1 Department of Cardiovascular Surgery, Qilu Hospital of Shandong University, 250012 Jinan, Shandong, China
2 Intensive Care Unit of Cardiovascular Surgery Department, Qilu Hospital of Shandong University, 250012 Jinan, Shandong, China
Abstract
Gastrointestinal bleeding (GIB) after cardiac surgery is a crucial complication. Therefore, this study aimed to develop and validate an early prediction model for postoperative GIB after cardiac surgery using routinely available preoperative data and early postoperative variables.
We conducted a retrospective cohort study of adult intensive care unit (ICU) patients after cardiac surgery using the Medical Information Mart for Intensive Care IV (MIMIC-IV) v3.1 database. The first 24 hours after ICU admission served as the prediction window, and preoperative characteristics and variables collected within this window were extracted using Structured Query Language (SQL). The cohort was randomly split into training and validation sets (7:3). Candidate predictors were selected using least absolute shrinkage and selection operator (LASSO) regression, combined with 1000 bootstrap resamples to improve stability. A multivariable logistic regression model was developed and presented as a nomogram. Model discrimination was evaluated using receiver operating characteristic (ROC) curves and the area under the curve (AUC), with 95% confidence intervals (CIs), in the training and validation cohorts and in prespecified surgical subgroups. Calibration was assessed by bootstrap resampling with the Brier score, and clinical utility was examined using decision curve analysis (DCA).
Among 10,611 included patients, 324 (3.1%) developed GIB. The final nomogram included 13 predictors: the first arterial partial pressure of oxygen (PaO2) after ICU admission, minimum PaO2, maximum blood urea nitrogen (BUN), activated partial thromboplastin time (APTT), red cell distribution width (RDW), red blood cell (RBC) transfusion within 24 h after ICU admission, mean peripheral oxygen saturation (SpO2), mean heart rate (HR), mean arterial pressure during the first 6 h after ICU admission, atrial fibrillation, hyperlipidemia, chronic heart failure, and the Simplified Acute Physiology Score II (SAPS II). The model demonstrated discrimination in the training set (AUC 0.770; 95% CI 0.737–0.803) and validation set (AUC 0.772; 95% CI 0.721–0.823). The AUCs across subgroups ranged from 0.764 to 0.785, suggesting stable discrimination across procedure types. Calibration plots showed agreement between predicted and observed risks, and DCA suggested potential clinical benefit across relevant threshold probabilities.
This study developed and validated a prediction model based on routinely available preoperative and early postoperative data that can identify patients at increased risk of GIB after open cardiac surgery.
Keywords
- gastrointestinal bleeding
- cardiac surgery
- MIMIC-IV
- prediction model
- LASSO
- multivariable logistic regression
- nomogram
Post–cardiac surgery gastrointestinal bleeding is closely associated with postoperative mortality [1, 2, 3]. Large cohort studies have reported in-hospital mortality rates of up to 17% among patients who develop post–cardiac surgery gastrointestinal bleeding (PCGIB) [3, 4, 5]. Beyond its direct clinical consequences, PCGIB can exacerbate downstream complications, including shock and infection, prolong hospital stay [2, 6], and increase healthcare utilization [4, 7].
Patients undergoing cardiac surgery are exposed to multiple perioperative stressors, including hypoperfusion, systemic inflammatory responses, and hemodynamic instability [8, 9]. These insults can precipitate stress-related mucosal injury and, when compounded by anticoagulant or antiplatelet therapy and perioperative coagulation disturbances, can progress to clinically significant gastrointestinal bleeding (GIB) [10, 11, 12, 13, 14].
Early postoperative vital signs, laboratory abnormalities (e.g., blood pressure, urine output, lactate, oxygenation, and coagulation parameters), and the intensity of pharmacologic support are routinely available in standard care and capture key physiological domains related to oxygen delivery, perfusion, and hemostasis, making them particularly informative for early bleeding risk stratification [8, 9, 11].
A bedside-applicable prediction model incorporating preoperative and early postoperative variables could facilitate timely risk stratification, individualized monitoring, and preventive measures (including prophylactic proton pump inhibitor therapy), while supporting balanced decisions on the initiation and intensity of anticoagulant and antiplatelet therapy. Using routinely available variables from the Medical Information Mart for Intensive Care IV (MIMIC-IV) database, we aimed to develop and validate an early prediction model for gastrointestinal bleeding after cardiac surgery and to assess its discrimination, calibration, and clinical utility.
This retrospective cohort study used data from MIMIC-IV v3.1 (PhysioNet), a publicly available, de-identified electronic health record database comprising intensive care unit (ICU) admissions at Beth Israel Deaconess Medical Center (BIDMC) from 2008 to 2022. Because the dataset is de-identified, the original data collection was approved by the institutional review boards and conducted with a waiver of informed consent [15].
The study was reported with reference to Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD) guideline [16].
We identified adult hospitalizations in MIMIC-IV in which patients underwent the
target open cardiac surgery procedures. The unit of analysis was the
hospitalization (hadm_id). Eligible admissions met the following criteria: age
We excluded admissions with diagnoses of esophageal/gastric varices (EV) or gastrointestinal malignancy during the index hospitalization (International Classification of Diseases, Ninth and Tenth Revision (ICD-9/10) code lists are provided in Supplementary Table 1). To preserve the prespecified prediction window, we additionally excluded admissions with chart-documented gastrointestinal bleeding within the first 24 h after ICU admission; an event was considered “within 24 h” if its chart time occurred within 24 h of ICU admission.
The primary outcome was gastrointestinal bleeding occurring during the ICU stay beyond the first 24 h after ICU admission. Gastrointestinal bleeding was defined as overt or occult bleeding into the gastrointestinal tract accompanied by a decrease in hematocrit and a positive fecal hemoglobin or fecal occult blood test [17, 18, 19]. Outcome events were ascertained using charted clinical evidence together with concordant ICD-9/10 diagnosis codes; the operational chart-based screening approach is detailed in Supplementary Methods 1.
Candidate predictive variables were selected based on prior literature, biological plausibility, and clinician input. Variables included baseline demographics, comorbidities, surgical procedure type, vital signs, and laboratory tests, perioperative blood product use, and medication exposures. All candidates were limited to information that could be obtained within the prediction window (the first 24 h after ICU admission).
Early postoperative hemodynamic and oxygenation variables were summarized within 0–6 h, 0–12 h, and 0–24 h windows after ICU admission and, together with the same baseline and perioperative covariates, were used to construct three candidate predictor datasets. Feature selection and model development were then performed separately in each dataset, and the optimal time window was chosen based on predictive performance and clinical practicality. To minimize temporal ambiguity and reverse causation, we excluded postoperative complications with uncertain onset times—including shock, low cardiac output syndrome, and cerebrovascular events—which are often identified primarily through ICD-9/10 coding and therefore cannot be reliably timed to the prespecified prediction window or distinguished from events occurring after bleeding onset.
Data extraction was performed using Structured Query Language (SQL). To maintain data integrity, we excluded patients with missing values in more than 10% of all candidate predictors, as well as individual variables with a missingness rate exceeding 10%. The final cohort was randomly partitioned into a training cohort (n = 7428) and a validation cohort (n = 3183) using a 7:3 ratio.
Missing data were addressed using multiple imputation (five imputations, 50 iterations each) with predictive mean matching for continuous variables (Supplementary Methods 2) [20, 21].
Univariable logistic regression analyses were performed to summarize unadjusted
associations and effect directions between candidate predictors and the outcome
(OR, 95% CI, and p value), and to provide an initial screening of
candidate predictors. Given the substantial correlations and redundancy among
early ICU physiologic variables, we applied a bootstrap resampling procedure
(1000 resamples) to improve the stability of predictor selection in the training
set [22, 23]. In each resampled dataset, least absolute shrinkage and selection
operator (LASSO) regression was performed, with the regularization parameter
selected to minimize the cross-validated error [24]. We quantified selection
stability using the bootstrap selection proportion (
A PCGIB risk prediction nomogram was developed based on regression coefficients from the final multivariable logistic regression model. We conducted sensitivity analyses across the 0–6 h, 0–12 h, and 0–24 h time-window datasets, and selected the optimal time window based on model performance and clinical applicability. Discrimination was assessed using receiver operating characteristic (ROC) curves and area under the curve (AUC) in both the training and validation cohorts, with prespecified subgroup analyses by procedure type: coronary artery bypass grafting (CABG)–only, valve–only, and CABG+valve surgery. Calibration was evaluated in the training set using bootstrap resampling (1000 iterations) to generate calibration plots and an optimism-corrected C-index; Brier scores were reported for both cohorts. Clinical utility was examined using decision curve analysis (DCA).
Continuous variables are presented as mean
A total of 12,173 ICU admissions undergoing open cardiac surgery were initially
identified (Supplementary Fig. 1). After restricting to the first
postoperative ICU admission, adults (age
As shown in Supplementary Table 2, baseline demographics,
comorbidities, and key perioperative characteristics were well balanced between
the training and validation cohorts (p
For descriptive purposes, Table 1 summarizes baseline, perioperative, and early
postoperative variables for the overall cohort, stratified by outcome (GIB vs
non-GIB). Overall, 324 patients (3.1%) were diagnosed with PCGIB. Building on
the balanced split (Supplementary Table 2), we compared characteristics
between the GIB and non-GIB groups in the overall cohort (Table 1). Patients with
GIB were older [71.00 (61.75–79.00) vs 68.00 (60.00–75.00) years; p
| Characteristic | Non-GIB (n = 10,287) | GIB (n = 324) | p value | ||
| Demographics and general characteristics | |||||
| Gender | |||||
| Female | 2950 (28.7) | 103 (31.8) | 0.223 | ||
| Male | 7337 (71.3) | 221 (68.2) | |||
| Age (years) | 68.00 (60.00, 75.00) | 71.00 (61.75, 79.00) | |||
| Height (cm) | 171.24 |
169.92 |
0.017 | ||
| Weight (kg) | 85.60 (74.10, 98.85) | 85.00 (72.60, 96.17) | 0.091 | ||
| BMI, kg/m2 | 28.98 (25.94, 32.71) | 28.98 (25.89, 32.40) | 0.538 | ||
| Smoking history, n (%) | 4034 (39.2) | 121 (37.3) | 0.497 | ||
| Alcohol use history, n (%) | 714 (6.9) | 36 (11.1) | 0.004 | ||
| Comorbidities and complications | |||||
| Acute myocardial infarction, n (%) | 1926 (18.7) | 80 (24.7) | 0.007 | ||
| Coronary artery disease, n (%) | 7623 (74.1) | 235 (72.5) | 0.525 | ||
| Hypertension, n (%) | 8044 (78.2) | 259 (79.9) | 0.454 | ||
| Chronic heart failure, n (%) | 1846 (17.9) | 102 (31.5) | |||
| HFrEF, n (%) | 1608 (15.6) | 82 (25.3) | |||
| HFpEF, n (%) | 983 (9.6) | 59 (18.2) | |||
| Low cardiac output syndrome, n (%) | 849 (8.3) | 63 (19.4) | |||
| Chronic kidney disease, n (%) | 1815 (17.6) | 113 (34.9) | |||
| Acute kidney injury, n (%) | 8005 (77.8) | 299 (92.3) | |||
| Liver disease, n (%) | 175 (1.7) | 12 (3.7) | 0.007 | ||
| Coagulopathy, n (%) | 2025 (19.7) | 88 (27.2) | |||
| Atrial fibrillation, n (%) | 4438 (43.1) | 195 (60.2) | |||
| Chronic lung disease, n (%) | 1785 (17.4) | 68 (21.0) | 0.090 | ||
| Anemia, n (%) | 6176 (60.0) | 228 (70.4) | |||
| Hyperlipidemia, n (%) | 7378 (71.7) | 189 (58.3) | |||
| Peripheral vascular disease, n (%) | 1715 (16.7) | 84 (25.9) | |||
| Diabetes without chronic complications, n (%) | 2763 (26.9) | 83 (25.6) | 0.619 | ||
| Diabetes with chronic complications, n (%) | 1188 (11.5) | 51 (15.7) | 0.021 | ||
| Delirium, n (%) | 538 (5.2) | 34 (10.5) | |||
| Stroke/cerebrovascular event, n (%) | 665 (6.5) | 45 (13.9) | |||
| Shock, n (%) | 602 (5.9) | 85 (26.2) | |||
| In-hospital mortality, n (%) | 169 (1.6) | 33 (10.2) | |||
| ICU severity scores and vital signs within 24 h after ICU admission | |||||
| Mean HR, beats/min | 80.57 (75.13, 87.08) | 82.16 (75.58, 90.31) | 0.003 | ||
| Mean MAP, mmHg | 74.34 (70.62, 78.29) | 73.72 (68.67, 78.43) | 0.024 | ||
| Mean SpO2, % | 97.86 (96.82, 98.76) | 97.71 (96.30, 98.83) | 0.118 | ||
| SAPS II | 36.00 (29.00, 43.00) | 40.00 (34.00, 48.00) | |||
| SOFA | 5.00 (3.00, 7.00) | 6.00 (4.00, 8.25) | |||
| Mean CVP, mmHg | 9.86 (7.94, 11.94) | 10.34 (9.86, 13.31) | |||
| Total drainage volume, mL | 560.00 (425.00, 730.00) | 560.00 (490.00, 803.75) | |||
| Vital signs within 6 h after ICU admission | |||||
| Mean MAP, mmHg | 76.40 (71.88, 81.14) | 76.40 (70.00, 80.03) | 0.007 | ||
| Mean HR, beats/min | 79.40 (74.00, 83.67) | 80.00 (76.05, 87.62) | |||
| Mean SpO2, % | 99.59 (98.67, 100.00) | 99.50 (97.20, 100.00) | |||
| Laboratory measurements within 24 h after ICU admission | |||||
| Maximum creatinine (mg/dL) | 0.90 (0.80, 1.20) | 1.10 (0.90, 1.70) | |||
| Maximum BUN (mg/dL) | 17.00 (13.00, 22.00) | 21.50 (16.00, 29.25) | |||
| Minimum arterial pH | 7.31 (7.28, 7.34) | 7.31 (7.26, 7.35) | 0.508 | ||
| First arterial pH | 7.40 (7.36, 7.44) | 7.39 (7.35, 7.43) | 0.061 | ||
| First arterial pH, n (%) | |||||
| 7.35–7.45 (normal) | 6674 (64.9) | 197 (60.8) | 0.121 | ||
| 1918 (18.6) | 75 (23.1) | ||||
| 1695 (16.5) | 52 (16.0) | ||||
| Maximum lactate (mmol/L) | 2.50 (2.00, 3.20) | 2.70 (1.90, 4.00) | |||
| First lactate (mmol/L) | 2.00 (1.50, 2.60) | 2.00 (1.30, 2.90) | 0.350 | ||
| Minimum PaO2 (mmHg) | 99.00 (81.00, 125.00) | 91.00 (68.00, 110.25) | |||
| First PaO2 (mmHg) | 325.00 (258.00, 383.00) | 306.00 (167.25, 377.00) | |||
| Maximum INR | 1.40 (1.30, 1.60) | 1.50 (1.30, 1.70) | |||
| Maximum PT, s | 15.50 (14.20, 17.20) | 16.30 (14.60, 18.60) | |||
| Maximum APTT (s) | 33.20 (29.40, 39.95) | 38.50 (31.15, 54.62) | |||
| Maximum blood glucose (mg/dL) | 173.00 (152.00, 199.00) | 172.50 (150.00, 207.00) | 0.934 | ||
| First blood glucose (mg/dL) | 136.00 (114.00, 163.00) | 135.00 (110.75, 167.00) | 0.361 | ||
| Maximum WBC count (×109/L) | 15.10 (11.90, 19.20) | 14.90 (10.67, 18.60) | 0.026 | ||
| Minimum hemoglobin (g/dL) | 8.90 (7.60, 10.10) | 8.00 (6.90, 9.80) | |||
| Minimum HCT, % | 27.30 (24.20, 30.60) | 25.40 (22.70, 29.82) | |||
| Minimum platelet count (×109/L) | 131.00 (105.00, 164.00) | 132.50 (96.75, 183.00) | 0.620 | ||
| Maximum RDW (%) | 13.80 (13.10, 14.80) | 15.20 (14.07, 16.50) | |||
| Postoperative therapies within 24 h after ICU admission | |||||
| Maximum VIS | 33.01 (0.00, 100.05) | 30.01 (0.00, 130.04) | 0.039 | ||
| Mean VIS | 13.90 (0.00, 43.92) | 9.67 (0.00, 54.34) | 0.214 | ||
| Norepinephrine use, n (%) | 1295 (12.6) | 80 (24.7) | |||
| Norepinephrine infusion duration, n (%) | |||||
| 0 h | 8992 (87.4) | 244 (75.3) | |||
| 0– |
429 (4.2) | 16 (4.9) | |||
| 6– |
256 (2.5) | 21 (6.5) | |||
| 610 (5.9) | 43 (13.3) | ||||
| Antiplatelet therapy within 24 h, n (%) | 7695 (74.8) | 205 (63.3) | |||
| Antiplatelet therapy within 12 h, n (%) | 981 (9.5) | 49 (15.1) | |||
| Anticoagulant therapy within 24 h, n (%) | 1942 (18.9) | 96 (29.6) | |||
| Anticoagulant therapy within 12 h, n (%) | 1515 (14.7) | 86 (26.5) | |||
| RBC transfusion, n (%) | 2672 (26.0) | 155 (47.8) | |||
| Plasma transfusion, n (%) | 1019 (9.9) | 68 (21.0) | |||
| Platelet transfusion, n (%) | 1323 (12.9) | 76 (23.5) | |||
| Surgical procedure, n (%) | |||||
| CABG plus valve surgery | 1413 (13.7) | 71 (21.9) | |||
| CABG only | 5255 (51.1) | 110 (34.0) | |||
| Valve surgery only | 2978 (28.9) | 111 (34.3) | |||
| Others | 641 (6.2) | 32 (9.9) | |||
Note: Continuous variables are expressed as mean
Laboratory and physiologic profiles also differed between groups. Patients with
PCGIB had higher Simplified Acute Physiology Score II (SAPS II) [40.00
(34.00–48.00) vs 36.00 (29.00–43.00); p
In univariable logistic regression analyses, increasing age was associated with
a higher risk of postoperative GIB [odds ratio (OR) 1.02, 95% confidence
interval (CI) 1.01–1.03; p
In the time-window sensitivity analysis (Supplementary Fig. 2), the 0–6 h model achieved the highest discrimination (AUC 0.770), compared with the 0–12 h (AUC 0.758) and 0–24 h models (AUC 0.725). Given its superior performance and the clinical advantage of earlier risk stratification, we selected the 0–6 h window for final model development.
Fig. 1 shows the LASSO coefficient path from a single fit on the full training cohort for the final selected 0–6 h window dataset.
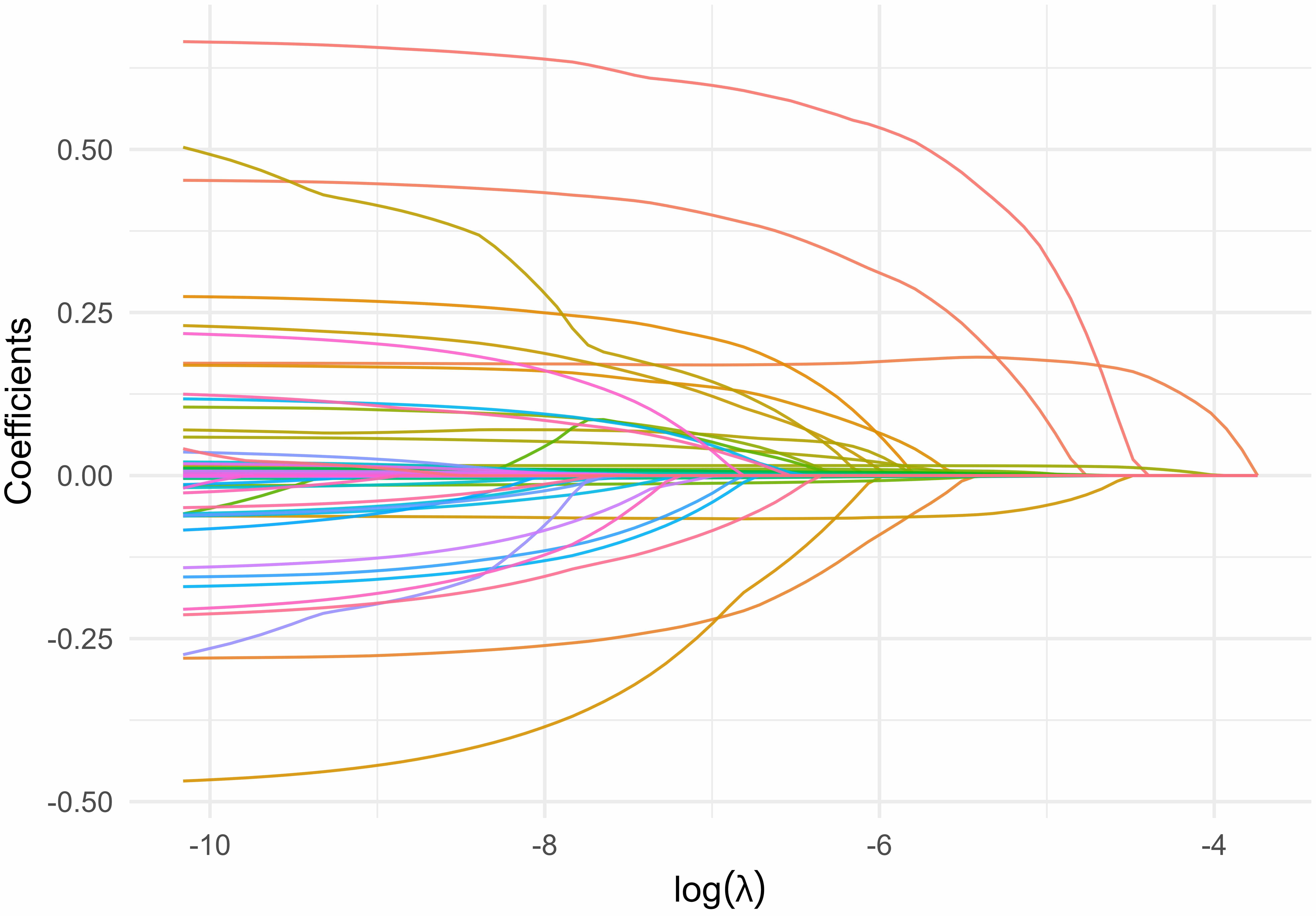 Fig. 1.
Fig. 1.
LASSO feature selection in the 0–6 h window dataset (training cohort). The plot shows the coefficient path from a single LASSO fit on the full training set. LASSO, least absolute shrinkage and selection operator.
Thirteen predictors were identified for the final model: first PaO2 after ICU admission, minimum PaO2 within 24 h, mean peripheral oxygen saturation (SpO2), heart rate (HR), and mean arterial pressure (MAP) within 0–6 h after ICU admission, maximum BUN, APTT, and RDW within 24 h, red blood cell (RBC) transfusion within 24 h, atrial fibrillation, hyperlipidemia, chronic heart failure, and SAPS II (Table 2).
| Predictor | b | OR (95% CI) | p value |
| First PaO2 (ICU) | –0.0020 | 0.998 (0.996–0.999) | 0.009 |
| Minimum PaO2 (24 h) | –0.0020 | 0.998 (0.994–1.001) | 0.263 |
| Mean HR (0–6 h) | 0.0070 | 1.007 (0.999–1.016) | 0.104 |
| Maximum APTT (24 h) | 0.0070 | 1.007 (1.002–1.011) | 0.002 |
| SAPS II | 0.0080 | 1.008 (0.996–1.019) | 0.170 |
| Maximum BUN (24 h) | 0.0149 | 1.015 (1.006–1.023) | |
| Mean MAP (0–6 h) | –0.0151 | 0.985 (0.969–1.000) | 0.060 |
| Mean SpO2 (0–6 h) | –0.0758 | 0.927 (0.875–0.985) | 0.012 |
| Maximum RDW (24 h) | 0.1848 | 1.203 (1.129–1.278) | |
| Chronic heart failure | 0.2111 | 1.247 (1.001–1.553) | 0.059 |
| Hyperlipidemia | –0.2510 | 0.772 (0.593–1.005) | 0.062 |
| Atrial fibrillation | 0.4606 | 1.585 (1.200–2.103) | 0.001 |
| RBC transfusion (24 h) | 0.6816 | 1.977 (1.462–2.674) |
Abbreviations: b, regression coefficient; OR, odds ratio; CI, confidence interval; APTT, activated partial thromboplastin time; BUN, blood urea nitrogen; HR, heart rate; ICU, intensive care unit; MAP, mean arterial pressure; PaO2, arterial partial pressure of oxygen; RBC, red blood cell; RDW, red cell distribution width; SAPS II, Simplified Acute Physiology Score II; SpO2, peripheral oxygen saturation; 0–6 h indicates 0–6 h after ICU admission; 24 h indicates the first 24 h after ICU admission.
Supplementary Table 4 summarizes the stability selection results based
on 1000 bootstrap resamples. All 13 predictors met the prespecified stability
criteria (
The multivariable logistic regression model was visualized as a nomogram (Fig. 2; OR in Table 2). Among the included predictors, RBC transfusion showed the
largest effect [OR 1.977, 95% confidence interval (CI) 1.462–2.674; p
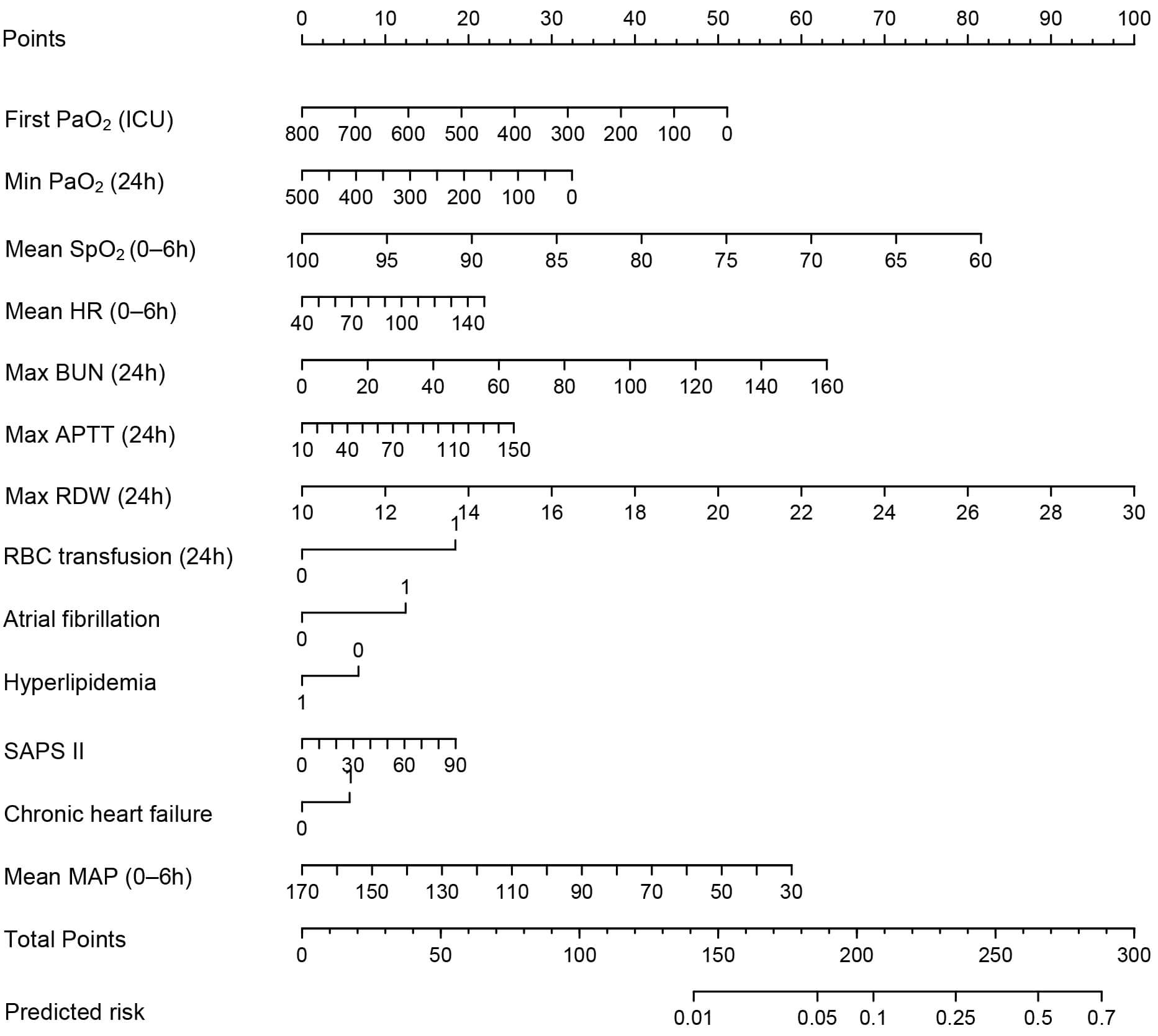 Fig. 2.
Fig. 2.
Nomogram for the final 13-predictor multivariable logistic regression model for predicting post–cardiac surgery gastrointestinal bleeding. Abbreviations: APTT, activated partial thromboplastin time; BUN, blood urea nitrogen; HR, heart rate; ICU, intensive care unit; MAP, mean arterial pressure; PaO2, arterial partial pressure of oxygen; RBC, red blood cell; RDW, red cell distribution width; SAPS II, Simplified Acute Physiology Score II; SpO2, peripheral oxygen saturation; 0–6 h indicates 0–6 h after ICU admission; 24 h indicates the first 24 h after ICU admission.
The model demonstrated good discrimination in the training set (AUC 0.770; 95% CI 0.737–0.803; Fig. 3a) and in the validation set (AUC 0.772; 95% CI 0.721–0.823; Fig. 3b).
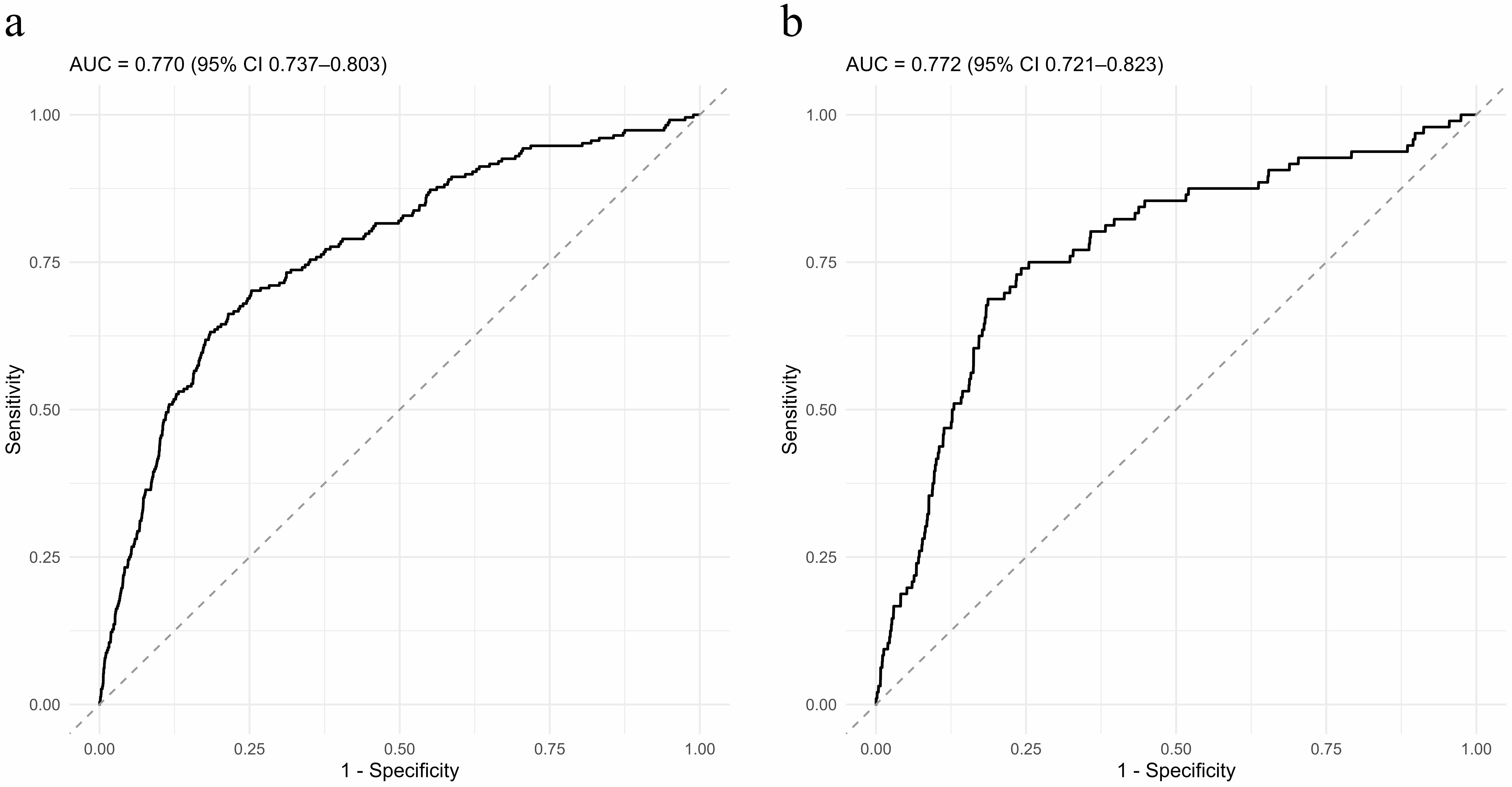 Fig. 3.
Fig. 3.
ROC curves of the prediction model in the training and validation cohorts. (a) AUC = 0.770 (95% CI 0.737–0.803) in the training cohort; (b) AUC = 0.772 (95% CI 0.721–0.823) in the validation cohort. Abbreviations: ROC, receiver operating characteristic; AUC, area under the curve.
In procedure-specific subgroup analyses, the AUC was 0.768 (95% CI 0.734–0.803) in the valve-only subgroup (n = 3089; GIB events = 111), 0.785 (95% CI 0.737–0.832) in the CABG+valve subgroup (n = 1484; events = 71), and 0.764 (95% CI 0.726–0.802) in the CABG-only subgroup (n = 5365; events = 110) (Fig. 4). AUCs were similar across subgroups (0.764–0.785), suggesting stable discrimination across procedure types.
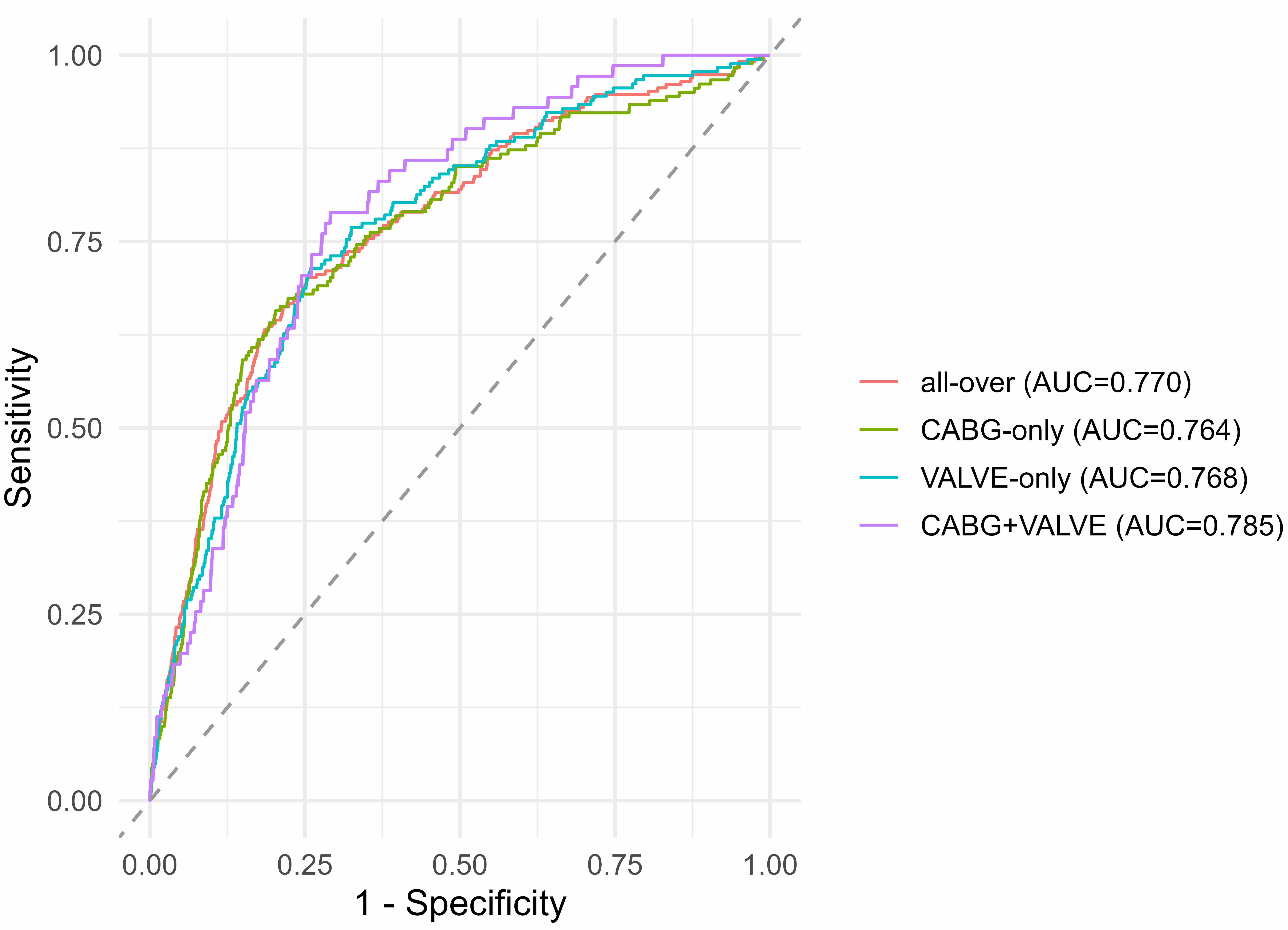 Fig. 4.
Fig. 4.
ROC curves of the prediction model across procedure subgroups.
The C-index was 0.798 (95% CI 0.747–0.849), with an optimism-corrected intercept of –0.234 and slope of 0.921. Brier scores were 0.0286 in the training set and 0.0273 in the validation set, indicating good agreement between predicted and observed risks. The bootstrap calibration curve in the validation set closely approximated the ideal line, supporting good calibration (Fig. 5). DCA suggested that the model provided greater net benefit when the threshold probability ranged from approximately 1% to 25% (Fig. 6).
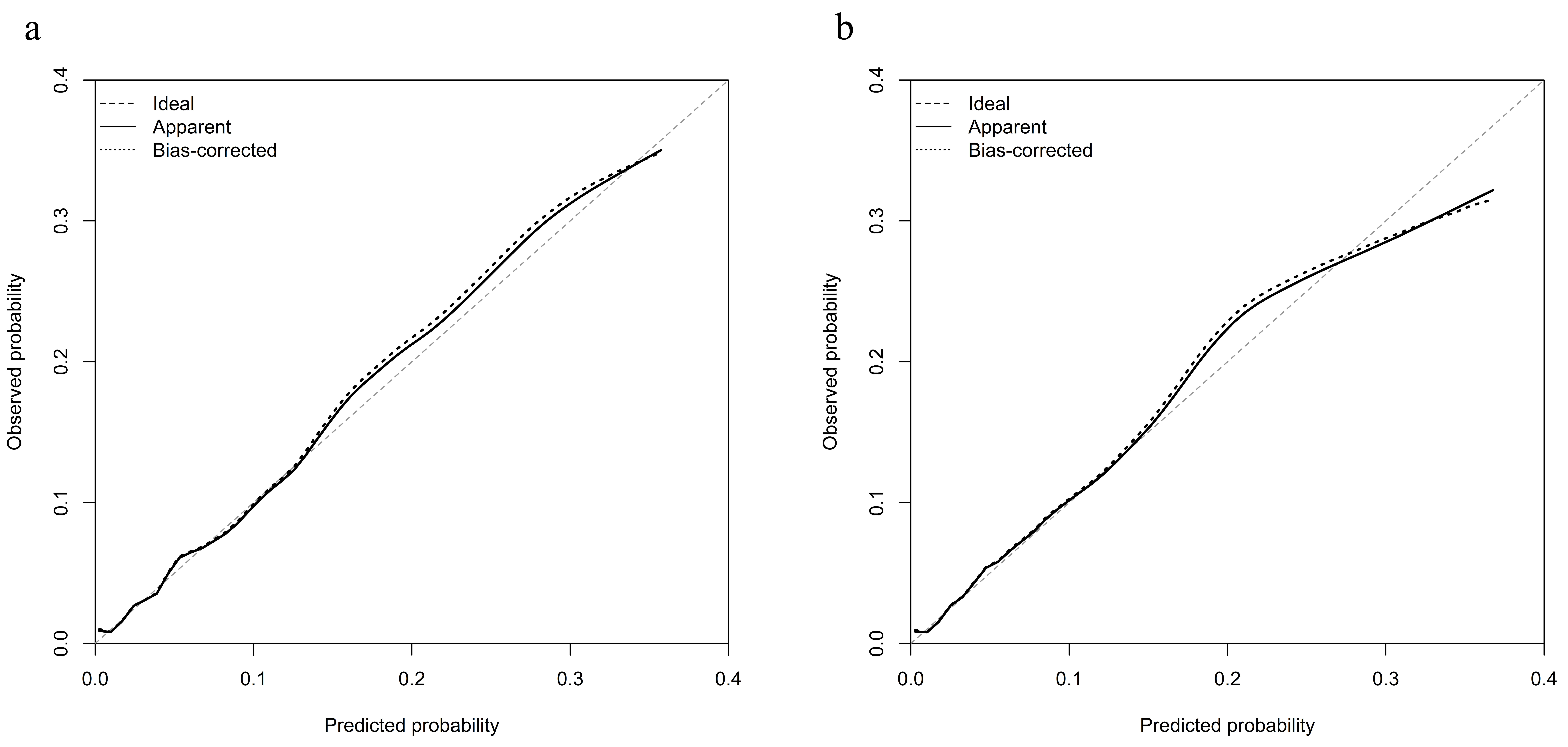 Fig. 5.
Fig. 5.
Calibration plot of the prediction model in the training and validation cohorts. (a) Calibration plot of the nomogram in the training cohort; (b) calibration plot in the validation cohort.
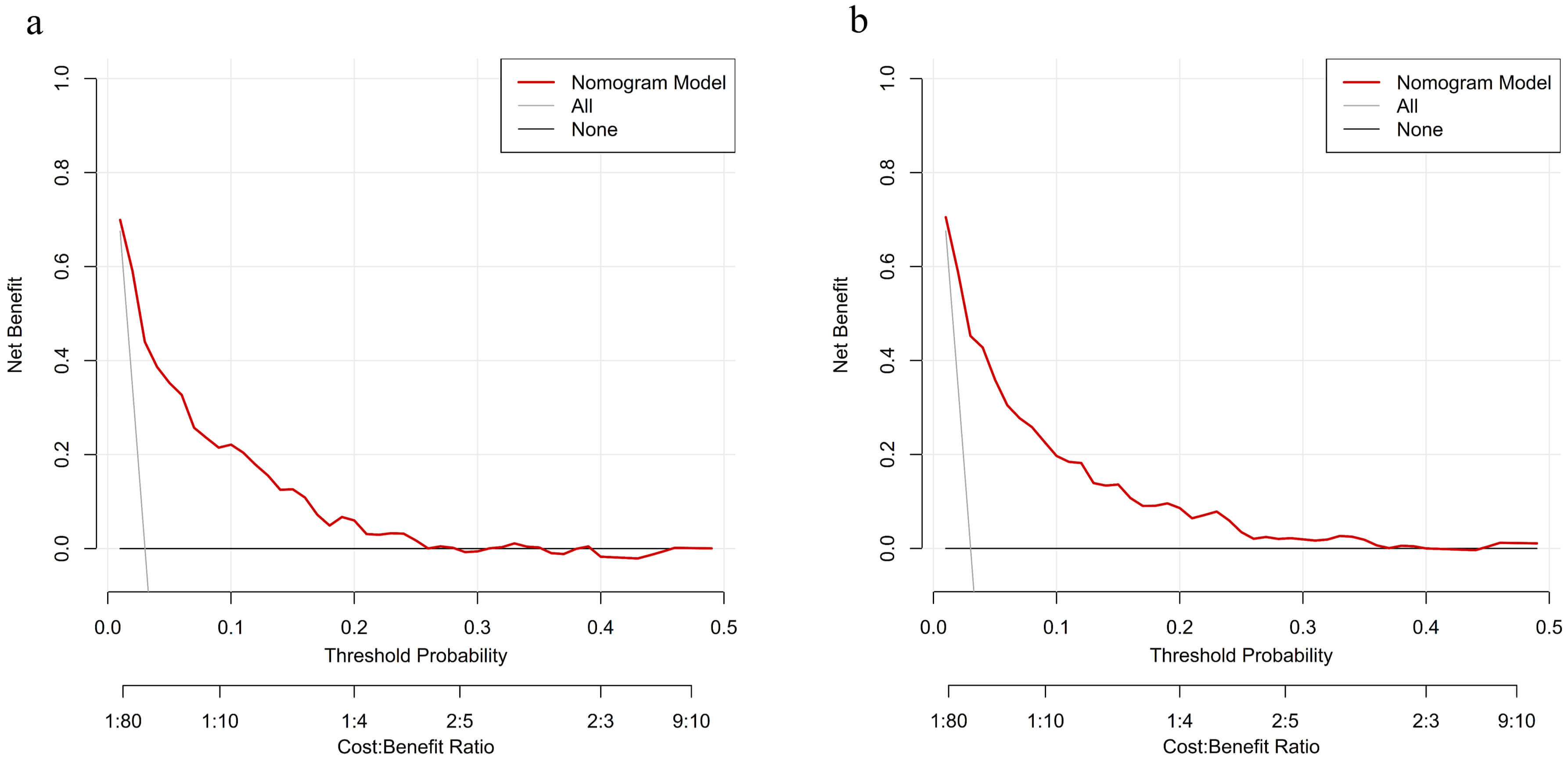 Fig. 6.
Fig. 6.
Decision curve analysis (DCA) of the nomogram in the training cohort and the validation cohort. (a) Decision curve analysis (DCA) of the nomogram in the training cohort; (b) DCA in the validation cohort.
The gastrointestinal tract is particularly vulnerable to ischemia–reperfusion injury and stress-related mucosal disease (SRMD) [12, 13]. After open cardiac surgery, patients are exposed to several converging insults—hypoperfusion, systemic inflammation, coagulopathy, and hemodynamic instability [8, 9], and gastrointestinal (GI) complications can occur across a broad clinical spectrum [10, 11]. In some cohorts, reported mortality among patients who develop postoperative GI complications reaches 21.5%–38% [26, 27, 28].
PCGIB is uncommon but carries substantial clinical consequences and is consistently associated with increased mortality, with in-hospital death rates reported at approximately 17% among affected patients in a large single-center cohort of 29,909 cases [1, 2, 3, 5]. PCGIB may also amplify the burden of infection and shock, prolong hospitalization [2, 6], and increase resource utilization [3, 4, 5]. These findings underscore the importance of early risk stratification.
Accordingly, we developed and internally validated an early prediction model for PCGIB after open cardiac surgery, focusing on clinical information available within 24 h after ICU admission, using MIMIC-IV, a publicly available, high-quality critical care database [15]. To our knowledge, this is the first model specifically developed for PCGIB after open cardiac surgery using MIMIC-IV.
Available evidence suggests that the occurrence of postoperative gastrointestinal bleeding indicates insufficient systemic perfusion, enhanced inflammatory response, or multiple organ dysfunction. Hypoperfusion and ischemia-reperfusion can increase the risk of SRMD [29], while coagulation dysfunction and anticoagulant and platelet aggregation treatments may lower the threshold for hemostasis [8], making mucosal damage more likely to manifest as clinically overt bleeding [30, 31, 32]. Therefore, relying solely on preoperative factors is insufficient to accurately predict the postoperative outcome.
The first 24 h after cardiac surgery represent a critical window of physiologic instability, as substantial clinical deterioration can occur during this period [9]. Rapid shifts in volume status, vascular tone, and myocardial performance can precipitate cardiogenic or vasoplegic shock, hypoperfusion, and impaired oxygen delivery. Early postoperative parameters capture both the immediate postoperative state and, in part, the cumulative effects of intraoperative hypoperfusion and inflammation.
Accordingly, early postoperative vital signs, laboratory abnormalities, and the intensity of pharmacologic data routinely collected in standard care, may capture key physiological domains (e.g., oxygen delivery, perfusion, and hemostasis) and provide a practical opportunity for early risk stratification [8, 33]. Leveraging such early postoperative information, rather than relying solely on preoperative variables, may improve clinical feasibility and predictive performance.
PCGIB may be delayed and clinically occult [1, 10, 34], and recognition can be
obscured by sedation, concurrent surgical bleeding, polypharmacy, and evolving
organ dysfunction [14]. Prior studies suggest that most postoperative GIB occurs
several days after surgery rather than immediately [7, 35, 36]. Bhat et
al. [35] reported that GIB occurred at a mean of 10.3
We used the first 24 h after ICU admission as the assessment window. During this early postoperative period—before most events occur—physiologic data are relatively comprehensive and readily accessible, enabling earlier identification of patients at risk. Focusing on early measurements also helps mitigate reverse causality by reducing the likelihood that predictors are downstream consequences of bleeding events.
The final model integrated comorbidity burden and early postoperative indicators of oxygenation and perfusion, coagulation, and transfusion requirements, reflecting the multifactorial pathophysiology of postoperative bleeding risk.
Clinically, heart rate, MAP, and early PaO2/SpO2-related indices likely capture early stability of oxygen delivery and perfusion [37, 38, 39, 40, 41]. Hypoxemia after cardiac surgery frequently coexists with hemodynamic instability, which may exacerbate tissue hypoxia and plausibly increase bleeding risk. A 6-h window is preferable because it supports actionable risk stratification early after ICU admission, facilitating timely surveillance and preventive interventions. A 24-h window may introduce information that is less useful for early decision-making and may be influenced by early postoperative events or management changes, thereby reducing the practical utility of the model.
Maximum APTT within 24 hours provided an objective signal of coagulopathy. Coagulation abnormalities may predispose patients to bleeding once mucosal injury develops. The 2024 Society of Critical Care Medicine (SCCM)/American Society of Hospital Pharmacists (ASHP) guideline identifies coagulopathy as a risk factor for clinically important stress-related upper GI bleeding [30].
Similarly, higher blood urea nitrogen (BUN) was associated with increased PCGIB risk, consistent with prior reports [42]. BUN may rise in upper GI bleeding due to absorption of blood proteins and increased urea production and may also reflect renal dysfunction and systemic stress, both of which are common in higher-risk patient [43].
RDW emerged as one of the most influential predictors and is readily available in routine testing. Similarly, prior studies have reported an independent association between higher RDW and postoperative GI bleeding risk in CABG populations (OR 2.83; 95% CI 1.46–5.51; p = 0.002) [44].
Blood transfusion may reflect a more complex operative course and more severe perioperative bleeding, and it may also interact with subsequent coagulation status and mucosal tolerance. Across cardiac and non-cardiac surgical cohorts, perioperative or postoperative RBC transfusion has been linked to higher risks of mortality, infection, ischemic complications, and greater resource utilization [45, 46, 47, 48].
Hyperlipidemia appeared protective in our model. This direction is consistent with emerging observational evidence suggesting that very low low-density lipoprotein cholesterol (LDL-C) levels are associated with higher in-hospital bleeding risk, including GI bleeding [49]. Studies in GI disease have also reported lower LDL-C in patients with peptic ulcer bleeding compared with those without bleeding [50], and lower LDL-C has been associated with bleeding outcomes in other clinical settings [51, 52].
Atrial fibrillation and chronic heart failure likely reflect a greater baseline cardiovascular disease burden, more frequent postoperative hemodynamic instability, and more exposure to anticoagulant and antiplatelet therapy, all of which may increase bleeding propensity [26, 53]. Chronic heart failure, HFrEF, and HFpEF are hierarchically related and correlated representations of the same underlying condition. In the stability-selection framework, the overall chronic heart failure indicator showed higher selection stability than the subtype variables, suggesting that it served as a more robust summary of heart failure status without introducing redundancy.
SAPS II, a widely used ICU severity score integrating physiologic derangement and multiorgan dysfunction, has been associated with adverse postoperative outcomes as well as clinically important bleeding risk in adult ICU populations [32].
This model has several strengths. We leveraged a large sample size, and incorporated multidimensional variables available early after surgery that more directly reflect postoperative physiology, and clear clinical interpretability. Predicted risk may be translated into actionable strategies, such as intensified mucosal protection, optimization of oxygenation and perfusion, individualized timing for resumption of antithrombotic therapy, and enhanced surveillance for occult bleeding.
Several limitations should be acknowledged. First, the model was developed using a single-center electronic health record (EHR) database (MIMIC-IV), and its generalizability to other cardiac surgical ICUs requires further validation. Although a 24-h postoperative landmark design was applied to mitigate reverse causation bias, occult bleeding may still have begun within the first 24 hours and could have influenced some predictors. Second, the low event rate (3.1%) limited outcome information content; in particular, the small number of events in higher-risk strata may reduce the stability of calibration estimates. To mitigate this, we performed rigorous internal validation (including bootstrap optimism correction [54] and procedure-stratified validation) and reported calibration and clinical net benefit in accordance with best practices [55]. Third, the study period spanned 2008–2022, during which surgical techniques, antithrombotic strategies, transfusion practices, and stress-ulcer prophylaxis may have evolved, introducing potential dataset drift. Furthermore, our reported regression coefficients and related precision measures may not fully reflect variability introduced by missing data and may be slightly optimistic. Future studies should evaluate alternative imputation specifications, conduct independent external validation in multicenter cohorts or other public databases, and evaluate the model’s generalizability and calibration stability in larger external datasets [56, 57, 58].
A 13-variable prediction model based on information available within the first 24 h after ICU admission in MIMIC-IV v3.1 achieved at least moderate discrimination in both the training and validation sets, with good calibration and measurable clinical net benefit. The model remained relatively robust across major cardiac surgery procedure types and may serve as an adjunct for early risk stratification and resource allocation. External validation and further refinement are needed before implementation as a clinical decision support tool.
AMI, acute myocardial infarction; APTT, activated partial thromboplastin time; ASHP, American Society of Health-System Pharmacists; AUC, area under the curve; BIDMC, Beth Israel Deaconess Medical Center; BMI, body mass index; BUN, blood urea nitrogen; C-index, concordance index; CABG, coronary artery bypass grafting; CAD, coronary artery disease; CI, confidence interval; CKD, chronic kidney disease; CVP, central venous pressure; DCA, decision curve analysis; EHR, electronic health record; EV, esophageal/gastric varices; GI, gastrointestinal; GIB, gastrointestinal bleeding; HCT, hematocrit; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; HR, heart rate; ICD-9, International Classification of Diseases, Ninth Revision; ICD-9/10, International Classification of Diseases, Ninth/Tenth Revision; ICU, intensive care unit; INR, international normalized ratio; IQR, interquartile range; LASSO, least absolute shrinkage and selection operator; LDL-C, low-density lipoprotein cholesterol; MAP, mean arterial pressure; MICE, multivariate imputation by chained equations; MIMIC-IV, Medical Information Mart for Intensive Care IV database; NE, norepinephrine; OR, odds ratio; PCGIB, post–cardiac surgery gastrointestinal bleeding; PT, prothrombin time; RBC, red blood cell; RDW, red cell distribution width; ROC, receiver operating characteristic; SAPS II, Simplified Acute Physiology Score II; SCCM, Society of Critical Care Medicine; SD, standard deviation; SOFA, Sequential Organ Failure Assessment; SQL, Structured Query Language; SRMD, stress-related mucosal disease; TRIPOD, Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis; UGIB, upper gastrointestinal bleeding; VIF, variance inflation factor; VIS, vasoactive-inotropic score; WBC, white blood cell; b, regression coefficient.
The data that support the findings of this study are derived from the MIMIC-IV database, which is publicly available at https://physionet.org/content/mimiciv/3.1/ for credentialed researchers who complete the required data use agreement.
Conceptualization, XL, QZ and XZ; methodology, XZ; software, XL and QS; validation, QS and ZL; formal analysis, XL, QS, and ZL; investigation, XL; resources, XL; data curation, XL; writing—original draft preparation, XL and QS; writing—review and editing, QZ, ZL and XZ; visualization, XL and QS; supervision, QZ and XZ; project administration, QZ. All authors meet the ICMJE authorship criteria, have read and agreed to the published version of the manuscript, and take responsibility for the integrity of the work.
The study used data from the publicly available, de-identified MIMIC-IV database, which was approved by the Institutional Review Boards of the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center; therefore, additional ethical approval and informed consent were waived.
We would like to express our gratitude to the Massachusetts Institute of Technology Laboratory for Computational Physiology and the Beth Israel Deaconess Medical Center for the MIMIC Project.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGpt-4.0 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF50244.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






