1 Department of Cardiovascular Surgery, Beijing Anzhen Hospital, Capital Medical University, 100011 Beijing, China
2 Beijing Institute of Heart, Lung and Blood Vessel Diseases, 100011 Beijing, China
3 School of Nursing, Capital Medical University, 100069 Beijing, China
Abstract
This study aimed to investigate the impact of the modified Morrow procedure, with or without concomitant mitral valve surgery, on Kansas City Cardiomyopathy Questionnaire (KCCQ) scores and quality of life (QoL) in patients with hypertrophic obstructive cardiomyopathy (HOCM), and to provide an evidence base for the development of perioperative management strategies.
A retrospective analysis was conducted on 82 patients with HOCM who underwent the modified Morrow procedure. Patients were divided into two groups: those who received the modified Morrow procedure alone (n = 41) and those who underwent the combined procedure (n = 41). Baseline characteristics, postoperative follow-up parameters, and changes in KCCQ scores were compared between the two groups.
The two groups were comparable at baseline, except for a significant difference in preoperative mitral regurgitation (p < 0.05). No significant differences were observed between the groups in postoperative KCCQ scores or cardiac function parameters (p > 0.05). Repeated-measures analysis of variance revealed a significant main effect of time (p < 0.001), but no significant main effect of surgical group or time-by-group interaction (p > 0.05).
The modified Morrow procedure significantly improves QoL in patients with HOCM. Concomitant mitral valve surgery does not impose an additional burden on KCCQ scores. Clinical efforts should focus on meticulous perioperative management throughout the care cycle and on personalized cardiac rehabilitation guidance to facilitate postoperative recovery. Ultimately, these measures support a closed-loop “assessment–intervention–monitoring–follow-up” management system that continuously safeguards the QoL and the long-term prognosis of patients undergoing the combined procedure.
Keywords
- modified Morrow procedure
- mitral valve surgery
- hypertrophic obstructive cardiomyopathy (HOCM)
- KCCQ score
- management strategies
Hypertrophic obstructive cardiomyopathy (HOCM) is a common inherited cardiomyopathy characterized by abnormal ventricular wall thickening and left ventricular outflow tract (LVOT) obstruction. The clinical presentation is often severe, predisposing patients to malignant arrhythmias and sudden cardiac death, significantly threatening their life and quality of life (QoL) [1, 2]. The modified Morrow procedure is the standard surgical treatment for drug-refractory, severely symptomatic HOCM patients, effectively relieving LVOT obstruction, improving clinical symptoms, and optimizing long-term prognosis [3, 4]. HOCM patients often present with mitral regurgitation (MR), primarily due to systolic anterior motion (SAM) of the mitral valve leaflet, while in some cases, it stems from intrinsic mitral valve pathology [5]. Whether to intervene on the mitral valve during surgery has long been a subject of debate.
From a clinical management perspective, surgical success is only one crucial dimension of evaluating HOCM treatment outcomes; patients’ postoperative QoL is equally a core concern in clinical practice [6]. The Kansas City cardiomyopathy questionnaire (KCCQ) is an internationally recognized, standardized self-administered instrument for assessing the health status of patients with heart failure. Its advantage lies in its sensitivity to capturing postoperative changes in symptom improvement, physical function recovery, social function adaptation, and overall QoL enhancement. However, studies utilizing the KCCQ to evaluate QoL in patients undergoing the modified Morrow procedure with concomitant mitral valve surgery are limited [7, 8, 9].
Based on this background, this study, from a clinical management perspective, aims to retrospectively analyze the clinical data of 82 HOCM patients who underwent surgical treatment at our hospital. It focuses on exploring whether concomitant mitral valve surgery during the modified Morrow procedure imposes an additional burden on postoperative QoL, as represented by KCCQ scores. The goal is to provide a scientific basis for optimizing the perioperative clinical management pathway assessment system, developing personalized health education plans, and implementing precise rehabilitation guidance for HOCM patients, thereby enhancing the quality of postoperative recovery and survival experience.
This study screened hospitalized HOCM patients who underwent either the modified Morrow procedure alone or the combined procedure at Beijing Anzhen Hospital, Capital Medical University, between January 2022 and December 2024, using the Hospital Information System (HIS).
Inclusion criteria: (1) Diagnosed with HOCM (diagnostic criteria based on the
“Chinese Guidelines for the Diagnosis and Treatment of Adult Hypertrophic
Cardiomyopathy 2023”) [10, 11]; (2) Resting or provoked LVOT gradient
Exclusion criteria: (1) Echocardiography not meeting HOCM diagnostic criteria; (2) Incomplete echocardiographic data; (3) Missing other baseline data; (4) Comorbid severe hepatic or renal dysfunction, malignancy, mental illness, or incomplete clinical data.
Surgical indications and group allocation: All patients underwent the modified
Morrow procedure for standard indications. Concomitant mitral valve surgery
(repair) was performed based on predefined criteria: (1) moderate/severe MR
(regurgitant jet area
Ultimately, 82 HOCM patients were enrolled and divided into two groups based on the surgical approach: the modified Morrow procedure alone group (n = 41) and the combined procedure group (n = 41). The Ethics Committee of Beijing Anzhen Hospital, Capital Medical University approved this study. All patients gave their informed consent in compliance with the principles of the Declaration of Helsinki.
All enrolled patients underwent baseline assessment within 24 hours of admission, and the following data were collected; three experienced clinicians and nurses independently reviewed the data to ensure accuracy: (1) Demographic and clinical baseline data: age, gender, height, body mass index, past medical history, and comorbidities.
(2) Echocardiographic parameters: Left atrial diameter (LAD), interventricular septum thickness (IVST), left ventricular end-diastolic volume (LVVd), left ventricular ejection fraction (LVEF), left ventricular fractional shortening (LVFS), maximum velocity across the LVOT (Vmax), peak pressure gradient across the LVOT (PG), and MR severity.
(3) Perioperative data: Surgical approach, key operative parameters, length of hospital stay.
QoL assessment: The first visit at Beijing Anzhen Hospital in January 2022 was defined as the follow-up starting point. Follow-up was completed at 6 months postoperatively via outpatient review, telephone call, or re-hospitalization. The short form of the Kansas City cardiomyopathy questionnaire (KCCQ-12) was used to assess QoL before surgery and at 6 months postoperatively, covering domains such as symptoms, physical limitations, social limitations, and quality of life. Scores range from 0 to 100, with higher scores indicating better QoL.
SPSS version 26.0 (version number: 26.0.0.0, IBM Corp., Armonk, New York, USA)
was used for statistical analysis. Normally distributed continuous data are
presented as mean
A total of 82 HOCM patients were enrolled and divided into the modified Morrow
procedure alone group (n = 41) and the combined procedure group (n = 41), the
combined procedure group consisted of patients undergoing isolated mitral
annuloplasty, shown in Table 1. In the isolated procedure group, the mean age was
47.39
| Variable | Total (n = 82) | Isolated procedure group (n = 41) | Combined procedure group (n = 41) | p value |
| Ages (years) | 50.00 |
47.39 |
52.67 |
0.051 |
| Male, n (%) | 51 (62.20) | 26 (63.41) | 25 (60.98) | 0.820 |
| HTN, n (%) | 24 (29.27) | 14 (34.15) | 10 (24.39) | 0.332 |
| T2D, n (%) | 3 (3.66) | 2 (4.88) | 1 (2.44) | |
| AF, n (%) | 5 (6.10) | 1 (2.44) | 4 (9.76) | 0.356 |
| CAD, n (%) | 12 (14.63) | 3 (7.32) | 9 (21.95) | 0.061 |
| KCCQ | 41.88 |
42.61 |
41.15 |
0.679 |
| LAD (mm) | 44.22 |
44.16 |
44.27 |
0.933 |
| IVST (mm) | 20.43 |
20.84 |
20.06 |
0.522 |
| LVPWT (mm) | 12.68 |
12.70 |
12.65 |
0.952 |
| LVVd (mL) | 81.18 |
78.68 |
83.67 |
0.517 |
| LVVs (mL) | 24.33 |
24.65 |
24.02 |
0.798 |
| LVEF, (%) | 67.82 |
67.68 |
67.95 |
0.857 |
| LVFS, (%) | 37.97 |
37.62 |
38.31 |
0.596 |
| Vmax (cm/s) | 363.61 |
364.68 |
362.62 |
0.946 |
| PG (mmHg) | 64.56 |
66.11 |
63.08 |
0.724 |
| MR (cm2) | 8.19 |
7.30 |
8.94 |
0.039 |
KCCQ, Kansas City cardiomyopathy questionnaire; LAD, left atrial diameter; IVST, interventricular septal thickness; LVPWT, left ventricular posterior wall thickness; LVVd, left ventricular end-diastolic volume; LVVS, left ventricular end-systolic volume; LVEF, left ventricular ejection fraction; LVFS, left ventricular fractional shortening; Vmax, maximum velocity across the left ventricular outflow tract; PG, peak pressure gradient across the left ventricular outflow tract; MR, mitral regurgitation (regurgitant jet area in cm2); HOCM, hypertrophic obstructive cardiomyopathy; HTN, hypertension; T2D, type 2 diabetes; AF, atrial fibrillation; CAD, coronary artery disease.
As shown in Table 2, statistically significant differences were observed between the two groups in aortic cross-clamp time (Z = –3.080, p = 0.002) and cardiopulmonary bypass time (Z = –2.781, p = 0.005). The median aortic cross-clamp time was 150.0 min (127.0–182.0 min) in the combined procedure group and 113.8 min (100.5–144.3 min) in the isolated procedure group. The median cardiopulmonary bypass time was 122.0 min (102.0–178.0 min) and 80.0 min (80.0–122.0 min), respectively. However, no significant differences were found between the two groups in the duration of mechanical ventilation (Z = –1.346, p = 0.178), surgical time (Z = –1.218, p = 0.223), length of intensive care unit (ICU) stay (Z = –1.729, p = 0.084), or total hospital stay (Z = –0.796, p = 0.426). These results indicate that although the combined procedure required longer intraoperative times due to the additional handling of the mitral valve, it did not significantly prolong postoperative recovery time or hospitalization.
| Variables | Total (n = 82) | 1 (n = 41) | 2 (n = 41) | Statistic | p |
| Hospital stay, quant median, M (Q1, Q3) | 16.000 (13.000, 23.000) | 16.000 (13.000, 23.000) | 16.000 (13.000, 16.000) | Z = –0.796 | 0.426 |
| Cross-clamp times, quant median, M (Q1, Q3) | 138.500 (107.125, 174.000) | 113.750 (100.500, 144.250) | 150.000 (127.000, 182.000) | Z = –3.080 | 0.002 |
| Cardiopulmonary bypass times, quant median, M (Q1, Q3) | 112.000 (96.500, 136.000) | 80.000 (80.000, 122.000) | 122.000 (102.000, 178.000) | Z = –2.781 | 0.005 |
| Duration of mechanical ventilation, quant median, M (Q1, Q3) | 25.000 (18.750, 49.500) | 18.750 (15.000, 25.000) | 25.000 (18.750, 49.500) | Z = –1.346 | 0.178 |
| Surgical time, quant median, M (Q1, Q3) | 6.000 (5.000, 7.000) | 6.000 (5.000, 7.000) | 6.000 (5.000, 7.000) | Z = –1.218 | 0.223 |
| Length of ICU stay, quant median, M (Q1, Q3) | 21.000 (18.000, 60.000) | 19.000 (15.000, 21.000) | 21.000 (19.000, 60.000) | Z = –1.729 | 0.084 |
ICU, intensive care unit; M (Q1, Q3), the median (first quartile, third quartile).
There were no significant differences between the two groups in postoperative
KCCQ scores, LAD, IVST, LVVd, LVEF, LVFS, Vmax, PG, or MR at the 6-month
follow-up (p
| Variable | Total (n = 82) | Isolated procedure group (n = 41) | Combined procedure group (n = 41) | p-value |
| KCCQ after surgery | 57.76 |
57.95 |
57.56 |
0.842 |
| LAD (mm) | 39.05 |
38.94 |
39.14 |
0.914 |
| IVST (mm) | 12.84 |
12.59 |
13.02 |
0.671 |
| LVVd (mL) | 36.03 |
36.61 |
35.48 |
0.914 |
| LVVs (mL) | 15.35 |
13.42 |
17.19 |
0.389 |
| LVEF (%) | 60.36 |
61.06 |
59.82 |
0.495 |
| LVFS (%) | 32.88 |
33.33 |
32.50 |
0.617 |
| Vmax (cm/s) | 135.17 |
143.00 |
129.30 |
0.236 |
| PG (mmHg) | 10.26 |
12.31 |
6.67 |
0.222 |
| MR* (n) | 0.474 | |||
| Without | 80 (97.56) | 39 (95.12) | 41 (100.00) | |
| With | 2 (2.44) | 2 (4.88) | 0 (0.00) |
KCCQ, Kansas City cardiomyopathy questionnaire; LAD, left atrial diameter; IVST,
interventricular septal thickness; LVVd, left ventricular end-diastolic volume;
LVVS, left ventricular end-systolic volume; LVEF, left ventricular ejection
fraction; LVFS, left ventricular fractional shortening; Vmax, maximum velocity
across the left ventricular outflow tract; PG, peak pressure gradient across the
left ventricular outflow tract; MR, mitral regurgitation; MR*, moderate or severe
mitral regurgitation. According to echocardiographic criteria, mitral
regurgitation is considered moderate to severe when the regurgitant jet area is
No significant intergroup difference was observed in preoperative KCCQ scores
(p = 0.679). A significant postoperative improvement in KCCQ scores was
noted in both the isolated (42.61
| Variable | Time point | Isolated procedure group (n = 41) | Combined procedure group (n = 41) | p value (groups) |
| KCCQ score | preoperative | 42.61 |
41.15 |
0.679 |
| postoperative | 57.95 |
57.56 |
0.842 | |
| p value (times) |
KCCQ, Kansas City cardiomyopathy questionnaire.
Further simple effect analysis showed that postoperative KCCQ scores increased
significantly compared to preoperative scores in both groups (p
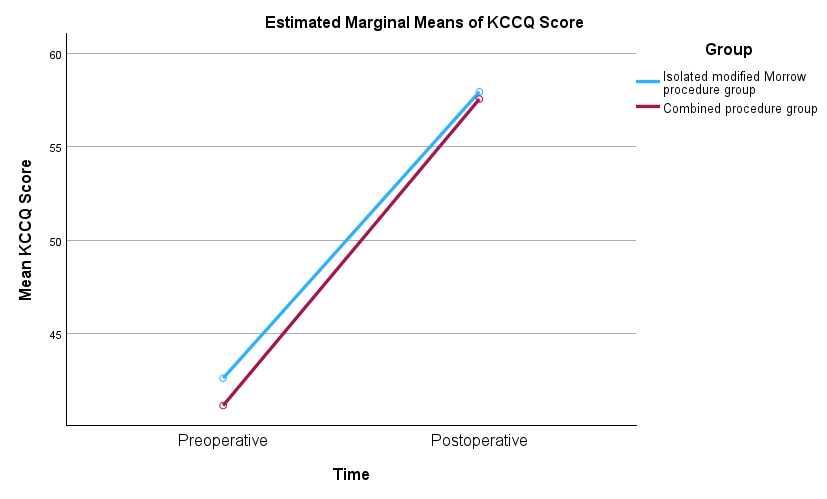 Fig. 1.
Fig. 1.
Estimated marginal means of KCCQ scores from the pre- to postoperative period, by group. KCCQ, Kansas City cardiomyopathy questionnaire.
Univariate and multivariate linear regression analyses were performed to explore factors associated with postoperative KCCQ score. Univariate analysis showed that preoperative interventricular septum thickness (IVST) was significantly associated with improvement in postoperative KCCQ score (p = 0.019). After adjusting for gender, age, preoperative KCCQ grade, and left ventricular structural and functional parameters in the multivariate analysis, IVST remained an independent predictor (p = 0.040) (Table 5). The results indicated that greater preoperative interventricular septum thickness was associated with higher postoperative KCCQ scores, and the difference was statistically significant.
| Variables | Univariate linear analysis | Multivariate linear regression | ||
| p | p | |||
| Ages (years) | –0.157 (–0.313 to 0.000) | 0.053 | ||
| KCCQ before surgery | 0.263 (0.156 to 0.370) | 0.198 (0.079–0.317) | 0.002 | |
| LAD (mm) | –0.006 (–0.330 to 0.317) | 0.969 | ||
| IVST (mm) | 0.484 (0.087 to 0.880) | 0.019 | 0.421 (0.028–0.814) | 0.040 |
| LVPWT (mm) | 0.167 (–0.470 to 0.804) | 0.610 | ||
| LVVd (mL) | –0.020 (–0.076 to 0.035) | 0.474 | ||
| LVVs (mL) | –0.101 (–0.279 to 0.077) | 0.270 | ||
| LVEF (%) | 0.232 (–0.071 to 0.534) | 0.137 | ||
| LVFS (%) | 0.303 (–0.054 to 0.660) | 0.100 | ||
| Vmax (cm/s) | 0.017 (0.002 to 0.032) | 0.026 | ||
| PG (mmHg) | 0.062 (0.006 to 0.118) | 0.034 | 0.060 (0.008–0.111) | 0.027 |
| MR (cm2) | –0.043 (–0.636 to 0.550) | 0.887 | ||
| Gender (%) | ||||
| Male | 0.000 (Reference) | |||
| Female | –1.527 (–5.470 to 2.416) | 0.450 | ||
| Procedure group | ||||
| Isolated procedure group | 0.000 (Reference) | |||
| Combined procedure group | –0.390 (–4.227 to 3.446) | 0.842 | ||
KCCQ, Kansas City cardiomyopathy questionnaire; LAD, left atrial diameter; IVST, interventricular septal thickness; LVPWT, left ventricular posterior wall thickness; LVVd, left ventricular end-diastolic volume; LVVS, left ventricular end-systolic volume; LVEF, left ventricular ejection fraction; LVFS, left ventricular fractional shortening; Vmax, maximum velocity across the left ventricular outflow tract; PG, peak pressure gradient across the left ventricular outflow tract; MR, mitral regurgitation.
The results of this study show that KCCQ scores significantly improved at the
6-month follow-up in HOCM patients, regardless of whether they underwent the
isolated modified Morrow procedure or the combined procedure (p
From a clinical management perspective, although early postoperative KCCQ scores did not differ between the combined surgery group and the isolated surgery group, the particularity of the former lies in the need for long-term anticoagulation (especially lifelong for those with mechanical valve replacements), stricter long-term monitoring of mitral valve function, and more stringent heart rate control. While these factors did not manifest as differences in KCCQ scores at 6 months, they are crucial for influencing long-term QoL and clinical outcomes. It is essential to construct a comprehensive management process tailored to their characteristics of “high long-term anticoagulation needs, stringent heart rate monitoring requirements, and need for long-term mitral valve function follow-up”.
Furthermore, the KCCQ, as a heart failure-specific tool, has been proven sensitive to perioperative interventions. Research by Sherrod et al. [9] showed that every 10-point increase in the KCCQ overall summary score is associated with an 18% reduction in the risk of heart failure hospitalization or death within one year. For combined surgery patients, irregular anticoagulation or significant heart rate fluctuations might offset the KCCQ improvement gained from surgery and even increase the risk of thrombosis/bleeding. Therefore, clinical management should not stop at the conclusion of “no difference in early postoperative KCCQ”. Instead, it must address the long-term management challenges specific to combined surgery patients. In clinical practice, using a “disease journey map” as a framework, precisely recording MR baseline and coagulation function preoperatively, managing international normalized ratio (INR) postoperatively via a “Valve Anticoagulation e-Assistant”, monitoring heart rate with wearable devices, and leveraging mini-programs and official accounts after discharge to achieve intelligent transitional care encompassing “anticoagulation self-management-KCCQ score tracking - re-examination reminders” are crucial. This ultimately forms a closed-loop “Assessment-Intervention-Monitoring-Follow-up” management system, building a management framework centered on “intelligent tools + full-process closed-loop” to continuously safeguard the QoL and long-term prognosis of patients undergoing combined mitral valve surgery.
Using the disease journey map as a framework, identify core management nodes in the preoperative phase. In addition to routine cardiac function assessment, KCCQ scoring, and admission evaluation, enhance dynamic monitoring of MR grade for combined surgery patients, record regurgitation jet area, valve leaflet morphology, and left atrial pressure via transthoracic echocardiography [14], and incorporate this data into the “preoperative baseline” module of the journey map. Simultaneously, complete baseline coagulation function testing. If preoperative MR is accompanied by left atrial thrombus, anticoagulation may need to be initiated preoperatively to strive for thrombus resolution.
Preoperative anxiety is the most common negative emotion in HOCM patients and can directly affect the speed of postoperative recovery and QoL. Nurses can implement stratified education based on KCCQ scores and anxiety scale assessment results [15]. Utilize visual aids, precise education, combined with videos and sharing previous successful cases to explain the protective effect of combined surgery on long-term cardiac function; alleviate patient concerns about “slower recovery with combined surgery”, help patients establish positive treatment expectations, and enhance treatment confidence. Simultaneously, through preoperative preparation videos combined with face-to-face education, instruct patients in breathing exercises and bed-bound defecation training; complete preoperative preparations, including surgical site skin preparation, and guide and assist patients with personal hygiene and oral care to reduce the risk of postoperative infection and promote recovery.
For combined mitral valve surgery patients, initiate anticoagulation therapy immediately postoperatively. Utilize the “Valve Anticoagulation e-Assistant” for fully digitalized management. Healthcare providers guide patients and family members to record daily warfarin administration time and dosage via a mini-program. The mini-program syncs INR test results and suggests dosage adjustments based on INR values. Nurses can log into the backend to review data. Patients can present their anticoagulation data during and follow-up visits, allowing healthcare providers to guide medication based on the data, avoiding bleeding or thrombotic risks.
The most common postoperative complication of the modified Morrow procedure is arrhythmia [6], most frequently conduction abnormalities. Closely monitor vital signs to promptly detect and manage complications [16]. Introduce wearable monitoring devices worn on the patient’s wrist to monitor heart rate and rhythm in real-time without restricting patient mobility. Simultaneously, keep a temporary pacemaker ready at the bedside, checking its threshold, battery level, and lead connection each shift to ensure immediate availability for emergency pacing. Adjust fluid infusion rates appropriately, guide early mobilization, implement enhanced recovery after surgery (ERAS) principles to improve patient compliance with early breathing exercises and ambulation, promoting cardiopulmonary recovery [17]. Employ multimodal analgesia to enhance patient comfort. Develop individualized rehabilitation plans based on patient condition, including exercise, diet, and lifestyle adjustments.
Use the teach-back method before discharge to assess understanding of the patient and family members. Emphasize the importance of long-term self-management, instruct patients to adhere to medication (especially beta-blockers, warfarin), schedule regular echocardiographic follow-ups, monitor weight changes, heart rate variations, and avoid strenuous exercise and dehydration. Re-train on the use of the “Valve Anticoagulation e-Assistant” to ensure patients can independently record medication intake and query INR information. Continue the use of wearable devices; the hospital can remotely monitor heart rate and rhythm for three months postoperatively, utilizing information platforms to provide transitional care, offering remote guidance to patients with abnormal heart rate changes. Guide patients to initiate self-monitoring at home later. Regularly follow up on KCCQ score changes via WeChat, phone calls, or mini-programs to promptly understand their QoL status and provide guidance, building an integrated “hospital-family” chronic disease management model [18].
The combined procedure was associated with longer aortic cross-clamp and cardiopulmonary bypass times. Although no significant increase in early postoperative complications was observed in this cohort, extended operative and perfusion time may elevate the risk of neurological, respiratory, and inflammatory complications. This underscores the importance of meticulous intraoperative management and postoperative monitoring in patients undergoing combined surgery. Future studies with larger samples and longer follow-up are needed to better evaluate the safety profile of the combined approach.
(1) Single-center, retrospective design, susceptible to selection bias and confounding factors; (2) Relatively small sample size of only 82 patients, which may be underpowered to detect small differences; (3) Follow-up endpoint was 6 months postoperatively, preventing assessment of the long-term stability of KCCQ scores; (4) The 6-month follow-up period was too short to allow for a meaningful comparison of major clinical events (e.g., mortality, heart failure rehospitalization) between the two surgical groups.
The modified Morrow procedure significantly improves KCCQ scores and QoL in HOCM patients. Concomitant mitral valve surgery does not add burden to patient KCCQ scores. Greater preoperative interventricular septum thickness is associated with higher postoperative KCCQ scores. This underscores the positive value of the surgery in improving patient QoL. In summary, when HOCM patients have coexisting mitral valve pathology requiring surgical intervention, surgeons can opt for the modified Morrow procedure combined with mitral valve surgery based on clear surgical indications, and patients need not excessively worry about potential negative impacts of this combined surgical approach on postoperative QoL.
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
MH, XTL, and HC conceived the study; YQC, XTL, XRZ, and KZ curated the data; KZ, XWZ and HC performed the formal analysis; YQL acquired the funding; MH, XL, and YQL conducted the investigation; XRZ, CWR and YQL developed the methodology; MH, QYL, and XTL administered the project; XRZ, YQL, HC, and CWR provided the resources; HC developed the software; YQL, CWR and HC supervised the study; HC validated the results; XL and XRZ wrote the original draft; MH, CWR, XTL, KZ, YQC, XWZ, HC, and YQL reviewed and edited the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study follows the Declaration of Helsinki. The Ethics Committee of Beijing Anzhen Hospital, Capital Medical University, approved this study (approval ID: 2025266X). This study is a retrospective analysis of existing medical records data. According to the approval from our Institutional Review Board (IRB) and Ethics Committee, informed consent was waived.
Not applicable.
This research was approved by the National Natural Science Foundation of China (Grant No. 82170374).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

