1 Department of Chemistry, University of Ibadan, 200284 Ibadan, Oyo State, Nigeria
Abstract
Dioscorea dumetorum (bitter yam) is an underutilized West African species valued for its nutritional and ethnomedicinal importance. While its tubers have been extensively studied, the essential oils of its aerial parts remain largely unexplored. This study investigated the chemical composition, antimicrobial activity, and antioxidant potential of essential oils extracted from the leaves and stems of Dioscorea dumetorum, to provide scientific insight into their bioactive properties and industrial relevance.
Leaves and stems were air-dried, pulverized, and hydro distilled using a Clevenger-type apparatus. Oil yields and physicochemical properties were determined, while chemical constituents were characterized by Gas Chromatography–Mass Spectrometry (GC-MS). Antimicrobial activity was evaluated against selected bacterial (Staphylococcus aureus, Escherichia coli, Bacillus subtilis, Salmonella typhi, Pseudomonas aeruginosa, Klebsiella pneumoniae) and fungal (Candida albicans, Aspergillus niger, Penicillium notatum, Rhizopus stolonifer) pathogens using the agar well diffusion method. Antioxidant potential was assessed by Ferric Reducing Antioxidant Power (FRAP) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assays.
The leaf and stem oils yielded 0.50% and 1.07%, respectively. GC-MS analysis revealed 23 and 20 compounds dominated by 2-pentadecanone (20.64%) and phytol (16.74%) in the leaf oil, and di-n-octyl phthalate (34.43%) and eicosanoic acid esters (15.97%) in the stem oil. Both oils exhibited broad-spectrum antimicrobial effects, with leaf oil showing larger inhibition zones (18–26 mm) than stem oil (14–24 mm). Antioxidant assays demonstrated concentration-dependent activities: leaf oil showed stronger ferric-reducing power, whereas stem oil displayed higher radical-scavenging potential.
These findings demonstrated that Dioscorea dumetorum essential oils possess significant antimicrobial and antioxidant properties with distinct phytochemical profiles, supporting their potential applications as natural sources of bioactive compounds in nutraceutical, pharmaceutical, and cosmetic formulations.
Keywords
- Dioscorea dumetorum
- volatile oils
- gas chromatography–mass spectrometry
- antimicrobial agents
- antioxidants
- plant extracts
- pharmacology
Yams (Dioscorea spp.) are tuber crops of significant nutritional, economic, and cultural importance across tropical regions, particularly in West Africa [1]. Among these, Dioscorea dumetorum (bitter yam), belonging to the family Dioscoreaceae, remains underutilized despite its rich nutritional and medicinal potential [2]. In southeastern Nigeria, it is traditionally consumed for its perceived health benefits and is regarded as a dietary option for diabetic patients. However, its limited use is largely attributed to its bitter taste, postharvest hardening, and spoilage tendencies [3]. Studies have suggested that processing methods such as soaking and extended cooking can improve its nutritional and phytochemical qualities [4, 5].
In recent years, interest in Dioscorea dumetorum has expanded beyond its nutritional composition to its phytochemical constituents and potential therapeutic applications. Plants within the Dioscorea genus are known to contain diverse bioactive metabolites, including alkaloids, flavonoids, saponins, and essential oils, which exhibit a wide range of pharmacological activities. Essential oils, in particular, are complex mixtures of volatile compounds synthesized by aromatic plants and have been utilized for centuries in traditional medicine, perfumery, and food preservation [6]. Their biological properties such as antimicrobial, antioxidant, anti-inflammatory, and anticancer activities make them promising candidates for natural therapeutic and industrial applications [7, 8].
The extraction and characterization of essential oils depend on the plant source and the extraction technique employed, including hydrodistillation, steam distillation, and supercritical CO2 extraction, among others [9, 10]. These oils are widely applied in the pharmaceutical, cosmetic, and agro-food sectors due to their lipophilicity, low toxicity, and biological efficacy [11]. In medicine, essential oils have been investigated for their therapeutic potential against infectious and inflammatory diseases [12], while in the food industry, they serve as natural preservatives and flavoring agents with antimicrobial and antioxidant functions [13, 14].
Hydro-distillation was selected for its efficiency in isolating volatile compounds without chemical degradation. Compared to solvent extraction and Soxhlet methods, hydro-distillation is environmentally friendly, requires minimal solvents, and better preserves thermolabile bioactives such as monoterpenes and sesquiterpenes. This technique also facilitates direct comparison with other medicinal plant essential oils extracted under similar conditions [12, 14].
Despite the established pharmacological relevance of Dioscorea species, most research has focused on the tubers of Dioscorea dumetorum, with little attention to its aerial parts (stems and leaves) which are often discarded as waste. Yet, these parts may harbor unique volatile phytochemicals with potential biological and industrial applications [15, 16]. Investigating their essential oil composition not only promotes resource optimization but also contributes to the broader understanding of the species’ phytochemical diversity and pharmacological potential.
Given the growing demand for natural alternatives to synthetic additives and therapeutics, this study aims to extract, characterize, and evaluate the bioactivities of the essential oil from Dioscorea dumetorum. The research specifically focuses on elucidating the chemical constituents using Gas Chromatography–Mass Spectrometry (GC-MS) and assessing the oil’s antioxidant and antimicrobial properties through standard in vitro assays. The findings will provide baseline data for future pharmacological and industrial applications of Dioscorea dumetorum, highlighting its potential as a source of natural bioactive compounds [12].
This is the first comprehensive study investigating both the chemical composition and the comparative antimicrobial and antioxidant potentials of Dioscorea dumetorum leaf and stem essential oils extracted using hydrodistillation. Unlike previous studies focused mainly on tubers, this work highlights the underexplored aerial parts of the plant as a potential source of bioactive compounds with therapeutic relevance [16].
Fresh leaves and stems of Dioscorea dumetorum (bitter yam) were collected from Fiditi, Oyo State, Nigeria, and authenticated at the Department of Botany, University of Ibadan (Voucher No.: UIH–23643). The materials were air-dried under shade at room temperature for eight days to prevent loss of volatile components, pulverized, and stored in airtight containers until extraction.
Essential oils were extracted from the powdered leaves (465 g) and stems (135 g) by hydrodistillation using a Clevenger-type apparatus. Each sample was immersed in distilled water and distilled for 3 h. The condensate was collected, and the oil layer separated, dried over anhydrous sodium sulfate, and stored in amber vials at 4 °C until analysis [6, 7].
The essential oil was visually observed for its color, consistency, and odor. The yield was calculated as a percentage using the formula:
Chemical profiling of the oils was performed using a Shimadzu GCMS-QP2010
equipped with an AOC-20i auto-sampler. Samples were diluted (1:100, v/v) in
n-hexane with n-decane as an internal standard. Separation was achieved on a
capillary column using helium as the carrier gas at 0.8 mL/min. The oven
temperature was programmed from 60 °C (1 min hold) to 240 °C at
13 °C/min, with a final hold of 1 min. The MS was operated in electron
impact (EI) mode (70 eV), scanning m/z 35–500. Compounds were identified by
comparison with NIST/Wiley libraries and confirmed via retention indices
(
The antimicrobial activity of the essential oils was tested against Staphylococcus aureus, Escherichia coli, Bacillus subtilis, Salmonella typhi, pseudomonas aeruginosa, Klebsiella pneumoniae, and fungal strains (Candida albicans, Aspergillus niger, penicillium notatum, and Rhizopus stolonifer). The agar well diffusion method was used on Nutrient Agar (bacteria) and Sabouraud Dextrose Agar (fungi). Gentamicin and tioconazole served as positive controls, while dimethyl sulfoxide (DMSO) served as the negative control. Zones of inhibition were measured after incubation (37 °C for 24 h for bacteria; 26–28 °C for 48 h for fungi). Minimum inhibitory concentration (MIC) values were determined by broth microdilution in 96-well plates [7, 8].
All antimicrobial assays were carried out in triplicate (n = 3). Essential oil concentrations of 250, 500, 1000, 2000, and 4000 µg/mL were tested using the agar well diffusion method. Gentamicin (for bacteria) and tioconazole (for fungi) served as positive controls, while dimethyl sulfoxide (DMSO) was used as the negative control to exclude solvent effects.
Antioxidant capacity was evaluated using 2, 2, diphenylpicrylhydrazyl (DPPH)
radical scavenging and Ferric Reducing Antioxidant Power (FRAP) assays. All
antioxidant assays were performed in triplicate (n = 3), and results were
expressed as mean
DPPH Assay: Essential oil samples (25–400 µg/mL) were reacted with 0.4 mM DPPH in methanol, incubated in the dark for 30 min, and absorbance read at 517 nm. The radical scavenging activity was expressed as percentage inhibition of the DPPH radical, calculated relative to the control (DPPH solution without extract) using the equation:
Where AC represents the absorbance of the control, AS represents the absorbance of the test sample. A higher percentage inhibition indicated a stronger radical scavenging capacity.
FRAP Assay: Samples were mixed with phosphate buffer, potassium
ferricyanide, and trichloroacetic acid, followed by ferric chloride addition.
Absorbance was measured at 700 nm, with increased absorbance indicating higher
reducing power. For the assay, different concentrations of the extract were mixed
with 2.5 mL of phosphate buffer (200 mM, pH 6.6) and 2.5 mL of 1% potassium
ferricyanide. The mixtures were incubated at 50 °C for 20 minutes, after
which 2.5 mL of 10% trichloroacetic acid was added to terminate the reaction.
The mixtures were then centrifuged at 650
The essential oils obtained from the leaves and stems were pale yellow with aromatic odor (Table 1).
| Plant part | Weight of dried sample (g) | Volume of oil (mL) | Oil yield (%) | Color | Odor |
| Leaves | 465 | 2.34 | 0.50 | Pale yellow | Aromatic |
| Stems | 135 | 1.45 | 1.07 | Pale yellow | Aromatic |
The physical characteristics and modest yields of Dioscorea dumetorum essential oils suggest potential applications in food, medicinal, and industrial sectors. The pale-yellow coloration, often linked to carotenoids and flavonoids, reflects antioxidant potential, while the aromatic odor implies the presence of volatile terpenoids with known antimicrobial and therapeutic properties [17]. Similar to other Dioscorea species, these bioactive constituents may enhance the functional value of the oils as natural preservatives or flavoring agents in food systems [18].
GC-MS profiling revealed 24 and 20 compounds in the leaf and stem oils, respectively. The leaf oil was dominated by phytol (16.74%), 2-pentadecanone (20.64%), and spiro ketone derivatives (12.77%), compounds known for antioxidant, antimicrobial, and aromatic properties. The stem oil contained di-n-octyl phthalate (34.43%), eicosanoic acid esters (11.87% and 4.10%), and 13-octadecenal (9.32%) as major constituents, reflecting a fatty acid-rich composition. Both oils shared acetophenone and phytol, indicating overlapping pharmacological potential. The chemical variation suggests the leaf oil may possess stronger antioxidant activity due to its higher proportion of oxygenated terpenes, while the stem oil’s composition favors antimicrobial and emollient applications.
The GC-MS analysis of the essential oil revealed 24 distinct compounds, representing 100% of the total oil composition. The identified compounds, their retention times (RT), percentages composition are presented in Table 2. Fig. 1 shows the GC-MS Analysis of Leaves.
| S/N | Retention time | %Composition | Name |
| 1 | 9.745 | 1.07 | 1-Octen-3-ol |
| 2 | 9.889 | 2.30 | 5-Hepten-2-one |
| 3 | 11.543 | 4.12 | Acetophenone |
| 4 | 13.673 | 1.05 | 1-Cyclohexene |
| 5 | 14.127 | 0.83 | Cyclohexanone |
| 6 | 14.600 | 0.61 | 1,5-Heptadiene |
| 7 | 15.538 | 1.44 | 2-Undecanone |
| 8 | 16.163 | 10.18 | 5,9-Undecadien-2-one |
| 9 | 16.860 | 4.72 | trans-.beta.-Ionone |
| 10 | 19.063 | 2.22 | Octadecanal |
| 11 | 19.147 | 1.07 | 1-Dodecanol |
| 12 | 19.241 | 1.81 | Spiro [4.5] dec-8-en-7-ol |
| 13 | 19.650 | 1.56 | Pentadecane |
| 14 | 19.735 | 12.77 | Spiro [4.5] dec-6-en-8-one |
| 15 | 20.083 | 0.96 | Tetracontane |
| 16 | 20.321 | 20.64 | 2-Pentadecanone |
| 17 | 20.689 | 1.15 | 2-methyltetracosane |
| 18 | 21.095 | 1.23 | Docosane |
| 19 | 21.232 | 8.91 | 5,9,13-Pentadecatrien-2-one |
| 20 | 22.902 | 1.09 | Heptadecane |
| 21 | 23.005 | 1.34 | 2-methyltetracosane |
| 22 | 23.358 | 16.74 | Phytol |
| 23 | 25.565 | 1.19 | Octacosane |
| 99.00 |
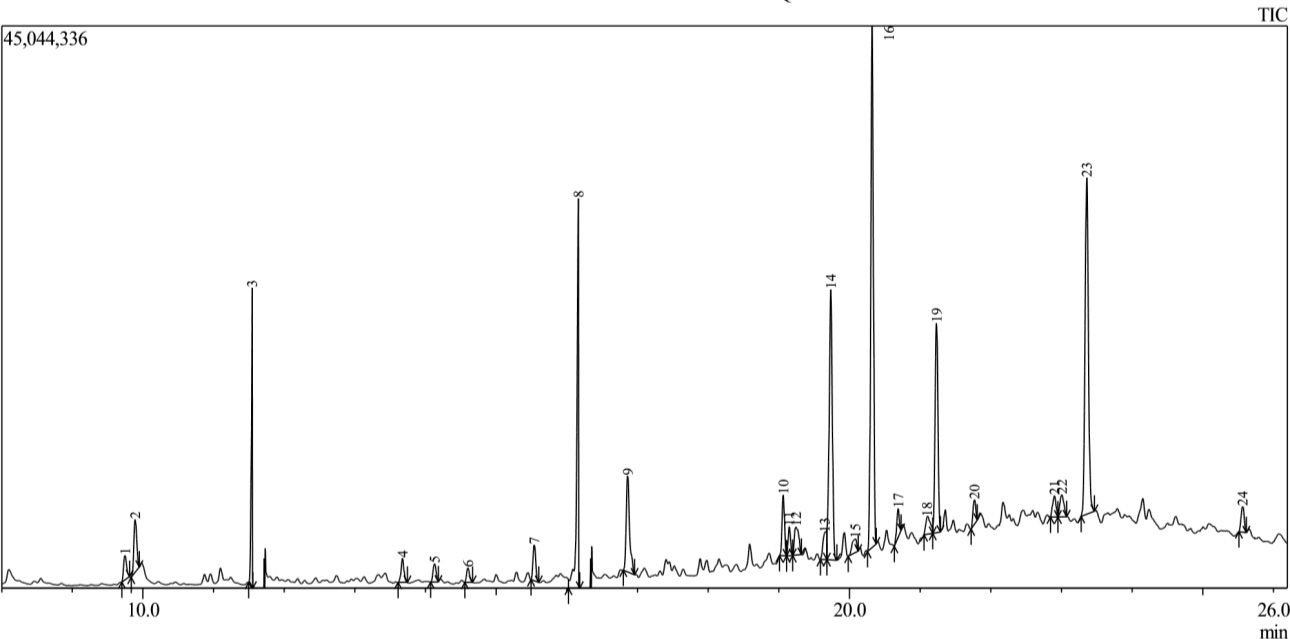 Fig. 1.
Fig. 1.
Chromatographic profile of Dioscorea dumetorum leaf extract. The chromatogram shows distinct peaks corresponding to the various phytochemical compounds present in the leaf extract, each peak representing a specific retention time and relative concentration.
The GC-MS analysis of the essential oil revealed 20 distinct compounds, representing 100% of the total oil composition. The identified compounds, their retention times (RT), peak areas relative percentages are presented in Table 3. Fig. 2 shows the GC-MS Analysis of Stem.
| S/N | Retention time | %Composition | Name |
| 1 | 11.635 | 6.60 | Acetophenone |
| 2 | 14.275 | 7.83 | 3-hydroxypropyl ester |
| 3 | 16.192 | 0.54 | cis-Z-.alpha.-Bisabolene epoxide |
| 4 | 16.770 | 9.32 | 13-Octadecenal |
| 5 | 17.982 | 0.66 | Tridecanal |
| 6 | 18.815 | 1.17 | Heptasiloxane |
| 7 | 18.960 | 34.43 | Di-n-octyl phthalate |
| 8 | 19.054 | 3.84 | Pentadecanal |
| 9 | 19.660 | 1.85 | 1-Decanol, 2-octyl- |
| 10 | 20.125 | 1.97 | Octadecanoic acid |
| 11 | 20.305 | 3.55 | 2-Pentadecanone |
| 12 | 21.043 | 1.73 | 9,17-Octadecadienal, (Z)- |
| 13 | 21.489 | 11.87 | 2,3-bis(acetyloxy)propyl ester |
| 14 | 21.969 | 0.48 | 5-methylhex-2-yl butyl ester |
| 15 | 22.170 | 0.50 | trans-2-undecenoic acid |
| 16 | 22.408 | 2.18 | 3-n-Butylthiophene-1,1-dioxide |
| 17 | 22.791 | 4.10 | Eicosanoic acid |
| 18 | 23.050 | 3.79 | Hexadecanoic acid |
| 19 | 23.339 | 2.79 | Phytol |
| 20 | 26.070 | 0.82 | 1-(2-aminoethoxy)hydro |
| 100.00 |
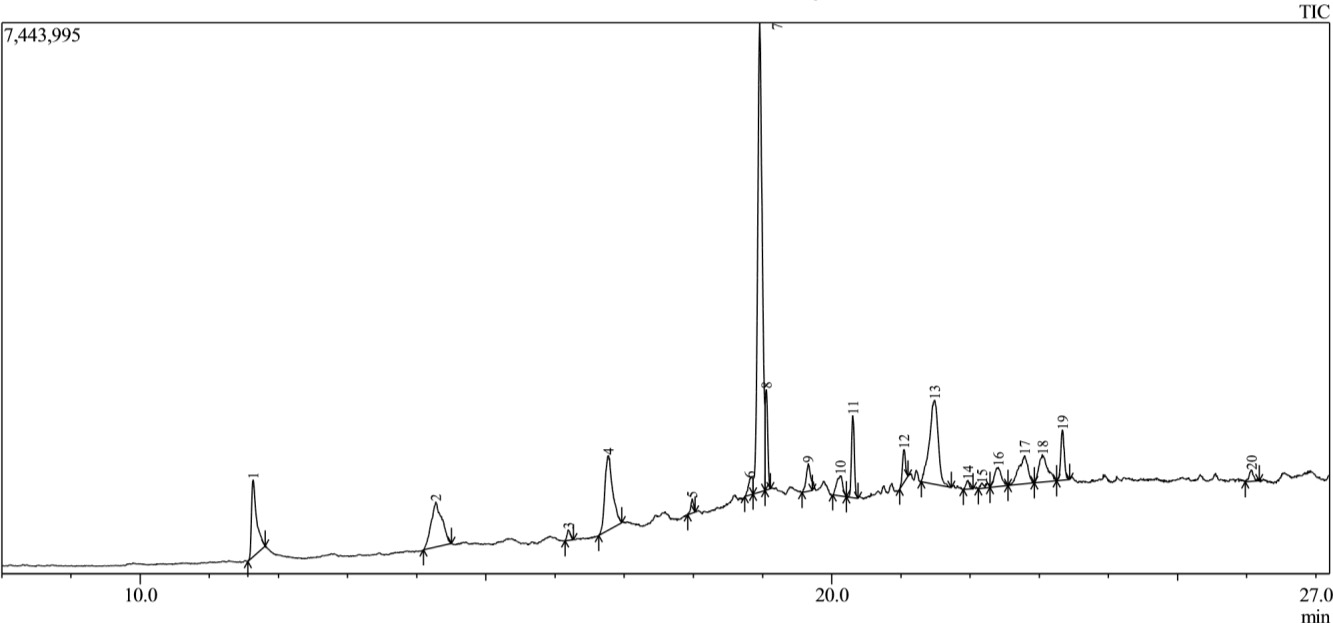 Fig. 2.
Fig. 2.
Chromatographic profile of Dioscorea dumetorum stem extract. The chromatogram displays distinct peaks corresponding to individual phytochemical compounds detected in the stem extract, each peak representing a specific retention time and relative abundance.
The leaf and stem essential oils exhibited broad-spectrum antimicrobial activity
against six bacterial and four fungal strains. The leaf oil produced larger
inhibition zones (18–26 mm) than the stem oil (14–24 mm). Statistical analysis
showed that the antimicrobial activity of the leaf oil was significantly higher
than that of the stem oil at the highest concentration (p
| S/N | S.aur | E.col | B.sub | P.aer | S.typ | K.pne | C.alb | A.nig | P.not | R.sto |
| 1. | 26 | 24 | 26 | 25 | 24 | 24 | 20 | 20 | 18 | 20 |
| 2. | 24 | 21 | 22 | 22 | 20 | 21 | 18 | 18 | 16 | 18 |
| 3. | 21 | 18 | 20 | 19 | 18 | 18 | 16 | 16 | 14 | 16 |
| 4. | 18 | 10 | 17 | 17 | 16 | 16 | 12 | 14 | 12 | 12 |
| 5. | 14 | 12 | 13 | 14 | 12 | 13 | 10 | 10 | 10 | 10 |
| –ve | - | - | - | - | - | - | - | - | - | - |
| +ve | 40 | 40 | 38 | 38 | 40 | 38 | 30 | 28 | 28 | 28 |
S.aur, Staphylococcus aureus; E.col, Escherichia coli; B.sub, Bacillus subtilis; P.aer, Pseudomonas aeruginosa; S.typ, Salmonella typhi; K.pne, Klebsiella pneumoniae; and fungal strains C.alb, Candida albicans; A.nig, Aspergillus niger; p.not, Penicillium notatum; and R.sto, Rhizopus stolonifer; –ve, negative control; +v, positive control (Gentamicin and tioconazole served as positive controls, while DMSO served as the negative control); DMSO, dimethyl sulfoxide.
| S/N | B.cer | E.col | B.sub | P.aer | S.typ | K.pne | C.alb | A.nig | P.not | R.sto |
| 1. | 22 | 24 | 23 | 24 | 24 | 23 | 20 | 18 | 14 | 16 |
| 2. | 18 | 21 | 20 | 21 | 21 | 20 | 18 | 16 | 12 | 14 |
| 3. | 16 | 18 | 17 | 18 | 18 | 16 | 16 | 12 | 10 | 10 |
| 4. | 14 | 10 | 14 | 15 | 16 | 14 | 12 | 10 | - | - |
| 5. | 10 | 12 | 11 | 12 | 13 | 12 | 10 | 10 | - | - |
| –ve | - | - | - | - | - | - | - | - | - | - |
| +ve | 40 | 40 | 38 | 38 | 40 | 38 | 30 | 28 | 28 | 28 |
S.aur, Staphylococcus aureus; E.col, Escherichia coli; B.sub, Bacillus subtilis; P.aer, Pseudomonas aeruginosa; S.typ, Salmonella Typhi; K.pne, Klebsiella pneumoniae; and fungal strains C.alb, Candida albicans; A.nig, Aspergillus niger; p.not, Penicillium notatum; and R.sto, Rhizopus stolonifer; –ve, negative control; +v, positive control (Gentamicin and tioconazole served as positive controls, while DMSO served as the negative control).
| Organisms | S.aur | E.col | B.sub | P.aer | S.typ | K.pne | C.alb | A.nig | P.not | R.sto |
| Leaf (mm) | 26 | 24 | 26 | 25 | 24 | 24 | 20 | 20 | 18 | 20 |
| Stem (mm) | 22 | 24 | 23 | 24 | 24 | 23 | 20 | 18 | 14 | 16 |
Table 6 shows a comparison of activity between the leaf and stem inhibition zones at the highest concentration across all ten microorganisms. These provide a straightforward comparison, emphasizing the greater activity of the leaf extracts relative to the stems. Fig. 3 also shows the comparison using the bar chart.
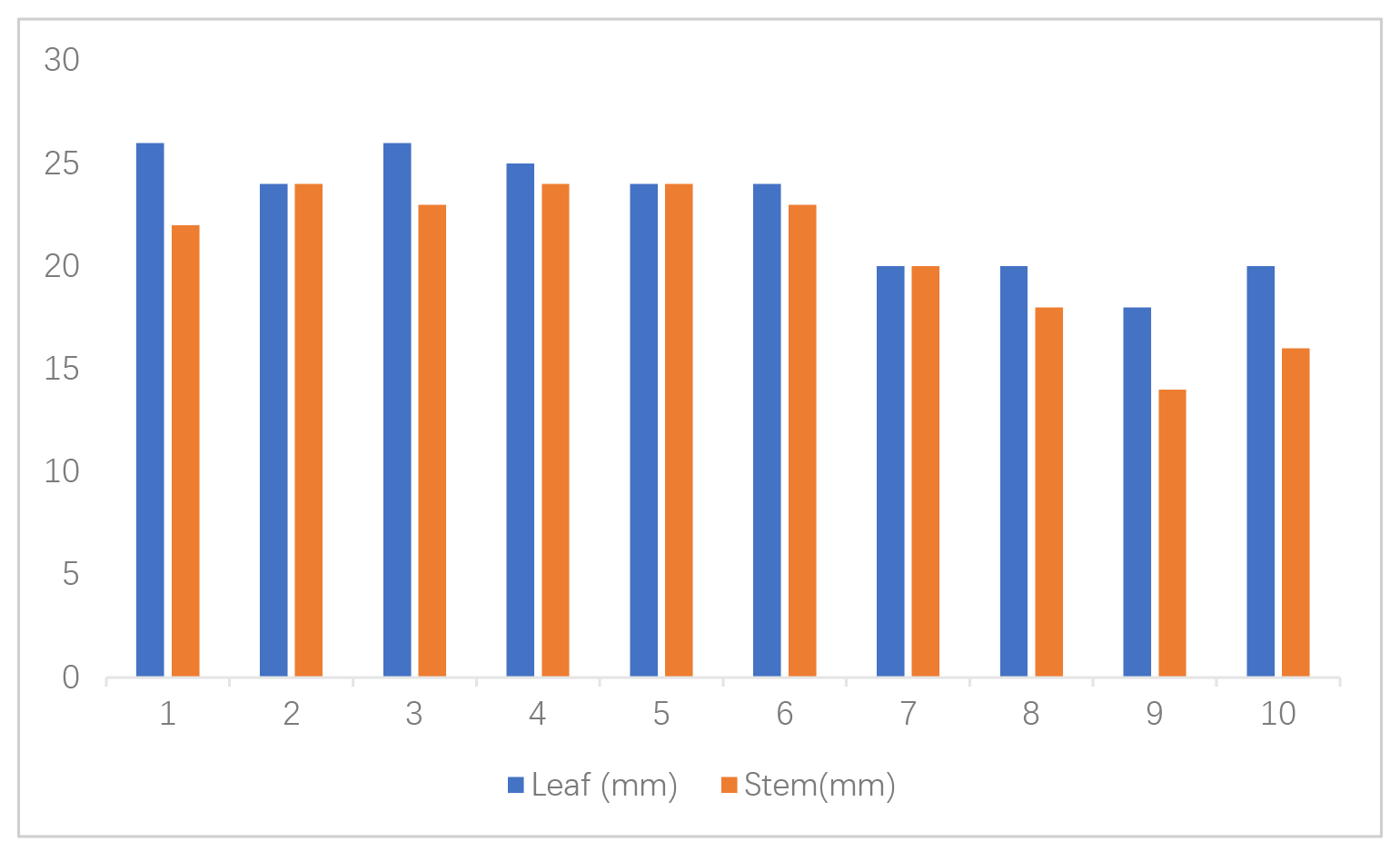 Fig. 3.
Fig. 3.
Comparison of leaf and stem essential oils against ten
microorganisms. Data represent mean
The values represent mean inhibition zones (mm) from three independent
experiments (n = 3), and differences between leaf and stem oils were
statistically significant at p
Both extracts demonstrated concentration-dependent ferric reducing power,
confirming their electron-donating and radical-quenching abilities. The leaf
extract showed higher FRAP values than the stem extract across all concentrations
tested. Statistical analysis confirmed that the FRAP values of the leaf extract
were significantly higher than those of the stem extract (p
| Concentration (µg/mL) | 1 | 2 | 3 | Mean | SD | Mean |
| 4000 | 18.009 | 18.040 | 18.009 | 18.019 | 0.018 | 18.019 |
| 2000 | 15.852 | 15.884 | 15.884 | 15.873 | 0.018 | 15.873 |
| 1000 | 14.478 | 14.478 | 14.478 | 14.478 | 0.000 | 14.478 |
| 500 | 11.978 | 12.040 | 12.035 | 12.018 | 0.034 | 12.018 |
| 250 | 10.415 | 10.665 | 10.665 | 10.582 | 0.144 | 10.582 |
| Concentration (µg/mL) | 1 | 2 | 3 | Mean | SD | Mean |
| 4000 | 14.103 | 14.103 | 14.165 | 14.124 | 0.036 | 14.124 |
| 2000 | 12.072 | 12.197 | 12.290 | 12.186 | 0.109 | 12.186 |
| 1000 | 10.822 | 10.822 | 10.790 | 10.811 | 0.018 | 10.811 |
| 500 | 10.103 | 9.478 | 9.478 | 9.686 | 0.361 | 9.686 |
| 250 | 8.290 | 8.290 | 8.290 | 8.290 | 0.000 | 8.290 |
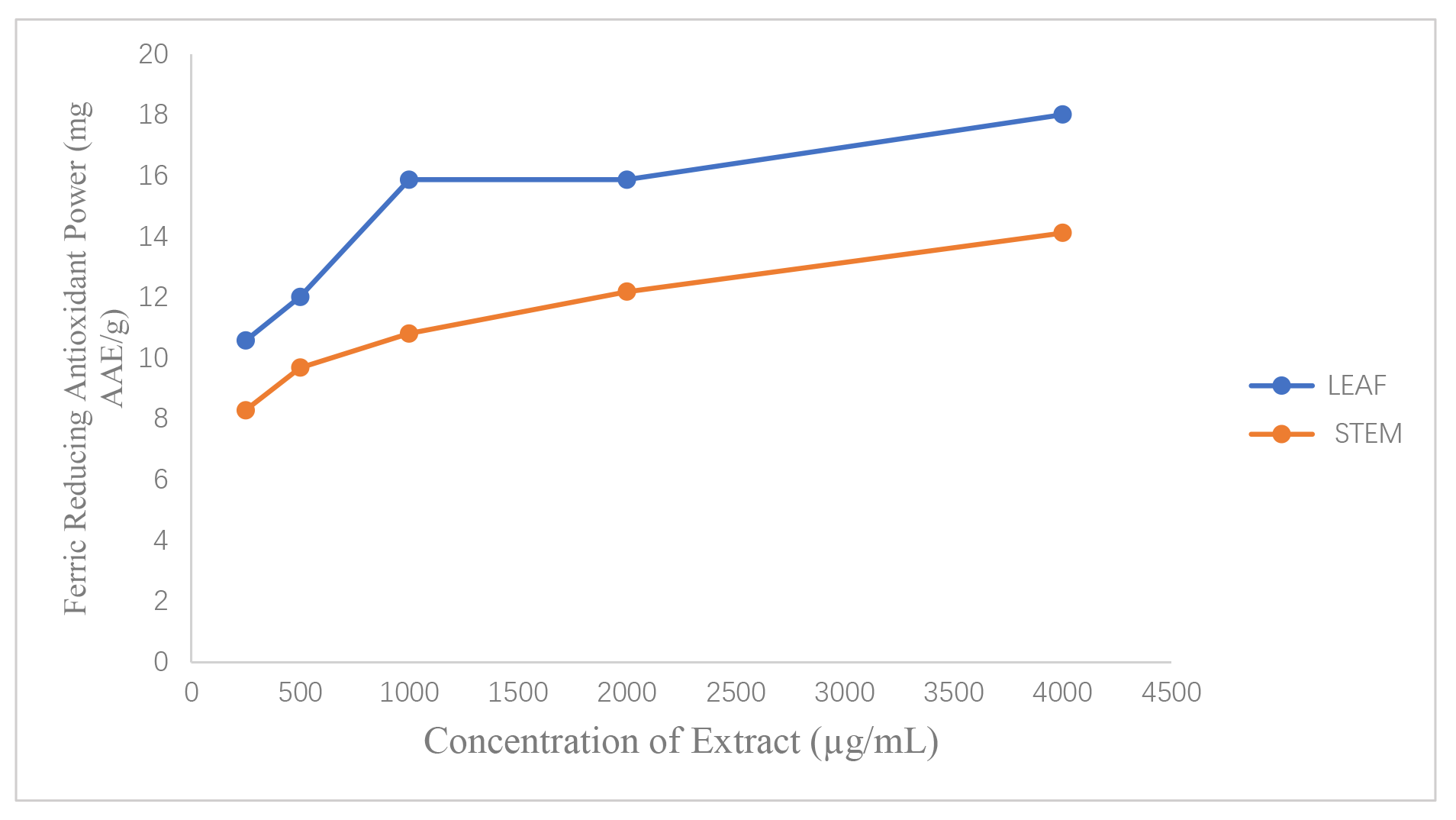 Fig. 4.
Fig. 4.
Comparative bar charts showing ferric reducing antioxidant power (FRAP) activity of leaf and stem extracts. X-axis: Concentration of Extract µg/mL. Y-axis: Ferric Reducing Antioxidant Power (mg AAE/g).
The DPPH assay confirmed that both extracts possess free radical scavenging activity in a concentration-dependent manner. Fig. 5 shows the leaf extract activity. Fig. 6 shows the stem extract activity. The stem extract exhibited a lower IC50 value (224.11 µg/mL) than the leaf extract (1288.52 µg/mL), suggesting stronger scavenging efficiency, though both were less potent than ascorbic acid (IC50 = 156.15 µg/mL). Fig. 7 shows the standard antioxidant activity. The antioxidant properties can be attributed to secondary metabolites such as terpenoids and phenolic compounds identified in the GC-MS analysis. These findings reinforce the potential of Dioscorea dumetorum essential oils particularly from the stem as moderate natural antioxidants suitable for applications in oxidative stress management and product stabilization. The DPPH radical scavenging activities are presented in Table 9 (leaf oil) and Table 10 (stem oil), while the standard is shown in Table 11. A comparison of IC50 values is provided in Table 12. Fig. 8 shows the experimental workflow schematic.
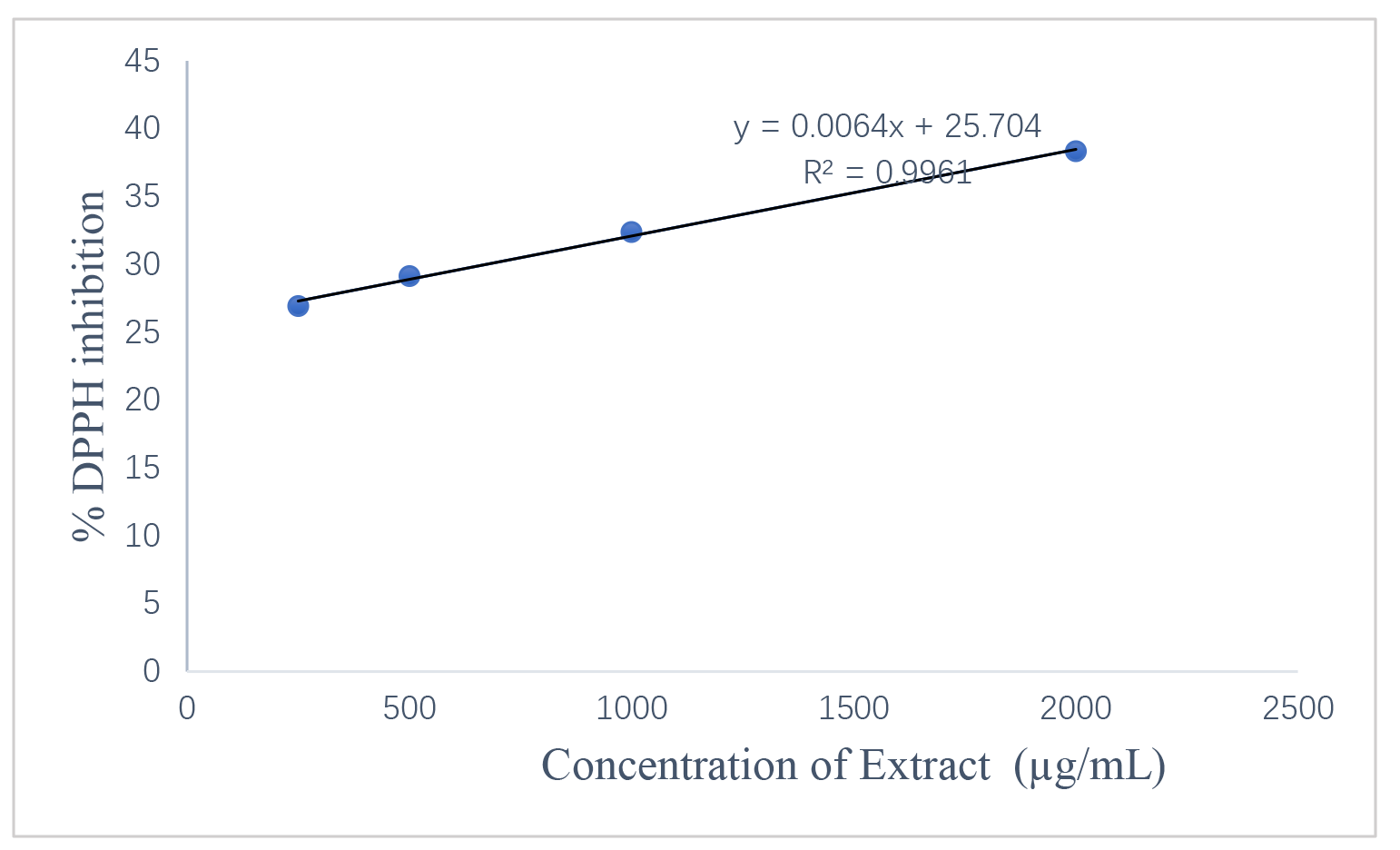 Fig. 5.
Fig. 5.
DPPH radical scavenging activity of Dioscorea
dumetorum leaf extract. Showing % DPPH inhibition (Y-axis) against
Concentration of Extract (µg/mL) (X-axis). Data represent mean
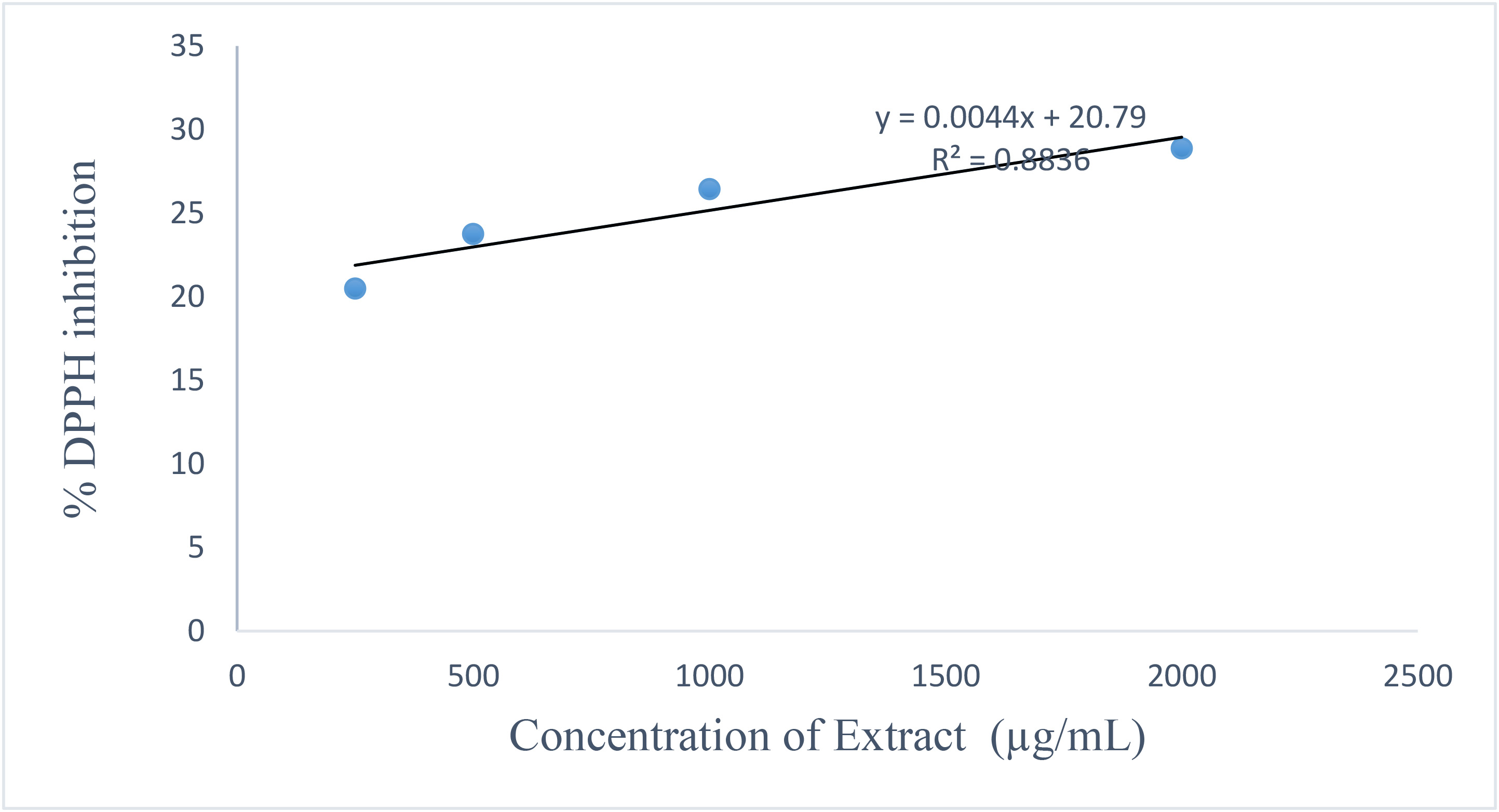 Fig. 6.
Fig. 6.
DPPH radical scavenging activity of Dioscorea
dumetorum stem essential oil. Showing % DPPH inhibition (Y-axis) against
Concentration of Extract (µg/mL) (X-axis). Data represent mean
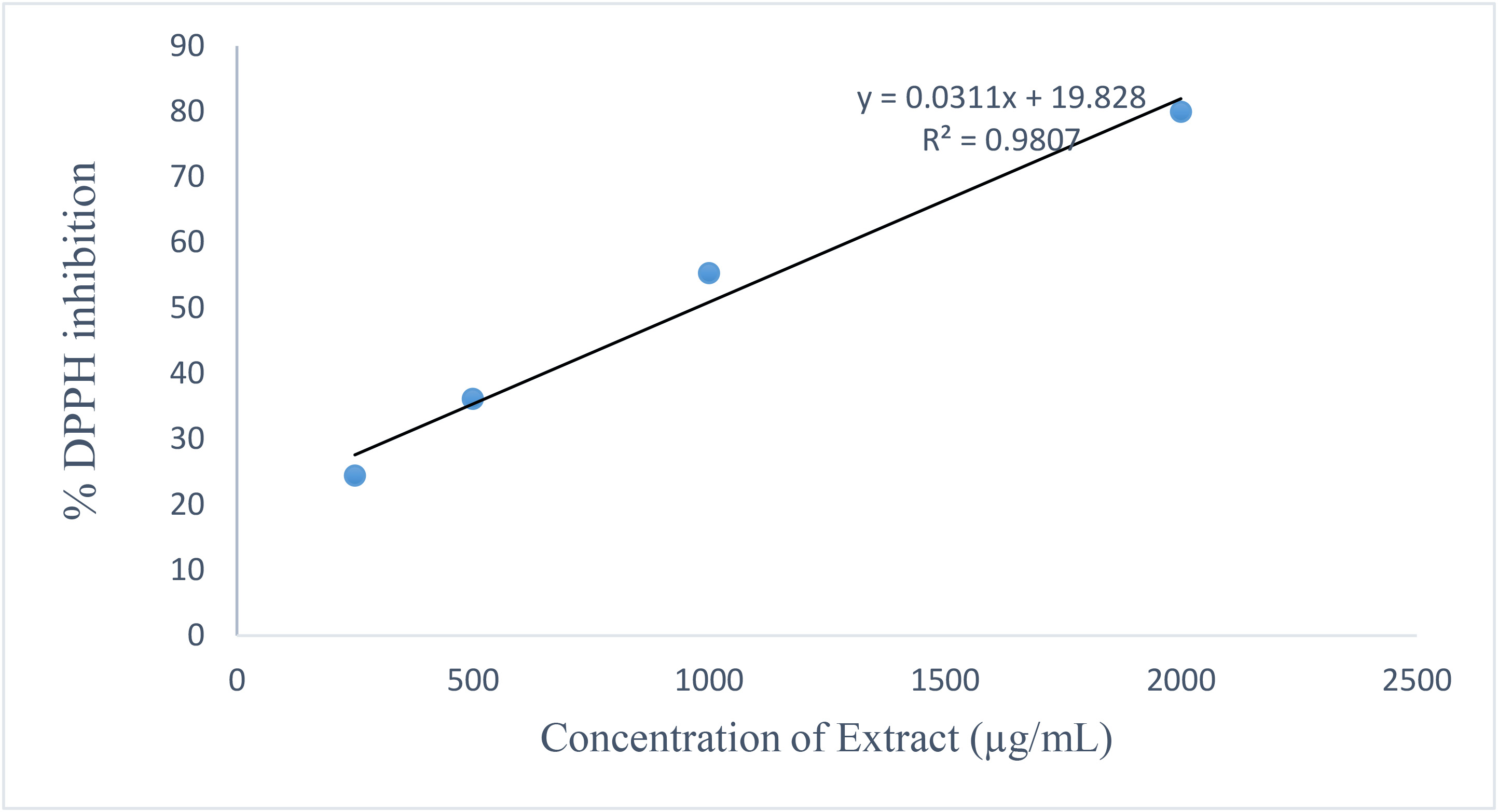 Fig. 7.
Fig. 7.
DPPH radical scavenging activity of the ascorbic acid standard.
Showing % DPPH inhibition (Y-axis) against Concentration of Extract
(µg/mL) (X-axis). Data represent mean
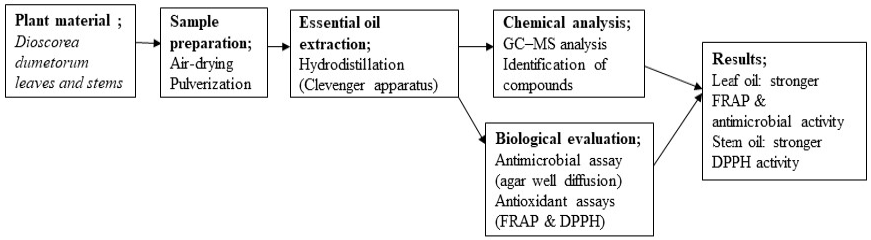 Fig. 8.
Fig. 8.
Schematic representation of the experimental workflow for the extraction, chemical characterization, and biological evaluation of Dioscorea dumetorum leaf and stem essential oils. The diagram summarizes sample preparation, hydrodistillation extraction, GC-MS analysis, antimicrobial screening, and antioxidant assays (FRAP and DPPH).
| Conc (µg/mL) | 1 | 2 | 3 | Mean | SD | Mean |
| 4000 | 41.39 | 41.23 | 41.15 | 41.26 | 0.12 | 41.26 |
| 2000 | 38.43 | 38.43 | 38.11 | 38.32 | 0.18 | 38.32 |
| 1000 | 32.27 | 32.50 | 32.35 | 32.37 | 0.12 | 32.37 |
| 500 | 29.31 | 29.15 | 28.92 | 29.13 | 0.20 | 29.13 |
| 250 | 26.81 | 27.00 | 27.00 | 26.94 | 0.11 | 26.94 |
DPPH, 2,2-diphenyl-1-picrylhydrazyl.
| Conc (µg/mL) | 1 | 2 | 3 | Mean | SD | Mean |
| 4000 | 30.32 | 30.48 | 30.71 | 30.50 | 0.20 | 30.50 |
| 2000 | 28.84 | 28.84 | 29.00 | 28.89 | 0.09 | 28.89 |
| 1000 | 26.35 | 26.58 | 26.35 | 26.43 | 0.13 | 26.43 |
| 500 | 23.77 | 23.77 | 23.70 | 23.75 | 0.04 | 23.75 |
| 250 | 20.58 | 20.34 | 20.58 | 20.50 | 0.14 | 20.50 |
| Conc (µg/mL) | 1 | 2 | 3 | Mean | SD | Mean |
| 4000 | 96.40 | 96.41 | 96.38 | 96.40 | 0.02 | 96.40 |
| 2000 | 79.86 | 80.02 | 79.96 | 79.95 | 0.08 | 79.86 |
| 1000 | 55.26 | 55.30 | 55.34 | 55.30 | 0.04 | 55.26 |
| 500 | 36.10 | 36.14 | 36.11 | 36.12 | 0.02 | 36.10 |
| 250 | 24.38 | 24.44 | 24.43 | 24.42 | 0.03 | 24.38 |
| Sample | IC50 (µg/mL) | Antioxidant potency (Lower = Stronger) |
| Standard Ascorbic Acid | 156.15 | Very strong |
| Dioscorea dumetorum Stem Essential Oil | 224.11 | Moderate |
| Dioscorea dumetorum Leaf Essential Oil | 1288.52 | Weak |
The GC-MS analysis of Dioscorea dumetorum essential oils showed distinct compositional differences between the leaf and stem oils, reflecting organ-specific biosynthetic activity and environmental influence on metabolite accumulation [19, 20].
The leaf oil contained 23 identified compounds, mainly oxygenated terpenoids and ketones, while the stem oil had 20 compounds dominated by fatty acid esters and aldehydes. 2-pentadecanone (20.64%) and phytol (16.74%) were the predominant constituents, in the leaf oil, followed by spiro [4.5] dec-6-en-8-one (12.77%) and 5,9-undecadien-2-one (10.18%). These oxygenated molecules are known for their antimicrobial, antioxidant, and aromatic properties [21, 22].
Phytol, a chlorophyll-derived diterpene, has been reported to exhibit strong antioxidant, anti-inflammatory, and antimicrobial effects [23, 24], while methyl ketones such as 2-pentadecanone enhance the oxidative stability and antimicrobial performance of essential oils [25, 26]. The presence of spiro ketones and ionone-type compounds further supports the high antioxidant potential of the leaf oil [26, 27].
Conversely, the stem oil was dominated by di-n-octyl phthalate (34.43%), 13-octadecenal (9.32%), and eicosanoic acid derivatives (11.87% and 4.10%), suggesting a fatty acid–rich fraction with emollient and preservative properties [21, 28]. Although phthalates are sometimes associated with laboratory contamination, precautions were taken during sample preparation, including the use of clean glassware and analytical-grade solvents. Therefore, its presence is reported with caution, and further studies are recommended to confirm its biological origin.
Both oils contained acetophenone and phytol, indicating shared pharmacological potential [23]. However, the higher oxygenated terpene content in the leaf oil likely accounts for its stronger ferric-reducing antioxidant power, whereas the fatty acid–based composition of the stem oil supports its superior radical-scavenging and antimicrobial activities [26]. Overall, the GC-MS results confirm that Dioscorea dumetorum essential oils are rich in bioactive compounds with promising antioxidant and antimicrobial potential, supporting their value for nutraceutical and pharmaceutical applications.
The extraction process significantly impacted both the yield and chemical
profile of the essential oils. Hydro-distillation tends to recover lighter
volatile molecules, such as
The antimicrobial evaluation of Dioscorea dumetorum revealed that both the leaf and stem essential oils exhibited inhibitory activity against a wide range of microorganisms, including both bacteria and fungi. All tests were done in triplicate (n = 3). Gentamicin and tioconazole were used as positive controls; DMSO as negative control. Extract concentrations ranged from 250–4000 µg/mL. However, the results consistently showed that the leaf extracts were more potent than the stem extracts, producing larger zones of inhibition across almost all test organisms. This suggests that the leaves contain higher concentrations of active phytochemicals responsible for antimicrobial effects [24]. Both extracts demonstrated a clear dose-dependent pattern, where antimicrobial activity decreased progressively with dilution, confirming that higher concentrations of the bioactive compounds enhance effectiveness [25]. Importantly, the extracts displayed broad-spectrum activity by inhibiting both Gram-positive and Gram-negative bacteria (e.g., Bacillus cereus, Escherichia coli, Salmonella typhi, and Pseudomonas aeruginosa) as well as pathogenic fungi such as Candida albicans and Aspergillus niger [21, 25]. Despite this broad antimicrobial profile, the potency of the crude extracts was lower than that of the standard drugs used as positive controls (gentamycin for bacteria and tioconazole for fungi). For instance, while the extracts produced inhibition zones in the range of 18–26 mm at the highest concentration, the standard antibiotics yielded much larger zones (38–40 mm for bacteria and 28–30 mm for fungi). Nevertheless, the significant activity of the oils, especially from the leaves, highlights their potential as sources of natural antimicrobial agents. These findings align with previous reports linking antimicrobial efficacy in Dioscorea species and other medicinal plants to terpenoids, phenolic compounds, and ketones identified in their essential oils [21, 23, 26, 29].
The antibacterial efficacy of the essential oils can be attributed to the
disruption of microbial membranes and increased permeability, leading to leakage
of cellular components and inhibition of enzymatic activity. This activity is
primarily linked to oxygenated monoterpenes such as 1,8-cineole and
Both the leaf and stem extracts of Dioscorea dumetorum demonstrated ferric reducing antioxidant power, confirming the presence of bioactive compounds capable of donating electrons to reduce Fe3+ to Fe2+ and thereby neutralize free radicals [27]. The antioxidant activity increased proportionally with concentration across all tested concentrations, reflecting a clear concentration–response relationship. However, the leaf extract consistently exhibited higher FRAP values than the stem extract at every concentration tested. This suggests that the leaves contain a greater abundance of antioxidant phytochemicals, likely including phenolics and flavonoids, which are well-established contributors to redox activity [24, 27]. In contrast, the stem exhibited moderate but lower reducing capacity, which may be attributed to its primarily structural role and reduced metabolic activity. These findings are consistent with previous studies on other medicinal plants, where leaves typically display stronger antioxidant activity due to higher levels of photosynthetically derived secondary metabolites [26, 27]. The results therefore highlight Dioscorea dumetorum leaves as a richer source of natural antioxidants with potential applications in functional foods, nutraceuticals, and pharmaceutical formulations targeted at managing oxidative stress [24].
The results clearly show that both the leaf and stem essential oils of Dioscorea dumetorum possess free radical–scavenging capacity as measured by the DPPH assay [26]. The activity was concentration dependent, with higher concentrations producing stronger radical-scavenging effects. Between the two plant parts, the stem extract exhibited greater potency (IC50 = 224.11 µg/mL) than the leaf extract (IC50 = 1288.52 µg/mL), despite the leaf showing slightly higher percentage inhibition at individual concentrations (e.g., 41.26% at 4000 µg/mL for leaf vs. 30.50% for stem). This indicates that the stem oil requires much lower concentrations to reach 50% inhibition, making it more effective overall. When compared to the ascorbic acid standard (IC50 = 156.15 µg/mL), both extracts demonstrated significantly lower antioxidant activity, confirming that while Dioscorea dumetorum has notable free radical–scavenging potential, it is less potent than pure ascorbic acid [31]. These findings are consistent with reports on other medicinal plants, where crude extracts typically show moderate antioxidant capacity compared to pure standards but still contribute significantly to overall antioxidant potential [27, 31]. The observed activity likely reflects the presence of secondary metabolites such as phenolic compounds and flavonoids, which are known for their radical-scavenging properties [24, 26, 27]. Thus, Dioscorea dumetorum extracts, particularly the stem, may serve as a source of natural antioxidants with potential applications in nutraceuticals, functional foods, and phytomedicine aimed at combating oxidative stress [24, 27].
The observed differences between FRAP and DPPH results highlight the assay-dependent nature of antioxidant evaluation. While FRAP measures electron-donating ability, DPPH assesses free radical scavenging efficiency. Therefore, the higher FRAP activity of the leaf oil and the stronger DPPH activity of the stem oil reflect differences in their phytochemical composition and antioxidant mechanisms rather than contradiction [32].
This study has several limitations. First, the antimicrobial and antioxidant activities were evaluated using in vitro assays only, which may not fully reflect in vivo biological effects. Second, the identification of compounds by GC-MS was based on spectral library matching, and further confirmation using advanced analytical techniques such as NMR is recommended. Third, the potential contamination of certain compounds such as di-n-octyl phthalate cannot be completely ruled out. Additionally, the study did not investigate the toxicity or safety profile of the essential oils. Future studies should include in vivo evaluations, toxicity assessments, and isolation of individual bioactive compounds.
This study successfully investigated the essential oils of Dioscorea dumetorum leaves and stems with respect to their yield, chemical composition, antimicrobial properties, and antioxidant potential. The oils were pale yellow with characteristic aromatic odor, and their yields, though relatively low (0.50% for leaves and 1.07% for stems), and are consistent with reports that Dioscorea species generally accumulate more starch than volatile oils.
Gas Chromatography–Mass Spectrometry (GC-MS) revealed a diverse array of bioactive constituents: 23 compounds in the leaf oil and 20 compounds in the stem oil. Leaf oil was rich in phytol (16.74%) and oxygenated ketones such as 2-pentadecanone (20.64%), both linked with strong antioxidant properties. Stem oil was dominated by di-n-octyl phthalate (34.43%) and long chain fatty acid esters, compounds associated with antimicrobial and emollient activities.
Biological assays confirmed that both oils possess broad-spectrum antimicrobial activity, inhibiting Gram-positive and Gram-negative bacteria as well as pathogenic fungi. However, the leaf extracts consistently produced larger inhibition zones than the stem extracts, though both were less potent compared to standard antibiotics and antifungal agents. In antioxidant assays, both oils demonstrated concentration-dependent activity. The leaf oil exhibited stronger ferric reducing power, while the stem oil showed higher radical scavenging potency (IC50 = 224.11 µg/mL) than the leaf oil (IC50 = 1288.52 µg/mL), but both were weaker than ascorbic acid (IC50 = 156.15 µg/mL).
Leaf oil has higher FRAP, indicating stronger reducing power, but stem oil has lower IC50 in DPPH, meaning stronger radical scavenging. These differences are due to the specific mechanisms of each assay.
Overall, the results provided scientific evidence that Dioscorea dumetorum contains bioactive compounds with both antimicrobial and antioxidant activities. The leaves appear more promising as a source of antioxidant agents, while the stems show better antimicrobial and radical scavenging potential. These findings support the ethnomedicinal use of Dioscorea dumetorum and highlight its relevance for further development in nutraceutical, pharmaceutical, and cosmetic applications.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
GO: Conceptualization, Methodology, Supervision, Investigation, Data analysis, Writing-original draft, Supervision. AB: Laboratory investigation, Practical, Result analysis, Conceptualization, Methodology. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The plant Dioscorea dumetorum used in this study was collected from Fiditi, Oyo State, Nigeria, with the consent of the landowner. The collection was carried out in accordance with local regulations and recognized ethical guidelines for the collection of plant materials.
The authors appreciate the Department of Chemistry, University of Ibadan for the use of UV-Visible Spectrophotometer used for the antioxidant screening and also the staff of Central Science Laboratory, University of Lagos for the use of GC-MS equipment.
This research received no external funding.
Given her role as the Editorial Board member, Ganiyat K. Oloyede had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Mehmet Ozaslan.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








