1 Department of Pharmacy, School of Pharmacy and Public Health, Independent University, Bangladesh (IUB), 1229 Dhaka, Bangladesh
2 Department of Medical Biotechnology, Faculty of Science, University of Technology Sydney (UTS), Sydney, NSW 2007, Australia
3 Department of Biomedical Science, School of Health and Life Sciences, Teesside University, TS1 3BX Middlesbrough, UK
4 Department of Pharmaceutical Technology, Faculty of Pharmacy, University of Dhaka, 1000 Dhaka, Bangladesh
†These authors contributed equally.
§These authors contributed equally.
Abstract
Diabetes mellitus (DM) is a chronic metabolic disorder characterized by persistent hyperglycemia and associated with severe complications, including cardiovascular diseases, neuropathy, nephropathy, and retinopathy. Although synthetic antidiabetic drugs are available, the side effects and limited long-term effectiveness of these medications highlight the urgent need for safer, more potent alternative therapies. Ginkgo biloba L., a traditional medicinal plant rich in flavonoids, terpenoids, and bilobalide, has attracted attention for its potential role in diabetes management. This review critically evaluates the antidiabetic potential of G. biloba by analyzing evidence from in vitro, in vivo, and clinical studies. Moreover, this review highlights the pharmacological actions of G. biloba and its key bioactive compounds, focusing on their mechanisms of action, including the activation of adenosine monophosphate-activated protein kinase (AMPK), the translocation of glucose transporter type 4 (GLUT4), and the inhibition of protein tyrosine phosphatases. The review also discusses the therapeutic implications of G. biloba supplementation and identifies gaps in clinical validation, optimal dosing, and safety profiling. Preclinical studies have demonstrated that G. biloba improves glycemic control by enhancing glucose uptake, regulating insulin secretion, inhibiting α-glucosidase activity, and exerting antioxidant and anti-inflammatory effects. Additionally, clinical trials suggest that supplementation with G. biloba can reduce oxidative stress, improve lipid profiles, and mitigate diabetes-related complications. However, despite these promising outcomes, inconsistencies remain in present study designs, dosages, and patient populations, which question the validity of results. Furthermore, studies related to the antidiabetic effect and underlying mechanisms of G. biloba, such as modulation of AMPK pathways and GLUT4 expression, also remain inadequate and warrant further systematic investigation. G. biloba may still be considered a complementary treatment approach in managing diabetes due to its broad pharmacological activities and favorable safety profile. However, well-designed, large-scale clinical trials are crucial for establishing standardized dosing regimens, confirming long-term safety, and fully elucidating the mechanisms of action. Integrating G. biloba into therapeutic strategies could offer a natural, and effective adjunct for enhancing glycemic control and reducing diabetes-related complications.
Keywords
- diabetes
- G. biloba
- glucose metabolism
- insulin resistance
- active compound
Diabetes mellitus (DM) is a common metabolic disorder that affects global
health, often leading to mild to severe complications [1]. It has become a
significant public health concern in the twenty-first century [2], and is usually
described as an emerging global health crisis. In 2021, approximately 537 million
people aged 20 to 79 had DM, with figures expected to reach 643 million by 2030
and 783 million by 2045, thus posing significant challenges for patients and
healthcare providers [3, 4]. DM is characterized by prolonged high blood glucose
levels resulting from defective insulin secretion, insulin function, or both [5].
Diabetes is primarily classified into type 1 (T1D) and type 2 (T2D), each
characterized by its differences in clinical and mechanistic features. T1D is
associated with autoantibodies, loss of
Medicinal plants have been valued in traditional medicine as alternative treatments for decades, mainly when supported by scientific validation. Compared to synthetic drugs, they are often more affordable, with fewer adverse effects, and therapeutic benefits [11]. Despite advancements in phytochemical-based therapies for diabetes, its global prevalence and associated complications remain high, underscoring the urgent need for discovering more effective therapeutic compounds. Phytochemicals derived from herbs are increasingly recognized for their potential in managing metabolic disorders, particularly dyslipidemia and hyperglycemia [12]. Ginkgo biloba, the oldest surviving gymnosperm, often called “the living fossil”, has been used in traditional Chinese medicine for centuries. This dioecious tree exhibits diverse pharmacological properties, including neuroprotective, antioxidant, anticancer, hepatoprotective, and cardiovascular benefits, attributed to its roles as a free radical scavenger, membrane stabilizer, and platelet-activating factor inhibitor [13, 14]. It is widely marketed as a dietary supplement for metabolic disorders; however, its regulatory status and safety can vary depending on the country, thus concerns over potential adverse effects have been raised in some contexts [15]. Numerous preclinical and clinical studies have explored G. biloba’s potential for improving metabolic parameters. However, evidence regarding its specific antidiabetic mechanisms, such as adenosine monophosphate-activated protein kinase (AMPK) activation and GLUT4 translocation, remains fragmented and inconclusive, highlighting the need for a comprehensive systematic review [16]. G. biloba contains unique bioactive constituents, including acylated flavonol glycosides (ginkgoghrelins), ginkgolic acids, biflavones (ginkgetin), ginkgotides, and terpene trilactones (ginkgolides), making it valuable in herbal medicine. Studies investigating its leaf extracts, seeds, and key compounds like ginkgolide B and C have demonstrated beneficial effects such as weight loss, reduced adipose tissue, improved lipid profiles, enhanced glucose and insulin regulation, and positively impacting kidney function [17]. However, while many investigations have confirmed G. biloba’s antioxidant and general metabolic benefits, its direct effects on hyperglycemia, insulin resistance, and glucose metabolism are inconsistently reported across different experimental models [18]. Moreover, specific modulation pathways such as AMPK activation and GLUT4 translocation have not been comprehensively reviewed in the context of G. biloba for diabetes therapy, representing a significant knowledge gap. Given the escalating global burden of diabetes and the limitations of current pharmacotherapies, a deeper understanding of plant-based alternatives like G. biloba could offer promising complementary strategies. Building on traditional medicine knowledge and emerging antidiabetic research, this study critically reviews G. biloba’s role in controlling hyperglycemia and modulating key metabolic pathways. It aims to provide clearer, detailed insights compared to previous general studies, while highlighting future directions for therapeutic development.
A comprehensive literature search was conducted using Google Scholar, PubMed, Cochrane Library, ScienceDirect, and Scopus databases. The search utilized the following keyword combinations with Boolean operators to maximize relevance and search coverage: “G. biloba” AND (“Diabetes Mellitus” OR “Diabetes” OR “Hyperglycemia”) AND (“antidiabetic” OR “insulin resistance” OR “glucose metabolism”). Additional keywords such as “isolated compounds” AND (“in vitro” OR “in vivo” OR “clinical trials”) were also employed to narrow the focus on studies that investigated the antidiabetic properties of compounds isolated from G. biloba. This review was limited to English-language studies published up to December 11, 2024, to ensure accessibility and ease of understanding. The selection process adhered to specific inclusion criteria, which included in vitro and in vivo studies, clinical trials, and research focusing on the antidiabetic effects of G. biloba and its isolated compounds. Key data extracted from the selected studies included: year of publication, tested compounds, experimental models (e.g., cell lines, animal models), observed outcomes, results, tested concentrations, and molecular mechanisms underlying the observed effects. Studies that did not meet these criteria or failed to report antidiabetic effects of G. biloba or its compounds were excluded from the review.
In this study, 85 articles reported the antidiabetic effects of G. biloba through in vitro, in vivo, and clinical trials. Most of these studies focused on G. biloba leaves or its isolated compounds. Tables 1A,1B,1C (Ref. [19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85]) provides an overview of the in vitro and in vivo studies examining the antidiabetic properties of G. biloba and its bioactive compounds.
| Test model | Parts | Dose/concentration | Route of administration | Mechanism of action | Formulation | Ref. |
| Leaves | 10, 25, and 50 mg/mL | In vitro, Enzyme | ↓ |
Water and Ethanol | [19] | |
| Leaves | - | In vitro, Enzyme | Potent inhibition of |
Undefined | [20] | |
| Seed | 20 mg/mL | In vitro, Enzyme | Alkali-soluble acid | [21] | ||
| AMPK | Leaves | 20 and 40 µg/mL | In vitro, Enzyme | ↑ GLUT4 translocation to the membrane and subsequent ↑ glucose uptake in C2C12 myotubes activated the AMPK pathway. | Petroleum ether | [22] |
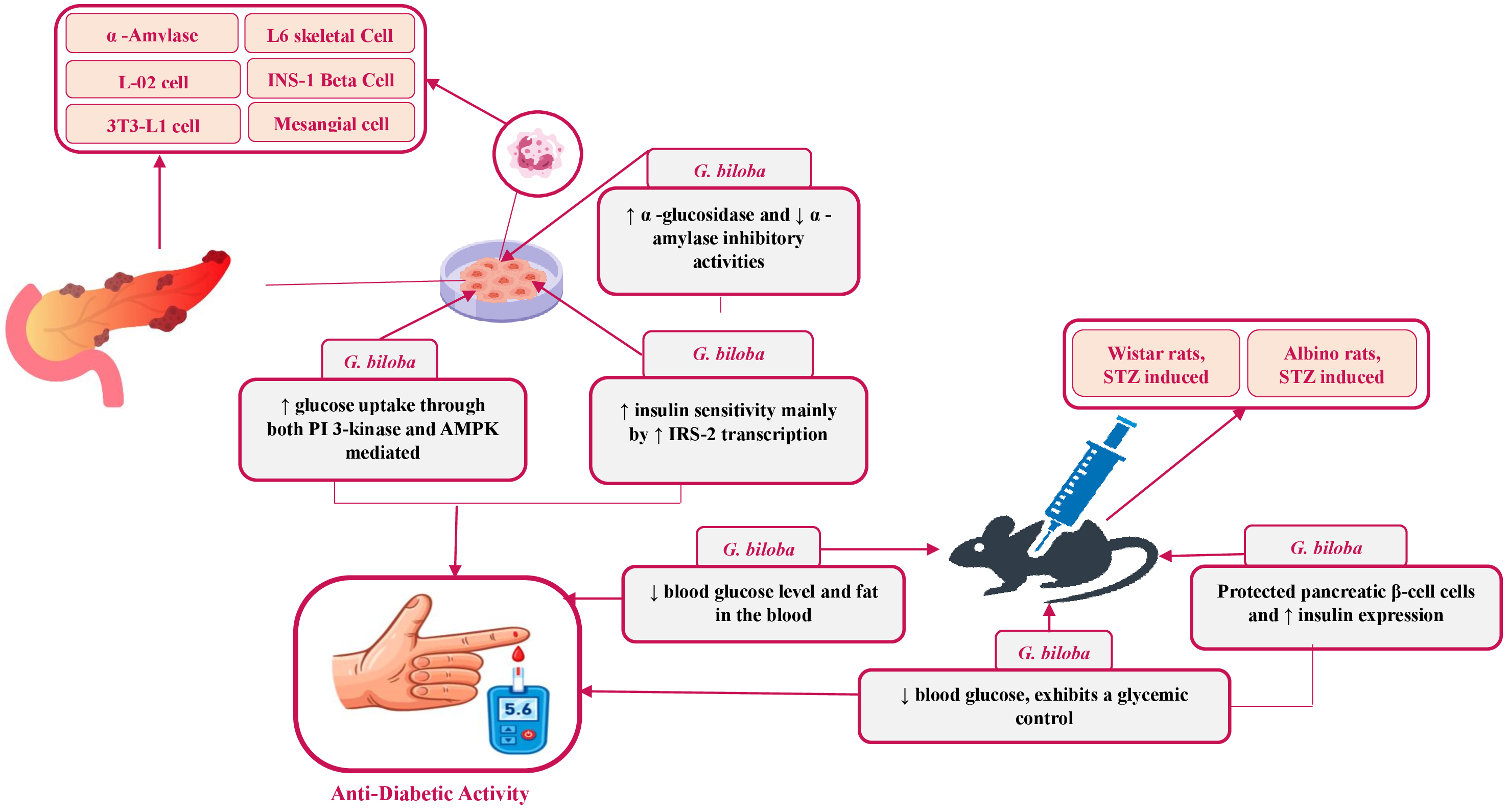
| L6 skeletal muscle cell | Leaves | 50 µg/mL | In vitro, Cell line | ↑ Glucose uptake through both PI 3-kinase and AMPK mediation leads to glucose homeostasis. | Methanol | [23] |
| SMC | Leaves | 0.25 µg/mL | In vitro, Cell line | Artificially mimic insulin’s effects on glucose metabolism and transport. | Ethanol | [24] |
| BMSCs | Leaves | 200 µg/mL | In vitro, Cell line | ↑ The effectiveness of BMSCs resulted in ↓ the reversal of blood glucose levels and reversing oxidative stress. | Ethanol | [25] |
| L-02 cell line | Leaves | 10 mg/L | In vitro, Cell line | ↑ Insulin sensitivity is mainly regulated by ↑ IRS-2 transcription. | Undefined | [26] |
| INS-1 rat beta cells | Leaves | 50 g/mL | In vitro, Cell line | ↑ Insulin secretion. | Undefined | [27] |
| Preadipocytes (Swiss 3T3-L1 cells) | Leaves | 0.75 and 1.0 mg/mL | In vitro, Cell line | ↑ Preadipocyte maturation, ↑ expression of mature adipocyte proteins. | Undefined | [28] |
| Mesangial cell | Leaves | 30 mmol/L | In vitro, Cell line | ↓ Levels of TGF- |
Undefined | [29] |
| HUVECs | Leaves | 25, 50, 100 µg/mL | In vitro, Cell line | ↓ IHG-induced DNA oxidation of endothelial cells. | G. biloba extract (EGb 761) | [30] |
| 3T3-L1 cells | Seed | 0.4% ginkgo vinegar | In vitro, Cell line | ↓ C/EBPδ and PPAR |
Ginkgo vinegar | [31] |
| HAECs | Leaves | 100 µg/mL | In vitro, Cell line | ↓ Adhesion molecule expression and endothelial adhesion by ↑ HO-1 via PI3K/Akt/eNOS and p38/MAPK pathways. | Undefined | [32] |
| HLEC | Leaves | 10, 20, and 40 µg/mL | In vitro, Cell line | ↓ Apoptosis is inhibited by inhibiting oxidative stress, ↓ the Bax/Bcl-2 ratio, and ↓ caspase-3 activity. | Undefined | [33] |
| EGCs | 25, 50, 100 µg/mL | In vitro, Cell line | ↓ Hyperglycemic. | Undefined | [34] |
AMPK, Adenosine monophosphate-activated Protein Kinase; SMC, Smooth muscle cells; BMSCs, Bone marrow mesenchymal stem cells; GLUT4, Glucose
transporter type 4; IRS-2, Insulin receptor substrate 2; TGF-
| Test model | Parts | Dose/concentration | Route of administration | Mechanism of action | Formulation | Ref. |
| Wistar rats, STZ induced | Leaves | 100, 200, and 300 mg/kg | I.P | Antihyperglycemic, antioxidant, and antihyperlipidemic activities. | Undefined | [35] |
| Wistar rats, STZ induced | Leaves | - | I.P | ↓ Serum glucose. | Undefined | [36] |
| Wistar rat, STZ induced | Leaves | 0.11 g/kg/day | P.O | Protected |
Ethanol | [37] |
| Wistar male rats, STZ induced | Leaves | 50, 100, 150 mg/kg/day | Orally | Anti-hyperglycemic effects by upregulated GLUT-4 and IRS-1 in hepatic tissues. | Undefined | [38] |
| Wistar rats, STZ induced | Leaves | 50 mg/kg | I.P | Cardioprotective effects against late complications of diabetes are helpful as an adjuvant therapy for the prevention of diabetic cardiomyopathy. | Ethanol | [39] |
| Wistar rats, STZ induced | Leaves | 200 mg/kg | P.O | Did not improve maternal glycemia, pregnancy rate, antioxidant enzymes, or fetal development in diabetic rats. | Undefined | [40] |
| Wistar rats, STZ induced | Leaves | 0.11 g/kg | I.P | ↓ Hyperlipidemia, uremia, oxidative stress, and renal dysfunction. | Undefined | [41] |
| Wistar rats, STZ induced | Leaves | 100 mg/kg | Intragastrically | ↓ Blood glucose. | Ethanol | [42] |
| Wistar rats, STZ induced | Leaves | 50 mg/kg | Gavage | Neuroprotective effects on the jejunum and ileum plexuses, supporting enteric nervous system integrity in diabetes. | Undefined | [43] |
| Male Wistar rats, STZ induced | Leaves | 100 mg/kg/day | P.O | ↓ Blood glucose and plasma drug concentrations. | Undefined | [44] |
| Male Wistar rats, STZ induced | Leaves | 100 mg/kg/day | P.O | Effects of diabetes on enzyme activities. | Undefined | [45] |
| Male ApoE−/− mice, STZ induced | Leaves | 200 or 400 mg/kg | Gavage | Blocking ERS, ↓ diabetic myocardial damage, including intramyocardial inflammation, interstitial fibrosis, and cardiomyocyte death. | Ethanol | [46] |
| Mice, STZ induced | Leaves | 50 mg/kg | P.O | ↓ Blood triglyceride levels, ↑ liver LPL, ↑ liver PPAR- |
Undefined | [47] |
| Rats, STZ induced | Leaves | 500 mg/kg GBE diluted in 2 mL 0.9% | Gavage | ↓ Hyperglycemia, dyslipidemia, ↓ food intake, and body adiposity. ↓ PTP-1B levels in the gastrocnemius muscle, ↑ insulin sensitivity, Akt phosphorylation, and IRS-1. | Undefined | [48] |
| Albino rats, STZ induced | Leaves | 120 mg/kg | P.O | ↓ The levels of glucose and fat in the blood. | Undefined | [49] |
| Albino rats, STZ induced | Leaves | 100 mg/kg | P.O | ↑ Prostatic changes produced by DM. | Undefined | [50] |
| Albino rats, STZ induced | Leaves | 100 mg /kg daily | Intraperitoneal | ↓ Blood glucose level. | The capsule was dissolved in saline | [51] |
| Albino Wistar rats, STZ induced | Leaves | 50 and 100 mg/kg/day, | Orally | ↓ FBS and ↑ in blood GSH. | Undefined | [52] |
| Albino rats, STZ induced | Leaves | 100 mg/kg/day | P.O | Protects against diabetes-induced prostate damage. | Undefined | [53] |
| DBA/2 mice, STZ induced | Leaves | low dose (50 mg/kg/day); high dose (200 mg/kg/day) | P.O | Protected podocytes from hyperglycemia and prevented DN through Nrf-2/HO-1 activation. | Undefined | [54] |
| OLETF rats | Leaves | 100 and 200 mg/kg | P.O | Protects against atherosclerosis and may aid in its prevention. | Undefined | [55] |
| Male ApoE−/− mice, STZ induced | Leaves | low-dose GBE group (200 mg/kg/day), and high-dose GBE group (400 mg/kg/day) | Intragastric | ↓ Serum lipid metabolism levels, blood glucose, and inflammatory cytokines. | Undefined | [56] |
| Normoglycemic male adult Wistar rats | Leaves | 100 mg/kg/day | Orally | ↓ Hyperglycemic activity. ↑ Glucose and lipid metabolism act on the expression of key genes. | Undefined | [57] |
| Wistar albino rats, STZ induced | Leaves | 100 mg/kg/day | Orally | Exhibiting antidiabetic, antihypertensive, and antilipidemic effects. | Undefined | [58] |
| Sprague-Dawley rats | Leaves | 40 mg/kg | P.O | Preventive effect against hyperlipidemia. | Undefined | [59] |
| Adult male Sprague‐Dawley rats, STZ induced | Leaves | 50, 100, and 200 mg/kg/day | Orally | ↓ Blood glucose exhibits glycemic control. | Dissolved in 1% CMC‐Na at 10, 20, and 40 mg/mL concentrations. | [60] |
| Sprague Dawley rats, STZ induced | Leaves | 200 mg/kg/day | Orally | Antihyperglycemic and antilipidemic effects, ↑ such as glucose metabolism in the liver by modulating ↑ the hepatic key genes. | Undefined | [61] |
| Male Sprague-Dawley rats | 200 mg/kg | Orally | ↑ Pancreatic |
Methanol | [62] | |
| HFD-induced Mice | Leaves | 100, 200 and 400 mg/kg | P.O | ↓ Insulin resistance and ameliorate other symptoms of the metabolic syndrome. | Undefined | [63] |
STZ, streptozotocin; I.P, intraperitoneally; P.O, oral administration; OLETF, Otsuka Long-Evans Tokushima Fatty; GLUT-4, glucose transporter type 4; IRS-1, insulin receptor substrate 1; GBE, Ginkgo biloba extract; FBS, fasting blood sugar; GSH, glutathione; Nrf-2, nuclear factor erythroid 2-related factor 2; HO-1, heme oxygenase-1; HFD, high-fat diet; CMC-Na, sodium carboxymethylcellulose.
| Isolated compounds | Test model | Parts | Dose/concentration | Route of administration | Mechanism of action | Formulation | Ref |
| Bilobalide (1) | Male Wistar rats, STZ | Leaves | 10 mg/kg | Intraperitoneal | ↓ Blood glucose and activating insulin secretion. | Undefined | [64] |
| 3T3-L1 preadipocyte | - | 0–200 µM | In vitro, Cell line | Inhibited adipogenesis and ↑ lipolysis in 3T3-L1 cells via AMPK activation. | Undefined | [65] | |
| Female NMRI mice, alloxan-induced | Leaves | 100 & 200 mg/kg | Administered orally by gavage (stomach tube) | ↑ Effect of glucose and tolbutamide. | Undefined | [66] | |
| 3T3-L1 preadipocytes | - | 10, 20 & 50 µM | In vitro, Cell line | ↓ Inflammatory adipokine secretion, ↑ adiponectin secretion, ↓ NF- |
Undefined | [67] | |
| 3T3-L1 preadipocytes | - | 10, 20, and 50 µM | In vitro, Cell line | Protected adipocytes from hypoxia by ↓ oxidative stress, mitochondrial damage, ↓ and cell death, and inflammation. | Undefined | [68] | |
| 3T3-L1 | Leaves | 25 µM, 50 µM | In vitro, Cell line | Potential novel obesity treatment. | Undefined | [69] | |
| adipocytes | |||||||
| Male Wistar rats | - | 2 mg/kg/day | In vivo, Streptozotocin | ↑ Glucose uptake is linked to glycogen synthesis. | Distilled water | [70] | |
| 2-hydroxy-6-(10′-hydroxypentadec-11′(E)-en-1-yl) benzoic acid (2), 2-hydroxy-6-(11′-hydroxypentadec-9′(E)-en-1-yl) benzoic acid (3), 2-hydroxy-6-tridecylbenzoic acid (4), 2-hydroxy-6-pentadecylbenzoic acid (5), 2-hydroxy-6-(12′-hydroxyheptadec-13′(E)-en-1-yl) benzoic acid (6), and 2-hydroxy-6-(11-hydroxyundecyl) benzoic acid (7) | PTP | Leaves | 50 µM | In vitro, Protein | Compounds 5–7 inhibited PTPN11, PTPN2, PTP1B, DUSP9, PTPRS, and PTPN9 by over 90%, while compounds 2 and 3 showed significant activity against PTPN11, PTPN2, PTP1B, and DUSP9, targeting PTPs related to insulin resistance. | Aqueous, methanol | [71] |
| Quercetin (8) | Leaves | 0.061 µM and 0.038 µM | In vitro, Enzyme | Potent |
Undefined | [72] | |
| Rutin (9) | Leaves | 0.043 and 0.037 µM | In vitro, Enzyme | Potent |
Undefined | [73] | |
| Ginkgolide C (10) | 3T3-L1 cells | Leaves | 3–100 µM | In vitro, Cell line | Inhibited adipogenesis-related factors, ↑ Sirt1/AMPK activity, ↑ lipolysis, ↑ metabolic syndrome, and insulin resistance. | Undefined | [74] |
| Kaempferol (11) | INS-1E cells and human islets | - | 0.1, 1, and 10 µM | In vitro, Cell line | ↑ |
Undefined | [75] |
| Ginkgetin (12) | 3T3-L1 cells | Leaves | 5 µM | In vitro, Cell line | Inhibited PPAR |
Undefined | [76] |
| C57BL/6 male mice | Leaves | 5 and 10 mg/kg/day | In vivo, intraperitoneal injection | Inhibited white adipose hypertrophy, showing potential as an anti-obesity drug. | Undefined | [77] | |
| Isoginkgetin (13), bilobetin (14), ginkgetin (12) and sciadopitysin (15) | pancreatic lipase | Leaves | 1, 10, and 100 µM | In vitro, Enzyme | Hypolipidemic effects. | Undefined | [78] |
| Isoginkgetin (13) | 3T3-L1 mouse fibroblasts | Leaves | 5 mM | In vitro, Cell line | ↑ Adiponectin production in adipocytes. ↑ Plasma adiponectin levels. | Undefined | [79] |
| Ginkgolide B (16) | HUVECs | Leaves | 0.2, 0.4, and 0.6 mg/mL | In vitro, Cell line | Inhibited TLR4-mediated inflammation and JAK2/STAT3, p38 MAPK signaling in high-glucose endothelial cells. | Undefined | [80] |
| MPC5 | Leaves | 0, 5, 10, 20, 40, 80, 100, 200 µM | In vitro, Cell line | ↓ Total cholesterol, hyperglycemia serum, lipid accumulation, and triglyceride concentrations. | Undefined | [81] | |
| Male ICR mice, streptozotocin | - | - | In vivo | Prevents endothelial dysfunction in the DM aorta. ↑ Aortic SOD1 activity and ↑ NO bioavailability in endothelial cells. | Undefined | [80] | |
| Ginkgolic acid (17) | Leaves | 13.8 and 40.1 µg mL (–1) | In vitro, Enzyme | ↑ |
Ethyl acetate, n-hexane, n-butanol, and chloroform | [81] | |
| Amentoflavone (18), bilobetin (14), sequoiaflavone (19), ginkgetin (12), sciadopitysin (15), isoginkgetin (13) | 3T3-L1 preadipocytes | Leaves | 0.005–100 µM | In vitro, Cell line | ↑ Lipolysis inhibits cAMP phosphodiesterase. | Undefined | [82] |
| (2E,4E,1′R,3′S,5′R,8′S)-dihydrophaseic acid 3′-O- |
Human hepatocarcinoma HepG2 cells | Stem bark | 10 ng/mL | In vitro, Cell line | Inhibited NF-κB activity and ↓ COX-2 and iNOS expression. | Methanol | [83] |
| Ginkgolides A, B, C (34, 16, 10), and bilobalides (1). | HFD-induced obese male Wistar rats | - | - | In vivo | ↓ Adipocyte. ↓ Acetate accumulation tended to ↓ [3H]-oleate incorporation into epididymal adipose tissue. | Undefined | [84] |
| Rutin (9), quercetin (8) | Adult male Sprague-Dawley rats, STZ | 90 mg/kg | In vivo | Inhibited the aldose reductase (AR) activity, ↑ production of glutathione, ↓ malondialdehyde (MDA), and advanced glycosylation end products (AGEs). | Undefined | [85] |
PTP, Protein tyrosine phosphorylation; GLUT-4,
Glucose transporter type 4; MPC5, Mouse renal podocytes; ICR, Institute of Cancer Research; NF-
The aqueous extract of Ginkgo leaves at a 50 mg/mL dose demonstrated significant
Skeletal muscle regulates blood glucose levels by facilitating glucose uptake
through insulin-dependent and insulin-independent pathways. It serves as the
primary site for glucose transport [22]. Two major independent signaling pathways
govern this process in response to various stimuli. PI3-kinase, activated by
insulin, is essential for promoting glucose uptake, with its impaired activation
in skeletal muscle leading to reduced glucose transport. AMPK, another key
regulatory protein primarily activated by cellular stress, plays a central role
in maintaining energy homeostasis. It consists of two catalytic subunit isoforms,
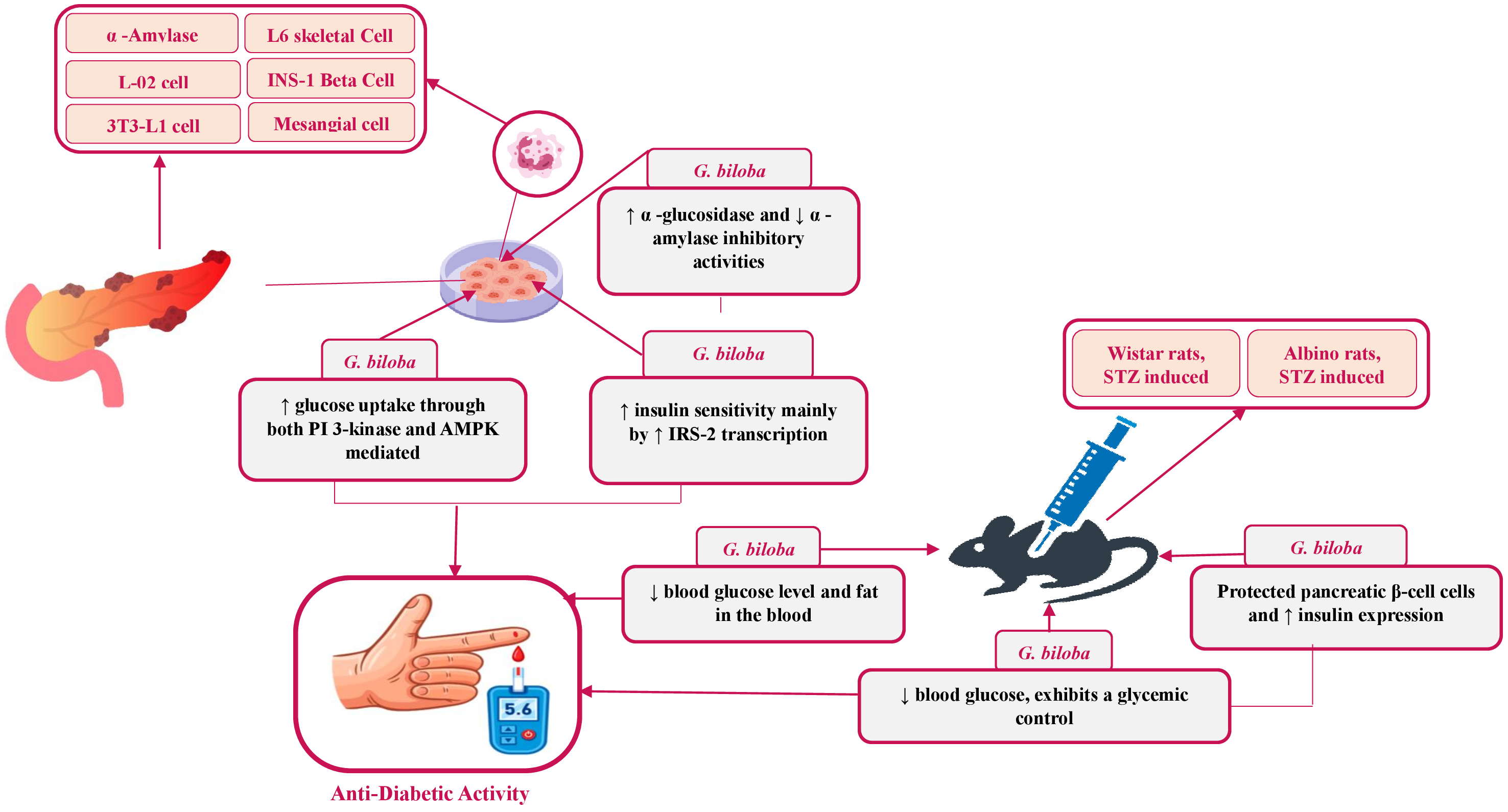 Fig. 1.
Fig. 1.
Proposed in vivo and in vitro mechanisms of action of G. biloba in antidiabetic activity. AMPK, Adenosine monophosphate-activated protein kinase; IRS-2, Insulin receptor substrate 2; PI 3-kinase, Phosphatidylinositol 3-kinase; STZ, Streptozotocin. The up sign (↑) indicates an increase, and the down sign (↓) indicates a decrease.
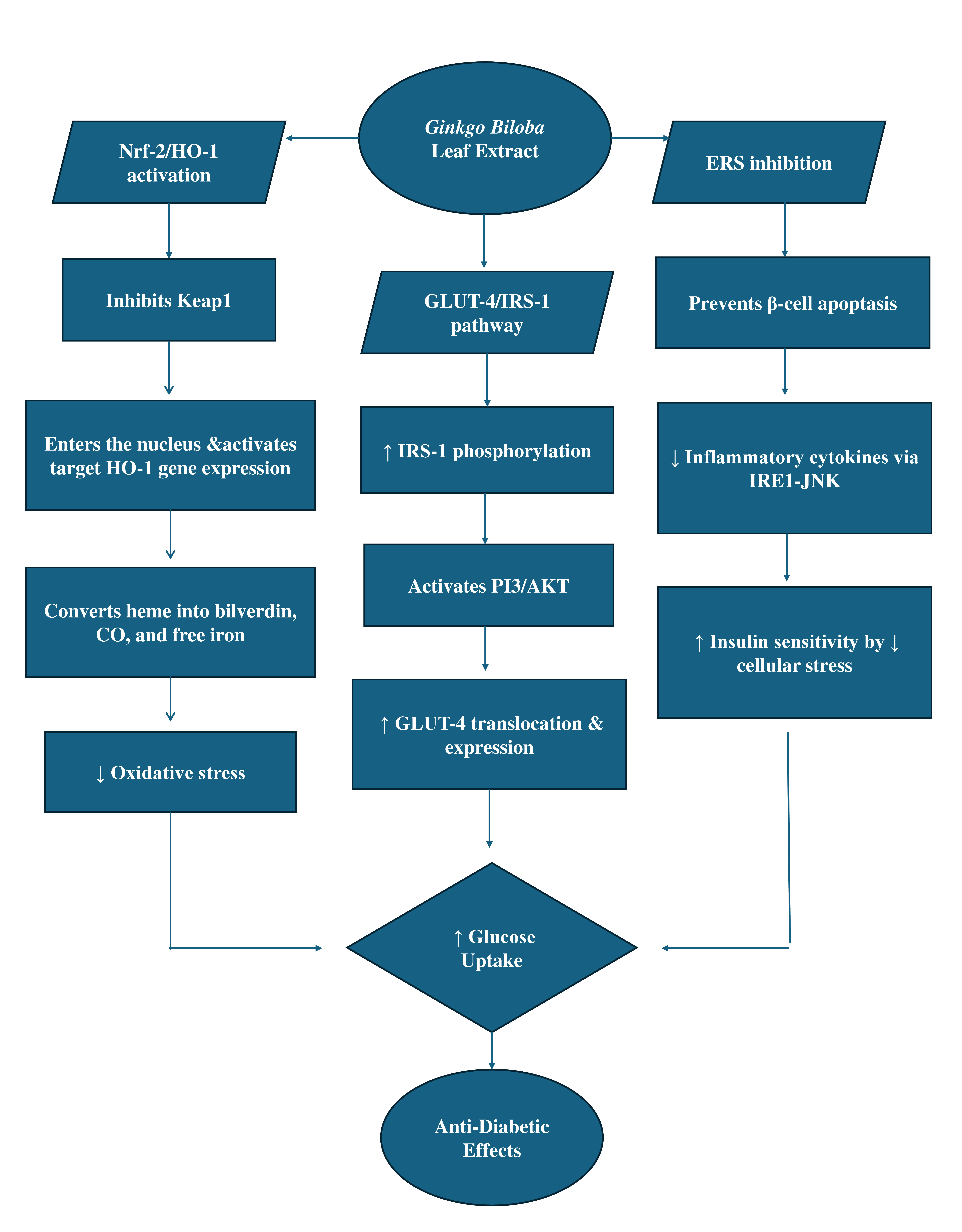
G. biloba leaf extract initiates its protective effects primarily
through activation of the Nrf-2/HO-1 pathway. The extract inhibits Keap1, a
negative regulator of Nrf-2, allowing Nrf-2 to translocate into the nucleus where
it binds to antioxidant response elements (ARE) and upregulates the expression of
the HO-1 gene. The HO-1 enzyme catalyzes the conversion of heme into biliverdin,
CO, and free iron, which collectively reduce oxidative stress in the cell.
Concurrently, G. biloba stimulates the GLUT-4/IRS-1 signaling pathway,
where it increases phosphorylation of IRS-1, activating downstream PI3K/AKT
signaling. This cascade promotes the translocation and expression of GLUT-4 on
the cell membrane, enhancing glucose uptake by cells. Additionally, G.
biloba inhibits endoplasmic reticulum stress (ERS), preventing apoptosis of
pancreatic
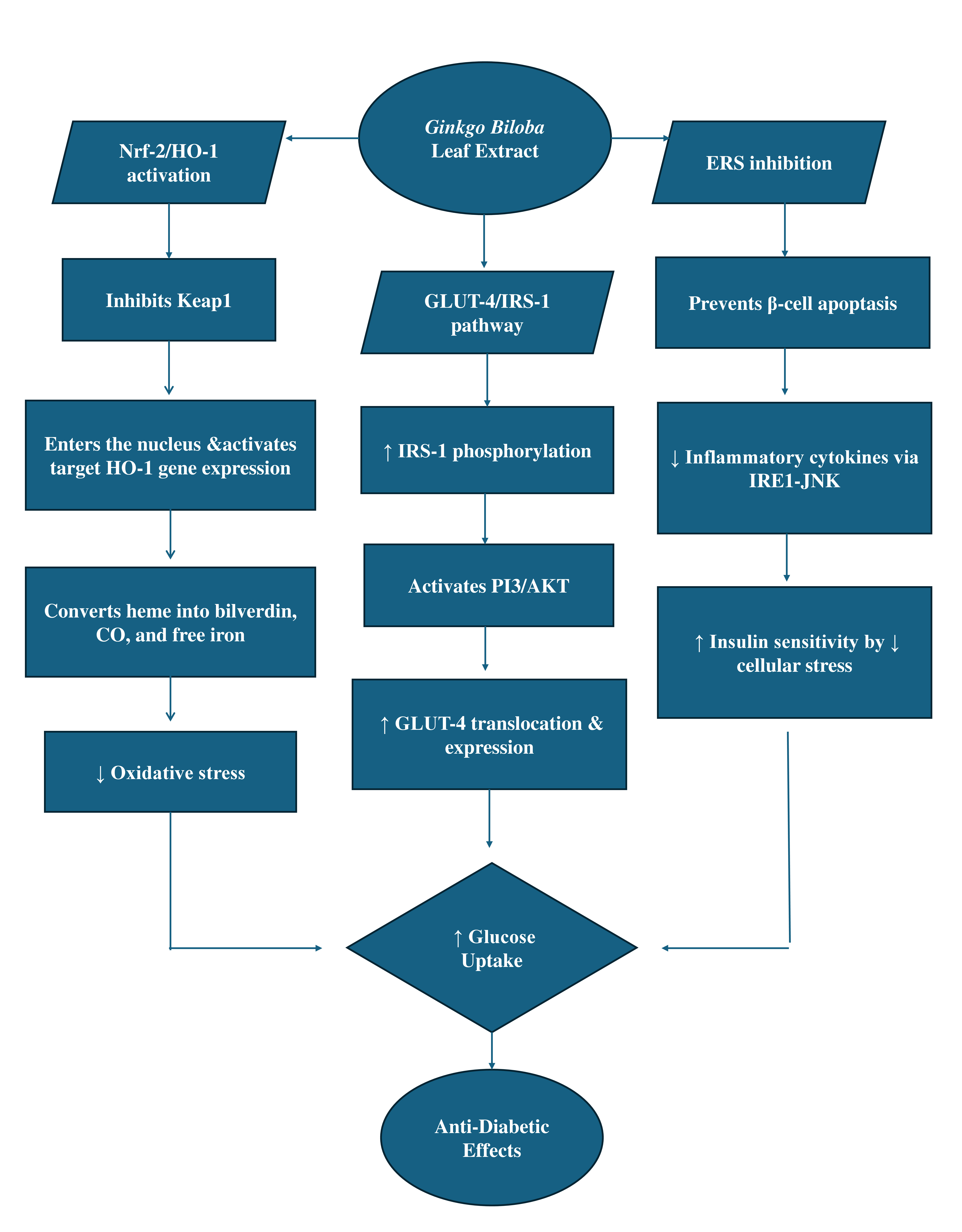 Fig. 2.
Fig. 2.
Mechanistic pathways of G. biloba in diabetes management, highlighting Nrf-2/HO-1-mediated antioxidant effects, enhancement of insulin signaling via GLUT-4/IRS-1, and ERS inhibition. GLUT4, Glucose transporter type 4; HO-1, Heme oxygenase-1; Nrf2, Nuclear factor erythroid 2-related factor 2; IRS-1, Insulin receptor substrate-1; ERS, Endoplasmic reticulum stress.
Another study reported that intraperitoneal administration of leaf extracts led
to a marked reduction in serum glucose levels. At the same time, the treated
diabetic rats maintained a higher body weight than the untreated group, further
highlighting the metabolic benefits of the extract [36]. Oral administration of
0.11 g/kg/day of ethanolic leaf extracts effectively preserved pancreatic
Intragastric administration of 100 mg/kg of ethanolic leaf extract in
streptozotocin (STZ)-induced diabetic Wistar rats significantly reduced blood
glucose levels, reinforcing its potential as an antidiabetic agent [42].
Similarly, gavage administration of 50 mg/kg of the extract exhibited a
neuroprotective effect on the jejunum submucous plexus and the myenteric plexus
of the ileum, suggesting its role in preserving enteric nervous system integrity
in diabetic conditions [43]. Oral administration of 100 mg/kg per day of the
extract in male Wistar rats reduced blood glucose levels and influenced plasma
drug concentrations, indicating potential pharmacokinetic interactions [44].
Additionally, another study reported that the same oral dosage modulated enzyme
activities affected by diabetes, suggesting a broader metabolic regulatory effect
[45]. In ApoE-/- diabetic mice, gavage administration of 200 or 400 mg/kg of
the ethanolic extract effectively reduced diabetic myocardial damage by blocking
endoplasmic reticulum stress (ERS), thereby preventing cardiomyocyte death,
interstitial fibrosis, and intramyocardial inflammation [46]. Furthermore, in
STZ-induced diabetic mice, 50 mg/kg of the extract significantly lowered blood
triglyceride levels while increasing peroxisome proliferator-activated
receptor-alpha (PPAR-
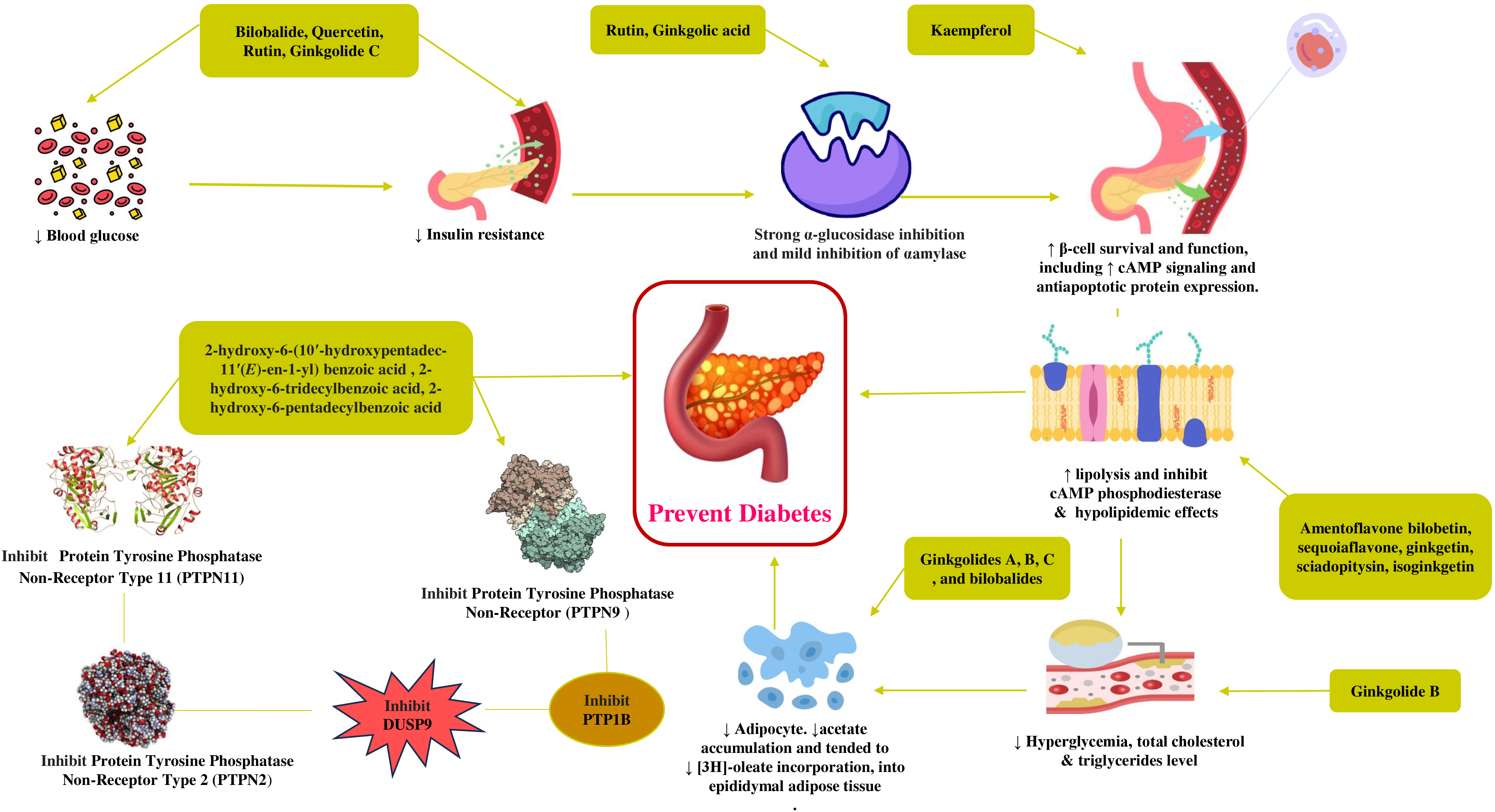
The bioactive derivatives such as bilobalide, quercetin, rutin, and ginkgolide C demonstrated lower blood glucose levels, improving the body’s sensitivity to insulin. The subsequent effect of lowered insulin resistance by rutin and ginkgolic acid was potentiated through slow release of glucose to blood after a meal by potent and mild inhibition of digestive enzymes such as alpha-glucosidase and alpha-amylase, respectively. Kaempferol from G. biloba protected beta cell damage by expressing antiapoptotic protein and improved insulin function by enhancing insulin signalling by cAMP. The bioactive derivatives 2-hydroxy-6-(10′-hydroxypentadec-11′(E)-en-1-yl) benzoic acid (2), 2-hydroxy-6-(11′-hydroxypentadec-9′(E)-en-1-yl) benzoic acid, 2-hydroxy-6-tridecylbenzoic acid, 2-hydroxy 6-pentadecylbenzoic demonstrated inhibition of multiple protein tyrosine phosphatases (PTPs) involved in insulin resistance such as PTPN11, PTPN2, PTPN9, PTP1B, and DUSP9. Ginkgolides A, B, C, and bilobalide derivatives, especially ginkgolide B, lowered hyperglycemic states, total cholesterol, triglyceride serum levels, and lipid accumulation associated with diabetes, thus maintaining adipose tissue. Amentoflavone, bilobetin, sequoiaflavone, ginkgetin, sciadopitysin, and isoginkgetin exerted hypolipidemic effects by reducing lipolysis and inhibiting the enzyme cAMP phosphodiesterase. A mechanism of action of G. biloba on glucose is given in Fig. 3.
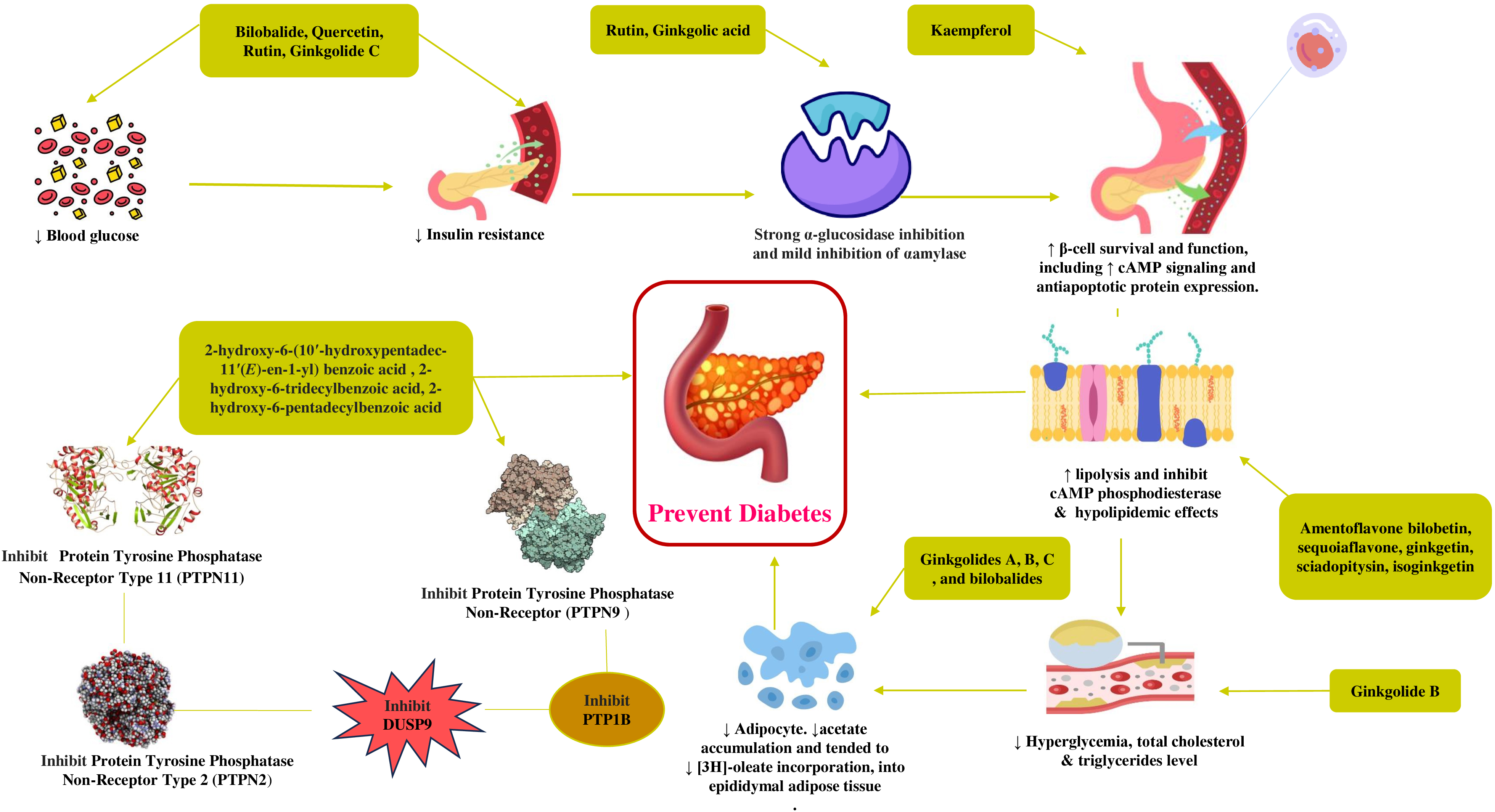 Fig. 3.
Fig. 3.
Proposed mechanism of action of G. biloba and its isolated compounds in exerting antidiabetic effects. cAMP, Cyclic adenosine monophosphate; PTPN2, Protein tyrosine phosphatase non-receptor type 2; PTPN9, Protein tyrosine phosphatase non-receptor type 9; PTPN11, Protein tyrosine phosphatase non-receptor type 11.
Bilobalide, when administered to male Wistar rats at a dose of 10 mg/kg
intraperitoneally, demonstrated a remarkable reduction in blood glucose levels,
activation of insulin secretion, and alleviation of liver damage caused by
streptozotocin (STZ)-induced diabetes. These findings suggest potential
therapeutic effects on diabetes-related complications [64]. In vitro,
bilobalide has been shown to inhibit adipogenesis and promote lipolysis in 3T3-L1
preadipocytes by activating the AMPK signaling pathway, further supporting its
role in modulating metabolic processes [65]. In female NMRI mice subjected to
alloxan-induced diabetes, bilobalide administered orally at doses of 100 and 200
mg/kg by gavage, in conjunction with EGb 761, enhanced the activity of beta cells
in the presence of glucose, indicating its synergistic effect on glucose
regulation and insulin sensitivity [66]. Additionally, bilobalide exhibited
protective properties in 3T3-L1 preadipocytes against hypoxia-induced
inflammation and insulin resistance. This was achieved by reducing inflammatory
adipokine secretion, enhancing adiponectin release, modulating the
NF-
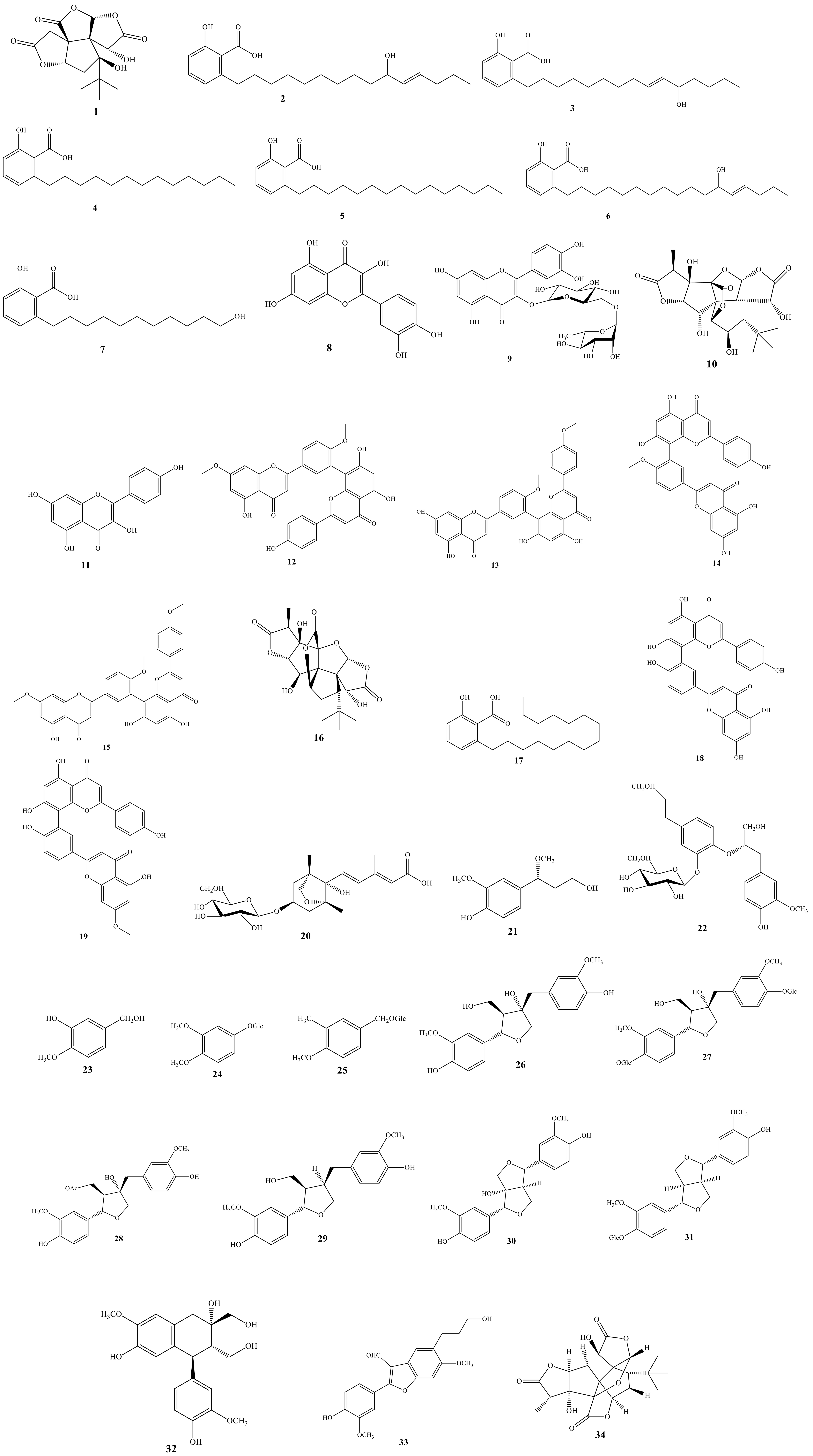
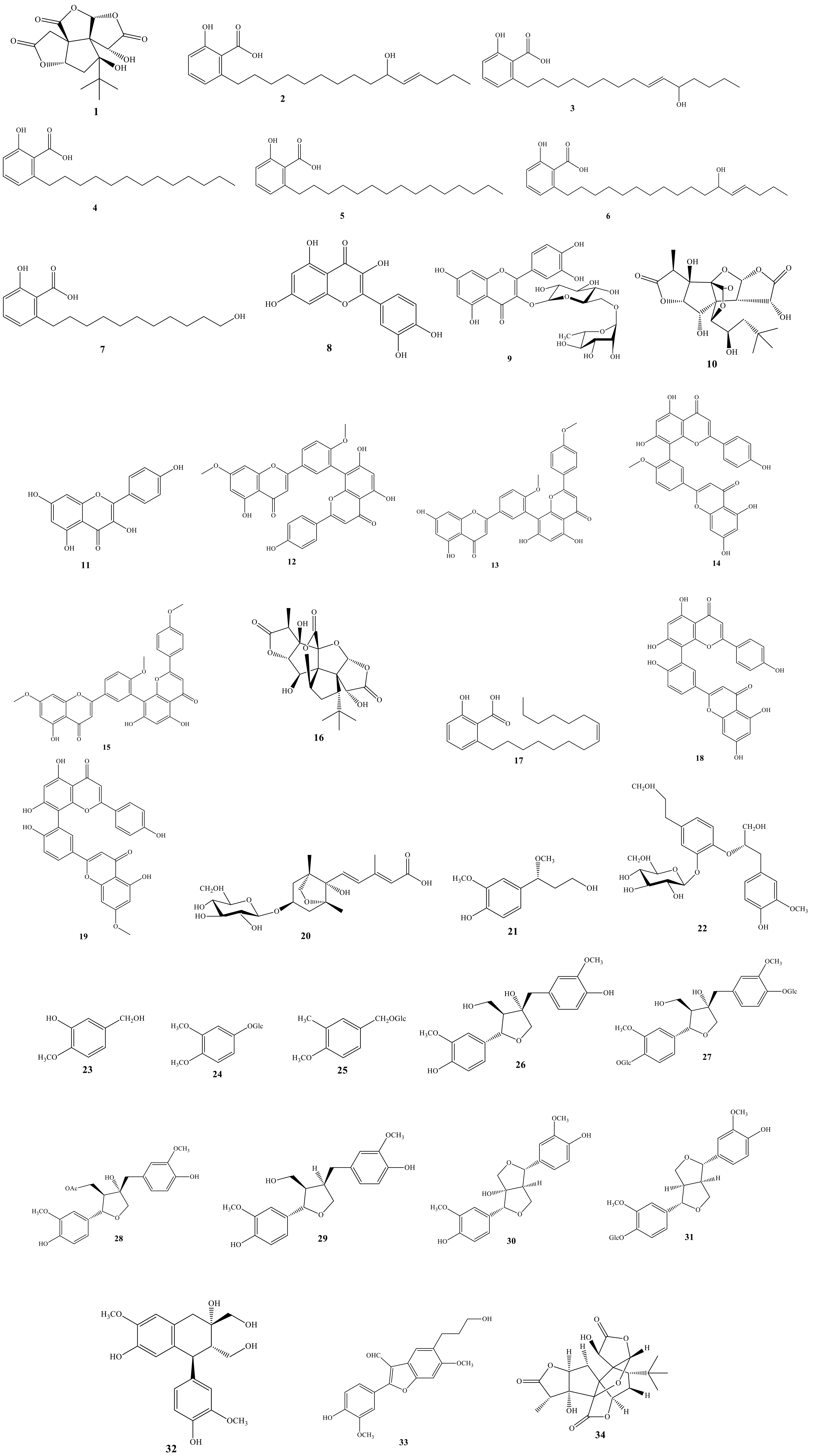 Fig. 4.
Fig. 4.
Chemical structures of compounds isolated from Ginkgo biloba.
Furthermore, bilobalide provided significant protection to adipocytes from the
adverse effects of hypoxia in a dose-dependent manner, inflammation,
mitochondrial damage, and attenuating oxidative stress. The treatment
significantly reduced cell death, with the highest dose (50 µM) showing a
10% reduction, highlighting its potential in mitigating cellular damage
associated with metabolic disorders [68]. In another study, bilobalide induced
apoptosis in mature 3T3-L1 adipocytes through a ROS-mediated mitochondrial
pathway, offering promising therapeutic avenues for obesity management [69].
Moreover, in vivo studies in male Wistar rats treated with bilobalide at
a dose of 2 mg/kg/day revealed increased glycogen content in the liver and
muscle, in normal and STZ-induced diabetic rats This effect suggests that
bilobalide may help mitigate impaired glucose utilization, possibly through its
antioxidant properties or by enhancing glucose uptake, further supporting its
role in managing diabetes [70]. The compounds
2-hydroxy-6-(10′-hydroxypentadec-11′(E)-en-1-yl) benzoic acid (2),
2-hydroxy-6-(11′-hydroxypentadec-9′(E)-en-1-yl) benzoic acid (3),
2-hydroxy-6-tridecylbenzoic acid (4), 2-hydroxy-6-pentadecylbenzoic acid (5),
2-hydroxy-6-(12′-hydroxyheptadec-13′(E)-en-1-yl) benzoic acid (6), and
2-hydroxy-6-(11-hydroxyundecyl) benzoic acid (7) were tested for their inhibitory
effects on protein tyrosine phosphatases (PTPs) relevant to insulin resistance.
These compounds exhibited potent enzyme inhibition, with 4–6 showing over 90%
inhibition against PTPN11, PTPN2, PTP1B, DUSP9, PTPRS, and PTPN9. Compounds 2 and
3 also displayed significant inhibitory activity against several PTPs, including
PTPN11, PTPN2, PTP1B, and DUSP9. These findings underscore the importance of the
side chain length and substitutions within the side chain of ginkgolic acid
derivatives in determining their inhibitory potency. These derivatives from
G. biloba leaves may serve as promising therapeutic candidates for
managing Type 2 diabetes mellitus (T2DM) due to their potential to inhibit PTPs
involved in insulin resistance [71]. Quercetin (8) and rutin (9), both derived
from the leaves, exhibited potent
Ginkgetin (12), a compound derived from G. biloba leaves, was found to
inhibit the expression of PPAR
The clinical trials conducted to date with G. biloba have primarily targeted its antidiabetic effects and improve lipid profiles, and mitigate diabetes-related complications (Table 2, Ref. [86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100]).
| Administered material | Study design | Number of Subjects | Health condition of subjects | Duration | Dosage regimen | Findings/Results | Ref |
| GKB extract | Randomized, placebo-controlled, double-blinded | 60 | T2DM patients | 90 days | 120 mg/day | ↓ Blood HbA1c, fasting serum glucose | [86] |
| GKB extract | Randomized, placebo-controlled, double-blinded | 24 | T2DM patients | - | 120 mg/day | Inhibit insulin resistance | [87] |
| G. biloba extract (EGb 761) | Randomized, placebo-controlled, double-blinded crossover study | 10 | T2DM patients | - | 120 mg/day | ↓ Glycosylated hemoglobin A(1c) levels | [88] |
| G. biloba extract injection | Control treatment | 60 | DN patients | - | (5 mL/amp) 20 mL | ↓ Urinary albumin excretion rate, hemorheology ↑, renal function, and regulated blood lipids | [89] |
| GKB extract | Randomized controlled study | 80 | NPDR patients | 6 months | 2 mL-orally, 3 times/day | ↓ Number of retinal micro‐aneurysms and areas of retinal hemorrhage, ↓ LDL cholesterol, triglycerides, total cholesterol, ↑ HDL, ↓ platelet adhesion rate, and platelet aggregation rate | [90] |
| G. biloba Tablets | Randomized, double-anonymized, multicenter, controlled trial | 600 | Type 2 diabetes | 24 months | 24 mg/day | Attenuate the deterioration of albuminuria in type 2 diabetes patients | [91] |
| Extract of G. biloba leaf (EGB) | Randomized controlled trial | 68 | DN | 3 months | 9.6 mg Orally, 3 times/day | ↓ Urinary mALB, alpha1-MG, IgG, TF, RBP, and NAG | [92] |
| GKB extract | Randomized, double-anonymized, placebo‐controlled | 210 | T2DM | 3 years | 24 mg, 3 times/day | Less ACR ↑ | [93] |
| GKB extract capsules | Randomized double blind placebo-controlled | 250 | T2DM | 27 months | For the first 9 months, one capsule 80 mg twice/day. For the second 9 months, one capsule 80 mg 3 times/day | Demonstrated a moderate effect on psychological state and Significant ↑ glycemic control in T2DM patients | [94] |
| Ginkgo biloba extract | 20 | Patients with normal glucose tolerance | 3 months | 120 mg/day | ↓ Blood pressure and an increase in the rate of insulin metabolic clearance | [95] | |
| G. biloba extract (EGb 761) | Not specific | 20 | NIDDM with hyperinsulinemia, NIDDM with pancreatic exhaustion | 3 months | 120 mg/day | ↓ Insulin-mediated glucose metabolism and ↑ blood glucose levels | [96] |
| G. biloba (EGb 761®) | Not specific | 120 | T2DM | 6 months | 240 mg/day | ↑ Brain function and QoL indicators in DM-2 by counteracting insulin resistance | [97] |
| G. biloba Pills | Randomized, double-blind, placebo-controlled | 12 | - | 58 weeks | Oral G. biloba pills or drops, three times/day, five at a time | Regulated glucose, ↓ diabetes risk, and ↓ CVD incidence and mortality | [98] |
| G. biloba extract | Double-blind, randomized, placebo-controlled crossover study | 30 | Healthy non-diabetic, glucose tolerant volunteers | 3-months | 120 mg as a single dose | Modulated hypothalamic-pituitary-adrenal axis, ↓ basal cortisol and stress-induced cortisol response | [99] |
| Capsule | Randomized double-blind, placebo-controlled trial | 60 | T2DM | 90 days | 120 mg | ↓ Lipid profile, ↓ inflammatory mediators, leptin level and ↑ the antioxidant status of T2DM patients | [100] |
T2DM, Type 2 diabetes mellitus; DN, Diabetic nephropathy; HbA1c, Hemoglobin A1c; EGb 761, Ginkgo biloba extract; mALB, Microalbuminuria; alpha1-MG, Alpha-1-microglobulin; IgG, Immunoglobulin G; TF, Transferrin; RBP, Retinol binding protein; NIDDM, Non-insulin-dependent diabetes mellitus; ACR, Albumin-creatinine ratio; CVD, Cardiovascular disease; QoL, Quality of life.
A randomized, placebo-controlled, double-blinded study conducted on 60 patients with type 2 diabetes mellitus (T2DM) over 90 days demonstrated that administering 120 mg/day of GKB extract significantly reduced blood HbA1c and fasting serum glucose levels [86]. Similarly, another randomized, placebo-controlled, double-blinded study involving 24 T2DM patients found that the exact dosage of GKB extract effectively inhibited insulin resistance [87]. In a randomized, double-blind, placebo-controlled crossover study conducted on 10 T2DM patients, G. biloba extract (EGb 761) at a dosage of 120 mg/day resulted in a reduction in glycosylated hemoglobin A1c (1c) levels [88]. Additionally, a control treatment study using G. biloba extract injection in 60 diabetic nephropathy (DN) patients revealed that administering 20 mL (5 mL per ampule) led to a decrease in urinary albumin excretion rate, regulation of blood lipids, and improvement in renal function and hemorheology [89]. A randomized controlled study with 80 non-proliferative diabetic retinopathy (NPDR) patients over a six-month duration showed that oral administration of 2 mL of GKB extract three times daily significantly decreased the number of retinal microaneurysms and areas of retinal hemorrhage, along with lowering serum total cholesterol, triglycerides, and low-density lipoprotein cholesterol while increasing high-density lipoprotein cholesterol. Furthermore, platelet aggregation and adhesion rates were also reduced [90]. A large-scale, randomized, double-blind, multicenter controlled trial involving 600 type 2 diabetes patients over 24 months indicated that daily intake of 24 mg of G. biloba tablets helped attenuate the deterioration of albuminuria in diabetic patients [91]. Similarly, a randomized controlled trial with 68 DN patients over three months showed that an oral dosage of 9.6 mg of G. biloba extract (EGB) taken three times daily led to significant reductions in urinary microalbumin (mALB), alpha1-microglobulin (alpha1-MG), immunoglobulin (IgG), transferrin (TF), retinal binding protein (RBP), and N-acetyl-beta-D-glycosaminidase (NAG) levels [92]. A long-term, randomized, double-blind, placebo-controlled study on 210 T2DM patients conducted over three years found that consuming 24 mg of GKB extract three times daily resulted in a lower albumin-to-creatinine ratio (ACR) increase [93]. Another randomized, double-blind, placebo-controlled trial on 250 T2DM patients over 27 months revealed that administering 80 mg of GKB extract capsules twice daily for the first nine months and thrice daily for the following nine months demonstrated moderate improvements in psychological states and significantly enhanced glycemic control [94]. A study on 20 patients with standard glucose tolerance over three months showed that taking 120 mg of G. biloba extract daily reduced blood pressure and increased insulin metabolic clearance rate [95]. Furthermore, research on 20 patients with non-insulin-dependent diabetes mellitus (NIDDM) with hyperinsulinemia or pancreatic exhaustion over three months found that daily intake of 120 mg of G. biloba extract reduced insulin-mediated glucose metabolism while elevating blood glucose levels [96]. A six-month study involving 120 T2DM patients revealed that consuming 240 mg/day of G. biloba (EGb 761®) positively affected insulin resistance-related factors in the brain, improving overall brain function and enhancing quality of life indicators [97]. A randomized, double-blind, placebo-controlled trial on 12 participants over 58 weeks assessed the impact of G. biloba pills, taken three times a day in groups of five, in controlling impaired glucose regulation, slowing diabetes progression, and reducing the incidence and mortality of cardiovascular diseases [98]. A randomized, double-blind, placebo-controlled crossover study on 30 healthy, glucose-tolerant volunteers over three months reported that administering 120 mg of G. biloba extract as a single dose affected the hypothalamic-pituitary-adrenal axis by reducing basal cortisol levels and cortisol production in response to acute hyperglycemic challenges [99]. Lastly, a double-blind, randomized, placebo-controlled trial on 60 T2DM patients over 90 days found that consuming 120 mg of G. biloba capsules significantly decreased inflammatory mediators and leptin levels while improving antioxidant status and lipid profiles [100].
G. biloba demonstrates a strong safety profile, with multiple animal and human studies reporting no significant toxicity or adverse effects, supporting its use as a safe natural therapeutic agent. In clinical trials, administration of G. biloba extract at a dose of 120 mg/day showed no adverse effects on treated individuals’ liver, kidney, or hematopoietic functions [86]. Similarly, another study using EGb 761 at the same dose for three months reported it to be safe and well-tolerated in both healthy and diabetic participants, with only minor, non-life-threatening side effects such as changes in appetite, mild headaches, and menstrual bleeding [88]. In animal studies, acute toxicity assessments revealed no toxic effects even at higher doses; for example, oral administration of GKB at 400 mg/kg showed no signs of toxicity [63]. Further supporting its safety, no adverse effects were noted after administering 200 mg/kg body weight over 60 days [61]. In experimental models, to further mitigate any risk of toxicity, high doses of GKB (500 mg/kg) were diluted in normal saline (0.9%) before administration, and no toxicological effects were reported [48]. Similarly, in another study, high doses of G. biloba extract, such as 400 mg/kg, were dissolved and delivered in normal saline to minimize potential toxicity [56]. While adverse events—including coronary artery disease, cerebrovascular accidents, and cancer have been reported, no direct causal relationship to G. biloba alone has been established. In combination therapy trials, such as those involving G. biloba and Liuwei Dihuang (LWDH), the absence of detailed subgroup analyses limits the ability to attribute these events to any single component [91]. These findings are consistent across different formulations and dosages, emphasizing G. biloba’s tolerability at both therapeutic and elevated doses. Nonetheless, further investigations, particularly regarding long-term use and potential interactions, are recommended to confirm its safety for broader clinical applications.
There is growing commercialization of products that combine concentrated bioactive compounds from natural sources, known as nutraceuticals. The health benefits of these compounds have attracted significant interest from the scientific community, with some tested through experimental studies [101]. Plant-derived dietary supplements, including antioxidants, essential fatty acids, lipid metabolism enhancers, vitamins, and trace elements, are marketed and recommended to patients as beneficial additions to a diabetic diet and conventional treatments. These supplements aim to enhance glycemic control and minimize the effects of chronic complications [102]. G. biloba products were among the top-selling medicinal items in U.S. health food stores. Unlike the leaves, Ginkgo nuts have a long history of use as both food and medicine, with their first mention in herbals around 1350 AD [103]. G. biloba leaves and seeds are rich in vitamin, carbohydrates, riboflavin, proteins, amino acid, minerals and other essential nutrients (Table 3, Ref. [103, 104, 105, 106]). These nuts are believed to provide health benefits, including diabetes, cancer prevention and treatment for neurological diseases [107]. Carbohydrate compounds in plants, such as oligosaccharides and polysaccharides, have been found to significantly enhance glucose and insulin metabolism in both healthy and diabetic individuals. A study compared three diets in diabetic patients: low-starch (43% carbs, 22% protein, 34% fat), high-starch (55% carbs, 15% protein, 30% fat), and a typical American diet (40% carbs, 20% protein, 40% fat). The low-starch diet resulted in significantly lower plasma glucose and insulin levels. Another trial of over 3200 patients showed that high-fiber, low-fat diets could help prevent diabetic complications. A clinical study also found that high-carb, high-fiber meals improved glycemic control and reduced post-meal lipids in type 2 diabetes patients [108].
| Class | Component | Leaves (mg/100 g) | Seeds (mg/100 g) | Ref |
| Macronutrients | Protein | 12,270 |
9000–13,000 | [103, 104, 105, 106] |
| Carbohydrate | 72,980 |
35,000–72,600 | ||
| Fat | 4750 |
2400 | ||
| Fiber | 2500 | - | ||
| Vitamins | Vitamin C | 79.2 | 15 | |
| Vitamin E | 59.3 | - | ||
| Thiamine (B1) | 1.53 | 0.22 | ||
| Riboflavin (B2) | 2.98 | 0.09 | ||
| Niacin (B3) | 2.44 | 0.16 | ||
| Vitamin (B6) | 3.57 | 0.33 | ||
| Minerals | Sodium | 1.65–2.34 | 7 | |
| Potassium | 4.33–21.11 | 510 | ||
| Calcium | 24.62–40.22 | 2 | ||
| Magnesium | 18.45–69.26 | 27 | ||
| Phosphorus | 4.90–32.65 | 124 | ||
| Iron | 2.63–6.67 | 1 | ||
| Zinc | 0.20–1.85 | 0.34 | ||
| Copper | 0.64–0.79 | 0.27 | ||
| Manganese | 0.63 | 0.11 | ||
| Amino Acids | Leucine | 3630 | 310 | |
| Lysine | 1390 | 200 | ||
| Valine | 2950 | 280 | ||
| Serine | 1510 | 290 | ||
| Proline | 2610 | 340 | ||
| Glycine | 2040 | 230 | ||
| Glutamic acid | 7670 | 830 | ||
| Histidine | 1510 | 100 | ||
| Phenylalanine | 1800 | 170 | ||
| Arginine | 4860 | 420 | ||
| Cystine | 160 | 20 | ||
| Alanine | 2910 | 240 | ||
| Methionine | 750 | 50 | ||
| Aspartic acid | 6420 | 540 | ||
| Isoleucine | - | 200 | ||
| Threonine | 2290 | 260 | ||
| Tryptophan | - | 70 | ||
| Tyrosine | 1320 | 60 | ||
| Others | Ash | 10,010 |
- | |
| Water | 55,200 | - |
The reviewed studies on the antidiabetic effects of Ginkgo biloba leaf
extracts exhibited significant heterogeneity, particularly in dosage,
administration routes, animal models, and outcome measures, thus making
comparisons and generalizations challenging. The dosage range varies considerably
for each bioactive derivative from G. biloba (Tables 1A,1B,1C (Ref. [19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85])). This discrepancy
raises concerns about the feasibility and safety of translating these doses to
human-equivalent doses (HEDs). For instance, a 200 mg/kg rat dose translates to
approximately 32 mg/kg in humans, highlighting the need for further research into
dose-response relationships and the therapeutic window for human applications.
This lack of dose translation between animal models and humans restricts applying
preclinical results to clinical practice. Moreover, the heterogeneity in
administration routes (e.g., intraperitoneal injection, oral gavage, and oral
administration) adds to the variability. For example, intraperitoneal
administration of leaf extracts at doses ranging from 50–500 mg/kg showed
significant antihyperglycemic, antioxidant, and hypolipidemic effects in
STZ-induced diabetic rats (Table 1B). In contrast, 100 mg/kg/day of ethanolic
leaf extract oral administration preserved pancreatic
G. biloba as a potential alternative therapy for diabetes mellitus (DM) has garnered significant attention due to its promising pharmacological effects, including its antioxidant, anti-inflammatory, and insulin-sensitizing properties. However, the current research scenarios about the antidiabetic activity of the herb present several challenges and gaps that need to be addressed in future studies to understand its full therapeutic potential better. One central area requiring further investigation is the standardization of G. biloba dosage. While numerous studies have demonstrated its efficacy in reducing blood glucose levels and improving insulin sensitivity, there is considerable variation in the dosages and forms of G. biloba used across clinical trials. This inconsistency makes it difficult to determine an optimal, universally applicable dose for effective diabetes management. Future clinical studies should focus on selecting the most effective doses while considering factors such as the bioavailability of active compounds, patient age, comorbidities, and the duration of treatment. While preclinical studies have provided valuable insights into the mechanisms through which G. biloba exerts its effects, such as AMPK activation, GLUT4 translocation, and inhibition of protein tyrosine phosphatases, the detailed molecular pathways remain insufficiently explored. To further validate the therapeutic antidiabetic activity of G. biloba, future research must focus on uncovering the exact mechanisms of action behind its potency in managing diabetes. Investigations around molecular interactions of key bioactive compounds, such as bilobalide and flavonoids, will provide deeper insights into their role in regulating glucose metabolism and insulin secretion. These mechanistic studies could lead to more specific therapeutic regimens, which can be used as adjunct therapy alongside other antidiabetic agents. Another possible direction for future research could be to explore the long-term safety and efficacy of G. biloba in diabetic populations with comorbid conditions, such as cardiovascular disease, neuropathy, and nephropathy. Focus should be placed on chronic toxicity, herb-drug interactions, and use during pregnancy. While initial clinical studies have suggested that G. biloba may help mitigate diabetes-related complications, large-scale, multicenter clinical trials are necessary to establish its role in preventing and treating complications over extended periods. Additionally, the potential of G. biloba in managing gestational diabetes and its effects during pregnancy remains largely unexplored, which calls for further investigation.
In conclusion, G. biloba L. holds considerable promise as a complementary therapy for diabetes management. Its multifaceted pharmacological activities, such as enhanced glucose uptake, improved insulin secretion, and reduced oxidative stress, make G. biloba an ideal candidate for improving glycemic control, thus helping prevent diabetes-related complications. Preclinical and clinical studies have shown encouraging results, suggesting that G. biloba can reduce blood glucose levels, improve lipid profiles, and protect against diabetes-induced nephropathy and retinopathy. However, to fully establish its role as an effective adjunct in diabetes management, future research must address the gaps in clinical trials, optimise dosing regimens, and further elucidate the underlying mechanisms of action. With well-designed, large-scale clinical trials and a deeper understanding of its molecular effects, G. biloba could become integral to diabetes treatment strategies, offering a natural, safe alternative to conventional therapies.
ACR, Albumin-to-creatinine ratio; AMPK, Adenosine Monophosphate-activated Protein Kinase; Alpha1-MG, Alpha1-microglobulin; ApoE, Apolipoprotein E; Bax-to-Bcl-2, ratio of Bcl-2-associated X (Bax) to B-cell lymphoma 2 (Bcl-2) protein expression; BMSCs, Bone marrow mesenchymal stem cells; C/EBP
Conceptualization, NHS; Formal Analysis, NHS, IP, and JAC; Funding Acquisition, NHS, TTas, and IP; Investigation, Resources, Writing - original draft, NHS, NATE, AAM, MDH, TTab, PS and TTas; Writing - review & editing, NHS, NATE, TTas, IP, and JAC; Supervision and Reviewing, NHS, IP, and JAC. All authors have contributed to the editorial changes made to the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.