1 Department of Cardiothoracic Surgery, University of Pittsburgh, Pittsburgh, PA 15213, USA
2 UPMC Heart and Vascular Institute, University of Pittsburgh Medical Center, Pittsburgh, PA 15213, USA
3 Division of Cardiology, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA
4 Heart, Vascular & Thoracic Institute, Cleveland Clinic Florida, Weston, FL 33331, USA
Abstract
Low-flow, low-gradient (LFLG) aortic stenosis (AS) is associated with high-risk physiology and poor outcomes; however, the optimal intervention strategy, surgical aortic valve replacement (SAVR) versus transcatheter aortic valve replacement (TAVR), remains uncertain.
A systematic review and pooled meta-analysis of studies reporting Kaplan-Meier outcomes after SAVR and TAVR in patients with LFLG AS, published through June 2025, was performed. Individual patient data were reconstructed from published curves. Analyses included Cox frailty models, restricted mean survival time (RMST), and two-stage meta-analyses. Adjusted analyses accounted for baseline differences, including age, comorbidities, and echocardiographic parameters.
Five studies comprising 1940 patients (721 SAVR and 1219 TAVR) were included. Patients undergoing TAVR were older and had higher rates of peripheral arterial disease, while other comorbidities and echocardiographic parameters were similar between groups. In unadjusted analyses, TAVR was associated with higher mortality compared with SAVR (hazard ratio (HR) 1.36, 95% confidence interval (CI) 1.11–1.65; p = 0.003). The estimated 5-year survival was 67% for SAVR versus 49% for TAVR, with an RMST of 0.64 years in favor of SAVR. Despite risk adjustment, TAVR was associated with higher mortality (HR 1.42, 95% CI 1.17–1.73), with a reduction in RMST of 1.06 years. Two-stage meta-analyses yielded consistent findings with low-to-moderate heterogeneity.
SAVR was associated with significantly lower mortality in patients with LFLG AS than TAVR, both before and after risk adjustment. While these findings suggest a potential survival benefit, the data are observational and hypothesis-generating, and residual confounding cannot be excluded.
Keywords
- aortic stenosis
- surgical aortic valve replacement
- transcatheter aortic valve replacement
Patients with severe aortic stenosis (AS) may present with a low transvalvular pressure gradient despite possessing a severely reduced aortic valve area (AVA), a condition termed low-flow, low-gradient (LFLG) AS [1]. This condition can occur with reduced left ventricular ejection fraction (LVEF; classical LFLG) or preserved ejection fraction (paradoxical LFLG), each reflecting distinct underlying pathophysiologies [2]. Classical LFLG typically results from impaired systolic function due to afterload mismatch or concomitant ischemic heart disease, whereas paradoxical LFLG arises from impaired diastolic filling, ventricular remodeling, or other comorbidities. In addition, patients commonly present with concomitant atrial fibrillation and mitral regurgitation, which contribute to low stroke volume index.
Gradient alone is insufficient to capture disease severity, as low flow can mask the hemodynamic burden of severe stenosis [3, 4, 5]. Consequently, stroke volume, flow status, and valve area provide complementary information that is critical for clinical decision-making [6, 7]. Current guidelines recommend aortic valve replacement (AVR) for symptomatic patients with severe LFLG AS; however, the optimal choice of intervention, surgical AVR (SAVR) versus transcatheter AVR (TAVR), remains uncertain [2, 8]. TAVR offers a less invasive alternative with potential hemodynamic advantages; meanwhile, SAVR provides durable outcomes, lower rates of paravalvular leak and conduction disturbances, and facilitates concomitant procedures such as coronary revascularization [9, 10].
Nonetheless, evidence directly comparing SAVR and TAVR in LFLG AS remains limited, as most available studies are often retrospective, small, or heterogeneous in patient characteristics [11]. Therefore, the impact of procedural choice on long-term survival in this high-risk population remains unclear. To address this gap, this study aimed to pool available data comparing mortality after TAVR versus SAVR in patients with LFLG AS to provide evidence for guiding individualized treatment decisions.
This systematic review and meta-analysis were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [12]. Ethical approval was not required for this study-level meta-analysis.
A structured literature search was performed in the MEDLINE, Embase, and Web of Science databases to identify studies published through June 1st, 2025. The search was guided by the PICO framework: Population: patients with low-flow, low-gradient aortic stenosis; Intervention: transcatheter aortic valve replacement (TAVR); Comparison: surgical aortic valve replacement (SAVR); Outcomes: all-cause mortality. Key search terms included “low gradient”, “paradoxical low flow low gradient”, “classical low flow low gradient”, “normal flow low gradient”, and “aortic stenosis”, combined using Boolean operators and tailored to each database. Moreover, reference lists of included studies and relevant reviews were also screened to ensure completeness. Two reviewers (X.J., M.P.S.) independently screened titles, abstracts, and full texts according to predefined eligibility criteria. Studies were included if the analysis enrolled patients with LFLG AS undergoing TAVR or SAVR and reported relevant outcomes with extractable data. Exclusion criteria included reviews, case reports, single-arm studies, and studies with patients without LFLG AS. When multiple reports from overlapping populations were identified, the most recent or most comprehensive study was retained.
Two reviewers (X.J., M.P.S.) independently extracted patient characteristics,
and Kaplan-Meier curves from each study were digitized using Web Plot Digitizer
software; individual patient data (IPD) were reconstructed from these plots,
including the total number of patients, total events, and the number at risk at
each time point for each treatment group [13]. The methodological quality and
risk of bias were also independently evaluated by the same reviewers using the
Cochrane Risk of Bias tool. Time-to-event outcomes were analyzed using a Cox
frailty model with robust standard errors to estimate hazard ratios (HRs) and
95% confidence intervals (CIs). Between-study heterogeneity was accounted for by
including a
Our initial search yielded 1146 unique citations, of which 10 publications were potentially relevant (Fig. 1). From these, five studies were included, encompassing 1940 patients, with 721 undergoing SAVR and 1219 undergoing TAVR (Table 1) [11, 14, 15, 16, 17]. Study periods ranged from 2002 to 2023. The included studies were conducted across multiple countries, including Austria, Canada, Switzerland, the United States, the United Kingdom, and multinational cohorts. Study designs were primarily retrospective observational analyses; one study reported a post hoc analysis of the Placement of Aortic Transcatheter Valves (PARTNER) 2A randomized trial and the PARTNER 2 SAPIEN 3 intermediate- and high-risk registry. All studies employed statistical adjustment methods to account for baseline differences, including multivariable regression, inverse probability of treatment weighting (IPTW), and propensity score weighting. Fig. 2 presents the qualitative assessment of the included studies using the ROBINS-I (Risk Of Bias In Non-randomized Studies of Interventions) tool, highlighting most concerns due to residual confounding, which was moderate in most studies. Overall, however, the risk of bias was judged to be low.
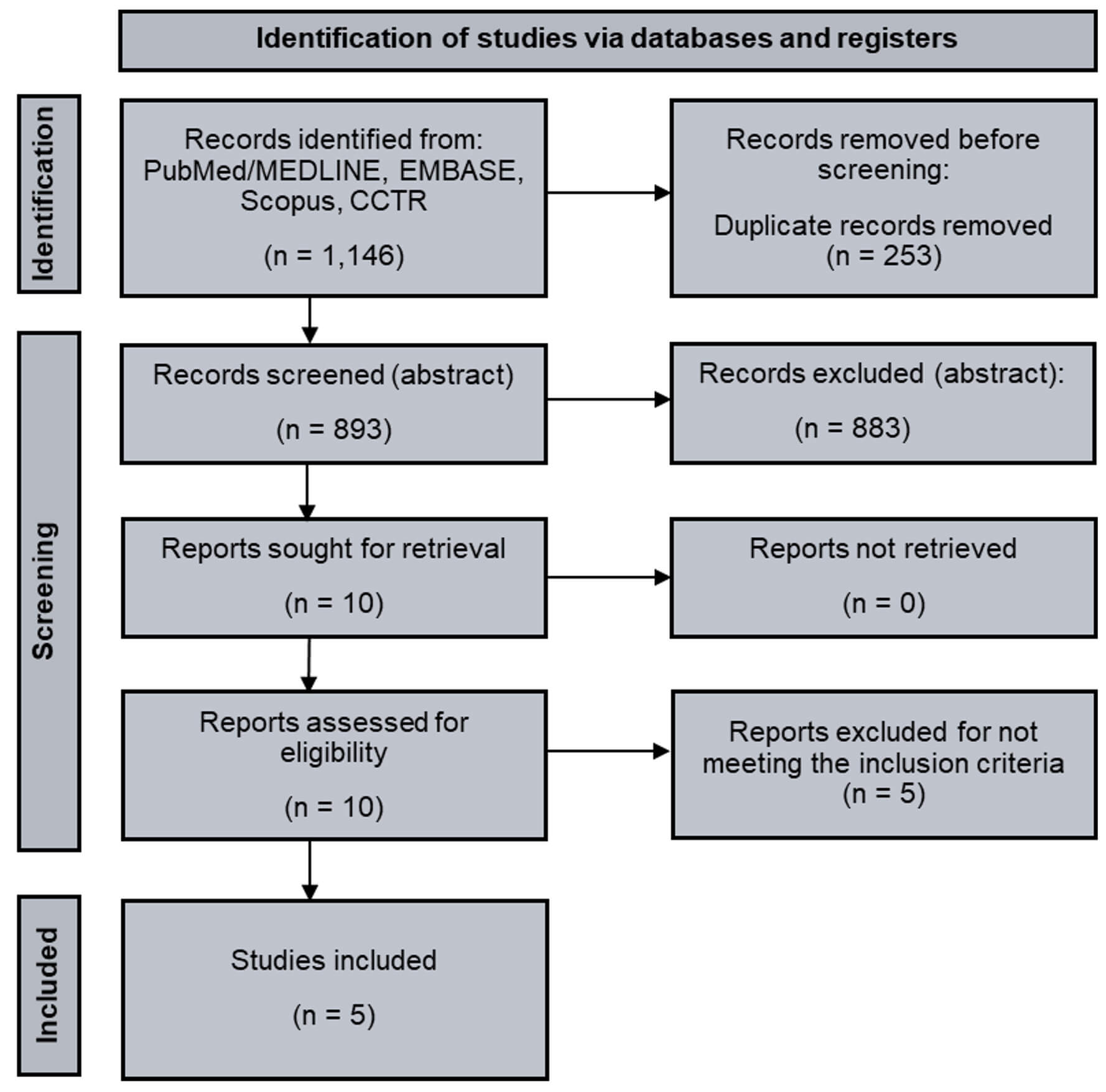 Fig. 1.
Fig. 1.
Study flowchart. Legend: PRISMA flow diagram of included studies. Abbreviations: CCTR, Cochrane Controlled Trials Register; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
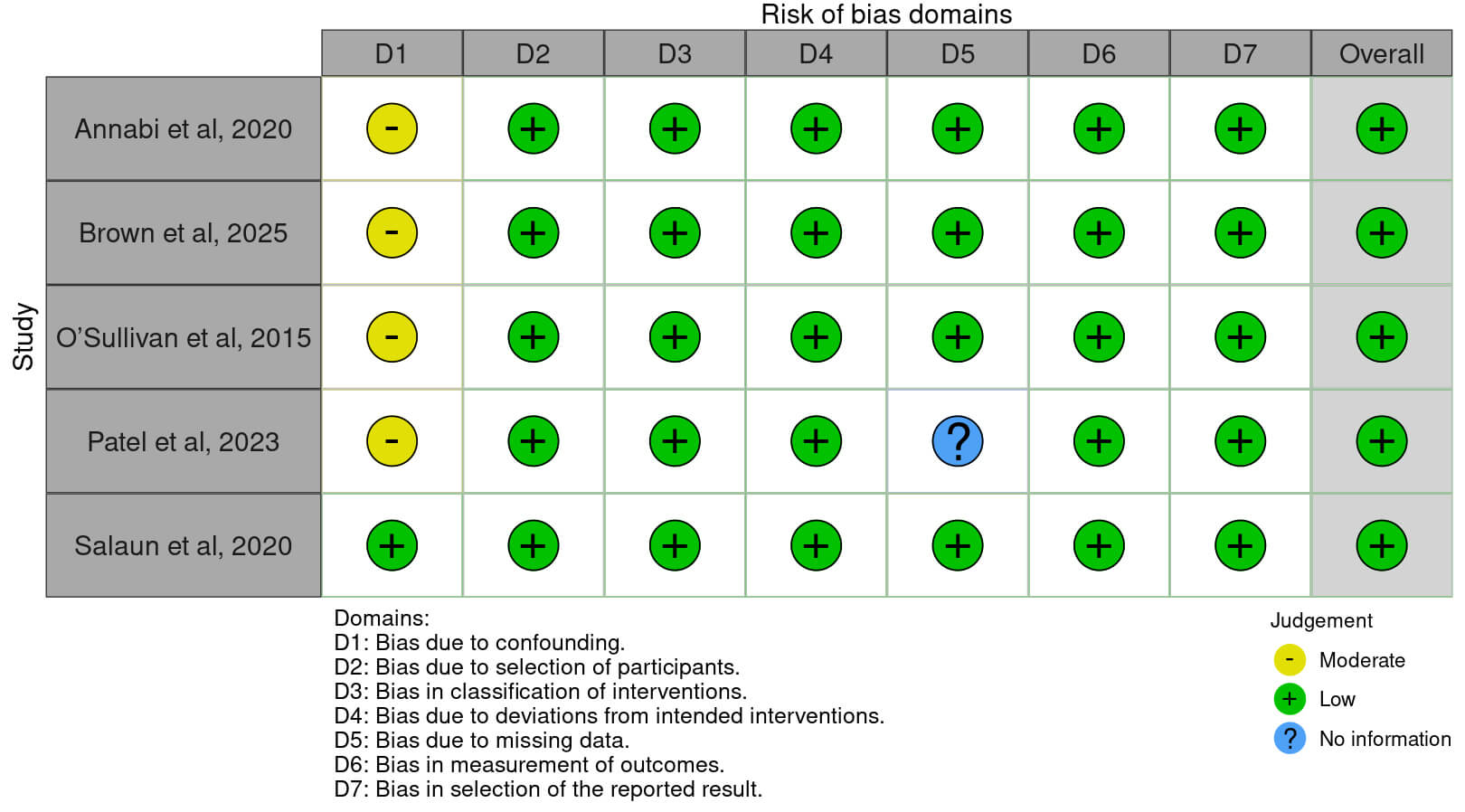 Fig. 2.
Fig. 2.
Risk of bias assessment for the included studies. Legend: Risk of bias was evaluated with the ROBINS-I (Risk Of Bias In Non-randomized Studies of Interventions) tool.
| Study | Country | Study design | Period | Total sample, n | SAVR, n | TAVR, n | Adjustment for confounders |
| Annabi et al., 2020 [17] | Austria and Canada | Retrospective from Prospective Registry | 2002–2016 | 274 | 176 | 98 | Multivariable and IPTW |
| Brown et al., 2025 [15] | United States | Retrospective observational | 2010–2023 | 248 | 131 | 117 | Multivariable adjustment |
| O’Sullivan et al., 2015 [11] | Switzerland | Retrospective observational | 2005–2012 | 160 | 52 | 108 | Multivariable adjustment |
| Patel et al., 2023 [14] | United Kingdom | Retrospective observational | 2015–2020 | 322 | 102 | 220 | Propensity score weighting |
| Salaun et al., 2020 [16] | Multiple | Post hoc RCT | 2012–2014* | 936 | 260 | 676 | Multivariable adjustment |
Legend: Data include study design, country, study period, total sample size, number of patients undergoing surgical aortic valve replacement (SAVR) or transcatheter aortic valve replacement (TAVR), and methods used to adjust for confounders. *For the PARTNER 2 Registry, data collection continued through 2017. Abbreviations: SAVR, surgical aortic valve replacement; TAVR, transcatheter aortic valve replacement; IPTW, inverse probability of treatment weighting; RCT, randomized controlled trial.
Baseline characteristics for the patients in the individual studies are reported
in Table 2 (Ref. [11, 14, 15, 16, 17]). Patients undergoing TAVR were generally older
than those undergoing SAVR, with mean ages ranging from 66 to 82.5 years (mean
difference (MD) +8.96 years, 95% CI +5.26 to +12.66; p
| Study | Age, years | Male, % | BMI, kg/m2 | HTN, % | DM, % | CAD, % | PAD, % | COPD, % | AVA, cm2 | MG, mmHg | Vmax, m/s | LVEF, % | Classical LFLG, % |
| Annabi et al., 2020 [17] | 71 |
127 (72)/64 (66) | 28.3 |
118 (67)/87 (90) | 61 (35)/36 (37) | 63 (36)/73 (75) | 4 (2)/18 (19) | 46 (27)/35 (38) | 0.76 |
28 |
3.5 |
35 [25–60]/30 [25–40] | 115 (65)/74 (76) |
| Brown et al., 2025 [15] | 66 [59–73]/79 [76–84] | 103 (78.6)/97 (82.9) | 31.1 [26.0–35.1]/28.4 [25.6–31.6] | 109 (83.2)/105 (89.7) | 43 (32.8)/39 (33.3) | 38 (29)/68 (58.1) | 10 (7.6)/27 (23.1) | NA | 0.8 [0.7–1.0]/0.7 [0.6–0.9] | 32 [27–36]/32 [27–36] | NA | 45 [33–58]/48 [35–60] | 81 (61.8)/59 (50.4) |
| O’Sullivan et al., 2015 [11] | 78.4 |
33 (63)/63 (58) | 26.4 |
43 (83)/87 (81) | 17 (33)/35 (32) | 40 (77)/80 (74) | 6 (12)/29 (27) | 7 (13)/17 (16) | 0.73 |
29.26 |
3.32 |
38.90 |
NA |
| Patel et al., 2023 [14] | 71 |
72 (71)/155 (71) | NA | 82 (80)/153 (70) | 33 (32)/62 (28) | 52 (51)/103 (47) | NA | NA | 0.8 [0.6–0.9]/0.7 [0.6–0.8] | 28 [22–36]/31 [24–35] | NA | NA | 102 (100)/220 (100) |
| Salaun et al., 2020* [16] | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA |
Legend: Reported parameters include demographic characteristics, comorbidities,
functional status, echocardiographic indices, and operative details. Continuous
variables are expressed as the mean
Four studies were included, comprising 618 patients who underwent SAVR and 998
who underwent TAVR, with a median follow-up of approximately 2 years (Q25–Q75:
1–3) and a total follow-up of 5 years. Patients treated with TAVR had a higher
risk of mortality than those treated with SAVR (HR 1.36, 95% CI 1.11–1.65;
p = 0.003). Estimated survival for SAVR patients was 82% (95% CI
79–85) at 1 year, 77% (95% CI 73–80) at 2 years, and 67% (95% CI 62–73) at
5 years, compared with 75% (95% CI 73–78), 64% (95% CI 61–67), and 49%
(95% CI 41–58), respectively, for TAVR patients (Fig. 3A). RMST analysis
indicated that TAVR was associated with a reduction in expected survival of 7.5
months compared with SAVR (RMST difference –0.64 years, 95% CI –0.88 to
–0.40; p
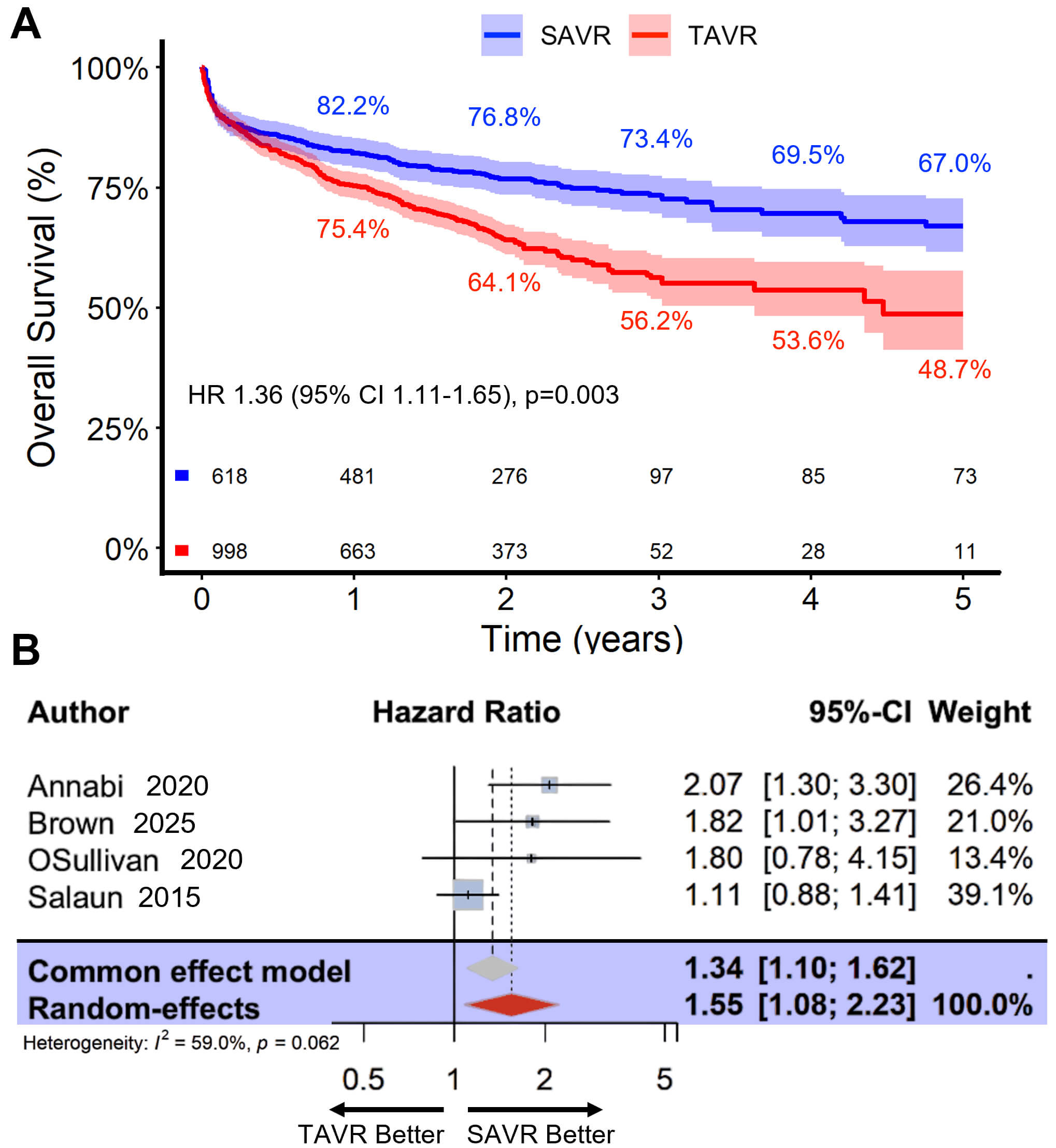 Fig. 3.
Fig. 3.
Comparative outcomes of TAVR versus SAVR in patients with LFLG AS. Legend: (A) Kaplan-Meier survival curves up to 5 years for patients who underwent TAVR versus SAVR, with survival estimates indicated at key time points. (B) Results of a two-stage meta-analysis summarizing the pooled effect of TAVR versus SAVR on long-term survival.
Four studies were included in the adjusted analysis, comprising 668 patients who
underwent SAVR and 641 who underwent TAVR, with a median follow-up of 1.89 years
(Q25–Q75: 0.17–2.37) and a total follow-up of 5 years. Even after adjustment
for baseline covariates, TAVR was associated with a significantly higher risk of
mortality compared with SAVR (HR 1.42, 95% CI 1.17–1.73; p
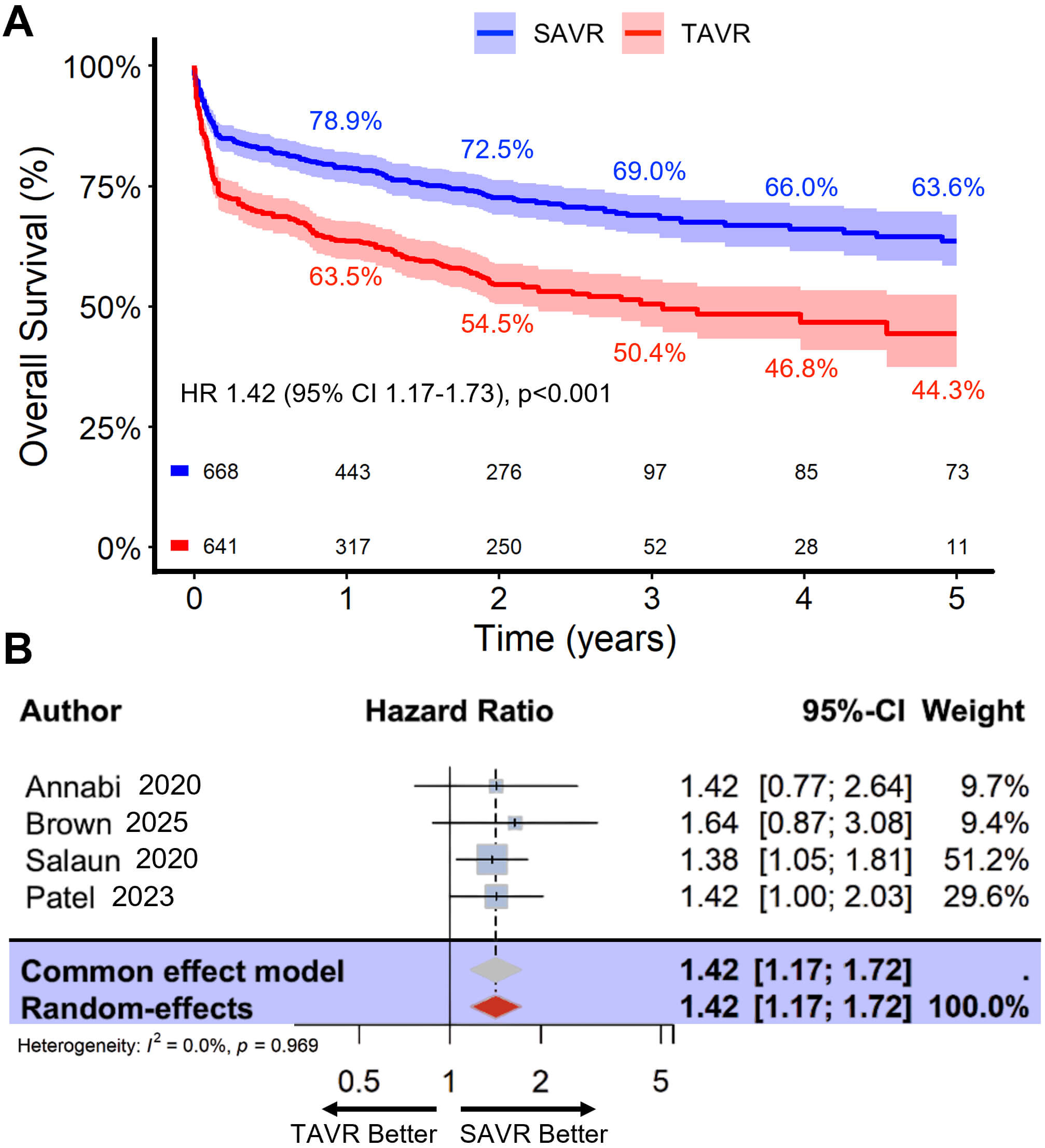 Fig. 4.
Fig. 4.
Adjusted comparative outcomes of TAVR versus SAVR in patients with LFLG AS. Legend: (A) Kaplan-Meier survival curves up to 5 years for patients who underwent TAVR versus SAVR, adjusted for confounders, with survival estimates shown at key time points. (B) Results of a two-stage meta-analysis summarizing the pooled, adjusted effect of TAVR versus SAVR on long-term survival.
In this meta-analysis of patients with LFLG AS, our findings suggest that TAVR was associated with significantly higher mortality than SAVR in both the unadjusted (HR 1.36, 95% CI 1.11–1.65) and adjusted analyses (HR 1.42, 95% CI 1.18–1.72). Survival at 1, 2, and 5 years was consistently higher among SAVR patients, and RMST analyses indicated an average loss of approximately one year of life expectancy over 5 years for those who underwent TAVR. However, these associations should be interpreted cautiously, given the observational nature of the included studies and the potential for residual confounding. Patients who underwent TAVR were generally older and had a higher prevalence of peripheral arterial disease, whereas most other comorbidities and echocardiographic parameters were similar. Overall, these results reflect associations rather than causal effects and highlight the need for individualized heart-team decision-making.
The importance of this work lies in recognizing that patients with LFLG AS constitute a distinct clinical entity compared with those with normal-flow, high-gradient AS. These patients have historically demonstrated poorer survival, even when LVEF is preserved. Importantly, this population is not rare, as LFLG AS accounts for approximately 5–10 percent of patients undergoing isolated, first-time AVR. Within this group, two subtypes are recognized: classical LFLG, characterized by reduced LVEF, and paradoxical LFLG, where LVEF is preserved. Classical LFLG is more common, whereas paradoxical LFLG is frequently associated with the worst outcomes among patients with AS. In addition to LVEF, low transvalvular flow has been identified as a strong predictor of outcomes following valve replacement. Although current guidelines recommend AVR for patients with severe symptomatic LFLG AS [2, 8], the optimal choice of intervention, SAVR or TAVR, remains an area of active investigation [18].
In a multicenter cohort of 204 patients with LFLG AS and concomitant coronary artery disease treated between 2005 and 2012, both SAVR and TAVR were associated with significantly improved survival compared with medical therapy (adjusted HR 0.16 and 0.30, respectively). While there were no significant differences in all-cause mortality between the two procedures, SAVR patients were more likely to undergo complete revascularization (35% vs. 8%). Notably, this finding is relevant, as patients with incomplete revascularization had more than a 2-fold increase in 1-year cardiovascular mortality [11]. In contrast, the PARTNER high-risk substudy found no statistically significant difference in mortality between TAVR and SAVR at 2 years among 105 patients with classical LFLG AS [16]. Furthermore, at the opposite end of the risk spectrum, the TOPAS registry reported significantly lower mortality with transfemoral TAVR compared with SAVR (HR 0.28, 95% CI 0.11–0.72) at 36 months [17]. Lastly, in a contemporary low-risk cohort, early outcomes, including 30-day mortality, stroke, and pacemaker rates, were comparable between SAVR and TAVR; however, paravalvular leak was more common with TAVR, and survival at 1 year was numerically higher with SAVR, consistent with trends observed in our pooled analysis [15]. In that study, the higher incidence of paravalvular leakage may partially explain these findings, as even mild leaks have been associated with adverse long-term outcomes following TAVR [19]. Another potential explanation for the observed differences in outcomes between TAVR and SAVR is the impact of patient-prosthesis mismatch (PPM), particularly in the LFLG population [20]. Although severe PPM is more frequent after SAVR, the impact of this condition is primarily observed in TAVR patients with low-flow physiology, in whom PPM is linked to higher cardiac mortality and rehospitalization [20, 21, 22]. Overall, these data suggest a possible survival benefit and more durable hemodynamic performance with SAVR in appropriately selected patients; however, the limited number of studies and risk of confounding prevent definitive conclusions. For example, smaller sample sizes in prior studies may have obscured a true survival benefit with SAVR. Conversely, up to 70% of the excess mortality observed with TAVR may reflect residual confounding from older age, higher comorbidity burden among TAVR recipients, differences in valve types, or an era effect [23]. This is particularly important because most studies were judged to have at least a moderate risk of bias in the domain of residual confounding, a key limitation for interpreting these pooled estimates.
Recent evidence further refines our understanding of factors influencing outcomes following TAVR in patients with LFLG AS. Observational studies have examined the impact of valve type, balloon-expandable versus self-expanding. While 30-day mortality was similar between groups, self-expanding valves were associated with lower mean gradients but higher rates of paravalvular leak. In contrast, balloon-expandable valves were associated with a reduced risk of heart failure readmission over a median follow-up of approximately 2.5 years [24]. The supra-annular design of many self-expanding valves contributes to lower post-TAVR gradients, especially in patients with small annuli or low-flow states, potentially improving left ventricular (LV) unloading [25, 26]. Similarly, annular size has been evaluated as a potential determinant of post-TAVR outcomes. In a cohort of 1866 patients, individuals with small annuli had higher mean gradients and a greater incidence of structural valve deterioration; however, intermediate-term mortality and heart failure rehospitalization were comparable to those with larger annuli across all flow-gradient subgroups, including classical and paradoxical LFLG AS [27]. Extending this to surgical practice, valve size has not been associated with either in-hospital or mid-term mortality, even for prostheses smaller than 21 mm [28]. TAVR valves typically achieve larger effective orifice areas and lower gradients at equivalent sizes, thereby potentially favoring hemodynamic effects on ventricular remodeling and early clinical outcomes. Sutureless surgical valves, which offer hemodynamics comparable to TAVR while maintaining lower rates of paravalvular leak, may represent an alternative strategy for selected patients with LFLG AS; however, prospective studies are needed to confirm these potential benefits [29, 30].
In patients with LFLG AS, recent advances in imaging have enabled computed tomography-derived aortic valve calcium (AVC) scoring to complement traditional functional assessments, particularly when dobutamine stress echocardiography (DSE) is inconclusive or not feasible [6, 31]. However, recent evidence shows that AVC has limited discriminatory power in this population, with low sensitivity (44.3%) and specificity (56.5%) for identifying true severe AS [32]. While DSE remains the reference standard, the use of this technique can be limited by arrhythmias, patient tolerance, and availability [33]. Thus, although AVC provides a noninvasive and reproducible measure of valve calcification, current Agatston score cutoffs cannot reliably replace DSE and should be applied with caution in clinical decision-making for these patients. Lastly, despite advances in both new-generation TAVR devices and SAVR techniques, clinical outcomes for patients with LFLG AS remain suboptimal [33]. These findings emphasize that AVR alone may not be sufficient to fully address the elevated risk in this vulnerable population [33]. Optimizing long-term outcomes will likely require a comprehensive, multidisciplinary strategy that combines valve replacement with intensive medical therapy, close management of comorbid conditions, and structured cardiac rehabilitation to enhance functional recovery and survival [7, 34].
This meta-analysis has several important limitations. Most of the included studies were retrospective observational cohorts; only one was a post hoc analysis of a randomized trial, in which flow status was not specifically accounted for. Therefore, residual confounding from unmeasured patient characteristics, procedural selection, and center-specific practices cannot be excluded despite statistical adjustments. Critical baseline variables, including frailty and surgical risk scores, were inconsistently reported across the included studies. These factors are particularly relevant, as patients undergoing TAVR in real-world practice are often at higher baseline risk than those receiving SAVR. Additionally, heterogeneity in LFLG subtypes (classical versus paradoxical), baseline comorbidities, and valve selection may have influenced outcomes. Given the important differences between LFLG subtypes, a formal subgroup analysis would be valuable. However, most studies did not report outcomes separately for classical and paradoxical LFLG AS, precluding such an analysis. Additionally, TAVR technology has evolved substantially over the study period, and earlier studies may not reflect contemporary practice, which could influence observed outcomes. Moreover, reconstruction of individual patient data from digitized Kaplan-Meier curves, although validated, may introduce minor inaccuracies relative to the original datasets. In addition, the use of varying adjustment methods across studies (IPTW, propensity scores, or multivariable regression) may have introduced further heterogeneity in the estimated effects. Finally, follow-up was limited to a maximum of 5 years, thereby limiting conclusions regarding long-term survival and valve durability. Despite these limitations, this analysis offers meaningful insights into the differential impact of TAVR versus SAVR in high-risk LFLG patients; meanwhile, prospective randomized controlled trials are awaited to provide more definitive evidence.
In patients with LFLG AS, SAVR was associated with significantly lower mortality than TAVR, both before and after adjustment for baseline characteristics. Across studies, TAVR patients experienced a reduction in expected survival of approximately 1 year over follow-up periods of up to 5 years. These findings represent observed associations rather than causal effects, and residual confounding cannot be excluded. Accordingly, these data are hypothesis-generating and should not be interpreted as definitive evidence of SAVR superiority, highlighting the need for careful procedural selection and individualized management in this high-risk population.
All data reported in this paper will be shared by the corresponding author upon request.
XJ, GR, and MPS contributed to the conception and design of the study, performed the research, and drafted the manuscript. IH, TO, DS-G, DK, CT, and AM contributed to data interpretation and critically revised the manuscript for important intellectual content. IS contributed to the conception and design of the study, contributed to data interpretation and critically revised the manuscript, and provided overall supervision. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT-4o in order to check spelling and grammar. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF47967.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




