1 College of Science, Department of Zoology, King Saud University, 11451 Riyadh, Saudi Arabia
2 Central Research Laboratory, Department of Medical Research on Experimental Animals, King Saud University, PO Box 22452 Riyadh, Saudi Arabia
Abstract
Berberine represents a promising supplement with several health benefits. Thus, this study aimed to investigate the efficacy of berberine in reducing mammary carcinoma induced by 7,12-dimethylbenz[a]anthracene (DMBA) in female mice.
A total of 40 virgin female mice were segregated into four groups: an untreated control group, a group treated with a daily oral dose of berberine (BBR) extract (25 mg/kg), a group injected with a single dose of 50 mg/kg DMBA in the breast fat pad, and a group treated with DMBA, similar to the third group. After 1 week of injections, the animals were treated with BBR extract, as in the second group.
Animals treated with DMBA for mammary carcinoma induction showed increases in liver enzymes (ALT, AST, and ALP), kidney biomarkers (BUN, UA, and CRT), and an oxidative stress marker (MDA). Meanwhile, DMBA also promoted a decrease in the antioxidant marker (GSH) and a minor elevation in estrogen levels. Conversely, the progesterone levels increased significantly, along with those of proinflammatory cytokines (interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α)). The histopathological analysis revealed carcinoma cells in the breast tissue, with a marked desmoplastic response. Meanwhile, the uterus showed hyperplasia and dysplasia of endometrial epithelia. The immunohistochemical analysis for Ki-67 expression revealed an intense immunoreaction associated with carcinoma proliferation in the breast. In contrast, the treatment with BBR extraction one week after the induction of mammary carcinoma by DMBA resulted in a significant decrease in liver enzymes and kidney biomarkers, as well as a reduced MDA level and a higher GSH level due to oxidative stress reduction, along with a variance in female hormone levels and a decrease in cytokines. Furthermore, the histopathological analysis showed a decline in the occurrence of carcinoma and desmoplastic response in the breast, as well as less hyperplasia and dysplasia of endometrial epithelia, and a lower incidence of Ki-67 expression in the breast.
This study observed that berberine extract possessed a potent effect in reducing the mammary carcinoma induced by DMBA in female mice.
Keywords
- mammary carcinoma
- DMBA
- berberine
- Ki-67
Mammary carcinoma is the most prevalent diagnosed carcinoma in women. Consequently, animal models of mammary adenocarcinoma have been developed as valuable tools for mammary cancer research to identify mechanisms of carcinoma development and associated treatment models [1]. Moreover, 7,12-dimethylbenz[a]anthracene (DMBA), a carcinogenic chemical composed of an aromatic cyclic hydrocarbon, is widely used to investigate mammary adenocarcinoma in animal models and has been shown to induce mammary carcinoma in rodent models [2]. Notably, DMBA activates the aryl hydrocarbon receptor, which represents the initial step in gene transcription, including cytochrome enzymes that play an essential role in DMBA metabolism [3]. Meanwhile, the risk factors associated with breast cancer may be non-modifiable, such as age, genetic mutations, family history, pregnancy, and breastfeeding, unlike modifiable factors, which include hormonal replacement therapy, physical activity, alcohol intake, smoking, and exposure to chemicals [4]. Clinical subtypes of breast cancer include progesterone receptor (PR)+, estrogen receptor (ER)+, HER2+, and triple-negative breast cancer (TNBC), which contribute to further complicating the diagnosis of mammary cancer [5]. Numerous histological patterns of mammary carcinoma have been observed; the most common was invasive ductal carcinoma; meanwhile, other patterns included lobular and cribriform [6].
A natural benzylisoquinoline alkaloid, berberine (BBR), which is mainly present in medicinal plants within the Ranunculaceae, Berberidaceae, and Rutaceae families, may also exist in the stem bark, rhizomes, and even roots of plants [7]. Berberine contains several natural bioactive agents, such as hydrochloride salt, which is used as an oral antibacterial agent [8]. Recently, these extracted herbal medicines, including berberine, were found to possess significant potential for treating vascular diseases [9]. Berberine is a yellowish powder with a melting point of up to 140 °C; berberine is soluble in warm water but insoluble in organic solvents [10]. Moreover, BBR has been postulated to promote a protective impact against liver injury induced by doxorubicin or tetrachloride [11]. Several investigations also showed that BBR increased the activity of antioxidant enzymes, such as catalase (CAT) and superoxide dismutase (SOD), but decreased the activity of oxidative stress markers, including nitric oxide (NO) levels and malondialdehyde (MDA) [12].
Therefore, this study aimed to determine the effect of BBR extract on the control of mammary adenocarcinoma induced by DMBA in female Albino mice.
Female Albino mice were supplied from the animal house of the central lab of King Saud University, Saudi Arabia. Animals weighed approximately 30 g and were about 14 to 15 weeks old. The mice were kept in clean plastic cages, at room temperature, and subjected to a 12 h day/night cycle. The mice were fed on commercial rodent pellets and had access to clean water.
Animals were randomly segregated into four groups with 10 mice per group: the
first group was the untreated control, the second group was treated with a daily
oral dose of BBR extract (25 mg/kg) (Paradise Company, Dania Beach, FL, USA) [13], the third group
was injected with a single dose of 50 mg/kg DMBA in the breast fat pad, the
fourth group was treated with DMBA similar to the third group; a week after the
injection, the animals were treated with BBR extract, as in the second group. The
experiment lasted for 28 days after the BBR extract treatment. Finally, the
animals were euthanized by CO2 flow with a flow rate of (2–3 L/min), the
anesthesia model was performed by placing the animal in a small chamber (40
Liver enzymes (ALT (Cat. No. 292 006 (100 T)), AST (Cat. No. 260 001, 260 002), and ALP (Catalog No. 216 001)) were determined in serum using spectrophotometer kits (Spectrum, Madrid, Spain).
Kidney biomarkers (BUN (Cat. No. 321 001), UA (Cat. No. 323 000), and CRT (Cat. No. 331 001)) in the serum were evaluated using commercial kits (Spectrum, Madrid, Spain).
Malondialdehyde (MDA, Cat. No.: E‑BC‑K025‑M) and glutathione reductase (GSH, Cat. No.: E‑BC‑K030‑S) in the serum were determined using d and spectrophotometry (Elabscience®, Wuhan, China).
Serum estrogen (Cat. No. E‑EL‑0152) and progesterone (Cat. No. E‑EL‑0154) levels were measured by enzyme-linked immunosorbent assay (ELISA) using commercial kits (Elabscience®, Wuhan, China) according to the manufacturer’s protocol.
Serum interleukin-6 (IL-6) (Cat. No.: E‑EL‑M0044) and tumor necrosis
factor-alpha (TNF-
Breast and uterus samples were collected and fixed in 10% formalin. Then, the
samples were dehydrated in ascending grades of alcohol. The samples were embedded
in paraffin wax, then sectioned at 6 µm and dried. Some sections
were stained using hematoxylin and eosin, and others were stained in Masson’s
trichrome. The sections were then imaged using light microscopy (Nikon, Tokyo,
Japan). The sections were graded according to the Nottingham histopathological
score [14] as follows: grade 0, scores 1–2; grade I, scores 3–5; grade II,
scores
Detection of Ki-67 expression in breast tissue using the Avidin biotin complex method (ABC). The sections were deparaffinized and rehydrated, then unmasked in a citrate buffer in a microwave for 5 min, washed with phosphate-buffered saline (PBS) buffer 3 times, incubated with a hydrogen peroxide blocking solution, and then a protein block was applied after washing. The samples were incubated with a primary antibody at –4 °C in the dark overnight. Simply, the sections were washed and incubated with a secondary antibody for 1.5 hours, then washed and incubated with the avidin–biotin complex for 1 hour. After washing, the sections were incubated with DAB for 30 min, then dehydrated and mounted. The sections were imaged using light microscopy (Nikon, H550S, Tokyo, Japan).
The presented study used five images per sample (fields were imaged at
400
Data are expressed as the mean
A single dose of 50 mg/kg DMBA significantly increased liver enzyme levels (ALT, AST, and ALP) compared with the control group (Fig. 1). Meanwhile, treatment with BBR after DMBA resulted in a significant decrease in liver enzyme levels compared to the DMBA group.
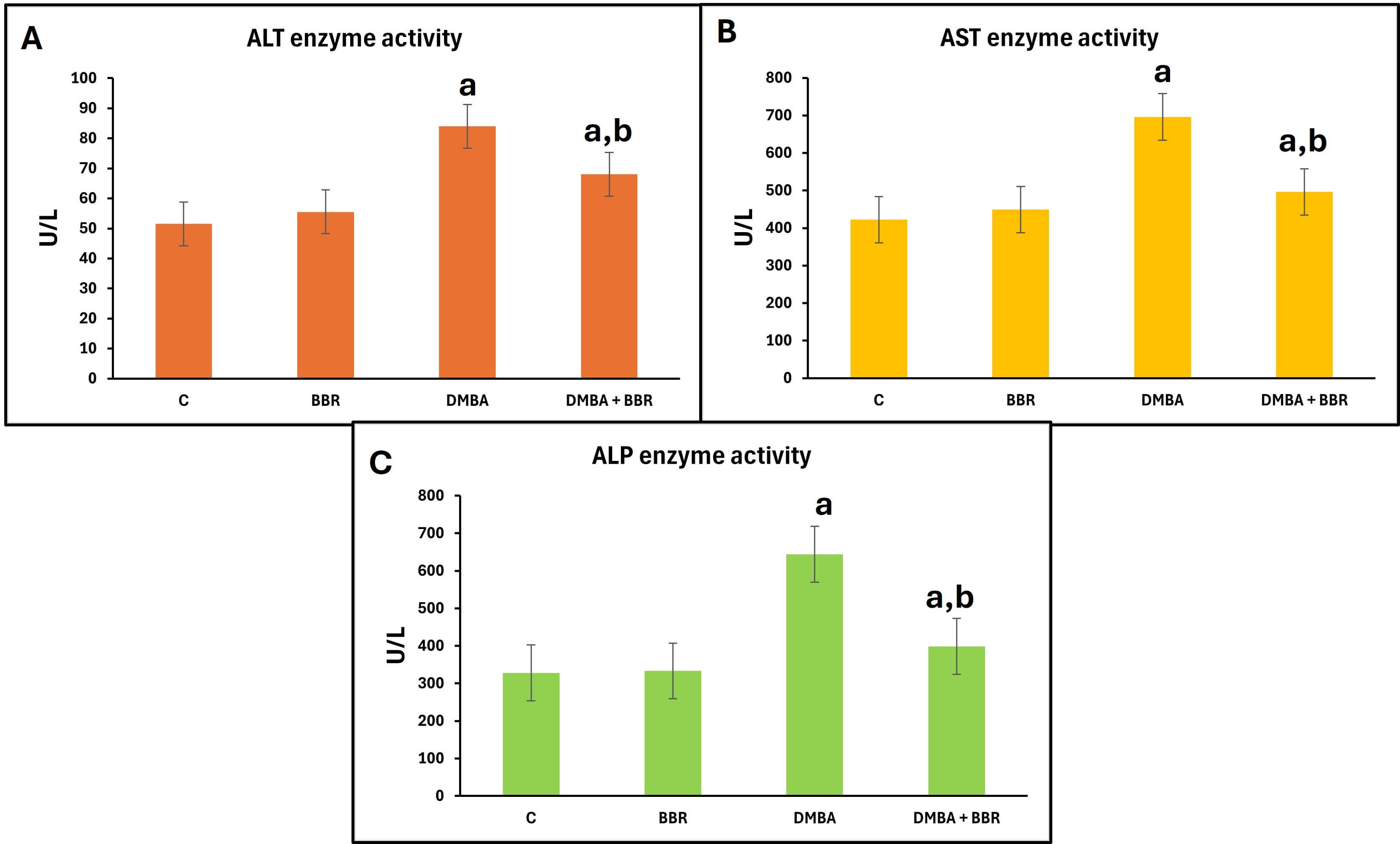 Fig. 1.
Fig. 1.
The effect of BBR extract on liver enzymes activities. Bar
graph of the liver enzyme activities: (A) ALT activity, (B) AST activity, (C) ALP
activity. DMBA promoted a significant increase in the liver enzymes, which
decreased with BBR extract treatment. Bars represent the mean; p
The investigation of kidney biomarkers showed a significant elevation in BUN and UA levels in the DMBA-treated group. Meanwhile, animals treated with BBR extract after DMBA presented a significant decline in BUN and UA levels compared to the DMBA group. The CRT levels did not change across experimental groups during the experiment (Fig. 2).
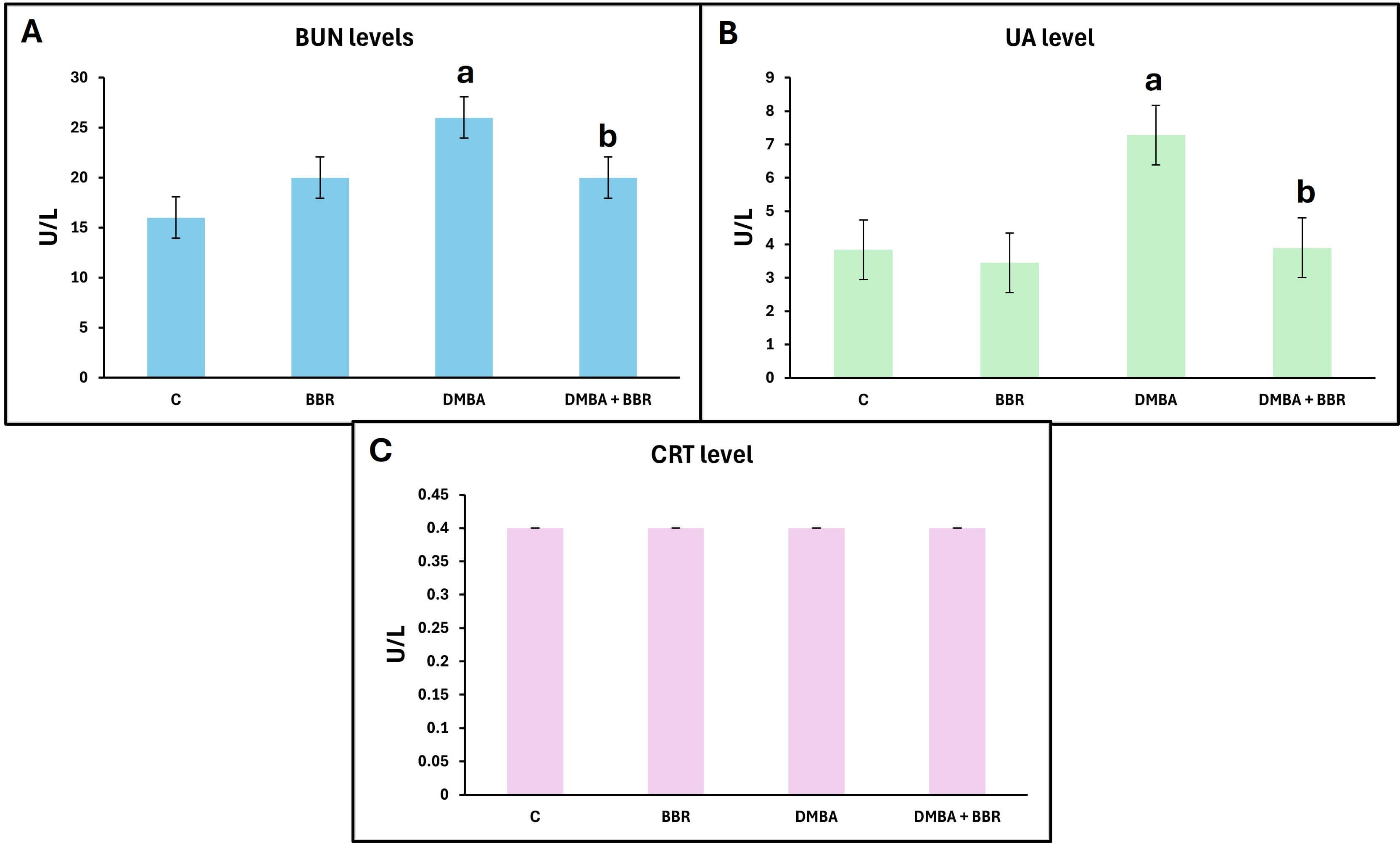 Fig. 2.
Fig. 2.
Effect of BBR extract on kidney biomarkers. Bar graph of the
kidney biomarkers. (A) BUN Blood Urea Nitrogen level, (B) UA Uric Acid level, (C)
CRT creatinine level, DMBA showed significant increases in kidney biomarkers,
with decreases following BBR treatment. Bars represent the mean;
p
DMBA markedly increased MDA levels, a lipid peroxide end product, indicating elevated oxidative stress. Moreover, the GSH levels were significantly decreased within the same group, indicating reduced antioxidant activity. However, treatment with BBR extract after DMBA-induced breast cancer significantly reduced MDA levels and increased GSH levels, indicating the ability of BBR extract to reduce oxidative stress and enhance antioxidant capacity (Fig. 3).
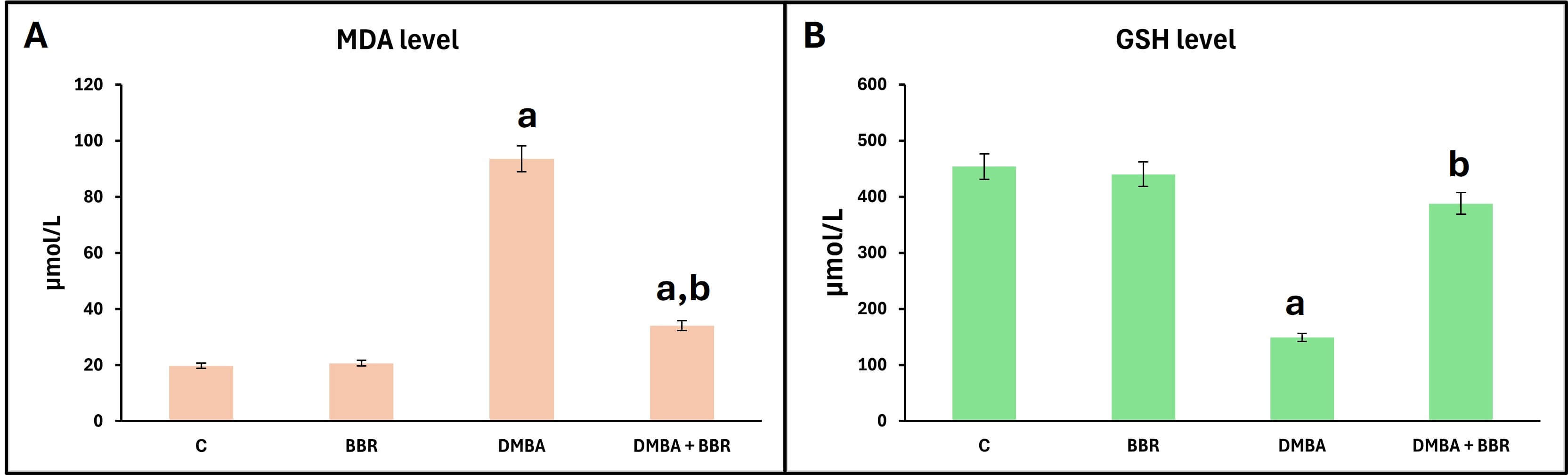 Fig. 3.
Fig. 3.
Effect of BBR extract on oxidative stress. Bar graph of the
oxidative stress biomarkers, (A) MDA level, (B) GSH level. DMBA promoted a
significant increase in MDA level and a decrease in GSH level. The BBR extract
significantly decreased MDA levels and increased GSH levels. Bars represent the
mean; p
The estimate of the estrogen hormone levels showed minor changes across the experimental groups (Fig. 4). Moreover, progesterone levels were significantly higher in the tumor group (animals treated with DMBA) than in the control group. In contrast, the groups treated with the BBR extract after tumor induction showed a significant decrease compared to the tumor group.
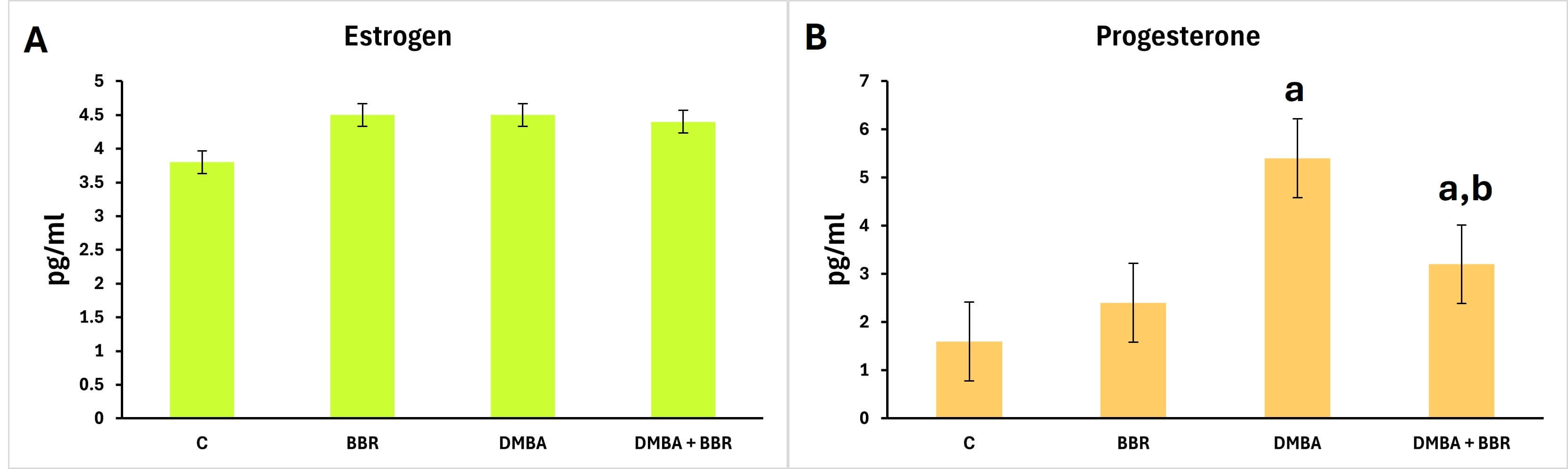 Fig. 4.
Fig. 4.
Effect of BBR extract on female hormones. Bar graph of
the hormone estimation. (A) Non-significant changes in estrogen hormone levels
are observed across groups; (B) a significant increase in progesterone in the
tumor group, and a significant decrease in the tumor group treated with BBR
extract. Bars represent the mean; p
The estimated cytokine levels (IL-6 and TNF-
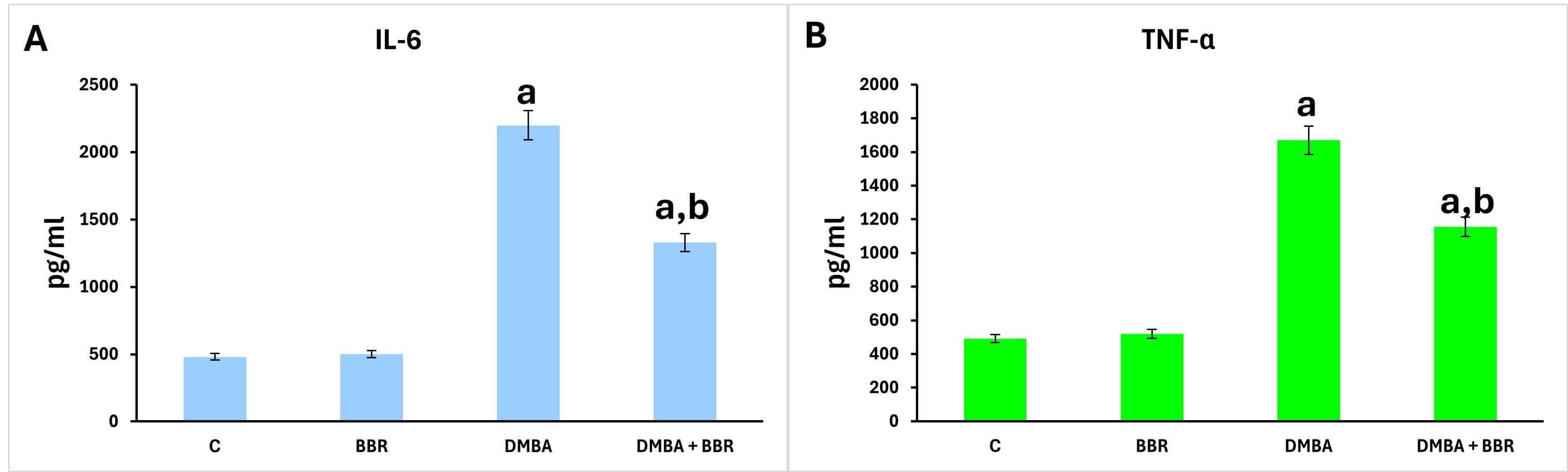 Fig. 5.
Fig. 5.
Effect of BBR extract on cytokines. Bar graph of the cytokine
determination. A significant rise in IL-6 and TNF-
The control breast images contained no view of the mammary glands in the adipose tissue (Fig. 6A); the breast images of the animals treated with BBR extract revealed no pathological changes, as in the control (Fig. 6B). Meanwhile, the breast images of the animals treated with DMBA demonstrated an induction of a mammary carcinoma manifested by hyperplasia of epithelial cells, as carcinoma cells formed along with desmoplastic stroma (Fig. 6C), which registered grade II and scored 7 (Table 1). Additionally, the breast images of the animals treated with BBR extract after induction of breast tumor by DMBA, showed a reduced presence of mammary carcinoma (Fig. 6D), which registered grade I and scored 5 (Table 1).
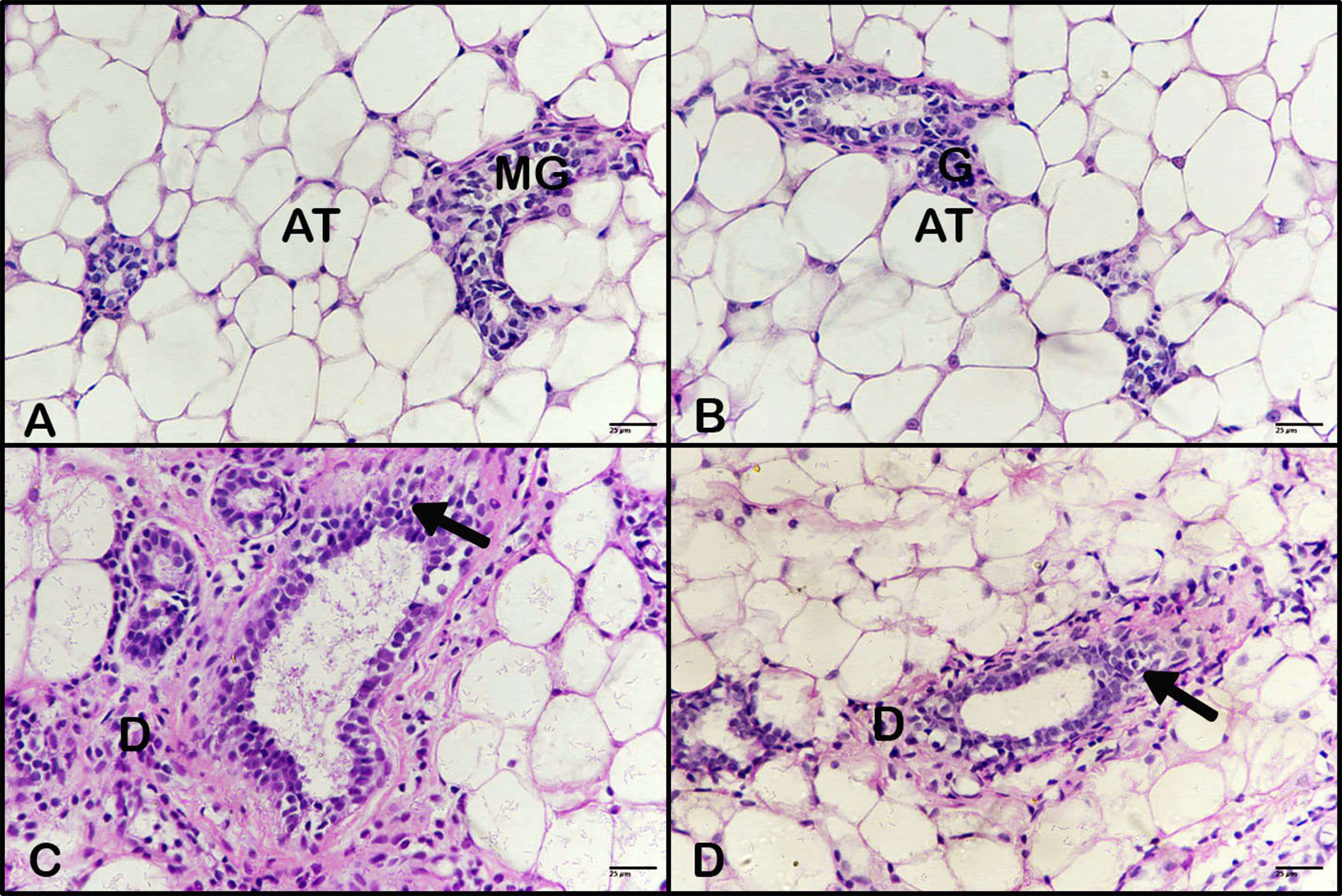 Fig. 6.
Fig. 6.
Effect of BBR extract on breast cancer. Photomicrograph of
breast tissues. (A) Control breast tissue and (B) breast tissue of an animal
treated with BBR extract revealed no pathological changes. (C) Breast tissue from
animals treated with DMBA showed carcinoma cells and a desmoplastic reaction (D).
Breast tissue from animals treated with BBR extract after tumor induction showed
fewer carcinoma cells and a reduced desmoplastic reaction. (H&E, 400
| Groups | Grades | Score |
| C | 0 | 0 |
| BBR | 0 | 0 |
| DMBA | II | 7 |
| DMBA + BBR | I | 5 |
Breast tissue stained by Masson trichrome showed no desmoplastic reactions in the control, as did the breast tissue of animals treated with BBR extract (Fig. 7A,B). Conversely, the breast tissues of animals treated with DMBA displayed an intense presence of desmoplastic stroma as a response to the mammary carcinoma induced (Fig. 7C), with a high percentage and optical density of stain (Fig. 8). Moreover, the breast tissues of animals treated with BBR extract after mammary carcinoma induction revealed a reduced desmoplastic response (Fig. 7D), with a lower percentage and optical density of staining (Fig. 8).
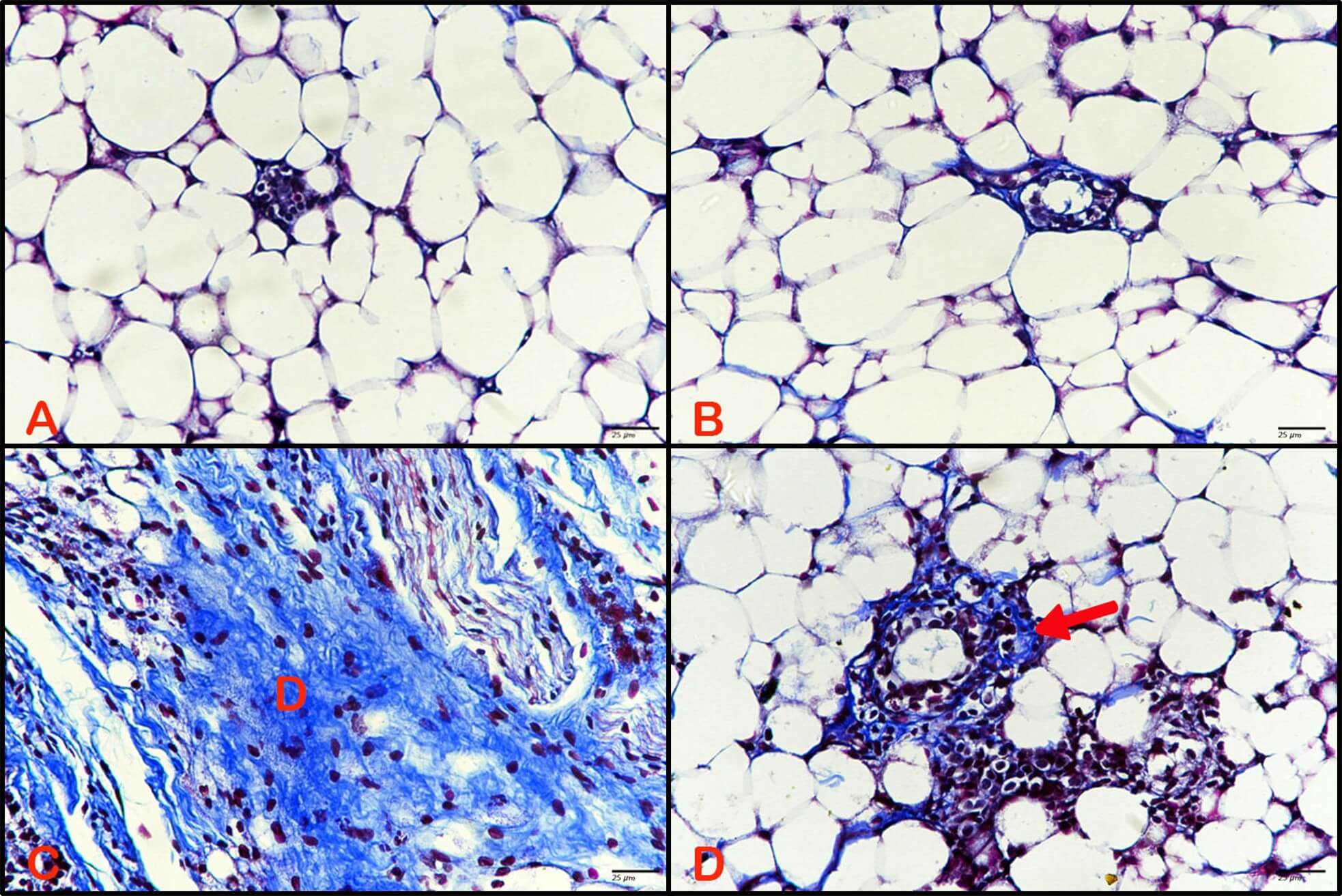 Fig. 7.
Fig. 7.
Effect of BBR extract on desmoplastoc reactions of
breast cancer. Photomicrograph of breast tissue stained with Masson trichrome.
(A) Control breast tissue and (B) breast tissue of animals treated with BBR
extract revealed no desmoplastic reactions. (C) Breast tissue of animals treated
with DMBA displayed (D) intense desmoplastic reaction. (D) Breast tissue from
animals treated with BBR extract after tumor induction showed less desmoplastic
stroma (red arrow). (Masson trichrome, 400
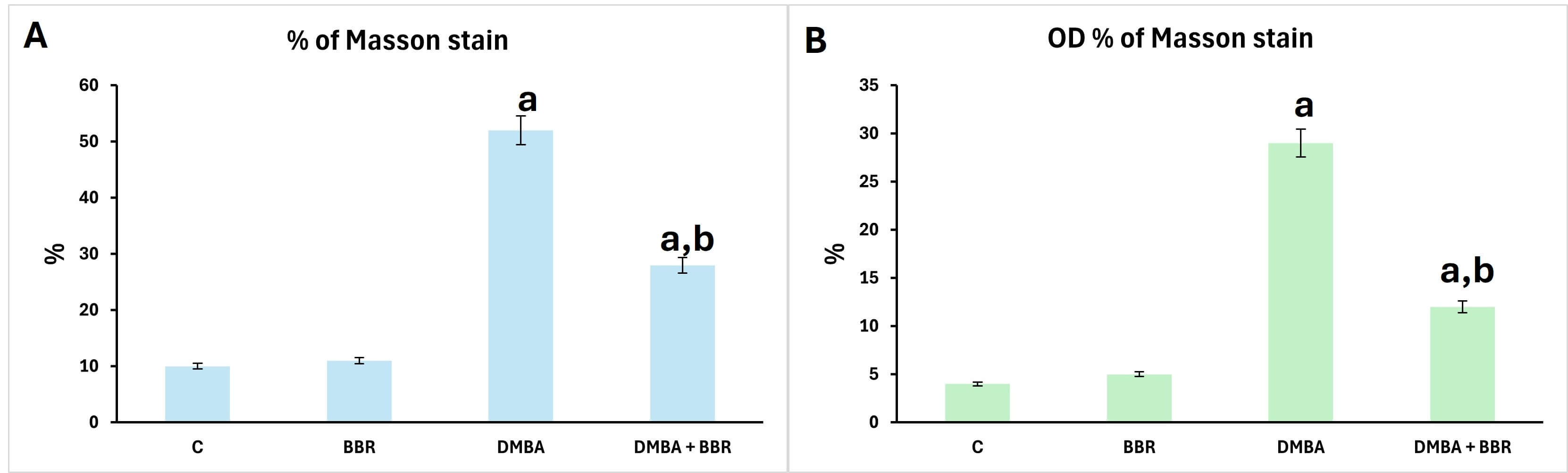 Fig. 8.
Fig. 8.
Morphometrics of Masson trichrome stain. Bar chart of Masson trichrome stain metrics showing significant increase of distribution percentage and optical density of Masson trichrome stain. (A) Percentage, (B) optical density. a, significant compared to control; b, significant compared to DMBA group.
The uterus of control animals showed a normal structure (Fig. 9A). Additionally, the uterus of animals treated with BBR extract showed no pathological alterations (Fig. 9B). However, the uterus of animals treated with DMBA revealed hyperplasia and dysplasia of epithelia, referring to early tumor formation (Fig. 9C). Furthermore, the uterus of animals treated with BBR extract after DMBA showed less hyperplasia and dysplasia of endometrial epithelia (Fig. 9D).
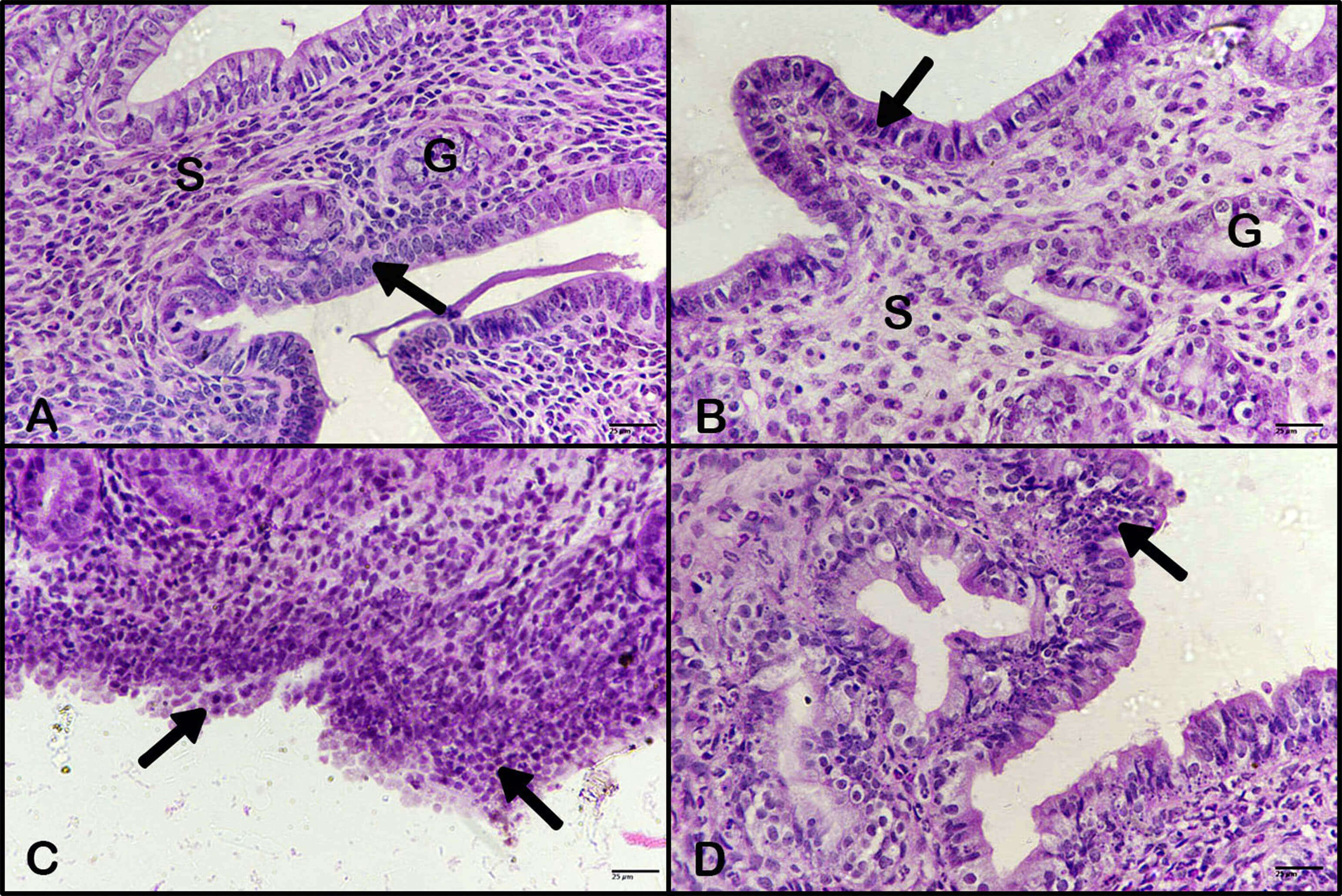 Fig. 9.
Fig. 9.
Effect of BBR extract on hyperplasia of uterus.
Photomicrographs of the uterus. (A) Control uterus, (arrow) epithelia, (G)
uterine gland, (S) stroma. (B) The uterus of animals treated with BBR extract
showed no pathological changes, stroma (S), uterine gland (G). Black arrow indicates epithelia. (C) The uteri of
animals treated with DMBA showed marked hyperplasia and dysplasia of the
endometrial epithelium (black arrows). (D) The uteri of animals treated with BBR
extract after DMBA treatment showed less hyperplasia of endometrial epithelium
(black arrow). (H&E, 400
Control and breast of animals treated with BBR extract showed a negative response of Ki-67 expression (Fig. 10A,B). However, the breast of animals treated with DMBA displayed an intense immune response (Fig. 10C), which is considered evidence of more proliferation of carcinoma cells, with a high percentage and optical density of expression stain (Fig. 11). However, the breast tissues of animals treated with BBR extract after mammary carcinoma induction by DMBA revealed a reduced immune response (Fig. 10D), referring to decreased activity of carcinoma cells in proliferation, with a lower percentage and optical density of Ki-67 expression (Fig. 11).
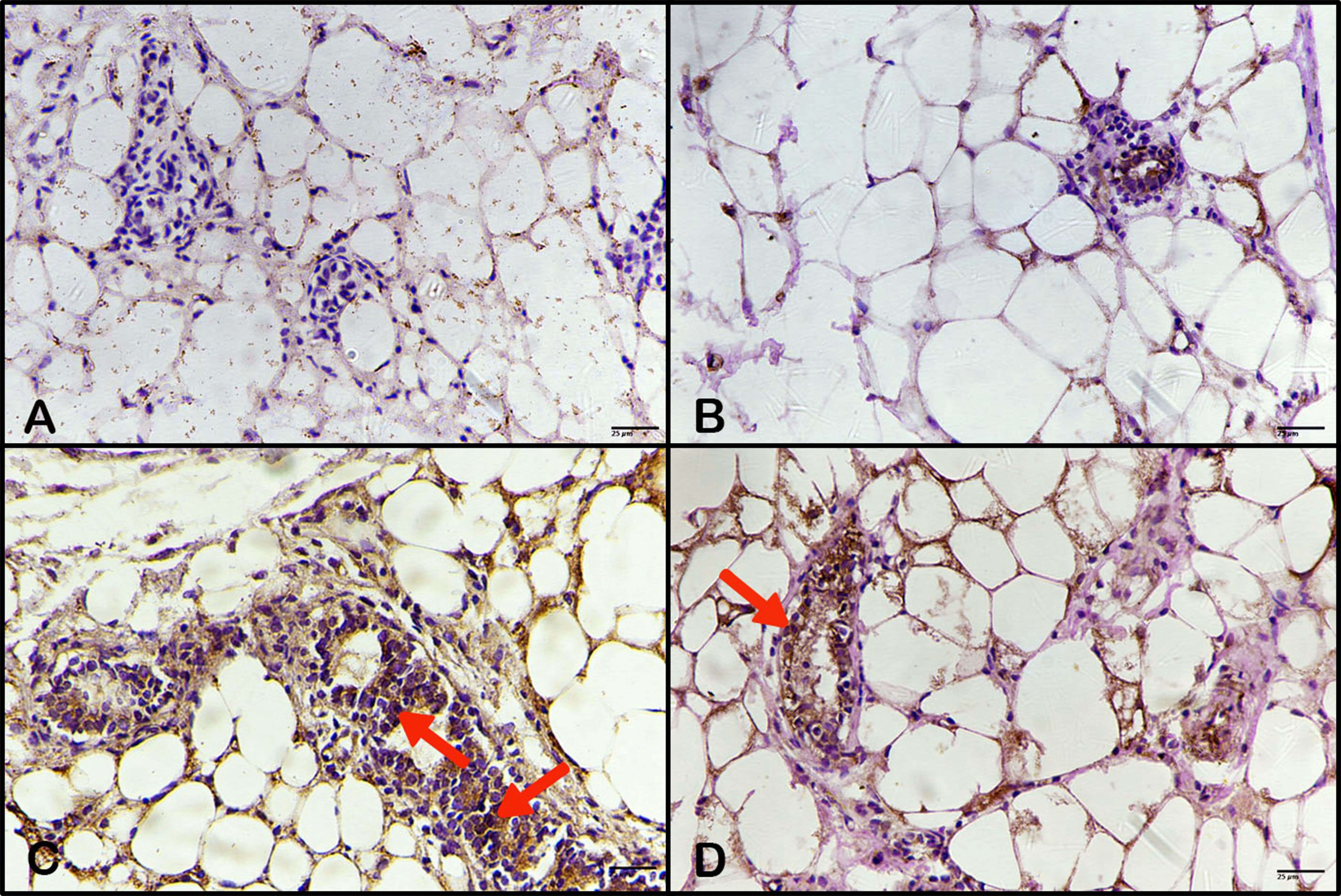 Fig. 10.
Fig. 10.
Effect of BBR extract on proliferation of tumor cells by
Ki-67 detection. Photomicrographs of mouse breast tissue stained
immunohistochemically for Ki-67 expression. (A) The control showed a
negative response. (B) Breast tissue of animals treated with BBR extract showed a
negative response; (C) breast tissue of animals treated with DMBA displaying an
intense immune response (arrows); (D) breast tissue of animals treated with BBR
extract after DMBA posting, showed a decreased immune response (arrow). (ABC,
400
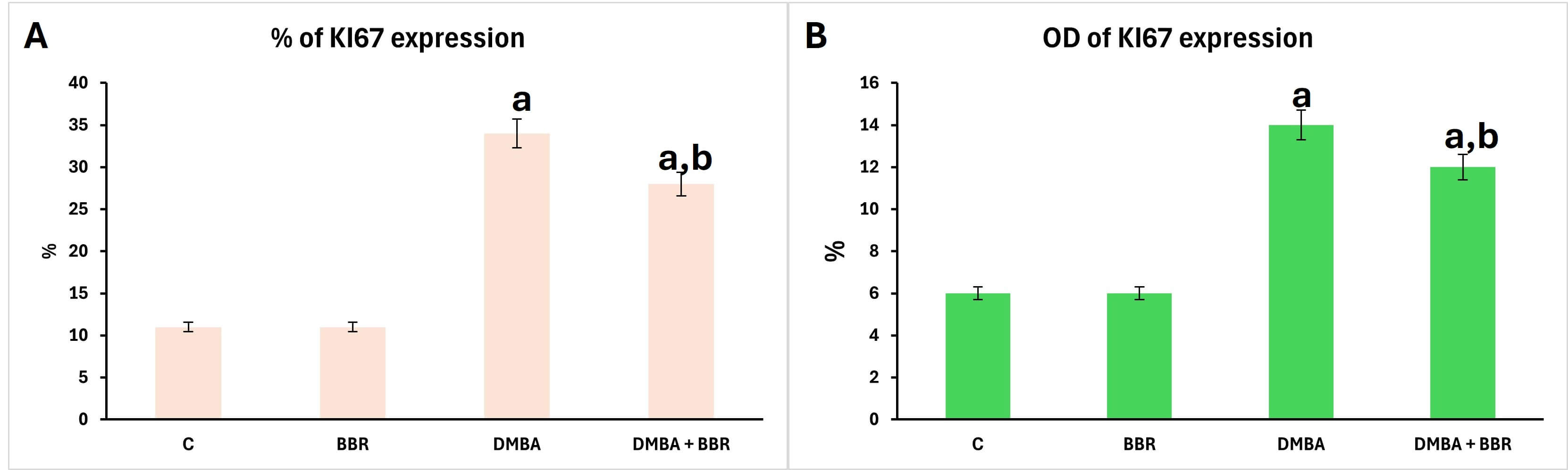 Fig. 11.
Fig. 11.
Morphometrics of Ki-67 expression immune stain. Bar chart of Ki-67 distribution percentage and optical density after treatment with BBR extract compared to DMBA group. (A) Percentage, (B) optical density. a, significant compared to control group; b, significant compared to DMBA group.
This study aimed to investigate the efficacy of berberine in reducing mammary carcinoma in female mice induced by DMBA. Mammary carcinoma can be induced in different rat and mouse strains after a single dose of carcinogens, such as DMBA, or other agents, including nitrosomethylurea (NMU). The induced tumors were found to be invasive to the breast fat pad. The induced tumor was highly hormone-dependent and, therefore, is widely used as an animal model of estrogen and progesterone-dependent breast cancers [2]. The present study revealed that a single dose of DMBA (50 mg/kg) promotes the onset of mammary carcinoma. Prior studies have suggested that DMBA mediates carcinogenesis through DNA destruction and the deregulation of critical genes involved in cell proliferation and survival [15]. DMBA has also been reported to activate the aryl hydrocarbon receptor (AhR), a factor that might regulate certain genes and contribute to cellular metabolism [16]. AhR-dependent could upregulate cytochrome P450 enzymes that metabolize DMBA into a mutagenic intermediate that induces DNA destruction and initiates tumorigenesis [17].
This work revealed that injecting DMBA causes a significant elevation in liver enzyme levels. A previous study reported that administering DMBA significantly increased ALT, AST, and GGT levels due to hepatocyte damage caused by DMBA, which destroyed hepatocyte DNA and led to the release of enzymes into the bloodstream, thereby elevating enzymatic activities [18]. Moreover, the present findings show that BBR extract treatment after the induction of breast tumor by DMBA caused a significant decrease in liver enzymes. Previous observations have shown that BBR extract could reduce these elevated liver enzymes associated with non-alcoholic fatty liver disease, as BBR might inhibit hepatocyte damage [19].
A polycyclic aromatic hydrocarbon causes several toxic effects; one of the most severe was nephrotoxicity, as kidneys exposed to this potent carcinogen showed a significant increase in BUN and CRT [20]. The present results also confirmed that injecting a single dose of DMBA into the breast fat pad to induce mammary carcinoma led to a marked increase in kidney biomarkers, especially BUN and UC. Additionally, the present results showed that administering BBR extract significantly reduced kidney biomarkers elevated by DMBA injection. Another study reported that BBR extract reduced kidney impairment and suppressed podocyte dysfunction, as evidenced by reduced kidney biochemical markers in diabetic animals, via targeting the phosphatidylinositol 3-kinase–protein kinase B pathway [21].
During DMBA-induced breast carcinogenesis, reactive oxygen species (ROS) and DMBA–DNA adducts are produced [2]. DMBA, then bound to cellular membranes, initiated peroxidation of polyunsaturated fatty acids, and statistically increased oxidative stress by raising MDA levels and decreasing antioxidant biomarkers such as CAT, SOD, and GSH [22]. Accordingly, this work showed that DMBA injection elevated MDA levels and reduced GSH levels, indicating intense oxidative stress. This work also showed that treatment with BBR extract after DMBA-induced carcinogenesis significantly reduced oxidative stress and enhanced antioxidant capacity. Furthermore, previous observations reported that MDA levels declined and glutathione (GSH) levels increased when BBR extract was co-administered with a high-fat diet; therefore, BBR could attenuate oxidative stress by activating antioxidative pathways [23].
The presented study revealed that DMBA used as a mammary carcinoma inducer
resulted in the increase of female hormones estrogen and progesterone, which
indicated that the tumor induced by DMBA was hormone-dependent. It was observed
in a prior study that, in virgin female Sprague-Dawley rats, the immune
expression of the prognostic factors estrogen receptor
This work showed that injecting DMBA into the breast fat pad significantly
increased proinflammatory cytokine levels (IL-6 and TNF-
Our results showed that a single injected dose of DMBA into the breast fat pad induced mammary carcinoma cells, accompanied by inflammatory cells and a marked desmoplastic reaction in the breast. Moreover, DMBA caused hyperplasia and dysplasia of the uterine endometrial epithelium, along with high Ki-67 expression in breast tissue, indicating a strong proliferative activity of carcinoma cells. Another study reported the formation of neoplastic tumor cells, along with lymphocytes, in the breast following DMBA injection into the breast fat pad [33]. Alternatively, these current observations showed that treatment with BBR extract after the induction of breast tumors resulted in reduced carcinoma, a desmoplastic response in the breast, less hyperplasia and dysplasia of epithelia in the uterus, and a lower Ki-67 expression in the breast. A previous study showed that BBR extract could reduce DMBA-induced ductal carcinoma and invasive carcinoma, likely due to active bioactive compounds in the extract [13].
This study concludes that the carcinogen DMBA could damage DNA and induce mutations in cells, leading to carcinogenesis. When localized in the breast through the fat pad injection, the DMBA induced mammary carcinoma, also causing an increase in liver enzymes, kidney biomarkers, oxidative stress, and female hormones, along with a heavy induction of mammary carcinoma in the breast with a desmoplastic response and hyperplasia in the endometrial epithelia of the uterus. However, berberine extract could modify biochemical markers and oxidative stress altered by DMBA due to its bioactive components; moreover, berberine extract decreased the presence of carcinoma, desmoplastic response in the breast, and the reduced hyperplasia of epithelia in the uterus.
The datasets used in this study are available from the corresponding author upon reasonable request.
DME, design of experiment, practical part, writing and revision. IAA, animal housing, injections, dissection and practical part. ASA, practical part and writing. WSAL-Q, practical part and statistical analysis. EA, practical part. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The experiment was carried out according to the Institutional Review Board (IRB), King Saud University, Riyadh, Saudi Arabia. The project was approved with the ethics reference No. KSU-SE-21-67, experiment have complied according to King Saud University for animals walefare, Saudi Arabia.
The authors express gratitude for the fund received from The Research Institute/Center Supporting Program (RICSP-26-3), King Saud University, Riyadh, Saudi Arabia. The content underwent thorough linguistic review and editing by HD Global Nexus Company., 6 October City, Giza, Egypt.
The project was funded by The Research Institute/Center Supporting Program (RICSP-26-3), King Saud University, Riyadh, Saudi Arabia.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.











