1 Department of Physiology, Faculty of Medicine, Kırşehir Ahi Evran University, 40100 Kırşehir, Türkiye
2 Department of Histology and Embryology, Faculty of Medicine, Kırşehir Ahi Evran University, 40100 Kırşehir, Türkiye
3 Department of Physiology, Faculty of Medicine, Balıkesir University, 10145 Balıkesir, Türkiye
Abstract
Alcohol consumption exerts toxic effects on the central nervous system, causing neurodegeneration and behavioral disorders. The neuroprotective potential of disulfiram (DSF), a widely used treatment for alcohol dependence, remains unclear. Thus, this study aimed to evaluate the effects of DSF on ethanol-induced neurodegeneration, behavioral impairments, and cardiovascular alterations.
A total of 28 rats were randomly assigned to four groups: control, ethanol (EtOH), EtOH + DSF, and DSF. Ethanol (5 g/kg) was administered orally three times per week for 14 days. The EtOH + DSF group received ethanol plus DSF (100 mg/kg, intraperitoneally, i.p.) while the DSF group received DSF alone. Blood pressure was measured using tail-cuff plethysmography. Behavioral alterations were analyzed through the open field test (OFT), elevated plus maze (EPM), and forced swim test (FST). Brain tissues were examined histopathologically using hematoxylin–eosin and toluidine blue staining. Data were analyzed by analysis of variance (ANOVA) and the Kruskal–Wallis test.
In the EPM, no significant differences were found among the groups in open-arm activity or risk-assessment behaviors. The OFT showed a significant reduction in locomotor activity in the EtOH group (p < 0.0001). In the FST, ethanol increased immobility (p < 0.0001) and decreased the latency to immobility (p = 0.0014). Ethanol markedly lowered systolic, diastolic, and mean arterial pressures, whereas DSF elevated systolic and mean arterial pressures without affecting heart rate. DSF treatment affects systolic and mean arterial blood pressures. So for systolic blood pressure p = 0.002. For mean arterial blood pressure p = 0.024. Histological analysis revealed neuronal damage in the EtOH group, while DSF significantly reduced the number of degenerating neurons (p < 0.001).
Ethanol exposure was associated with depression-like behavior, reduced locomotor activity, and hypotension. DSF treatment may improve behavioral outcomes, partially counteract ethanol-induced hypotension, and confer neuroprotective effects.
Keywords
- anxiety
- depressive disorder
- disulfiram
- ethanol
- neurodegeneration
- histology
Chronic alcohol consumption is a major global health problem associated with high mortality and morbidity [1]. However, only a small proportion of individuals who consume alcohol become chronic drinkers [2], whereas binge drinking is more prevalent [3]. Binge drinking is typically defined as the consecutive consumption of one or more alcoholic beverages within a short period during the previous six months [3]. Furthermore, binge drinking induces pathophysiological changes in multiple organ systems in both humans and animals, particularly in the central nervous system, causing effects such as neuroinflammation, neurotoxicity, and oxidative stress-induced damage [4]. In addition to these pathological alterations, binge drinking is associated with cognitive and neurobehavioral impairments [5, 6].
Operationally, binge drinking is characterized as the intake of 4–5 alcoholic drinks within approximately two hours, depending on gender, leading to an elevated blood alcohol concentration of 0.08% or higher [7]. In animal models that mimic binge-like drinking patterns, ethanol is commonly administered to rats via oral gavage [8]. In such models, repeated doses of ethanol exposure have been observed to induce neurodegeneration in specific brain regions and to elicit behavioral changes [9]. The findings from studies suggest that binge drinking in rat models produces anxiety- and depression-like behaviors [10, 11], which could serve as an experimental basis for the development of new pharmacological therapies aimed at treating anxiety- and depression-like disorders that arise in humans following binge drinking.
Disulfiram (DSF) inhibits aldehyde dehydrogenase, the enzyme that catalyzes the conversion of acetaldehyde to acetate during alcohol metabolism [12]. The accumulation of acetaldehyde leads to a DSF–ethanol reaction, which can produce mild, moderate, or severe symptoms in humans [13]. In the literature, the co-administration of DSF with other pharmacological agents has recently been proposed as a therapeutic strategy for alcohol-dependent patients with comorbid psychiatric conditions, including depression, post-traumatic stress disorder, bipolar disorder, schizophrenia, schizoaffective disorder, and cocaine addiction [14]. However, despite the previous use of DSF, an older agent, the neurobiological effects of DSF remain a subject of ongoing research [15].
Indeed, the protective role of DSF has been primarily attributed to the DSF–ethanol reaction, which results in aversive acetaldehyde accumulation by inhibiting aldehyde dehydrogenase, thereby producing conditioned behavioral avoidance [16]. In addition to this well-known mechanism, DSF has been reported to exert neuroprotective effects through several other pathways. One proposed mechanism involves modulation of pyroptosis pathways, thereby preserving blood–brain barrier integrity [17]. DSF has also been shown to reduce oxidative stress-mediated damage, contributing to neuroprotection [18]. Furthermore, the anti-inflammatory effects of DSF have been associated with inhibition of the HSP70/TLR4/NLRP3 signaling pathway [19]. Nonetheless, studies investigating the potential histological, behavioral, and cardiovascular effects of DSF in binge-like drinking animal models remain limited [20, 21]. Most available evidence regarding DSF has been derived from models of chronic alcohol consumption or clinical populations with alcohol use disorder [22].
The relationship between low to moderate alcohol consumption and cardiovascular disease remains controversial. While some earlier studies have suggested potential cardioprotective effects, more recent studies do not support this view [23]. Abstinence from alcohol or a significant reduction in alcohol intake has been shown to influence elevated blood pressure [24]. Alcohol consumption increases blood lipid levels, thereby raising the risk of coronary artery disease, increasing the risk of thrombosis, and leading to acute or chronic hypertension [25]. Additionally, existing literature indicates an association between alcohol consumption and ischemic heart disease, arrhythmia, cardiomyopathy, stroke, and heart failure [26].
Thus, the novelty of the present study lies in the use of a binge-like ethanol paradigm combined with a comprehensive assessment integrating behavioral, cardiovascular, and histopathological outcomes within a single experimental model. In contrast to previous studies that have primarily focused on chronic alcohol exposure or the aversive mechanisms of DSF [27, 28], this study explores the potential protective effects of DSF against binge-like drinking alongside the neurobehavioral, neurodegenerative, and cardiovascular effects of this agent. Therefore, the findings may extend the current understanding of DSF beyond the role as an alcohol-aversive agent and provide new insights into additional potential neuroprotective and physiological benefits.
In light of the limited data regarding the effects of DSF in the context of binge-like ethanol drinking, this study aimed to investigate the impact of DSF on anxiety- and depression-like behavioral disturbances, histopathological alterations in brain tissue, and cardiovascular parameters induced by binge-like ethanol exposure. To this end, the open field test (OFT), elevated plus maze (EPM), and forced swim test (FST) were employed to evaluate behavioral outcomes; meanwhile, cardiovascular measurements were obtained using the tail-cuff plethysmography technique. Histopathological changes were analyzed using hematoxylin–eosin (H&E) and toluidine blue (TB) staining techniques.
The use of laboratory animals in this study at Kırşehir University (Approval Number: 2025/8-3) was approved by the Local Ethics Committee. All procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals.
A total of 28 adult male Wistar albino rats (200–250 g, 8–10 weeks old) were used. The animals were obtained from the Kırşehir Ahi Evran University Laboratory Animal Production and Research Center, and were housed in the animal facility under controlled environmental conditions, with the temperature maintained at 22–25 °C and a 12 h light/12 h dark cycle (lights on at 07:00 and off at 19:00). The rats were kept in specially designed cages, and the bedding material was replaced daily. All animals received the same standard laboratory rat chow, and water and food were provided ad libitum [29]. Only male rats were included to minimize hormonal variability associated with the estrous cycle, which may confound behavioral, neurobiological, and cardiovascular outcomes in the ethanol-exposure model. This approach allowed for a more controlled assessment of the effects of DSF in a binge-like ethanol drinking paradigm. The animals were randomly assigned to four experimental groups (n = 7 per group):
Control group (n = 7): Rats received oral gavage of 0.9% saline (volume-matched to EtOH) three times per week at 12-hour intervals for 14 days.
Ethanol (EtOH) group (n = 7): Rats received ethanol orally at a dose of 5 g/kg three times per week at 12-hour intervals for 14 days [30].
Ethanol + disulfiram (EtOH + DSF) group (n = 7): Rats received ethanol orally at a dose of 5 g/kg three times per week at 12-hour intervals for 14 days. DSF (100 mg/kg) diluted in 0.9% saline was administered intraperitoneally once every 7 days, immediately after the final ethanol administration on that day [30, 31].
Disulfiram (DSF) group (n = 7): Rats received only DSF at a dose of 100 mg/kg intraperitoneally once every 7 days [31]
Noninvasive blood pressure measurements in rats were obtained using a tail-cuff
system with volume pressure recording (MAY-NIBP250, Ankara, Türkiye). Cuff
integrity was routinely verified before each experiment. Measurements were
performed in a quiet, temperature-controlled environment (22
In the EPM, anxiety-related behavior was evaluated by measuring time spent in the open arms, number of open-arm entries, head-dipping frequency, and stretch-attend postures. In the OFT, locomotor activity was assessed by recording the total distance traveled. In the FST, depressive-like behavior was analyzed by measuring immobility duration and latency to first immobility.
The EPM test was performed to record anxiety-like behavior [34]. The apparatus
consisted of two open arms (50
The OFT, a widely used method for evaluating general locomotor activity and
anxiety-like behavior, was performed once per animal. All experiments were
performed during the light phase of the light–dark cycle, with all groups tested
within the same time frame to minimize circadian influences. On the test day,
rats were transported to the experimental room and allowed to acclimate in their
home cages for 1 hour prior to the session. The open field apparatus consisted of
a square Plexiglas arena measuring 60
Rats were individually introduced into a transparent cylindrical glass tank (50
cm height
Prior to decapitation, all rats were deeply anesthetized with a ketamine (60 mg/kg)–xylazine (10 mg/kg) mixture to minimize pain and distress. This combination of agents was administered to the animals at a volume of 1 mL/kg. Adequacy of anesthesia was confirmed by the absence of both pedal and corneal reflexes before proceeding.
Brain tissues were fixed in 10% neutral buffered formaldehyde, dehydrated through a graded alcohol series, cleared in xylene, and embedded in paraffin blocks. Sections of 5 µm thickness were prepared and stained with H&E to evaluate overall morphology and cellular alterations. In addition, TB staining was performed to assess Nissl granules in neurons of the cerebral cortex, thereby enabling the identification of degenerative neurons and comparison among experimental groups. Images of the stained sections were obtained using a light microscope (Nikon Eclipse Si, Tokyo, Japan).
In this study, brain tissue was evaluated for the presence of pyknotic neurons,
vacuolization, edema, and microhemorrhage (Nikon Eclipse Si, Tokyo, Japan).
Histopathological alterations were assessed semiquantitatively using a 0–3
scoring system based on the estimated percentage of the affected area within each
examined field: 0 = no pathological finding; 1 = mild changes affecting
All statistical analyses were performed using GraphPad Prism 9 (GraphPad
Software, San Diego, CA, USA). Data distribution was assessed using the
Shapiro–Wilk test before inferential analysis. Variables that met the assumption
of normality (p
Blood pressure values were measured at the end of the behavioral experiments and
compared among the groups. Systolic blood pressure in the EtOH group (44.46
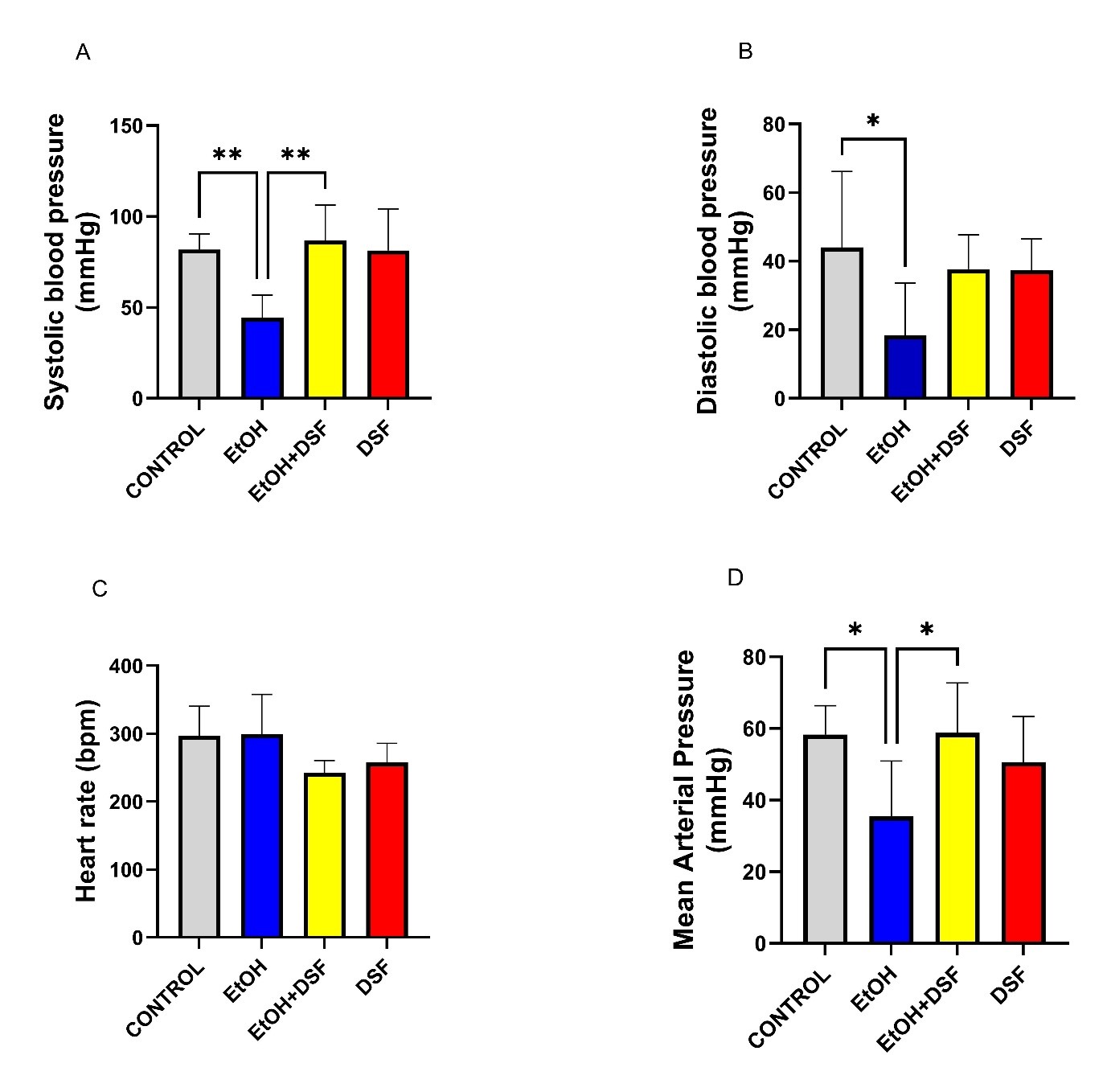 Fig. 1.
Fig. 1.
Effects of ethanol (EtOH) and disulfiram (DSF) treatments on
cardiovascular parameters (n = 7). (A) Systolic blood pressure (mmHg), (B)
diastolic blood pressure (mmHg), (C) heart rate (bpm), and (D) mean arterial
pressure (MAP; mmHg) in control, EtOH, EtOH + DSF, and DSF groups. Data are
presented as mean
In the EPM test, no significant differences were observed among the groups in
the time spent in the open arms (control: 51.43
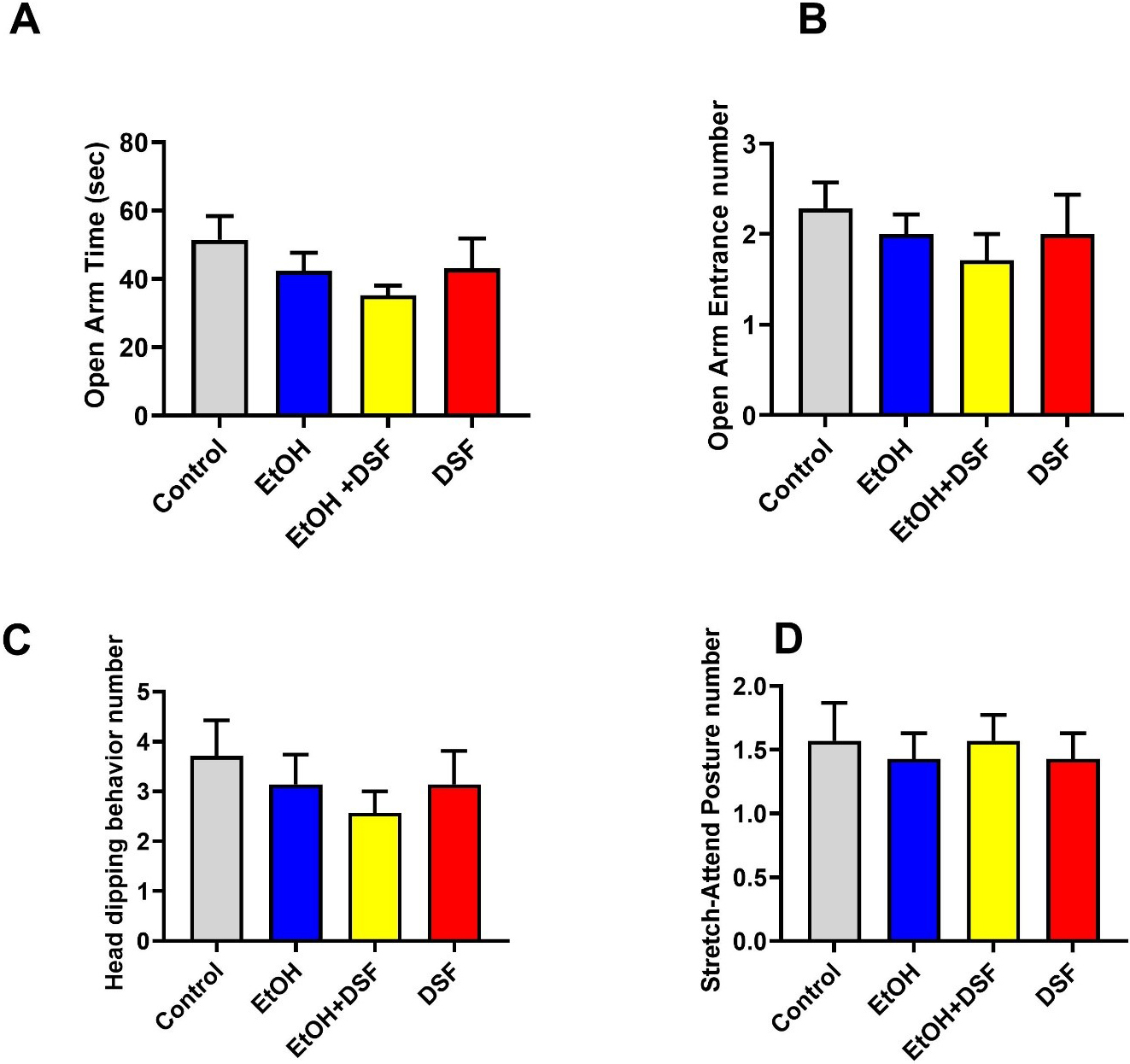 Fig. 2.
Fig. 2.
Effects of EtOH and DSF treatments on anxiety-related behaviors
in the elevated plus maze (EPM) test (n = 7). (A) Open arm time (sec), (B) open
arm entrance number, (C) head dipping behavior number, and (D) stretch-attend
posture number in control, EtOH, EtOH + DSF, and DSF groups. Data are presented
as mean
In the OFT, the total distance traveled was significantly reduced in the EtOH
group (325.7
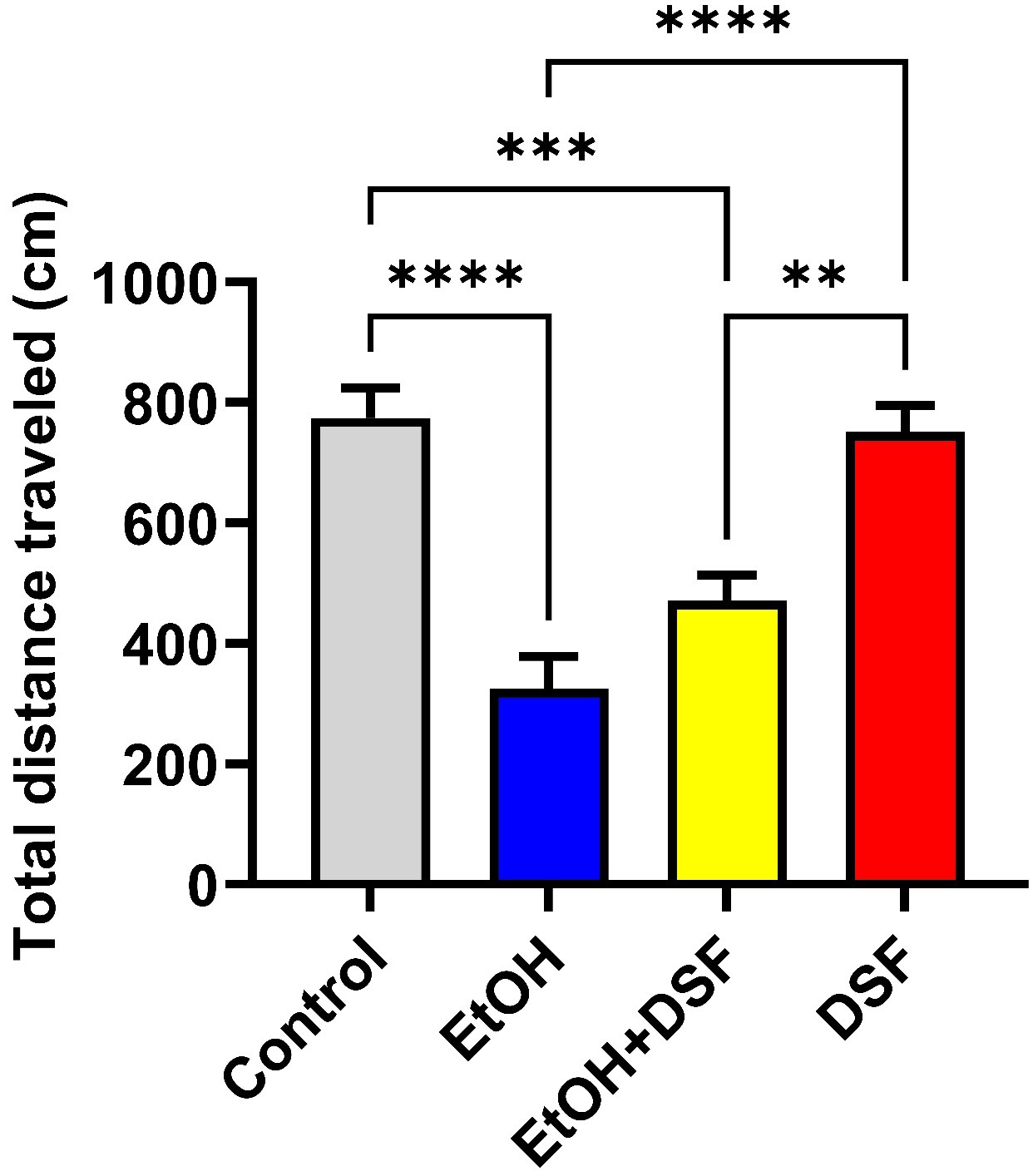 Fig. 3.
Fig. 3.
Effects of EtOH and DSF treatments on anxiety-related behaviors
in the open field test (OFT) (n = 7). Data are presented as mean
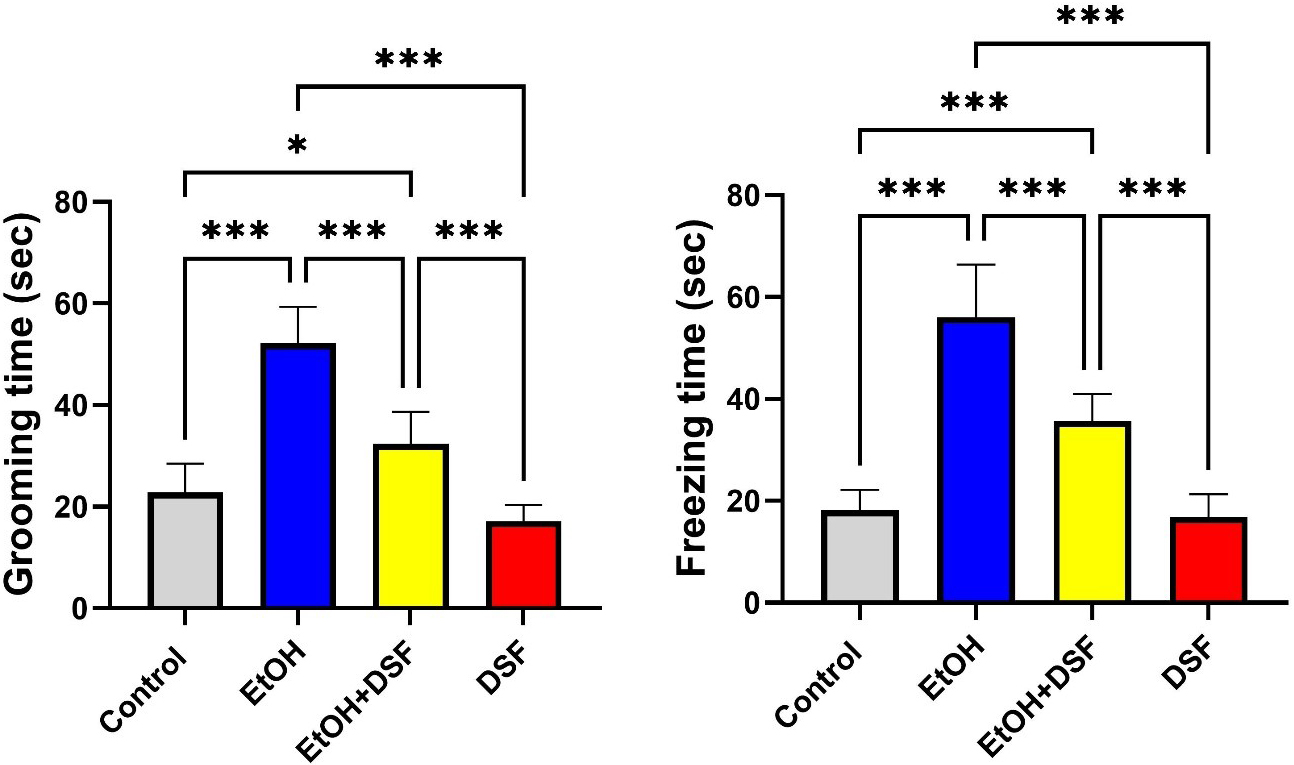 Fig. 4.
Fig. 4.
Effects of EtOH and DSF treatments on anxiety-related behavioral
parameters in the OFT (n = 7). Grooming time and freezing time are presented as
mean
In the intergroup evaluation of the FST, immobility time in the EtOH group
(176.0
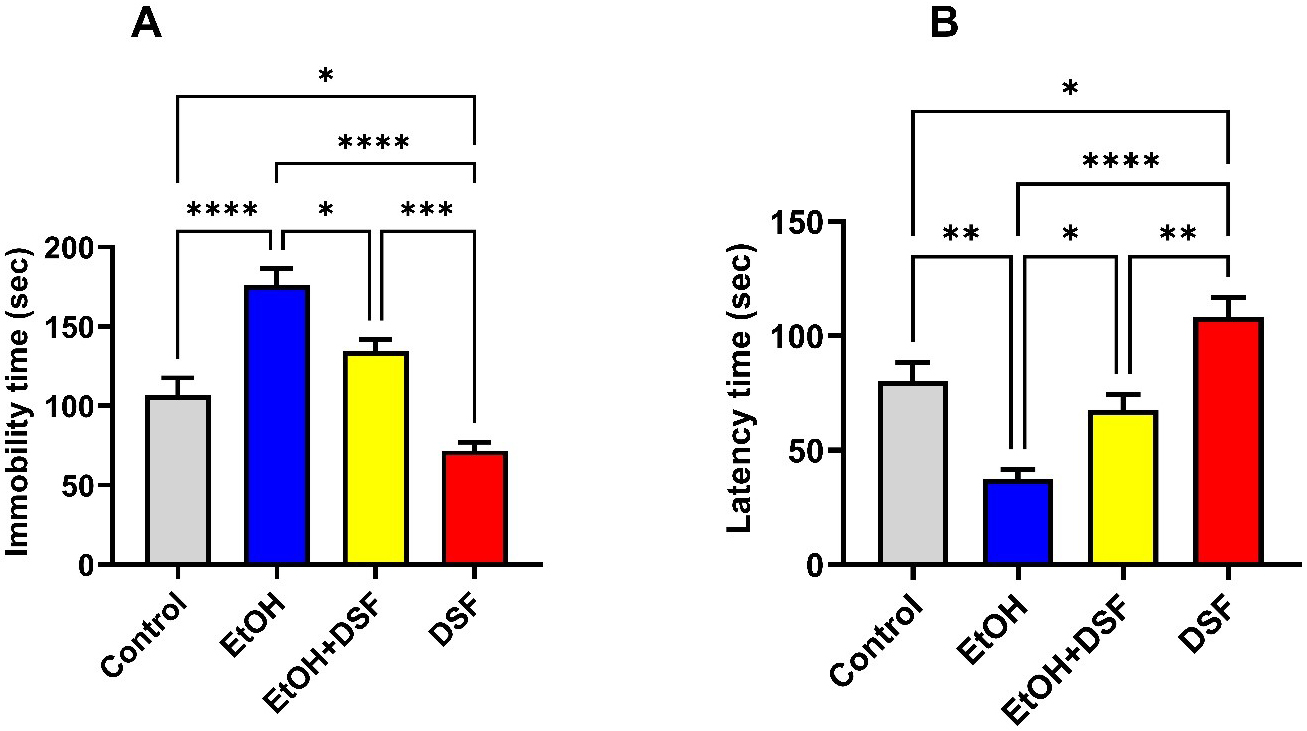 Fig. 5.
Fig. 5.
Effects of EtOH and DSF treatments on depression-related
behaviors in the forced swimming test (FST) (n = 7). Data are presented as mean
Fig. 6 illustrates the histopathological appearance of brain tissue following
H&E staining. In the control group, neurons in the cerebral cortex exhibited
normal architecture, with euchromatic nuclei and visible nucleoli, indicating
preserved cellular morphology. The neuronal arrangement was regular, and the
intercellular spacing appeared normal. Cytoplasmic integrity was maintained, with
no evidence of vacuolization. In addition, glial cell density was within normal
limits. In the EtOH group, widespread cellular damage was observed in the
cerebral cortex compared with the control group. In this group, darkly stained
pyknotic neurons with shrunken nuclei (p
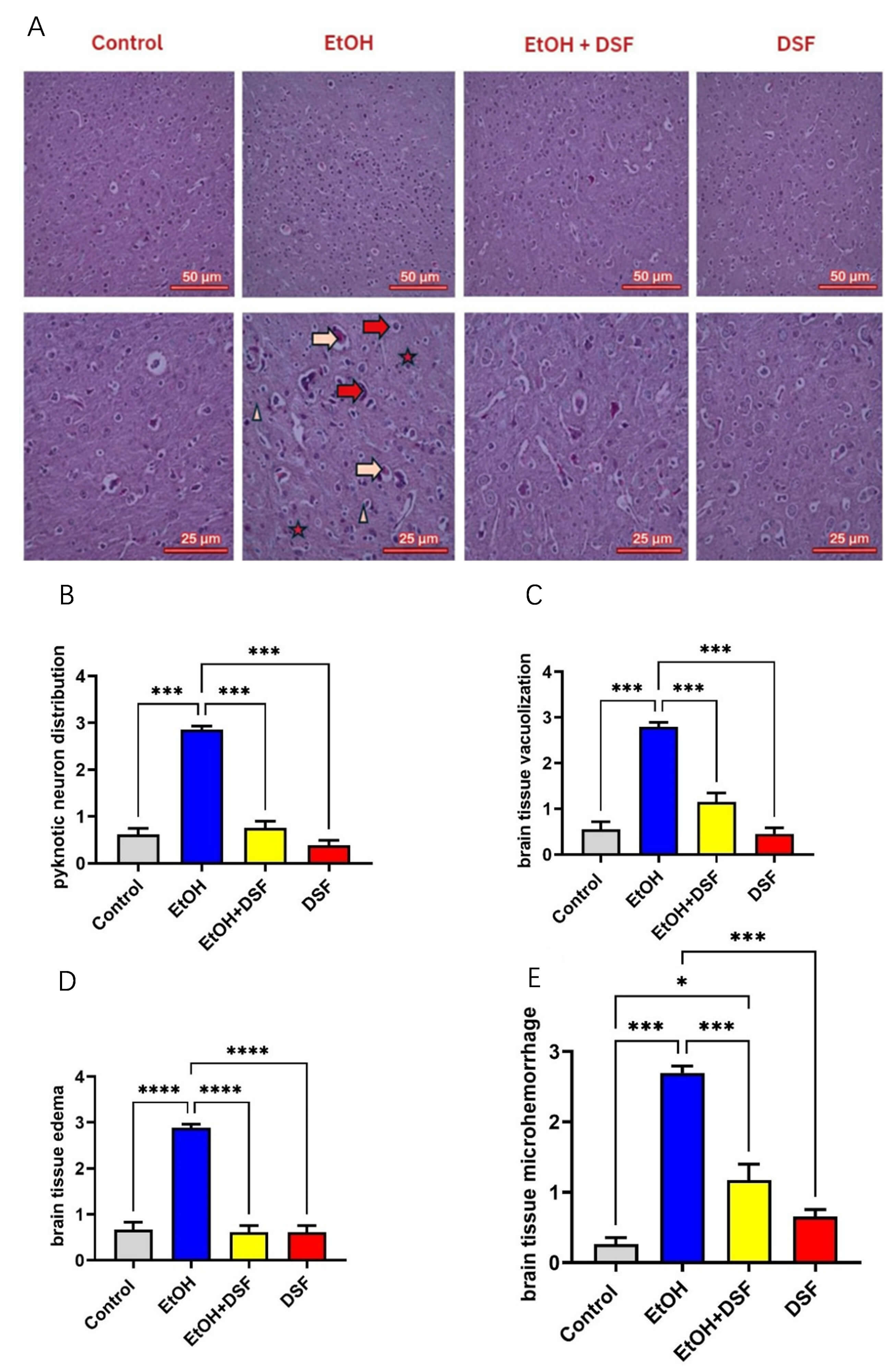 Fig. 6.
Fig. 6.
Representative photomicrographs of brain tissue from Control,
EtOH, EtOH + DSF, and DSF groups (n = 7) at 200
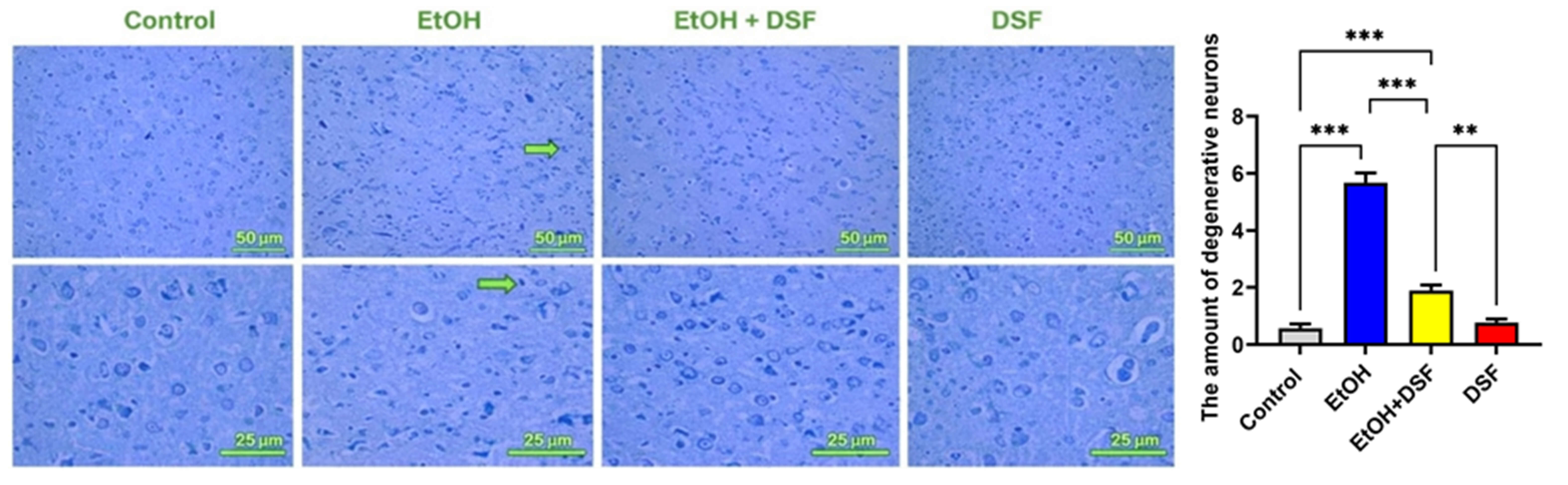 Fig. 7.
Fig. 7.
Representative photomicrographs of brain tissue sections from
control, EtOH, EtOH + DSF, and DSF groups (n = 7) at 200
In this study, rats exposed to binge-like ethanol showed a significant reduction in both systolic and mean arterial blood pressure. In contrast, DSF administration appeared to restore systolic and mean arterial pressure without markedly affecting diastolic pressure or heart rate. Behavioral assessments revealed that ethanol induced depressogenic and anxiogenic-like effects, as evidenced by increased immobility and decreased latency in the FST and reduced locomotor activity in the OFT. DSF partially mitigated these behavioral effects. EPM parameters were not significantly affected by any treatment. Histopathological analysis of the cerebral cortex in ethanol-treated rats revealed neuronal damage, including pyknotic neurons, vacuolization, edema, and microhemorrhages. In addition to these changes, DSF treatment reduced neuronal degeneration as assessed by histopathological examination. Overall, these findings suggest that DSF exerts a protective effect against ethanol-induced cardiovascular, behavioral, and neurohistological alterations.
Our EPM findings indicate that neither ethanol nor DSF had a significant effect in this binge-like ethanol consumption model. This is consistent with previous studies reporting that ethanol does not reliably induce anxiety-like behavior in the EPM test [38, 39]. However, DSF has been reported to produce anxiolytic-like effects in rodents under specific conditions [40, 41]. For instance, Saitoh et al. [40] demonstrated that DSF increased the time spent in the open arms of the EPM in a dose- and time-dependent manner. Conversely, Escrig et al. [42] reported that DSF alone did not alter open-arm exploration, whereas co-administration of DSF (60–90 mg/kg) with ethanol induced anxiogenic effects [42]. Our EPM results may be related to the dose, duration, and route of DSF administration. Additionally, the absence of significant alterations in the ethanol group may be attributable to the relatively short duration of ethanol exposure, as longer or more chronic exposure periods could lead to more pronounced behavioral changes.
The OFT results showed that ethanol exposure reduced locomotor activity and increased anxiety-related behaviors, whereas DSF partially reversed these effects [43]. Saitoh et al. [40] reported that DSF administration did not significantly alter locomotor activity compared with the control group. Similarly, Kim and Souza-Formigoni [44] reported that mice treated with DSF (15 mg/kg) or placebo, in combination with ethanol or saline for 10 days, showed no significant change in locomotor activity following acute DSF administration; however, ethanol-sensitized mice exhibited higher locomotor activity compared with saline-treated controls [44]. Consistent with the findings of Kim and Souza-Formigoni [44], the dose and duration of DSF administration in our study mitigated ethanol-induced anxiety-like behaviors.
Our FST results demonstrated increased immobility and decreased latency to immobility in the ethanol group, indicative of depression-like behavior. This finding is consistent with previous studies linking binge-like ethanol exposure to depressive-like phenotypes [10, 45]. Repeated binge-like ethanol exposure in adolescent female rats has been shown to elicit both anxiety- and depression-like behaviors [46], and chronic binge-like alcohol exposure similarly promotes depressive-like behaviors in adolescent rats [11]. In the present study, DSF treatment reversed ethanol-induced changes in immobility and latency times. While previous studies have primarily focused on the biological effects or locomotor activity of DSF, our binge-like drinking model suggests that DSF may exert a protective effect by attenuating ethanol-induced depression-like behaviors.
Binge-like drinking generates acetaldehyde and downstream acetate, both centrally and peripherally [28]. These metabolites have significant dose- and tissue-dependent effects, including direct control of vascular tone, hemodynamic alterations [47], and sympathetic activation [48]. In our study, systolic and mean arterial blood pressure were reduced in the ethanol group, indicating ethanol-induced hypotension. Consistent with this, a previous chronic alcohol study reported the onset of hypotension in rats beginning in the third week of ethanol exposure [47]. In a study by Yao and Abdel-Rahman [49], ethanol administration was also associated with hypotension in rats. Similarly, in an 8-week chronic ethanol study conducted in female rats, ethanol-induced hypotension was observed [50]. In contrast to our findings, Jensen and Faiman [51] reported that combined administration of ethanol and DSF induced hypotension in rats. DSF inhibits aldehyde dehydrogenase, leading to marked accumulation of acetaldehyde when ethanol is consumed. Acetaldehyde causes vasodilation, increased capillary permeability, and hypotension, producing flushing, tachycardia, and low blood pressure [52]. A study has also reported a hypertensive effect of DSF in the absence of alcohol dependence. The effect of DSF on blood pressure may not depend solely on ethanol metabolism; DSF may also exert direct vascular regulatory effects via autonomic and catecholaminergic mechanisms [53]. The findings of our study suggest that DSF may partially attenuate the hypotensive response and may contribute to cardiovascular autonomic regulation, possibly through vasoconstrictive effects in rats, depending on the dose and duration of administration.
The cerebral cortex of ethanol-exposed rats exhibited significant cellular damage and degenerative changes, whereas brain tissue from the control group maintained a healthy structure. Consistent with our findings, a previous 12-week ethanol exposure study also reported degenerative lesions and vacuolization in the brains of rats [54]. Another study reported that alcohol exposure in rats was associated with hemorrhage, congestion, necrosis, and cellular infiltration in the brain [55]. In a previous study, intranasally administered DSF was investigated as a potential therapeutic agent for glioblastoma treatment [56]. In our binge-like ethanol drinking model, DSF treatment mitigated these neurodegenerative changes and, particularly in TB staining, markedly reduced the number of degenerative neurons. Histologically, DSF promoted the recovery of ethanol-damaged neurons.
This study has certain limitations. First, it was not possible to assess sex-related differences in the responses of the rats to ethanol and DSF because the study was limited to male rats. Second, only the OFT, EPM, and FST assessments were used as behavioral evaluations; additional cognitive and memory-related tests would have provided a more comprehensive understanding of the neurobehavioral effects. Third, biochemical and molecular analyses that could have elucidated the underlying mechanisms of the reported protective effects, such as oxidative stress markers, neurotransmitter levels, and inflammatory cytokines, were not conducted. Fourth, the study focused only on early-stage injury and behavioral alterations, which may not fully reflect the long-term consequences of the condition. Finally, the lack of blood ethanol concentration (BEC) measurements represents an important limitation. The absence of BEC data precludes direct verification of intoxication levels and assessment of inter-animal variability in ethanol exposure.
In summary, our findings demonstrate that binge-like ethanol exposure induces cardiovascular dysfunction, neurobehavioral impairments, and cortical neuronal damage in rats. DSF treatment exerted protective effects by reversing ethanol-induced hypotension, ameliorating depressive-like behaviors, and reducing neuronal degeneration. While DSF did not significantly influence anxiety-related parameters in the EPM, this agent partially improved locomotor activity and depression-like outcomes. These results suggest that DSF may serve as a potential therapeutic candidate against the deleterious cardiovascular, behavioral, and neurohistological effects of binge-like ethanol consumption. However, although our findings indicate potential neuroprotective and behavioral benefits of DSF in a binge-like exposure model, the clinical feasibility of using this agent in this population may be limited without appropriate patient selection, monitoring, and education. Thus, future clinical studies should focus on identifying subgroups of binge drinkers who may benefit from DSF-based interventions, optimizing dosing strategies, and evaluating safety outcomes under controlled conditions. Integrating pharmacological approaches with behavioral interventions may also improve adherence and reduce risks in this population. Moreover, further studies involving both sexes, extended treatment periods, and molecular analyses are warranted to clarify the underlying mechanisms and long-term impact of DSF.
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
SK: Conceptualization, Methodology, Investigation, Data curation, Formal analysis, Writing – original draft, Supervision. KTK: Histopathological analysis, Investigation, Data curation, Visualization, Writing – review & editing. HÇ: Methodology, Behavioral experiments, Formal analysis, Writing – review & editing, Supervision. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The experiment was carried out according to Local Ethics Committee for the Use of Laboratory Animals at Kırşehir University (Approval Number: 2025/8-3). All procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







