1 Xizang University Medical College, 850000 Lhasa, Xizang, China
†These authors contributed equally.
Abstract
The incidence of altitude-related diseases has increased as more and more people have begun living in places at high altitudes. High-altitude polycythemia (HAPC) is a very widespread disease in high-altitude areas. Salvia miltiorrhiza has high medicinal value, with anti-tumor, anti-inflammatory, anti-myocardial ischemia, anti-oxidative stress, anti-thrombosis, and vascular dilation effects, as well as anti-atherosclerosis effects, thereby protecting against brain tissue damage, improving microcirculation, and activating the immune system. This study aimed to predict the latent molecular mechanisms through which the main active ingredients of Salvia miltiorrhiza treat HAPC disease using bioinformatics methods, including network pharmacology and molecular docking.
The Traditional Chinese medicine system pharmacology (TCMSP) database was used to screen for effective compounds and protein targets of Danshen, and the chemical structures of these compounds were downloaded. Using ‘high-altitude polycythemia’ as the keyword, the GeneCards and Online Mendelian Inheritance in Man (OMIM) databases were searched for potential disease targets, and the overlapping targets were identified through an intersection analysis. The protein-protein interaction (PPI) network was analyzed using Cytoscape 3.80. The Metascape database was utilized for Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses. 3D structures of the major proteins were obtained from the Protein Data Bank (PDB) database, and molecular docking was conducted between the main compounds and the core proteins, after which binding energies were plotted as a heatmap. The binding scenarios of the top two binding energies were visualized.
Danshen has anti-HAPC effects on the major compounds MOL000006, MOL007154, MOL007145, MOL007100, MOL007041, MOL007049, MOL007108, and MOL007088; the core targets are RAC-alpha serine/threonine-protein kinase (AKT1), interleukin-6 (IL6), tumor necrosis factor (TNF), vascular endothelial growth factor A (VEGFA), matrix metalloproteinase-9 (MMP9), nitric oxide synthase 3 (NOS3), estrogen receptor (ESR1), peroxisome proliferator-activated receptor gamma (PPARG), and interleukin-10 (IL10). The GO and KEGG results revealed that Danshen may exert anti-HAPC activity by regulating the binding of ligand-activated transcription factors, nuclear receptor activity, signaling receptor regulator activity, and the binding of certain other molecules to cellular compounds such as receptor complexes, the side of the membrane, the endoplasmic reticulum lumen, and the response to hormones, thereby achieving the regulation of epithelial cell migration, blood pressure, and other biological process (BP) entries. The signaling pathways related to the mechanism of action of Danshen mainly include the cancer pathway, the Hypoxia-inducible factor 1 (HIF-1) signaling pathway, and the Phosphatidyqinositol-3 kinase-RAC-alpha serine/threonine-protein kinase (PI3K-Akt) signaling pathway. The molecular docking results showed that the active components bound more stably to the targets, especially NOS3, TNF, and AKT1.
This research predicted the primary active compounds, targets of action, and signaling pathways involved in predicting the efficacy of Danshen in treating HAPC, using network pharmacology and molecular docking methods. This research provides the theoretical basis for subsequent experimental studies on the use of Danshen in clinical practice.
Keywords
- network pharmacology
- molecular docking simulation
- polycythemia
- Salvia miltiorrhiza
- pharmacokinetics
Salvia miltiorrhiza Bunge, also known as Danshen (DS) in Chinese, was first documented in Shennong’s Classic of Materia Medica (Shennong Bencao Jing) and is derived from the roots and stems of Salvia miltiorrhiza Bge. The family Labiatae is bitter and cold in nature and reportedly removes blood stasis and promotes blood circulation [1]. The main chemical compounds in Danshen are water-soluble phenolic acids and lipophilic tanshinone compounds, which exert antithrombotic effects, lipid-regulating functions, and vasodilating activities [2, 3, 4]. According to Liu et al. [5], Danshen preparations can effectively treat primary hypertension by assisting Western medicine with antihypertensive drugs, contributing to the improvement in the levels of low-density lipoprotein, high-density lipoprotein, and triglycerides, and subsequently regulating blood pressure. In addition, Li and Zhang [6] revealed that Danshen tablets enhance myocardial contractility, increase coronary blood flow, inhibit platelet aggregation, reduce the number of anginal episodes, improve blood lipid levels, and regulate blood pressure within a reasonable range in patients with stable angina pectoris.
High-altitude polycythemia (HAPC) is a chronic mountain sickness affected by the compensatory overproliferation of blood erythrocytes due to the hypoxia of the plateau and is often observed in residents living at altitudes of more than 2500 m permanently [7]. The characteristics of the plateau environment include hypobaric hypoxia, a wide range of temperatures between day and night, and a high level of ultraviolet radiation. Hypoxia is the primary hazardous factor affecting people physical and mental health and life activities, especially at high altitudes. Excessive erythrocyte proliferation leads to increased erythrocyte, hemoglobin, and erythrocyte cumulative pressure, higher blood viscosity, and thrombosis, which collectively decrease the ability of the blood to deliver oxygen to all organs in the body. As a result, organismal damage to multiple organs, such as the kidneys, heart, and lungs, occurs in the affected patients [8, 9]. The pathogenesis of HAPC is complex; however, studies have shown that the onset of HAPC is related to the plateau environment. With the increasing number of people moving to the plateau, the incidence of HAPC is gradually increasing [10, 11, 12]. Modern medicine has not yet elucidated the pathogenesis of HAPC, and the therapeutic effect is also unsatisfactory, with no safe or effective therapeutic drugs for HAPC available in China or abroad. Consequently, it is of practical importance to find relevant treatment measures to reduce the harm caused by HAPC to the physical and mental health of individuals.
Network pharmacology enables predicting the mode of action of drugs for disease through the establishment of a “drug-disease-target-pathway” network. Molecular docking technology can provide workable ideas and methods for exploring the pharmacologic substance basis and molecular mechanism of Danshen’s action [13]. Owing to the anti-thrombotic, blood pressure-lowering, lipid-regulating, and vasodilating effects of Danshen reported in the literature, Danshen was hypothesized as a potential drug for achieving the improvement and the treatment for plateau erythropoietic syndrome (HAPC). This research explored the active compounds and action targets of Danshen when used in the therapy of HAPC using network pharmacology and molecular docking methods. This study predicted the mechanism of action of Danshen in the treatment of HAPC at the theoretical level, illustrating the value of Danshen in HAPC prevention and treatment and providing a theoretical basis for subsequent fundamental research on HAPC treatment using Danshen and its clinical validation. The specific research process is presented in Fig. 1.
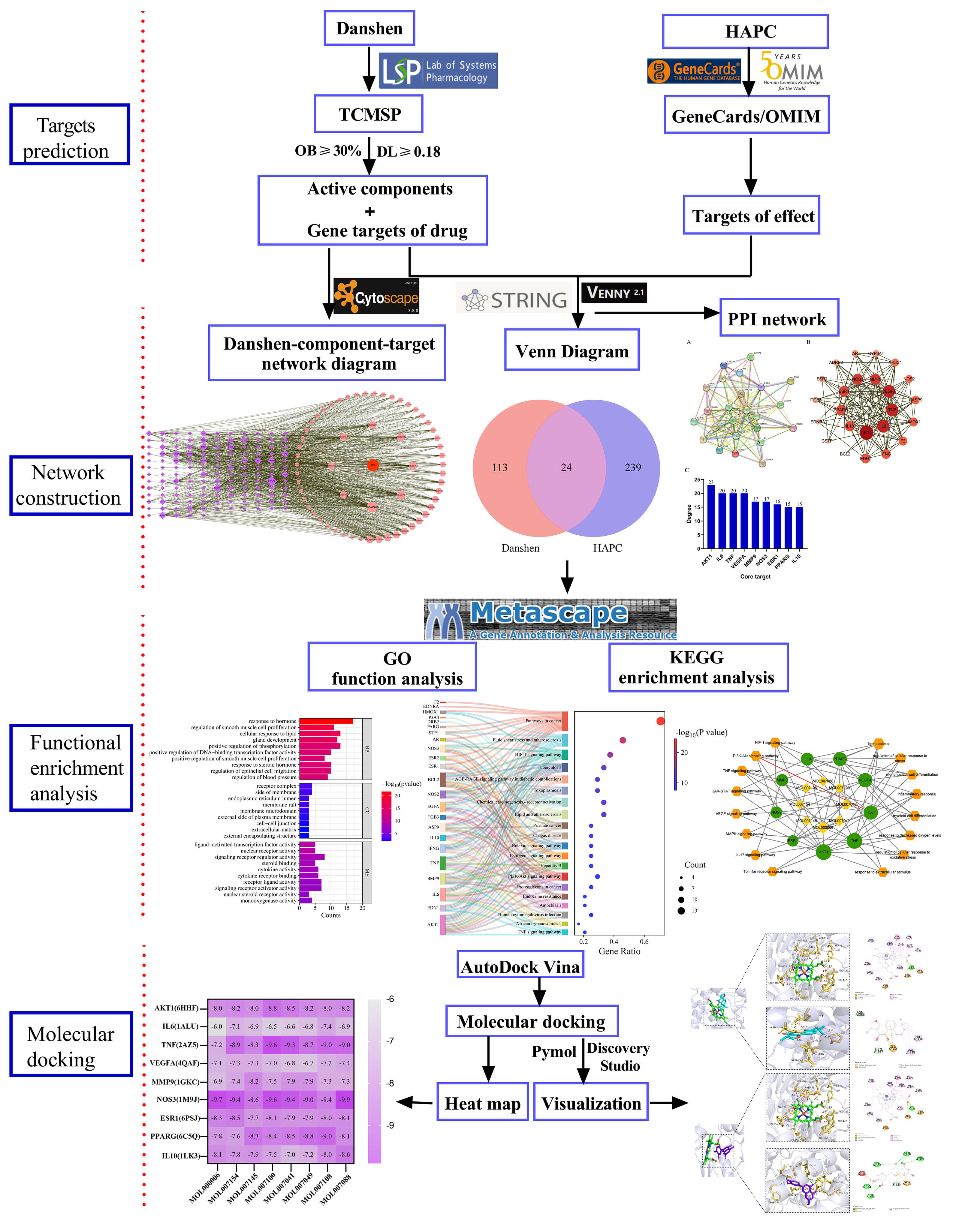 Fig. 1.
Fig. 1.
Flowchart of the research. HAPC, High-altitude polycythemia; TCMSP, Traditional Chinese medicine system pharmacology; OMIM, Online Mendelian Inheritance in Man; OB, Oral bioavailability; DL, Drug likeness; PPI, protein-protein interaction; GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes.
Oral drug likeness (DL)
The type file and network file containing the drugs, active compounds, targets, and classification information were created and input Cytoscape 3.8.0 software (https://cytoscape.org/release_notes_3_8_0.html) to establish the drug-ingredient-target spot network. The shape, size, or color about the target spots was adjusted by the degree of the drug ingredient or gene. The size of drug compounds or genes was correlated to their degree of connectivity within the network. Specifically, larger drug compounds or genes tended to exhibit a greater degree of connectivity, suggesting a greater likelihood of these compounds being the key active ingredients or functional genes involved in the mechanisms of Danshen’s action.
Using ‘high-altitude polycythemia’ as the keyword, the target spots of disease action were obtained from the GeneCards (https://www.genecards.org/) [15] and Online Mendelian Inheritance in Man (OMIM) [16] (https://www.omim.org/search/advanced/geneMap) websites. Construct a Venn diagram through the bioinformatics website (http://www.bioinformatics.com.cn/), and the intersecting targets of Danshen-HAPC were identified as potential targets through which Danshen exerts its anti-HAPC effects.
The screened “Danshen-HAPC” intersecting target spots were imported to the String platform (https://string-db.org), the species was set to “Homo sapiens”, and the “.tsv” format file was downloaded, and was imported into Cytoscape 3.80 software to analyze the core targets.
The Metascape database [17] (http://Metascape.org/) was carried out to conduct Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis and Gene Ontology (GO) function analysis of the intersection genes. GO function was analyzed at the molecular function (MF), cellular component (CC), and biological process (BP) levels [18]. The intersecting genes were copied into the Metascape database, and the species was set to “Homo sapiens”. A personalized analysis was performed, and the GO and KEGG data were downloaded. In the BP, CC, and MF analyses, the entries with the top 10 p values were chosen to construct bar graphs on the bioinformatics website. KEGG pathways with the top 20 p values were filtered out and used to construct a Sankey dot plot. Finally, through the above two plots, conducting KEGG enrichment analysis and GO functional analysis, and the results were analyzed and visualized.
Discovery Studio 2019 Client software (Dassault Systemes Biovia, https://www.3ds.com/products/biovia/discovery-studio), AutoDock Tools 1.5.7 (https://autodocksuite.scripps.edu/) and PyMOL (https://pymol.org/) were performed to conduct simulations of the interaction conformations of drug small molecules with target proteins and to predict their affinity. The 3D structure of the active ingredient of the drug was downloaded from the TCMSP database. In the docking simulation, torsion bonds and torsion centers were checked, and combined with the UniProt (https://www.uniprot.org/) database, obtain the 3D molecular structure files of the target proteins from the PDB database (https://www1.rcsb.org/). About the target protein structures, water molecules were deleted, and the proteins and drug molecules were subjected to hydrogenation treatment using the AutoDock software. The sites with the original inhibitors of the proteins were used for molecular docking, and at the sites with no inhibitor of the ligand, active pocket sites were created throughout the whole protein. Eventually, AutoDock Vina was employed to perform molecular docking and confirm the best binding structure of the active ingredients. The docking protocol utilized a semi-flexible mode, with an exhaustiveness value of 8. The maximum number of output conformations set to 10. PyMOL software was conducted to visualize the target-compound complexes. Molecular docking of the core targets with the main active ingredients was performed, and heatmap was established with the lowest binding energy. The AutoDock Vina results and macromolecules were integrated together into the PyMOL software and keep in PDB format. The docking results were analyzed with the Discovery Studio software, and the protein-compound complexes were visualized.
202 chemical components were identified from the TCMSP website, and among these,
a total of 65 chemical compounds met the selection criteria of DL
The network file containing Danshen along with the active components and target
spots of Danshen, and the type file containing the classification message were
executed and then imported them input Cytoscape 3.8.0 software to build the
drug-ingredient-target spot network (Fig. 2). Pink circles, purple circles, and
red circles represent the active compounds of the drug, the targets of action of
Danshen, and Danshen, respectively, which might be involved in the process of
drug action. The active compounds with the top 8 degrees were MOL000006
(luteolin), MOL007154 (tanshinone IIA), MOL007145 (salviolone), MOL007100
(dihydrotanshinlactone), MOL007049 (4-methylenemiltirone), MOL007041
(2-isopropyl-8-methylphenanthrene-3,4-dione), MOL007108 (isocryptotanshi-none),
and MOL007088 (cryptotanshinone), all with degree values
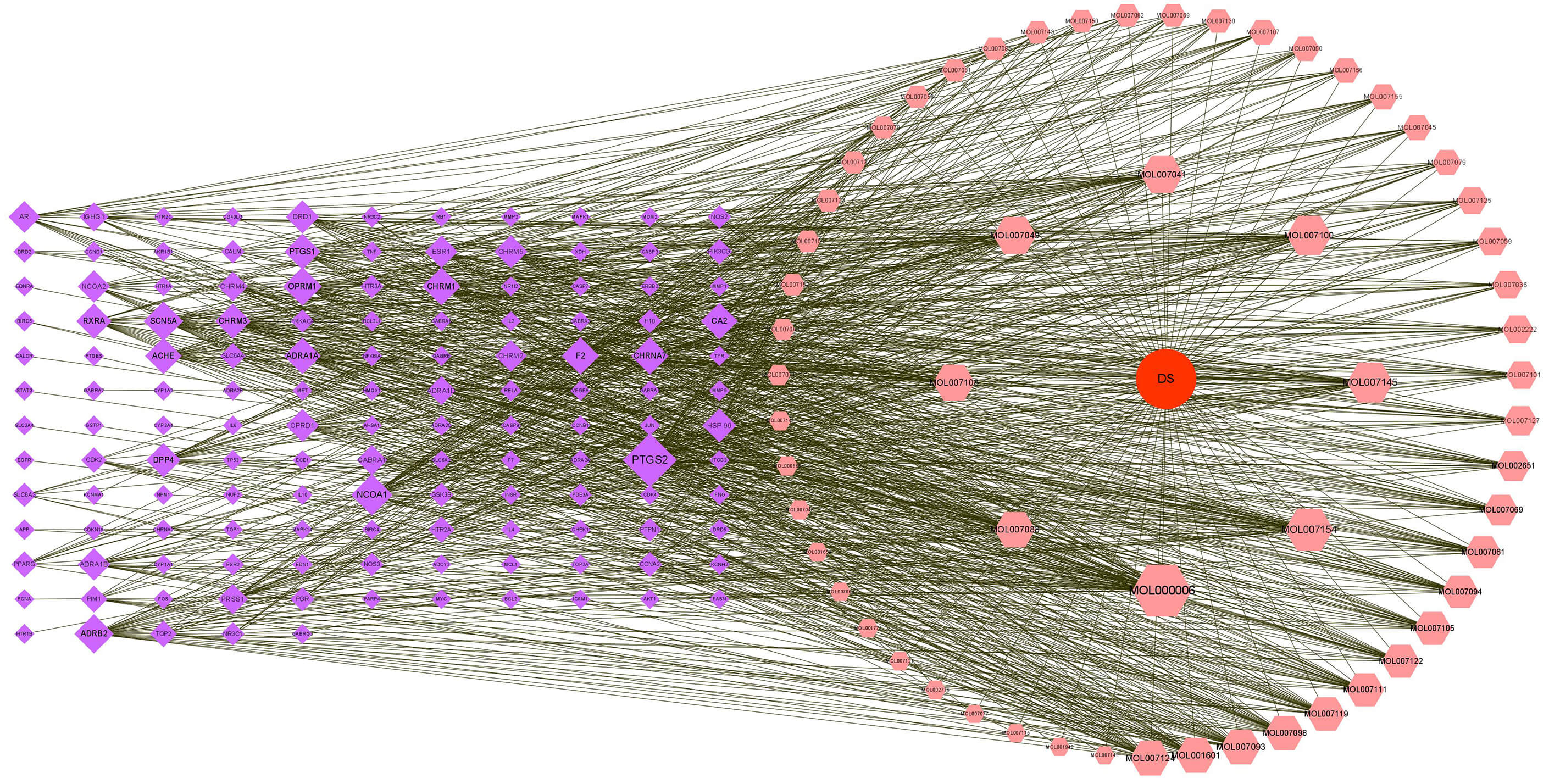 Fig. 2.
Fig. 2.
Danshen-component-target diagram.
| ID | Name | OB (%) | DL | Degree | Structure |
| MOL000006 | luteolin | 36.16 | 0.25 | 58 | 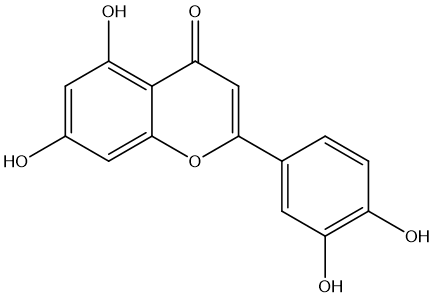 |
| MOL007154 | tanshinone iia | 49.89 | 0.4 | 42 | 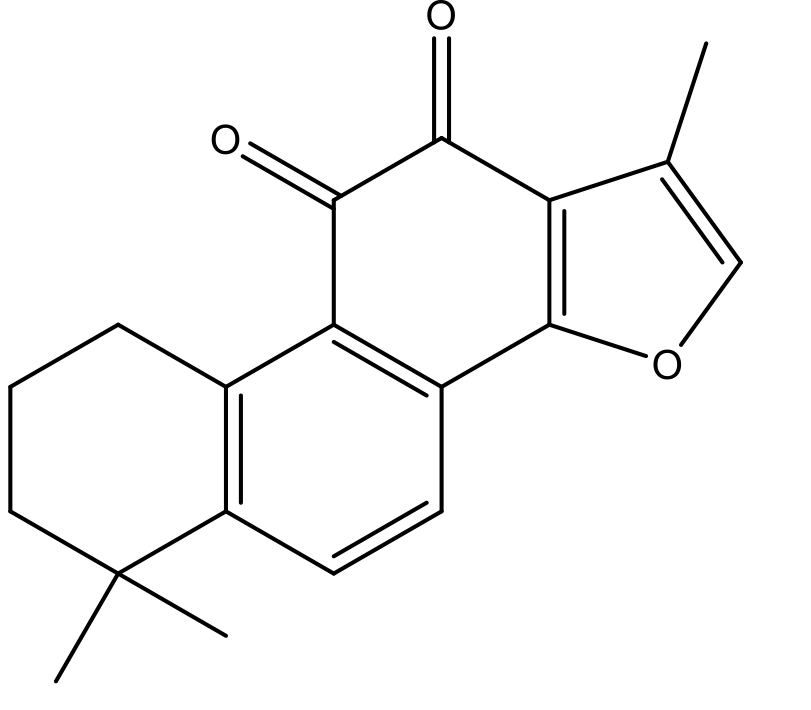 |
| MOL007145 | salviolone | 31.72 | 0.24 | 39 | 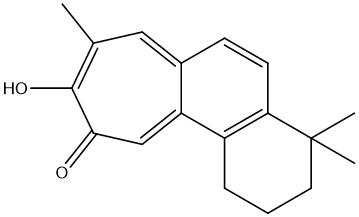 |
| MOL007100 | dihydrotanshinlactone | 38.68 | 0.32 | 37 | 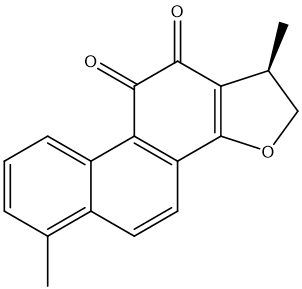 |
| MOL007041 | 2-isopropyl-8-methylphenanthrene-3,4-dione | 40.86 | 0.23 | 34 | 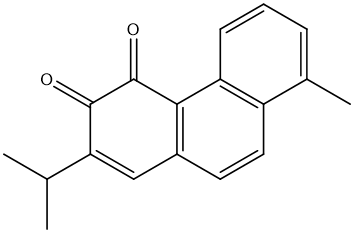 |
| MOL007049 | 4-methylenemiltirone | 34.35 | 0.23 | 34 | 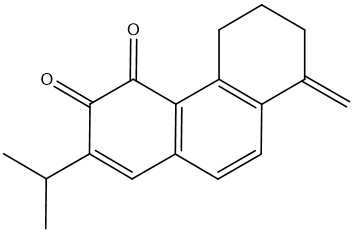 |
| MOL007108 | isocryptotanshi-none | 54.98 | 0.39 | 32 | 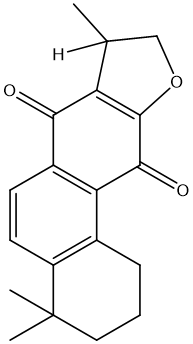 |
| MOL007088 | cryptotanshinone | 52.34 | 0.4 | 31 | 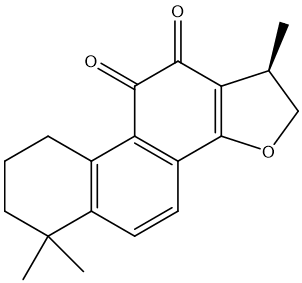 |
OB, Oral bioavailability; DL, Drug likeness.
Using the keywords “high altitude polycythemia” in the GeneCards and OMIM databases to obtain the disease targets, and these databases each contained 210 and 57 gene targets. After deleting the duplicates, 263 relevant targets were retained. A Venn diagram (Fig. 3) was made through the Microbiotics website, and thus, 24 intersecting targets of Danshen-HAPC were obtained.
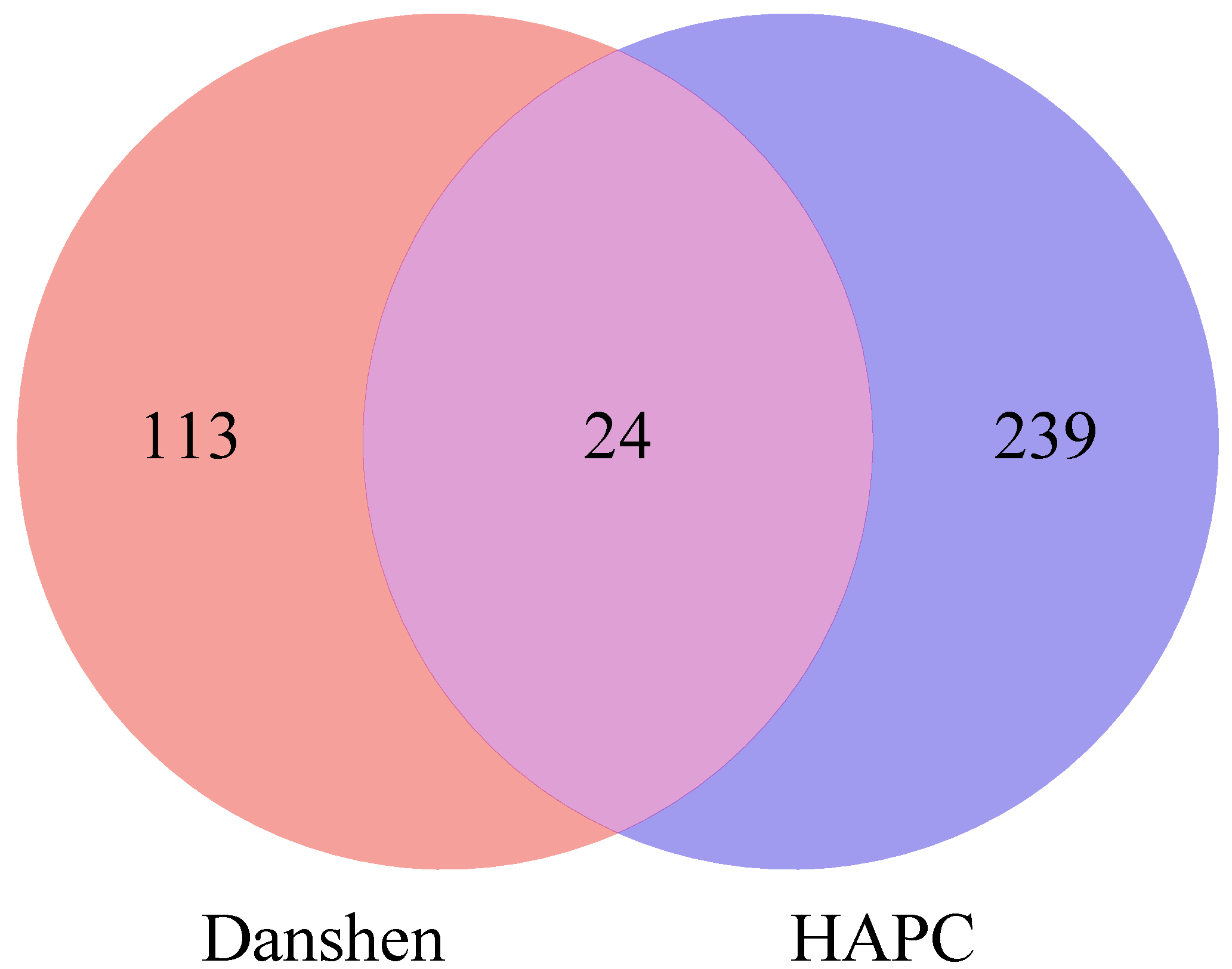 Fig. 3.
Fig. 3.
Venn diagram.
The screened intersection genes were imported into the String platform, and the “PNG” format file of these genes was downloaded to obtain the file’s portable network graphic. There were 24 nodes and 164 edges in the protein-protein interaction (PPI) network (Fig. 4A). After the “.tsv” file was downloaded, and Cytoscape 3.80 software was used to screen the core targets and thus establish the PPI network (Fig. 4B). The top 9 active targets are AKT1 (RAC-alpha serine/threonine-protein kinase, degree = 23), IL6 (interleukin-6, degree = 20), TNF (tumor necrosis factor, degree = 20), VEGFA (vascular endothelial growth factor A, degree = 20), MMP9 (matrix metalloproteinase-9, degree = 17), NOS3 (nitric oxide synthase 3, degree = 17), ESR1 (estrogen receptor, degree = 16), PPARG (peroxisome proliferator-activated receptor gamma, degree = 15), and IL10 (interleukin-10, degree = 15) (Fig. 4C). In addition, the core target might be one of these 9 inner-proteins through which Danshen exerts its therapeutic effects on HAPC.
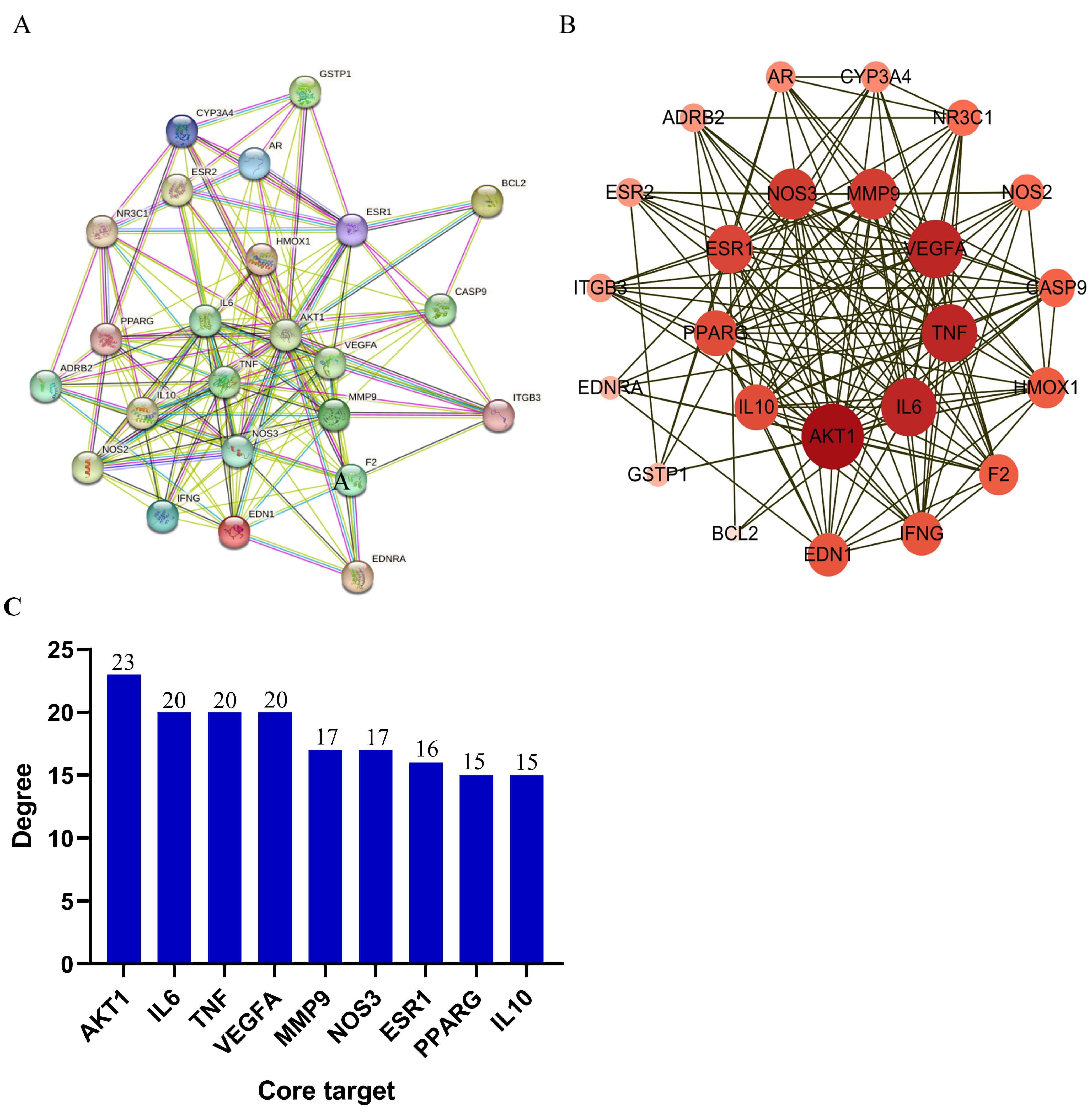 Fig. 4.
Fig. 4.
PPI network analysis. (A) was a grid diagram downloaded from the String platform, (B) was analyzed from Cytoscape 3.8.0 software, and (C) showed the degree values of the core genes. The above three figures all displayed the situation of the intersecting genes and subsequently derive the core genes. PPI, Protein-protein interaction; AKT1, RAC-alpha serine/threonine-protein kinase; TNF, tumor necrosis factor; VEGFA, vascular endothelial growth factor A; MMP9, matrix metalloproteinase-9; NOS3, nitric oxide synthase 3; ESR1, estrogen receptor; PPARG, peroxisome proliferator-activated receptor gamma; IL10, interleukin-10.
Twenty-four Danshen-HAPC intersection genes were imported into the Metascape platform, and the GO functions were analyzed from three aspects, namely, BP, CC, and MF. Among the top 10 terms with the smallest p values, BP was associated with a total of 688 terms, which were enriched in biological processes such as response to hormone, regulation of smooth muscle cell proliferation, regulation of epithelial cell migration, and regulation of blood pressure and others. CC was associated with 9 terms, which were associated mainly with receptor complex, side of membrane, cell-cell junction, extracellular matrix. MF was associated with 36 terms, which were associated mainly with ligand-activated transcription factor activity, signaling receptor regulator activity, nuclear receptor activity, and cytokine activity (Fig. 5). The length of the entry indicates the targets quantity included in this item, and the color scale (blue to red) corresponds to each item’s p value magnitude. A smaller p value represents a stronger correlation between the entry and Danshen for the treatment of HAPC. The functional analysis results of GO revealed that Danshen may regulate the binding of the molecules, such as those related to the ligand-activated transcription factor activity, nuclear receptor activity, and signaling receptor regulator activity, to cellular compounds such as receptor complex, side of membrane, and the endoplasmic reticulum lumen. These crossing genes may also be participate in BP process, the response to hormone, control the epithelial cell migration and regulation of blood pressure to exert anti-HAPC effects, for instance.
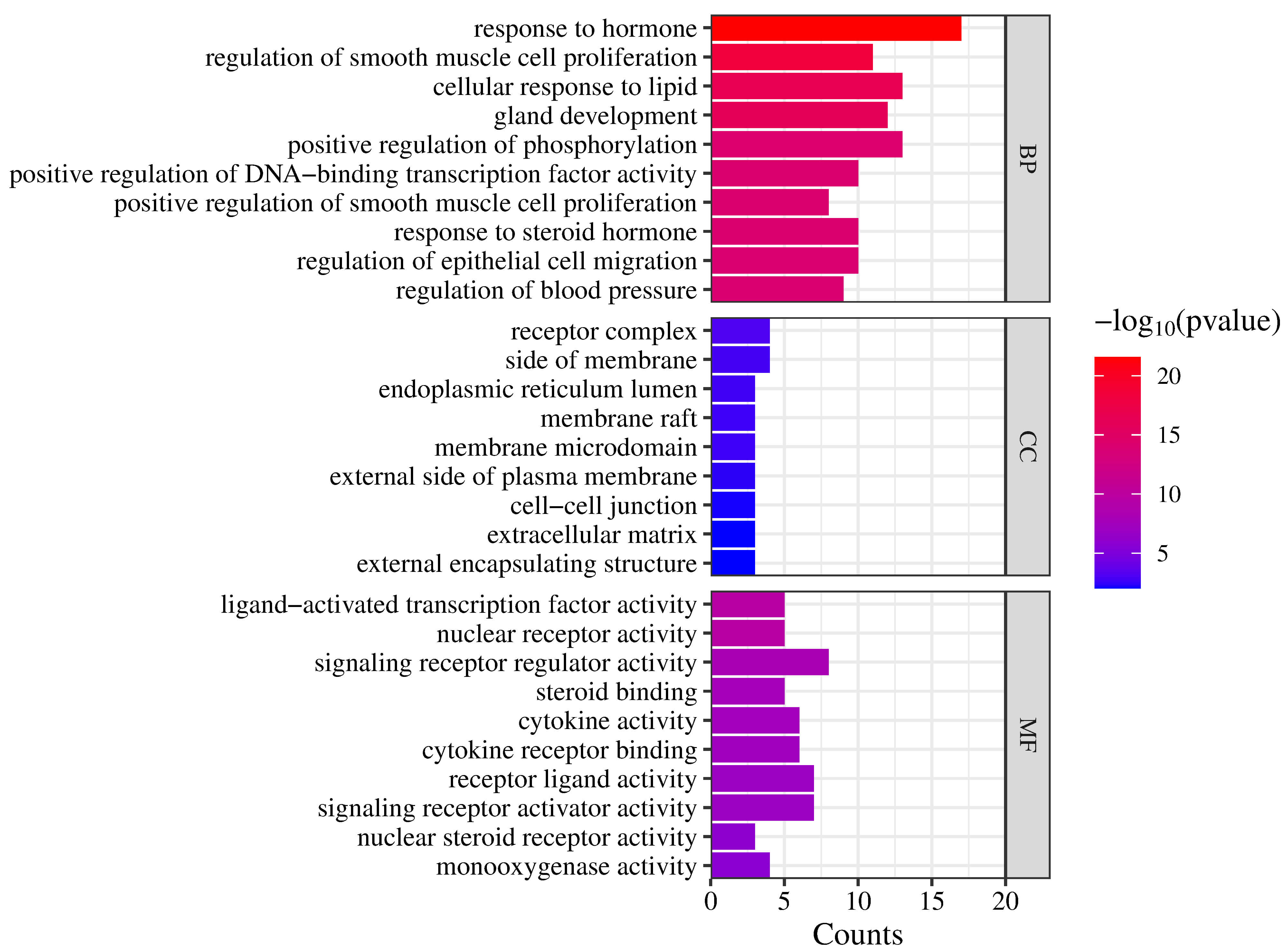 Fig. 5.
Fig. 5.
GO functional analysis.
KEGG analysis revealed 91 signaling pathways, following which screening of the
top 20 signaling pathways with smaller p values was performed. Sankey
bubble plots were generated (Fig. 6). The size of the circles represents the
number of genes involved in the pathway. The color scale (blue to red) indicates
the size of the ‘-log(p value)’ of pathway, and a small p value
indicates that the pathway is more significant in the anti-HAPC effects of
Danshen. According to the figure, the pathways in which Danshen exerts anti-HAPC
activity were enriched mainly in the Pathways in cancer, Fluid shear
stress and atherosclerosis, the Hypoxia-inducible factor 1 (HIF-1) signaling
pathway, and the Phosphatidyqinositol-3 kinase- RAC-alpha
serine/threonine-protein kinase (PI3K-Akt) signaling pathway. These pathways
mainly involved the targets AKT1, TNF, VEGFA, and IL6, which suggests that the
Danshen’s active compounds may play role in anti-HAPC via multiple
target-multiple signaling pathways, and the HIF-1 signaling pathway is regarded
as the most important pathway among these. HIF-1 is a heterodimer composed of
HIF-1
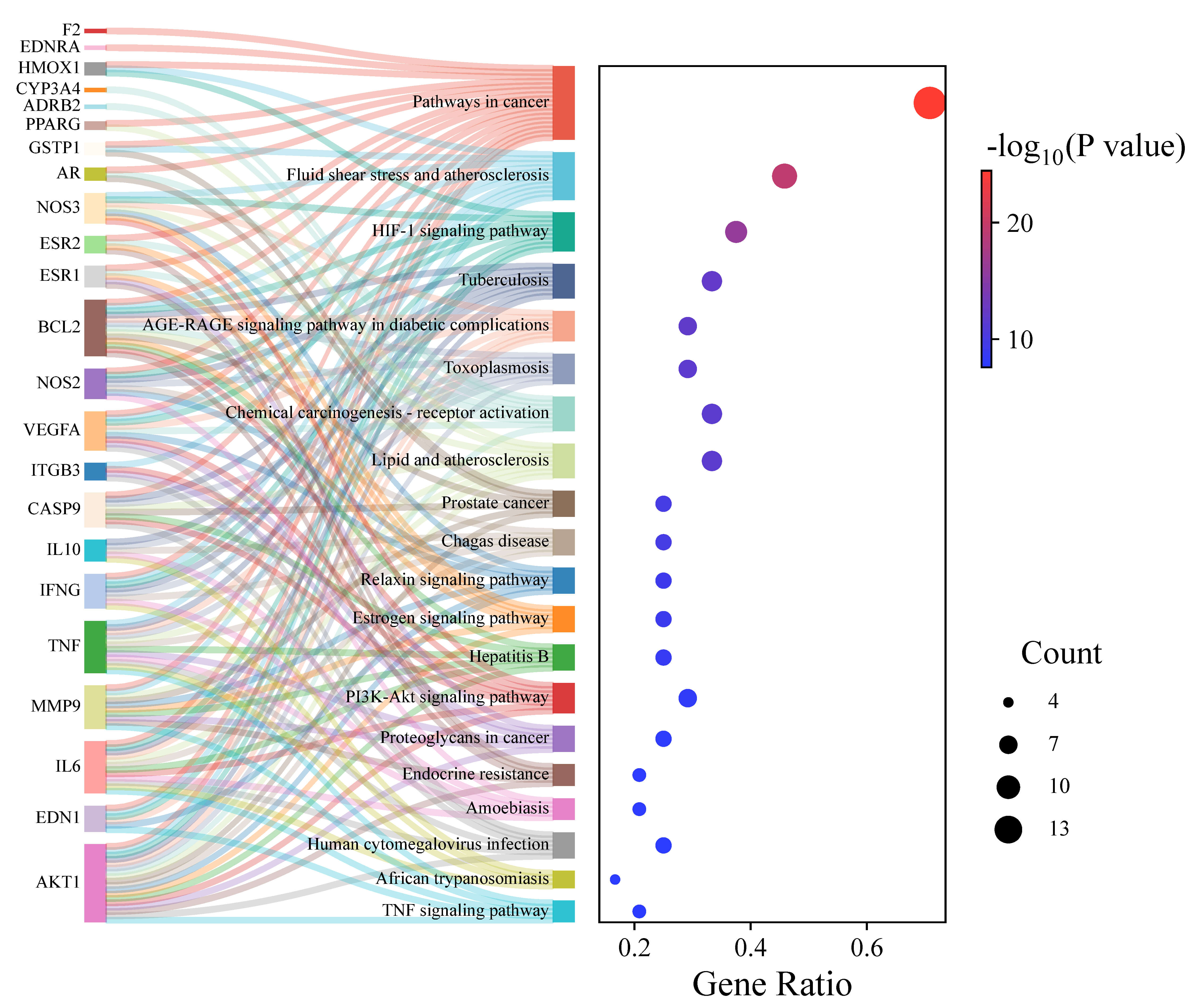 Fig. 6.
Fig. 6.
Sankey dot plot pathway enrichment.
The biological processes and signaling pathways involved in HAPC, such as
anti-inflammation, hypoxia, oxidation, and cell proliferation, were also screened
in this study, and 9 core targets were analyzed. The HIF1
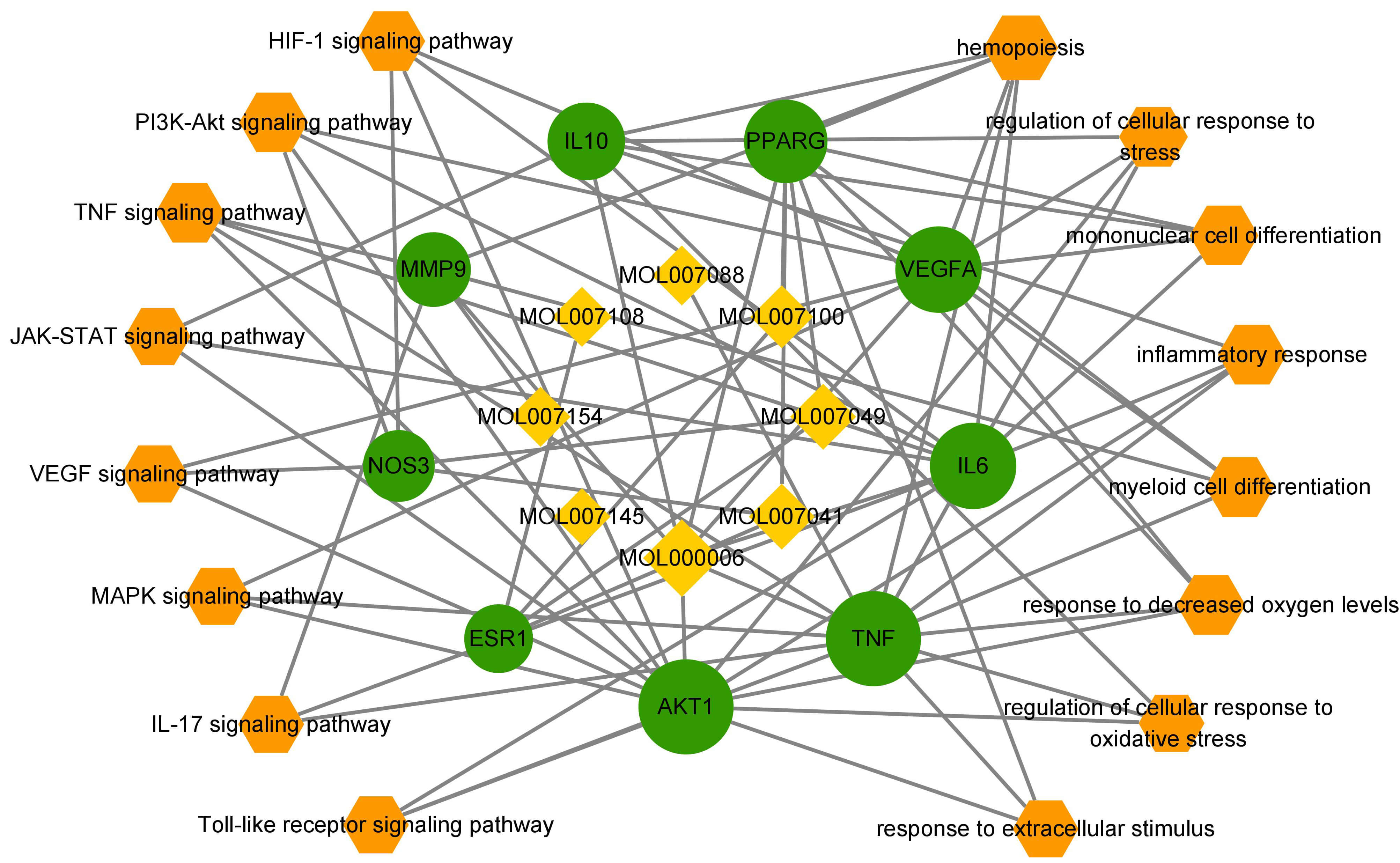 Fig. 7.
Fig. 7.
GO-BP analysis and KEGG analysis of the core target spots.
Molecular docking process between the drug-disease core target spots and the
main active compounds was performed. Using AutoDock Vina 1.5.7, PyMOL, and
Discovery Studio 4.5 Client software to analyze the mutual effect between major
core targets and the potential active components. The top 8 effective ingredients
are MOL000006, MOL007154, MOL007145, MOL007100, MOL007041, MOL007049, MOL007108,
and MOL007088. Subsequently, the 3D structures of the 9 core proteins, including
AKT1 (PDB ID: 6HHF), IL6 (PDB ID: 1ALU), TNF (PDB ID: 2AZ5), VEGFA (PDB ID:
4QAF), MMP9 (PDB ID: 1GKC), NOS3 (PDB ID: 1M9J), ESR1 (PDB ID: 6PSJ), PPARG (PDB
ID: 6C5Q), and IL10 (PDB ID: 1LK3) were obtained from the PDB database. AutoDock
Vina 1.5.7 software was used to calculate the binding energies, and a heatmap was
generated (Fig. 8). A lower binding energy represented better binding of the drug
compounds to the target protein. Generally, the binding free energy is less than
–5 kcal
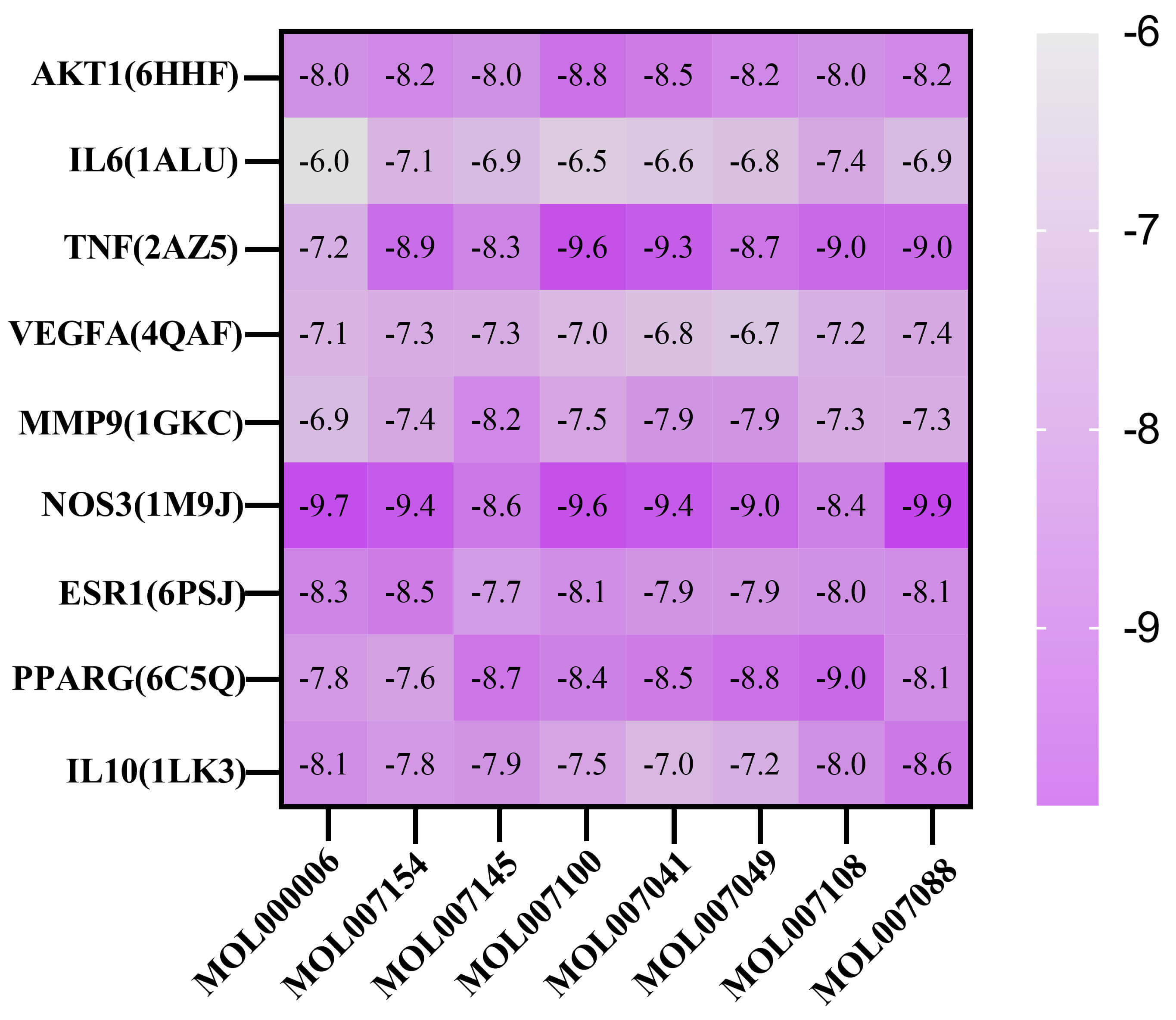 Fig. 8.
Fig. 8.
Heatmap for core genes and active compounds.
The results of molecular docking were visualized. Considering the first 2 groups with the lowest binding energies as examples (cryptotanshinone-NOS3 and luteolin-NOS3), the docking data are shown in Fig. 9. It was revealed that the intermolecular forces involved in the interactions between active site residues and the potential active compounds NOS3 mainly included Pi-anion bonds and Pi-donor hydrogen bonds. The cryptotanshinone-NOS3 compound was stabilized by a carbon-hydrogen bond with the chain ASN:366, GLN:247 was stabilized by a Pi-donor hydrogen bond, and PRO:334 and VAL:336 were stabilized by an alkyl bond (Fig. 9A). Moreover, the Luteolin-NOS3 complex demonstrated that five amino acids on the chain, namely, PRO:334, PHE:353, TYR:357, ASN:366, and ARG:372, formed conventional hydrogen bonds, while GLN:247 formed a Pi-donor hydrogen bond with the drug compound (Fig. 9B).
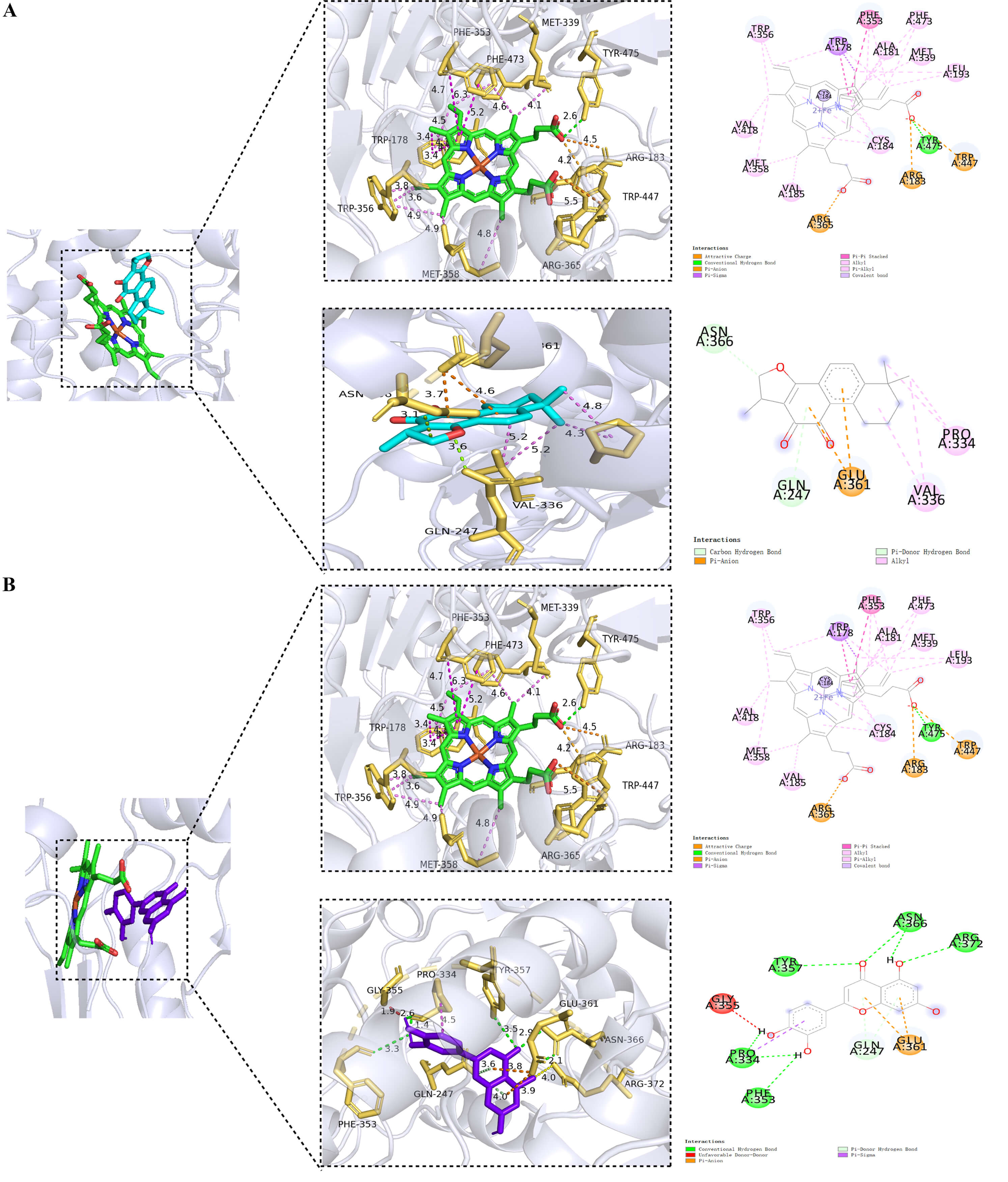 Fig. 9.
Fig. 9.
Visualization of the molecular docking data. (A) The first figure represented the interaction of the inhibitor carried by the protein NOS3 itself, and the second figure represented the interaction of cryptotanshinone with NOS3. (B) The first figure represented the interaction of the inhibitor carried by the protein itself, and the second figure represented the interaction of luteolin with NOS3. Green: inhibitor carried by the protein itself; Light gray: NOS3 protein; Cyan: cryptotanshinone; Purple: luteolin. The specific coordinate information of the NOS3 docking box is center_x = 14.45, center_y = 10.62, center_z = 54.04.
HAPC is a common chronic disease with a relatively high incidence in
high-altitude areas, characterized by an increase in red blood cells (males,
hemoglobin [Hb]
Salvia miltiorrhiza Bunge is typically refered to as Danshen in China and is used as medicinal materials in the form of its dried roots or rhizomes [21]. The first record of this medicine and its effects in China is the ancient medical treatise, “Shennong’s Classic of Materia Medica (Shennong Bencao Jing)” [22]. Danshen belongs to the liver and kidney meridians and is cold in nature. It has a slightly bitter taste. Danshen is distributed mainly in the northern, eastern, and northwestern regions of China. The active compounds of Danshen mainly consist of two parts: danshenolic acid and tanshinones, represented by danshenolic acid B and tanshinone IIA, respectively. Danshen can cool the blood to reduce swelling, activate the blood to remove blood stasis, and nourish the blood to tranquilize the mind [23]. Danshen is a Chinese frequently-used herbal medicine and it is applied in the treatment of many diseases, including diseases of blood vessel [24], Alzheimer’s disease [25], Parkinson’s disease [26], cancer [27], and bone loss [28].
In this study, 59 active compounds in Danshen were identified, and 137 potential anti-HAPC targets were predicted using network pharmacology. A “medicine-compound-target” diagram was constructed, which revealed that Danshen may exert its anti-HAPC activity through multiple targets and compounds. The active compounds of Danshen could be categorized into two main groups: tanshenolic acid and tanshinones. Tanshinones are lipophilic active compounds in Danshen, which are unstable to light and are classified as diterpenoids, mainly contains several important components, such as dihydrotanshinone, tanshinone IIA, cryptotanshinone and tanshinone I. Among these, tanshinone IIA has the most significant activity [29, 30]. The top 8 active compounds of Danshen that exert anti-HAPC effects include tanshinone IIA, salviolone, isocryptotanshinone, and cryptotanshinone; in particular, tanshinone IIA has a variety of biological function, including antioxidation [31], anti-inflammatory [32], anti-fibrotic [33], antiviral [34], and anti-tumor effects [35]. It has a great protective effect against cardiovascular diseases [36]. Salviolone exerts some beneficial effects on melanoma by hindering cell cycle progression, the malignant phenotype, and Signal transducer and activator of transcription 3 (STAT3) signaling [37]. There are few reports on the biological activity of isocryptotanshinone, although it has been proven to be able to restrain the proliferation of breast cancer cells and gastric cancer cells [38, 39]. Cryptotanshinone has a wide variety of pharmacological effects, including anti-tumor activity [40, 41], anti-inflammatory function [42], immunoregulation activity [43], and anti-fibrotic effects [44].
PPI network analysis in this study revealed that AKT1, IL6, TNF, VEGFA, MMP9, NOS3, ESR1, PPARG, and IL10 could be the core targets through which Danshen exerts its anti-HAPC effects. As a serine/threonine protein kinase, AKT1 regulates a variety of processes, including metabolic proliferation, cell survival, growth, and angiogenesis [45]. IL-6 plays a significant role in bone homeostasis and blood vessels by inducing VEGF and further enhancing vascular permeability and angiogenic activity [46]. TNF exert its action in angiogenesis through synergistically inducing VEGF production along with IL1B and IL6 [47]. MMP9 plays a significant role in the proteolysis of extracellular matrix components and the migration of white blood cells [48]. NOS3 has been linked to the production of NO. NO causes the relaxation of vascular smooth muscle through the cyclic guanosine monophosphate (cGMP)-mediated signal transduction pathway, induces angiogenesis in coronary vessels through VEGF, and promotes blood coagulation by activating platelets [49]. ESR1, a nuclear hormone receptor, is involved in the regulation of eukaryotic gene expression. It affects cellular proliferation and differentiation in target tissues and is also involved in the activation of NOS3 and endothelial NO production. It is reported that increasing the levels of circulatory NO and NO metabolite is helpful for acclimatization to high altitudes [50, 51]. PPARG achieves the maintenance of the angiogenic potentiality in mature PMVECs via E2F1-mediated gene regulation [52]. IL-10 plays a role in many immune system cells and is a very important immunoregulatory cytokine and can limit the excessive damage caused by inflammation to the body, thereby protecting the body [53]. Notably, these genes are associated with angiogenesis and inflammation through functions related to HAPC occurrence; therefore, it was hypothesized in this study that Danshen may exert its anti-HAPC activity mainly by affecting angiogenesis and inhibiting the inflammatory responses.
In this study, the 24 target spots were conducted to analysis of the GO function
and KEGG enrichment to identify the roles of the intersecting Danshen-HAPC
targets in signaling pathways and gene functions. In the GO analysis, 688 entries
were enriched in BP, 9 entries in CC, and 36 entries in MF. These research
results reveled that Danshen may exert anti-HAPC effects through regulating the
binding of compounds related to ligand-activated transcription factor activity,
nuclear receptor activity, and signaling receptor regulator activity to the
receptor complex, the side of the membranes, and the lumen of the endoplasmic
reticulum in response to hormones, regulating the migration of epithelial cells,
and regulating blood pressure. KEGG pathway analysis revealed 91 pathways
potentially related to the anti-HAPC effects of Danshen. These studies focused on
pathways involved in cancer, the HIF-1 signaling pathway, the PI3K-Akt signaling
pathway, and others, and mainly involved the targets AKT1, TNF, VEGFA, IL6, and
others, which suggested that the anti-HAPC activity of Danshen may be realized
through the multi-target and multi-signaling pathways of the active compounds,
especially, the HIF-1 signaling pathway may be of great significance. In
addition, through molecular docking it was shown that all the active compounds of
Danshen were more stably bound to protein targets, especially the three target
proteins, NOS3, TNF, and AKT1 which bound to the main active compounds with lower
binding energies and stronger stability, and the binding energies were all
In summary, Danshen may exert its anti-HAPC effect through 8 main active compounds that act on 24 targets. The GO function analysis included 733 entries related to nuclear receptor, signaling receptor regulator, cytokine, epithelial cell migration regulation, and blood pressure regulation. The enriched KEGG pathways included 91 signaling pathways, such as the pathways relevant to cancer, the HIF-1 signaling pathway, and the PI3K-Akt signaling pathway. Specifically, the main active compounds, including MOL000006, MOL007154, MOL007145, MOL007100, MOL007041, MOL007049, MOL007108, and MOL007088, may bind to core proteins, such as AKT1, IL6, TNF, VEGFA, MMP9, NOS3, ESR1, PPARG, and IL10, and exert anti-HAPC effects by regulating the biological processes, such as epithelial cell migration regulation, blood pressure regulation, Pathways in cancer, the HIF-1 signaling pathway, and the PI3K-Akt signaling pathway. Finally, through molecular docking, three target proteins, NOS3, TNF, and AKT1, which exhibited greater binding ability to the key active components of Danshen, were identified. This study further substantiated the positive effects of the traditional Chinese medicine Danshen, on HAPC in theory through the use of network pharmacology strategies and molecular docking methods, and the findings would guide the relevant clinical and experimental studies in the future.
DS, Danshen; BP, Biological process; CC, Cellular component; HAPC, High-altitude polycythemia; MF, Molecular function; DL, Drug likeness; GO, Gene ontology; OB, Oral bioavailability; PPI, Protein-protein interaction; KEGG, Kyoto Encyclopedia of Genes and Genomes; TCMSP, Traditional Chinese medicine system pharmacology.
Protein structures for key targets were obtained online from the PDB Protein Data Bank (https://www.rcsb.org/), including AKT1 (PDB ID: 6HHF), IL6 (PDB ID: 1ALU), TNF (PDB ID: 2AZ5), VEGFA (PDB ID: 4QAF), MMP9 (PDB ID: 1GKC), NOS3 (PDB ID: 1M9J), ESR1 (PDB ID: 6PSJ), PPARG (PDB ID: 6C5Q) IL10 (PDB ID: 1LK3). The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
JR acquired the data online, analyzed the data and revised the manuscript. WY made figures, tables and searched references, outlined the overall structure of the article and wrote the first draft of the article. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Our research focused on the molecular mechanism prediction of Danshen with the anti-HAPC effect, without involving animal experiments. So ethical approval was not applicable.
We would like to express my gratitude to all those who helped me during the writing of this manuscript, and thanks to all the peer reviewers for their opinions and suggestions.
The present study was supported by the Xizang Natural Science Foundation of China (no. XZ202401ZR0010), the Central Guidance on Local Science and Technology Development Fund of Xizang (grand no. XZ202301YD0040C) and Science and Technology Department’s Major Scientific Project of Xizang (no. XZ202201ZD0001G).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









