1 School of Medical Technology, Guangdong Medical University, 523808 Dongguan, Guangdong, China
2 The Affiliated Dongguan Songshan Lake Central Hospital, Guangdong Medical University, 523000 Dongguan, Guangdong, China
†These authors contributed equally.
Abstract
Tuberculosis (TB) remains one of the deadliest infectious diseases worldwide. The substantial reservoir of latent infections, the emergence of multidrug-resistant and extensively drug-resistant TB (MDR/XDR-TB), and the prolonged duration and toxicity of current therapeutic regimens underscore the urgent need for novel treatment strategies. Therapeutic vaccines, an innovative immunotherapeutic approach, are designed to be administered alongside standard pharmacological treatments. By enhancing pathogen-specific immune responses in the host, these vaccines aim to eradicate persistent bacteria, reduce treatment duration, and prevent disease recurrence, thus representing a promising strategy for addressing MDR-TB. This review systematically summarizes the leading therapeutic vaccine candidates currently in clinical trials, detailing the associated mechanisms of action and preclinical and clinical efficacy data. Additionally, the review critically examines existing challenges and outlines future research directions. Overall, this comprehensive analysis seeks to enhance understanding of the potential and development pathways of therapeutic vaccines for TB treatment, contributing to the global effort against this widespread health threat.
Keywords
- TB
- therapeutic vaccines
- immunotherapy
- clinical research
- latent infection
- MDR-TB
Tuberculosis (TB) is caused by Mycobacterium tuberculosis (Mtb) and is transmitted between individuals via respiratory contact. It primarily affects the lungs but can potentially damage any organ or tissue. Despite sustained global efforts to control TB, it continues to pose a significant threat to public health. According to the World Health Organization’s (WHO) 2024 Global TB Report, approximately 10.8 million new TB cases occurred worldwide in 2023, reflecting an incidence rate of 134 per 100,000 individuals, a 0.2% increase compared to 2022 [1]. Currently, intradermal administration of the Bacillus Calmette-Guérin (BCG) vaccine remains the most widely used approach for TB prevention. Nevertheless, the protective efficacy following BCG vaccination in newborns, effective in preventing severe forms such as miliary and meningeal TB, persists only for about 10 years and gradually diminishes thereafter, resulting in insufficient protection among adults [2].
In recent decades, following the widespread application of anti-TB drugs, the prevalence of drug-resistant and multidrug-resistant strains has markedly increased. Experimental findings indicate that among 2367 isolates, 273 (11.5%) exhibited resistance to at least one anti-TB drug, while 221 (9.3%) were classified as multidrug-resistant TB (MDR-TB) [3]. For example, in Pakistan, approximately 518,000 TB cases were reported, including around 15,000 MDR-TB cases [4]. Drug-resistant mutations arise due to inadequate or inappropriate treatment. If these mutations persist, continued selection pressure from improper treatment will eventually lead to the emergence of MDR-TB within the population [5].
The current treatment approach for drug-susceptible TB involves a six-month regimen comprising four drugs, which is associated with significant side effects and suboptimal patient adherence. Conversely, therapeutic regimens for multidrug-resistant cases are more complex, potentially requiring the administration of up to seven drugs over periods ranging from 9 to 24 months. Currently, the clinical development of a single novel anti-TB drug typically requires at least six years, while the sequential development of new combination regimens can extend to approximately 20 years or longer [6]. Such efforts incur substantial research and development costs, and global capacity to support these activities remains limited [7]. The management of latent tuberculosis infection (LTBI) also faces challenges related to adherence and hepatotoxicity. Most studies utilize a nine-month isoniazid regimen for LTBI treatment; however, utilization rates and treatment adherence demonstrate substantial variability (18% to 100%), as assessed by treatment completion rates. The most frequently reported adverse events include hepatotoxicity, gastrointestinal intolerance, and neuropathy, with incidence rates ranging from 1% to 48% [8], as shown in Fig. 1.
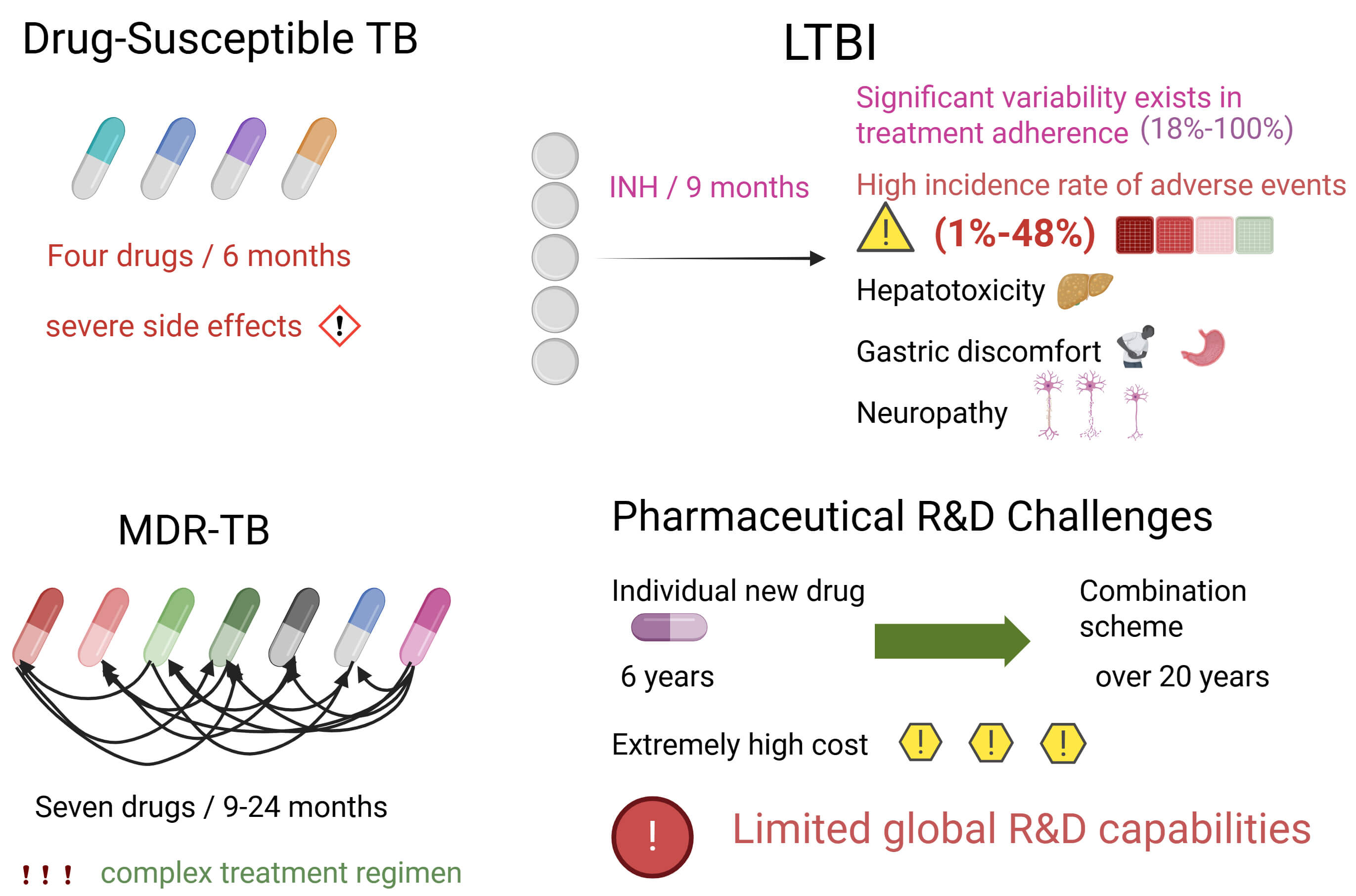 Fig. 1.
Fig. 1.
Current treatment approaches for drug-susceptible TB. Created in BioRender. LIN, Q. (2025) https://BioRender.com/de31uyj. TB, tuberculosis; LTBI, latent tuberculosis infection; MDR-TB, multidrug-resistant TB.
Given the global context of more than 10 million new TB cases annually, including approximately 500,000 drug-resistant cases, there is an urgent need for innovative therapeutic interventions to achieve the strategic goals outlined in the End TB Strategy [9]. This strategy aims to reduce TB incidence by 80% and mortality by 90% by 2030, while concurrently eliminating catastrophic expenditures for affected households [10]. Despite the availability of antibiotics with potential efficacy against TB, the disease continues to cause substantial morbidity and mortality globally, particularly in developing countries. Following the completion of the whole-genome sequencing of Mtb (approximately 4000 genes), researchers have identified numerous small molecules exhibiting potent activity against both drug-susceptible (DS) and drug-resistant (DR) strains, subsequently characterizing and validating their targets.
To address challenges posed by drug resistance, many researchers have shifted their focus from conventional targets of existing anti-TB antibiotics to newly identified drug targets within Mtb. Recent targets of interest include GyrA/B, ATP synthase, QcrB, DprE1, FadD32, Pks13, and MmpL3. Nevertheless, several candidate compounds targeting these mechanisms have encountered limitations, such as toxicity, inadequate in vivo efficacy, or unfavorable elimination half-lives [11].
Particularly, the treatment of MDR-TB has long posed a formidable challenge in global medicine, with adverse reactions such as ototoxicity being especially prominent. To effectively address this issue, the World Health Organization (WHO) introduced a new short treatment regimen (STR) in 2020. This regimen replaces traditional injectable aminoglycosides with the oral drug bedaquiline, often administered in combination with pretomanid, linezolid, and moxifloxacin [12]. This innovative regimen offers significant advantages. In terms of safety, it substantially reduces the risk of ototoxicity, fundamentally enhancing treatment safety and minimizing unnecessary physical harm to patients. Regarding administration, the oral mode is more convenient, significantly improving patient adherence and facilitating smoother treatment progression. This not only alleviates patient suffering but also enhances therapeutic efficacy. However, the new regimen is not without limitations and introduces new challenges. Potential cardiac-related adverse reactions, such as QT prolongation, necessitate regular electrocardiogram (ECG) monitoring of patients [13]. To ensure patient safety and treatment effectiveness, it is urgent to establish and improve national pharmacovigilance systems and patient safety data collection frameworks. These systems and data flows can promptly and accurately collect and analyze adverse drug reaction information, providing robust data support for the continuous evaluation and optimization of MDR-TB treatment [14]. In promoting the new regimen, significant attention must be paid to safety monitoring challenges, and relevant systems and mechanisms must be continually improved. Only through these measures can the benefits of the new regimen be fully realized, delivering improved treatment outcomes and quality of life to MDR-TB patients.
Despite considerable efforts to develop more effective anti-TB therapeutics, only three drugs with novel mechanisms of action have been approved since 2013, and all are associated with notable adverse effects [11]. Consequently, there is an urgent need for more efficacious vaccines that can confer immunity against all forms of TB across the entire population, along with novel anti-TB medications surpassing existing regimens in efficacy, tolerability, and treatment duration, to cure TB and reduce its transmission. Therapeutic vaccines precisely meet these requirements.
The onset and progression of TB are closely associated with host immunological status, and the risk of developing TB is significantly elevated in specific populations such as individuals with diabetes mellitus. Diabetes not only induces metabolic dysregulation but also impairs the functions of granulocytes, monocytes and lymphocytes, thereby increasing bacterial infection susceptibility, promoting progression from latent to active disease, and resulting in poorer treatment outcomes [15]. Therapeutic vaccines, which function by enhancing pathogen-specific immune responses in individuals already infected to control or eliminate TB infection, have emerged as a promising adjunctive therapy and a major global research focus. In contrast to prophylactic vaccines, which aim to prevent Mtb infection or its progression to active disease, therapeutic vaccines serve as auxiliary treatments for TB or as measures to prevent recurrence after clinical cure [16]. Prophylactic and therapeutic vaccines for TB differ significantly in key immune responses, reflecting their distinct immunological goals and mechanisms. Prophylactic vaccines aim to establish immunological barriers against Mtb infection by stimulating Th1/Th17 cellular immunity, CD8+ T-cell responses, and protective antibody production (e.g., anti-LAM antibodies), thereby forming immunological memory. This memory acts as a “sentinel”, enabling rapid pathogen recognition and defense upon invasion. In contrast, therapeutic vaccines primarily target individuals already infected. During infection, components such as lipoarabinomannan (ManLAM) induce immunosuppression, and therapeutic vaccines aim to reverse this condition. They focus on restoring T-cell functionality, inhibiting regulatory T-cell (Treg) expansion, and utilizing CD1 molecules to present glycolipid antigens (e.g., Ac2SGL) to activate specific T-cells. These actions enhance infected cell clearance while regulating inflammation to prevent excessive immune responses [17, 18].
Overall, preventive vaccines emphasize preemptive immune preparation and memory formation, whereas therapeutic vaccines prioritize immune restoration and pathological modulation after infection. Both play distinct yet critical roles in TB prevention and control. This review systematically summarizes recent advancements in therapeutic vaccines for TB treatment, highlighting their mechanisms of action and clinical translation results.
Upon entering macrophages, Mtb ensures its survival primarily by
inhibiting phagosome–lysosome fusion and acidification. It secretes protein
kinase G (PknG) to interfere with intracellular signaling pathways, suppressing
NF-
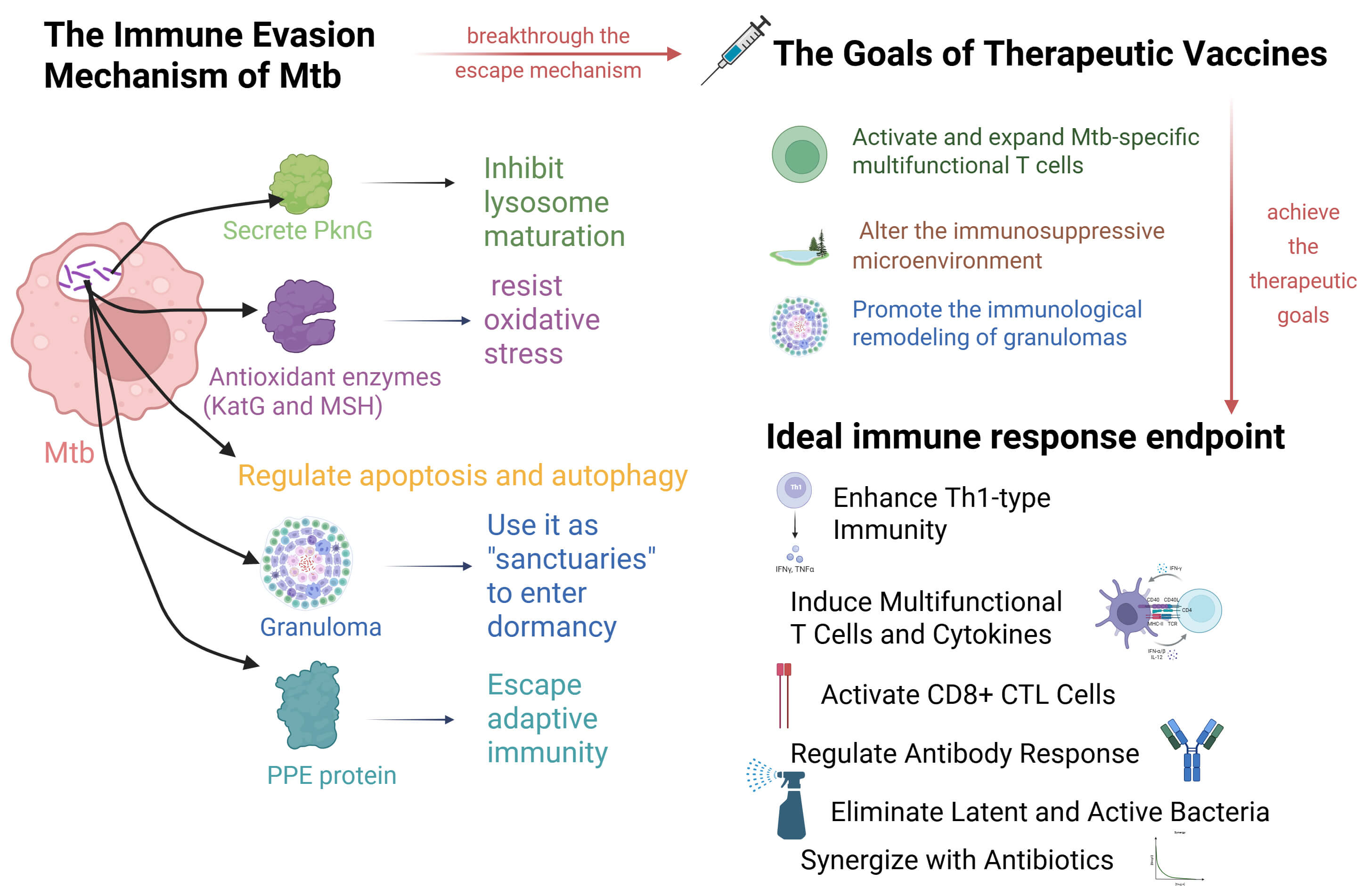 Fig. 2.
Fig. 2.
Mechanisms of action and immunological foundations of therapeutic vaccines. Created in BioRender. LIN, Q. (2025) https://BioRender.com/b9vth9a.
Furthermore, Mtb regulates host cell metabolism through the manipulation of iron, hydrogen, and calcium ions, further supporting its survival. When the host forms granulomas to restrict bacterial dissemination, Mtb exploits these structures as protective niches and enters a metabolically quiescent state, thereby evading immune-mediated clearance. To escape adaptive immunity, Mtb adopts multiple strategies, including modulation of Toll-like receptor (TLR) signaling, inhibition of major histocompatibility complex class II (MHC-II) via proline–proline–glutamic acid (PPE) proteins to suppress antigen presentation, and induction of interleukin-10 (IL-10) production to disrupt cytokine balance. Collectively, these mechanisms enable Mtb to establish long-term latent infection and reactivate when host immunity declines [19].
Therapeutic vaccines aim to activate and enhance Mtb-specific multifunctional T cells (e.g., Th1 cells and CD8+ cytotoxic T lymphocytes [CTLs]), thereby increasing bactericidal capacity [16]. Additionally, these vaccines modify the immunosuppressive microenvironment, such as by modulating regulatory T (Treg) cell function or inhibiting immune checkpoints, including programmed cell death protein 1 (PD-1) [20]. They also facilitate the immunological reconstruction of granulomas, enabling bactericidal immune cells to infiltrate infection sites and eliminate pathogens [20], as shown in Fig. 2.
The ideal immune response endpoints for therapeutic TB vaccines encompass
multiple dimensions. These vaccines should augment Th1-type cellular immunity,
trigger robust multifunctional T-cell responses with the secretion of cytokines
(IFN-
TB/FLU-04L (V7) is a novel intranasal TB vaccine candidate based on an attenuated influenza A virus vector encoding two mycobacterial antigens: Ag85A and ESAT-6, as shown in Fig. 3 and Table 1 (Ref. [21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54]). The vaccine design mimics the respiratory infection route, thereby inducing robust mucosal and systemic immune responses via intranasal immunization [55]. Clinical Objective: Adjunctive therapy aimed at shortening treatment duration. Current Status: The initial Phase II trial was limited by a relatively small sample size, yet positive trends in sputum smear conversion and weight improvement were still observed [21]. In another subsequent Phase II trial, the V7 group demonstrated more prominent results, with a sputum smear conversion rate of 72.2%, significantly higher than the 19% of the placebo group, and a notable increase in body weight [22]. The large-scale Phase III trial further confirmed its advantages. The V7 vaccine increased the overall sputum smear conversion rate to 68% within one month and significantly improved indicators such as inflammation, anemia, and liver damage, showing great potential for accelerating the treatment course and comprehensively optimizing clinical outcomes [23]. The results of a series of Phase II and Phase III clinical trials conducted on the oral V7 vaccine indicate that this vaccine is both safe and effective as an adjuvant immunotherapy for TB.
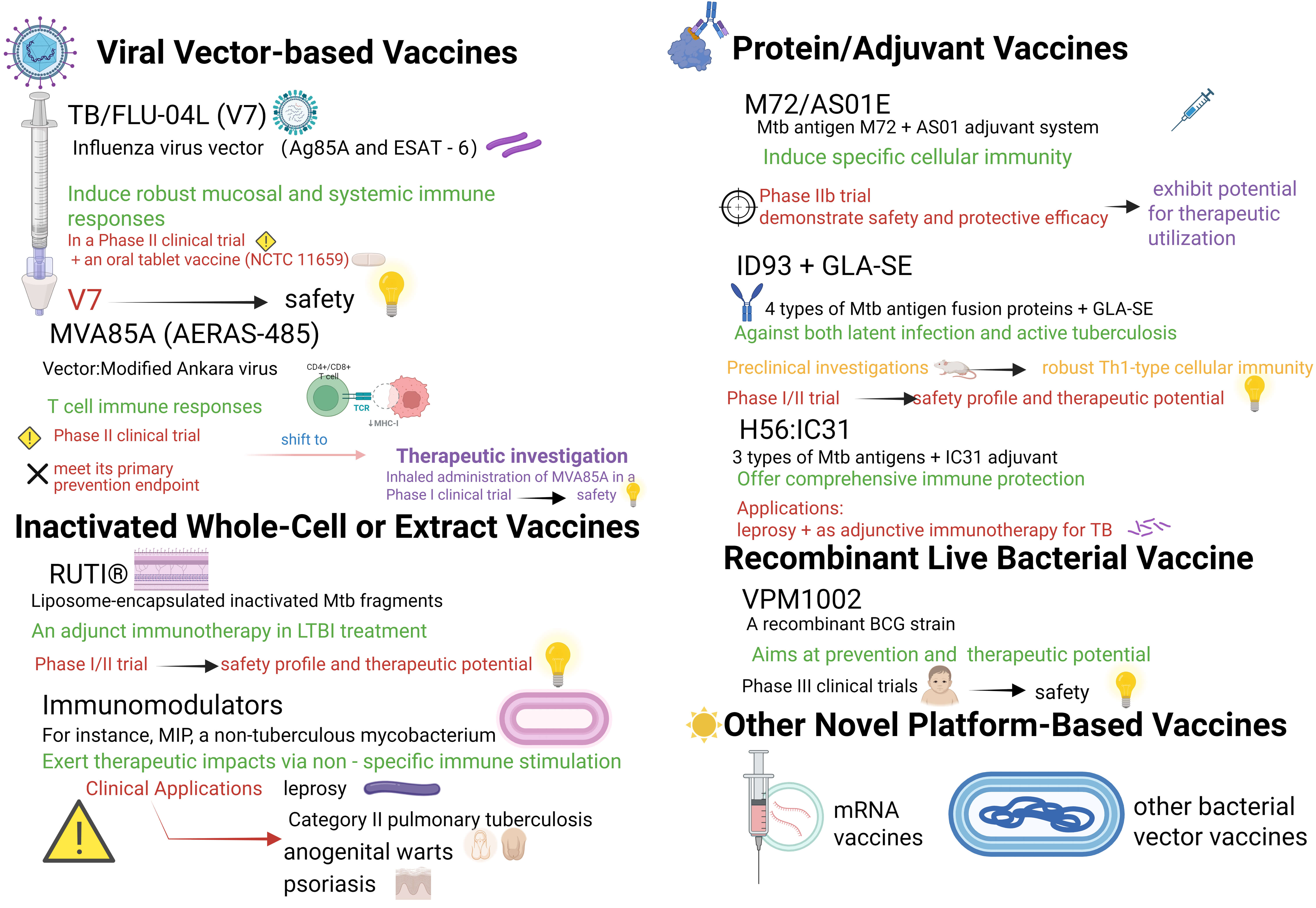 Fig. 3.
Fig. 3.
Research advancements in principal therapeutic vaccine candidates. Created in BioRender. LIN, Q. (2025) https://BioRender.com/hzhm0kk.
| Vaccine | Vaccine components | Implementing institution | Manufacturer | Manufacturer’s location | Phase | Clinical objective | Study population | Trial status | Reference |
| TB/FLU-04L (V7) | Ag85A and ESAT-6 + 10 µg heat-killed Mycobacterium vaccae. | Lisichansk TB Dispensary, Ukraine | Immodulon Therapeutics Ltd | London, UK. | II | To evaluate its safety and efficacy as an adjunct immunotherapy, aiming to enhance the effectiveness of standard chemotherapy and potentially shorten the treatment duration. | 41 patients with active pulmonary TB, Patients included various types: drug-sensitive, retreated, drug-resistant, and HIV-coinfected. | Completed (1-month trial) | [22] |
| Lischansk Regional TB Dispensary (Ukraine); Kharkiv National Medical University; Luhansk State Medical University; Immunitor Inc. (Canada) | Anhui Longcom Biologic Pharmacy Co., Ltd. | 93 Kexue Avenue, National New High-Tech Industrial Development Zone, Hefei, Anhui Province 230088, China. | II | To evaluate safety and efficacy as adjunct immunotherapy in sputum smear-positive pulmonary TB patients, with primary endpoint of sputum smear clearance after one month. | 43 sputum smear-positive pulmonary TB patients (22 treatment, 21 placebo), including newly diagnosed, re-treated, and MDR-TB cases. | Completed | [21] | ||
| Sponsor: Immunitor LLC Main Implementing Sites: Mongolia: Misheel Clinic of Lung Surgery, Sukhbaatar District TB Dispensary, Bayanzurkh District TB Dispensary, etc. |
BioEos Ltd. | Marden, Kent, UK. | III | To evaluate the efficacy and safety as an adjunct immunotherapy, aiming to shorten the TB treatment duration, reduce chemotherapy side effects (e.g., liver damage), and improve clinical indicators. | 152 patients with active pulmonary TB, patients included drug-sensitive and multidrug-resistant TB types. | Completed (The conclusions still require follow-up validation) | [23] | ||
| Ukraine: Lisichansk Regional TB Dispensary, Kharkiv National Medical University, Special TB Dispensary No. 1, etc. | |||||||||
| International Collaborators: Stellenbosch University, London School of Hygiene & Tropical Medicine, etc. | |||||||||
| MVA85A (AERAS-485) | MVA virus as a vector to express antigen Ag85A. | SATVI, University of Cape Town; Centre for Clinical Vaccinology and Tropical Medicine, University of Oxford (UK). |
Not explicitly stated (MVA85A was developed by the University of Oxford.) | Not explicitly stated | I | To evaluate the safety and immunogenicity of MVA85A as a booster vaccine in BCG-primed, healthy adults exposed to mycobacteria but uninfected with Mtb. | 24 healthy, BCG-vaccinated adults from a TB-endemic area in South Africa. | Completed (1-year follow-up) | [43] |
| Multiple centers in the UK, including the University of Oxford, Oxford University Hospitals NHS Trust, University of Birmingham, Royal Free Hospital, King’s College Hospital, etc. | IDT Biologika GmbH | Dessau-Roßlau, Germany. | I | To evaluate the safety and immunogenicity of MVA85A delivered by aerosol inhalation in healthy adults with LTBI. | Healthy UK adults aged 18–55 with LTBI. | Completed | [25] | ||
| SATVI, Aeras, Oxford-Emergent Tuberculosis Consortium (OETC), University of Cape Town, University of Oxford, Emergent BioSolutions, etc. | Not explicitly stated (MVA85A was developed by the University of Oxford in collaboration with Emergent BioSolutions.) | Not explicitly stated | IIb | To assess the safety, immunogenicity, and efficacy of MVA85A against TB and Mtb infection in BCG-vaccinated infants. | Healthy, HIV-negative infants aged 4–6 months who had previously received BCG vaccination. | Completed (enrollment from July 15, 2009, to May 4, 2011; follow-up completed October 2012) | [24] | ||
| Primarily conducted by South African institutions. | Not explicitly stated (MVA85A was developed by the University of Oxford.) | Not explicitly stated | II | To evaluate the safety and immunogenicity of a novel strategy using MVA85A as a prime vaccine in HIV-exposed newborns, followed by selective, deferred BCG vaccination at 8 weeks of age. | 248 HIV-exposed newborns | Completed (Follow-up to 52 weeks/1 year for safety and immunogenicity assessment) | [44] | ||
| MVA85A-IMX313 (fusion vaccine) | Modified Vaccinia Ankara virus expressing Ag85A fused with IMX313 (oligomerization domain from chicken. C4b-binding protein) | The Jenner Institute, University of Oxford; School of Cancer Sciences, University of Birmingham; IMAXIO, Lyon, France. | IDT Biologika GmbH | Germany | I | To assess the safety and immunogenicity of MVA85A-IMX313 compared with MVA85A in BCG-vaccinated adults (first-in-human evaluation of IMX313). | Healthy BCG-vaccinated UK adults aged 18–55 years, without LTBI. | Completed | [26] |
| ChAdOx1 85A–MVA85A | ChAdOx1 85A (Chimpanzee adenovirus-vectored vaccine expressing Ag85A); MVA85A (Modified vaccinia Ankara virus expressing Ag85A). | The Jenner Institute, University of Oxford; Institute of Immunology and Immunotherapy, University of Birmingham. | ChAdOx1 85A: Clinical Biomantufacturing Facility (CBF), University of Oxford, UK; MVA85A: IDT Biologika GmbH, Germany. | ChAdOx1 85A: Oxford, UK; MVA85A: Germany. | I | To evaluate the safety and immunogenicity of ChAdOx1 85A alone and in a prime-boost regimen with MVA85A in BCG-vaccinated healthy UK adults. | Healthy UK adults aged 18–55 years, previously vaccinated with BCG ( |
Completed | [27] |
| MRC/UVRI and LSHTM Uganda Research Unit; The Jenner Institute, University of Oxford. | ChAdOx1 85A: Clinical Biomanufacturing Facility, University of Oxford, Oxford, UK; MVA85A: IDT Biologika, Dessau-Rosslau, Germany; BCG vaccine (Moscow strain 361-I): Serum Institute of India, Pune, India. | ChAdOx1 85A: Oxford, UK; MVA85A: Dessau-Rosslau, Germany; BCG: Pune, India. | IIa | To evaluate safety and immunogenicity of ChAdOx1 85A–MVA85A compared with BCG revaccination in Ugandan adolescents. | Ugandan adolescents aged 12–17 years, BCG-vaccinated at birth, without active TB or recent exposure. | Completed | [28] | ||
| M72/AS01E | M72 recombinant fusion protein combined with the Adjuvant System AS01. | GSK, Aeras, and multiple research centers in Kenya, South Africa, and Zambia. | GSK | Wavre and Rixensart, Belgium. | IIb | To evaluate the safety and efficacy of the M72/AS01 candidate vaccine in preventing bacteriologically confirmed active pulmonary TB disease in HIV-negative adults with latent Mtb infection. | HIV-negative adults aged 18–50 years with latent Mtb infection. | Primary analysis completed | [45] |
| SATVI; University of Cape Town, in collaboration with GSK. | GSK Vaccines | Rixensart, Belgium. | II | To evaluate the safety, reactogenicity, and immunogenicity of the candidate vaccine M72/AS01 in healthy, HIV-negative adolescents living in a TB endemic setting, regardless of Mtb infection status. | Healthy, HIV-negative adolescents aged 13–17 years living in a TB endemic area. | Completed | [46] | ||
| YRG CARE Medical Centre, Chennai, India. | GSK | Not explicitly stated | II | Assess long-term safety and immunogenicity up to 3 years post-vaccination in HIV+ and HIV- adults. | HIV+ adults (ART-stable and ART-naive) and HIV- adults, aged 18–59 years. | Completed (2011–2015), long-term follow-up completed | [47, 48] | ||
| GSK Vaccines (Belgium), National Taiwan University Hospital (Taiwan), Tartu University Hospital Lung Clinic (Estonia), Far Eastern Memorial Hospital (Taiwan). | GSK Vaccines | Wavre and Rixensart, Belgium. | II | To assess safety, reactogenicity, and immunogenicity of M72/AS01E in adults with or without history of TB treatment. | 3 cohorts: TB-naïve adults, adults previously treated for TB, adults who completed intensive phase of TB treatment. | Terminated prematurely due to high incidence of large local injection site reactions in TB-treatment cohort | [29] | ||
| The commentary does not list the specific implementing institutions for the phase 3 trial. The authors are affiliated with: Victorian Tuberculosis Program, Royal Melbourne Hospital, at the Peter Doherty Institute for Infection and Immunity; Department of Infectious Diseases, University of Melbourne, at the Peter Doherty Institute for Infection and Immunity; Victorian Infectious Disease Service, Royal Melbourne Hospital, at the Peter Doherty Institute for Infection and Immunity. | GSK Vaccines | Not explicitly stated | III | Optimizing the phase III trial of the M72/AS01E TB vaccine candidate based on the results of the phase 2b trial. | NA | NA | [30] | ||
| ID93 + GLA-SE | ID93: Recombinant fusion protein antigen; GLA-SE: TLR4 agonist adjuvant in stable emulsion. | Infectious Disease Research Institute (IDRI), Aeras, University of Washington, Ragon Institute, etc. | IDRI (now AAHI), University of Iowa. | Seattle, WA; Iowa City, IA, USA. | I | To evaluate safety, tolerability, and immunogenicity of ID93 + GLA-SE in healthy adults. | 60 BCG-naïve, QuantIFERON-negative, healthy adults in the US. | Completed | [49] |
| Aeras and the Paul G Allen Family Foundation. | ID93 + GLA-SE was developed and manufactured by the Infectious Disease Research Institute (Seattle, WA, USA). | Seattle, WA, USA. | I | To assess safety and immunogenicity of ID93 + GLA-SE in BCG-vaccinated adults in a TB-endemic country. | 66 HIV-negative, BCG-vaccinated healthy adults (aged 18–50 years). | Completed | [31] | ||
| Saint Louis University Center for Vaccine Development; Infectious Disease Research Institute (IDRI)/Access to Advanced Health Institute (AAHI). | IDRI (now AAHI), University of Iowa, Lyophilization Technology, Inc. | Seattle, WA; Iowa; Warminster, PA, USA. | I | To evaluate the safety, tolerability, and immunogenicity of a thermostable single-vial lyophilized presentation compared to a non-thermostable two-vial presentation in healthy adults. | 93 screened healthy adults, 48 enrolled (18–48 years, predominantly white females). | Completed | [50] | ||
| Three clinical sites near Cape Town, South Africa; funded by Wellcome Trust. | ID93 + GLA-SE was developed and manufactured by the Infectious Disease Research Institute (Seattle, WA, USA). | Seattle, WA, USA. | IIa | To assess safety and immunogenicity of ID93 + GLA-SE as a therapeutic vaccine in patients with previously treated TB. | 61 BCG-vaccinated, HIV-uninfected adults (aged 18–60 years) with recently treated drug-sensitive pulmonary TB in South Africa. | Completed | [32] | ||
| Quratis Inc. (Seoul, Republic of Korea); Seattle Children’s Research Institute; HDT Bio; Yonsei University Severance Hospital, Ajou University Hospital, Chung-Ang University Hospital (clinical sites). | Quratis Inc. | Seoul, Republic of Korea. | II | To evaluate safety and immunogenicity of ID93 + GLA-SE in HIV-negative, previously BCG-vaccinated, QFT-negative healthy adults in South Korea. | 107 healthy, BCG-vaccinated, QFT-negative adult healthcare workers (aged 19–65 years) in South Korea. | Completed | [33] | ||
| H56:IC31 | H56 (recombinant fusion protein of Ag85B, ESAT-6, and Rv2660c) + IC31 adjuvant | University of Cape Town Lung Institute, Aurum Institute, TASK Clinical Research Centre. | Statens Serum Institut (SSI) manufactured the H56 fusion protein, IC31 adjuvant, and placebo. | Copenhagen, Denmark. | I | To evaluate safety and immunogenicity of H56:IC31 in adults recently successfully treated for drug-susceptible pulmonary TB. | Adults aged 18–60 years, HIV-negative, recently completed treatment for drug-susceptible pulmonary TB, confirmed Mtb-negative at end of treatment. | Completed | [34] |
| Emavundleni Clinical Research Site in Crossroads, Cape Town, South Africa. | H56:IC31 provided by Statens Serum Institut (SSI); H4 antigen provided by Sanofi Pasteur, IC31 adjuvant by Valneva. | SSI: Copenhagen, Denmark; Sanofi Pasteur: USA; Valneva: Austria. | Ib | To assess safety and immunogenicity of H4:IC31, H56:IC31, and BCG revaccination; to optimize immune assays for correlates of protection analyses. | 84 healthy, QFT-negative, HIV-uninfected adolescents (aged 12–17 years) in Cape Town, previously BCG-vaccinated at birth. | Completed | [51] | ||
| Oslo University Hospital and Haukeland University Hospital, Norway. | Statens Serum Institut (SSI), Denmark. | Copenhagen, Denmark. | I/II | To assess safety and immunogenicity of H56:IC31 vaccination with or without adjunctive cyclooxygenase-2-inhibitor (etoricoxib) in patients with drug-sensitive TB. | Adults ( |
Completed | [35] | ||
| SATVI, University of Cape Town; Statens Serum Institut (Copenhagen, Denmark); Aeras (Rockville, MD, USA). | Statens Serum Institut (SSI), Denmark. | Copenhagen, Denmark. | I/IIa | To select optimal dose and schedule of H56:IC31 in HIV-uninfected, BCG-vaccinated adults in a TB-endemic setting, stratified by Mtb infection status (QFT-positive/negative). | 98 healthy, HIV-uninfected, BCG-vaccinated South African adults (aged 18–50 years). | Completed | [52] | ||
| Six clinical trial sites: five in South Africa (TASK, SATVI, UCTLI, Aurum Institute in Tembisa and Klerksdorp) and one in Tanzania (NIMR, Mbeya). | Statens Serum Institut (SSI), Denmark. | Copenhagen, Denmark. | IIb | To evaluate the efficacy, safety, and immunogenicity of the H56:IC31 vaccine in reducing the rate of TB disease recurrence in HIV-negative adults successfully treated for drug-susceptible pulmonary TB. | HIV-negative adults (aged 18-60 years) who had completed |
Completed | [36] | ||
| RUTI® | Detoxified and liposomed cellular fragments of Mtb | The study was conducted at the Hospital Universitari “Germans Trias i Pujol” in Badalona, Catalonia, Spain, which served as the investigational centre. | RUTI® was developed under Good Manufacturing Practices (GMP) by Archivel Farma in Badalona (Catalonia, Spain). | Badalona, Catalonia, Spain. | I | To evaluate safety and T-cell immune responses of RUTI® after subcutaneous inoculation in healthy volunteers. | Healthy volunteers (recruited at one site) | Completed | [37] |
| Archivel Farma, Parexel International, Synexa Life Sciences, Janus Developments, ITS Develop, The Aurum Institute, Unitat de Tuberculosis Experimental (UTE). | Archivel Farma | Badalona, Catalonia, Spain. | II | To evaluate safety, tolerability, and immunogenicity of three doses of RUTI vaccine in subjects with LTBI. | HIV-positive and HIV-negative adults (18–50 years) with LTBI. | Completed | [38] | ||
| MIP | Heat-killed Mycobacterium indicus pranii (5 × 108 bacilli per dose) | All India Institute of Medical Sciences (AIIMS), National JALMA Institute, National Institute of TB and Respiratory Diseases, National Institute of Research in TB, SMS Medical College, NHL Municipal Medical College, Mahavir Hospital and Research Centre, RBTB Hospital, Catalyst Clinical Services, Cadila Pharmaceuticals Ltd., Department of Biotechnology, PGIMER, National Institute of Immunology, Chest Clinic and Hospital. | Cadila Pharmaceuticals Ltd. | Ahmadabad, India. | III | To evaluate the safety and efficacy of MIP as an adjunct to Anti-Tubercular Treatment (ATT) in Category II pulmonary TB patients, focusing on sputum culture conversion and cure rate. | Category II pulmonary TB patients (treatment failure, relapse, or default after previous treatment), sputum smear-positive, aged 18–60 years. | Completed | [39] |
| Indian Council of Medical Research (ICMR) – National Institute for Research in TB (NIRT), Chennai; All India Institute of Medical Sciences (AIIMS), Delhi; National Institute of Tuberculosis and Respiratory Diseases (NITRD), Delhi; ICMR-National AIDS Research Institute (NARI), Pune; Regional Medical Research Centre, Bhubaneshwar; National Institute of Tuberculosis (NTI), Bengaluru; Bhagwan Mahavir Medical Research Centre (BMMRC), Hyderabad; St. John Medical College (SJMC), Bengaluru; with multiple subsites across India. | VPM1002 is manufactured by Serum Institute of India; Immuvac (MIP) is manufactured by Cadila Pharmaceuticals. | VPM1002: Pune, India (Serum Institute of India); Immuvac (MIP): Ahmadabad, India (Cadila Pharmaceuticals). | III | To evaluate efficacy and safety of VPM1002 and MIP in preventing microbiologically confirmed TB among healthy household contacts of sputum smear-positive pulmonary TB patients over 3 years; assess immunogenicity via flow cytometry and Luminex assays at months 2 and 6 post-vaccination. | 12,000 healthy household contacts (aged 6–99 years) of newly diagnosed sputum smear-positive pulmonary TB patients across multiple sites in India. | Recruitment completed; follow-up ongoing; results to be disseminated through publications and presentations | [53] | ||
| VPM1002 | Genetically modified Mycobacterium bovis BCG with deletion of urease C gene (ureC) and expression of listeriolysin (Hly) from Listeria monocytogenes | the clinical trial sponsor and research centers involved. | Not explicitly stated | Not explicitly stated | I | To assess safety and immunogenicity of a single dose of VPM1002 in healthy adults, including both BCG-naïve and BCG-immune individuals. | Healthy male volunteers aged 18–55 years, divided into BCG-naïve (n = 40) and BCG-immune (n = 40) groups. | Completed | [40] |
| Stellenbosch University, Vakzine Projekt Management GmbH, HJ Clinical Trials Consultancy, Max Planck Institute for Infection Biology, Desmond Tutu TB Center, Fam-Cru, and other members of the VPM Study Group. | Not explicitly stated | Not explicitly stated | II | To evaluate safety and immunogenicity of VPM1002 compared to BCG in HIV-unexposed newborn infants in a high TB prevalence setting. | Healthy, HIV-unexposed newborn infants ( |
Completed | [41] | ||
| Four health centers in South Africa; sponsored and manufactured by Serum Institute of India. | Serum Institute of India | Pune, India. | II | To compare safety and immunogenicity of VPM1002 versus BCG in HIV-exposed and HIV-unexposed newborn babies. | 416 newborn babies (aged |
Completed | [54] | ||
| NA | NA | NA | III | NA | Newborn infants | Ongoing | [42] |
TB, tuberculosis; HIV, Human Immunodeficiency Virus; MDR-TB, multidrug-resistant TB; MVA, Modified Vaccinia Ankara; LTBI, latent tuberculosis infection; BCG, Bacillus Calmette-Guérin; GSK, GlaxoSmithKline; SATVI, South African Tuberculosis Vaccine Initiative; ART, antiretroviral therapy; GLA-SE, Glucopyranosyl Lipid Adjuvant - Stable Emulsion; QFT, QuantiFERON-TB; MIP, Mycobacterium indicus pranii/ Mycobacterium w (Mw).
MVA85A employs a highly attenuated modified vaccinia Ankara (MVA) virus vector expressing the principal Mtb antigen, Ag85A. Its underlying mechanism leverages the robust immunogenicity of MVA to augment T-cell immune responses against Ag85A, aiming to control or eradicate latent bacteria, as shown in Fig. 3 and Table 1. Preclinical studies indicate its potential to elicit potent antigen-specific CD4+ and CD8+ T-cell responses. Nevertheless, a Phase IIb clinical trial investigating MVA85A as a preventive vaccine demonstrated good tolerability and moderate immunogenicity but failed to show significant efficacy against TB or Mtb infection [24]. Currently, due to its ability to elicit highly persistent T-cell responses in immunocompetent subjects, the developmental focus has shifted toward therapeutic applications [56]. Investigations are ongoing into its combination with antibiotics to reduce treatment duration and prevent relapse. Concurrent clinical studies are evaluating its safety and immune adjuvant potential. For instance, in a Phase I trial, aerosol administration of MVA85A showed good efficacy and safety in two volunteers with latent TB infection, inducing a specific immune response [25].
In the new research area of MVA85A, the focus has expanded from single vaccines to multi-strategy combined immunization programs and molecular adjuvant fusion vaccines, with a clear chronological development and iterative strategies. In 2016, the research first explored the fusion vaccine of MVA85A and the molecular adjuvant IMX313 (MVA85A - IMX313). A Phase I trial was conducted on BCG - vaccinated adults in the UK. Although it was confirmed that the safety was comparable to that of the parental MVA85A and it could induce specific antibodies, it failed to significantly enhance the cellular immune response. This was an initial exploration of enhancing the efficacy of MVA85A through protein engineering modification [26]. In 2020, the research shifted to the heterologous prime - boost strategy. A Phase I trial showed that the prime - boost regimen of ChAdOx185A priming followed by MVA85A boosting could safely induce a stronger and broader immune response, verifying the crucial role of MVA85A as a booster [27]. In 2024, the key Phase IIa trial indicated that in adolescents in Uganda, the ChAdOx185A–MVA85A regimen had good safety and significantly superior immunogenicity compared to BCG revaccination [28]. Overall, the research results of MVA85A have laid a solid clinical evidence chain for it to become the next - generation TB booster vaccine.
M72/AS01E consists of the Mtb antigen M72 (a fusion protein) and GlaxoSmithKline’s AS01 adjuvant system. AS01 is a liposomal adjuvant containing MPL and QS-21 that enhances Th1-type cellular immune responses. The vaccine is designed to prevent TB resulting from reactivation of latent infection or new infection by inducing antigen-specific cellular immunity, as shown in Fig. 3 and Table 1. Its primary clinical objective is prophylaxis. However, it has also demonstrated therapeutic potential. Current status: Phase II clinical trials have shown acceptable safety and strong immunogenicity in latently infected individuals, with approximately 50% efficacy against pulmonary TB in IGRA-positive adults, indicating a meaningful efficacy signal [29]. A Phase III trial plans to enroll approximately 18,000 IGRA-positive participants and will focus on efficacy in high-burden settings [30]. Although primarily intended for disease prevention, this vaccine also shows potential therapeutic utility, possibly facilitating latent infection clearance through enhanced immune regulation.
ID93 + GLA-SE comprises a fusion protein of four Mtb antigens combined with the TLR4 agonist adjuvant glucopyranosyl lipid adjuvant–stable emulsion (GLA-SE). This formulation is designed to provide protection against both latent infection and active TB. The TLR4 agonist is included to strengthen immune responses, as shown in Fig. 3 and Table 1.
Preclinical studies indicate that the vaccine induces durable and robust
Th1-type cellular immunity in murine models. This includes antigen-specific
multifunctional CD4+ T cells that secrete IFN-
H56:IC31 comprises three Mtb antigens (Ag85B, ESAT-6, and Rv2660c) combined with the adjuvant IC31. It is designed to provide comprehensive immune protection against active bacterial replication and latent TB infection, as shown in Fig. 3 and Table 1. The clinical objective is to prevent recurrence of pulmonary TB after treatment completion. Current status: As a therapeutic vaccine, Phase I/IIa clinical trials demonstrated acceptable safety and good tolerability among adults recently cured of drug-sensitive pulmonary TB. It effectively elicited an immune response predominantly characterized by antigen-specific CD4+ T cells. These findings confirm its significant immunogenicity and support its potential use as a therapeutic vaccine [34, 35]. Further transcriptomic studies have shown that adjunctive vaccination during standard treatment can trigger significant early gene expression perturbations, including the upregulation of genes related to the interferon signaling pathway, pattern recognition receptors, and small GTPases. This suggests that the vaccine elicits extensive innate and adaptive immune responses. Notably, the research also found that, two months after the end of treatment, the expression levels of ACS - COR transcriptional signature genes associated with the progression and recurrence risk of TB were significantly lower in vaccine recipients (especially the “responders” who generated potent cellular and humoral immunity) than in the non - vaccinated control group. This indicates at the molecular level that the vaccine may help consolidate the therapeutic effect and potentially reduce the risk of recurrence [58]. However, in terms of preventing recurrence, a Phase IIb clinical trial showed that vaccination with H56:IC31 following pulmonary TB treatment did not reduce the risk of disease recurrence [36].
RUTI® is a liposome-encapsulated vaccine composed of inactivated
Mtb fragments, primarily designed as an adjunct immunotherapy in LTBI
treatment to shorten therapy duration and enhance therapeutic efficacy [20]. Its
mechanism involves delivering multiple antigens to stimulate robust Th1-type
cellular immune responses, notably the production of IFN-
Preclinical studies in murine and guinea pig models have demonstrated the prophylactic and therapeutic potential of RUTI® against TB [59]. Phase I clinical trials confirmed its safety and immunogenicity [37]. Phase II trials reported acceptable safety and tolerability in LTBI subjects, with localized injection-site reactions being the primary adverse event. The vaccine induced strong multi-antigen-specific T-cell responses, particularly in HIV-positive populations. Notably, a single 25 µg dose triggered a potent immune response without requiring a booster. These findings support subsequent clinical trials utilizing a single-dose administration at higher doses, subject to expanded safety evaluation [38].
An example is Mycobacterium indicus pranii (MIP), a non-tuberculous mycobacterium that exerts therapeutic effects through non-specific immune stimulation [20]. Its clinical objective is to function as an immunomodulator/immunotherapy to assist TB treatment in combination with anti-TB drugs, as shown in Fig. 3 and Table 1. In clinical applications, India’s National Leprosy Eradication Programme (NLEP) integrated the MIP vaccine for immunotherapy during its pilot phase in 2016 [60]. According to the “National Leprosy Strategic Plan and Roadmap (2023–2027)”, MIP is recognized as a safe and effective immunoprophylactic agent against leprosy. It is administered to contacts of index cases, with subsequent monitoring. When combined with multidrug therapy (MDT), MIP accelerates bacterial and granuloma clearance, reducing reaction severity and incidence. Over the past four decades, MIP has been evaluated extensively across tertiary care centers and field settings, gaining broad patient acceptance due to favorable outcomes [61]. In patients with Category II pulmonary TB, MIP is used as an adjunctive immunotherapy alongside anti-TB medications. It significantly accelerates sputum culture conversion, particularly in refractory cases characterized by drug resistance, a high bacterial load, or bilateral cavitary lesions [39]. Additional applications include treatments for anogenital warts and psoriasis [62].
VPM1002 is a recombinant BCG strain engineered by introducing two genes (urease C and listeriolysin O) into the original BCG strain [63, 64]. Its primary aim is prevention, but it also holds potential as a therapeutic vaccine, as shown in Fig. 3 and Table 1. Phase I clinical trials have demonstrated its safety and immunogenicity in adults [40]. Phase II trials have shown that VPM1002 has improved safety compared to BCG in newborn populations [41]. Currently, the vaccine is undergoing Phase III clinical trials targeting newborn infants [42].
In preclinical studies using mouse models, mRNA vaccines have demonstrated protective efficacy against TB [65]. Currently, these vaccines are still at the preclinical research stage and have not yet entered human clinical trials as shown in Table 1. Furthermore, bacterial vector vaccines have also been investigated as potential therapeutic vaccines [66].
TB bacteria manifest in multiple states, including latent and active phases [67]. Scientists face the critical challenge of identifying optimal antigen combinations for these distinct disease states. Existing animal models (mice and guinea pigs) cannot fully replicate the pathological processes and immune responses of human TB. Consequently, vaccines with promising outcomes in animal studies often fail to achieve satisfactory efficacy in clinical trials.
Selecting appropriate endpoint indicators poses multiple challenges, including microbiological conversion, clinical efficacy, and immunological markers. Each indicator has distinct strengths and limitations. The patient population is heterogeneous, including both drug-sensitive and MDR-TB patients, with varying treatment requirements and responses [68]. Additionally, the timing of immunotherapy integration with chemotherapy regimens requires careful consideration. Initiating immunotherapy either too early or too late may compromise treatment outcomes.
Inducing a robust immune response in infected individuals may involve risks, such as immunopathological damage. Therefore, it is essential to precisely regulate the intensity and direction of immune responses during treatment. An excessively activated immune system may cause inflammation in the lungs and other organs, potentially worsening the patient’s condition rather than improving it. Immunotherapy, as part of combination therapy, follows different approval and marketing pathways compared to traditional drugs or vaccines. Thus, new regulatory standards and procedures are necessary to ensure its safety and efficacy.
Developing personalized vaccination strategies tailored to distinct disease stages (latent infection, active disease, and MDR-TB) could more accurately meet patient needs. Exploring novel adjuvant systems, such as TLR agonists and STING agonists, may optimize immune response quality and improve therapeutic efficacy. Conducting biomarker-based clinical trials to identify predictive immunological correlates of treatment efficacy would enable early assessment of therapeutic outcomes. Promoting combined immunotherapeutic approaches, such as integrating vaccines with immune checkpoint inhibitors, could generate synergistic effects. Strengthening global collaboration to conduct pivotal clinical trials in high TB-burden countries would broaden patient access and accelerate the global implementation of immunotherapy.
As an adjunctive intervention to pharmacological treatments for TB, therapeutic vaccines have significant potential to reduce treatment duration, overcome drug resistance, and eliminate latent infections. They are poised to transform the TB treatment paradigm from the traditional antibiotic-only approach toward a novel era of antibiotic-immune synergy. Currently, numerous vaccine candidates, based on diverse platforms and utilizing various antigens, are undergoing clinical evaluation.
Despite notable challenges in their development and application, including complex immune mechanisms and individual variability, several therapeutic vaccine candidates, including RUTI®, have shown favorable safety profiles and promising signs of immunogenicity and efficacy. This indicates substantial potential for further advancement of therapeutic vaccines in TB treatment.
Looking forward, the success of therapeutic vaccines will rely on interdisciplinary cooperation, innovative trial designs, and sustained funding. Integrating expertise from various disciplines will provide a more comprehensive perspective on vaccine development. Innovative trial designs may accelerate the optimization process, while strong financial support remains essential for ongoing research. A breakthrough in this area could establish therapeutic vaccines as a critical tool for achieving the ambitious goal of ending the TB epidemic, offering new hope to patients globally.
SLiu, QZ, and SLin designed the research. FY and JL developed the overall concept. QY integrated and refined the key highlights. JP conducted the literature search and drafted the manuscript. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by Natural Science Foundation of Guangdong Province (2023A1515140072), Science and Technology Project of Dongguan (20231800940512), College Student Innovation and Entrepreneurship Training Program Project (202510571027, S202510571083), Undergraduate Innovation and Entrepreneurship Education Base Project of Guangdong Medical University (JDXM2024083F, JDXM2024067, JDXM2024121), Special Project for Clinical and Basic Sci & Tech Innovation of Guangdong Medical University (GDMULCJC2025161).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



