1 Department of Cardiovascular Surgery, Beijing Chao-Yang Hospital, Capital Medical University, 100020 Beijing, China
†These authors contributed equally.
Abstract
Gastrointestinal bleeding events represent a serious complication after coronary revascularization. In recent years, hybrid coronary revascularization (HCR) has gradually become one of the main surgical methods for multivessel coronary artery disease. This study aimed to identify risk factors for gastrointestinal bleeding following one-stop HCR.
This single-center retrospective study included 231 patients with coronary artery disease who underwent one-stop HCR between April 2018 and April 2023. Baseline conditions and perioperative parameters of patients were collected from electronic medical records; gastrointestinal bleeding events were documented, and retrospective statistical analyses were performed.
Among the 231 patients, 12 experienced gastrointestinal bleeding events, yielding an incidence rate of 5.2%; three patients died due to gastrointestinal bleeding, corresponding to a mortality rate of 25.0% among patients with bleeding events. Compared with patients without bleeding events, those with gastrointestinal bleeding had significantly higher rates of prior cerebrovascular disease (50.0% vs. 21.0%), preoperative creatinine (defined as the last serum creatinine measured within 24 h before surgery) (78.5 vs. 67.7 μmol/L), perioperative total blood transfusion volume during the period (600 vs. 0 mL), perioperative blood transfusion >5 units (33.3% vs. 8.7%), intraoperative bleeding volume (400 vs. 200 mL), total postoperative drainage volume (1453 vs. 1160 mL), mechanical ventilation time (18.0 vs. 16.0 h), and mortality (25.0% vs. 3.2%) (all p < 0.05). Single-factor and multi-factor regression analyses identified prior cerebrovascular disease (CVD) as an independent risk factor for gastrointestinal bleeding (odds ratio (OR) = 3.754, 95% confidence interval (CI) = 1.202–11.724; p = 0.023). Mortality was significantly higher in the gastrointestinal bleeding group than in the control group.
Previous cerebrovascular disease is a risk factor for gastrointestinal bleeding after one-stop HCR. In such patients, antiplatelet strategies should be appropriately adjusted to prevent and reduce gastrointestinal bleeding. However, the number of events in our study was limited, and future studies should incorporate larger sample sizes to validate these findings.
Keywords
- gastrointestinal bleeding
- hybrid coronary revascularization
- risk factor
- dual antiplatelet therapy
- cerebrovascular disease
The occurrence of gastrointestinal bleeding after coronary revascularization represents a serious complication of cardiac surgery, associated with high mortality and disability rates [1]. Several retrospective studies have reported various bleeding-related complications after coronary revascularization [1, 2]. However, to our knowledge, no studies have specifically evaluated the risk factors for upper gastrointestinal bleeding after hybrid coronary revascularization (HCR), a combination of stent percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG), primarily used to revascularize multivessel coronary artery disease [3]. HCR combines the minimally invasive features of PCI with the long-term patency of CABG grafts, effectively reducing the occurrence of postoperative major adverse cardiovascular and cerebrovascular events (MACCEs). HCR also shortens intensive care unit (ICU) and overall hospital stays, reduces ventilation time, lowers the perioperative period, decreases the need for blood transfusions, and reduces the incidence of atrial fibrillation; thus, HCR has become one of the main revascularization methods currently used to treat multivessel coronary artery disease [4]. As the clinical application of HCR increases, the safety of the associated perioperative management has attracted more attention, especially the balance between antithrombotic therapy and bleeding risk. Dual antiplatelet therapy (DAPT) is recommended after both CABG and PCI [5]. Similarly, antiplatelet therapy is commonly prescribed after percutaneous (transcatheter) heart valve replacement (i.e., transcatheter aortic valve replacement/implantation, TAVR/TAVI) to reduce thrombotic events and recurrent ischemic complications, with the choice between single and DAPT based on patient-specific ischemic and bleeding risk as well as current evidence and guidelines [6]. However, this increases the risk of bleeding-related complications after cardiac surgery. Although gastrointestinal bleeding after general coronary revascularization has been of concern, upper gastrointestinal bleeding, as a specific subtype characterized by more rapid progression and poorer prognosis, has not been specifically studied in the HCR population. Existing studies on bleeding complications after HCR have primarily focused on cardiovascular-related bleeding, such as mediastinal and wound bleeding, with limited systematic exploration of upper gastrointestinal bleeding. Consequently, the incidence rates, risk factors, and clinical impact of upper gastrointestinal bleeding in HCR patients remain unclear. Therefore, identifying the risk factors associated with upper gastrointestinal bleeding is necessary. Thus, this study aimed to review and collect relevant preoperative, intraoperative, and postoperative data from patients to identify risk factors for upper gastrointestinal bleeding after HCR, thereby helping surgeons identify high-risk patients early and implement timely, effective interventions to improve survival rates and quality of life.
This single-center retrospective study included 163 men and 68 women who underwent one-stop HCR at Beijing Chaoyang Hospital, affiliated with Capital Medical University, from April 2018 to April 2023. HCR is suitable for patients with multivessel disease involving the left main coronary artery (LMCA) and/or the proximal left anterior descending artery (LAD), in whom CABG and PCI are high-risk or difficult, or when a single method cannot achieve the best outcome. A total of 231 patients were included in the study: 12 (5.2%) experienced gastrointestinal bleeding and 219 (94.8%) did not. According to the follow-up protocol, discharged patients were required to attend the outpatient clinic for re-examination at 6 weeks and 6 months after discharge, and then annually. In addition to routine follow-up visits, researchers contacted all discharged patients by telephone again before October 2023. For patients who experienced gastrointestinal bleeding and were admitted to the hospital, the emergency and inpatient medical records from that visit were reviewed. If the patient had been treated at another hospital, the patient was asked about their medical records to clarify the diagnosis of gastrointestinal bleeding. Since this was a retrospective study, the requirement for informed consent was waived.
(1) Patient selection: adults (age
(2) Coronary anatomy: multivessel coronary artery disease (
(3) Surgical indications: patients deemed high-risk candidates for conventional CABG or PCI by a multidisciplinary heart team.
(4) Data completeness: availability of complete perioperative records, including antiplatelet/anticoagulant regimens, bleeding events, and imaging data.
(5) Baseline stability: no evidence of active gastrointestinal bleeding, coagulopathy, or other uncontrolled bleeding risks preoperatively.
(1) Preoperative bleeding risk: active gastrointestinal bleeding or a history of gastrointestinal bleeding within the preceding 6 months, including a history of peptic ulcer disease, prior upper gastrointestinal bleeding, or gastric ulcer.
(2) Organ dysfunction: severe hepatic insufficiency (Child–Pugh class C) or
renal insufficiency (estimated glomerular filtration rate (eGFR)
(3) Anticoagulant use: long-term use of oral anticoagulants (e.g., warfarin, direct oral anticoagulants) that cannot be temporarily discontinued perioperatively.
(4) Missing data: incomplete records (e.g., undocumented antiplatelet strategies, loss to follow-up, or failure to complete at least one postoperative outpatient visit).
(5) Intraoperative conversion: patients requiring intraoperative conversion to conventional CABG or emergency PCI due to technical failure.
(6) Malignancy: active malignancy or life expectancy
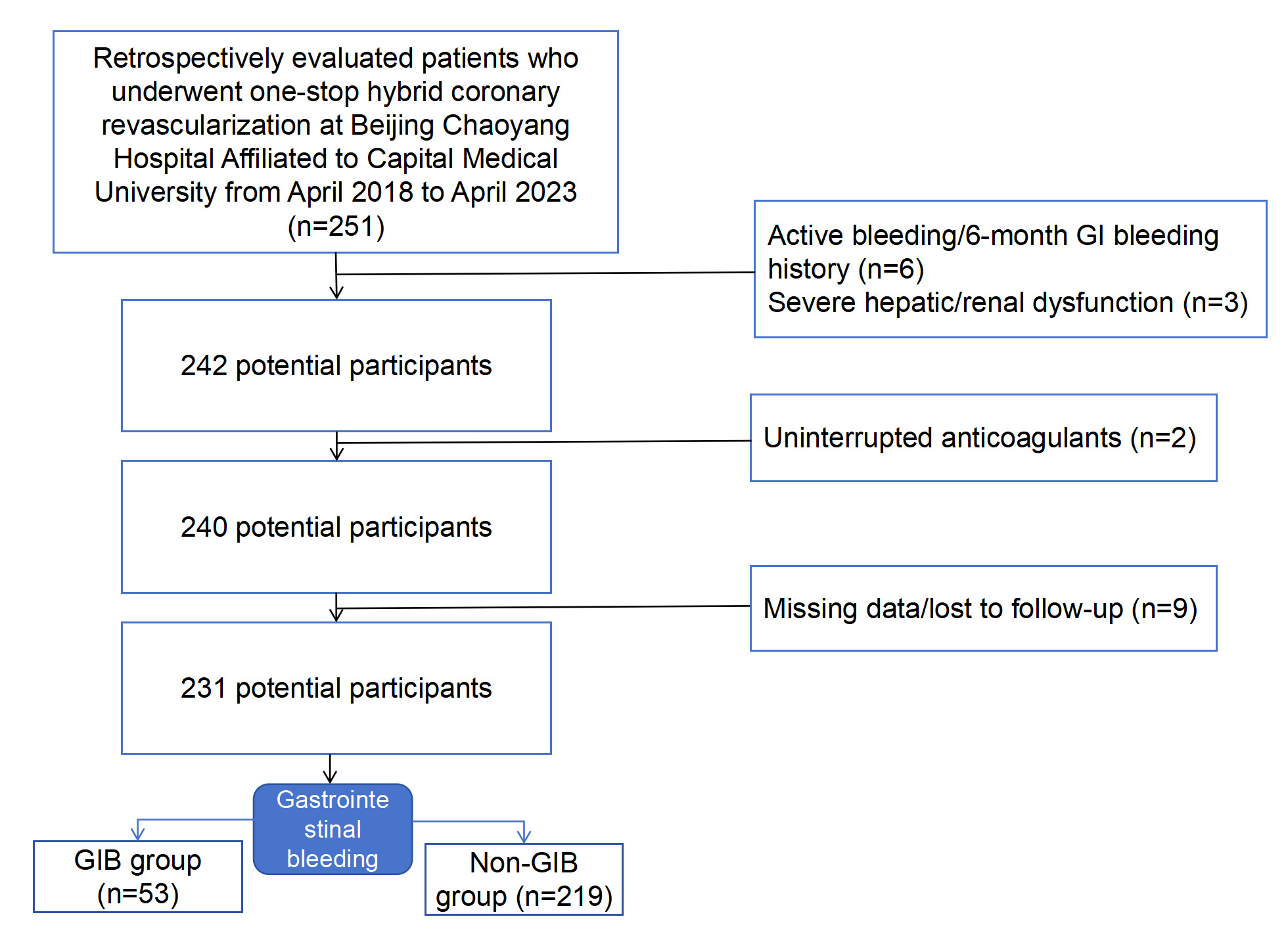 Fig. 1.
Fig. 1.
Inclusion and exclusion criteria flowchart. GIB, gastrointestinal bleeding.
(7) High baseline gastrointestinal (GI)-bleeding risk: During screening, 9
patients who met one or more of the above exclusion criteria (mainly criterion 1,
and in some cases criteria 2 and/or 3) were excluded because recent or active GI
bleeding, severe renal dysfunction (eGFR
In HCR surgery, minimally invasive direct CABG (MIDCABG) and PCI are performed
simultaneously in a hybrid operating room. Generally, MIDCABG is performed first,
followed by PCI. To maintain the integrity of the sternum, the left internal
mammary artery (LIMA) is harvested under direct vision through a small 5–7 cm
incision on the anterolateral side of the fourth and fifth intercostal spaces,
with the heart kept beating at normal temperature. The LIMA–LAD anastomosis is
completed. After the anastomosis is completed, transit-time flow measurement
(TTFM) is used to evaluate the graft flow and anastomosis quality [7]. PCI is
performed on other non-anterior descending diseased vessels. A drug-eluting stent
(DES) is generally selected. In selected cases, a drug-coated balloon (DCB)
strategy may be considered after adequate lesion preparation, provided the final
angiographic result shows Thrombolysis in Myocardial Infarction (TIMI) 3 flow, no
flow-limiting dissection, and
From 2018 to 2023, the detailed perioperative antithrombotic therapies with
potential impacts on bleeding risk were as follows: (1) Preoperative medication:
All patients continued oral aspirin (100 mg/day) until the morning of surgery to
maintain baseline antiplatelet activity. Clopidogrel was discontinued 5–7 days
preoperatively to reduce intraoperative bleeding risk while avoiding excessive
attenuation of antiplatelet effects; (2) intraoperative anticoagulants and
antiplatelet agents: During surgery, unfractionated heparin was administered at a
dose of 100–120 IU/kg body weight to maintain an activated coagulation time
(ACT)
Perioperative baseline variables are shown in Table 1. Preoperative, intraoperative, and postoperative parameters included age, gender, body mass index (BMI), smoking and drinking history, hypertension, diabetes history, hyperlipidemia history, peripheral vascular disease, previous cerebrovascular disease, previous myocardial infarction (MI), previous PCI, left main disease, preoperative left ventricular ejection fraction (LVEF), left ventricular end-diastolic diameter (LVED), history of bleeding disorders, any previous digestive tract disease, history of malignant tumors, number of diseased coronary arteries, preoperative D-dimer, preoperative hemoglobin, preoperative platelet count, preoperative Cr, total perioperative blood transfusion, intraoperative heparin dosage, length of hospital stay, mechanical ventilation time, ICU length of stay, drainage volume within 24 hours after surgery, total postoperative drainage volume, mortality, postoperative MI, and repeat revascularization. The above data were obtained from the electronic medical records of Chaoyang Hospital by designated personnel who were not involved in this study to minimize bias and ensure data authenticity and reliability.
| Variables | All patients (n = 231) | GIB group (n = 12) | Non-GIB group (n = 219) | p-value |
| Age (years) | 65.0 |
68.8 |
64.8 |
0.181 |
| Male | 163 (70.6%) | 10 (83.3%) | 153 (69.9%) | 0.502 |
| BMI (kg/m2) | 25.6 |
24.5 |
25.6 |
0.220 |
| Previous smoking | 117 (50.6%) | 8 (66.7%) | 109 (49.8%) | 0.254 |
| Previous alcohol drinking | 80 (34.8%) | 6 (50.0%) | 74 (33.9%) | 0.409 |
| Hypertension | 167 (72.3%) | 9 (75.0%) | 158 (72.1%) | 1.000 |
| Diabetes mellitus | 142 (61.5%) | 7 (58.3%) | 135 (61.6%) | 1.000 |
| Hyperlipidemia | 91 (39.4%) | 5 (41.7%) | 86 (39.3%) | 1.000 |
| CRD | 10 (4.3%) | 1 (8.3%) | 9 (4.1%) | 0.420 |
| PVD | 18 (7.8%) | 2 (16.7%) | 16 (7.3%) | 0.238 |
| CVD | 52 (22.5%) | 6 (50.0%) | 46 (21.0%) | 0.047 |
| Previous MI | 48 (20.8%) | 5 (41.7%) | 43 (19.6%) | 0.143 |
| Previous PCI | 62 (26.8%) | 4 (33.3%) | 58 (26.5%) | 0.852 |
| Left main disease | 89 (38.5%) | 6 (50.0%) | 83 (37.9%) | 0.593 |
| LVEF (%) | 65.0 (60.0, 70.0) | 65.0 (56.8, 69.3) | 65.0 (60.0, 70.0) | 0.782 |
| LVED | 48.1 |
47.5 |
48.1 |
0.731 |
| Bleeding events | 5 (2.2%) | 0 (0.0%) | 5 (2.3%) | 1.000 |
| Any GID | 50 (21.7%) | 4 (33.3%) | 46 (21.0%) | 0.522 |
| Malignant | 9 (3.9%) | 2 (16.7%) | 7 (3.2%) | 0.073 |
| Diseased branches | 3 (3, 3) | 3 (3, 3) | 3 (3, 3) | 0.657 |
| Preoperative D-dimer | 0.29 (0.19, 0.51) | 0.30 (0.14, 0.83) | 0.29 (0.19, 0.51) | 0.748 |
| Preoperative HGB | 133.7 |
132.5 |
133.8 |
0.820 |
| Preoperative PLT | 204 (172, 241) | 176 (157, 215) | 204 (174, 242) | 0.082 |
| Preoperative Cr | 68.5 (58.6, 81.5) | 78.5 (76.1, 95.0) | 67.7 (58.4, 81.2) | 0.011 |
| Transfusion | 103 (44.6%) | 7 (58.3%) | 96 (43.8%) | 0.079 |
| Total transfusion (mL) | 0 (0, 400) | 600 (50, 1350) | 0 (0, 400) | 0.009 |
| Transfusion |
23 (10.0%) | 4 (33.3%) | 19 (8.7%) | 0.022 |
| Heparin dosage | 120 (100, 150) | 130 (115, 150) | 120 (100, 150) | 0.569 |
| Bleeding volume (mL) | 200 (200, 350) | 400 (300, 650) | 200 (200, 300) | 0.001 |
| Operation time (min) | 300 (255, 330) | 300 (240, 390) | 300 (263, 330) | 0.421 |
| Reoperation for bleeding | 7 (3.0%) | 1 (8.3%) | 6 (2.7%) | 0.315 |
| Hospital days (d) | 20.0 (16.0, 24.0) | 20.5 (15.0, 27.3) | 20.0 (17.0, 24.0) | 0.666 |
| Ventilator time (h) | 16.0 (15.0, 18.0) | 18.0 (17.0, 37.5) | 16.0 (15.0, 17.0) | 0.012 |
| ICU days (d) | 4.0 (2.0, 6.0) | 4.0 (3.3, 6.8) | 3.0 (2.0, 6.0) | 0.243 |
| Drainage, 24 h | 845.0 (625, 1090) | 980.0 (871, 1218) | 825.0 (610, 1070) | 0.095 |
| Total drainage (mL) | 1170 (805, 1540) | 1453 (1363, 1565) | 1160 (800, 1535) | 0.028 |
| Mortality | 10 (4.3%) | 3 (25.0%) | 7 (3.2%) | 0.011 |
| Postoperative MI | 10 (4.3%) | 2 (16.7%) | 8 (3.7%) | 0.088 |
| Repeat revascularization | 19 (8.2%) | 1 (8.3%) | 18 (8.2%) | 1.000 |
Note: Continuous and normally distributed variables between the two groups were analyzed using an independent-samples t-test. A signed-rank test was applied to such data with a non-normal distribution. GIB, gastrointestinal bleeding; CRD, chronic respiratory disease; PVD, peripheral vascular disease; CVD, cerebrovascular disease; LVEF, left ventricular ejection fraction; LVED, left ventricular end diastolic diameter; GID, gastrointestinal disease; HGB, hemoglobin; PLT, platelets; ICU, intensive care unit; MI, myocardial infarction; Cr, creatinine; PCI, percutaneous coronary intervention; BMI, body mass index.
Continuous variables are expressed as the mean
A total of 231 patients who underwent HCR were included in this study. The mean
age was 65.0
All clinical factors related to gastrointestinal bleeding were included in a
binary logistic regression analysis. Several statistically significant risk
factors were identified in the univariate analysis, including prior
cerebrovascular disease, ICU length of stay, total operation time, preoperative
D-dimer level, perioperative blood transfusion
| Variables | Regression coefficient | SEM | Wald | OR | 95% CI | p-value |
| CVD | 1.341 | 0.560 | 5.730 | 3.822 | 1.275–11.458 | 0.017 |
| Preoperative Cr | 0.000 | 0.017 | 0.000 | 1.000 | 0.968–1.033 | 0.986 |
| Ventilator time (h) | 0.001 | 0.002 | 0.111 | 1.001 | 0.997–1.005 | 0.739 |
| ICU length of stay (d) | 0.061 | 0.025 | 5.819 | 1.062 | 1.011–1.116 | 0.016 |
| Operation time | 0.011 | 0.005 | 4.594 | 1.011 | 1.001–1.021 | 0.032 |
| Pre-D-dimer | 0.502 | 0.253 | 3.922 | 1.652 | 1.005–2.715 | 0.048 |
| Transfusion |
1.402 | 0.639 | 4.820 | 4.063 | 1.162–14.205 | 0.028 |
| Bleeding volume | 0.002 | 0.001 | 6.000 | 1.002 | 1.000–1.004 | 0.014 |
| Total drainage | 0.001 | 0.000 | 4.752 | 1.001 | 1.000–1.001 | 0.029 |
| Total transfusion | 0.001 | 0.000 | 7.042 | 1.001 | 1.000–1.002 | 0.008 |
Note: SEM, standard error of the mean; CVD, cerebrovascular disease; OR, odds ratio; CI, confidence interval; ICU, intensive care unit; Cr, creatinine.
| Variables | Regression coefficient | SEM | Wald | OR | 95% CI | p-value |
| CVD | 1.323 | 0.581 | 5.185 | 3.754 | 1.202–11.724 | 0.023 |
| Total drainage | 0.001 | 0.000 | 5.714 | 1.001 | 1.000–1.001 | 0.017 |
Note: SEM, standard error of the mean; CVD, cerebrovascular disease; OR, odds ratio; CI, confidence interval.
Gastrointestinal bleeding is defined as a reduction in hemoglobin of at least 2
g/dL, accompanied by hematemesis or black stool, and is diagnosed accordingly
[9]. In this study, the incidence of gastrointestinal bleeding in patients
undergoing HCR was 5.2%, and the mortality rate among those who exhibited
gastrointestinal bleeding was 25.0%. There was a significant difference in
mortality between patients with and without gastrointestinal bleeding (25.0% vs.
3.2%; p = 0.011). Previous studies have reported an incidence of
gastrointestinal bleeding after CABG of 1.1% [10]. Meanwhile, the incidence of
gastrointestinal bleeding in patients treated with PCI was 0.6%. Compared with
patients without gastrointestinal bleeding, the mortality rate was higher in
those with gastrointestinal bleeding (9.7% vs. 1.1%; p = 0.001), as
well as a longer median hospital stay (5.8 vs. 1.6 days) [11]. The incidence and
mortality of gastrointestinal bleeding in this study were higher than those
previously reported after CABG and PCI, potentially because HCR differs from
traditional surgery; thus, HCR patients will be administered oral aspirin before
the operation. Furthermore, heparin will be used on the day of operation to
maintain an ACT
Gastrointestinal bleeding after cardiac surgery is associated with increased mortality, prolonged ventilator use, pneumonia, wound infection, sepsis, acute renal injury, atrial fibrillation, MI, ICU treatment time, and prolonged hospital stay [12]. Previous studies have suggested several factors that increase the risk of bleeding during DAPT treatment, including a history of bleeding, oral anticoagulants, women, advanced age, low body weight, chronic renal insufficiency, diabetes, anemia, and long-term use of steroids or nonsteroidal anti-inflammatory drugs (NSAIDs) [11, 13, 14]. Intraoperative blood loss and postoperative total drainage may lead to gastrointestinal perfusion deficiency, vasoconstrictive catecholamine release, weakening of the mucus–bicarbonate barrier, gastric acid emptying, and other factors, such as gastrointestinal mucosal damage and gastrointestinal ulcers, which may lead to gastrointestinal bleeding in the later stage [15]. After multivariate analysis, prior cerebrovascular disease and total drainage were established as independent predictors of postoperative gastrointestinal hemorrhage in patients undergoing HCR. Cerebrovascular diseases included hemorrhagic stroke and ischemic stroke. In this study, 6 of 12 patients with gastrointestinal hemorrhage had a history of cerebral infarction and had been administering antiplatelet drugs for a long time. Therefore, it is considered that patients with previous cerebrovascular diseases may increase the risk of postoperative gastrointestinal bleeding due to long-term use of antiplatelet drugs before the operation. Indeed, long-term antiplatelet therapy (e.g., aspirin) may impair gastrointestinal mucosal repair and increase the risk of bleeding by disrupting the mucus–bicarbonate barrier and inhibiting platelet aggregation [16]. However, this mechanism must be considered alongside alternative explanations, such as generalized atherosclerosis, vascular frailty, and unmeasured confounders. Meanwhile, increased total drainage can also lead to a higher risk of postoperative gastrointestinal hemorrhage, primarily by inducing perioperative hypovolemia, which exacerbates gastrointestinal hypoperfusion, impairs mucosal barrier function, and promotes stress ulcer formation.
The results of this study are similar to those of previous studies, which have focused more on preoperative baseline data [17]. In contrast, this study includes many factors, such as intraoperative anticoagulant use and operation time, perioperative blood transfusion and postoperative drainage, and postoperative ICU length of stay. The results show that patients with previous cerebrovascular diseases have a relatively high risk of gastrointestinal bleeding after HCR. For these patients, attention should be paid to adjusting antiplatelet and prevention strategies to reduce the risk of gastrointestinal bleeding after the operation and discharge. Furthermore, caution is required in interpreting these findings. The wide CIs of the OR and the limitations of this study (small sample size, low event rate, unmeasured confounding factors) mean that the observed association cannot be definitively attributed solely to long-term antiplatelet therapy. Instead, these findings may reflect a combination of direct (mucosal damage caused by antiplatelet drugs) and indirect (systemic vascular dysfunction, unmeasured confounding factors) effects.
The use of dual antiplatelet drugs after coronary revascularization can reduce mortality and the incidence of ischemic events, but also increase the corresponding risk of bleeding [18]. DAPT generally uses aspirin and clopidogrel. Previous studies and guidelines recommend that aspirin be continued indefinitely after CABG, and that in patients with acute coronary syndrome (ACS) who are treated with DAPT and subsequently undergo CABG, P2Y12 inhibitor therapy be resumed after surgery to complete 12 months of DAPT following the ACS event. DAPT has been shown to reduce the risk of in-stent thrombosis by 0.4%, MI by 1.1%, and moderate-to-severe bleeding by 1.2% [13]. Since HCR combines CABG and PCI, balancing anticoagulation and bleeding risk is a major challenge for both surgeons and anesthesiologists.
After coronary revascularization, the generally recommended antiplatelet
strategy is DAPT, which can significantly reduce mortality and MACCEs. Common
DAPT regimens include aspirin plus ticagrelor or clopidogrel. Aspirin plus
ticagrelor is the best combination for reducing bridge restenosis, mortality, and
MACCEs, but this combination is associated with increased risk of bleeding during
use [19]. Aspirin directly acts on the phospholipid layer of gastric mucosa,
destroys the hydrophobic protective barrier of gastric mucosa [20], promotes the
release of cytotoxic substances such as leukotrienes, resulting in direct mucosal
loss; meanwhile, aspirin also reduces prostaglandin synthesis, gastric mucosal
blood flow, and mucosal protection by inhibiting mucosal cyclooxygenase (COX)-1
and COX-2 activity [21]. Clopidogrel noncompetitively inhibits adenosine
diphosphate (ADP) receptors and platelet aggregation. However, it has been
reported to delay gastrointestinal mucosal healing, which may contribute to an
increased risk of gastrointestinal bleeding [22]. The AUGUSTUS trials have shown
that aspirin use for 30 days or longer can seriously increase the risk of
bleeding. After 30 days, continued aspirin therapy further increases the risk of
bleeding without reducing the risk of ischemic events [23]. Almost all patients
receiving antiplatelet therapy have varying degrees of gastrointestinal injury.
The bleeding risk of aspirin or clopidogrel alone was lower than that of aspirin
combined with clopidogrel (0.6% vs. 5.4%; p
Among the patients undergoing HCR surgery, those with a history of cerebrovascular diseases exhibited a higher risk of postoperative gastrointestinal bleeding. For these patients, antithrombotic strategies should be adjusted, and appropriate preventive measures should be implemented to reduce the risk of gastrointestinal bleeding.
Most previous studies have focused on CABG and PCI; to our knowledge, there are currently no relevant studies on gastrointestinal bleeding after HCR. This study, as the first report analyzing risk factors for gastrointestinal bleeding after HCR, provides new insights and lays the foundation for future preventive strategies. However, this study has certain limitations. First, the critically low number of gastrointestinal bleeding events (n = 12) leads to insufficient statistical power for definitive analysis. The event-per-variable (EPV) ratio in the multivariable logistic regression model is only ~6, which is below the commonly recommended threshold of 10 EPVs. This may lead to model instability, potential overfitting, overestimated odds ratios, and wide confidence intervals, rendering the findings preliminary and unreliable for definitive conclusions. Second, this was a single-center retrospective study with a small overall sample size (n = 231). Moreover, patient selection and perioperative management may be specific to our institution, limiting the generalizability of the results. Third, due to the small number of events and sample size, we were unable to conduct subgroup analyses (e.g., differentiating types of cerebrovascular disease) or adjust for all potential confounders (e.g., severity of comorbidities, non-steroidal anti-inflammatory drug use, or gastrointestinal mucosal status). Additionally, the analysis of factors such as gender, age, and smoking history was inconsistent with previous studies, possibly due to insufficient statistical power required to detect true associations.
ACT, Activated Clotting Time; CABG, Coronary Artery Bypass Grafting; CI, Confidence Interval; CRD, Chronic Respiratory Disease; CVD, Cerebrovascular Disease; DAPT, Dual Antiplatelet Therapy; DCB, Drug-Coated Balloon; DES, Drug-Eluting Stent; eGFR, estimated Glomerular Filtration Rate; GIB, Gastrointestinal Bleeding; HCR, Hybrid Coronary Revascularization; HGB, Hemoglobin; ICU, Intensive Care Unit; LAD, Left Anterior Descending Artery; LIMA, Left Internal Mammary Artery; LMCA, Left Main Coronary Artery; LVED, Left Ventricular End-Diastolic Diameter; LVEF, Left Ventricular Ejection Fraction; MACCEs, Major Adverse Cardiovascular and Cerebrovascular Events; MIDCABG, Minimally Invasive Direct Coronary Artery Bypass Grafting; OR, Odds Ratio; PCI, Percutaneous coronary intervention; PLT, Platelet Count; PPI, Proton Pump Inhibitor; PVD, Peripheral Vascular Disease; SD, Standard Deviation; SEM, Standard Error of the Mean; TTFM, Transit-Time Flow Measurement.
The datasets used and analysed during the current study were available from the corresponding author on reasonable request.
PS designed the study, secured funding, and oversaw the implementation of the project. ZF and YX managed the data. ZF collected demographic characteristics and wrote the initial draft, YX and PS revised it. All authors have accessed and verified the data and collectively decided to submit the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Ethics Committee of the Beijing Chao-Yang Hospital, Capital Medical University (Approval No.: 2023-673). All methods were carried out in accordance with Declaration of Helsinki, and all of the participants provided signed informed consent.
Not applicable.
This work was supported financially by National clinical key specialty construction project Cardiovascular Surgery.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

