1 Pharmaceutical Care Department, Ministry of National Guard—Health Affairs, Jeddah 21423, Saudi Arabia
2 King Abdullah International Medical Research Center, Jeddah 22384, Saudi Arabia
3 College of Medicine, King Saud bin Abdulaziz University for Health Sciences, Jeddah 22384, Saudi Arabia
4 Department of Pharmacology, Faculty of Medicine, King Abdulaziz University, Rabigh 25732, Saudi Arabia
Abstract
Despite nephrotoxicity concerns, liposomal amphotericin B (L-AMB) remains essential for treating invasive fungal infections in immunocompromised patients. Therefore, this study aimed to evaluate the incidence of L-AMB-associated nephrotoxicity, assess the effectiveness of nephroprotective premedication, determine treatment efficacy, and identify risk factors for adverse outcomes.
A retrospective analysis was conducted on adult hematology–oncology patients who received intravenous L-AMB for ≥7 days between January 2017 and December 2020. Patients with pre-existing dialysis dependency or acute kidney injury were excluded. The primary endpoint was the incidence of acute kidney injury within two weeks of L-AMB administration. Secondary endpoints included treatment efficacy, biomarker associations, predictors of nephrotoxicity, and the effectiveness of nephroprotective strategies.
Among the 90 analyzed patients, 46.7% (42/90) developed nephrotoxicity within two weeks of L-AMB treatment, whereas 53.3% (48/90) did not experience nephrotoxicity. Treatment efficacy was high, with 83.3% of patients avoiding breakthrough fungal infections. Median serum creatinine levels were significantly higher in the nephrotoxicity group both before and after treatment (p < 0.001). Logistic regression identified advancing age as significantly associated with higher odds of nephrotoxicity (odds ratio (OR) = 1.032; p = 0.017). Concomitant use of colistin (OR = 10.10; p = 0.008) and cyclosporine (OR = 9.01; p = 0.027) significantly increased nephrotoxicity risk. No significant association was found between the galactomannan/β-D-glucan results and breakthrough infections (p = 0.131) or between nephroprotective premedication protocols and nephrotoxicity prevention (p = 0.798).
L-AMB-associated nephrotoxicity affected 46.7% of the included hematology–oncology patients while maintaining acceptable antifungal efficacy (83.3% without breakthrough infections). Advancing age, colistin, and cyclosporine were identified as independent risk factors with additive nephrotoxic mechanisms. The absence of demonstrable benefits from premedication strategies likely reflects methodological limitations. These findings support enhanced renal monitoring for patients receiving concurrent nephrotoxic agents and highlight the need for prospective studies to optimize nephroprotective strategies.
Keywords
- liposomal amphotericin B
- nephrotoxicity
- hematology-oncology
- invasive fungal infections
- acute kidney injury
Invasive fungal infections cause severe morbidity and mortality, particularly in immunocompromised patients [1]. Patients at risk include those receiving cancer chemotherapy, patients with HIV infection, and those with acute leukemia or hematopoietic stem cell transplant recipients who experience prolonged neutropenia [2].
Amphotericin B is a polyene antifungal drug exhibiting broad-spectrum efficacy against yeasts, molds, and the protozoan parasite Leishmania species [3]. This extensive range of activity led to its recognition as the gold standard of antifungal therapy for many years [1]. However, the use of amphotericin B has been limited by its association with high incidences of infusion-related adverse events and significant toxicity, such as nephrotoxicity, liver disorder, or hypokalemia [1, 4].
Nephrotoxicity is the most clinically significant side effect of amphotericin B [5]. Key characteristics of amphotericin B-induced nephrotoxicity include elevated serum creatinine levels, diminished glomerular filtration rate (GFR), urine potassium depletion, hypokalemia, urinary magnesium depletion, and hypomagnesemia [6]. Normal-saline loading and slow-rate administration of amphotericin B are thought to decrease acute renal damage, hypokalemia, and deadly arrhythmias [5, 7].
Liposomal amphotericin B (L-AMB) is a lipid-associated formulation of the broad-spectrum polyene antifungal agent amphotericin B derived from Streptomyces nodosus [8], which was developed to reduce the toxicity of amphotericin B without compromising its antifungal activity [4]. This specific liposomal formulation reduces tissue distribution to the kidneys and drug-associated nephrotoxicity [9, 10]. In vitro and in vivo investigations have demonstrated that liposomal amphotericin B accumulates at fungal infection sites, where it adheres to fungal cell membranes and induces cell death [11, 12]. Furthermore, L-AMB remains strongly bound to liposomes in circulation, thereby reducing the risk of nephrotoxicity and infusion-related toxicity associated with conventional amphotericin B [11, 13].
Despite the advancement of liposomal formulations designed to mitigate toxicity, L-AMB remains linked to clinically significant nephrotoxicity [14]. Although certain patients may exhibit transient increases in serum creatinine that resolve post-treatment, a previous study has shown that a significant percentage of patients develop acute renal injury during therapy. Electrolyte imbalances, especially hypokalemia, are commonly observed and may occur independently of changes in serum creatinine levels. These findings collectively suggest that nephrotoxicity associated with L-AMB is reduced compared to conventional amphotericin B; however, it is not entirely eliminated, and remains a significant therapeutic concern, especially among high-risk populations [15].
A study using standardized criteria such as the RIFLE classification in patients with hematological malignancies demonstrated that approximately one-third of patients treated with L-AMB experience some degree of renal injury, with 19.4% at risk, 13.6% with injury, and 5.8% with failure, while severe renal failure is primarily seen in association with multiorgan failure [16].
Notably, most of the available data on the incidence and risk factors for L-AMB nephrotoxicity originate from Western settings [16, 17]. There is a lack of contemporary, region-specific studies applying standardized nephrotoxicity definitions in Middle Eastern settings, and little is known about the influence of local patient characteristics, clinical practices, and co-administered nephrotoxic agents in this context. Addressing this knowledge gap is vital for informing clinical practice and optimizing the management of invasive fungal infections in hematology-oncology patients in Saudi Arabia. Given insufficient local data, this study aimed to retrospectively evaluate the occurrence of nephrotoxicity among hematology-oncology patients who received L-AMB for invasive fungal infections and to assess risk factors in patients who developed nephrotoxicity at King Abdulaziz Medical City in Jeddah.
Ethical approval for this study was granted by the Ethics Committee at the King Abdullah International Medical Research Center (KAIMRC), under approval code NRJ23J/340/12. The study was designed as a retrospective chart review, utilizing clinical data extracted from the BestCare® electronic health record system. This analysis included all patients who received intravenous L-AMB from January 1, 2017, through December 31, 2020, at King Abdulaziz Medical City in Jeddah (KAMC-J).
The study cohort comprised adult hematology-oncology in patients who received
L-AMB therapy. Eligibility criteria required a minimum age of 18 years,
irrespective of gender, with documented L-AMB administration for
To assess the incidence of acute kidney injury (AKI) linked to the administration of L-AMB in the hematology-oncology cohort at King Abdulaziz Medical City-Jeddah.
This work utilized IBM SPSS Statistics version 25.0 (IBM Corp., Armonk, NY, USA)
for statistical analysis. The normality of continuous variables was evaluated by
histograms and the Shapiro-Wilk test. Demographic characteristics were presented
as frequencies and percentages for categorical variables, whereas continuous data
were expressed as means
Univariate binary logistic regression was performed for all demographic,
clinical, and medication variables (n = 35 medications). Variables with
p
This study compared the demographic and clinical characteristics of patients
with nephrotoxicity to those without nephrotoxicity (Table 1). In the
nephrotoxicity group, 69% were male, whereas the no nephrotoxicity group
comprised 60.4% males (p = 0.4). The median age was significantly
higher in the nephrotoxicity group (59 years, IQR: 35.3–73.3) compared to the no
nephrotoxicity group (47 years, IQR: 26.5–60.5, p = 0.03). Body weight
was similar between groups, with median values of 70.4 kg in the nephrotoxicity
group and 71.5 kg in the no nephrotoxicity group (p = 0.9). The median
dosage of L-AMB and treatment duration were similar between groups, exhibiting no
significant difference (p = 0.3 and p = 0.7, respectively).
Median serum creatinine levels were significantly elevated in the nephrotoxicity
group relative to the no nephrotoxicity group, both before (68.5 vs. 55
µmol/L, p = 0.01) and after to L-AMB administration (102.5
vs. 53 µmol/L, p
| Variable | Nephrotoxicity group (n = 42) | No nephrotoxicity group (n = 48) | p-value | |
| Mean | ||||
| Age | 59 (35.3–73.3) | 47 (26.5–60.5) | 0.03 | |
| Gender | 0.4 | |||
| Male | 29 (69) | 29 (60.4) | ||
| Female | 13 (31) | 19 (39.6) | ||
| Body weight (kg) | 70.40 (55.2–81.6) | 71.5 (56.8–78) | 0.9 | |
| Dose of L-AMB (mg/kg/day) | 5 (5–5) | 5 (3.7–5) | 0.3 | |
| Dose category | 0.5 | |||
| Dose |
8 (19) | 11 (22.9) | ||
| Dose |
31 (73.8) | 36 (75) | ||
| Dose |
3 (7.2) | 1 (2.1) | ||
| Duration of L-AMB (days) | 16 (6.8–28.5) | 17 (9–30) | 0.7 | |
| SCr (µmol/L), pre-L-AMB (−2 weeks) | 68.5 (52.8–90.8) | 55 (48.3–65.8) | 0.01 | |
| CrCl (mL/min), pre-L-AMB (−2 weeks) | 94.4 (48.5–120.3) | 124 (106–153) | ||
| SCr (µmol/L), post-L-AMB (+2 weeks) | 102.5 (57.3–138.5) | 53 (45.3–69.5) | ||
| CrCl (mL/min), post-L-AMB (+2 weeks) | 61 (48.4–97) | 120 (95–153.50) | ||
| K+ (mmol/L), pre-L-AMB (−24 h) | 3.9 (3.5–4.5) | 3.8 (3.5–4.2) | 0.6 | |
| K+ (mmol/L), post-L-AMB (+24 h) | 3.7 |
3.7 |
0.6 | |
| Mg2+ (mmol/L), pre-L-AMB (−24 h) | 0.83 (0.73–0.96) | 0.78 (0.69–0.87) | 0.1 | |
| Mg2+ (mmol/L), post-L-AMB (+24 h) | 0.82 (0.77–0.94) | 0.83 (0.77–0.92) | 0.7 | |
| Premedication strategy | 0.8 | |||
| Hydration + Infusion Prophylaxis | 37 (88.1) | 40 (83.3) | ||
| Infusion Prophylaxis Only | 4 (9.5) | 6 (12.5) | ||
| Hydration Only | 1 (2.4) | 2 (4.2) | ||
| Breakthrough fungal infection | 0.4 | |||
| No Breakthrough Infection | 33 (78.6) | 42 (87.5) | ||
| Breakthrough Infection | 9 (21.4) | 6 (12.5) | ||
| Indications | 0.5 | |||
| Candida species | 18 (42.9) | 20 (41.7) | ||
| Aspergillus species | 9 (21.4) | 7 (14.6) | ||
| Other fungal species | 6 (14.3) | 13 (27.1) | ||
| Empirical treatment | 9 (21.4) | 8 (16.7) | ||
Values are presented as mean
The incidence of nephrotoxicity within two weeks of L-AMB administration was 46.7%, with 42 out of 90 patients developing nephrotoxicity. In contrast, 53.3% of patients (48 out of 90) did not experience nephrotoxicity within this timeframe.
Treatment efficacy, assessed by the absence of breakthrough fungal infections
during and post-L-AMB therapy, revealed that 83.3% of patients (75 out of 90)
did not experience breakthrough infections. However, 16.7% (15 patients)
experienced breakthrough infections. Among those with breakthrough infections, a
variety of Candida species were identified, with Candida auris being the most
frequently reported, alongside Candida albicans, Candida parapsilosis, Candida
glabrata, and Candida krusei. There were also instances of possible invasive
candidiasis and heavy growth of Candida species, some associated with pneumonia.
Further analysis by L-AMB dose category did not reveal a statistically
significant association between dose level and the occurrence of breakthrough
infections (
A backward stepwise logistic regression (Backward LR) analysis was conducted to
determine factors associated with the likelihood of breakthrough fungal
infection. The analysis identified the Charlson Comorbidity Index (CCI) as a
significant predictor across multiple steps. In the final model (Step 8), a
higher CCI was associated with an increased risk of breakthrough fungal infection
(B = 0.337, p = 0.002), with an odds ratio (Exp(B)) of 1.401, indicating
that each unit increase in CCI raised the odds of breakthrough infection by
approximately 40%. Age, gender, galactomannan or
A chi-square analysis was conducted to examine the association between
Galactomannan/
To assess the risk factors associated with nephrotoxicity among patients
receiving L-AMB, a univariate (crude) binary logistic regression was initially
performed for demographic and clinical variables, including age, gender, weight,
BMI, L-AMB dose, premedication use, CCI, and all 35 concomitant nephrotoxic
medications individually. Among demographic and clinical variables, only age
showed a trend toward significance (p
The final multivariate model was adjusted for age, colistin, cyclosporine, ceftazidime-avibactam, and Piperacillin/tazobactam use. In the adjusted analysis, advancing age was significantly associated with higher odds of nephrotoxicity (adjusted OR = 1.032; 95% CI: 1.005–1.059; p = 0.017), with each additional year increasing the odds by 3.2%. Among the evaluated nephrotoxic agents, colistin use showed the strongest association with nephrotoxicity, with patients receiving colistin having 10.10 times higher odds of developing nephrotoxicity compared to those who did not (adjusted OR = 10.10; 95% CI: 1.85–55.56; p = 0.008). Cyclosporine use was also significantly associated with increased odds of nephrotoxicity (adjusted OR = 9.01; 95% CI: 1.29–62.50; p = 0.027). Although ceftazidime-avibactam showed a trend toward increased risk (adjusted OR = 5.99; 95% CI: 0.62–58.82; p = 0.122), this association did not reach statistical significance. In addition, Piperacillin/tazobactam use was not statistically significant (adjusted OR = 0.43; 95% CI: 0.10–1.81; p = 0.249) (Fig. 1).
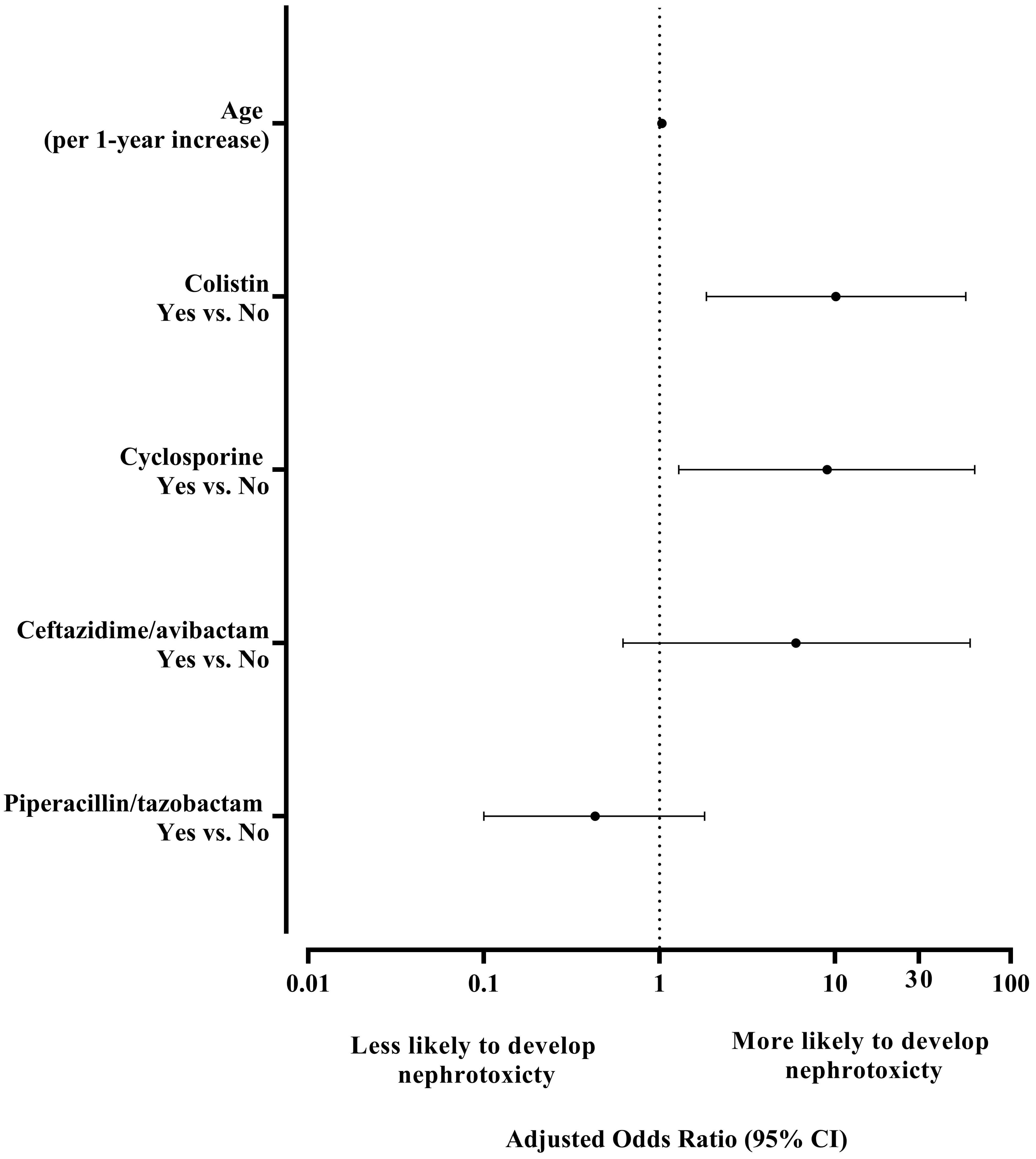 Fig. 1.
Fig. 1.
Forest plot of adjusted odds ratios from multivariate logistic regression for nephrotoxicity within 2 weeks. The vertical dashed line indicates OR = 1 (no effect). Error bars represent 95% confidence intervals.
The assessment of nephroprotective premedication protocols Prior to L-AMB
administration revealed variable distributions across nephrotoxicity groups.
Among patients who developed nephrotoxicity (n = 42), the majority received both
hydration and infusion prophylaxis (n = 37, 88.1%), followed by infusion
prophylaxis only (n = 4, 9.5%), and hydration only (n = 1, 2.4%). Similarly,
among patients without nephrotoxicity (n = 48), most received both hydration and
infusion prophylaxis (n = 40, 83.3%), while 6 (12.5%) received infusion
prophylaxis only and 2 (4.2%) received hydration only. Statistical analysis
showed no significant association between premedication strategy and
nephrotoxicity incidence (
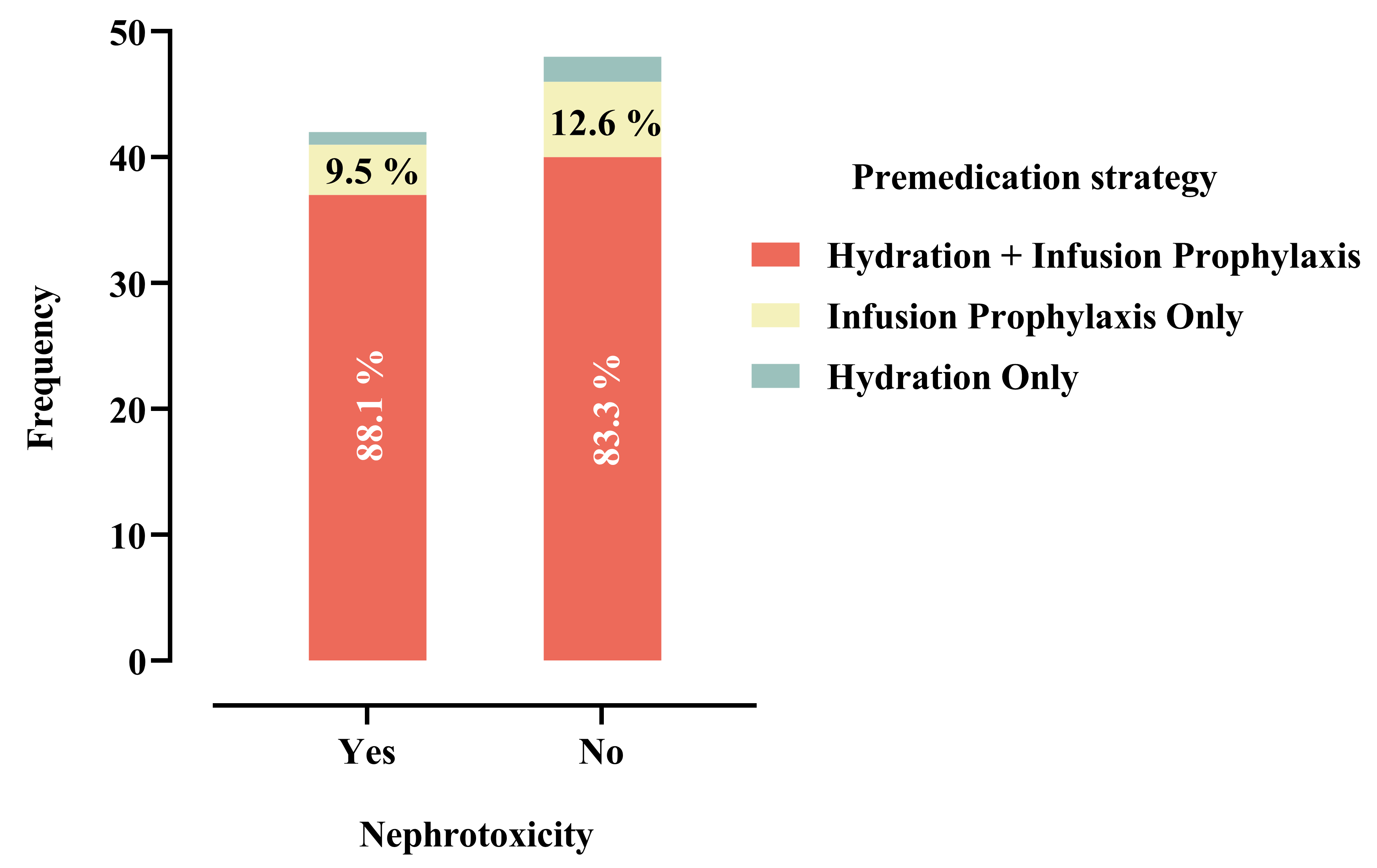 Fig. 2.
Fig. 2.
Distribution of premedication strategies among patients with and without nephrotoxicity (N = 90). Percentages shown within bars represent proportions within each group.
The primary finding of this study was that nearly half of the patients (46.7%) undergoing L-AMB treatment developed nephrotoxicity within two weeks of treatment initiation. A critical observation was that patients who subsequently developed nephrotoxicity had significantly elevated baseline serum creatinine levels (68.50 µmol/L) compared to those who remained nephrotoxicity-free (55 µmol/L, p = 0.01). Regarding treatment efficacy, L-AMB demonstrated good antifungal effectiveness, with 83.3% of patients (75 out of 90) experiencing no breakthrough fungal infections, while 16.7% (15 patients) developed breakthrough infections predominantly involving Candida species, with Candida auris being the most frequently reported pathogen. The Charlson Comorbidity Index emerged as a significant predictor of breakthrough infections (B = 0.337, p = 0.002), with each unit increase in CCI associated with a 40% increase in odds of treatment failure (OR = 1.401), highlighting the critical impact of underlying comorbidities on therapeutic outcomes. Nephrotoxicity was influenced by concomitant medications, particularly colistin (adjusted OR = 10.10, p = 0.008), which emerged as the strongest independent risk factor, followed by cyclosporine (adjusted OR = 9.01, p = 0.027). Age also contributed to nephrotoxicity risk, with each additional year associated with a 3.2% increase in odds of developing renal complications (adjusted OR = 1.032, p = 0.017). Notably, nephroprotective pre-medication protocols demonstrated minimal effectiveness in preventing nephrotoxicity, with no statistically significant association observed between full pre-medication administration and renal protection (p = 0.798).
The observed nephrotoxicity incidence of 46.7% in our cohort represents a substantial proportion of patients experiencing renal complications during L-AMB therapy. This finding aligns with contemporary reports demonstrating variable nephrotoxicity rates across different patient populations and clinical settings. A retrospective analysis of 75 patients receiving L-AMB at doses of 1, 3, and 5 mg/kg reported an overall nephrotoxicity rate of 56% [27]. Similarly, a cohort study of 103 treatment courses in patients with hematological malignancies found that 38.3% of patients developed some degree of renal injury according to RIFLE criteria, with 19.4% classified as at risk, 13.6% meeting injury classification, and 5.8% developing renal failure [16]. In contrast, a large Japanese multicenter study involving 507 patients reported a 37% incidence of acute kidney injury defined by KDIGO criteria, suggesting that nephrotoxicity definitions and patient populations significantly influence reported incidence rates [28]. The gender distribution in our study (64.4% male, 35.6% female) reflects typical demographic patterns in patients requiring antifungal therapy, though gender itself did not emerge as a significant predictor of nephrotoxicity development. These findings collectively underscore that L-AMB nephrotoxicity remains a clinically significant concern despite improved safety profiles compared to conventional amphotericin B formulations.
The high nephrotoxicity rate observed in our cohort likely reflects the unique vulnerability of hematology-oncology patients due to several compounding factors: profound and prolonged immunosuppression from underlying malignancies and cytotoxic therapies, polypharmacy involving multiple concurrent nephrotoxic agents (chemotherapy, calcineurin inhibitors, broad-spectrum antimicrobials), additional metabolic insults including tumor lysis syndrome and sepsis-related acute kidney injury, and the clinical imperative for prolonged high-dose antifungal therapy without the option of treatment discontinuation despite emerging toxicity [16].
Treatment efficacy results demonstrated acceptable therapeutic outcomes, with 83.3% of patients remaining free from breakthrough infections, which aligns favorably with historical L-AMB efficacy data. A pivotal randomized trial comparing L-AMB with conventional amphotericin B reported significantly fewer breakthrough fungal infections (3.2% vs. 7.8%, p = 0.009), with candidemia representing the predominant breakthrough pattern [20]. Recent pharmacokinetic/pharmacodynamic study suggests that standard L-AMB doses (3 mg/kg) may provide suboptimal coverage against C. auris isolates, with higher doses (5 mg/kg) potentially required for adequate therapeutic exposure [29]. The absence of dose-dependent associations with breakthrough infections in our analysis (p = 0.187) suggests that factors beyond dosing, particularly host-related variables, may be more critical determinants of treatment failure. The identification of the CCI as a significant predictor of breakthrough infections (OR = 1.401, p = 0.002) represents a clinically important finding with substantial prognostic implications. This association is consistent with extensive literature demonstrating the CCI’s utility in predicting adverse outcomes across diverse infectious disease scenarios, including candidemia, where combined comorbidity and severity scores effectively stratify mortality risk [30]. The 40% increase in breakthrough infection odds per unit CCI increase underscores the profound impact of underlying comorbidities on antifungal treatment success, likely reflecting compromised immune function, altered drug metabolism, and reduced physiological reserves in patients with multiple concurrent conditions.
The absence of statistically significant association between
galactomannan/
Multivariable logistic regression identified age, colistin, and cyclosporine as independent predictors of nephrotoxicity. advancing age was significantly associated with nephrotoxicity (adjusted OR = 1.032; p = 0.017), consistent with diminished renal functional reserve in older patients [39]. Colistin demonstrated the strongest association (adjusted OR = 10.10; p = 0.008). The mechanistic basis for this synergistic toxicity is well-established: colistin induces proximal tubular necrosis through membrane permeability changes and oxidative stress [40, 41], while amphotericin B causes renal vasoconstriction and direct tubular damage [42]. Concurrent administration produces additive nephrotoxic effects, with clinical studies reporting AKI rates exceeding 50% when polymyxins are combined with other nephrotoxic agents [41]. Cyclosporine was also independently associated with nephrotoxicity (adjusted OR = 9.01; p = 0.027). Cyclosporine induces afferent arteriolar vasoconstriction through enhanced endothelin-1 production and reduced prostaglandin synthesis, decreasing renal blood flow [43, 44]. This hemodynamic compromise potentiates amphotericin B tubular toxicity, and both agents independently deplete magnesium and potassium, compounding electrolyte disturbances [45]. The risk associated with ceftazidime-avibactam (adjusted OR = 5.99; 95% CI: 0.62–58.82; p = 0.122) reflects contemporary observations of enhanced nephrotoxicity in real-world clinical practice compared to controlled trials [46]. Piperacillin/tazobactam was not significantly associated (p = 0.249).
The absence of a statistically significant association between premedication
strategy and nephrotoxicity (Phi = 0.071, p = 0.798) warrants careful
interpretation and should not be construed as evidence that hydration is
ineffective. This finding contrasts with established evidence supporting
aggressive hydration strategies in amphotericin B therapy. Previous controlled
study has consistently demonstrated that saline loading with 1 liter of 0.9%
normal saline significantly reduces nephrotoxicity compared to dextrose
administration [47]. The landmark study by Mayer et al. [48] involving
61 patients with hematologic malignancies showed that massive hydration to ensure
urine output
Several methodological factors likely explain the discordance between our findings and established literature, preventing definitive conclusions regarding hydration efficacy. First, our premedication classification combined nephroprotective hydration with infusion-reaction prophylaxis into composite categories, potentially obscuring the independent protective effect of hydration. The majority of patients (85.6%) received both interventions simultaneously, limiting our ability to isolate the specific contribution of saline hydration to renal protection. Second, the retrospective design precluded standardized assessment of critical variables including actual volumes of intravenous fluids administered versus ordered, timing of hydration relative to L-AMB infusion, achievement of target urine output, and patient adherence to oral fluid intake recommendations. Electronic health record documentation may not accurately reflect actual fluid administration or physiological hydration status. Third, the very small number of patients in the ‘infusion prophylaxis only’ (n = 10) and ‘hydration only’ (n = 3) groups severely limits statistical power to detect a protective effect. This sample size limitation likely contributes to a Type II error, wherein a true nephroprotective benefit of hydration may have gone undetected. Fourth, in our high-risk hematology-oncology population with multiple concurrent nephrotoxic exposures and complex clinical conditions (including frequent fluid restrictions due to cardiac or pulmonary complications), the independent effect of hydration may be difficult to isolate.
These findings highlight the need for prospective studies with standardized, protocol-driven hydration regimens and objective measures of hydration adequacy to properly evaluate nephroprotective strategies during L-AMB therapy, rather than suggesting that hydration is ineffective.
This study has several limitations. The retrospective methodology limits causal
inference and may introduce selection bias, while the single-center setting may
limit generalizability to institutions with diverse patient populations or
clinical practices. The small sample size (n = 90 patients with 42 nephrotoxicity
events) may limit statistical power and increase the risk of overfitting in
multivariable analysis. A structured variable-selection technique was utilized to
mitigate this risk. Preliminary univariate (crude) logistic regression analyses
were performed for demographic and clinical factors, encompassing all 35
evaluated concurrent nephrotoxic medications. Variables with a p-value
less than 0.25 and/or demonstrated biological plausibility were deemed eligible
for inclusion in the multivariable model. Medications with very small sample
sizes (n
While our structured approach to variable selection enhanced model stability, the relatively small number of patients exposed to certain medications resulted in wide confidence intervals for some associations. For instance, the adjusted odds ratio for colistin was 10.10 (95% CI: 1.85–55.56), indicating considerable uncertainty in the precise magnitude of effect despite statistical significance. These wide intervals reflect the limited precision inherent to small sample studies and underscore the preliminary nature of our findings. Larger multicenter studies with greater numbers of exposed patients are needed to validate these associations, narrow confidence intervals, and provide more definitive estimates of nephrotoxic risk.
Additionally, potential confounders not uniformly documented in medical records, such as hydration status, timing of drug administration, or other unmeasured patient-specific variables may have affected the observed relationships. Although nephrotoxicity was defined using accepted criteria, this definition may not capture subclinical renal injury or the complete spectrum of renal failure. The lack of long-term follow-up prevents the evaluation of chronic kidney disease development or enduring renal damage.
This study demonstrates that L-AMB-associated nephrotoxicity remains clinically significant, affecting 46.7% of hematology-oncology patients. Advancing age, colistin, and cyclosporine were identified as independent risk factors in the final adjusted model, with mechanistic evidence supporting additive tubular and hemodynamic toxicity when these agents are combined with L-AMB. Ceftazidime-avibactam showed a non-significant trend toward increased risk requiring further investigation. The absence of demonstrable benefit from premedication strategies likely reflects methodological limitations rather than true ineffectiveness. L-AMB maintained acceptable efficacy (83.3% without breakthrough infections), with the Charlson Comorbidity Index predicting treatment failure. These findings support enhanced renal monitoring for patients receiving concurrent nephrotoxic agents, particularly colistin or cyclosporine, and highlight the need for prospective studies to optimize nephroprotective strategies.
Data is available on request from the corresponding author, owing to privacy/ethical restrictions.
MAA contributed to conceptualization, writing—original draft, and writing—review and editing. SA contributed to conceptualization and validation. AA contributed to conceptualization and data collection. AYA contributed to data collection. RAA contributed to methodology. MIM contributed to validation. AFA contributed to formal analysis, investigation, and resources. YAA contributed to data curation and visualization. AMA contributed to the conception and design of the study, writing—review and editing, supervision, and project administration. All authors have read and agreed to the final version of the manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki. This study was approved by the ethics committee at King Abdullah International Medical Research Center (KAIMRC) with approval number (NRJ23J/340/12). Given the retrospective design, which involved the analysis of existing anonymized data, the ethics committee granted a waiver for obtaining informed consent from participants.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/IJP47889.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


