1 Department of Pharmacology and Toxicology, College of Pharmacy, Qassim University, 52571 Buraydah, Saudi Arabia
Abstract
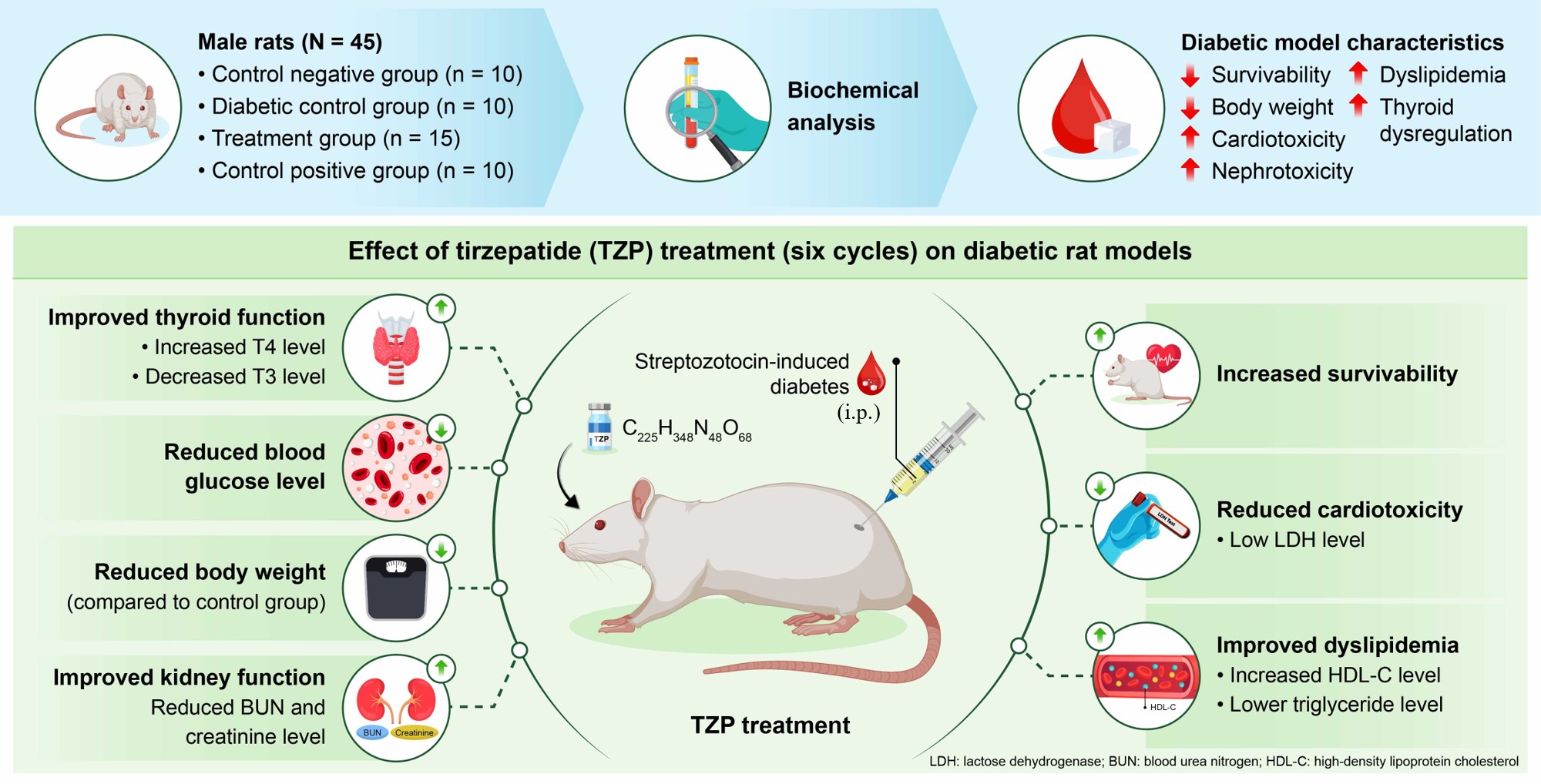
Tirzepatide (TZP), a dual glucose-dependent insulinotropic polypeptide (GIP)/glucagon-like peptide 1 (GLP-1) receptor agonist, has shown potential in improving glycemic control. However, its effects on diabetes mellitus (DM)-related systemic complications remain less defined. This study investigated the impact of TZP on metabolic, cardiovascular, renal, and thyroid parameters in a rat model of DM.
Experimental DM was induced in rats via a single intraperitoneal injection of streptozotocin (50 mg/kg). Animals were allocated into four groups: control (saline), DM, TZP alone (1.35 mg/kg, cyclic dosing), and DM+TZP (streptozotocin followed by five TZP injections). Survival, body-weight, and serum biochemical markers were monitored, including glucose, insulin, troponin I, Creatine kinase-MB (CK-MB), lactate dehydrogenase (LDH), blood urea nitrogen (BUN), creatinine, cholesterol, triglycerides (TG), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), thyroid-stimulating hormone (TSH), thyroxine (T4), and triiodothyronine (T3).
Survival was preserved in the control, TZP alone, and DM+TZP groups, whereas mortality reached 10% in the DM group. Body weight decreased significantly in the DM and DM+TZP groups, with a modest reduction in the TZP alone group. TZP markedly increased hyperglycemia and LDH levels without altering troponin I or CK-MB levels. DM-associated elevations in BUN and creatinine were partially attenuated by TZP, suggesting renoprotective effects. TZP had no impact on cholesterol or LDL-C, but significantly decreased triglycerides and increased HDL-C. Additionally, TZP modulated thyroid hormone levels, as reflected by increased T4 and decreased T3 concentrations compared with diabetic rats.
In this experimental DM model, TZP improved glycemic control, ameliorated renal dysfunction, and favorably modulated lipid metabolism and thyroid axis activity, with no evidence of overt myocardial necrosis observed based on troponin I and CK-MB; additional studies incorporating histology and/or functional assessments are needed to evaluate subclinical cardiac effects. These findings indicate the therapeutic potential of TZP in mitigating DM-associated complications beyond glucose regulation.
Graphical Abstract
Keywords
- diabetes mellitus
- tirzepatide
- cardiotoxicity
- nephrotoxicity
- dyslipidemia
- thyroid dysregulation
Diabetes mellitus (DM) is a chronic endocrine disorder marked by the gradual development of insulin resistance at the cellular level, leading to hyperglycemia. While DM is commonly diagnosed later in life, a notable increase in cases has been observed among younger populations, largely due to the rising rates of obesity in adolescents. Recent reports from the International Diabetes Federation estimate that approximately one in nine individuals aged 20–79 years worldwide is affected by DM [1, 2]. The disorder is linked to a heightened risk of cardiovascular diseases, compromised renal function, and dyslipidemia. Adults with DM are at a significantly increased risk of myocardial infarction and stroke. Additionally, there is a significant association between DM and kidney failure, as well as disruptions in blood lipid levels [3].
Approximately 30% of individuals with DM may experience proteinuria, renal impairment, and increased risk of cardiovascular diseases [4, 5]. Over time, they are at risk of developing diabetic cardiomyopathy and nephropathy, which pose significant long-term health challenges. Diabetes is frequently associated with thyroid dysfunction, likely due to impaired metabolic homeostasis, altered hepatic deiodinase activity, and disrupted energy metabolism. Emerging evidence suggests that glucagon-like peptide 1 (GLP-1) receptor agonists may indirectly influence thyroid hormone homeostasis through weight loss-associated metabolic changes, modulation of hepatic metabolism, and regulation of peripheral T4 to T3 conversion. Additionally, DM-associated dyslipidemia is characterized by elevated triglycerides and low-density lipoprotein cholesterol (LDL-C), along with reduced high-density lipoprotein cholesterol (HDL-C), contributing to increased cardiovascular risk [6, 7]. Certain pharmacological interventions effectively address the complications linked to DM [8]. For example, GLP-1 agonists, such as semaglutide, have demonstrated efficacy in this context [8, 9]. Research indicates that semaglutide can reduce the risk of cardiomyopathy and nephropathy while also ameliorating dyslipidemia [10].
GLP-1 receptor agonists administered via injection are effective in improving glycemic control and promoting weight loss, with a low risk of hypoglycemia [11]. Additionally, these agonists significantly improve cardiovascular and renal health [12]. When oral antidiabetic medications do not yield satisfactory results, GLP-1 agonists are recommended as a secondary therapeutic strategy [13]. Semaglutide, in particular, has demonstrated substantial efficacy in managing DM [14]. Furthermore, these agents mitigate the toxicities associated with specific therapies, such as chemotherapy [15, 16, 17]. Tirzepatide (TZP) is a dual incretin receptor agonist that activates both glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptors [18]. Through this combined mechanism, TZP enhances glucose-dependent insulin secretion, suppresses inappropriate glucagon release, and improves insulin sensitivity, leading to robust antihyperglycemic activity. In addition, incretin signaling influences broader cardiometabolic physiology, including gastric emptying, appetite regulation/weight change, and downstream effects on lipid handling and systemic inflammation/oxidative stress pathways. These mechanisms provide a biological rationale for investigating whether TZP may modify diabetes-associated complications beyond glycemic control, including renal dysfunction, dyslipidemia, and endocrine disturbances such as thyroid hormone dysregulation [19].
STZ-induced hyperglycemia is a well-established experimental model to study diabetes-associated systemic complications, including cardiac and renal injury. Therefore, the present study aimed to evaluate the efficacy and safety of TZP in an experimental rat model of DM, with particular emphasis on its effects on cardiac and renal function, lipid metabolism, and thyroid hormone homeostasis. Although TZP has demonstrated promise in improving glycemic control in diabetes, its effects on DM-related systemic complications, particularly cardiovascular, renal, lipid, and thyroid dysfunctions, remain less well defined. Addressing this gap, the current investigation seeks to provide new insights into the broader therapeutic potential of TZP beyond glycemic regulation.
To the best of our knowledge, this is among the first studies to evaluate tirzepatide’s effects on multiple diabetes-associated complications simultaneously, integrating biochemical indices of cardiac injury, renal dysfunction, dyslipidemia, and thyroid hormone homeostasis within a single STZ-induced diabetic rat model. This integrated approach is important because diabetes-related morbidity is multisystemic, and therapeutic agents may exert beneficial and/or adverse effects across different organs. Therefore, clarifying tirzepatide’s broader profile beyond glucose lowering is necessary to better inform its translational relevance and safety considerations.
The study protocol was approved by the Scientific Ethics Committee at Deanship of Graduate Studies and Scientific Research, Qassim University under Approval No. 25-20-01. All experimental procedures were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Forty male rats weighing 170–200 g were used in this study. Rats were housed on standard bedding and with environmental enrichment under controlled temperature and humidity conditions, with a 12-h light–dark cycle at 25 °C. They were given free access to food and water. The rats were observed daily to assess survival, and their body weights were measured every other day. Animals were randomly assigned to four experimental groups, and biochemical analyses were performed by investigators blinded to group allocation. The negative control group was administered physiological (0.9%) saline (n = 10). The DM group was given streptozotocin (STZ; 50 mg/kg, i.p.) to induce DM (n = 10). The positive control group (i.e., TZP group) was administered five injections of TZP (1.35 mg/kg, i.p.) with physiological saline (n = 10). In the treatment group (i.e., DM+TZP group), rats with DM induced by STZ (50 mg/kg, i.p.) were treated with five injections of TZP (1.35 mg/kg, i.p.) (n = 10).
Rats received a single injection (i.p.) of STZ (50 mg/kg) [20], freshly prepared
in 0.1 M citrate buffer (pH 4.2). Two days post-injection, DM was confirmed in
rats from the DM and treatment groups by measuring blood glucose levels. Rats
with a blood glucose level
The routine surveillance of survival rates yielded essential insights for this research. Cages were cleaned every 2 days, ensuring the immediate removal of deceased specimens. Consistent monitoring of body weight was imperative for assessing the animals’ overall health. Consequently, body weight was measured every 2 days, enabling the detection of minor fluctuations and aiding in the early recognition of potential health issues.
On day 10, nine animals per group were selected for biochemical analysis to
ensure equal group sizes. Animals that died during the experimental period were
excluded from the study prior to sample collection. At the end of the treatment
period, the surviving rats were lightly anesthetized using isoflurane delivered
via inhalation (~2–4% for induction, maintained at 1–3%).
Blood samples were subsequently collected from the retro-orbital venous plexus of
rats in the control, DM, TZP, and DM+TZP groups using sterile capillary tubes and
transferred into clean, labeled collection tubes [23]. Animals were then humanely
euthanized by CO2 inhalation in accordance with the AVMA Guidelines for the
Euthanasia of Animals (2020 Edition). Rats were placed individually into a
transparent euthanasia chamber containing ambient room air (gradual-fill method;
the chamber was not pre-charged with CO2). Compressed 100% CO2 gas
(
Blood was taken from the tail vein of rats that had fasted for 12 h. Glucose levels were estimated using the Contour® Next meter (Bayer, Leverkusen, Germany).
Serum insulin concentrations were determined using a rat insulin enzyme-linked immunosorbent assay (ELISA) kit (cat. no. RK09278, ABclonal Technology, Woburn, MA, USA) according to the manufacturer’s instructions. Serum samples were obtained as described in the sample preparation section [25].
The blood serum from all sample groups, control, DM, TZP, and DM+TZP, was meticulously analyzed using a cutting-edge automated analyzer. This advanced instrument leverages Electrochemiluminescence immunoassay (ECL) technology to measure a range of biomarkers linked to cardiac toxicity, including troponin I, CK-MB, BUN, creatinine, LDH, TG, HDL-C, LDL-C, and TC, as well as thyroid-related hormones, including thyroid-stimulating hormone (TSH), thyroxine (T4), and triiodothyronine (T3) [26]. The ECL technique was performed using the COBAS INTEGRA 400 plus system, strictly following the manufacturer’s guidelines (Roche Diagnostics, Germany).
The data are presented in this article as the mean
Fig. 1 illustrates the effect of DM and TZP treatment on rat survival. All rats in the control and TZP-treated groups survived throughout the experimental period. In contrast, the DM group exhibited a mortality rate of 10% by the end of the study. Notably, no mortality was observed in the DM+TZP group. These findings indicate that TZP treatment prevented DM-associated mortality in this experimental model (Fig. 1).
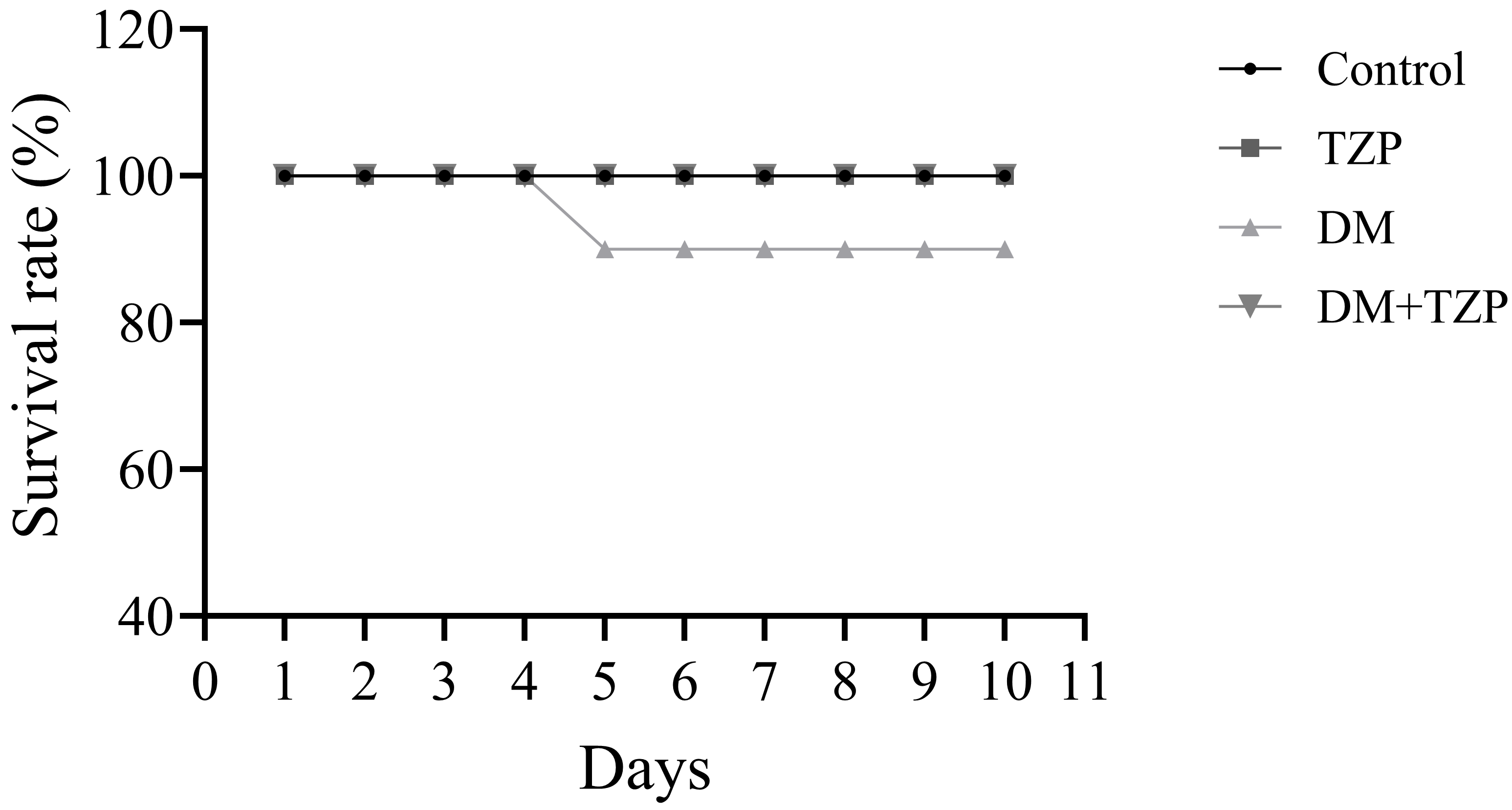 Fig. 1.
Fig. 1.
Effects of DM and TZP treatment on rat survival (n = 10). Abbreviations: Control, saline-treated rats; TZP, tirzepatide-treated rats; DM, diabetic rats; DM+TZP, diabetic rats treated with tirzepatide.
Body weight was recorded on days 1, 3, 5, 7, and 9. DM+TZP and DM groups exhibited a reduction in body weight on days 7 and 9 compared to the control group. In addition, the TZP-treated group showed a modest 10% reduction in body weight compared to the control (Fig. 2).
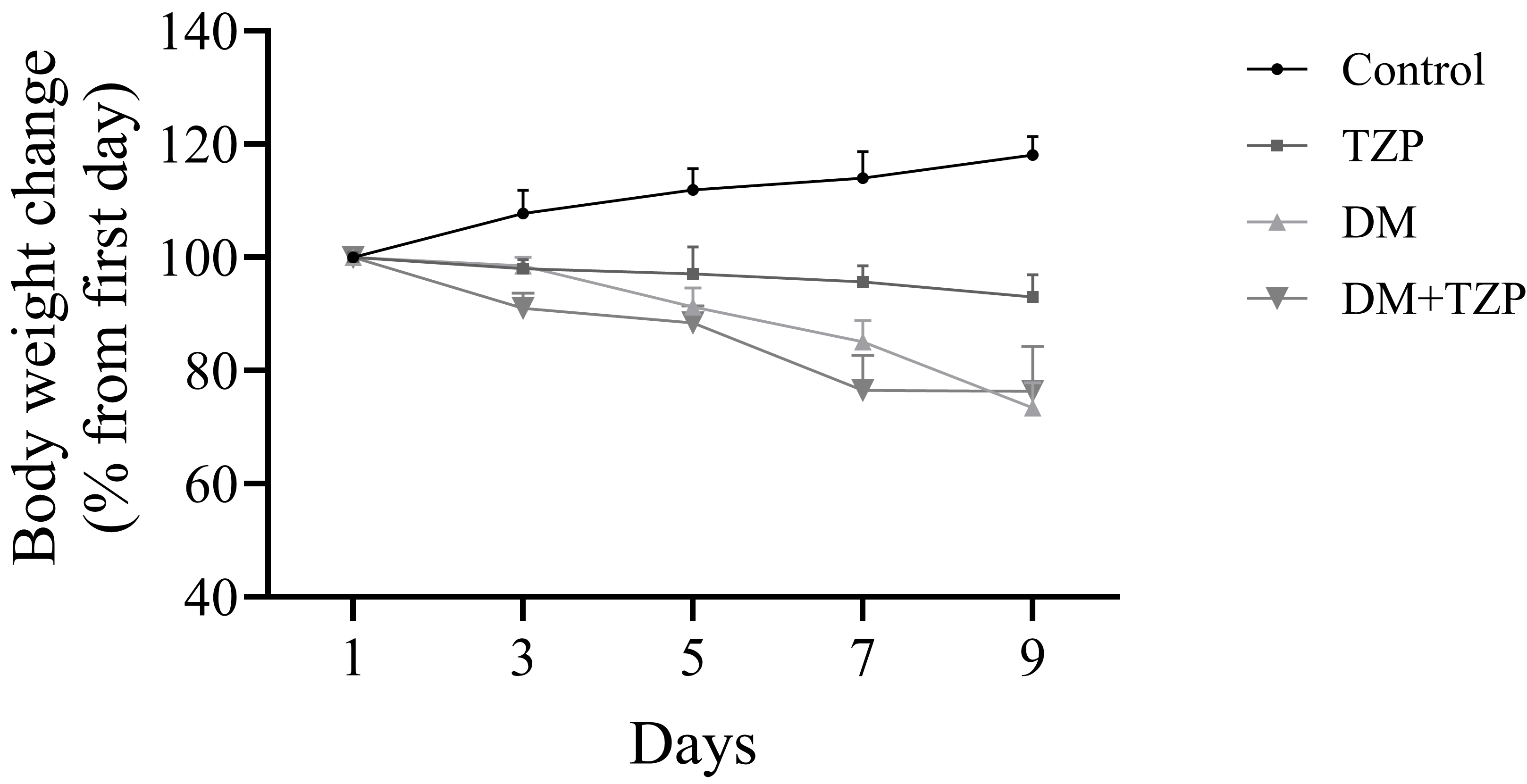 Fig. 2.
Fig. 2.
Effect of TZP on body weight changes in rats over 9 days. Abbreviations: Control, negative control; TZP, tirzepatide only; DM, DM control; DM+TZP, rats with DM treated with tirzepatide.
The effect of TZP on glycemic status in diabetic rats was evaluated by measuring
blood glucose levels on day 10. Diabetic rats exhibited significantly elevated
blood glucose levels compared with control rats. TZP treatment significantly
reduced blood glucose levels in diabetic rats compared with untreated diabetic
rats (Fig. 3A). Serum insulin levels were significantly reduced in diabetic rats
compared with controls and did not return to control levels following TZP
treatment (Fig. 3B). To further investigate insulin sensitivity, the Homeostatic
Model Assessment for Insulin Resistance (HOMA-IR) was calculated using the
formula: HOMA-IR = [fasting glucose (mg/dL)
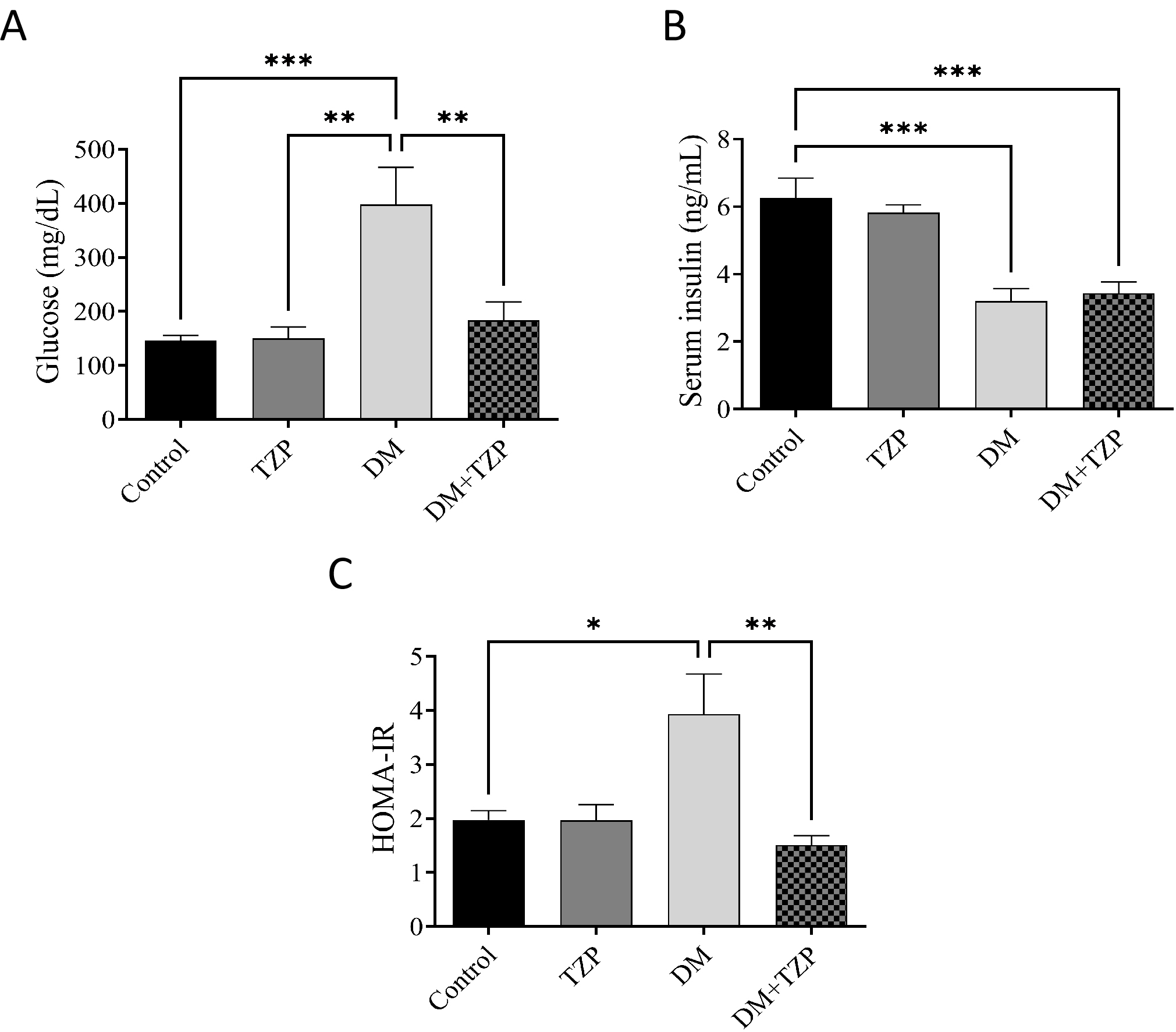 Fig. 3.
Fig. 3.
Effect of TZP on (A) glucose concentrations, (B) serum
insulin levels, and (C) HOMA-IR index in diabetic rats. The data represent the
mean
No significant differences in serum troponin I levels were observed among the experimental groups (Fig. 4A). Similarly, CK-MB levels did not differ significantly across groups (Fig. 4B), indicating no evidence of overt myocardial necrosis based on serum troponin I and CK-MB within the study timeframe. In contrast, LDH levels were significantly increased in non-diabetic rats treated with TZP compared with other groups (Fig. 4C), suggesting altered systemic or metabolic enzyme activity rather than specific cardiac injury.
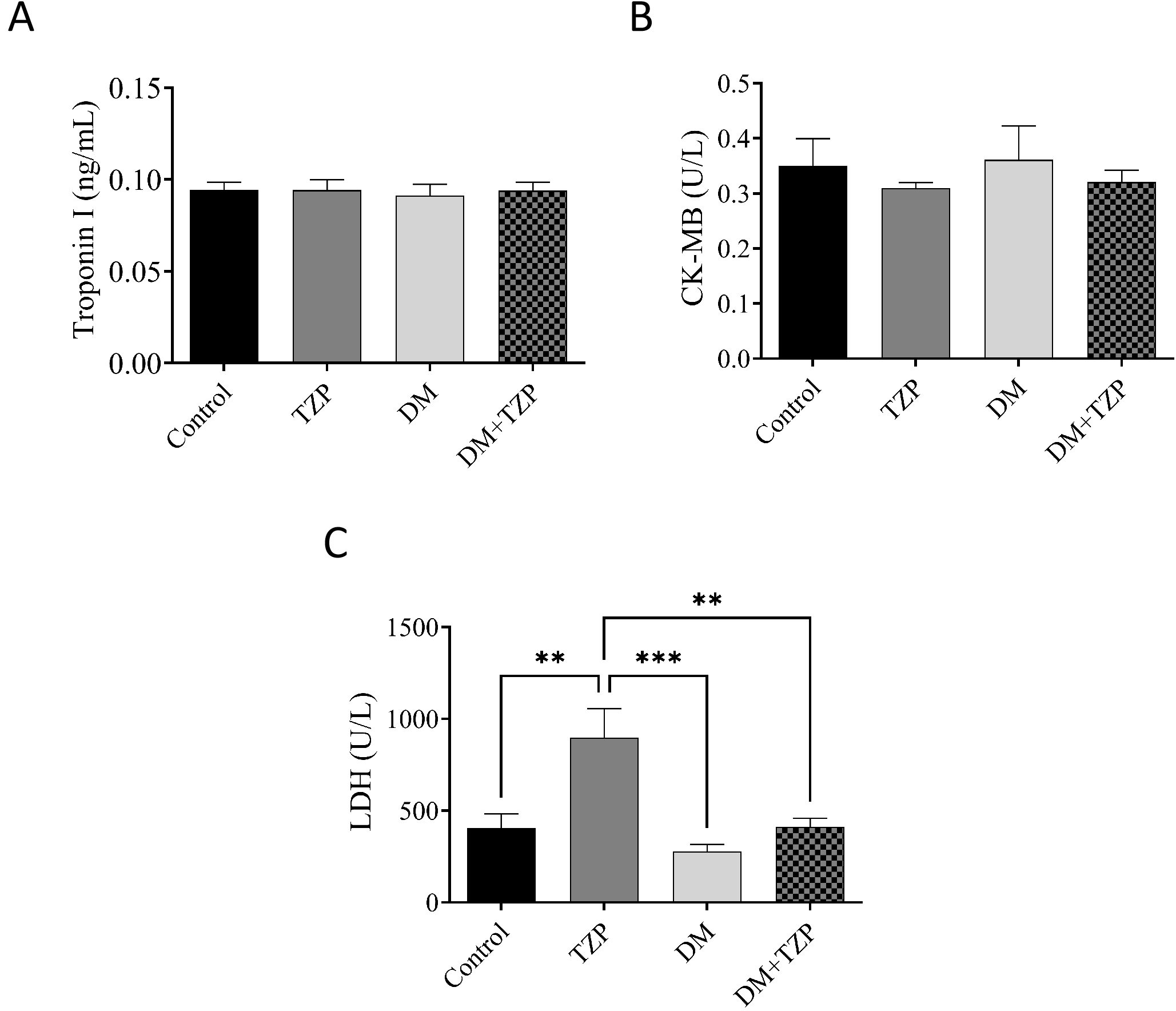 Fig. 4.
Fig. 4.
Effect of DM and TZP on cardiac-related biomarkers. (A)
troponin I, (B) CK-MB, and (C) LDH. **p
Rats with DM exhibited significantly elevated BUN, creatinine, and
BUN/creatinine ratios compared with control rats, indicating renal dysfunction
(Fig. 5; p
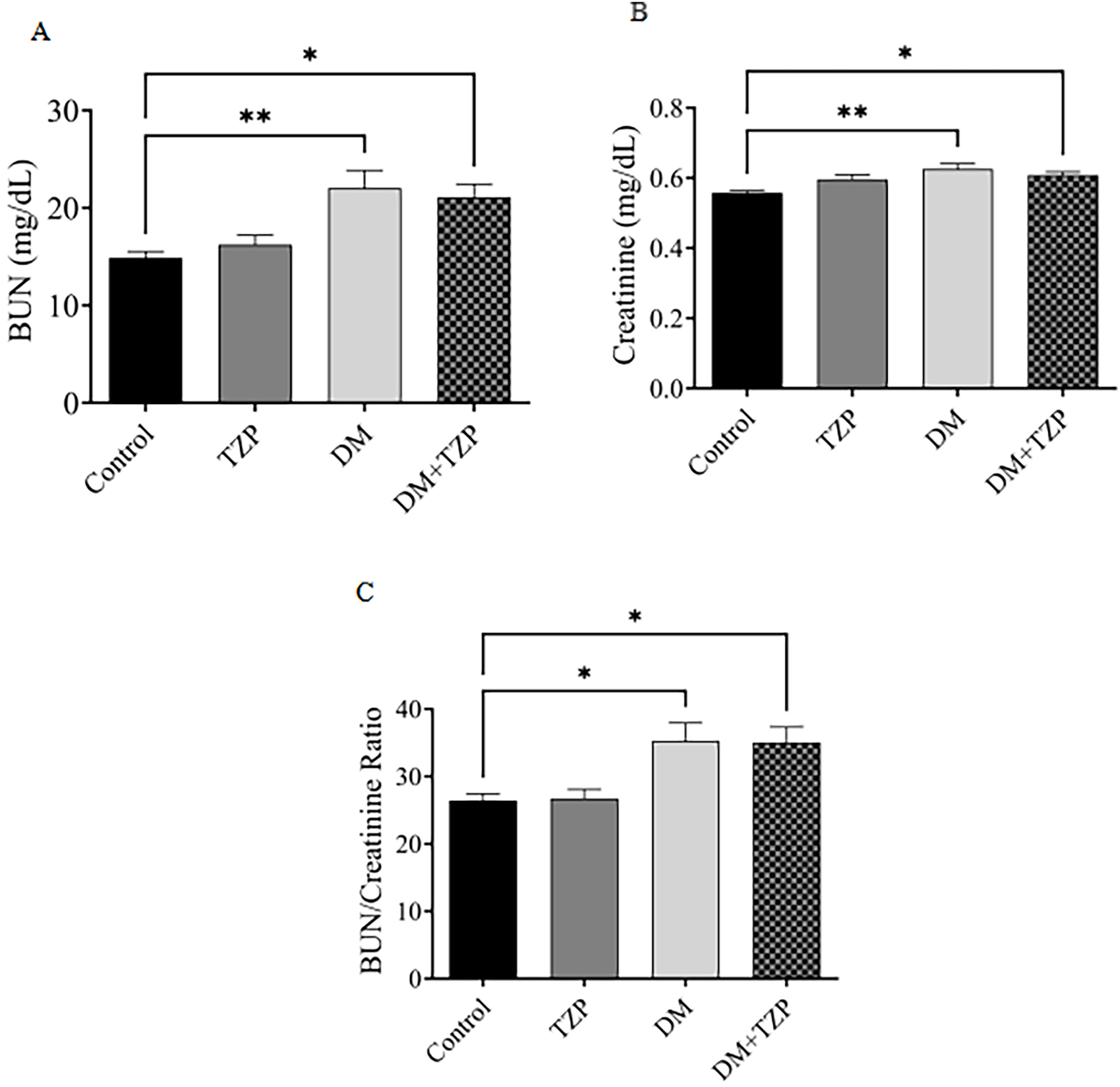 Fig. 5.
Fig. 5.
Effect of DM and TZP on renal function evaluated by
measuring the (A) BUN level, (B) creatinine level, and (C) BUN/creatinine ratio. **p
TC, TG, LDL-C, and HDL levels were measured to assess lipid changes in DM and
the effect of TZP on these changes (Fig. 6). No significant differences in total
cholesterol or LDL-C levels were observed among the groups (Fig. 6A,B). In
contrast, serum triglyceride levels were significantly reduced in both the TZP
and DM+TZP groups compared with the DM group (p
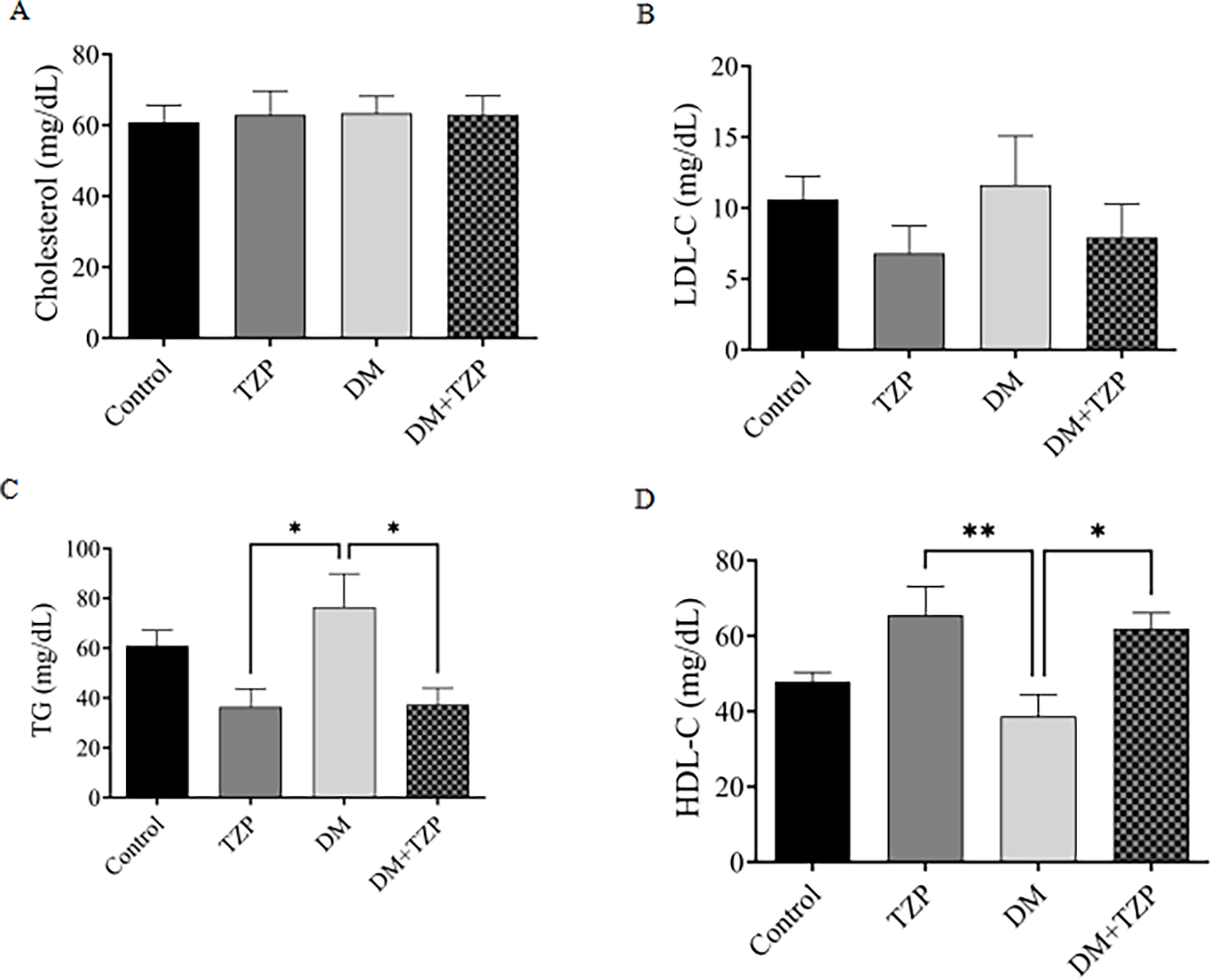 Fig. 6.
Fig. 6.
Effect of DM and TZP on lipid profile: (A) TC, (B)
LDL-C, (C) TG, and (D) HDL-C. *p
Serum TSH levels did not differ significantly among the experimental groups, indicating that neither DM nor TZP treatment markedly affected TSH secretion (Fig. 7A). Diabetic rats exhibited significantly lower T4 levels than controls, and TZP treatment significantly attenuated this reduction (Fig. 7B). Conversely, T3 levels were significantly elevated in diabetic rats and were partially normalized following TZP treatment (Fig. 7C). Collectively, these results suggest that TZP modulates diabetes-associated thyroid hormone dysregulation.
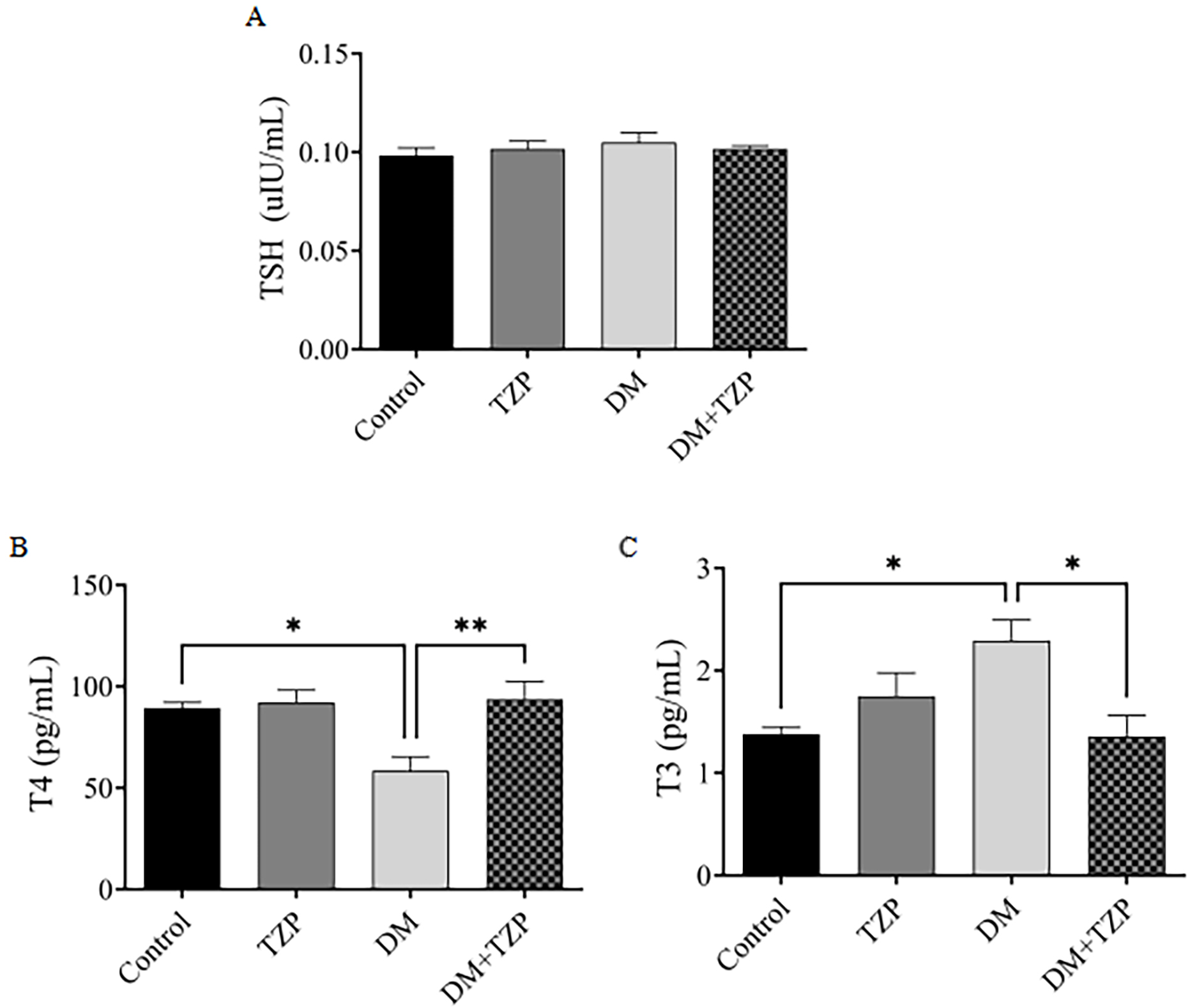 Fig. 7.
Fig. 7.
Effect of DM and TZP on thyroid hormone levels: (A) TSH, (B) T4,
and (C) T3. *p
This study investigated the effects of tirzepatide (TZP) in a streptozotocin (STZ)-induced diabetic rat model, assessing its impact across a comprehensive range of metabolic, renal, endocrine, and cardiovascular parameters. The findings demonstrate that TZP confers significant multisystem benefits, preventing diabetes-associated mortality, improving glycemic control, ameliorating dyslipidemia, partially attenuating renal dysfunction, and modulating thyroid hormone homeostasis. However, the study also revealed complex biochemical changes, such as elevated lactate dehydrogenase (LDH), that warrant careful interpretation. This discussion will interpret these findings by integrating a parameter-specific analysis with a broader mechanistic and comparative assessment, while also critically appraising the study’s limitations.
One of the most notable observations was the effect of TZP on survival. While a modest mortality rate was observed in untreated diabetic rats, no mortality occurred in those treated with TZP, suggesting that TZP prevented diabetes-associated mortality in this experimental setting rather than enhancing survival beyond normal levels. This protective effect is likely attributable to the compound’s multifaceted metabolic benefits [27, 28]. Interestingly, despite this survival advantage, TZP did not reverse the body weight loss observed in diabetic rats, indicating that its protective effects were not mediated by weight restoration but rather by systemic metabolic and endocrine modulation.
Regarding glycemic control, TZP significantly reduced blood glucose levels in
diabetic rats while exerting no effect in non-diabetic animals, consistent with
the glucose-dependent mechanism of incretin-based therapies [8, 29]. Despite the
reduction in hyperglycemia, TZP did not restore serum insulin levels, suggesting
that its antihyperglycemic action in this
Serum troponin I and creatine kinase-MB (CK-MB) levels, widely used indicators of cardiomyocyte necrosis [31, 32], remained unchanged across all experimental groups. Their stability suggests that neither diabetes nor TZP treatment induced overt myocardial cell injury during the 10-day study period. However, it is important to recognize that these markers may lack sufficient sensitivity to detect mild or subclinical cardiac stress [33, 34]. Therefore, while the data show no evidence of overt cardiotoxicity, they cannot exclude subtle myocardial alterations [35, 36].
In contrast, LDH activity was markedly elevated following TZP administration in non-diabetic rats. As a ubiquitous enzyme present in multiple tissues including the liver, kidney, and skeletal muscle [37, 38, 39, 40, 41], the elevation of LDH may reflect systemic tissue stress or altered metabolic enzyme activity rather than specific cardiotoxicity. The data revealed a complex interplay, with diabetes alone being associated with lower LDH activity, while the DM+TZP group showed intermediate levels. This suggests that the diabetic state may alter drug metabolism or cellular susceptibility, thereby attenuating the effect of TZP. This disparity between cardiac-specific markers and LDH underscores the importance of a multi-marker approach in evaluating drug safety, as reliance on one class of biomarkers alone may underestimate systemic effects [42].
The partial attenuation of elevated BUN and creatinine levels in diabetic rats suggests a modest renoprotective effect, likely secondary to improved metabolic control [43, 44]. However, the benefits may extend beyond simple glycemic management. Recent research indicates that TZP can normalize diabetic nephropathy by suppressing oxidative stress through the PI3K/AKT signaling pathway [45]. Activation of this pathway in podocytes enhances antioxidant capacity, mitigating high-glucose-induced cellular injury. This is further supported by post-hoc analyses of the SURPASS-4 trial, which demonstrated that TZP significantly reduced the urine albumin-to-creatinine ratio (UACR) and slowed the decline of eGFR compared to insulin glargine, suggesting direct renal benefits [10]. The observed improvements in our study are likely attributable to a combination of improved systemic metabolic control and these direct, tissue-specific protective mechanisms.
Diabetes produced the expected dyslipidemic profile of increased triglycerides (TG) and reduced HDL-C. TZP treatment favorably modulated this profile by significantly reducing TG and increasing HDL-C, a finding of high clinical relevance as these lipid fractions are strong predictors of atherogenesis and cardiovascular events [46, 47, 48]. The mechanisms underlying these effects are multifaceted and highlight the advantage of TZP’s dual-agonist profile. GLP-1 receptor agonism is known to reduce hepatic production of very-low-density lipoprotein (VLDL), a primary carrier of triglycerides. The addition of GIP receptor agonism provides a synergistic effect by enhancing lipoprotein lipase activity in adipose tissue, which promotes the clearance of triglyceride-rich lipoproteins from circulation [49]. This dual action likely explains the robust effects on triglycerides and HDL-C observed in our study and in large-scale clinical trials, which consistently show TZP’s superiority over selective GLP-1 agonists in improving atherogenic lipid profiles [49].
This study also sheds light on the complex interplay between TZP and thyroid hormone homeostasis. The pattern of low T4 and high T3 in untreated diabetic rats may reflect a state of enhanced peripheral conversion of T4 to the active hormone T3, a potentially maladaptive response to hyperglycemic stress driven by changes in deiodinase enzyme activity. TZP’s ability to normalize this pattern without altering TSH levels points towards a peripheral, rather than central, regulatory action, possibly by normalizing hepatic deiodinase activity secondary to improved metabolic health [50, 51, 52]. This finding is consistent with literature on GLP-1 receptor agonists and adds to the understanding of TZP’s broader endocrine effects [53].
This study’s findings contribute to a growing body of evidence demonstrating the superior efficacy of TZP as a dual GIP/GLP-1 receptor agonist compared to selective GLP-1 receptor agonists like semaglutide. The landmark SURPASS series of clinical trials consistently showed that TZP achieves greater reductions in HbA1c , which is the average amount of glucose (sugar) attached to hemoglobin in your red blood cells over the past two to three months, and body weight compared to semaglutide [27]. The results of our study, showing robust improvements across multiple metabolic parameters, align with these clinical data and underscore the added value of GIP receptor co-activation. The GIP component contributes to enhanced insulin secretion, improved lipid clearance, and has direct effects on appetite regulation in the central nervous system, creating a synergistic effect that surpasses what can be achieved with GLP-1 agonism alone [54]. The survival benefit observed in our TZP-treated diabetic rats provides compelling preclinical evidence for the profound and multifaceted benefits of this dual-agonist approach in mitigating the severe complications of diabetes.
While this study provides valuable insights, its limitations must be carefully
considered. A primary limitation is the use of the STZ-induced diabetes model,
which, by destroying pancreatic
Several methodological factors also limit the direct translatability of the results. The intraperitoneal (IP) route of administration differs from the subcutaneous (SC) route used clinically, which could lead to different pharmacokinetic profiles [56]. Furthermore, the short study duration (10 days) means the observed biomarker shifts represent acute responses. It is crucial to avoid extrapolating these to infer long-term organ protection, and future studies with longer observation periods are essential.
A significant and acknowledged limitation is the absence of histopathological analysis. While biochemical markers provide valuable functional data, they do not offer insight into the underlying structural integrity of organs. The elevation of LDH, a non-specific marker, highlights this gap; without histology, it is impossible to localize the source of this enzyme release. Future studies must incorporate comprehensive histopathological evaluation, including H&E, PAS, and Masson’s trichrome staining, to assess general morphology, glycogen deposition, and fibrosis in key tissues like the kidney and heart. Such analyses are critical to confirm whether the biochemical improvements translate into tangible structural protection, thereby providing definitive evidence for TZP’s organ-protective effects.
In this STZ-induced diabetic rat model, tirzepatide prevented diabetes-associated mortality and demonstrated a wide range of favorable metabolic effects, including improved glycemic control, reduced triglycerides, increased HDL-C, partial attenuation of renal dysfunction markers, and normalization of thyroid hormone imbalances. The dual agonism of GIP and GLP-1 receptors provides a powerful, synergistic mechanism that offers advantages over selective GLP-1 therapies. While no evidence of overt myocardial necrosis was observed, the elevation of LDH in non-diabetic rats warrants further investigation into systemic safety. Although constrained by the inherent limitations of the STZ model and the absence of histological data, this study provides a strong rationale for further investigation. Long-term studies incorporating histopathological and functional assessments are essential to fully elucidate the mechanisms of TZP’s organ-protective effects and to confirm its long-term benefits in clinical practice.
ANOVA, Analysis of variance; BUN, Blood urea nitrogen; CK-MB, Creatine kinase–MB; DM, Diabetes mellitus; ECL, Electrochemiluminescence immunoassay; GIP, Glucose-dependent insulinotropic polypeptide; GLP-1, Glucagon-like peptide-1; HDL-C, High-density lipoprotein cholesterol; i.p., Intraperitoneal; LDH, Lactate dehydrogenase; LDL-C, Low-density lipoprotein cholesterol; SEM, Standard error of the mean; STZ, Streptozotocin; T3, Triiodothyronine; T4, Thyroxine; TC, Total cholesterol; TG, Triglycerides; TSH, Thyroid-stimulating hormone; TZP, Tirzepatide.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
AMA and AHA carried out the experiments, analyzed and interpreted, and also prepared the scientific manuscript. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Scientific Ethics Committee at Deanship of Graduate Studies and Scientific Research, Qassim University under Approval No. 25-20-01. All procedures were conducted in accordance with the relevant Qassim University guidelines for animal care and use.
Not applicable.
The authors gratefully acknowledge Qassim University, represented by the Deanship of Graduate Studies and Scientific Research, on the financial support for this research under the number (QU-J-PG-2-2025- 54140) during the academic year 1446 AH/2024 AD.
The authors declare no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







