, 87, 96, 98, 100, 101, 102, 175, 176, 177] and Table 4 Ref. [33, 37, 39, 54, 64, 67, 69, 92, 175, 178, 179]). Research should focus on precision supplementation, innovative drug delivery systems, synergistic combinations with regenerative medicine, large-scale clinical trials, and the elucidation of epigenetic and anti-aging mechanisms. Addressing these research gaps will enable the development of more effective, personalized, and evidence-based strategies for enhancing surgical outcomes, promoting tissue regeneration, and improving post-surgical recovery.
| B-Vitamin | Primary Role in Surgery | Mechanisms & Effects | Key Findings | References |
|---|---|---|---|---|
| Thiamine (B1) | Postoperative nerve function and cellular resilience | Supports carbohydrate metabolism, ATP production, and neuronal function; modulates oxidative stress and reduces neuropathic pain. | Enhances ATP synthesis for neuronal recovery, reduces oxidative stress, and improves nerve conduction and Schwann cell function. | [5, 27, 87, 176] |
| Niacinamide (B3) | Skin restoration, anti-aging, and post-laser recovery | Enhances mitochondrial function, NAD⁺ production, and DNA repair; improves skin barrier integrity, hydration, and pigmentation control. | Stimulates ceramide production for skin hydration, inhibits melanosome transfer to reduce hyperpigmentation, and enhances post-laser wound healing. | [30, 31, 175] |
| Pantothenic Acid (B5) | Wound healing and scar management | Essential for CoA synthesis, accelerating cellular repair, modulating inflammatory responses, and supporting collagen synthesis. | Dexpanthenol promotes keratinocyte proliferation and fibroblast activity, reduces oxidative stress, and improves healing outcomes in various wound models. | [51, 52, 53] |
| Pyridoxine (B6) | Neuroprotection and neurotransmitter synthesis | Functions as a coenzyme in neurotransmitter production; modulates excitotoxicity and supports pain modulation. | Regulates glutamate release to prevent excitotoxic damage, supports serotonin and dopamine synthesis, and alleviates postoperative neuropathic pain. | [101, 102, 177] |
| Biotin (B7) | Hair regeneration and follicular support | Supports keratin synthesis, enhances follicular proliferation, and contributes to scalp microbiome balance. | Applied in hair transplant recovery to improve follicular survival; enhances hair regrowth in alopecia and reduces drug-induced hair loss. | [66, 68, 70] |
| Cobalamin (B12) | Nerve regeneration and postoperative recovery | Essential for myelin sheath maintenance, axonal repair, and nerve impulse conduction; reduces nerve damage and facilitates recovery. | High-dose methylcobalamin accelerates nerve regeneration, reduces neuropathic pain by inhibiting pro-inflammatory cytokines, and enhances Schwann cell proliferation. | [96, 98, 100] |
| Vitamin | Study Type & Sample | Intervention | Key Findings | |
| Alemanno et al. [92] | Thiamine (B1) | Retrospective clinical study; 110 patients undergoing interscalene block for shoulder surgery | 2 mg/kg thiamine hydrochloride added to 0.75% levobupivacaine vs. levobupivacaine alone | Prolonged postoperative analgesia duration (17.6 h vs. 11.4 h; p |
| Lee et al. [33] | Niacinamide | Randomized Controlled Trial (split-face); 25 patients | ADSC-CM combined with 2% niacinamide post-laser | Improved wrinkles, pigmentation, and collagen levels. |
| Hakozaki et al. [39] | Niacinamide | Randomized Controlled Trial; 18 Japanese women | 5% niacinamide vs. vehicle | Reduced hyperpigmentation and increased skin brightness. |
| Navarrete‐Solís et al. [37] | Niacinamide | Randomized Controlled Trial; 27 women | 4% niacinamide vs. 4% hydroquinone | Effectively treated melasma with fewer side effects than hydroquinone. |
| Chiu et al. [175] | Niacinamide & Kinetin | Randomized Controlled Trial (split-face); 52 subjects | 4% niacinamide combined with kinetin vs. placebo | Produced synergistic improvement in skin spots, pores, and wrinkles. |
| Heise et al. [54] | Panthenol | Randomized Controlled Trial; 38 patients | Dexpanthenol ointment vs. petroleum jelly | Accelerated wound healing and improved cosmetic outcomes. |
| Li et al. [64] | Panthenol, Madecassoside & Trace Elements | Randomized Controlled Trial (split-face); 43 women | Cream containing 5% panthenol, madecassoside, and trace elements | Improved healing post-laser treatment and reduced erythema and TEWL. |
| Samadi et al. [69] | Biotin & Dexpanthenol | Randomized Controlled Trial (double-blind); 50 patients | Intramuscular biotin combined with dexpanthenol | Increased hair density and improved terminal/vellus hair ratio; Bayer product was more effective. |
| El‐Esawy et al. [67] | Biotin & Zinc | Observational study; 60 men with androgenetic alopecia | Serum biotin and zinc level comparison | Found lower biotin and zinc levels in AGA patients; suggested correlation with hair loss. |
| Stücker et al. [178] | Cobalamin (B12) | Randomized Controlled Trial (placebo-controlled); 49 patients with atopic dermatitis | Topical vitamin B12 cream vs. base cream | Significantly improved symptoms and showed excellent tolerability. |
| Elgharably et al. [179] | Cobalamin (B12) | Clinical supplementation report | 1000 mcg sublingual vitamin B12 for dermatologic support | Improved skin and hair complaints associated with vitamin B12 deficiency. |
ADSC-CM, adipocyte-derived stem cell-conditioned media; AGA, androgenetic alopecia.
The metabolic processing and physiological effects of B vitamins vary significantly among individuals due to genetic polymorphisms, enzymatic activity differences, dietary factors, and gut microbiota composition [180]. This variability necessitates a shift from generalized supplementation protocols toward precision medicine approaches that tailor B vitamin intake based on individual metabolic profiles and surgical requirements [180, 181, 182]. Investigating genomic markers such as MTHFR mutations, which affect folate and cobalamin metabolism, to determine optimal dosages for enhanced skin repair, nerve protection, and collagen synthesis [183]. Additionally, nutrigenomic and metabolomic studies should assess how gut microbiota influences the absorption and bioavailability of B vitamins, particularly biotin and pantothenic acid, which play key roles in wound healing and scar management [184, 185]. Developing biomarker-based diagnostic tools for preoperative vitamin B screening can ensure patients receive adequate supplementation to enhance post-surgical recovery while preventing deficiencies that may lead to delayed wound healing, poor neural regeneration, and increased scar formation [186].
While the therapeutic benefits of B vitamins are well recognized, their rapid metabolism and excretion present challenges in maintaining sustained therapeutic efficacy. To overcome these limitations, further studies should explore the development of advanced drug delivery systems, including liposomal encapsulation, nanoemulsions, and polymeric microneedles, to enhance targeted delivery, sustained release, and localized action in plastic and cosmetic surgery applications [187, 188]. Liposomal formulations, for instance, have shown improved penetration into dermal and neural tissues, making them valuable in postoperative wound healing, scar modulation, and skin rejuvenation [188, 189]. Microneedle patches infused with niacinamide and dexpanthenol offer a promising minimally invasive approach for enhancing skin barrier function and pigmentation control in post-laser treatments and hyperpigmentation disorders [190, 191]. Additionally, hydrogel-based carriers loaded with cobalamin and pantothenic acid could serve as bioactive wound dressings, accelerating tissue regeneration by promoting fibroblast proliferation, extracellular matrix (ECM) remodeling, and angiogenesis [192, 193]. More research should focus on transdermal and implantable formulations that ensure prolonged vitamin release, reducing the need for frequent supplementation and optimizing therapeutic outcomes in aesthetic and reconstructive procedures.
The integration of vitamin B complex with regenerative medicine techniques holds significant potential for advancing plastic and reconstructive surgery. Several studies suggest that niacinamide enhances mitochondrial function and stem cell viability, making it a valuable adjunct in MSC therapy for skin rejuvenation and hair follicle regeneration [127, 194]. Additionally, vitamin B derivatives such as cobalamin and pyridoxine have an important role in Schwann cell proliferation and nerve regeneration, indicating their potential for incorporation into biodegradable nerve conduits for peripheral nerve repair [176, 195]. Future research should investigate how vitamin B complex interacts with PRP and ECM scaffolds to synergistically promote tissue regeneration, modulate inflammatory responses, and accelerate post-surgical recovery. Furthermore, exploring the role of vitamin B-enriched bioinks in 3D bioprinting could pave the way for engineered skin grafts and customized reconstructive implants that optimize wound healing and aesthetic outcomes in plastic surgery. Nevertheless, current dosing recommendations for vitamin B supplementation are based primarily on nutritional requirements rather than the specific demands of perioperative care and tissue repair. Therefore, more researches need to focus on defining optimal dosages, duration of supplementation, and mode of administration for different surgical procedures, ensuring maximum efficacy with minimal risk [196, 197]. Efforts should be made to elucidate the metabolic effects of chronic vitamin B supplementation and identify safe upper intake levels for patients undergoing prolonged reconstructive and cosmetic treatments [167, 198]. Additionally, post-surgical clinical trials should evaluate objective measures of functional recovery, such as scar elasticity, skin hydration, and nerve conduction velocity, to determine the long-term benefits and risks of vitamin B complex therapy in plastic surgery patients [199, 200]. Finally, future studies should explore how NAD⁺ precursors modulate fibroblast lifespan, collagen biosynthesis, and dermal remodeling, particularly in post-surgical scar prevention and anti-aging therapies [201, 202].
The vitamin B complex offers substantial therapeutic potential in enhancing the outcomes of plastic and cosmetic surgery. Its benefits extend beyond basic metabolic functions, aiding in skin restoration, wound healing, anti-aging therapies, and nerve protection. While the evidence supporting its role in these areas is promising, further research is needed to optimize dosing regimens, delivery methods, and the precise targeting of deficiencies based on individual patient profiles. Future studies should focus on integrating vitamin B complex into regenerative medicine and precision medicine frameworks, ensuring personalized care that maximizes therapeutic outcomes. Given its safety profile and multifaceted therapeutic roles, the vitamin B complex remains a critical element in advancing the clinical practice of plastic and cosmetic surgery.
Conceptualization: HL, FD, and AA; Data collection & analysis: AA, XZ, and WZ; Supervision: HL, and AA; Writing original draft: FD, and AA; Writing - review & editing: XZ, HL, and WZ. All authors have contributed to the editorial changes made to the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



