1 School of Basic Medical Sciences, Qujing University of Medicine & Health Sciences, 655100 Qujing, Yunnan, China
2 School of Pharmacy, Qujing University of Medicine & Health Sciences, 655100 Qujing, Yunnan, China
3 School of Clinical Medicine, Qujing University of Medicine & Health Sciences, 655100 Qujing, Yunnan, China
4 School of Traditional Chinese Medicine, Qujing University of Medicine & Health Sciences, 655100 Qujing, Yunnan, China
5 School of Medical Technology, Qujing University of Medicine & Health Sciences, 655100 Qujing, Yunnan, China
†These authors contributed equally.
Abstract
Renal interstitial fibrosis (RIF) is a prevalent and irreversible process that drives the progression of chronic kidney disease (CKD) to end-stage renal failure and is characterized by significant deposition of fibrotic matrix proteins. The unilateral ureteral obstruction (UUO) model induces rapid tubulointerstitial fibrosis and is widely used to study antifibrotic interventions. Scutellarin, an active flavonoid derived from Erigeron breviscapus, has demonstrated efficacy in alleviating symptoms of various chronic diseases, including stroke, neurodegenerative disorders, and cardiovascular and renal diseases. However, the impact of scutellarin on renal fibrosis in obstructive nephropathy remains unclear. Thus, this study evaluated the effects of scutellarin on fibrosis and renal function in a UUO mouse model.
Male C57BL/6 mice underwent UUO surgery and received oral scutellarin (21 mg/kg/day) for 3 days before and 5 days after surgery. Renal fibrosis was assessed by immunohistochemistry (IHC; collagen I) and Western blotting (WB; alpha-smooth muscle actin (α-SMA), fibronectin (FN), collagen I). Kidney function was evaluated by urinary protein (BCA assay), serum blood urea nitrogen (BUN), and serum creatinine (Scr). Data were analyzed using GraphPad Prism 8; p < 0.05 was considered statistically significant.
UUO induced significant interstitial fibrosis, as evidenced by increased collagen I deposition and elevated protein levels of α-SMA, FN, and collagen I. Scutellarin treatment significantly attenuated these changes, reducing the collagen I IHC-positive area to 0.63-fold and the α-SMA, FN, and collagen I protein levels (WB, normalized to glyceraldehyde-3-phosphate dehydrogenase) to 0.21-, 0.45-, and 0.54-fold of UUO levels, respectively (all p < 0.01). Concurrently, scutellarin significantly lowered proteinuria, BUN, and Scr compared with the UUO group (all p < 0.0001).
Scutellarin ameliorated renal fibrosis and dysfunction in the UUO mouse model, associated with downregulation of key fibrosis-related proteins. These findings support further preclinical investigation of scutellarin as a potential antifibrotic agent in kidney disease.
Keywords
- renal fibrosis
- scutellarin
- Erigeron breviscapus
- ureteral obstruction
- antifibrotic agents
- alpha-SMA protein
- fibronectin
- collagen type I
Chronic kidney disease (CKD) affects millions worldwide and frequently progresses to end-stage renal disease, requiring dialysis or transplantation. Renal interstitial fibrosis (RIF) characterized by excessive extracellular matrix (ECM) deposition and loss of functional parenchyma is the final common pathway underlying CKD progression, regardless of the initial etiology [1]. RIF disrupts renal architecture through fibroblast activation and ECM accumulation, leading to impaired glomerular filtration and tubular function [2]. Given its irreversible nature and central role in CKD, halting or reversing RIF has become a critical focus in nephrology research.
Myofibroblast activation is a pivotal event in RIF pathogenesis [3]. These
specialized cells, characterized by the expression of alpha-smooth muscle actin
(
The unilateral ureteral obstruction (UUO) mouse model is a well-established and widely used experimental system in nephrology research [8]. It rapidly induces renal fibrosis, inflammatory cell infiltration, oxidative stress, and ECM accumulation in the kidney, providing a reproducible platform for evaluating antifibrotic interventions [9, 10]. Although UUO does not fully recapitulate the slow progression of human CKD, it reliably models key aspects of fibrogenesis relevant to multiple CKD etiologies.
Scutellarin, a bioactive flavonoid derived from the traditional Chinese
medicinal herb Erigeron breviscapus, has attracted considerable
attention in recent years due to its diverse pharmacological properties.
Initially recognized for its cardiovascular benefits, including enhancing cardiac
function and exerting antithrombotic effects [11], scutellarin has since been
shown to possess a wide range of therapeutic activities in various preclinical
models. Studies have demonstrated its anti-inflammatory [12, 13], antioxidative
[14, 15], and antiapoptotic [16] effects, which contribute to its protective role
in multiple disease states. In the context of fibrosis, scutellarin has shown
promising results in ameliorating pulmonary fibrosis [17]. It achieves this by
reducing collagen deposition in lung tissues and inhibiting the expression of
inflammatory factors such as NLR family pyrin domain containing 3 (NLRP3),
interleukin 1
Based on this evidence, we hypothesized that scutellarin may attenuate renal fibrosis and dysfunction in the UUO model. Consequently, we investigated its effects on functional parameters, histological fibrosis, and the expression of key ECM and fibroblast activation markers.
Six-week-old male C57BL/6J mice were purchased from the Experimental Animal
Center of the Heshuo Business Department (Kunming, Yunnan, China). All animals
were housed in a specific pathogen-free facility under controlled conditions
(temperature: 22
Scutellarin (purity
The UUO model was established by ligation of the left ureter at two sites using 6-0 silk suture through a left flank incision under anesthesia with sodium pentobarbital (50 mg/kg, i.p., 10 mg/mL). Sham-operated mice underwent identical surgical exposure without ureteral ligation. On day 6 post-surgery, all mice were euthanized with sodium pentobarbital (150 mg/kg, i.p., 10 mg/mL), and the left kidneys, serum, and urine were collected for subsequent analyses.
All procedures were approved by the Institutional Animal Care and Use Committee and conducted in accordance with the ARRIVE guidelines.
Mouse renal tissues were fixed in 4% paraformaldehyde, dehydrated through graded ethanol, and embedded in paraffin. Four-micrometer sections were deparaffinized, rehydrated, and incubated in 3% hydrogen peroxide for 30 min to block endogenous peroxidase activity. Antigen retrieval was performed by heating slides in citrate-based antigen unmasking solution (boiling, 5 min). Sections were blocked in 5% goat serum for 60 min at room temperature and then incubated overnight at 4 °C with primary antibody against collagen Ⅰ (1:200, 14695-1-AP; Proteintech, Wuhan, Hubei, China). After washing three times with phosphate-buffered saline, sections were incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG secondary antibody (1:500, ab6721; Abcam, Cambridge, UK) for 50 min at room temperature. Immunoreactivity was visualized using a diaminobenzidine (DAB) chromogen substrate kit (ZSGB-BIO, Beijing, China), followed by hematoxylin counterstaining. Stained slides were imaged using the Leica microscope system (Leica Microsystems, Wetzlar, Germany), and collagen Ⅰ positive areas (%) were quantified using ImageJ software (National Institutes of Health [NIH], Bethesda, MD, USA).
Renal cortical tissues were homogenized in ice-cold lysis buffer containing
protease and phosphatase inhibitors (KeyGen Biotech, Nanjing, Jiangsu, China).
The bicinchoninic acid (BCA) assay was implemented to quantify the extracted
protein concentrations. Equal amounts of protein were separated by sodium dodecyl
sulfate polyacrylamide gel electrophoresis and electrotransferred to PVDF
membranes (Roche Applied Sciences, Penzberg, Bavaria, Germany). Membranes were
blocked in 5% non-fat milk in Tris-buffered saline containing 0.1% Tween 20
(TBST) for 1 h at room temperature and then incubated overnight at 4 °C with
primary antibodies targeting
At the end of the experiment, urine and blood samples were collected from all mice. Urinary total protein, blood urea nitrogen (BUN) and serum creatinine (Scr) were measured to assess renal function. Total urinary protein concentration was determined using the Pierce BCA Protein Assay Kit (23225; Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. BUN and Scr levels were quantified using an automated biochemical analyzer (Hitachi 7080, Tokyo, Japan) with commercially available enzymatic reagents.
Data are presented as the mean
Renal fibrosis in the UUO model is characterized by excessive deposition of ECM
components. Immunohistochemistry (IHC) analysis revealed that the interstitial
area positive for collagen I was significantly increased in UUO kidneys compared
with sham-operated controls (p
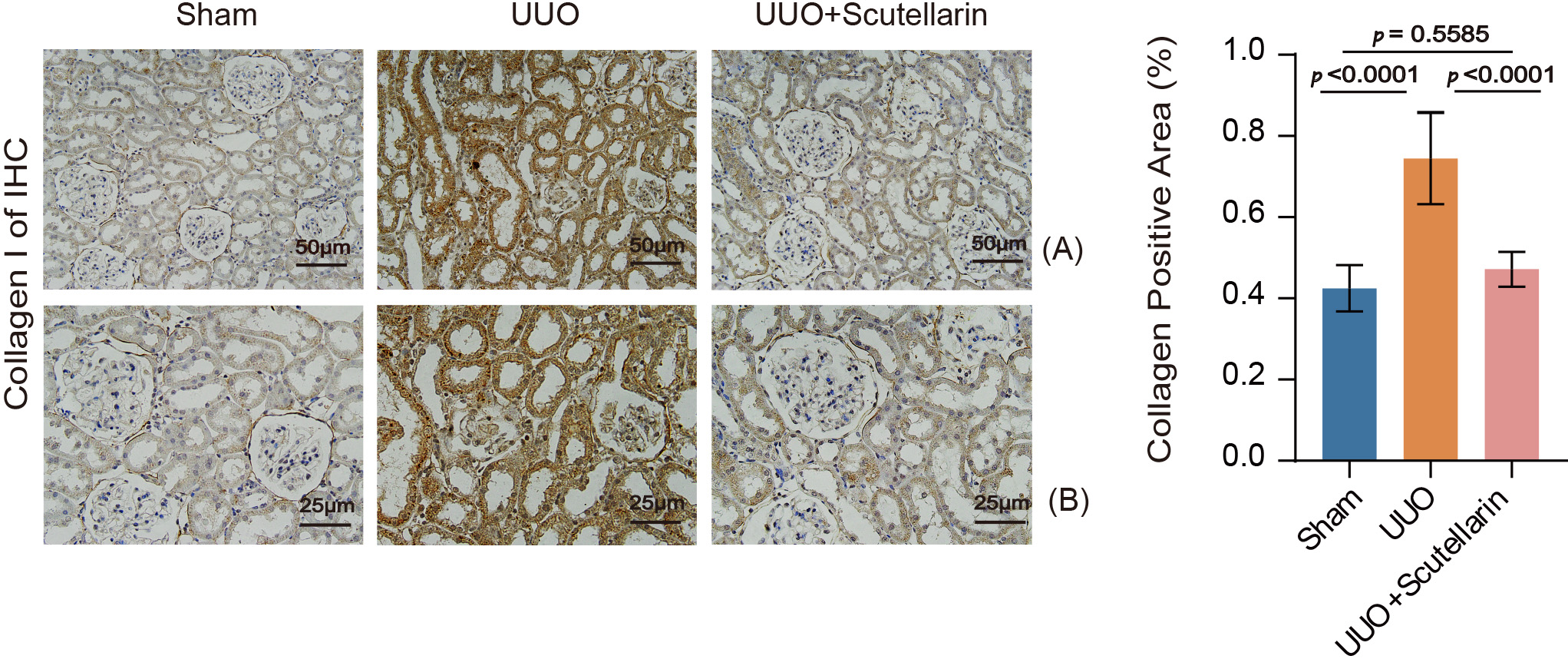 Fig. 1.
Fig. 1.
Scutellarin therapy alleviated collagen deposition caused by UUO
in mouse kidneys. Collagen positive area (%) was quantified by IHC. Data are
shown as the mean
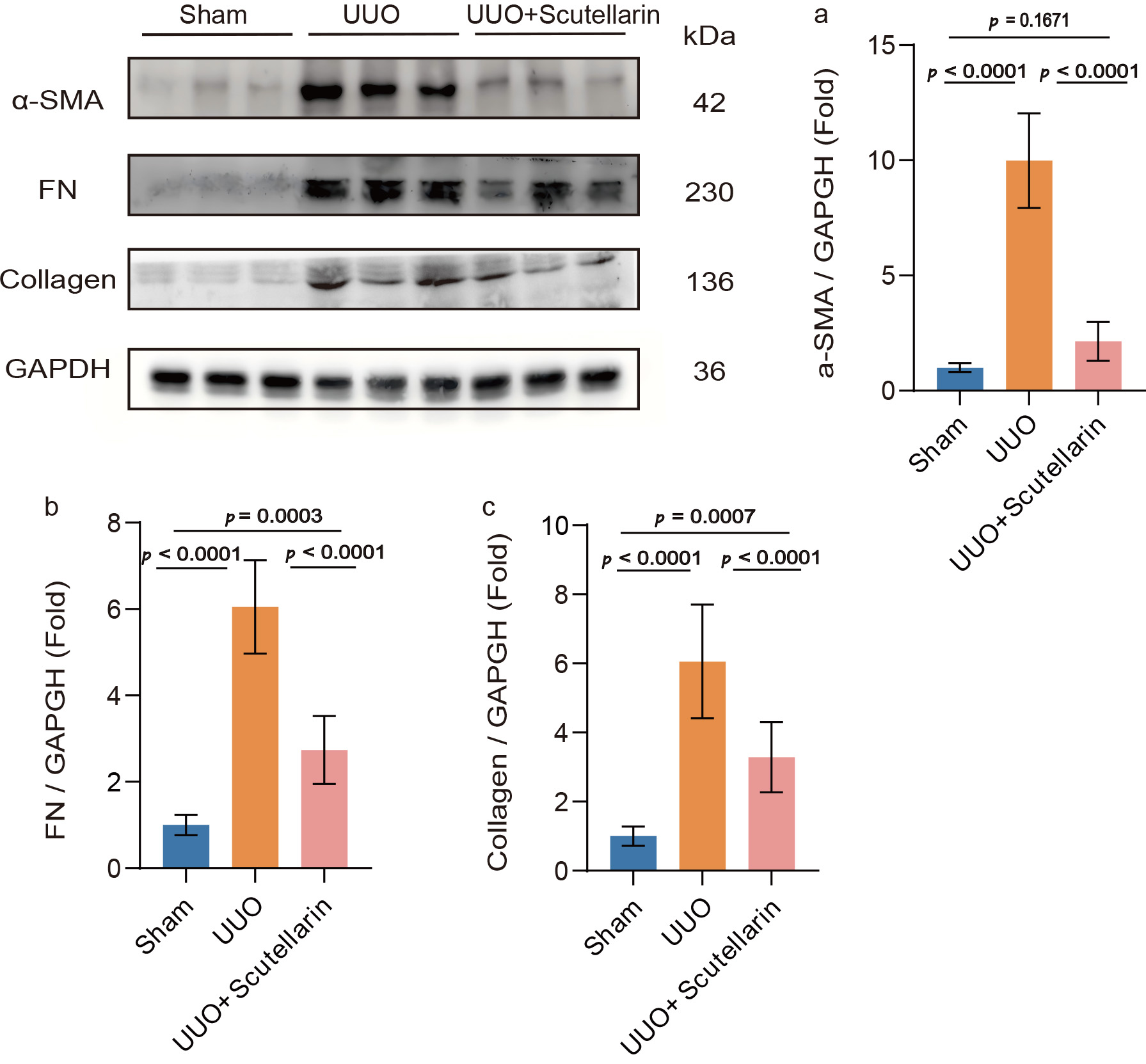 Fig. 2.
Fig. 2.
Scutellarin treatment decreased the levels of fibrosis-related
proteins in UUO mouse kidneys. Western blot analysis of (a)
| Parameter | Sham | UUO | UUO+Scutellarin | Fold change | Fold change | p value |
| (Mean |
(Mean |
(Mean |
(UUO/Sham) | (Scut/UUO) | (Scut vs. UUO) | |
| Collagen IHC (%) | 0.44 |
0.73 |
0.46 |
1.66 | 0.63 | |
| 1.00 |
9.99 |
2.14 |
9.99 | 0.21 | ||
| FN/GAPDH | 1.00 |
6.05 |
2.74 |
6.05 | 0.45 | |
| Collagen/GAPDH | 1.00 |
6.06 |
3.29 |
6.06 | 0.54 | |
| Proteinuria (mg/mL) | 0.11 |
2.88 |
1.70 |
26.51 | 0.59 | |
| BUN (mmol/L) | 10.51 |
24.91 |
17.20 |
2.37 | 0.69 | |
| Scr (µmol/L) | 37.10 |
87.37 |
61.73 |
2.36 | 0.71 |
Data are shown as the mean
We further assessed two additional fibrosis-associated proteins:
Collectively, scutellarin treatment attenuates UUO-induced renal fibrosis, as
evidenced by significant reductions in interstitial collagen I deposition and the
expression of key fibrosis-related proteins, including
Urinary protein, BUN, and Scr are well-recognized biochemical indicators commonly employed to evaluate the extent of kidney damage and assess functional alterations in renal pathologies.
In the UUO group, all three parameters were significantly elevated compared with
sham-operated controls (p
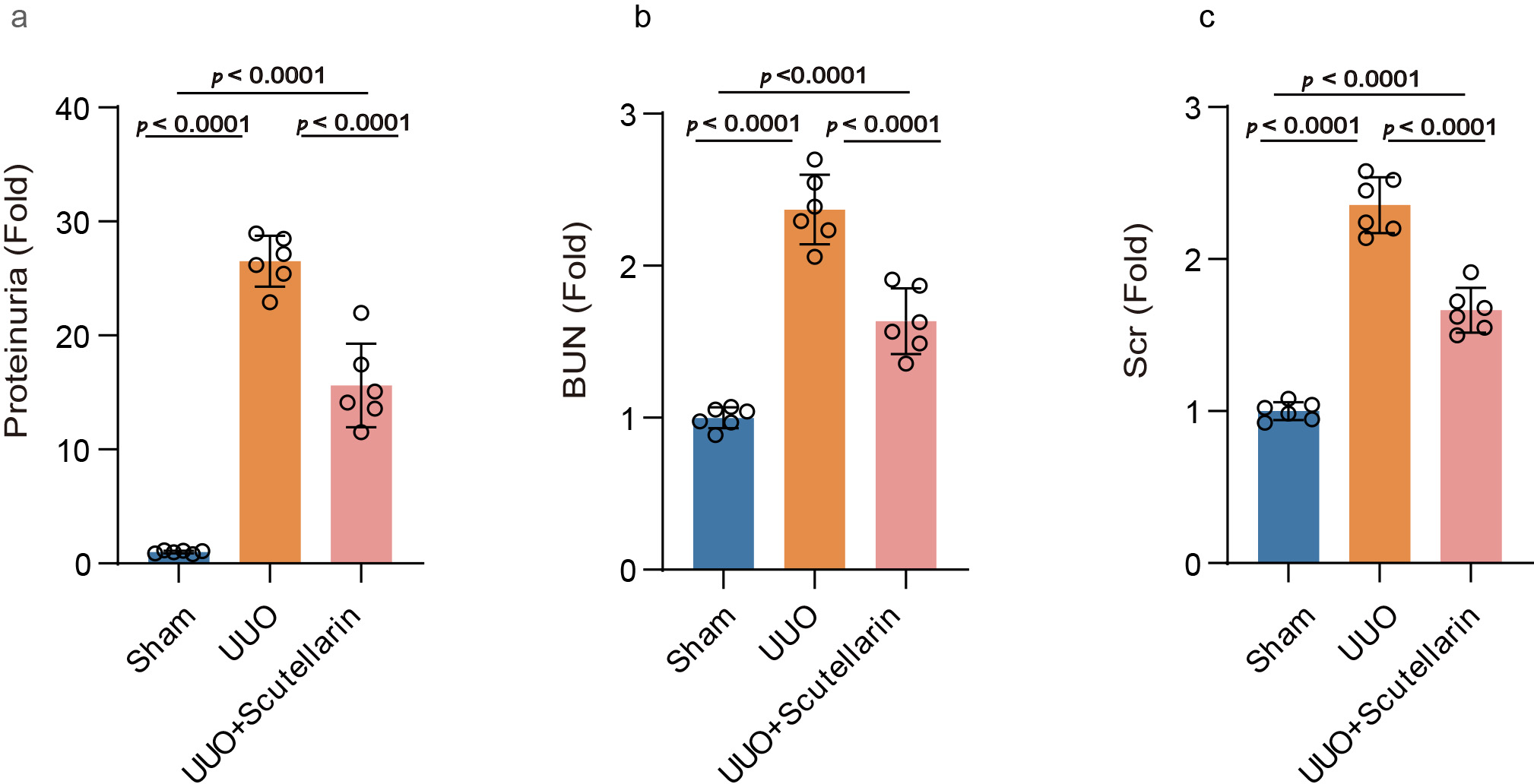 Fig. 3.
Fig. 3.
Scutellarin treatment mitigated renal impairment in UUO mouse
kidneys. Levels of (a) urinary protein, (b) BUN, and (c) Scr are expressed as
fold change relative to the sham group. Data are shown as the mean
These results demonstrated that scutellarin treatment ameliorated UUO-induced renal dysfunction, as evidenced by the significant reduction in key biochemical markers of kidney injury.
In this study, we demonstrated that scutellarin, a major active ingredient
derived from the traditional Chinese herb Erigeron breviscapus—exerts
potent antifibrotic effects on an UUO-induced mouse model of chronic
tubulointerstitial fibrosis. Treatment with scutellarin significantly improved
kidney function, as reflected by reductions in proteinuria, BUN, and Scr.
Histologically, this was accompanied by decreased interstitial collagen
deposition, confirmed by both IHC quantification of collagen I (%) and Western
blot analysis showing downregulation of collagen I, FN, and
The coordinated suppression of
This multimodal activity places scutellarin within a broader class of natural
compounds derived from traditional Chinese medicine that target renal fibrosis
through shared and distinct mechanisms. For
instance, baicalin (from Scutellaria baicalensis) [22, 23]
and curcumin (derived from Curcuma longa) [24, 25] also suppress
TGF-
It is important to acknowledge that scutellarin possesses documented anti-inflammatory and antioxidant properties in other organ systems [10, 14, 15, 18], and given that inflammation and oxidative stress can amplify fibrogenic signaling in the obstructed kidney, such ancillary effects may indirectly contribute to the observed benefit. Since direct measurements of redox status, inflammatory cytokines, or inflammasome components were not conducted in our model, the involvement of these pathways is speculative and warrants future investigation.
Unlike current standard-of-care agents (e.g., angiotensin-converting enzyme inhibitors or angiotensin receptor blockers) that primarily modulate hemodynamics to slow rather than reverse fibrosis [29, 30, 31], scutellarin therapeutically targets key structural components of the fibrotic response, evidenced by decreased collagen I deposition and reduced myofibroblast marker expression.
This study has several limitations that should be acknowledged. First, while the
observed downregulation of
Despite the limitations acknowledged above, Our findings demonstrate that
scutellarin attenuates RIF and improves functional parameters in the UUO mouse
model, associated with reduced expression of collagen I,
The datasets used in the current study are available from the corresponding author upon reasonable request.
LG designed the research and contributed to manuscript writing. LG, ZZ, QL and DC performed the research and conducted experiments. LG, DC, CC and ZZ performed data analysis. TY, YW, RC participated in research design, data collection and analysis, interpretation of results, and manuscript writing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Institutional Animal Ethics Committee of Qujing University of Medicine & Health Sciences approved (No. DL2025003) and conducted the study according to the Regulations on the Administration of Laboratory Animals (State Council of China), the Guiding Opinions on the Humane Treatment of Laboratory Animals (MOST, 2006), the Guidelines for Ethical Review of Laboratory Animal Welfare (GB/T 35892-2018), and the ARRIVE 2.0 guidelines.
Not applicable.
This research was supported by the following grants: (1) the 2025 Science Research Project of Yunnan Provincial Department of Education, “Research of Scutellarin by Inhibiting NLRP3 Inflammasome to Delay Pyroptosis of Diabetic Nephropathy Podocytes” (2025J1657); and (2) the Applied Basic Research Joint Special Funds of Chinese Medicine from Yunnan Provincial Science and Technology Department (202401AZ070001-097).
The authors declare no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



