1 Clinical Laboratory, Haikou Affiliated Hospital of Central South University XiangYa School of Medicine, 270208 Haikou, Hainan, China
Abstract
Hepatic fibrosis (HF) is a major global health burden with limited effective therapies. Ferroptosis, an iron-dependent form of cell death, and inflammatory pathways are implicated in HF progression. Salvia miltiorrhiza (Danshen) has demonstrated antifibrotic effects, but its underlying mechanisms remain unclear.
To elucidate the mechanisms of Danshen in treating HF through network pharmacology and experimental validation, focusing on the JAK/STAT pathway, iron metabolism, and ferroptosis.
Using the Traditional Chinese Medicine Systems Pharmacology (TCMSP) database, we identified 92 common targets among Danshen components, HF-related genes, and iron metabolism genes. A CCl4-induced HF rat model was established (n = 70) and treated with Danshen (3 or 6 g/kg/d) for 4 weeks. Hepatic fibrosis was assessed by Masson staining. Serum markers including Fe2+, transferrin (TRF), ferritin (SF), type III procollagen (PCIII), hydroxyproline (HyP), malondialdehyde (MDA), and glutathione (GSH) were measured. Western blotting evaluated hepatic expression of IL-6, P-JAK2, p-STAT3, and GPX4.
Network pharmacology revealed JAK2/STAT3 as top hub genes among 92 intersection targets. High-dose Danshen decreased IL-6, P-JAK2, and p-STAT3 expression by 45–60% (p < 0.05), increased serum Fe2+ and TRF by 1.8-fold, and reduced SF, HyP by >40% and PCIII by >15% (p < 0.05). Additionally, Danshen restored GPX4 protein expression and suppressed ferroptosis by decreasing MDA while increasing GSH (p < 0.01). Masson staining showed 55% reduction in collagen deposition with high-dose treatment.
Danshen alleviates hepatic fibrosis by modulating the JAK/STAT-iron-ferroptosis axis, establishing a novel multi-target therapeutic strategy for HF.
Graphical Abstract
Keywords
- Danshen
- liver fibrosis ferroptosis
- iron metabolism disorders
- network pharmacology
Hepatic fibrosis (HF) is a disease characterized by abnormal activation of hepatic stellate cells, an imbalance between the synthesis and degradation of extracellular matrix proteins, and excessive deposition that disrupts the normal structure of the liver resulting from various chronic liver injuries [1]. The global incidence rate of HF is between 4.5% and 9.0%. Liver injury first occurs during the progression of HF [2]. As injury accumulates, the body initially activates pro-inflammatory mechanisms. With the onset of inflammatory responses, the normal structure and physiological functions of liver tissue are gradually disrupted, leading to scar tissue formation and fibrosis [3]. As the degree of HF worsens, it can progress to irreversible cirrhosis and hepatocellular carcinoma, posing a significant challenge to global health [4].
Ferroptosis is a newly discovered form of iron-dependent cell death that has been studied in recent years, primarily involving abnormal iron ion metabolism, accumulation of lipid peroxides, and weakening of the antioxidant defense system. Numerous studies have indicated that inducing ferroptosis in hepatic stellate cells (HSCs) or alleviating ferroptosis in the liver can improve HF and reduce liver damage [5]. The central link in HF is the activation of HSCs; other liver cells also influence HSC activation [6]. In the pathological states of liver injury, ferroptosis affects multiple liver cell types and accelerates the progression of HF [7].
Currently, there is a lack of effective drugs for the treatment of HF in clinical practice. With the widespread application of traditional Chinese medicine (TCM) in various chronic liver diseases, its advantages such as fewer side effects, high safety, and the unique characteristics of being comprehensive and multi-targeted have gradually emerged [8]. Compared with Western medicine, TCM has unique advantages in the treatment of chronic diseases and can effectively reverse HF. However, the specific mechanisms of action are not yet fully understood and require further exploration [9]. Therefore, researching effective new diagnostic and treatment methods for HF is significant and of economic value.
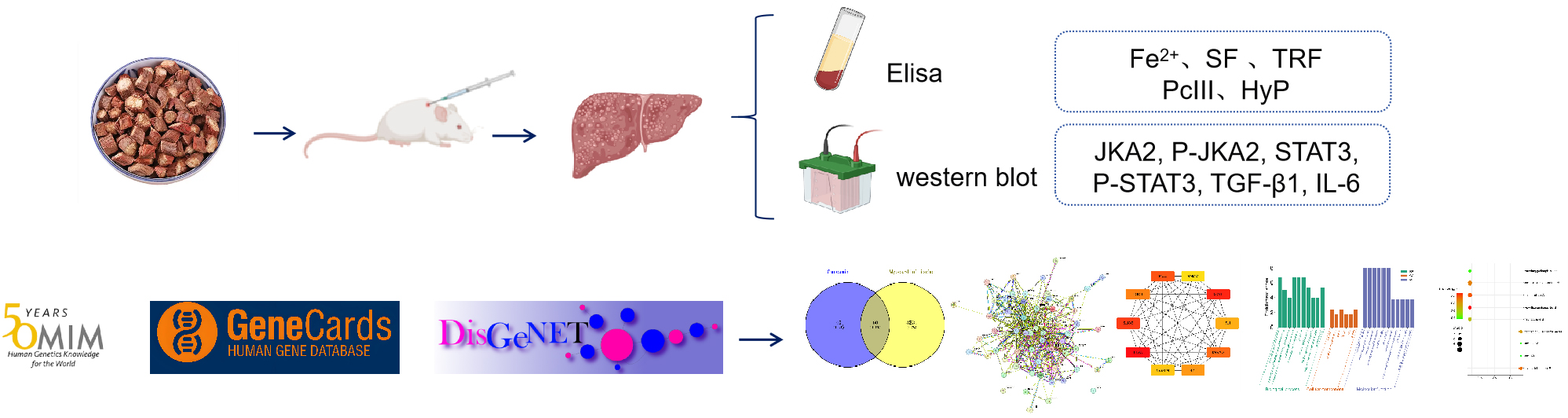
Salvia miltiorrhiza, commonly known as Danshen, is known for its effects and functions such as promoting blood circulation to remove blood stasis, reducing liver inflammation, inhibiting the proliferation of hepatic stellate cells, improving liver function, and anti-HF [10]. This study aimed to construct a rat model of HF by injecting a mixture of carbon tetrachloride and vegetable oil into the main limbs of rats to provide scientific evidence for the treatment of HF with Danshen. Additionally, this study will combine network pharmacology methods to identify the main active components of Danshen and further analyze its potential targets and mechanisms in anti-HF (Fig. 1).
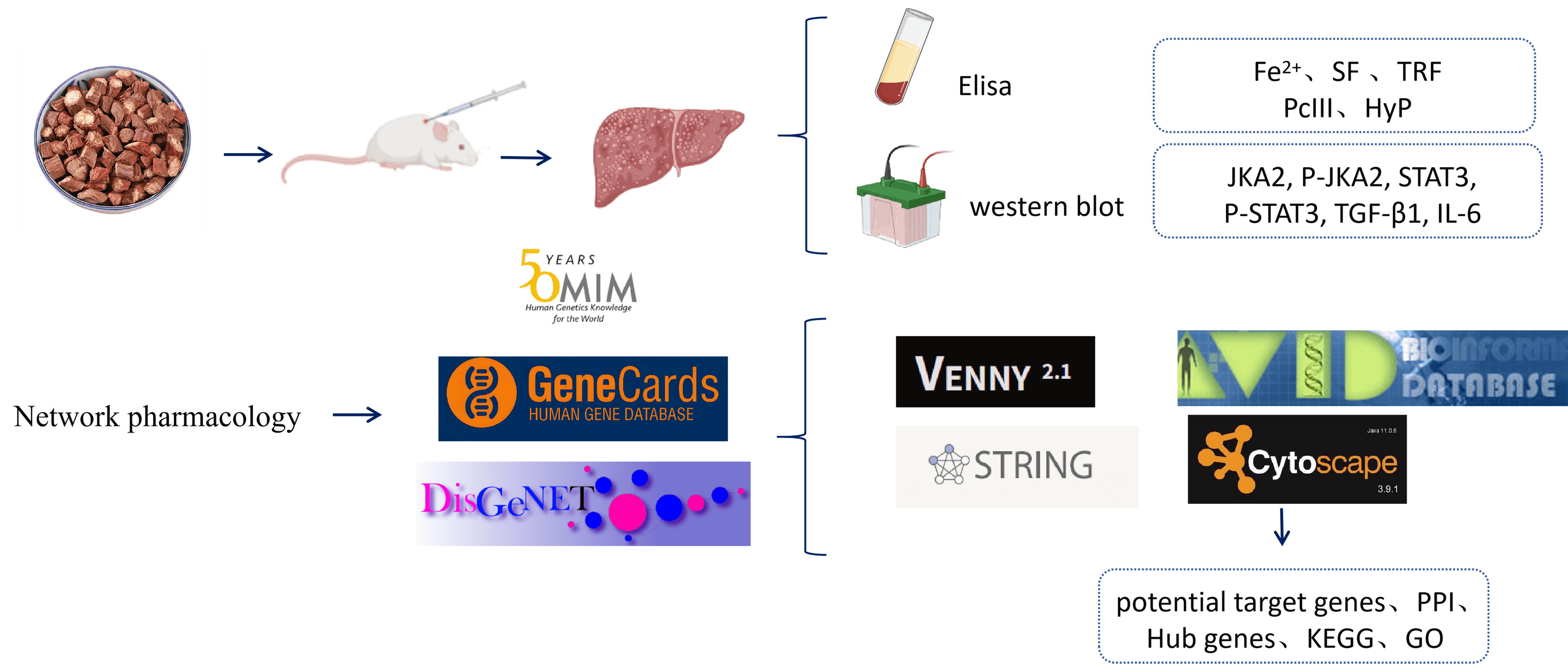 Fig. 1.
Fig. 1.
Detailed design flow chart of the current study. Created in BioRender. https://BioRender.com/al17dt9.
Sibe Biotechnology Co., Ltd. (Beijing) provided 70 SPF-grade male Sprague-Dawley
(SD) rats, weighing (200
Danshen was purchased from Guangdong Yifang Pharmaceutical Co., Ltd. (Foshan,
Guangdong, China), Masson (Macklin-M774677), and paraformaldehyde
(Macklin-P885233) were purchased from Shanghai Macklin Biochemical Technology
Co., Ltd. (Shanghai, China);
After a 1-week acclimatization period, 70 male SD rats were randomly assigned to
four groups using a random number table: a normal control group with 10 rats, a
model group with 20 rats, and two Danshen treatment groups with 20 rats each
receiving low (3 g
Search for “Danshen” in the Traditional Chinese Medicine Systems Pharmacology
Database (TCMSP, https://tcmsp-e.com/), and set the Absorption, Distribution,
Metabolism, Excretion (ADME) parameters with OB
Using “HF” and “iron metabolism” as search terms, disease targets were identified using the GeneCards database (https://www.genecards.org/), and Venn diagrams were used to identify the common targets of the two diseases.
Translate the main pharmacological components, targets, and corresponding target genes related to diseases of Danshen into Venny 2.1 (http://bioinfogp.cnb.csic.es/tools/venny/index.html), which are potential target genes for Danshen in the treatment of HF and iron metabolism.
Import the obtained common target genes into the STRING database, select the species “Homo sapiens”, and set the minimum interaction score to the highest confidence level of 0.40. We then obtained an overlapping gene Protein-Protein Interaction (PPI) network diagram. The PPI network diagram was imported into Cytoscape 3.9.1 software (San Francisco, CA, USA), and the maximum clique centrality (MCC) algorithm in the cytoHubba plugin was used to identify the top ten most significant hub genes.
Utilize The DAVID database (http://david.abcc.ncifcrf.gov) was used to perform Gene Ontology (GO) functional analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment for the common targets of Danshen in the treatment of HF and iron metabolism, thereby obtaining biological processes (BP), cellular components (CC), molecular functions (MF), and signaling pathways that are closely associated with the therapeutic effects of Danshen on HF and iron metabolism.
Liver tissues were taken from each group of rats, fixed with formaldehyde, and paraffin sections were prepared. The sections were dewaxed routinely. The sections were stained with hematoxylin for 10 min. The mixture was rinsed with distilled water three times. The sections were stained with Masson’s trichrome and picric acid for 30 min. Differentiate and dehydrate directly with anhydrous alcohol and mount with neutral resin. Images were obtained under a microscope to assess the degree of HF.
Rat liver tissue was used to detect the protein expression of IL-6, JAK, and
STAT by western blotting. Lysis buffer was added to extract proteins,
homogenized, and centrifuged, and the total protein concentration was measured
with the BCA method, boiled, loaded samples, transferred to a membrane, and
blocked; diluted mouse-sourced IL-6 (1:500), JAK (1:500), STAT3 (1:500), and
Orbital venous blood was collected from the rats, centrifuged at 3000 rpm for 10–15 minutes to separate the serum, and then centrifuged again at 3000 rpm for 5 min. Fe2+, TRF, and ferritin levels were detected according to the instructions of the ELISA kit.
Graph Pad Prism software (version 9.0, Boston, MA, USA) was used for the data
processing. All data were expressed as mean
By querying the TCMSP database, we obtained 20 pharmacologically active compounds from Danshen and their corresponding 188 targets. Information regarding the main pharmacologically active compounds is presented in Table 1.
| MOL ID | NAME | OB | DL |
| MOL001601 | 1,2,5,6-tetrahydrotanshinone | 38.75 | 0.36 |
| MOL001659 | Poriferasterol | 43.83 | 0.76 |
| MOL001771 | poriferast-5-en-3beta-ol | 36.91 | 0.75 |
| MOL001942 | isoimperatorin | 45.46 | 0.23 |
| MOL002222 | sugiol | 36.11 | 0.28 |
| MOL002651 | Dehydrotanshinone II A | 43.76 | 0.4 |
| MOL002776 | Baicalin | 40.12 | 0.75 |
| MOL000569 | digallate | 61.85 | 0.26 |
| MOL000006 | luteolin | 36.16 | 0.25 |
| MOL006824 | alpha-amyrin | 39.51 | 0.76 |
| MOL007036 | 5,6-dihydroxy-7-isopropyl-1,1-dimethyl-2,3-dihydrophenanthren-4-one | 33.77 | 0.29 |
| MOL007041 | 2-isopropyl-8-methylphenanthrene-3,4-dione | 40.86 | 0.23 |
| MOL007045 | 3 |
44.93 | 0.44 |
| MOL007048 | (E)-3-[2-(3,4-dihydroxyphenyl)-7-hydroxy-benzofuran-4-yl]acrylic acid | 48.24 | 0.31 |
| MOL007049 | 4-methylenemiltirone | 34.35 | 0.23 |
| MOL007050 | 2-(4-hydroxy-3-methoxyphenyl)-5-(3-hydroxypropyl)-7-methoxy-3-benzofurancarboxaldehyde | 62.78 | 0.4 |
| MOL007051 | 6-o-syringyl-8-o-acetyl shanzhiside methyl ester | 46.69 | 0.71 |
| MOL007058 | formyltanshinone | 73.44 | 0.42 |
| MOL007059 | 3-beta-Hydroxymethyllenetanshiquinone | 32.16 | 0.41 |
| MOL007061 | Methylenetanshinquinone | 37.07 | 0.36 |
| MOL007063 | przewalskin a | 37.11 | 0.65 |
| MOL007064 | przewalskin b | 110.32 | 0.44 |
| MOL007068 | Przewaquinone B | 62.24 | 0.41 |
| MOL007069 | przewaquinone c | 55.74 | 0.4 |
| MOL007070 | (6S,7R)-6,7-dihydroxy-1,6-dimethyl-8,9-dihydro-7H-naphtho[8,7-g]benzofuran-10,11-dione | 41.31 | 0.45 |
| MOL007071 | przewaquinone f | 40.31 | 0.46 |
| MOL007077 | sclareol | 43.67 | 0.21 |
| MOL007079 | tanshinaldehyde | 52.47 | 0.45 |
| MOL007081 | Danshenol B | 57.95 | 0.56 |
| MOL007082 | Danshenol A | 56.97 | 0.52 |
| MOL007085 | Salvilenone | 30.38 | 0.38 |
| MOL007088 | cryptotanshinone | 52.34 | 0.4 |
| MOL007093 | dan-shexinkum d | 38.88 | 0.55 |
| MOL007094 | danshenspiroketallactone | 50.43 | 0.31 |
| MOL007098 | deoxyneocryptotanshinone | 49.4 | 0.29 |
| MOL007100 | dihydrotanshinlactone | 38.68 | 0.32 |
| MOL007101 | dihydrotanshinoneI | 45.04 | 0.36 |
| MOL007105 | epidanshenspiroketallactone | 68.27 | 0.31 |
| MOL007107 | C09092 | 36.07 | 0.25 |
| MOL007108 | isocryptotanshi-none | 54.98 | 0.39 |
| MOL007111 | Isotanshinone II | 49.92 | 0.4 |
| MOL007115 | manool | 45.04 | 0.2 |
| MOL007118 | microstegiol | 39.61 | 0.28 |
| MOL007119 | miltionone I | 49.68 | 0.32 |
| MOL007120 | miltionone II | 71.03 | 0.44 |
| MOL007121 | miltipolone | 36.56 | 0.37 |
| MOL007122 | Miltirone | 38.76 | 0.25 |
| MOL007123 | miltirone II | 44.95 | 0.24 |
| MOL007124 | neocryptotanshinone ii | 39.46 | 0.23 |
| MOL007125 | neocryptotanshinone | 52.49 | 0.32 |
| MOL007127 | 1-methyl-8,9-dihydro-7H-naphtho[5,6-g]benzofuran-6,10,11-trione | 34.72 | 0.37 |
| MOL007130 | prolithospermic acid | 64.37 | 0.31 |
| MOL007132 | (2R)-3-(3,4-dihydroxyphenyl)-2-[(Z)-3-(3,4-dihydroxyphenyl)acryloyl]oxy-propionic acid | 109.38 | 0.35 |
| MOL007140 | (Z)-3-[2-[(E)-2-(3,4-dihydroxyphenyl)vinyl]-3,4-dihydroxy-phenyl]acrylic acid | 88.54 | 0.26 |
| MOL007141 | salvianolic acid g | 45.56 | 0.61 |
| MOL007142 | salvianolic acid j | 43.38 | 0.72 |
| MOL007143 | salvilenone I | 32.43 | 0.23 |
| MOL007145 | salviolone | 31.72 | 0.24 |
| MOL007149 | NSC 122421 | 34.49 | 0.28 |
| MOL007150 | (6S)-6-hydroxy-1-methyl-6-methylol-8,9-dihydro-7H-naphtho[8,7-g]benzofuran-10,11-quinone | 75.39 | 0.46 |
| MOL007151 | Tanshindiol B | 42.67 | 0.45 |
| MOL007152 | Przewaquinone E | 42.85 | 0.45 |
| MOL007154 | tanshinone iia | 49.89 | 0.4 |
| MOL007155 | (6S)-6-(hydroxymethyl)-1,6-dimethyl-8,9-dihydro-7H-naphtho[8,7-g]benzofuran-10,11-dione | 65.26 | 0.45 |
| MOL007156 | tanshinone VI | 45.64 | 0.3 |
Notes: OB, Oral Bioavailability; DL, Drug-Likeness; ADME, Absorption, Distribution, Metabolism, Excretion.
After mapping the targets corresponding to Danshen’s active compounds, targets corresponding to HF, and targets corresponding to iron metabolism using a Venn diagram, 92 common targets were identified (Fig. 2).
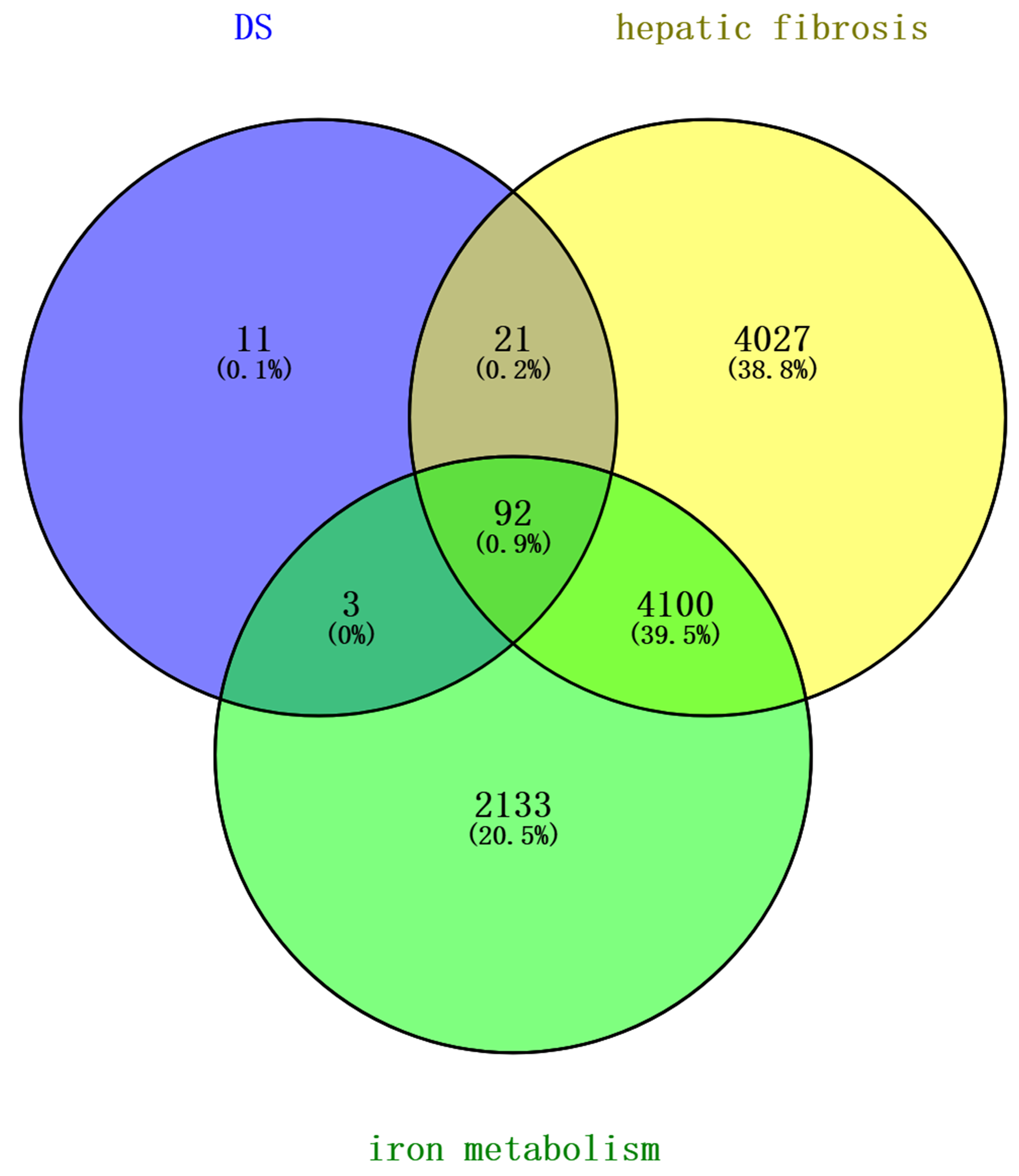 Fig. 2.
Fig. 2.
Venn diagram of targets related to Danshen and HF and iron metabolism. HF, Hepatic fibrosis.
The interaction data of the common targets for Danshen’s treatment of HF and iron metabolism in the STRING database were used to obtain the overlapping gene PPI network diagram (Fig. 3). There were 92 nodes and 1543 edges. The PPI network diagram into Cytoscape 3.9.1 software and the MCC algorithm in the cytoHubba plugin was used to identify the top 10 most significant hub genes (Fig. 4).
 Fig. 3.
Fig. 3.
PPI network of Danshen in the treatment of HF and iron metabolism. PPI, Protein-Protein Interaction.
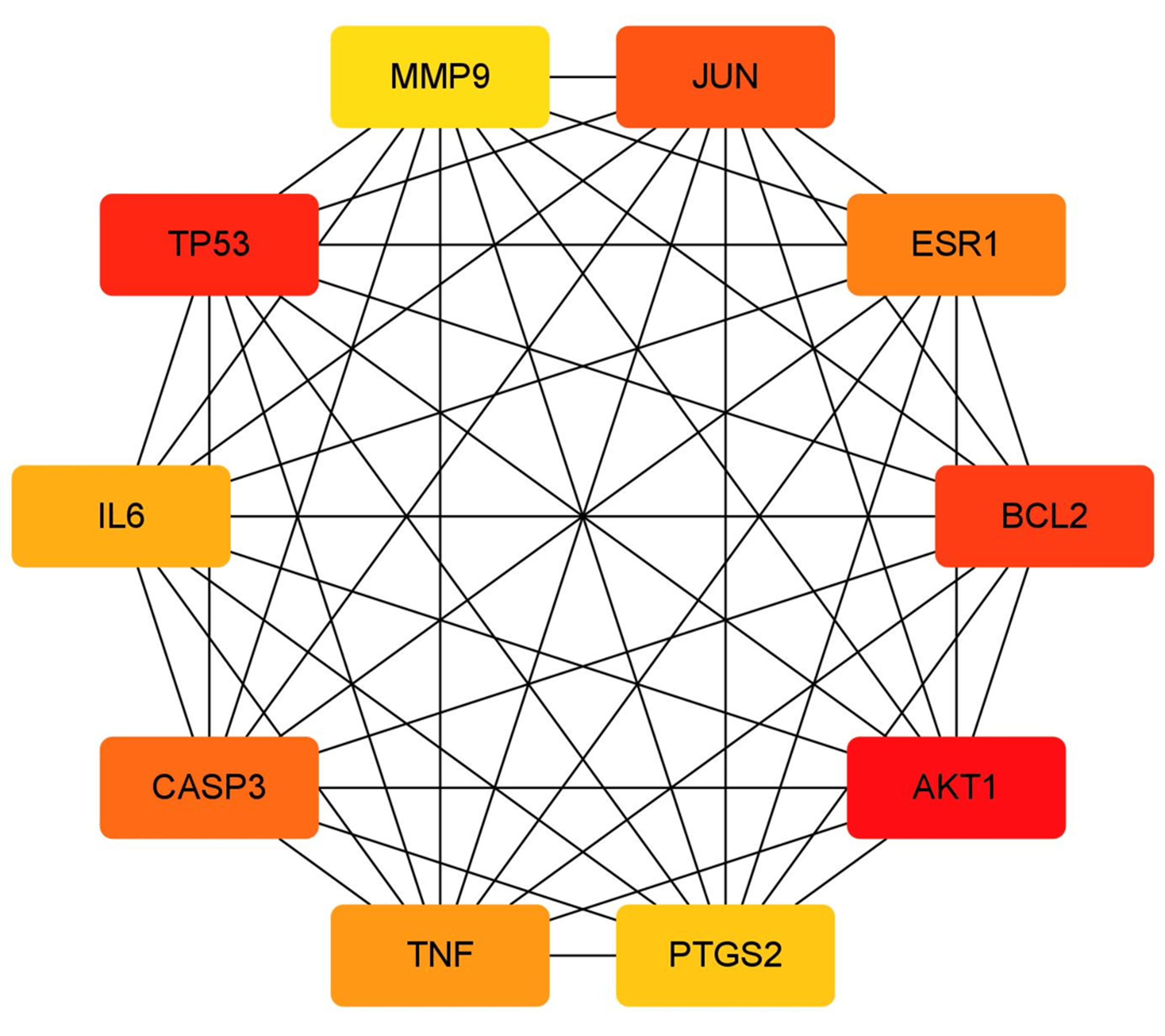 Fig. 4.
Fig. 4.
Top 10 hub genes in the treatment of HF and iron metabolism by Danshen.
The DAVID database was used to perform GO functional analysis of the common targets of Danshen in the treatment of HF and iron metabolism. In terms of BP (Biological Process), select the top 10 ranked entries, which mainly involve cellular response to glucocorticoids, cellular response to nicotine, and extrinsic apoptotic signaling pathway in the absence of ligand. For CC (Cellular Component), select the top 10 ranked entries, which mainly include nucleoplasm, nucleus, and receptor complexes. For MF (Molecular Function), select the top 10 ranked entries, which mainly involve nuclear receptor activity, estrogen response element binding, and steroid binding, among others. The above results were input into the Bioinformatics website (http://www.bioinformatics.com.cn), and bubble charts for BP, CC, and MF were generated (see Fig. 5, Table 2).
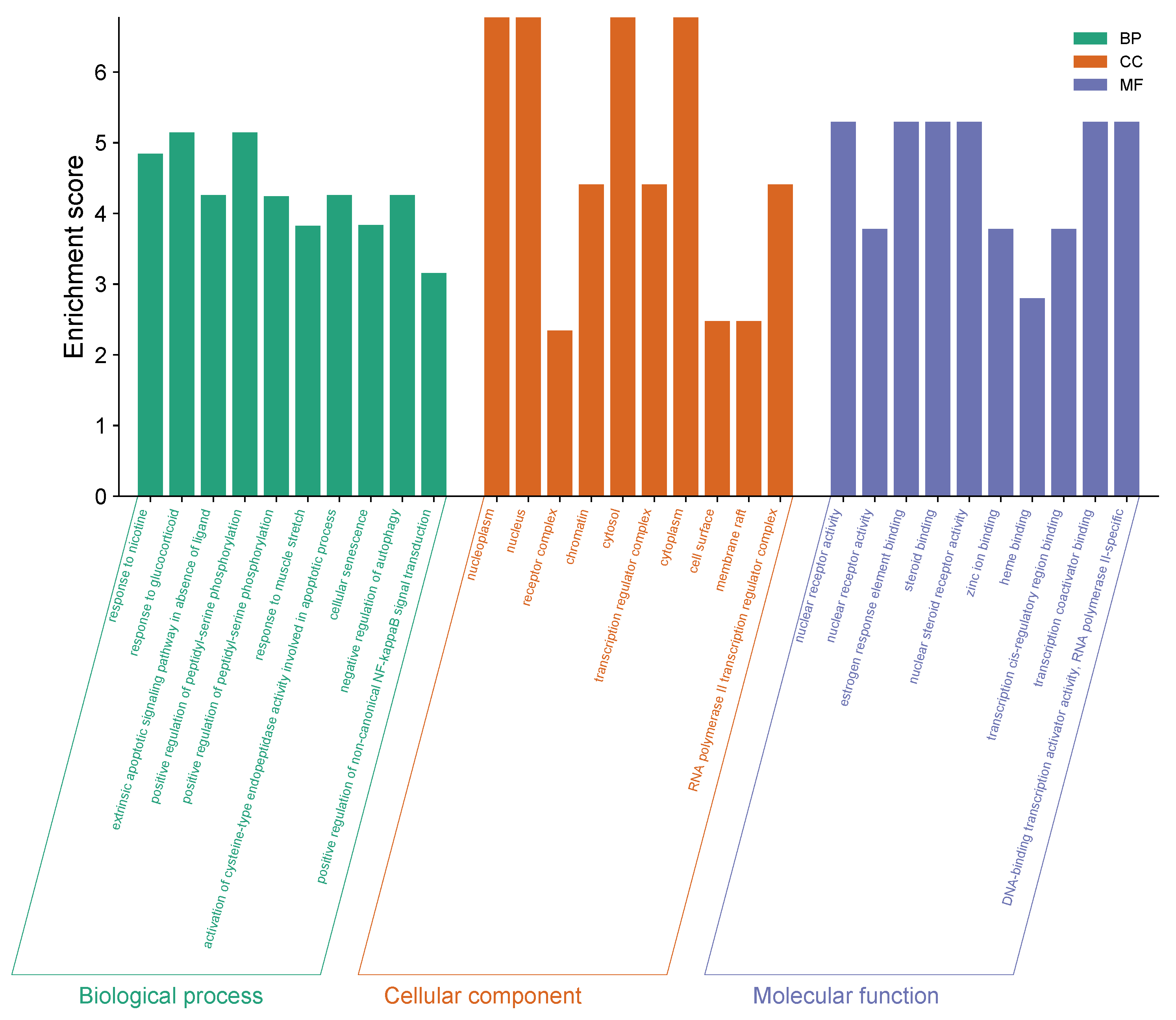 Fig. 5.
Fig. 5.
GO functional enrichment bar chart for Danshen for the treatment of HF and iron metabolism. GO, Gene Ontology.
| Types | Term | Count | Enrichment Score | p value |
| BP | response to nicotine | 7 | 4.844933797 | 3.73 × 10−8 |
| BP | response to glucocorticoid | 7 | 5.145466488 | 1.10 × 10−7 |
| BP | extrinsic apoptotic signaling pathway in absence of ligand | 6 | 4.257211096 | 6.28 × 10−7 |
| BP | positive regulation of peptidyl-serine phosphorylation | 7 | 5.145466488 | 1.08 × 10−6 |
| BP | response to muscle stretch | 5 | 3.822972582 | 1.56 × 10−6 |
| BP | activation of cysteine-type endopeptidase activity involved in apoptotic process | 7 | 4.257211096 | 1.73 × 10−6 |
| BP | cellular senescence | 6 | 3.83324354 | 6.47 × 10−6 |
| BP | negative regulation of autophagy | 6 | 4.257211096 | 9.06 × 10−6 |
| BP | positive regulation of non-canonical NF-kappaB signal transduction | 6 | 3.153129541 | 1.44 × 10−5 |
| CC | nucleoplasm | 45 | 6.770523464 | 3.58 × 10−10 |
| CC | nucleus | 54 | 6.770523464 | 1.16 × 10−8 |
| CC | receptor complex | 10 | 2.339313467 | 6.07 × 10−7 |
| CC | chromatin | 19 | 4.410314366 | 2.10 × 10−6 |
| CC | cytosol | 46 | 6.770523464 | 4.27 × 10−6 |
| CC | transcription regulator complex | 9 | 4.410314366 | 1.21 × 10−5 |
| CC | cytoplasm | 44 | 6.770523464 | 4.68 × 10−5 |
| CC | cell surface | 12 | 2.47850866 | 1.32 × 10−4 |
| CC | membrane raft | 6 | 2.47850866 | 0.002309073 |
| CC | RNA polymerase II transcription regulator complex | 5 | 4.410314366 | 0.00231675 |
| MF | nuclear receptor activity | 9 | 5.295334913 | 1.55 × 10−10 |
| MF | nuclear receptor activity | 9 | 3.779701442 | 1.55 × 10−10 |
| MF | estrogen response element binding | 6 | 5.295334913 | 1.61 × 10−9 |
| MF | steroid binding | 7 | 5.295334913 | 6.22 × 10−9 |
| MF | nuclear steroid receptor activity | 5 | 5.295334913 | 3.82 × 10−6 |
| MF | zinc ion binding | 17 | 3.779701442 | 4.02 × 10−6 |
| MF | heme binding | 8 | 2.797217185 | 8.65 × 10−6 |
| MF | transcription cis-regulatory region binding | 9 | 3.779701442 | 2.00 × 10−5 |
| MF | transcription coactivator binding | 5 | 5.295334913 | 7.67 × 10−5 |
| MF | DNA-binding transcription activator activity, RNA polymerase II-specific | 11 | 5.295334913 | 1.09 × 10−4 |
GO, Gene Ontology; BP, biological processes; CC, cellular components; MF, molecular functions.
The DAVID database was used to perform KEGG functional enrichment analysis of
the common targets of Danshen in the treatment of HF and iron metabolism. The top
20 most significantly enriched pathways (p
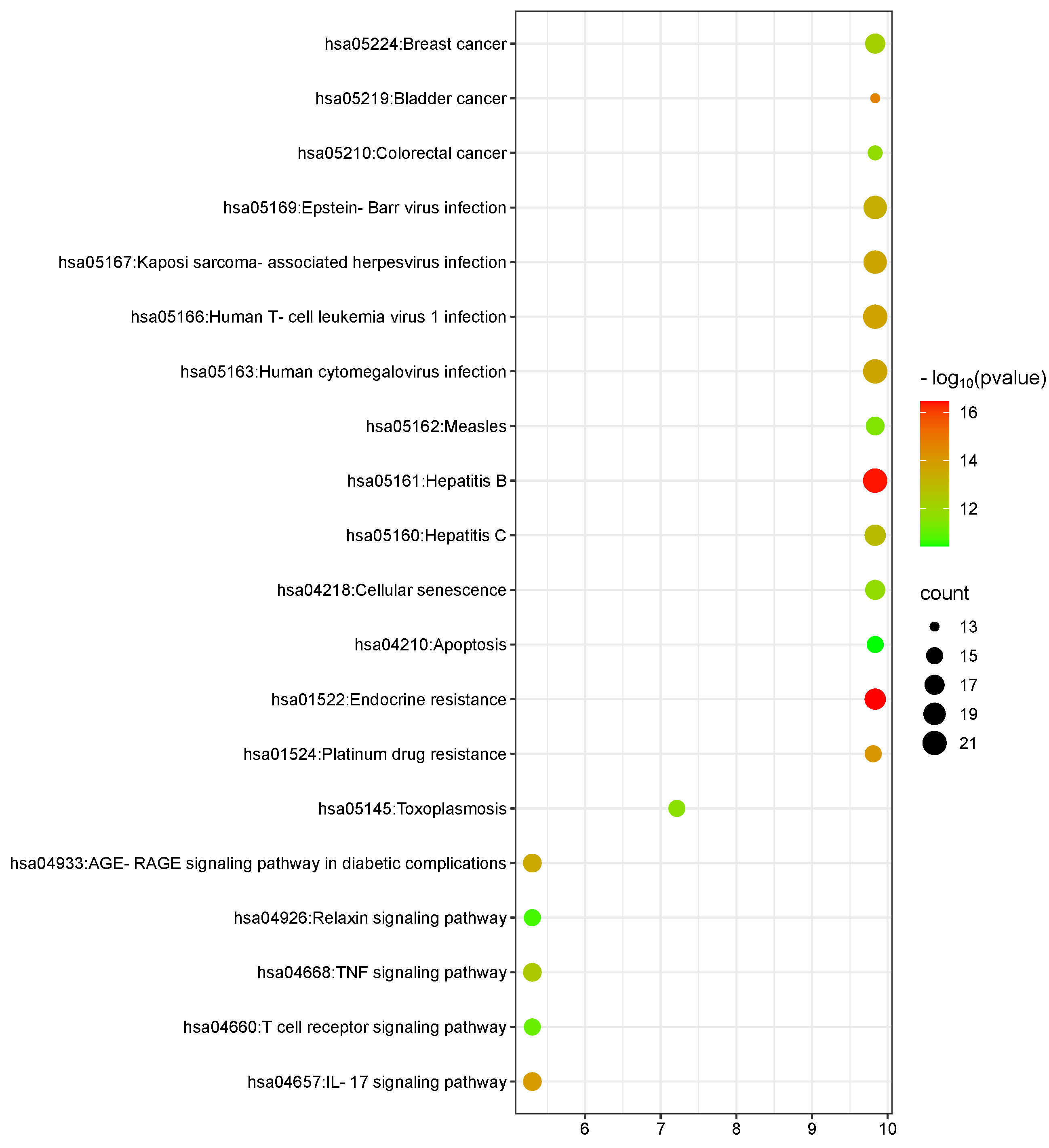 Fig. 6.
Fig. 6.
KEGG pathway enrichment analysis bubble chart (the size of the bubbles in the chart represents the number of genes annotated in the corresponding entries, and the color of the bubbles corresponds to the corrected p-value). KEGG, Kyoto Encyclopedia of Genes and Genomes.
| Term | Enrichment Score | p value | Count |
| Endocrine resistance | 9.835259674 | 3.38 × 10−17 | 18 |
| Hepatitis B | 9.835259674 | 3.88 × 10−17 | 21 |
| Bladder cancer | 9.835259674 | 2.00 × 10−15 | 13 |
| Platinum drug resistance | 9.809474273 | 7.85 × 10−15 | 15 |
| IL-17 signaling pathway | 5.302992243 | 1.10 × 10−14 | 16 |
| Human T-cell leukemia virus 1 infection | 9.835259674 | 1.95 × 10−14 | 21 |
| Kaposi sarcoma-associated herpesvirus infection | 9.835259674 | 2.31 × 10−14 | 20 |
| Human cytomegalovirus infection | 9.835259674 | 2.53 × 10−14 | 21 |
| AGE-RAGE signaling pathway in diabetic complications | 5.302992243 | 2.83 × 10−14 | 16 |
| Epstein-Barr virus infection | 9.835259674 | 4.41 × 10−14 | 20 |
| Hepatitis C | 9.835259674 | 1.25 × 10−13 | 18 |
| TNF signaling pathway | 5.302992243 | 3.44 × 10−13 | 16 |
| Breast cancer | 9.835259674 | 5.96 × 10−13 | 17 |
| Cellular senescence | 9.835259674 | 1.50 × 10−12 | 17 |
| Colorectal cancer | 9.835259674 | 1.57 × 10−12 | 14 |
| Toxoplasmosis | 7.21390789 | 2.24 × 10−12 | 15 |
| Measles | 9.835259674 | 3.48 × 10−12 | 16 |
| T cell receptor signaling pathway | 5.302992243 | 8.39 × 10−12 | 15 |
| Relaxin signaling pathway | 5.302992243 | 2.02 × 10−11 | 15 |
| Apoptosis | 9.835259674 | 3.74 × 10−11 | 15 |
KEGG, Kyoto Encyclopedia of Genes and Genomes.
Comparison of HF histopathology among the groups: In the normal group, the liver lobule structure was intact, hepatocytes were neatly arranged, and there was no evidence of hepatocyte degeneration, necrosis, inflammatory cell infiltration, or fibrous cord formation. In the model group, the liver lobule structure was significantly disrupted with blurred boundaries, disordered cell cords, swollen hepatocytes, and cytoplasmic steatosis. Compared with the model group, the Danshen high-dose group showed a relatively intact liver lobule structure and cell morphology, with improved HF, reduced fibrous cords, and decreased degrees of cellular edema, necrosis, and steatosis. Compared with the model group, the Danshen low-dose group did not show significant pathological changes in tissue, with multiple pseudo-lobules, and cells exhibited varying degrees of edema, necrosis, inflammatory reactions, and steatosis. See Fig. 7.
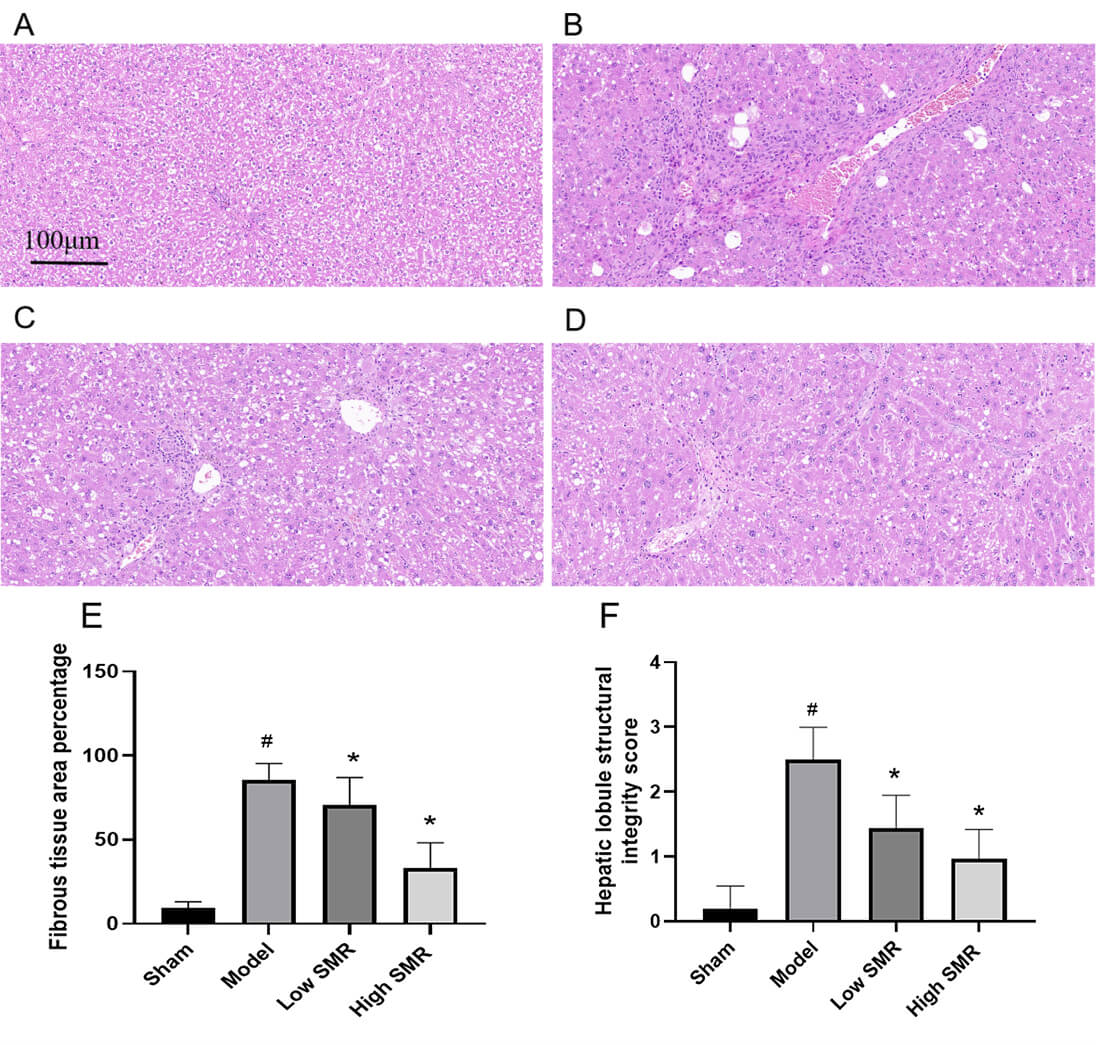 Fig. 7.
Fig. 7.
The Impact of Danshen on liver tissue fibrosis. Effect of
Danshen on liver tissue fibrosis: (A) sham group; (B) model group; (C) Danshen
low-dose group; and (D) Danshen high-dose group. Scale bar = 100 µm.
Percentage of fibrous tissue area (E), hepatic lobule structural integrity score
(F). #p
Western blot results showed that compared with the blank group, the expression
of IL-6, P-JAK2, and TGF-
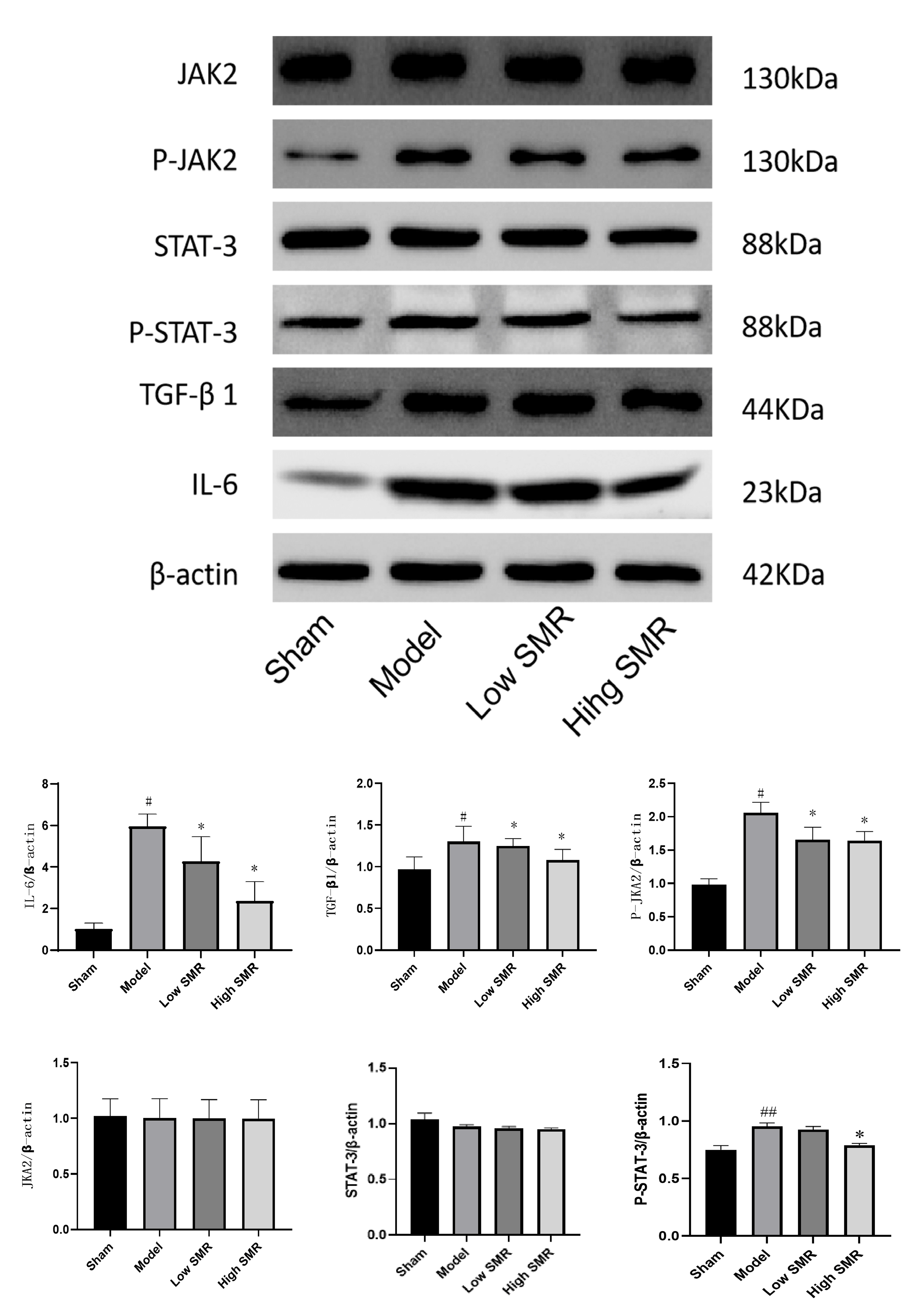 Fig. 8.
Fig. 8.
The effect of Danshen on the proteins JAK2, P-JAK2, STAT3,
P-STAT3, TGF-
To further determine the occurrence of ferroptosis, serum Fe2+, serum
ferritin (SF), TRF, type III procollagen (PcIII), and hydroxyproline (HyP) levels
were measured. The results showed that (see Fig. 9): compared with the blank
group, the levels of Fe2+ and TRF in the serum of the model group were
significantly decreased, while the levels of SF, PcIII, and HyP were
significantly increased (p
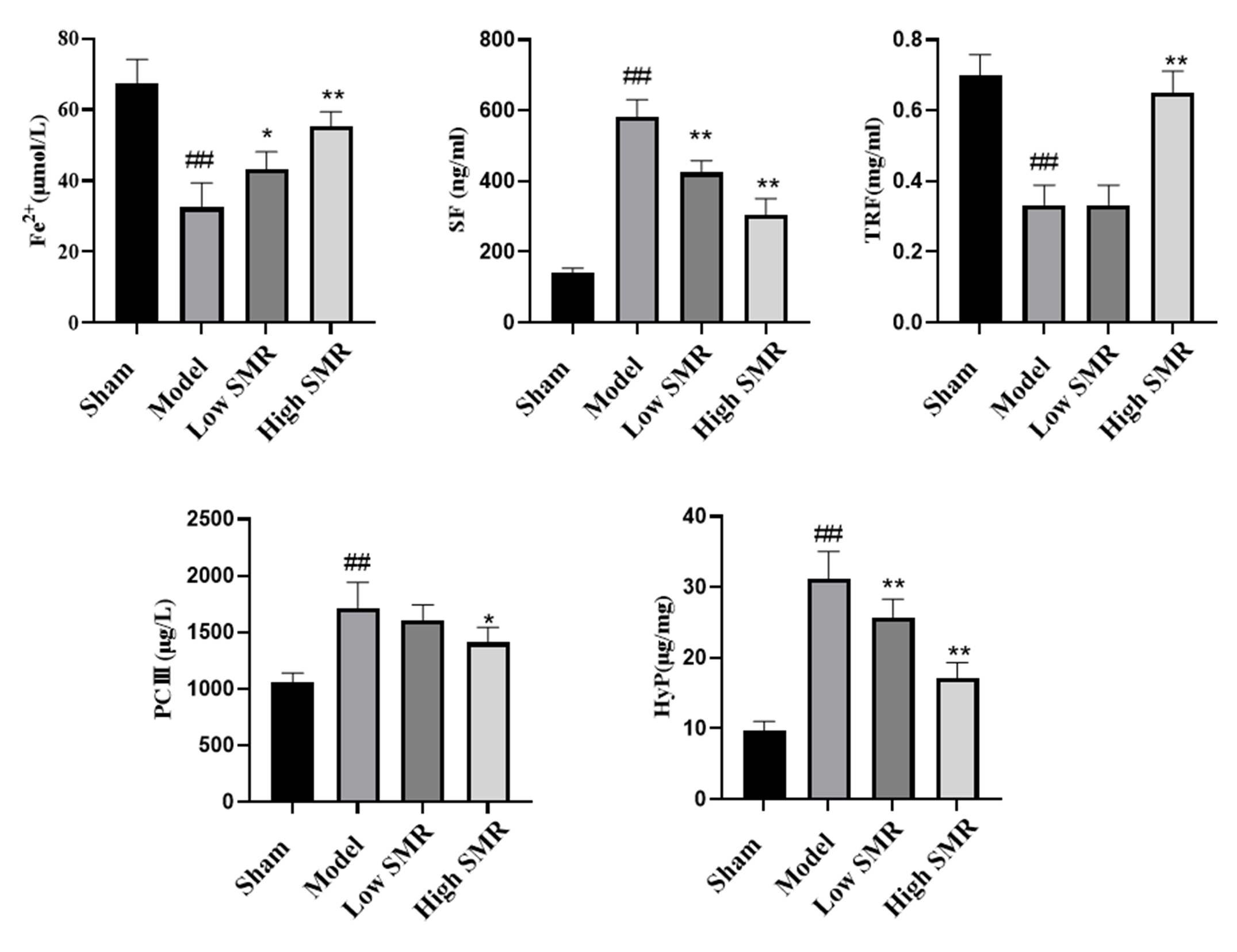 Fig. 9.
Fig. 9.
Effect of danshen on blood Fe2+, SF, TRF, PcIII, and HyP in
HF rats. ##p
At the level of iron metabolism, a significant increase in blood Fe2+ and TRF levels indicates the restoration of normal iron utilization and storage in the body.
To determine whether Danshen ameliorates liver fibrosis through the ferroptosis pathway, we measured serum MDA and GSH levels and hepatic GPX4 protein expression (see Fig. 10). MDA, the core oxidative stress product of HF tissue, was significantly elevated in serum, while Danshen attenuated this increase. GSH, the antioxidant stress marker of HF tissue, was significantly decreased, while Danshen ameliorated this decline. Western blot analysis revealed significantly decreased GPX4 protein expression in fibrotic liver tissue, which was reversed by Danshen intervention; high-dose treatment restored GPX4 levels to near-normal. These findings demonstrate that Danshen ameliorates fibrotic liver injury by inhibiting ferroptosis-related pathways and suppressing oxidative stress.
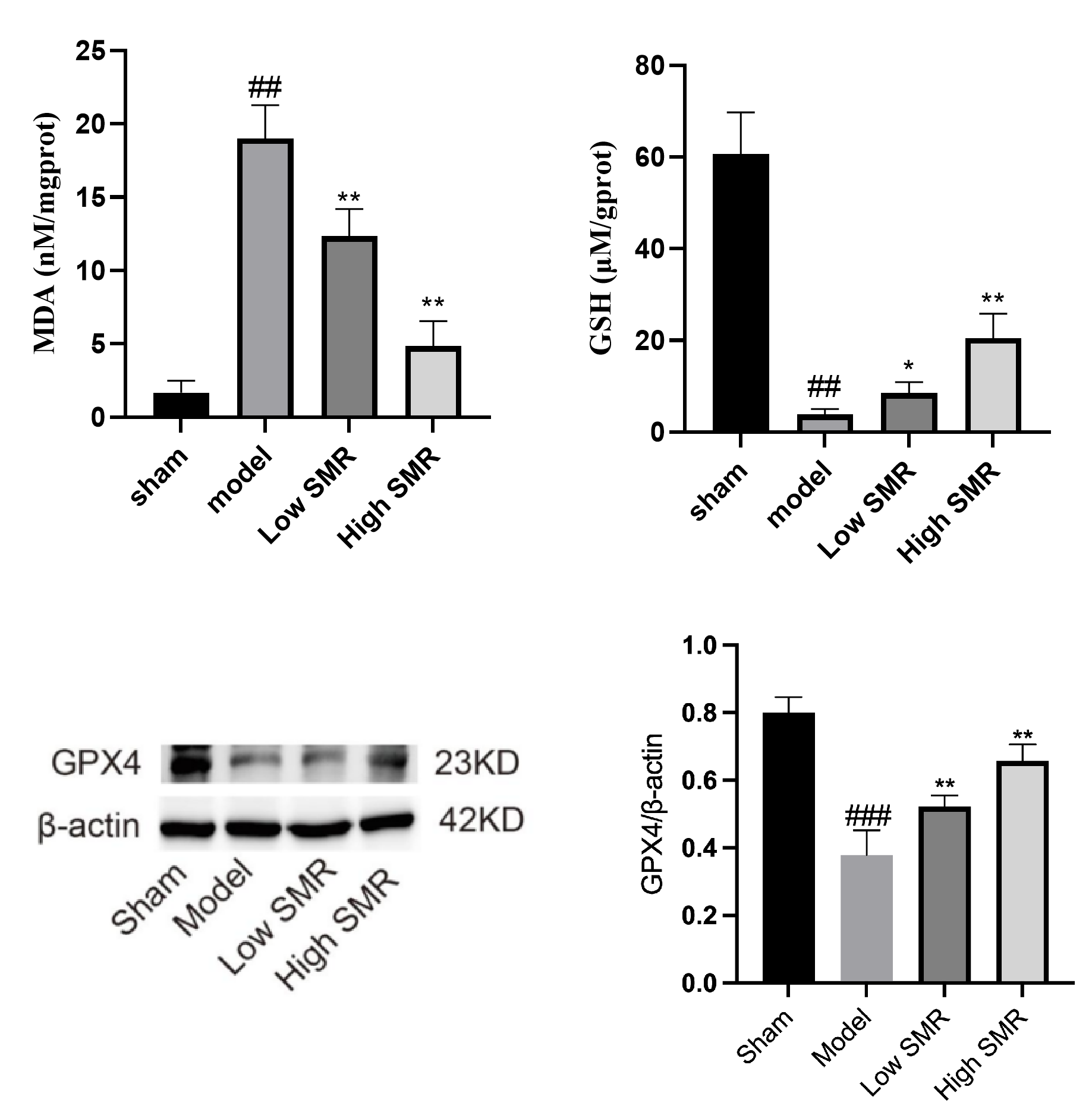 Fig. 10.
Fig. 10.
Effects of Danshen on serum MDA and GSH levels and hepatic GPX4
protein expression in liver fibrotic tissue. ##p
This study establishes a mechanistic framework wherein Danshen ameliorates CCl4-induced hepatic fibrosis through coordinated suppression of the IL-6/JAK/STAT3 inflammatory axis, restoration of iron homeostasis, and inhibition of ferroptosis. By integrating network pharmacology with experimental validation, we demonstrate that Danshen’s therapeutic effects extend beyond conventional anti-inflammatory actions to encompass a novel “inflammation-iron-ferroptosis” regulatory network.
Our computational analysis identified 92 nodes at the intersection of Danshen,
hepatic fibrosis, and iron metabolism, with JAK2/STAT3 emerging as top-ranking
hub genes (Figs. 2,3,4). The significant enrichment of AGE-RAGE, TNF, and IL-17
pathways (Fig. 6) aligns with established fibrogenic mechanisms, while the
prominence of “cellular senescence” and “apoptosis” pathways reflects the
pleiotropic nature of Danshen’s bioactive compounds. Notably, tanshinone IIA and
cryptotanshinone—key diterpenoids exhibiting OB
The JAK/STAT3 pathway represents a critical convergence point for inflammatory and iron-regulatory signals in hepatic fibrosis [13]. Our data demonstrate that CCl4-induced fibrosis is characterized by IL-6 overexpression and STAT3 hyperphosphorylation, consistent with HSC activation and hepcidin-mediated iron sequestration [14]. Danshen dose-dependently suppressed IL-6 expression and STAT3 phosphorylation while preserving total STAT3 protein levels (Fig. 8), suggesting targeted modulation rather than non-specific cytotoxicity. This is mechanistically significant because: (1) IL-6 is a master regulator of hepcidin transcription via STAT3 binding to the HAMP promoter [15]; (2) STAT3-driven hepcidin elevation impairs iron export via ferroportin degradation, creating a hypoxic microenvironment that perpetuates fibrogenesis [16]. By interrupting this feedforward loop, Danshen restores iron bioavailability, as evidenced by increased serum Fe2+, transferrin (TRF) and decreased ferritin (SF) (Fig. 9). The observed 1.8-fold elevation in TRF is particularly noteworthy, as transferrin not only facilitates iron delivery but also acts as an antioxidant by sequestering free iron that would otherwise catalyze Fenton reactions [17].
Our data reveal that CCl4-induced fibrosis produces a ferroptosis-permissive environment marked by GPX4 downregulation, GSH depletion, and MDA accumulation (Fig. 10), corroborating recent reports linking iron metabolism dysfunction to lipid peroxidation-driven cell death in chronic liver disease [18, 19]. Danshen’s restoration of GPX4 expression to near-normal levels represent a critical intervention point, as GPX4 is the sole enzyme capable of detoxifying membrane phospholipid hydroperoxides (PLOOHs) [20]. The reciprocal changes in MDA and GSH-core oxidative stress products and the primary GPX4 cofactor, respectively-demonstrate that Danshen reconstitutes the entire GPX4-GSH antioxidant axis. This finding resonates with recent observations that tanshinones can activate Nrf2, the transcriptional master regulator of GPX4 and GCLM (GSH synthesis) [21, 22]. Importantly, our study places ferroptosis within the broader JAK/STAT signaling context: STAT3 activation transcriptionally represses Nrf2, creating a vicious cycle of oxidative stress [23]; Danshen’s dual inhibition of STAT3 and restoration of GPX4-GSH likely disrupts this reciprocal inhibition.
We propose a novel feedback mechanism wherein Danshen-induced normalization of
iron metabolism reinforces anti-fibrotic efficacy (Fig. 9). Elevated
bioavailable Fe2+ satisfies the requirement of prolyl hydroxylases for
proper collagen cross-linking, reducing abnormal matrix deposition [24].
Simultaneously, increased intracellular iron may suppress IL-6 transcription via
ROS-dependent inhibition of NF-
While our integrated approach provides compelling evidence, several limitations warrant acknowledgment. First, the mechanistic validation remains at the protein expression level; functional indispensability of the JAK/STAT axis should be confirmed using specific inhibitors (e.g., Ruxolitinib) or hepatocyte-specific Stat3 knockout mice. Second, although we demonstrate GPX4 restoration, direct evidence that Danshen inhibits ferroptosis requires validation with GPX4 pharmacological inhibitors (RSL3/ML162) or conditional Gpx4 knockout models to establish causal dependency. Third, our study focused on male rats; sex-specific differences in iron metabolism and ferroptosis sensitivity necessitate future investigations in female cohorts [26]. Fourth, the network pharmacology analysis relied on databases with inherent annotation biases; experimental validation of additional hub genes (e.g., STAT1, ACVR1) would provide a more comprehensive understanding of Danshen’s polypharmacology. Finally, translating these findings to clinical practice requires multi-center, double-blind trials evaluating Danshen’s effects on serum PCIII, transient elastography, and patient-reported outcomes.
This study demonstrates that Danshen attenuates CCl4-induced hepatic fibrosis by downregulating the IL-6/JAK/STAT3 inflammatory axis, restoring iron homeostasis, and inhibiting ferroptosis through GPX4-GSH pathway activation. The identification of a “JAK/STAT-iron-ferroptosis” regulatory network provides a novel mechanistic framework for Danshen’s anti-fibrotic effects and highlights therapeutic potential for dual-targeting inflammation and iron-dependent cell death. Future studies leveraging genetic models and clinical translation will further solidify these findings and establish Danshen as a viable multi-modal therapy for hepatic fibrosis.
All data reported in this paper will also be shared by the lead contact upon request.
WO are responsible for the conception and design of articles, collection and collation of research materials, and drafting the manuscript. CL are responsible for the design the research study, review of the manuscript, overall supervision and Financial support. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The animal study protocol was approved by the Experimental Animal Ethics Committee of Haikou Affiliated Hospital of Central South University XiangYa School of Medicine (i.e., Haikou people’s Hospital). The ethical lot number: 2020- (Lunshen) -247. All experimental procedures were conducted in accordance with the relevant guidelines and regulations for the care and use of laboratory animals, including but not limited to the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize animal suffering and to reduce the number of animals used.
We gratefully acknowledge the assistance and instruction from Haikou Affiliated Hospital of Central South University XiangYa School of Medicine and Hainan Medical University.
This research was funded by Hainan Provincial Natural Science Foundation of China, grant number 821MS0846.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.










