1 Department of Pharmacology and Toxicology, College of Pharmacy, Taibah University, 41477 Madinah, Saudi Arabia
2 Department of Pharmacy Practice, College of Pharmacy, Taibah University, 41477 Madinah, Saudi Arabia
3 Department of Clinical Laboratory Sciences, College of Applied Medical Sciences, Taibah University, 41477 Madinah, Saudi Arabia
4 Health and Life Research Center, Taibah University, 41477 Madinah, Saudi Arabia
Abstract
As a major metabolic organ, the liver is constantly exposed to toxic substances. Although the flesh and seeds of Ajwa dates have been established to have antioxidant and anti-inflammatory properties, the hepatoprotective potential of Ajwa seed oil (ASO) against CCl4-induced damage has yet to be ascertained.
Twenty-four adult male mice were randomly assigned to one of four groups (n = 6 per group): control (oral saline for 12 days), CCl4-treated [saline for 12 days plus a single dose of CCl4 dissolved in corn oil 1:1 (1 mL/kg)], CCl4 + ASO (100 mg/kg ASO administered daily for 12 days, followed by CCl4), and CCl4 + silymarin (100 mg/kg silymarin administered daily for 12 days, followed by CCl4). Blood and liver samples were collected 24 h after CCl4 administration for biochemical and histological analyses.
ASO treatment substantially reduced CCl4-induced liver damage by restoring normal serum liver enzyme levels (alanine aminotransferase, aspartate aminotransferase, and lactate dehydrogenase; p < 0.05), lowering elevated levels of cholesterol and triglycerides, and increasing albumin levels (p < 0.05). The treatment significantly reduced lipid peroxidation, as evidenced by decreased malondialdehyde levels (p < 0.0001), and concurrently restored the total antioxidant capacity. Histological analysis revealed the preservation of liver architecture, reduced collagen deposition, and maintenance of glycogen storage (p > 0.05).
Our findings in this study revealed that ASO has beneficial hepatoprotective effects against CCl4-induced injury, which can be attributed largely to its antioxidant properties, as evidenced by restored total antioxidant capacity and reduced lipid peroxidation. Anti-inflammatory mechanisms may also contribute, as suggested by the histological reduction in inflammatory cell infiltration; however, direct measurement of inflammatory biomarkers warrants investigation in future studies.
Keywords
- Ajwa seed oil
- hepatoprotective
- carbon tetrachloride
- oxidative stress
- liver injury
As a major metabolic organ, the liver plays pivotal roles in detoxification, protein synthesis, and the regulation of glucose levels [1]. However, although the liver has unique regenerative capacities, the excessive consumption of alcohol or drugs and ingestion of other environmental xenobiotics can potentially cause severe hepatic damage, including hepatic fibrosis, cirrhosis, and hepatocellular carcinoma [2, 3]. Oxidative stress and inflammation have been established to be the primary factors contributing to the induction and propagation of liver injury, and thus suppressing these events may provide the key to controlling and mitigating their damaging effects [4, 5, 6].
A mounting body of evidence indicates that different types of liver injury can
be prevented using antioxidant-rich natural products, owing to their capacities
to eliminate oxidative stress and hence suppress inflammatory responses [7, 8, 9]. In recent years, a growing number of studies have specifically
demonstrated the hepatoprotective potential of plant-derived oils, phenolic
extracts, and flavonoid-rich fractions in experimental models of liver toxicity
[10, 11]. These phytoingredients have been shown to attenuate CCl4-induced
hepatotoxicity through multiple mechanisms, including activation of the
Nrf2/Keap1 antioxidant signaling pathway, suppression of NF-
Carbon tetrachloride (CCl4) is often used experimentally to induce hepatic injury in animals for evaluation of the hepatoprotective properties of drugs of interest. The toxicity induced by CCl4 is associated with the generation of trichloromethyl free radicals, which contribute to oxidative damage, lipid peroxidation, and death of hepatic cells [18, 19]. In this study, we assessed the capacity ASO to confer protection against CCl4-induced acute liver damage, with the efficacy being compared with that of silymarin, a well-established hepatoprotective drug [20]. Collectively, the findings of this study will contribute to enhancing our understanding of the properties of ASO as a source of antioxidants in managing liver injury and thereby augment the growing body of evidence on the potential use of natural products as hepatoprotective agents.
Twenty-four adult male BALB/c mice were selected from the animal house facility
at the College of Pharmacy, Taibah University, Madinah. Prior to the initiation of
experimental procedures, the mice were habituated for 1 week under controlled
environmental conditions (25
The experimental design was adapted from established CCl4-induced hepatotoxicity models [18, 19] and our previous work with date seed oil [21], with modifications specific to our objectives
The 24 mice were randomly assigned to one of the following four groups (n = 6 per group). Group 1 mice (serving as controls) received saline orally for 12 consecutive days. Group 2 mice received saline for 12 days, and 2 h after the final dose, the mice were administered CCl4 in corn oil (1:1) via intraperitoneal injection (1 mL/kg). Group 3 mice received a daily oral dose of ASO (100 mg/kg) for 12 days, followed by a single intraperitoneal dose of CCl4 (1 mL/kg) 2 h after the final dose. The 100 mg/kg dosage was selected based on preliminary dose-assessment studies (evaluating doses ranging from 50 to 200 mg/kg) conducted in our laboratory and previously published research [21]. This dose has been shown to provide substantial antioxidant activity in rodent models in the absence of any marked toxicity. Group 4 mice received a daily oral dose of silymarin (100 mg/kg) for 12 days, followed by a single CCl4 injection 2 h after the final dose. At 24 h post-CCl4administration, the animals were humanely euthanized under anesthesia induced with ketamine (100 mg/kg IP) and xylazine (10 mg/kg IP). Euthanasia was performed using a gradual-fill CO2 procedure in accordance with institutional and international guidelines. Cervical dislocation was applied as a secondary physical method to ensure death. Blood was collected by intracardiac puncture, and serum was subsequently separated. Liver samples were collected and rinsed in saline, and thereafter divided into two portions, one of which was used for tissue homogenization to measure the level of malondialdehyde (MDA) and total antioxidant (TAO) activity and the other for histopathological investigation.
A 12-day pre-treatment period was selected based on the previously established time considered necessary for the bioactive constituents of ASO to reach steady-state tissue levels and optimize the efficacy of endogenous antioxidant defense systems in hepatic tissue [21, 22]
CCl4 was obtained from Sigma-Aldrich and silymarin was sourced from Indena (Settala, Italy). Unless otherwise stated, all other chemicals and reagents were procured from Sigma-Aldrich.
Botanical Authentication: Ajwa dates (Phoenix dactylifera L., family Arecaceae) were acquired from a certified local supplier in Al-Madinah Al-Munawwarah, Saudi Arabia. Ripe whole fruits free from any irregularities, infections or damage were selected and cleaned for the purpose of seed collection. Following a previously described procedure [21], the seeds were thoroughly cleaned to eliminate any remaining fruit pulp, followed by washing and drying at moderate heat. Thereafter, the seeds were ground into a fine powder, which was maintained at 4 °C prior to use.
Having initially been sifted, the date seed powder was extracted overnight in a sealed container by mixing 20 g of powder with 500 mL of methanol at 40 °C on a magnetic stirrer. This extraction process was performed a total of three times, following which, the solvent was evaporated using a Soxhlet apparatus over 6 h, with an oil yield of approximately 8% relative to the seed powder dry weight.
Serum was separated from clotted samples of blood collected from mice by
centrifuging at 3000
Extracted liver tissues were initially fixed overnight in 10% formalin (w/v in phosphate-buffered saline) at 4 °C. Tissues underwent conventional histological processing prior to being fixed in paraffin and sectioned at a thickness of 5 µm. Sections were deparaffinized, rehydrated in a graded alcohol series, and stained with hematoxylin and eosin (H&E) to assess overall liver morphology and with Masson’s trichrome to determine collagen deposition and fibrosis. Stained sections were examined under a light microscope to assess differences in hepatocellular damage and structural changes among the experimental groups.
Liver tissue sections were evaluated by a blinded, board-certified pathologist using a semi-quantitative scoring system. Histopathological parameters were scored from 0 to 3 as follows: 0 = absent/normal, 1 = mild, 2 = moderate, and 3 = severe. The assessed parameters included hepatocellular necrosis, inflammatory cell infiltration, sinusoidal congestion, vacuolar degeneration, and collagen deposition in Masson’s trichrome-stained sections.
Glycogen content in PAS-stained sections was scored using a reverse scale: 0 = severe depletion, 1 = mild depletion, 2 = moderate retention, and 3 = abundant/normal glycogen.
Total histopathological scores were calculated by summing the individual
parameter scores for each animal. For each parameter, at least 10 random
high-power fields (
| Parameter | Control | CCl4 | CCl4 + ASO | CCl4 + Silymarin |
| Hepatocellular necrosis | 0.2 |
2.8 |
0.4 |
0.3 |
| Inflammatory infiltration | 0.3 |
2.6 |
0.5 |
0.4 |
| Sinusoidal congestion | 0.1 |
2.4 |
0.3 |
0.3 |
| Vacuolar degeneration | 0.2 |
2.7 |
0.4 |
0.4 |
| Early collagen accumulation* | 0.3 |
2.3 |
0.5 |
0.4 |
| Glycogen content | 2.9 |
0.4 |
2.7 |
2.8 |
| Total pathology score | 1.1 |
12.8 |
2.1 |
2.0 |
*Collagen accumulation observed at 24 hours post-CCl4 administration
represents an acute-phase extracellular matrix response and early hepatic
stellate cell activation, not established fibrosis. True hepatic fibrosis
requires repeated or chronic CCl4 exposure over several weeks. This
parameter is therefore interpreted as an early fibrogenic signal, and the term
‘collagen deposition’ is used descriptively to reflect Masson’s trichrome
staining findings rather than to imply frank fibrosis. Values represent mean
All data are expressed as the means
To assess hepatocyte integrity, the levels of ALT, AST, and LDH were measured in
the serum of blood collected from mice in the different treatment groups.
Compared with levels detected in the control group mice, treatment with CCl4 was observed to promote significant increases in the levels of serum ALT,
AST, and LDH (p
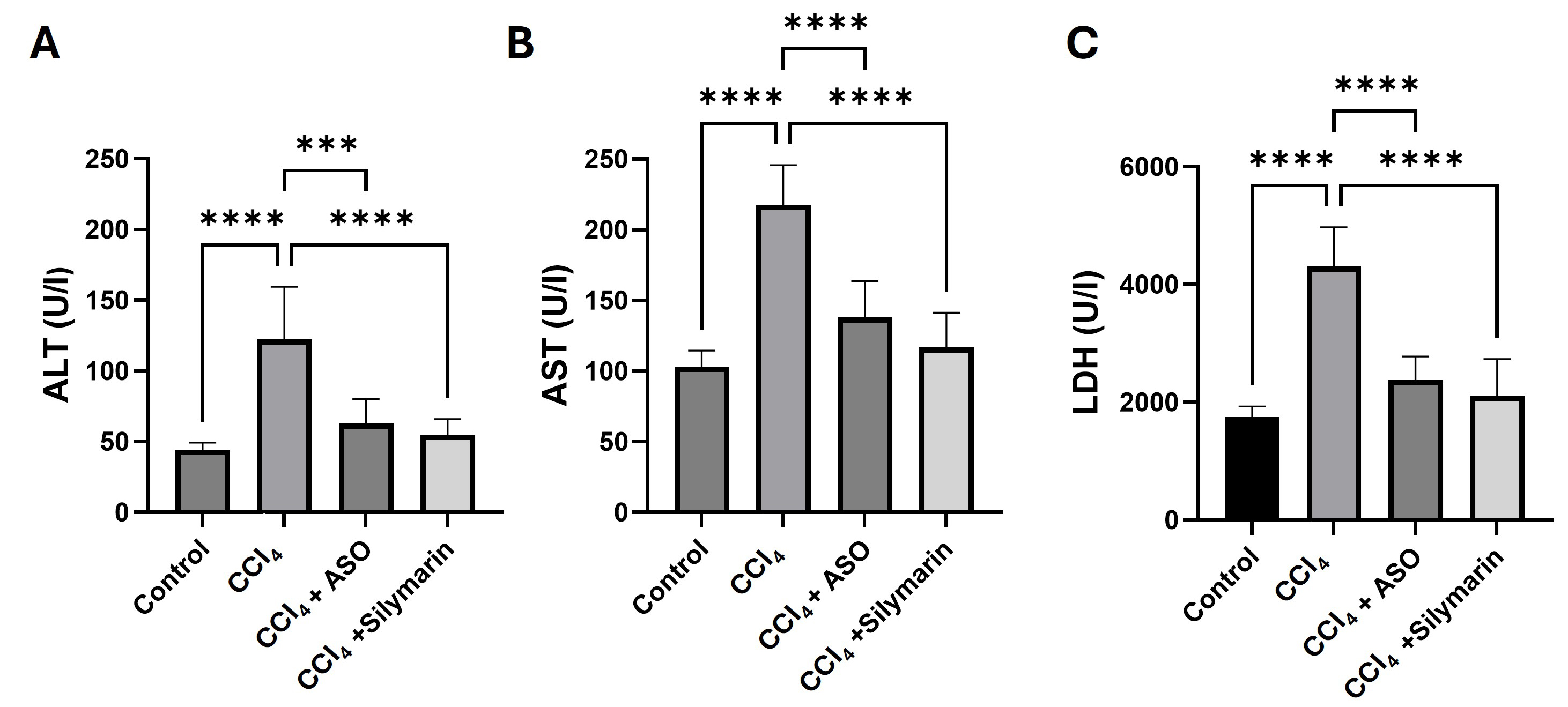 Fig. 1.
Fig. 1.
Effects of ASO on serum liver biomarkers. (A–C) Serum
biomarkers of different treatment groups. Data are presented as the
means
Serum levels of cholesterol, triglycerides, and albumin, which are widely
examined as biomarkers indicative of hepatocellular injury and impaired synthetic
function, were measured to determine the effects of different treatments on liver
biosynthetic capacity. Administration of CCl4 led to marked increases in the
levels of blood cholesterol and triglyceride (p
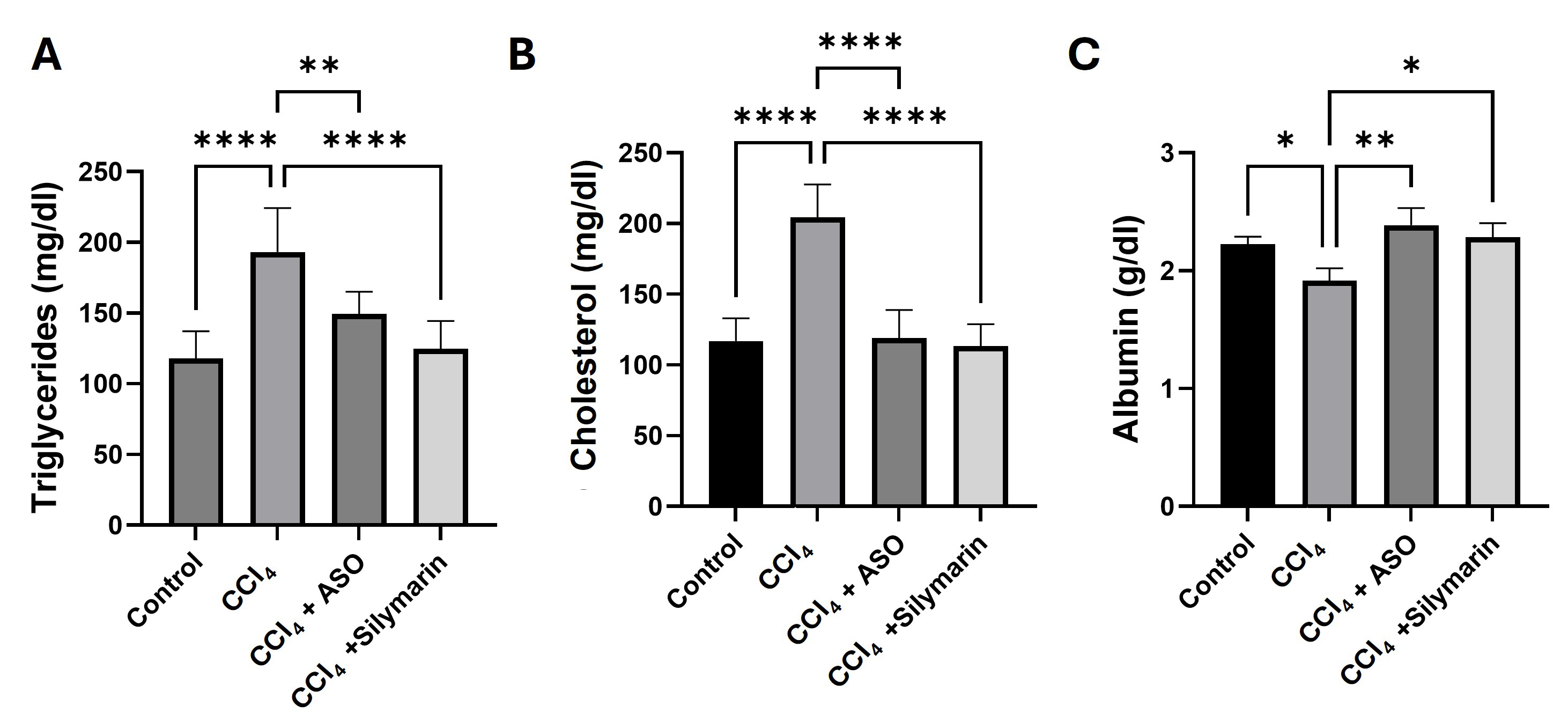 Fig. 2.
Fig. 2.
Effects of ASO on serum cholesterol, triglycerides, and albumin
levels. (A–C) Serum biomarkers in different treatment groups. Data are
presented as the means
Serum MDA levels, a well-established biomarker of lipid peroxidation, were
measured to assess the degree of CCl4-induced oxidative stress in hepatic
tissue. Relative to the control mice, the administration of CCl4 was
observed to promote marked increases in serum levels of MDA (p
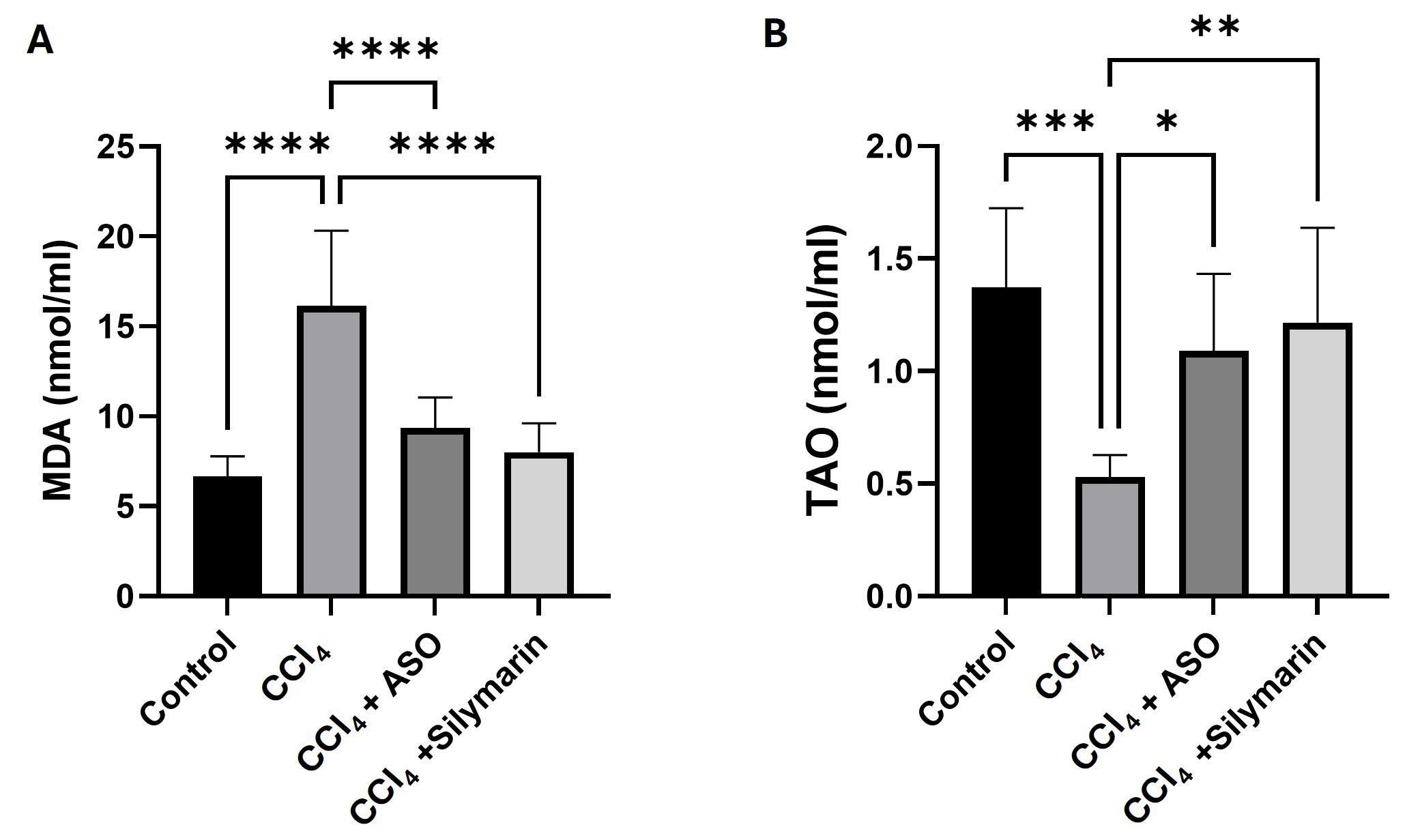 Fig. 3.
Fig. 3.
Effects of ASO on serum malondialdehyde (MDA) levels and total
antioxidant (TAO) activity. (A,B) Serum biomarkers for the different
treatment groups. Data are presented as the means
To assess the antioxidant defence capacity of liver, we also determined the
levels of TAO activity in mouse sera. CCl4 treatment was observed to result
in a significant depletion in TAO activity, reflecting the heightened level of
oxidative stress (p
To examine the histoarchitecture and structural integrity of liver tissues, we prepared hematoxylin- and eosin-stained sections from liver tissues collected from mice in the different treatment groups. Tissues obtained from the control mice were observed to have a normal morphology and architecture, with well-preserved hepatocytes (Fig. 4A). In contrast, tissues obtained from the CCl4-treated mice showed evidence of substantial damage, including portal inflammation, venous congestion, and fatty changes. including hepatocyte vacuolization and fatty droplet accumulation (Fig. 4B). Comparatively, tissues derived from mice treated with either ASO or silymarin were characterized by a marked amelioration of the CCl4-induced liver damage, with hepatic architecture restored to virtually normal levels (Fig. 4C,D).
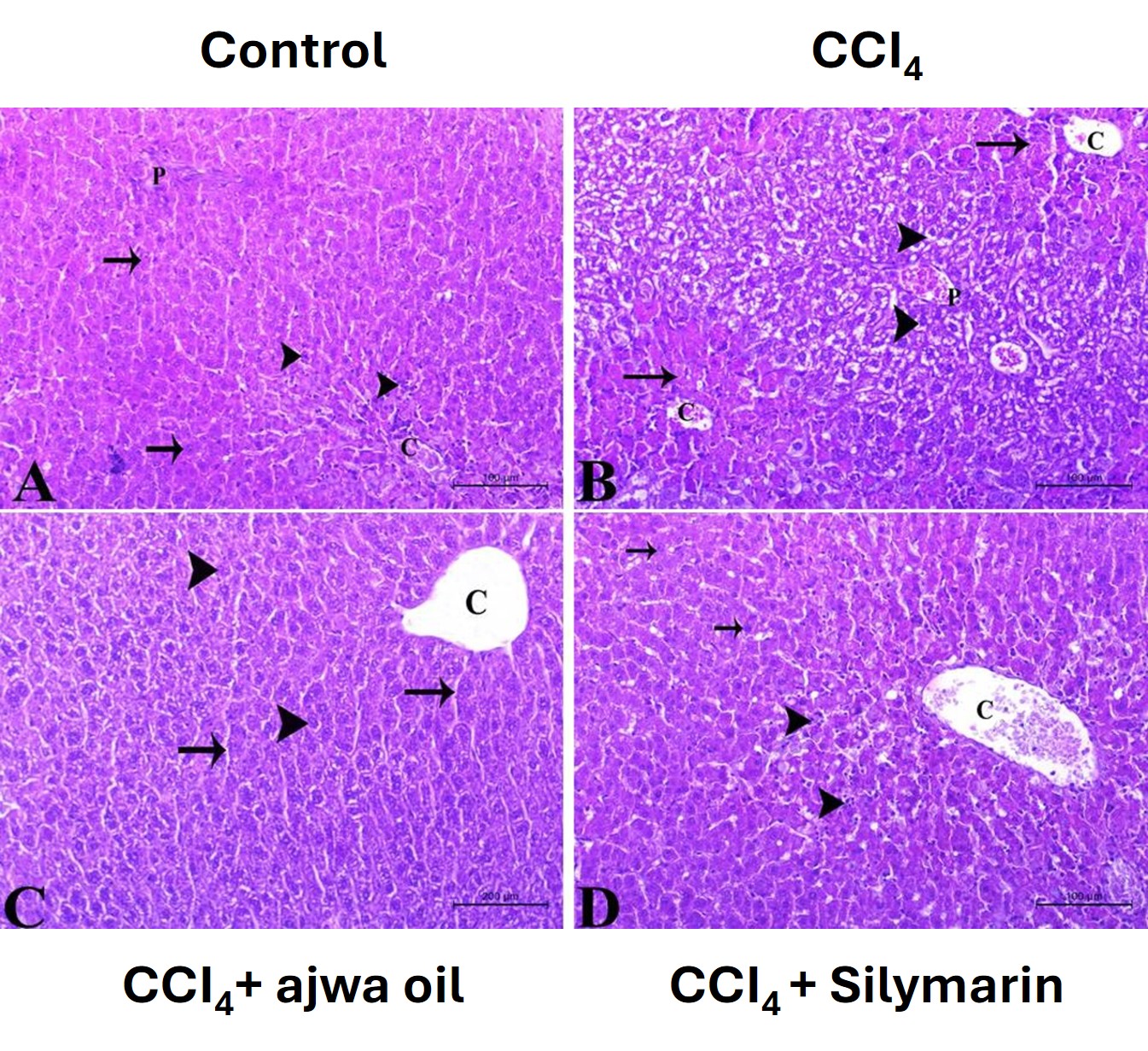 Fig. 4.
Fig. 4.
Photomicrographs of hematoxylin- and eosin-stained sections used
to assess the histological architecture of liver tissues in mice in the different
treatment groups (
To assess the effects on the different treatments on collagen fiber deposition and fibrosis, we performed Masson’s trichrome staining. Sections of tissues obtained from the control mice were characterized by a limited accumulation of collagen and an intact liver architecture (Fig. 5A). Comparatively, tissues obtained from CCl4-treated mice showed evidence of a substantial deposition of collagen, the development of fibrous septa, and an altered liver architecture (Fig. 5B). Notably the administration of both ASO and silymarin contributed to marked reductions in collagen deposition and also restored the aberrant hepatic architecture (Fig. 5C,D).
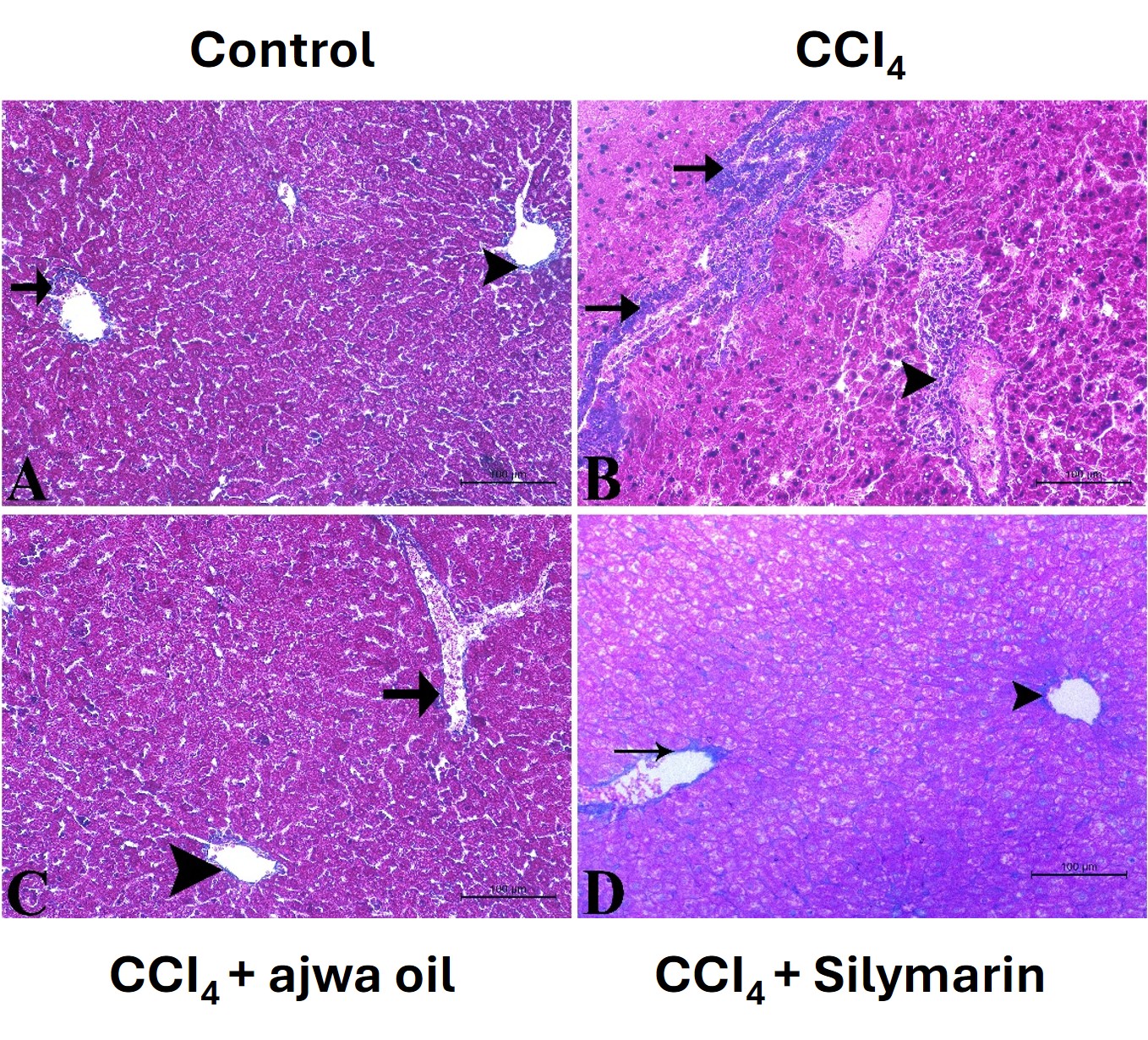 Fig. 5.
Fig. 5.
Photomicrographs of liver tissue sections stained with
Masson’s trichrome for assessment of collagen deposition in mice in the different
treatment groups (magnification
Periodic acid-Schiff (PAS) staining was used to identify glycogen and determine hepatocyte functional integrity. Whereas liver sections prepared for control animals revealed normal hepatocyte glycogen levels (Fig. 6A), those from the CCl4-administered mice showed reductions in the intensity of PAS staining, indicative of a depletion in glycogen caused by hepatocellular injury (Fig. 6B). Administration of ASO or silymarin restored glycogen reserves, as indicated by an enhanced intensity of PAS staining (Fig. 6C,D).
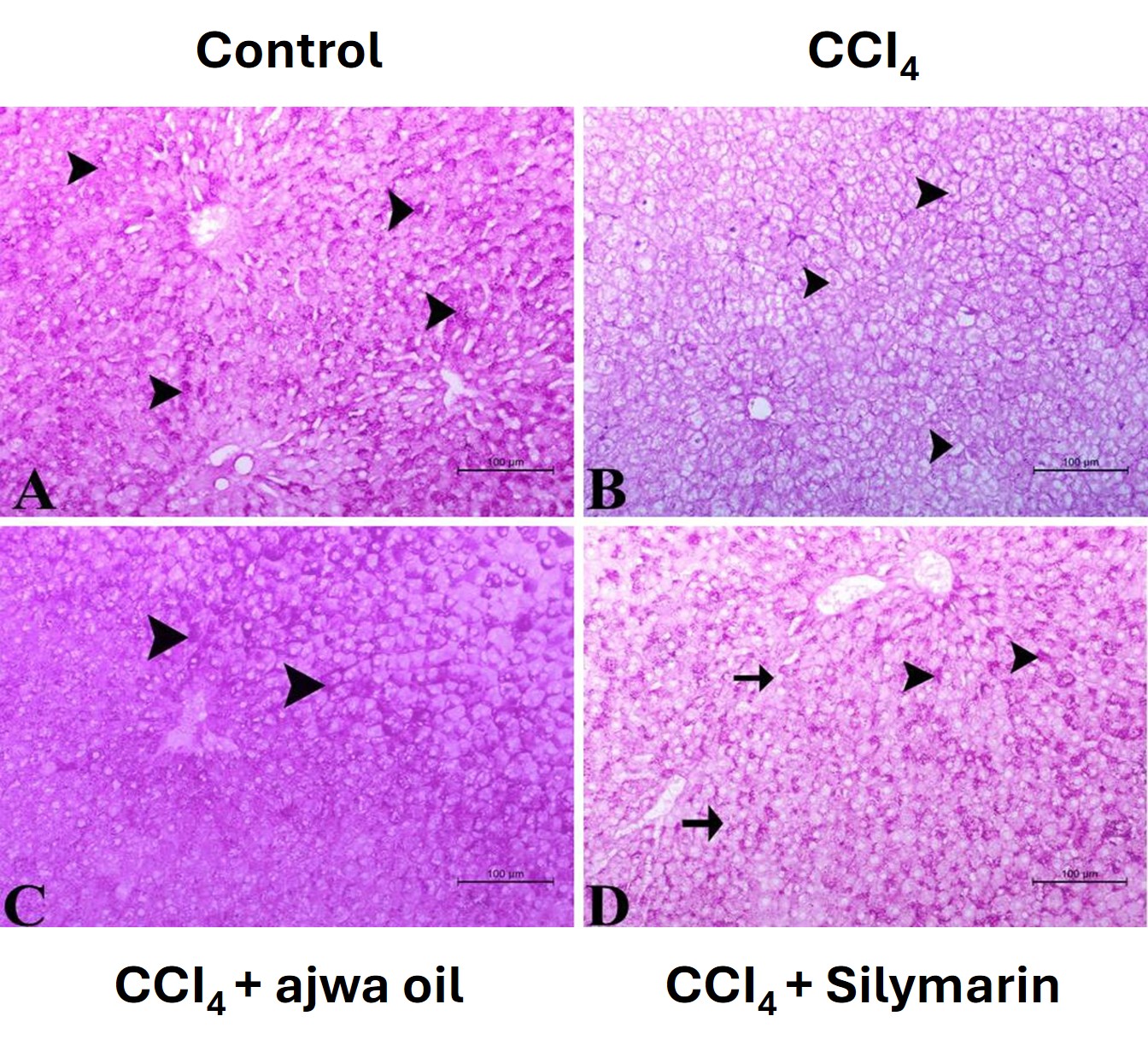 Fig. 6.
Fig. 6.
Photomicrographs of liver sections stained with periodic acid-Schiff (PAS) for assessment of glycogen storage. (A) Section from a control mouse showing pronounced PAS staining of hepatocytes (arrowhead). Scale bar = 100 µm. (B) Section from a CCl4-treated mouse showing minimal PAS staining of hepatocytes (arrowhead). Scale bar = 100 µm. (C) Section from a CCl4 + Ajwa seed oil-treated mouse showing strong PAS staining of hepatocytes (arrowhead). Scale bar = 100 µm. (D) Section from a CCl4 + silymarin-treated mouse showing moderate to strong PAS staining of hepatocytes (arrowhead), with a few hepatocytes showing weaker staining (arrow). Scale bar = 100 µm.
Table 1 summarizes the semi-quantitative histopathological scores for all
evaluated parameters. ASO treatment significantly reduced all CCl4-induced
pathological changes (p
The liver, as a key metabolic organ, is continuously exposed to multiple chemicals and is consequently particularly prone to oxidative stress. In this regard, CCl4, a well-established hepatotoxic agent, is widely used in experimental models to induce liver damage and fibrosis [18, 19]. Although the flesh and seeds of Ajwa dates have been demonstrated to have antioxidant and anti-inflammatory properties [10, 16], the hepatoprotective effects of ASO against CCl4-induced liver damage has yet to be assessed. In this study, we accordingly sought to determine the protective effects of ASO against CCl4-induced hepatotoxicity in mice using biochemical and histological techniques.
CCl4-treated mice were observed to have sustained substantial liver damage, characterized by elevated levels of serum parameters such as ALT, AST, LDH, cholesterol, and triglyceride, as well as reductions in albumin levels, which are features indicative of hepatocellular damage and compromised liver functions. CCl4 also perturbed hepatic oxidative status, depleting total antioxidant capacity and inducing lipid peroxidation, thereby contributing to heightened levels of oxidative stress. Moreover, histological analyses revealed structural damage to liver tissues, characterized by cellular degeneration, elevated levels of collagen deposition, and a depletion of glycogen, changes that are consistent with liver injury and early fibrosis and are frequently seen in the study of hepatotoxicity [5, 18].
The administration of ASO was found to ameliorate CCl4-induced liver damage at both the biochemical and histological levels, contributing to a normalization of the serum concentrations of ALT, AST, LDH, lipids, and albumin, whilst restoring hepatic antioxidant capacity. Similarly, histological evaluations of liver tissues obtained from ASO-treated mice revealed reductions in CCl4-induced hepatocellular injury and fibrosis [22]. Furthermore, PAS staining revealed that mice receiving ASO were characterized by a well-preserved hepatic architecture with reductions in the depletion of glycogen, thereby providing evidence of the hepatoprotective efficacy of this oil.
To a large extent, the observed hepatoprotective effects of ASO can be attributed to its antioxidant properties. In this regard, previous studies have reported that the antioxidant activities of ASO are associated with its phenolic and flavonoid contents [23]. Antioxidant-rich compounds have previously been shown to protect against the hepatocellular damage induced by oxidative stress [8, 9], mitigating oxidative stress in hepatocytes, facilitating cellular recovery, and minimizing the evidence of intoxication. The hepatotoxicity attributable to CCl4 is primarily mediated via the generation of reactive oxygen species, leading to oxidative stress [6, 24], causing lipid peroxidation, membrane damage, DNA damage, and inflammation [5, 18]. The amelioration of liver damage markers and reduction in the indicators of fibrosis observed in ASO-treated mice provides evidence that this oil facilitates hepatoprotection via mechanisms associated with the elimination of free radicals [25].
Chronic administration of hepatotoxic agents such as CCl4 can induce hepatic damage via activation of hepatic stellate cells that promote collagen synthesis and deposition, resulting in liver fibrosis [26]. Although established hepatic fibrosis typically develops following repeated or chronic CCl4 exposure rather than a single acute dose, the elevated collagen deposition observed at 24 hours in our model represents an early fibrogenic response, likely reflecting acute hepatic stellate cell activation and extracellular matrix remodeling rather than true fibrosis. The ability of ASO to attenuate this early collagen deposition suggests a potential anti-fibrogenic effect that warrants validation in chronic CCl4 models with repeated dosing over several weeks [27].
In addition to the aforementioned conditions, by mediating the release of
different pro-inflammatory cytokines at the site of injury, oxidative stress also
plays a key role in inducing inflammation, as has previously been reported with
respect to CCl4-induced liver damage [19]. The protective effects of ASO
could thus also be explained in terms of its capacity to replenish endogenous
antioxidant capacity and scavenge free radicals, thereby ameliorating the
inflammatory response and preventing the progression of fibrosis [16]. These
findings are consistent with a growing body of recent literature demonstrating
that phenolic-rich plant extracts and seed oils ameliorate CCl4-induced
hepatotoxicity through Nrf2 pathway activation, restoration of
glutathione-dependent antioxidant enzymes, and suppression of
NF-
On the basis of our findings in this study and those reported in the existing
literature, we propose a multi-faceted mechanism for the hepatoprotective effects
of ASO against CCl4-induced liver injury. Firstly, it is assumed that the
phenolic compounds and flavonoids identified in ASO (gallic acid, catechin,
epicatechin, and ferulic acid) act as direct free radical scavengers,
neutralizing the trichloromethyl radicals generated by the metabolism of
CCl4. Secondly, ASO appears to enhance the efficacy of endogenous
antioxidant defense systems, as evidenced by the restoration of total antioxidant
capacity, potentially mediated via an upregulation of antioxidant enzymes such as
superoxide dismutase, catalase, and glutathione peroxidase. Thirdly, the high
contents of unsaturated fatty acids (particularly oleic and linoleic acids) in
ASO may contribute to stabilizing hepatocyte membranes and reduce the levels of
lipid peroxidation, as indicated by a significant reduction in serum MDA levels.
Fourthly, it is plausible that the anti-inflammatory properties of ASO are
associated with a suppression of the production of pro-inflammatory cytokine and
inhibition of inflammatory cell infiltration into hepatic tissues. Finally, the
reductions in collagen deposition observed in ASO-treated mice indicate that this
oil may prevent hepatic stellate cell activation, thereby inhibiting the early
stages of fibrogenesis. Consequently, future studies employing molecular pathway
analysis, including assessments of Nrf2 signaling, NF-
Our finding that with respect to all evaluated parameters, the observed efficacy
of ASO was comparable to that of the reference hepatoprotective agent silymarin,
is particularly relevant. Specifically, ASO-treated animals showed no significant
difference from those treated with silymarin regarding the levels of liver
enzymes, lipid profiles, oxidative stress markers, and histological features
(p
Although in this study we utilized multiple complementary methods to assess liver damage and protection, encompassing biochemical indicators, oxidative stress parameters, and comprehensive histological examinations, there are certain limitations that should be acknowledged.
Notably, we primarily examined the preventive effects of ASO administered prior to exposing mice to CCl4, rather than assessing its therapeutic potential subsequent to the induction of liver damage. A key limitation of this study is the use of a single-dose acute CCl4 model with tissue collection at 24 hours. True hepatic fibrosis, characterized by progressive collagen cross-linking and fibrous septum formation, requires repeated or chronic CCl4 administration typically over 4–8 weeks. Therefore, the collagen deposition observed in this study should be interpreted as an early fibrogenic signal rather than established fibrosis, and the anti-fibrotic claims of ASO must be validated using a chronic liver injury model. Moreover, the molecular mechanisms underlying the hepatoprotective effects of ASO, including specific signaling pathways, gene expression profiles, and detection of the bioactive compounds associated with these effects, have yet to be thoroughly elucidated. For example, we did not examine the levels of inflammatory cytokine or markers of hepatic stellate cell activation, which would contribute to enhancing our understanding of the anti-inflammatory and anti-fibrotic mechanisms of ASO. Similarly, we have yet to address dose–response relationships or potential adverse effects associated with the prolonged administration of ASO.
In further research, it will be necessary to focus on evaluating the therapeutic efficacy of ASO in established liver injury models, conduct studies on chronic administration, perform detailed mechanistic investigations, and assess the safety profile of this oil for potential clinical translation.
In this study, we established that ASO has notable hepatoprotective effects against CCl4-induced liver damage, as evidenced by a normalization of serum biochemical markers and restoration of hepatic architecture. The observed improvements in oxidative stress markers and histological features highlight the potential utility of ASO as a promising natural therapeutic agent for protecting the liver against oxidative stress-induced damage and subsequent fibrosis. Further studies are required to elucidate the molecular mechanisms underlying ASO-mediated hepatoprotective effects and to evaluate its long-term efficacy using chronic liver injury models.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
THA, AFA, SWA, and FSA performed the experiments, critically revised the manuscript, and approved the final version. MHA, LAA, and AGA contributed to data analysis, revised the manuscript, and approved the final version. HMZ, AAB, and BDA, contributed to results interpretation, critically revised the manuscript, and approved the final version. AAA and HE performed experiments, wrote the original draft, and approved the final manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The research obtained ethical clearance from the Research Ethics Committee of the Faculty of Pharmacy at Taibah University in Madinah, Kingdom of Saudi Arabia (Project Reference Number COPTU-REC-122-20250128). Animal experiments adhere to the 3Rs principle: substitution, reduction, and optimization.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






