1 Acropolis Institute of Pharmaceutical Education and Research, 453771 Indore, India
2 Chameli Devi Institute of Pharmacy, 452010 Indore, India
Abstract
The therapeutic potential of herbal and natural compounds has long been recognized; meanwhile, the role of these compounds in neuropharmacology is gaining increasing attention due to their promising biochemical effects. The rising prevalence of neurodegenerative disorders, including Alzheimer’s disease, Parkinson’s disease, and depression, has underscored the need for novel, effective treatments with minimal side effects. Notably, natural products derived from plants, fungi, and other organisms offer a vast array of bioactive molecules that can interact with neuronal pathways to modulate cognitive function, neuroprotection, and synaptic plasticity. Furthermore, recent advancements in neuropharmacology have revealed the molecular mechanisms underlying the therapeutic efficacy of herbal compounds, such as flavonoids, alkaloids, terpenoids, and phenolic acids, in mitigating oxidative stress, inflammation, and neuroinflammation. Moreover, these compounds have shown promise in enhancing neurotransmitter systems, such as serotonin, dopamine, and acetylcholine. The integration of cutting-edge technologies, including high-throughput screening, metabolomics, and molecular docking, has facilitated the discovery of novel natural neuroprotective agents. Artificial Intelligence (AI) and nanotechnology are transforming the use of phytobioactive compounds in modern neuropharmacology by enabling precise prediction of molecular targets, optimizing drug-likeness, and enhancing mechanistic understanding. Additionally, nanocarriers such as polymeric nanoparticles, liposomes, and nanoemulsions, are improving the brain delivery, stability, and bioavailability of plant-derived molecules that traditionally suffer from poor pharmacokinetics. Together, these technologies can accelerate the development of next-generation neuroprotective therapeutics from natural products with greater efficiency, specificity, and translational potential. This review provides a comprehensive overview of the latest biochemical innovations in herbal and natural compounds for neuropharmacology, emphasizing the potential of these compounds as safe and effective alternatives or adjuncts to conventional neurotherapeutics.
Keywords
- neuropharmacology
- phytobioactive compounds
- biochemical mechanism
Neuropharmacology has become one of the most active fields of biomedical research, focusing on neurological and mental illnesses that together make up a large part of the world’s health problems. Conventional drug research has encountered challenges such as limited efficacy, unpleasant side effects, and elevated attrition rates in clinical trials, despite decades of advancement [1]. Conditions like Alzheimer’s disease, Parkinson’s disease, epilepsy, depression, and neuropathic pain are still only partially treated by current medications [2]. The central nervous system (CNS) is tremendously complicated, with many different neurotransmitter systems, synaptic plasticity, and neuronal circuits. This has made it hard to find drugs that work on just one target [3]. Consequently, there is an increasing interest in investigating multi-target and pathway-specific therapies, especially those derived from natural sources, to address these constraints [4].
Herbal medications and natural substances offer an extensive array of neuroactive molecules that engage with various biochemical pathways concurrently [5]. Many herbal substances have many effects, such as acting as antioxidants, changing the levels of neurotransmitters, and controlling neuroinflammation [6]. This is different from synthetic chemicals that are made to work on only one target. Numerous phytochemicals, including curcumin, ginsenosides, resveratrol, and flavonoids, have demonstrated potential in preclinical and clinical investigations for their neuroprotective and cognitive-enhancing effects [7]. Improvements in metabolomics, network pharmacology, and computational biology are making it easier to find new herbal candidates and their molecular targets [8]. The growing convergence of ethnomedicine and contemporary neuroscience underscores the translational capacity of natural substances in neuropharmacology [9].
This chapter examines the function of herbal and natural substances in neuropharmacology, focusing on their biochemical advancements. It combines ideas from both traditional medicine and modern research, focuses on how synaptic modulation, neuroinflammation, ion channel regulation, and epigenetic reprogramming work, and looks at how important phytochemicals can help treat major neurological illnesses. By bringing together current achievements, we hope to give a full picture of how herbal bioactive might help with neuropharmacological methods for the next generation [10].
Ayurveda and Traditional Chinese Medicine (TCM) are two of the most well-known systems for using medicinal plants to improve neurological health. In Ayurveda, herbs like Withania somnifera (Ashwagandha) and Bacopa monnieri (Brahmi) have been used for a long time to help with memory, cognition, and stress management [11]. On the other side, TCM focuses on herbs like Ginkgo biloba, Panax ginseng, and Rhodiola rosea, which were utilized in the past to bring “Qi” back into balance and clear the mind [12]. Ethnomedicinal traditions in Africa and South America exhibit significant utilization of plants for neurological health, including Tabernaemontana divaricata and Uncaria tomentosa [13]. These traditions emphasize the global applicability of herbal methods for brain health, reflecting a collective acknowledgment of natural substances in enhancing mental wellness [14].
While initially informed by empirical observation, the evolution of herbal medicine into contemporary neuropharmacology has been advanced by stringent experimental confirmation. Standardized preparations of Ginkgo biloba, which were originally mostly used as a folk treatment, have now been investigated in clinical trials for their ability to improve memory and prevent dementia [15]. Likewise, Bacopa monnieri, historically utilized as a “Medhya Rasayana” in Ayurveda, has demonstrated the capacity to enhance synaptic plasticity and memory in both animal models and clinical trials [16]. Improvements in phytochemistry have made it possible to separate active compounds such bacosides, ginsenosides, and curcuminoids, which are currently used as models for neuroactive medicines [17]. Furthermore, ethnopharmacological records have been digitized into global databases, facilitating interaction with computational drug discovery methodologies [18]. These advancements signify the transition from anecdotal applications to mechanistic and evidence-based neuropharmacology [19].
The discovery of early neuroactive chemicals from plants was very important for the development of herbal neuropharmacology. Alkaloids from Rauwolfia serpentina resulted in the identification of reserpine, an innovative antipsychotic medication that transformed psychiatry in the mid-20th century [20]. The extraction of galantamine from Galanthus woronowii (snowdrop) established a framework for acetylcholinesterase inhibitors in the treatment of Alzheimer’s disease [21]. Flavonoids and polyphenols extracted from green tea (Camellia sinensis) and turmeric (Curcuma longa) yielded initial insights into natural substances exhibiting antioxidant and anti-inflammatory properties pertinent to neurodegeneration [22]. These initial milestones demonstrate how conventional knowledge has spurred contemporary advancements in neuropharmacology, establishing a basis for subsequent mechanistic investigation [23].
Fig. 1 shows how natural substances nourish the brain and nervous system in
multiple manners. They influence neurotransmitters (like serotonin, GABA, and
norepinephrine) and ion channels that regulate (Nav1.7) to keep nerve signalling
in control. They prevent neuroinflammation by blocking NF-
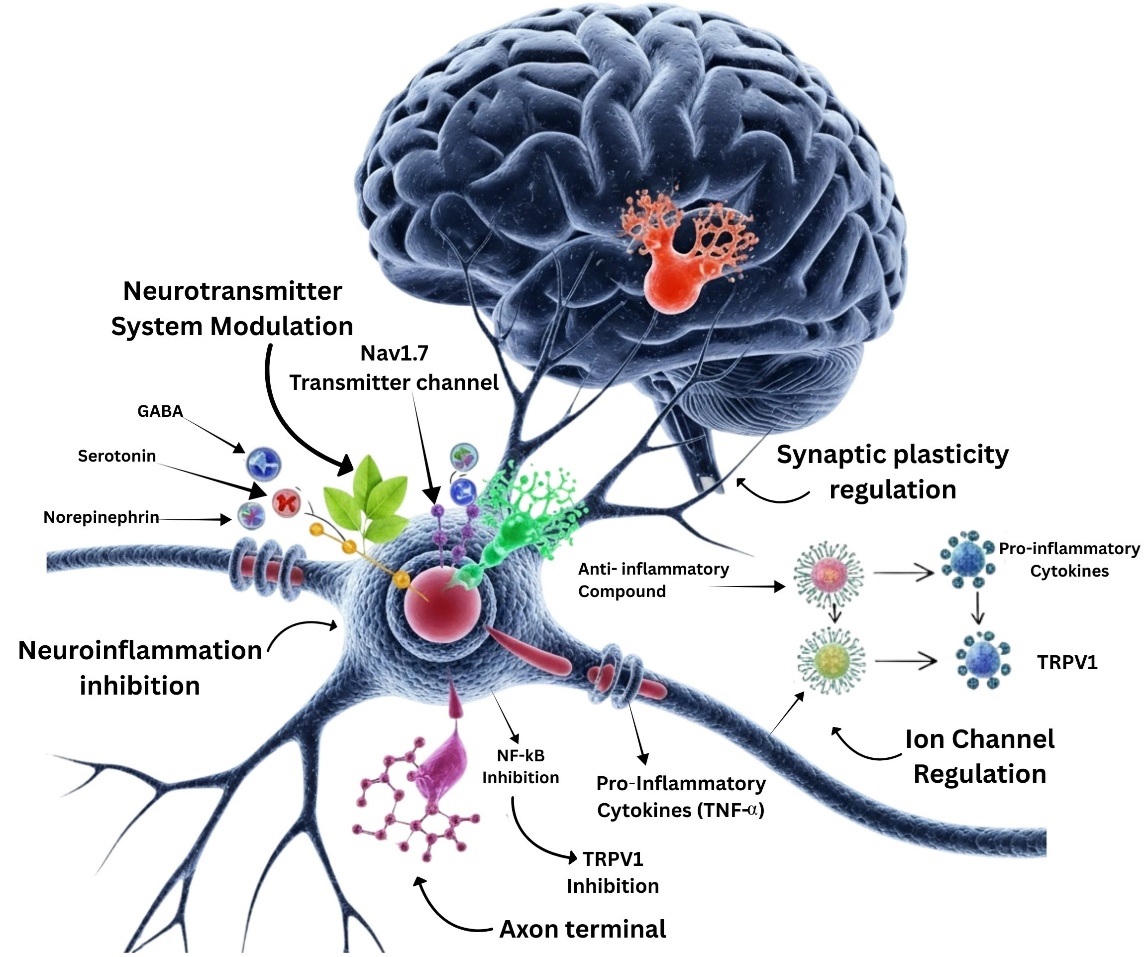 Fig. 1.
Fig. 1.
Biochemical pathway of natural substances.
Oxidative stress and chronic neuroinflammation are pivotal in the advancement of
neurodegenerative disorders, including Alzheimer’s and Parkinson’s diseases.
Herbal agents abundant in polyphenols, flavonoids, and alkaloids have
demonstrated the capacity to scavenge reactive oxygen species (ROS) and enhance
the expression of endogenous antioxidant enzymes, such as superoxide dismutase
(SOD) and catalase [24]. For instance, curcumin derived from Curcuma
longa inhibits NF-
Various herbal substances exhibit neuropharmacological effects via influencing neurotransmitter systems. Ginsenosides derived from Panax ginseng augment dopaminergic and cholinergic neurotransmission, facilitating enhancements in learning and memory [28]. Bacosides from Bacopa monnieri raise levels of serotonin and acetylcholine, which is linked to better synaptic communication in the hippocampus [29]. Alkaloids such as huperzine A also stop acetylcholinesterase, which keeps acetylcholine levels high, which is important for cognitive function [30]. These mechanistic findings illustrate the direct impact of traditional medicines on neurotransmitter circuits [31].
Apoptotic neuronal death is a defining characteristic of neurodegeneration, but compromised neurogenesis hinders recovery. Herbal chemicals like resveratrol stimulate SIRT1, which stops pro-apoptotic proteins (Bax, caspase-3) and boosts anti-apoptotic markers (Bcl-2) [32]. Withanolides from ashwagandha have been demonstrated to encourage neurogenesis in the hippocampus by encouraging synaptic remodeling and dendritic development [33]. Polysaccharides from Ganoderma lucidum also boost the expression of brain-derived neurotrophic factor (BDNF), which helps neurons stay alive and grow again [34]. These pathways underscore the potential of herbal medicines not only in avoiding neuronal death but also in promoting recovery [35].
Mitochondrial failure and reduced ATP production substantially enhance neuronal susceptibility. Herbal substances, such as gypenosides derived from Gynostemmapentaphyllum, have demonstrated the ability to stabilize mitochondrial membrane potential and augment ATP generation [36]. Curcumin and quercetin also protect mitochondria by lowering lipid peroxidation and changing the electron transport chain [37]. These effects help neurons stay strong when they’re under stress, which is good for long-term brain health [38].
Recent research underscores the significance of herbal medicines in epigenetic regulation. Polyphenols like EGCG and resveratrol control histone acetylation and DNA methylation, which in turn affect genes that are linked to neural plasticity and how the body reacts to stress [39]. For example, curcumin has been demonstrated to activate the Nrf2/ARE pathway, hence augmenting the expression of antioxidant response genes [40]. These results demonstrate that herbal substances serve as both biochemical modulators and epigenetic regulators of cerebral activity [41].
Pyroptosis, an inflammatory form of programmed cell death mediated by gasdermin activation, is increasingly recognized as a key contributor to neuroinflammation and neurodegeneration, suggesting that natural products with anti-inflammatory or inflammasome-modulating properties may exert therapeutic effects through this pathway. Similarly, ferroptosis, an iron-dependent lipid peroxidation-driven cell death mechanism, has emerged as a critical factor in neuronal loss in conditions such as Parkinson’s and Alzheimer’s diseases; yet, the potential of natural antioxidants, metal chelators, and lipid peroxidation inhibitors to modulate ferroptotic signaling remains insufficiently explored. Cuproptosis, a recently characterized copper-dependent cell death pathway linked to mitochondrial dysfunction, offers another promising frontier where natural products with metal-binding, mitochondrial-protective, or redox-stabilizing activities may play an influential role. The lack of integration of these modern mechanistic insights into current research limits our understanding of how natural compounds truly interact with neuronal survival pathways, indicating a major research gap and an opportunity for more advanced neuropharmacological investigations.
The Fig. 2 demonstrates that herbal compounds can be used in neuropharmacological advancements, such as enhancing memory functions and improving brain connections. Panax ginseng contains neuronal excitability stabilizers, while Bacopa monnieri and Curcuma longa enhance memory. Polyphenols like Epigallocatechin gallate (EGCG) and baicalein protect against neurodegenerative processes. Phytochemicals like curcumin, resveratrol, and green tea activate multiple signaling responses, suggesting a potential synergistic relationship between brain health and interventions.
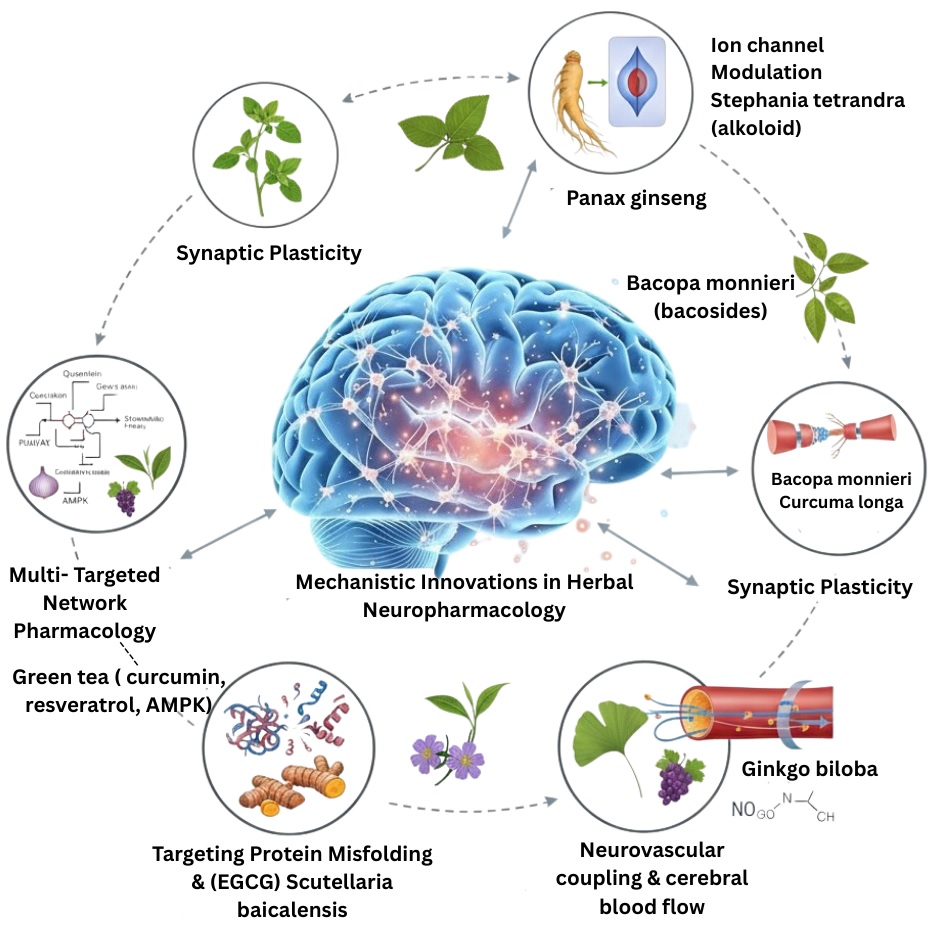 Fig. 2.
Fig. 2.
Mechanistic innovation in herbal neuropharmacology.
One of the new discoveries in herbal neuropharmacology is that phytochemicals can control how easily neurons become excited by interacting with ion channels. For example, ginsenosides from Panax ginseng control calcium and potassium channels, which keeps neurons firing and lowers excitotoxicity [42]. Likewise, alkaloids derived from Stephania tetrandra inhibit voltage-gated sodium channels, which are essential for regulating hyperexcitability associated with neuropathic pain [43]. These results indicate that herbs may function as natural templates for the creation of ion channel modulators with reduced negative effects compared to synthetic pharmaceuticals [44].
Herbal compounds have also been proven to increase synaptic plasticity, which is very important for learning and memory. Bacosides derived from Bacopa monnieri augment long-term potentiation (LTP) in hippocampal neurons through the modulation of NMDA receptor activation [45]. Curcumin enhances the phosphorylation of CREB, hence facilitating synaptic strengthening and plasticity [46]. Ginkgo biloba extract EGb761 increases the release of synaptic vesicles, which improves neurotransmission [47]. These findings collectively demonstrate the potential of herbal substances to enhance cognitive performance through the manipulation of plasticity pathways [48].
Herbal compounds also have an effect on neurovascular coupling, which makes sure that blood flows well to active areas of the brain. Ginkgo biloba extract enhances endothelial nitric oxide synthase (eNOS) activity, resulting in vasodilation and increased cerebral blood flow [49]. Resveratrol boosts the production of mitochondria in endothelial cells, which makes blood vessels even better at supporting neural activity [50]. These processes demonstrate that herbs influence not only neurons but also the vascular network that is crucial for brain function.
The formation of amyloid-beta plaques in Alzheimer’s disease and other forms of
protein misfolding and aggregation are important neurodegenerative processes.
Curcumin has demonstrated the ability to suppress amyloid-beta fibril production
and disrupt pre-existing clumps [51, 52]. Epigallocatechin-3-gallate (EGCG)
redirects harmful protein aggregates into non-toxic forms, which makes them less
harmful to the nervous system [53]. Additionally, baicalein derived from
Scutellaria baicalensis suppresses
Herbal substances work on more than one pathway at a time, which is different from synthetic medications that only work on one target. This is called a systems pharmacology approach. Research on network pharmacology has shown that drugs such as quercetin, curcumin, and resveratrol affect more than one signaling node, such as the PI3K/Akt, MAPK, and AMPK pathways [56]. This method that targets numerous pathways gives a complete therapeutic benefit, especially for complicated conditions like Alzheimer’s and Parkinson’s disease, where many harmful processes come together [57].
Numerous clinical trials have assessed herbal extracts for Alzheimer’s disease (AD) and dementia. Ginkgo biloba extract EGb761 is still the most researched herbal treatment. It has shown small but significant improvements in cognitive function and daily activities in people with mild to moderate AD [58]. A randomized controlled study (RCT) indicated that EGb761 mitigated cognitive decline similarly to donepezil in older people [59]. Bacopa monnieri supplementation similarly improved memory function and attention span in older people with mild cognitive impairment (MCI) [60]. Curcumin, despite its low bioavailability, has demonstrated decreases in amyloid-beta deposition in preliminary trials, indicating its promise in Alzheimer’s disease therapy [61].
Herbal treatments have also been tried on people with Parkinson’s disease (PD). Mucuna pruriens, a natural source of L-DOPA, enhanced motor symptoms and diminished “off” times in Parkinson’s disease patients, similar to traditional L-DOPA therapy but with a reduced incidence of dyskinesia episodes [62]. Clinical research also showed that polyphenols in green tea delayed the start of motor problems in people with early PD [63]. These results suggest that herbal medications may enhance or replace synthetic pharmaceuticals in the management of Parkinson’s disease [64].
Many studies have looked examined St. John’s Wort (Hypericum perforatum) as a treatment for depression. Meta-analyses indicate that it is comparably beneficial to selective serotonin reuptake inhibitors (SSRIs) in cases of mild to moderate depression, with fewer adverse effects [65]. In double-blind experiments, ashwagandha (Withania somnifera) has been proven to reduce cortisol levels and make people more resilient to stress [66]. These findings underscore the efficacy of herbal medicines as safer alternatives to traditional antidepressants [67].
Clinical investigations on neuroprotection post-stroke have demonstrated favorable effects of herbal substances. Ginkgo biloba extract enhanced cognitive recovery post-stroke and diminished neurological impairments in extensive randomized controlled trials [68]. Danshen (Salvia miltiorrhiza) extract has been utilized in China to enhance cerebral circulation, demonstrating a reduction in infarct volume and an improvement in recovery among ischemic stroke patients [69]. Resveratrol administration increased vascular function and cognitive recovery in post-stroke patients [70].
The table 1 highlights various herbal compounds such as Ginkgo biloba, curcumin, Bacopa monnieri, and resveratrol with neuroprotective properties that are either commercially available or being tested in clinical trials. It summarizes their sources, therapeutic potential against neurological disorders, and current development status as natural product-based interventions. Herbal supplements may help older people with normal cognitive abilities, according to trials conducted on those without dementia. A study on grape seed extract high in polyphenols found that it helped older people pay attention and remember things [71]. Supplementing with curcumin made elderly persons who didn’t have dementia feel better and remember things better [72]. Panax ginseng has been linked to enhanced psychomotor performance and less mental tiredness in healthy aging individuals [73]. These studies collectively suggest that herbal neuroprotectives can postpone cognitive deterioration associated with aging [74].
| Product (common name) | Botanical source | Indication/neuro target | Market status (example) | Representative clinical trial (NCT/ref) | Typical dose used in clinical studies | Proposed neuroprotective mechanisms |
| Ginkgo biloba (EGb 761®) | Ginkgo biloba leaf extract (standardized EGb 761) | Cognitive impairment, mild–moderate dementia, vascular cognitive impairment | Widely available as OTC supplement; EGb 761 is an approved phytomedicine for cognitive symptoms in parts of Europe. | Multiple trials—e.g., clinical efficacy studies (see NCT03090516/EGb 761 literature). | 120–240 mg/day of standardized extract in many trials. | Antioxidant, mitochondrial support, microcirculation enhancement, anti-inflammatory and anti-amyloid effects. |
| Curcumin (turmeric extract) | Curcuma longa (curcumin, standardized extracts, nanoformulations) | Alzheimer disease, age-related cognitive decline, neuroinflammation | Sold as dietary supplement; multiple enhanced formulations (nanoparticles, phospholipid complexes) in development/early clinical testing. | Several clinical studies and NCTs (e.g., NCT01383161 and other AD/cognition trials). | Typical trial ranges 500–2000 mg/day (varies by formulation; lower for enhanced-bioavailability forms). | Potent antioxidant and anti-inflammatory; modulates amyloid aggregation, metal chelation, NF-κB and Nrf2 pathways. |
| Bacopa monnieri (Brahmi) | Bacopa monnieri whole-plant standardized extracts (e.g., Bacosides) | Cognitive enhancement, memory, mild cognitive impairment | Widely marketed as nootropic/cognitive supplement globally; used in Ayurvedic formulations. | Multiple RCTs/meta-analyses (see clinical trial papers and reviews). | 300–450 mg/day of standardized extract (typical formulations 300 mg/day containing standardized bacosides). | Antioxidant, modulation of cholinergic systems, synaptic plasticity (BDNF), reduction of oxidative stress. |
| Huperzine A | Alkaloid from Huperzia serrata | Alzheimer’s disease—symptomatic cognitive improvement (acetylcholinesterase inhibition) | Marketed as supplement in some countries; used clinically in China (prescription formulations). | Multiple trials, e.g., NCT00083590 and phase II RCTs. | Typical clinical trial doses: 200–400 µg/day (varies; often 0.2–0.4 mg/day). | Reversible acetylcholinesterase inhibitor; may reduce glutamate excitotoxicity and oxidative stress. |
| Ashwagandha (Withania somnifera) | Withania somnifera root extracts (withanolides) | Cognitive dysfunction, stress-related cognitive decline, neuroprotection in AD models | Widely available as supplement (Ashwagandha extracts); several clinical trials ongoing for cognition/stress. | Examples: NCT04092647 and other trials assessing cognitive outcomes. | Typical studied doses: 300–600 mg/day of standardized root extract (depending on formulation). | Anti-inflammatory, antioxidant, modulation of neurotrophic factors (BDNF), mitochondrial stabilization, anti-amyloid effects in preclinical models. |
| Saffron (Crocus sativus) | Crocus sativus stigmas/standardized saffron extracts (crocin, safranal) | Mild–moderate Alzheimer’s disease, depressive symptoms in neuropsychiatric contexts | Available as supplement and culinary spice; standardized extracts used in clinical trials in Iran and elsewhere. | Several RCTs in AD/mild–moderate cognitive impairment (e.g., Akhondzadeh 2010 and later trials). | Typical clinical doses used in AD trials: 30 mg/day of saffron extract. | Antioxidant, anti-inflammatory, inhibition of A |
| Resveratrol | A polyphenol present in grapes, red wine; found in extracts | Alzheimer’s disease, aging-related cognitive decline, neuroinflammation | Sold as supplement; multiple clinical trials have tested high-dose resveratrol in AD populations. | Example trial: NCT01504854 and other pilot/phase II studies in AD. | Doses in trials vary widely (e.g., 500 mg–2 g/day or higher depending on study). | Sirtuin activation (SIRT1), antioxidant, anti-inflammatory, mitochondrial and metabolic modulation. |
Herbal formulations are ideal candidates for network pharmacology methods due to
their complexity and the fact that they contain several bioactive phytochemicals.
Researchers can map how different compounds affect interconnected signaling
pathways by looking at how they interact with more than one target. This can help
them understand how these compounds work together [75]. For instance, systems
biology models have shown that chemicals from Withania somnifera control
neuroinflammation through both the NF-
Herbal bioactives like curcumin, resveratrol, and quercetin still have big problems with solubility and bioavailability. Nanotechnology offers novel treatments by enhancing stability, targeted delivery, and penetration of the blood–brain barrier (BBB) [80]. For example, polymeric nanoparticles loaded with curcumin greatly improved brain absorption and stopped amyloid accumulation in models of Alzheimer’s disease [81]. Likewise, resveratrol encapsulated in lipid-based carriers exhibited enhanced neuroprotective effects in individuals with Parkinson’s disease [82]. Dendrimers and nanomicelles have been designed to co-deliver herbal polyphenols alongside synthetic medicines, resulting in synergistic therapeutic effects [83]. Furthermore, nanoparticles produced from plant extracts have demonstrated inherent neuroprotective characteristics [84].
AI and ML are changing the way herbal drugs are discovered by making it possible to forecast how phytochemicals will work. Machine learning (ML) algorithms can accurately anticipate how well phytocompounds will cross the blood-brain barrier (BBB), how poisonous they will be, and how well they will bind to their targets [85]. Deep learning-based virtual screening has discovered new neuroprotective alkaloids and flavonoids from extensive herbal chemical libraries [86]. AI-driven molecular docking and pharmacophore modeling have sped up the process of finding herbal neuroprotectives that work against both Alzheimer’s disease and Parkinson’s disease [87]. AI can also combine clinical trial data with molecular data to make herbal treatments work better [88]. These computational methods cut down on development time by a lot while still making sure that the product works and is secure.
Numerous case studies demonstrate the effective implementation of these technologies in practice. Nano-curcumin formulations have commenced clinical studies for Alzheimer’s Disease and have shown enhanced cognition scores relative to unformulated curcumin [89]. A network pharmacology-guided investigation of Ginkgo biloba revealed multitarget interactions with amyloid-beta and tau pathways, hence affirming its therapeutic significance [90]. In a similar vein, AI-assisted study of Panax ginseng metabolites uncovered novel ginsenosides exhibiting neuroprotective properties, currently undergoing evaluation in preclinical Parkinson’s disease models [91]. A synergistic application of omics and artificial intelligence to Bacopa monnieri clarified its influence on synaptic plasticity genes, correlating computational forecasts with clinical enhancements in memory [92]. These translational endeavors underscore the prospective integration of pharmacological, nanotechnological, and computational advancements into herbal neuropharmacology.
A major problem in herbal neuropharmacology is that there are no standard formulations. Herbal medicines often demonstrate considerable variability in chemical composition due to differences in plant species, geographic origin, harvesting time, and extraction methods [93]. These variances impede reproducibility in both preclinical and clinical research, leading to difficulties in determining suitable therapy dosages. Synthetic drugs contain clear active ingredients, while herbal formulations may include many bioactive ingredients that work together or against each other in ways that are not well known. The absence of internationally accepted pharmacopoeial standards exacerbates these dosing difficulties [94]. Recent progress in metabolomics and chemometric fingerprinting has been suggested as effective methods for improving uniformity; nevertheless, their practical application is still limited.
Even if they may seem safe, herbal substances can nevertheless be harmful to your health. The study has shown that certain herbal neuroactive compounds can be toxic to the liver and kidneys, and that the amount of the chemical taken can affect how toxic it is [95]. Moreover, the combinations between herbal medicines and conventional pharmaceuticals present a considerable therapeutic challenge. Ginseng and St. John’s Wort are known to interfere with cytochrome P450 enzymes, which changes how medications are broken down and raises the likelihood of bad effects [96]. A comprehensive investigation revealed that more than 30% of recorded herb-drug interactions include patients undergoing treatment for neurological or psychiatric conditions. Toxicity is linked to contaminants such as heavy metals, pesticides, and adulterants commonly present in poorly regulated herbal products [97]. To reduce these safety problems, it is important to create complete preclinical toxicological profiles and pharmacovigilance programs [98].
While herbal substances have encouraging neuroprotective properties in preclinical investigations, their conversion into clinically sanctioned pharmaceuticals is still constrained. The main problems are that preclinical models are not always the same, there aren’t many large-scale randomized controlled trials, and it’s hard to safeguard intellectual property [99]. In many situations, herbal medicines don’t get past the early clinical phases since there isn’t enough information about how they work in the body and how well they are absorbed. Regulatory challenges further postpone their approval, as herbal formulations frequently do not conform to traditional drug review criteria. Furthermore, cultural and regional disparities in the acceptance of herbal medicines affect patient adherence and clinical implementation. Bridging this gap necessitates multidisciplinary methodologies that incorporate ethnopharmacology, systems biology, and sophisticated clinical trial design [100].
There is still a lack of uniformity in the regulatory framework for herbal neuropharmacology worldwide. Due to their classification as dietary supplements rather than medicinal medicines, herbal substances in Western countries are not subject to thorough clinical testing. On the other hand, countries like India and China who have strong traditional medicine systems control herbal medications under specific rules, although they have trouble meeting international standards [101]. Ethical considerations also come up when there is biopiracy, when indigenous groups don’t get enough of the benefits, and when traditional knowledge is used without proper credit [102]. To build global trust in herbal neuropharmacology, it is important to protect intellectual property rights, have clear clinical trials, and have consistent regulatory procedures [103].
Herbal neuropharmacology is rapidly emerging as a bridge between traditional medicine and advanced biological science, revealing how plant-derived compounds can precisely modulate complex neuronal pathways. Modern tools such as nanotechnology, network pharmacology, and AI are transforming the discovery and delivery of these phytochemicals, helping overcome long-standing challenges in bioavailability, standardization, and multi-target interactions. Despite remaining regulatory and translational hurdles, the field is progressing toward globally harmonized, evidence-based practices, with the most promising pathways centered on antioxidant, anti-inflammatory, mitochondrial-protective, and metal-regulated cell-death mechanisms—positioning herbal therapeutics as a forward-looking, holistic strategy for future neurological care.
It is through combining age-old knowledge with cutting-edge research that herbal neuropharmacology hopes to realize its full potential. The use of genomic, metabolomic, and microbiome analysis to inform personalized herbal medicine has the potential to revolutionize treatment approaches by tailoring herbal substances to each patient’s unique requirements. Such customized interventions could transcend “one-size-fits-all” methodologies, guaranteeing both safety and efficacy across varied populations. New technologies in biotechnology, such CRISPR-based editing and multi-omics platforms, should speed up the discovery of new plant-based compounds and make their mechanistic paths clearer. This will make it possible to create formulations that are specific to certain pathways and can target many nodes in complicated neurological networks. Artificial intelligence and machine learning will push this frontier even farther by giving us predictive models for how herbs and drugs interact, how hazardous they are, and how to make clinical trials better. Nanotechnology and new ways to deliver drugs will also change how herbal compounds work in the body, solving long-standing problems with low bioavailability and stability. These methods will let strong natural substances like curcumin, resveratrol, and ginsenosides get to therapeutic levels in the brain with few side effects throughout the body. In the future, worldwide regulatory harmonization and ethical frameworks will be very important for making sure that new herbal treatments can be used safely in clinical practice. Herbal neuropharmacology is on the verge of a “biochemical renaissance” by encouraging cooperation between ethnopharmacology, current neuroscience, and computer sciences. In this time, substances that are inspired by nature may play a big role in creating the next generation of neurological drugs, combining old ways with new, precise medicine.
SD and PC writing original draft, editing and analyzed the data. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
Given his role as the Editorial Board member, Sumeet Dwivedi had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Mehmet Ozaslan. The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGpt in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


