1 Cardiac Surgery, Cruces University Hospital, 48903 Barakaldo, Spain
2 3D Printing and Bioprinting Platform, Biobizkaia Research Institute, 48903 Barakaldo, Spain
3 Cardiovascular Surgery, Hospital Universitario Quirónsalud Madrid, 28223 Pozuelo de Alarcón, Madrid, Spain
4 Cardiology, Cruces University Hospital, 48903 Barakaldo, Spain
5 Radiology, Cruces University Hospital, 48903 Barakaldo, Spain
Abstract
Hypertrophic cardiomyopathy (HCM) is defined as an abnormal thickening of the left ventricular myocardium, greater than 15 mm in one or more segments as measured by imaging modalities, in the absence of an identifiable secondary cause. The surgical treatment, the septal myectomy, remains the gold standard for invasive treatment. However, it is considered one of the most challenging surgeries, not only to learn, but also to teach. We identified four major challenges associated with HCM: the limited number of patients requiring surgical intervention, the limited intraoperative visibility, the heterogeneity of anatomical presentations, and the overall complexity in understanding the disease. 3D printing is a type of technology that converts virtual designs into physical objects through additive layering of materials. Approaching HCM with printed hearts allows to understand better the pathology and to guide the surgery by addressing the challenges associated with HCM. The application of 3D printing in HCM is a novel approach to understand the disease and planning surgery. This technology holds the potential to optimize preoperative strategies, enabling more precise resections and reducing perioperative complications.
Keywords
- 3D model
- HCM
- hypertrophic cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is defined as an abnormal thickening of the left ventricular myocardium, greater than 15 mm in one or more segments as measured by imaging modalities such as echocardiography, magnetic resonance imaging (MRI), or computed tomography (CT), in the absence of an identifiable secondary cause [1, 2].
The diagnosis of HCM is mainly verified by echocardiography, which enables the assessment of the hypertrophy as well as the left ventricular outflow tract (LVOT) obstruction. According to the European and American Guidelines (2023), MRI and CT scans are recommended for further morphological and functional detail [3, 4].
This pathology holds significant clinical relevance, as it is the leading cause of sudden death in young athletes [1]. Recent studies on the natural history of the disease estimate an annual mortality rate of approximately 1% among affected individuals [1, 5].
Septal myectomy remains the gold standard for invasive treatment. It involves targeted resection of the hypertrophied interventricular septum. In many cases, myocardial hypertrophy is associated with additional structural abnormalities, such as papillary muscle anomalies, which may require complementary surgical techniques to achieve optimal outcomes.
In 2020, a published article recognized septal myectomy as one of the most challenging surgeries, not only to learn, but also to teach. This highlights the crucial role of simulation and structured mentoring in surgical training [6].
In our experience, four fundamental challenges shape management of obstructive cardiomyopathy (HOCM): (1) the limited number of patients requiring surgery, (2) poor intraoperative visibility, (3) variability in phenotypic presentation, and (4) the complexity of understanding the disease as a multidimensional entity. These challenges were identified through longitudinal observation of our clinical and educational experience in surgical planning for HOCM, particularly in the context of the well-documented complexity of teaching and learning this procedure [6].
As we are in an era defined by digital planning and personalized medicine, 3D modeling offers a new lens through which to approach HOCM. 3D models can help to reshape how we see, learn and decide.
HCM is a condition with relatively few surgical indications. Consequently, surgical experience is often limited and concentrated in high-volume referral centers. As Birkmeyer et al. [7] demonstrated, operative outcomes directly correlate with surgical volume.
In this context, 3D models provide a valuable tool to address the limitations posed by the low number of eligible surgical cases. These models enable simulation of the septal myectomy in a controlled, risk-environment [8]. They facilitate a detailed understanding of patient-specific anatomy and support precise surgical planning (Fig. 1A).
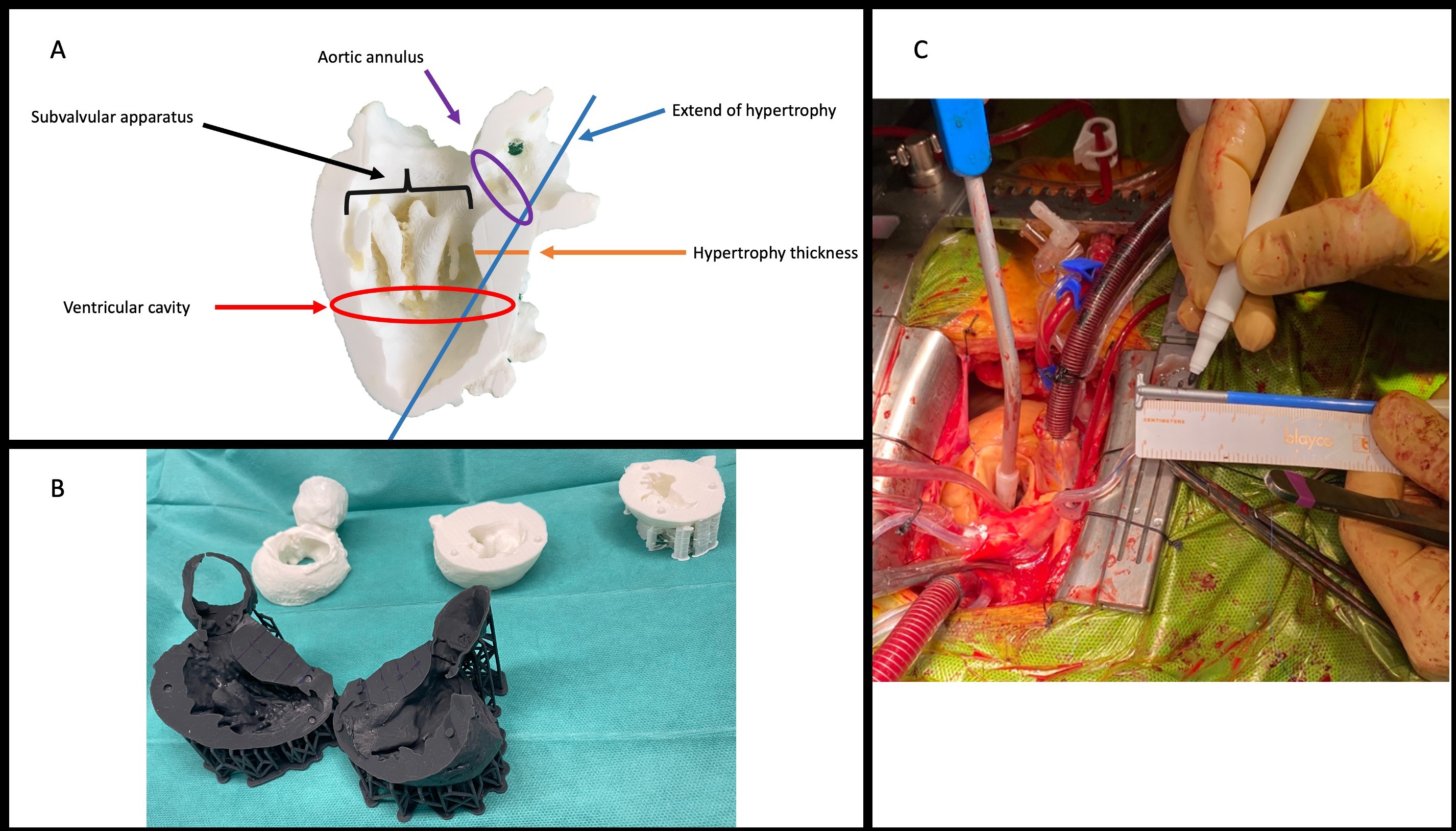 Fig. 1.
Fig. 1.
Patient-specific anatomical models for a Morrow procedure. (A) Key anatomical landmarks relevant to septal myectomy. (B) Patient-specific 3D-printed heart models used for preoperative planning and surgical guidance. (C) Intraoperative view showing reference markings on the electrocautery loop, applied to reproduce the preoperative measurements during septal resection.
The pathophysiology of LVOT obstruction in HOCM is inherently complex. Septal hypertrophy often coexists with other structural anomalies, such as elongated mitral valve leaflets, displaced papillary muscles, or fibromuscular bands that contribute to dynamic obstruction [9]. Therefore, surgical success relies on a comprehensive preoperative understanding of the ventricular anatomy to plan the appropriate strategy for relieving the obstruction, enlarging the left ventricular cavity and excising abnormal intraventricular structures.
The classical technique of septal myectomy, or Morrow procedure, involves the surgical resection of the hypertrophied interventricular septum responsible for LVOT obstruction [10]. Traditionally performed through a transaortic approach using a scalpel, its success depends not only on the depth but also on the length of the resection [6, 11]. Other techniques of resection, such as using an electrocautery with a diathermic loop are often used [12].
However, the aortic valve orifice provides a narrower access window than the ventricular cavity itself, and during cardioplegic arrest, the collapse of the ventricular wall further limits visualization. These factors complicate the identification of key intraventricular structures and increase the risk of suboptimal or excessive resection.
In this context, 3D modeling introduces a fundamental change in perspective [13]. Preoperative analysis of patient-specific 3D-printed hearts allows surgeons to determine the optimal depth and extent of the resection, anticipate anatomical obstacles, and plan complementary maneuvers involving the mitral or subvalvular apparatus (Fig. 1).
Furthermore, septal thickness can be preoperatively measured and marked on the 3D model and then translated intraoperatively. Reference marking on the electrocautery loop can serve as a visual and tactile guide—effectively transforming the preoperative plan into an intraoperative roadmap for safer, more precise resection.
A major challenge in HOCM lies in its broad phenotypic variability. While asymmetric septal hypertrophy is the most common form, hypertrophy may also involve other myocardial segments, resulting in diverse anatomical and functional patterns [14].
Anatomical evaluation often relies on echocardiography, but its operator dependence and limited reproducibility can lead to diagnostic discrepancies. For this reason, clinical guidelines recommend complementing echocardiography assessment with cardiac magnetic resonance or computed tomography, especially in cases with suboptimal imaging or residual obstruction. Comparative studies have showed that echocardiography tends to overestimate septal thickness relative to MRI and CT, with potential implications for diagnosis, risk assessment, and therapeutic decisions [4, 15, 16].
Several morphological classifications have been proposed to categorize HCM phenotypes [17]. However, these systems are rarely used in clinical practice and often fail to capture the full anatomical complexity of the disease. They generally overlook associated structural anomalies.
3D modeling offers a reproducible and integrative approach to characterizing the hypertrophic phenotype. By accurately reconstructing the myocardium, 3D models allow for objective assessment of hypertrophic distribution and associated anomalies. This approach also lays the foundation for a new morphological classification of HCM.
A comprehensive understanding of HOCM remains an ongoing challenge due to its multifactorial nature, encompassing genetic background, electrophysiological features, sociodemographic variables, and previous cardiac interventions.
HCM is predominantly a genetic disorder with autosomal dominant inheritance, carrying a 50% transmission risk to offspring [15]. Integrating 3D-modeling with genetic analysis could enable re-evaluation of genotype-phenotype correlations and help identify familial expression patterns predictive of disease onset, progression, and surgical complexity. This integration may ultimately contribute to a new phenotypic-genotypic classification of HCM.
Nearly all patients with HCM exhibit electrocardiographic abnormalities, regardless of obstruction severity [18]. Although a normal ECG does not exclude HCM, it may indicate a milder phenotype [17]. ECG findings remain useful for early detection, risk assessment, and, in some cases, for inferring underlying genetic variants [19].
Sex and ethnicity influence disease presentation and outcomes. Men tend to be diagnosed earlier and often present more pronounced hypertrophy [20], whereas women, due to smaller ventricular size, may reach diagnostic thresholds later but show higher rates of heart failure progression and mortality [21]. Registry data (ShaRE) also reveal a greater prevalence of pathogenic mutations in women [21]. Ethnic differences are evident as well; for instance, the apical variant, is more frequent in the Asian population (15% in Japan vs 3% in US) [22].
More than 80% of patients present at least one cardiovascular risk factor, such as hypertension, diabetes, or obesity, all of which can worsen symptoms and surgical outcomes [23]. Prior cardiac surgery increases perioperative risk; in such cases, 3D cardiac models offer a valuable means to visualizing intracardiac anatomy that may be obscured by scarring or altered geometry.
Patients with previous alcohol septal ablation present particular challenges. While both ablation and surgical myectomy relieve obstruction, their anatomical effects differ: ablation causes localized necrosis, often leading to right bundle branch block, whereas myectomy produces a controlled resection of the basal septum, more commonly resulting in left bundle branch block [15, 24]. Prior ablation also increases the risk of complete atrioventricular block, frequently necessitating permanent pacemaker implantation [25, 26].
HCM carries an inherent arrhythmogenic potential and remains the leading cause of sudden cardiac death (SCD) in young athletes. Risk stratification based on European guidelines an imaging-derived models - guides implantable cardioverter defibrillator (ICD) implantation [27]. Although septal myectomy does not eliminate SCD risk entirely, it has been shown to substantially reduce it, especially in patients with obstructive forms or adverse remodeling phenotypes [27, 28].
The use of 3D modeling in HOCM represents more than technological progress, it marks a shift in surgical perspective. Where the management of HOCM once relied primarily on experience, it is now complemented by simulation; visualization, once confined to the operative field, now begins in the preoperative setting; and anatomical variability, once a source of uncertainty, can now be leveraged to enhance surgical precision.
Each of the four challenges (scarcity, poor visibility, wide variability, and complexity) can be reframed not as barriers, but as opportunities for innovation. The 3D model is not merely a replica of the heart; it is a bridge between imaging and intuition, theory and practice, knowledge and mastery.
While virtual visualization can provide sufficient anatomical information in selected cases, 3D printing remains particularly valuable when a tangible and realistic surgical scenario is required, such as for surgical planning, procedural validation, intraoperative guidance, and educational purposes for both residents and patients. At present, clear criteria defining when virtual visualization alone is sufficient have not yet been established, and the decision should be tailored to the complexity of the pathology and the intended clinical application.
As cardiac surgery advances towards personalization, embracing such tools will define not only how we operate, but how we think.
In conclusion, the application of 3D printing in HCM offers a novel approach to understanding the disease and planning surgery. It has the potential to transform preoperative strategies by addressing key challenges through improved spatial orientation, personalized anatomical insights, and enhanced surgical precision.
CIPM, AF, DR, PP, AG, ACP, RG, IJ, CDA and RVS participated in the conception and writing of this manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work to take public responsibility for relevant parts of the content, and agree to be accountable for all aspects of the research, ensuring its accuracy and integrity.
Not applicable.
The authors acknowledge the contributions of the Departments of Radiology and Cardiology, as well as the engineering team, for their valuable contributions.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

