1 Technology Industry Development Center, Chongqing Medical and Pharmaceutical College, 400030 Chongqing, China
2 College of Pharmaceutical Sciences, Southwest University, 400715 Chongqing, China
Abstract
The present study aimed to elucidate the mechanism of action of the dried roots of Vladimiriae Radix against benign prostatic hyperplasia (BPH) using network pharmacology, molecular docking, and cell-level experimental verification technologies, thereby providing experimental evidence for basic research, clinical application, and the modernization research of Tibetan-Chinese medicine integrated medication.
Firstly, the active components of Vladimiriae Radix were screened using the TCM Systems Pharmacology Database and Analysis Platform (TCMSP) and PubChem, with the criteria of oral bioavailability (OB) ≥30% and drug-likeness (DL) ≥0.18. Subsequently, the SwissTargetPrediction database was used to identify potential targets for the components, and an overlap analysis was conducted on the BPH-related targets from GeneCards, Online Mendelian Inheritance in Man (OMIM), and the Therapeutic Target Database (TTD) to identify the common targets. Then, STRING and Cytoscape 3.10.3 analyses were used to construct the protein-protein interaction (PPI) network and the “Chinese medicine-component-target-disease” network for screening core targets. Gene ontology (GO)/kyoto encyclopedia of genes and genomes (KEGG) enrichment analyses were performed using the Database for Annotation, Visualization and Integrated Discovery (DAVID) database and bioinformatics platforms. Discovery Studio 2019 was used to verify the binding between components and targets, and AutoDockTools1-2 was employed to calculate the binding energy. Cell-level experiments (CCK-8 assay and RT-PCR) were conducted using BPH-1 cells to validate the effect of the representative component oleanolic acid.
A total of 235 common targets were identified between Vladimiriae Radix and BPH, and 6 core targets, including AR, CYP17A1, CYP19A1, ACHE, F2, and HMGCR, were further screened. These core targets are mainly involved in biological functions such as steroid hormone response, cellular response to nutrient levels, and regulation of membrane potential, and are enriched in BPH-related pathways including lipid and atherosclerosis, cholinergic synapse, and AGE-RAGE. Molecular docking verification found that the active components form stable bindings with the core targets. Cell experiments found that oleanolic acid significantly inhibits BPH-1 cell proliferation and regulates the mRNA expression of the six core targets at concentration of 10 μM, 20 μM, 40 μM (significantly downregulated the mRNA expression of AR and HMGCR (p < 0.05), significantly upregulated the mRNA expression of CYP17A1, CYP19A1, and ACHE (p < 0.05), and had no significant effect on F2).
Costunolide, dehydrocostus lactone, luteolin, quercetin, taraxasterol and oleanolic acid are the main bioactive ingredients in Vladimiriae Radix. Among them, oleanolic acid exhibited the highest binding energy with 6 core targets and exhibits anti-BPH properties. The present study fills the research gap in the anti-BPH mechanism of Vladimiriae Radix, validates the efficacy of the active components in Vladimiriae Radix at the cellular level, and provides clear targets and theoretical support for subsequent pharmacological verification, active component development, and clinical translation.
Keywords
- Vladimiriae Radix
- network pharmacology
- molecular docking
- benign prostate hyperplasia
- pharmacological action
Benign prostatic hyperplasia (BPH) is a common benign disease among elderly men [1]. Its pathological features include the proliferation of epithelium, fibromuscular tissue, glands, and stromal cells in the transition zone of the prostate and the perurethral area, which in turn leads to benign enlargement of the prostate. This condition is often accompanied by bladder outlet obstruction and clinically manifests as lower urinary tract symptoms (LUTS) [2]. The combination of these two is collectively referred to as BPH/LUTS. Specifically, LUTS includes frequent urination, urgent urination, urinary incontinence, increased nocturia, delayed urination, poor urine flow, and a feeling of incomplete bladder emptying [3]. These symptoms not only significantly reduce patients’ quality of life but also easily trigger negative emotions, impair physical and mental health, and affect family and social harmony. From an epidemiological perspective, the incidence of BPH shows a significant positive correlation with age. The incidence rate is approximately 50% among men over 50 years old, reaches 83% among those over 80 years old, and about 90% of men over 90 years old have varying degrees of prostatic hyperplasia. the incidence rate among men over 40 years old is around 8% [4]. With the intensification of population aging, clinical attention to this condition has continued to increase. Currently, clinical treatment mainly relies on surgery and medications. The first-line pharmacotherapeutic regimen for BPH/LUTS involves the use of alpha-blockers, 5-alpha-reductase inhibitors, either alone or in combination [5]. However, these medications are not effective for all BPH/LUTS patients, they also tend to cause adverse reactions such as sexual desire disorders and insomnia, and some patients experience drug tolerance issues. These factors limit their clinical application, making it of great significance to explore safe and low-toxicity treatment regimens. In the system of traditional chinese medicine (TCM), BPH is classified under the category of “Long Bi” (dribbling and retention of urine). Its core pathogenesis lies in the dysfunction of bladder qi transformation, with the main disease locations in the kidney and bladder. Syndrome differentiation revolves around kidney deficiency, dampness-heat, and blood stasis [6]. TCM treatment includes internal therapy (oral administration of herbal medicines) and external therapy (such as drug enema, acupuncture, and acupoint application) [7]. It has the advantages of treating both the root cause and symptoms, significant therapeutic effects, and few adverse reactions. TCM treatment not only improves LUTS symptoms in patients but also reduces blood stasis and edema in prostate tissue, thereby assisting in reducing prostate volume [8], which provides an important supplementary approach for BPH treatment.
Vladimiriae Radix is a Tibetan medicine with ethnic characteristics,
and it is also a cross-used medicinal species in TCM and Tibetan medicine [9].
Its main producing areas are concentrated in Aba Tibetan autonomous prefecture,
Liangshan Yi autonomous prefecture, Xichang, Baoxing, Ya’an (all in Sichuan
Province, China), as well as Yunyang, Kaixian, and Nanchuan (all in Chongqing
Municipality, China). Its traditional indications align closely with the
therapeutic needs of BPH. In Chinese medicine the herb is classified as a
qi-regulating drug (pungent, bitter, warm) that promotes flow, assists bladder qi
transformation, dispels blood stasis to reduce glandular swelling, and warms to
transform damp turbidity in the urethra—directly addressing the “long-bi”
(urinary retention) mechanism of BPH. Formulas containing it, such as “Xiang-sha
Liu-jun-zi Tang”, have been shown to relieve accompanying symptoms [10]. In
Tibetan medicine it is known as “Ma-nu”; it harmonizes the nyes-pa, opens the
urethra, disperses the “flesh nodule” of the prostate, and is commonly
incorporated into preparations like “Wu-wei Mu-xiang San”. Contemporary
clinical studies found that Chinese- and Tibetan-compound prescriptions
containing Vladimiriae Radix (e.g., Mu-xiang Dan-shen Yin combined with
tamsulosin) can significantly improve IPSS scores, residual urine volume, and
maximum urinary flow rate, reduce prostate volume, and cause fewer adverse
effects—thereby providing traditional theoretical support for the present study
and validating the herb’s candidacy for BPH therapy. The Vladimiriae
Radix herb is cylindrical or semi-cylindrical with longitudinal grooves,
slightly curved, measuring 10–30 cm in length and 1–3 cm in diameter. Its
surface is yellowish-brown or dark brown, with longitudinal wrinkles; where the
outer skin peels off, reticulate fine vascular bundles (resembling a loofah
sponge) are visible, and the root head occasionally has an “oil head” (a black,
sticky gelatinous substance). The herb is light in weight, hard and brittle, and
easy to break, the fracture surface is yellowish-white or yellow, containing
sparse dark yellow oil spots and cracks (Fig. 1A,B). The xylem is broad with
radial textures, and the center of some samples is withered. It has a slight
aroma, a bitter taste, and feels sticky when chewed [11]. Modern research has
shown that costunolide and dehydrocostus lactone are the core active components
in Vladimiriae Radix. Both can significantly inhibit the proliferation
of six types of human-derived tumor cells, and the exocyclic double bond at the
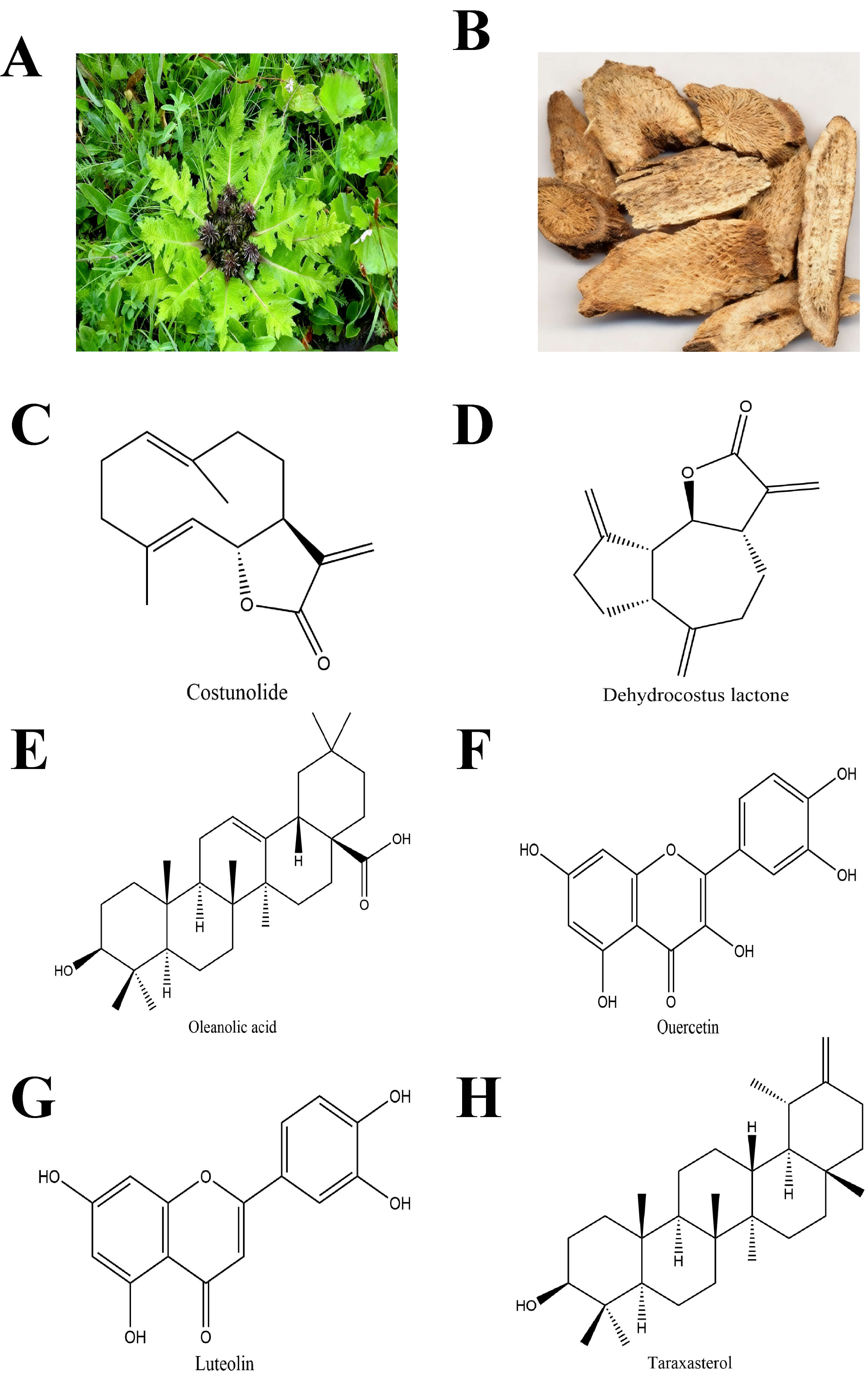 Fig. 1.
Fig. 1.
Original plant, dried medicinal material and main active components of Vladimiriae Radix. (A) The original plant of Vladimiriae Radix (the plant with rhizomes, roots and leaves). (B) Dried medicinal material of Vladimiriae Radix, Core active components which mainly including (C) costunolide, (D) dehydrocostus lactone, (E) oleanolic acid, (F) quercetin, (G) luteolin, and (H) taraxasterol from Vladimiriae Radix.
For the screening of active components of Vladimiriae Radix,
“Vladimiriae Radix” was used as the search term to log into the TCM
Systems Pharmacology Database and Analysis Platform (TCMSP,
https://www.tcmsp-e.com/#/home). The screening
criteria were set as oral bioavailability (OB)
| Name of database and software | Website address |
| TCM Systems Pharmacology Database and Analysis Platform (TCMSP) | https://www.tcmsp-e.com/#/database/ |
| PubChem Database | https://pubchem.ncbi.nlm.nih.gov |
| SwissTarget Prediction Database | https://www.swisstargetprediction.ch/ |
| GeneCards Database | https://www.genecards.org/ |
| Online Mendelian Inheritance in Man (OMIM) | https://omim.org/ |
| Therapeutic Target Database (TTD) | https://db.idrblab.net/ttd/ |
| Venny 2.1 Online Software | https://bioinfogp.cnb.csic.es/tools/venny/index.html |
| STRING 12.0 Online Database | https://string-db.org/ |
| DAVID 6.8 Database | https://davidbioinformatics.nih.gov/ |
| Microbiological Bioinformatics Platform | https://www.bioinformatics.com.cn/ |
| Lianchuan BioCloud | https://www.omicstudio.cn/tool |
| PDB Database | https://www.rcsb.org/?ref=nav_home |
Using “BPH” as the search term, search for BPH-related targets in the GeneCards database (GeneCards, https://www.genecards.org/), Online Mendelian Inheritance in Man database (OMIM, https://omim.org/), and Therapeutic Target Database (TTD, https://db.idrblab.net/ttd/) respectively, so as to ensure the acquisition of sufficient target information. Using the VENNY 2.1 plotting platform (https://bioinfogp.cnb.csic.es/tools/venny/index.html), a Venn diagram of the targets related to Vladimiriae Radix and the targets related to BPH was generated. By taking the intersection of the two sets of targets, the common therapeutic targets were screened out, thereby clarifying the target association between Vladimiriae Radix and BPH.
The disease-drug overlapping targets obtained via the Venn diagram and the core targets were imported together into Cytoscape 3.10.3 (Cytoscape Consortium, Seattle, WA, USA) to construct a “drug active component-common target” network. The data were then analyzed to screen proteins based on degree values, followed by visualization of the network.
To further investigate and visualize the interactions among overlapping targets, first upload the overlapping targets to the search tool for the retrieval of interacting genes/proteins (STRING) database (STRING, https://string-db.org/), set the organism as “Homo sapiens” to acquire data. Then import the obtained data into Cytoscape 3.10.3 software for PPI network analysis and visualization.
The overlapping targets were processed using the Database for Annotation,
Visualization and Integrated Discovery (DAVID) database
(https://davidbioinformatics.nih.gov/):
set “official gene symbol” as the select identifier, “gene list” as the list
type, and “Homo sapiens” as the organism. With p
To verify the interaction between the active components of Vladimiriae Radix and the overlapping targets of BPH screened via network pharmacology, the 3D structures (in SDF format) of the main active components of Vladimiriae Radix were downloaded from the PubChem database. Meanwhile, the 3D structures (in PDB format) of the overlapping targets were obtained from the RCSB (Research Collaboratory for Structural Bioinformatics) Protein Data Bank (PDB, https://www.rcsb.org/?ref=nav_home). The above-mentioned component and target structures were uploaded to Discovery Studio 2019 software for docking. Based on the energy values of the binding conformations between ligands and receptor proteins, the docking conformation with the lowest energy and the most stable structure was screened. Furthermore, PyMOL 3.0.3 software (Schrödinger, Inc., New York, NY, USA) was used for visual analysis and processing of the optimal docking results to intuitively present the interaction relationship between the two (the components and targets). Meanwhile, AutoDockTools1-2 (The Scripps Research Institute, La Jolla, CA, USA) was employed to perform a series of operations (such as water removal and hydrogen addition) on the macromolecular protein crystal structures, and finally the binding energy of each component was obtained.
Among the 6 core active components obtained from the network pharmacology
analysis of Vladimiriae Radix, oleanolic acid was selected as the
representative component for this cellular verification, for the following
reasons: Molecular docking results showed that oleanolic acid has good binding
ability to all key targets of BPH (docking energy
BPH-1 cells were purchased from Shanghai Jinyuan Biotechnology Co., Ltd. and cultured in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS) in a constant-temperature incubator at 37 °C with 5% CO2. When the cells reached the logarithmic growth phase, they were digested with 0.25% trypsin for passage. Experiments were performed when the cells were passaged to the 3rd generation. BPH-1 cells were validated by short tandem repeat (STR) profiling and tested negative for mycoplasma.
Frozen BPH-1 cells were retrieved and rapidly thawed in a 37 °C water bath. After centrifugation to remove the cryopreservation medium, the cells were resuspended in DMEM medium containing 20% FBS and double antibodies (penicillin/streptomycin). The cells were seeded into culture dishes and incubated in a 37 °C, 5% CO2 incubator. When the confluence of BPH-1 cells reached 80%–90%, the culture medium was discarded, and 3 mL of PBS was added to remove dead cells and excess medium, followed by 3 rounds of PBS washing.
Dihydrotestosterone (DHT) powder was dissolved in anhydrous ethanol to prepare a
10-3 M stock solution, which was aliquoted and stored at –20 °C in the
dark. During the experiment, the stock solution was diluted to the target
concentration of 10-7 M with serum-free DMEM medium, ensuring the final
ethanol concentration was
Logarithmic-phase BPH-1 cells were digested and resuspended in medium to form a
single-cell suspension with a concentration adjusted to 1
Six groups were set up in the experiment: blank control group (medium without
drugs), model group (500 nM DHT), positive drug group (500 nM DHT + 40 µM
finasteride), low-concentration drug group (500 nM DHT + 10 µM oleanolic
acid), medium-concentration drug group (500 nM DHT + 20 µM oleanolic acid),
and high-concentration drug group (500 nM DHT + 40 µM oleanolic acid). Each
group had at least 3 replicate wells. The plate was returned to the incubator for
continuous culture of 48 h. 10 µL of CCK-8 reagent was added to each well,
and after gently shaking the 96-well plate, it was incubated in the incubator for
another 1–2 h. The absorbance value (OD value) of each well at 450 nm wavelength
was detected using a microplate reader. The cell proliferation inhibition rate
(R) was calculated according to Formula (1): R = (1 – ODexperiment /
ODcontrol)
BPH-1 cells were lysed in 1 mL of Trizol, and total RNA was extracted according to the Trizol kit instructions. cDNA templates were synthesized by reverse transcription using a quantitative real-time PCR kit, and RT-PCR experiments were performed on an ABI7500 real-time PCR system. The reaction conditions were as follows: pre-denaturation at 95 °C for 10 min, followed by 40 cycles of denaturation at 95 °C for 10 s, annealing at 60 °C for 20 s, and extension at 72 °C for 34 s. After the reaction, the mRNA expression levels of 6 core proteins (AR, CYP17A1, CYP19A1, ACHE, F2, and HMGCR) were analyzed respectively. The specific primer components for qRT-PCR are listed in Table 2.
| Target genes | Primer sense (5′-3′) | Primer antisense (5′-3′) |
| ACHE | CTTCTCCTCCTCCTCTGGCT | ATGCCCAGGAAAGCAGAGAC |
| AR | ACACCAAAGGGCTAGAAGGC | GTAGTCGCGACTCTGGTACG |
| CYP17A1 | TCCTGCTGCACAATCCTCAG | ATAGTTGGTGTGCGGCTGAA |
| CYP19A1 | TGCGAGTCTGGATCTCTGGA | AGTTTGCTGCCGAATCGAGA |
| F2 | AGATGGGCTGGATGAGGACT | AGCCAAAGGTCCTCGGATTG |
| HMGCR | TGTGTGTGGGACCGTAATGG | GCAAGCTCCTTGGAGGTCTT |
All data were statistically analyzed using SPSS 21.0 software (IBM Corp.,
Chicago, IL, USA). The results were expressed as mean
A total of 6 active components were obtained through multi-source data
collection and collation (Table 3). The active components of Vladimiriae
Radix were screened based on the parameters of molecular weight (MW), OB, and
drug likeness (DL). These 6 active components included costunolide (Fig. 1C),
dehydrocostus lactone (Fig. 1D), oleanolic acid (Fig. 1E), quercetin (Fig. 1F),
luteolin (Fig. 1G), and taraxasterol (Fig. 1H) respectively. The six core
targets—AR, CYP17A1, CYP19A1, ACHE, F2, and HMGCR
(HMGCR is the rate-limiting enzyme of cholesterol synthesis; its
aberrantly elevated activity leads to cholesterol accumulation in prostatic
tissue, which provides excess precursors for androgen synthesis
[cholesterol
| Mol ID | Molecule Name | OB (%) | DL | MW |
| MOL010825 | Costunolide | 29.07 | 0.11 | 232.35 |
| MOL001298 | Dehydrocostus lactone | 58.57 | 0.14 | 230.33 |
| MOL000263 | Oleanolic acid | 29.02 | 0.76 | 456.78 |
| MOL000006 | Luteolin | 36.16 | 0.25 | 286.25 |
| MOL000098 | Quercetin | 46.43 | 0.28 | 302.25 |
| MOL004085 | Taraxasterol | 8.19 | 0.74 | 468.84 |
OB, Oral bioavailability; DL, drug-likeness; MW, molecular weight.
Using “BPH” as the keyword, search for prostate cancer-related targets in the
GeneCards database and OMIM database respectively re-extracted from GeneCards
using a relevance score threshold
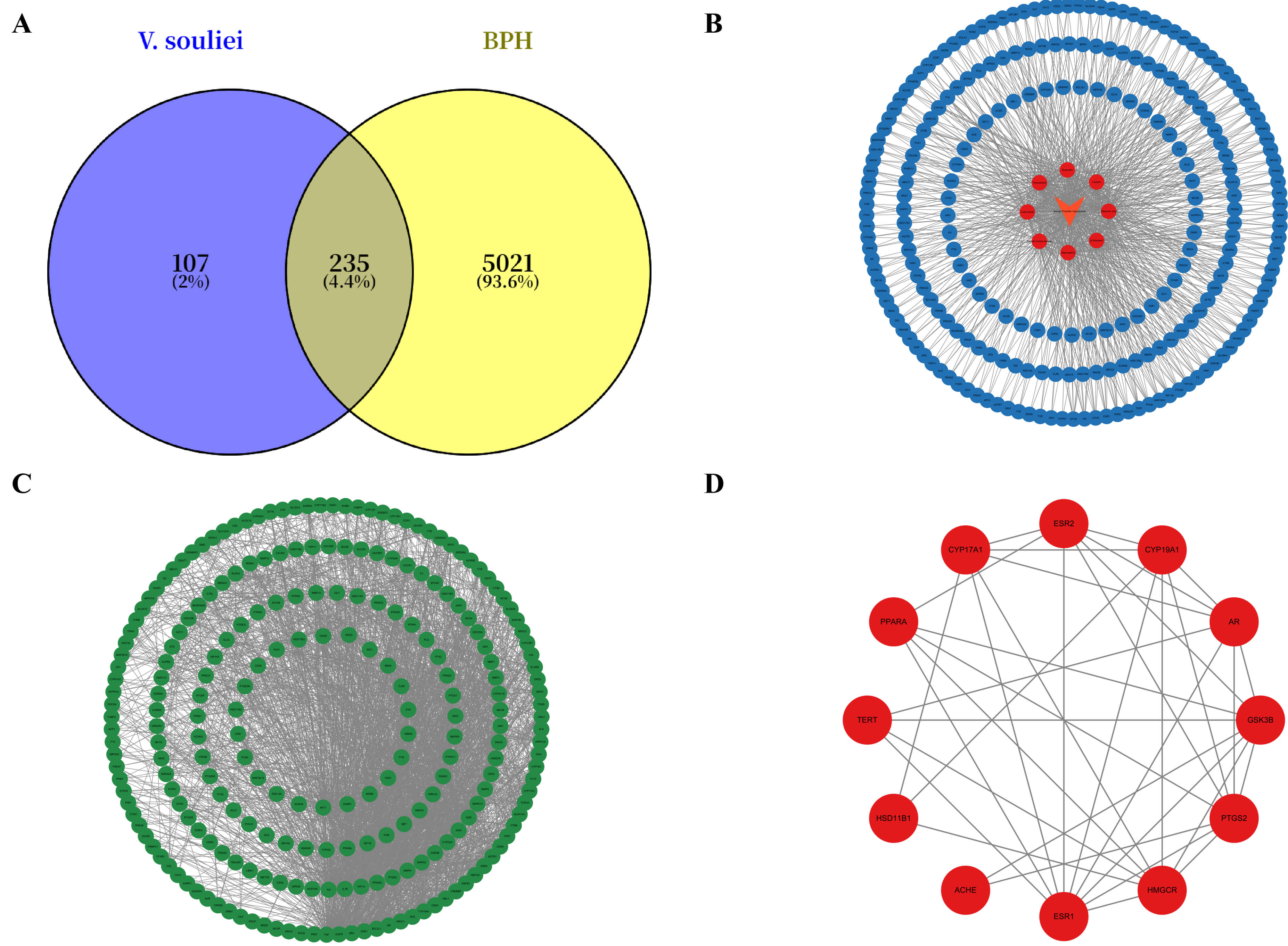 Fig. 2.
Fig. 2.
Common targets, interaction network, core targets and PPI network of Vladimiriae Radix against BPH. (A) Venn diagram of common targets between Vladimiriae Radix and BPH. (B) Interaction network of targets between Vladimiriae Radix and BPH. (C) Fifteen core targets of Vladimiriae Radix in treatment of BPH. (D) Protein PPI network diagram. BPH, benign prostatic hyperplasia; PPI, protein-protein interaction.
To more intuitively present the targets of the active components of Vladimiriae Radix in BPH, as well as the interactions between their common targets, this study imported the data into Cytoscape 3.10.3 software to construct a “drug-active component-disease-target” interaction network diagram and complete the visualization process (Fig. 2B). Among these, 247 protein-protein intersection nodes and 917 interaction edges between target proteins were obtained. The degree of connectivity between targets reflects their importance, with higher connectivity indicating greater importance. Additionally, the top 15 core targets with the highest degree values were screened out using the CytoHubba plugin (Fig. 2C).
The intersection targets of Vladimiriae Radix and BPH were uploaded to
the STRING 12.0 database. The species was set to “Homo sapiens” (human), and
the minimum interaction score was set to “highest confidence (
GO functional enrichment analysis was performed on the intersection targets of Vladimiriae Radix and BPH using the DAVID database, yielding a total of 678 terms related to BP, 72 terms related to CC, and 225 terms related to MF. The top 10 terms with the highest enrichment significance in each of the three categories were selected and imported into the Online MicroBioInfo Analysis Platform for visualization (Fig. 3A). The specific results are as follows: For BP: The core processes include cellular response to nutrient levels, response to drugs, transport of organic hydroxyl compounds, response to steroid hormones, regulation of membrane potential, response to metal ions, positive regulation of gene expression, response to exogenous stimuli, response to hypoxia, and positive regulation of miRNA transcription. These findings suggest that Vladimiriae Radix may exert therapeutic effects by regulating cellular physiological responses through multiple dimensions. For CC: The targets are mainly concentrated in membrane-related structures such as membrane rafts, membrane microdomains, and membrane regions, and also cover the extracellular space, extracellular region, and cell surface, which reflects the spatial distribution characteristics of the ta3rgets’ actions. For MF: The functions are dominated by amide binding, drug binding, phosphatase binding, and G protein-coupled amine receptor activity, and also involve enzyme binding, identical protein binding, and steroid binding, which demonstrates the mechanism of the targets’ actions at the molecular level. The results obtained from the GO analysis were further imported into the Online MicroBioInfo Analysis Platform for KEGG pathway enrichment visualization (Fig. 3B). The results showed that the core pathway of Vladimiriae Radix in treating BPH was dominated by “Pathways in cancer”, and this pathway as well as other related pathways contained multiple important targets associated with the pathological mechanism of BPH (Fig. 3C).
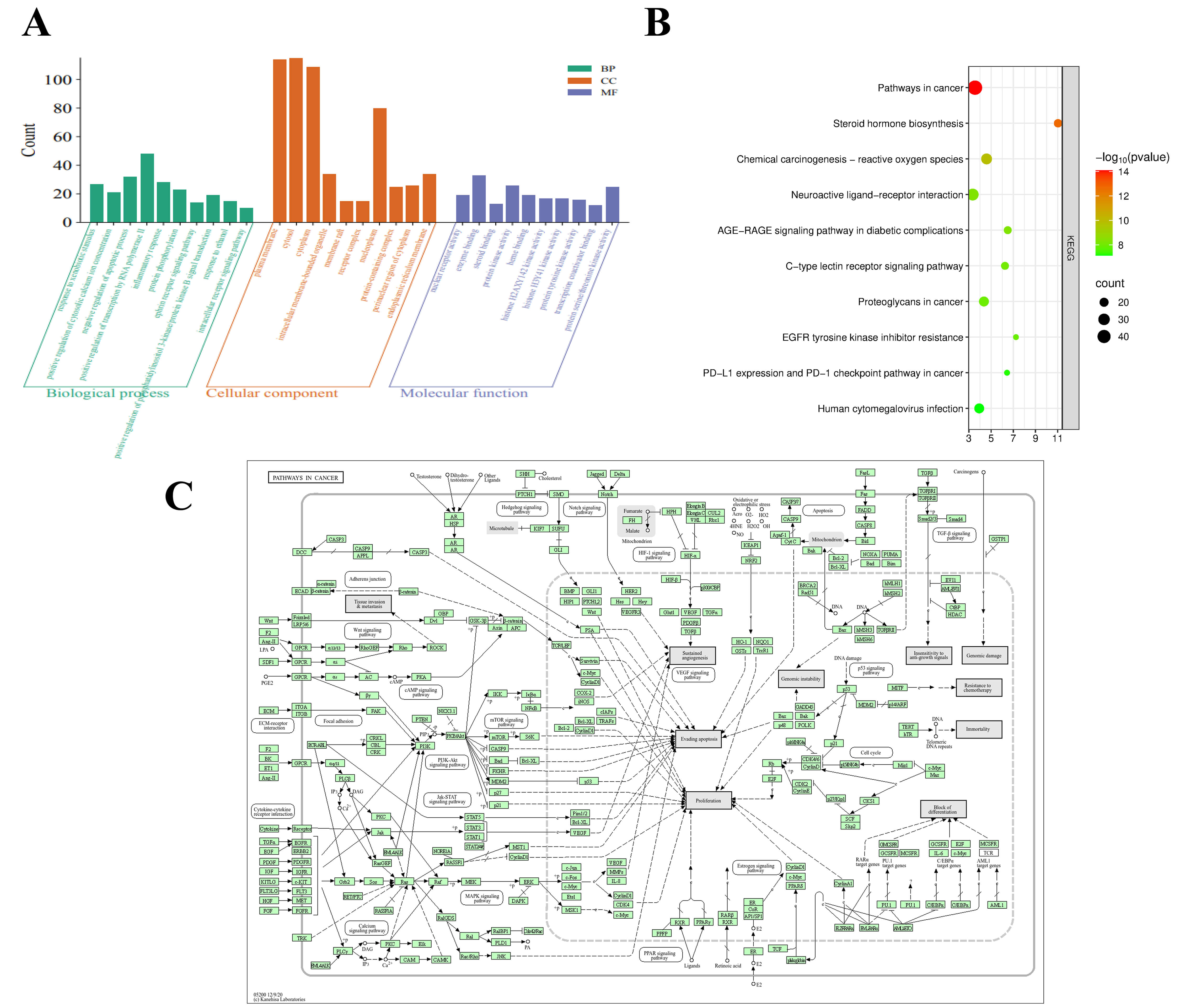 Fig. 3.
Fig. 3.
GO enrichment, KEGG pathway enrichment and cancer-related pathway of core targets. (A) GO Enrichment analysis of key targets of Vladimiriae Radix. (B) KEGG enrichment pathways of genes. (C) Pathways in cancer. GO, Gene ontology; KEGG, kyoto encyclopedia of genes and genomes.
Download the SDF-format 3D structure diagrams of the main active components of Vladimiriae Radix from PubChem, obtain the PDB-format 3D structure diagrams of intersection targets from the PDB (Protein Data Bank), upload them to Discovery Studio 2019 for docking, screen the optimal conformations based on the binding conformation energy values of ligands and receptor proteins, and then use PyMOL to conduct visual analysis of the interaction between the two.
The potential binding sites of oleanolic acid and ACHE include amino acid residues such as CLY-345 and SER-347, and the compound forms bonds with amino acids like carbon hydrogen bonds and conventional hydrogen bonds (Fig. 4A). The potential binding sites of oleanolic acid and AR include amino acid residues such as LEU-93, ILE-119, TRP-189, TRP-182, MET-118, and TYR-185, and the compound forms bonds with amino acids including carbon hydrogen bonds, pi-alkyl bonds, alkyl bonds, and covalent bonds (Fig. 4B). The potential binding sites of oleanolic acid and CYP17A1 include amino acid residues such as PHE-446, ARG-109, ARG-138, ARG-451, CYS-453, and ALA-459, and the compound forms interactions with amino acids such as van der waals forces, pi-donor hydrogen bonds, salt bridges, attractive charges, amide-pi stacked bonds, conventional hydrogen bonds, alkyl bonds, pi-alkyl bonds, carbon hydrogen bonds, covalent bonds, and unfavorable acceptor-acceptor interactions (Fig. 4C). The potential binding sites of oleanolic acid and CYP19A1 include amino acid residues such as ALA-438, ARG-375, MET-303, MET-364, PHE-430, and ARG-115, and the compound forms bonds with amino acids including salt bridges, attractive charges, conventional hydrogen bonds, carbon hydrogen bonds, pi-donor hydrogen bonds, pi-pi T-shaped bonds, amide-pi stacked bonds, alkyl bonds, pi-alkyl bonds, and covalent bonds (Fig. 4D). The potential binding sites of oleanolic acid and F2 include amino acid residues such as VAL-27, LYS-24, THR-25, LEU-23, TRP-31, and LEU-28, and the compound forms bonds with amino acids like unfavorable bumps, unfavorable donor-donor interactions, alkyl bonds, and pi-alkyl bonds (Fig. 4E). The potential binding sites of oleanolic acid and HMGCR include amino acid residues such as LYS-692, ASN-755, LEU-857, HOH-1268, LEU-853, and LEU-562, and the compound forms bonds with amino acids including water hydrogen bonds, conventional hydrogen bonds, unfavorable donor-donor interactions, unfavorable acceptor-acceptor interactions, alkyl bonds, and pi-alkyl bonds (Fig. 4F).
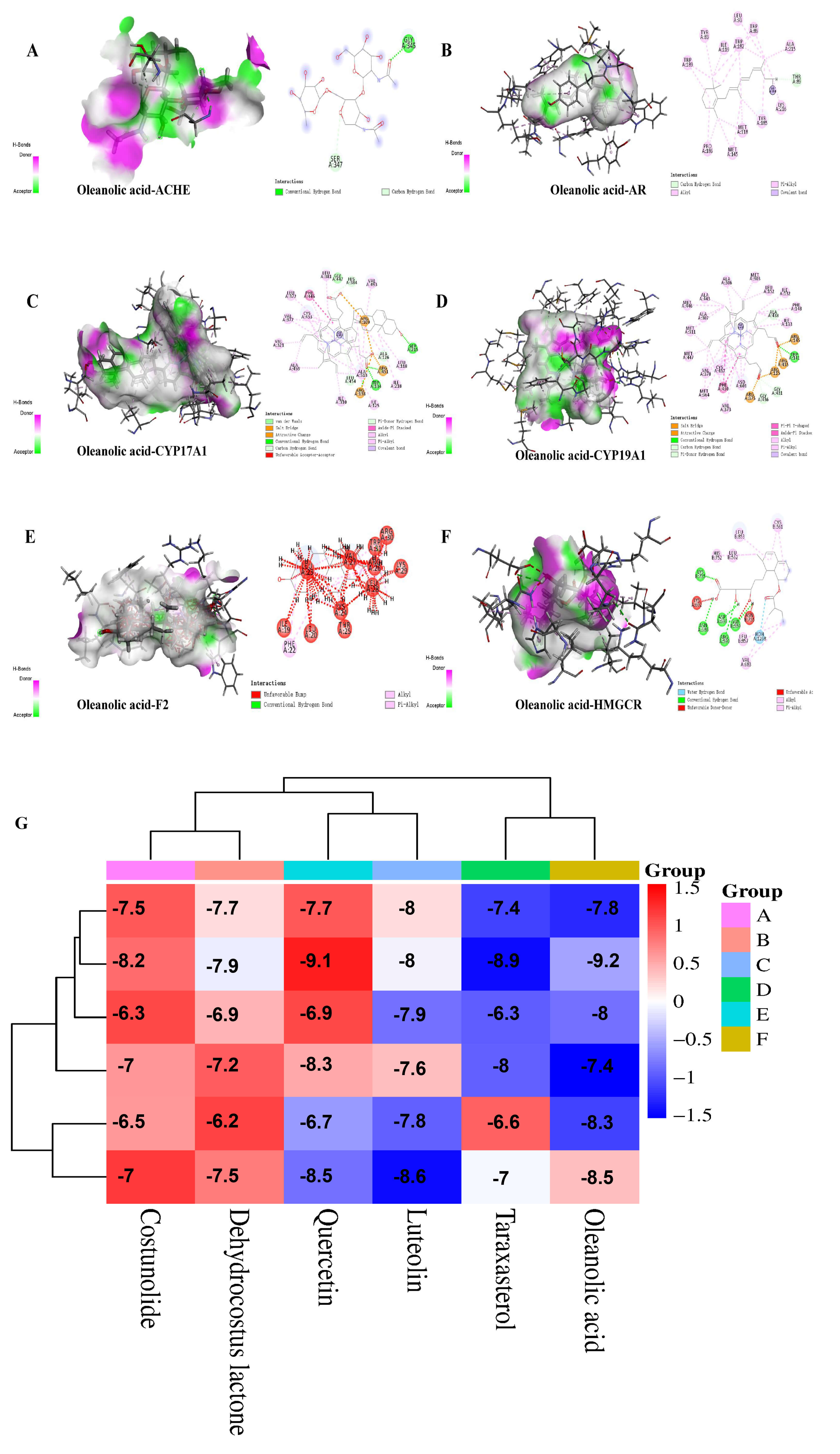 Fig. 4.
Fig. 4.
Interaction of oleanolic acid with core targets and docking energy heatmap. Interaction between oleanolic acid and protein targets (A) ACHE, (B) AR, (C) CYP17A1, (D) CYP19A1, (E) F2, (F) HMGCR. (G) Docking energy heatmap.
Based on the binding energy results obtained via AutoDockTools1-2, a heatmap was generated. The results show that 6 active components of Vladimiriae Radix exhibit good docking performance with 6 core targets of BPH, and the active components can form stable bindings with specific amino acid residues of the targets, confirming that the two have good binding activity Especially for oleanolic acid, its binding energy with various proteins is better than other active compounds, which is also the reason why we will continue to choose it for vitro experimental verification in the next step (Fig. 4G).
Microscopic image analysis showed that compared with the blank control group,
BPH-1 cells in the model group proliferated significantly. After treatment with
oleanolic acid at different concentrations (10–40 µM), the cell survival
rate decreased, with a significant inhibitory effect in the oleanolic acid groups
(p
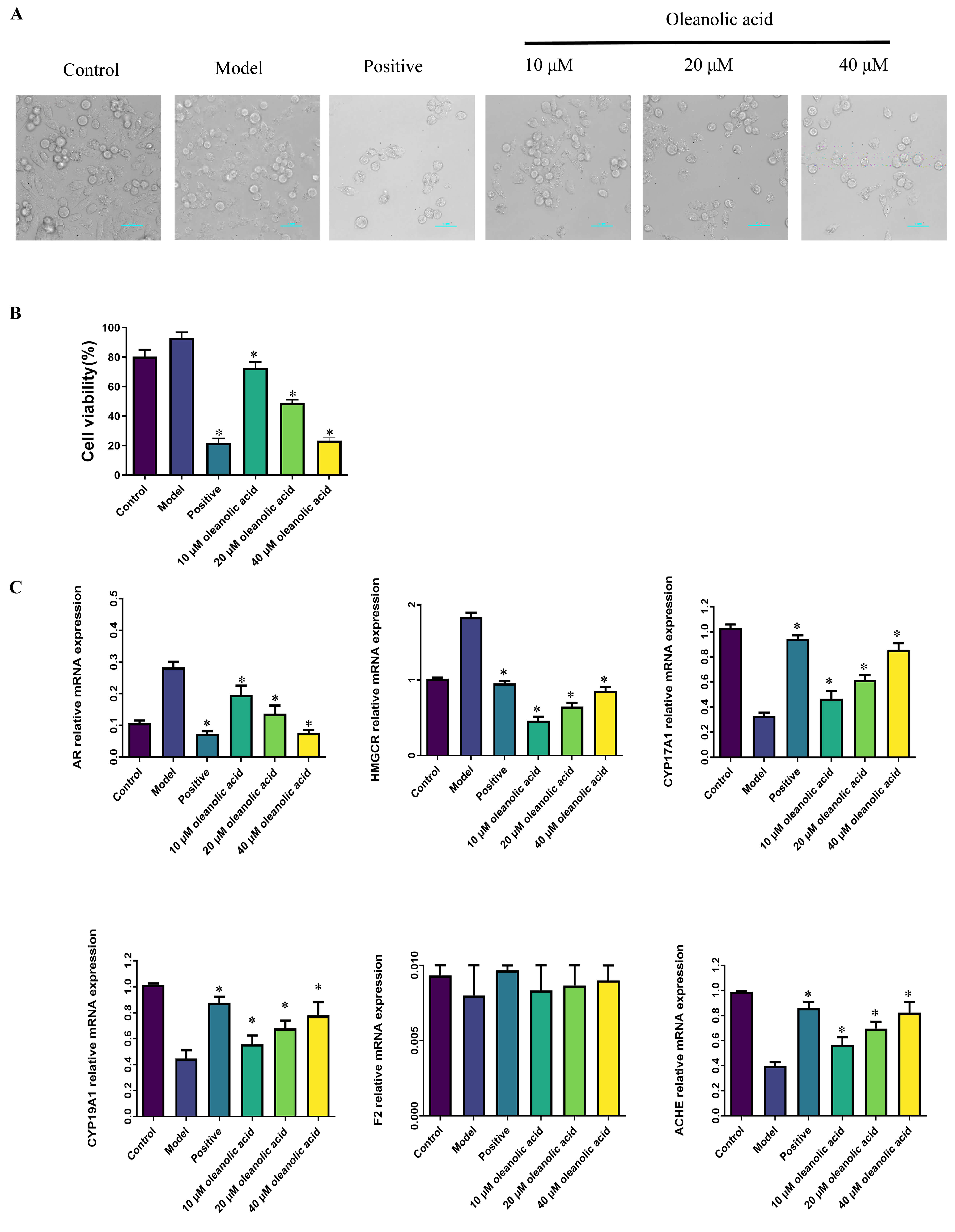 Fig. 5.
Fig. 5.
Effect of oleanolic acid on BPH-1 cell viability and
core gene mRNA expression. (A,B) Effect of oleanolic acid on cell viability.
Cells were treated with varies concentrations of oleanolic acid for 48 h. Scale bar = 50 μm. (C)
Effect of oleanolic acid on AR, HMGCR, CYP17A1,
CYP19A1, F2 and ACHE mRNA levels in BPH-1 cells. Data
were presented as the mean
To clarify the mechanism of action of oleanolic acid on BPH-1 cells, RT-PCR was
used to detect the mRNA expression levels
of AR, CYP17A1, CYP19A1, ACHE, F2,
and HMGCR genes. The results showed that compared with the control
group, the mRNA expression levels of AR and HMGCR in the BPH-1
cell model group established with 500 nM DHT were significantly increased
(p
To systematically elaborate the scientific mechanism underlying the anti-BPH
effect of the dried roots of Vladimiriae Radix, this study employed
network pharmacology and molecular docking techniques as core methods. From the
three-dimensional perspective of “component-target-pathway”, it explored the
pharmacodynamic material basis and action logic of D. souliei. This not only
addresses the practical challenges in clinical treatment of BPH but also provides
a new paradigm for the modernization research on the integrated application of
Tibetan medicine and TCM [17]. BPH, a common benign disease of the urinary system
in elderly men, exhibits distinct “multidimensional intertwined”
characteristics in its pathological mechanism [18]. Specifically, the
uncontrolled proliferation of epithelial and stromal cells in the prostatic
transition zone represents the core pathological manifestation [19], the
imbalance of the hormonal microenvironment mediated by DHT serves as the key
driving factor [20], and chronic non-bacterial inflammatory infiltration and
dysregulation of urethral cholinergic nerve modulation respectively exacerbate
tissue damage and LUTS [21]. These three factors together form the complex
pathological network of BPH. In current first-line clinical treatment regimens,
although
Costunolide, dehydrocostus lactone, oleanolic acid, luteolin, quercetin, and
taraxasterol are bioactive components of Vladimiriae Radix with
potential anti-BPH properties. AR, CYP17A1, CYP19A1,
ACHE, F2, and HMGCR were the core targets which
related to the bioactive components in anti-BPH. Oleanolic acid exhibits the good
binding energy with 6 core targets. Cell-level verification showed that oleanolic
acid may significantly inhibits the proliferation of DHT-induced BPH-1 cells
(p
The datasets generated and analysed during the current study are available upon request from the corresponding author.
JXM conceived and designed the research. XDF, JW, and JXM carried out the data analysis and wrote the paper in the present study. XDF and JW finished the drawing and experiment. JXM carried out the manuscript revision work. All authors participated in the editing and revision of the manuscript. All authors have read and approved the final manuscript. All authors fully participated in this research and agree to be accountable for all aspects of the study.
Not applicable.
Not applicable.
This work was financially supported by Key Scientific and Technological Research Project of Chongqing Municipal Education Commission (No. KJZD-K202302801), Scientific Research Project of Chongqing Medical and Pharmaceutical College (ygz2022104, ygzrc2024104, ygzrc2024101), Chongqing Municipal Education Commission Youth Project (KJQN202402816) respectively.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





