1 Department of Anesthesiology and Reanimation, Istanbul Basaksehir Cam and Sakura City Hospital, 34480 Istanbul, Türkiye
2 Department of Anesthesiology and Reanimation, University of Health Science, Bursa Yüksek Ihtisas Training and Research Hospital, 16310 Bursa, Türkiye
3 Department of Anesthesiology and Reanimation, Istanbul Health Science University Kanuni Sultan Süleyman Education and Training Hospital, 34303 Istanbul, Türkiye
Abstract
Postoperative cognitive dysfunction (POCD) remains a common neurological complication after cardiac surgery, particularly among older patients. Intraoperative cerebral oxygen desaturation during cardiopulmonary bypass (CPB) may reflect impaired cerebral perfusion and is proposed as a contributing factor to postoperative cognitive impairment. This study aimed to evaluate the association between intraoperative cerebral oxygen desaturation and postoperative cognitive outcomes in patients undergoing cardiac CPB surgery.
In this prospective observational study, 74 patients undergoing elective cardiac surgery with CPB between March 2023 and March 2024 were included. Cognitive function was assessed using the Mini-Mental State Examination (MMSE) preoperatively and on postoperative days (PODs) 4, 7, and 30. Bilateral frontal regional cerebral oxygen saturation (rSO2) was continuously monitored intraoperatively using near-infrared spectroscopy (NIRS). Cerebral desaturation was defined as a reduction of >20% in rSO2 from baseline. A standardized intervention protocol was applied when this threshold was exceeded.
The incidence of POCD was 32.4% during the first postoperative week and decreased to 20.2% by POD 30. Patients who developed POCD were significantly older than those without POCD (median (interquartile range [IQR]): 63 [58–68] vs. 59 [52–65] years; p = 0.026). Intensive care unit (ICU) and total hospital stays were significantly longer in the POCD group (median ICU stay: 74 [51.3–96] vs. 50 [46–60] hours; p < 0.001; median hospital stay: 9 [8–15.5] vs. 7 [7–9] days; p < 0.001). Intraoperative rSO2 values were significantly lower in the POCD group following the initiation of CPB, particularly during the hypothermic and rewarming phases (T2–T4). Percentage decreases in rSO2 at T2, T3, and T4 showed significant positive correlations with MMSE scores on POD 4 (r = 0.283, p = 0.014; r = 0.312, p = 0.007; r = 0.312, p = 0.007, respectively) and POD 30 (r = 0.314, p = 0.006; r = 0.270, p = 0.020; r = 0.349, p = 0.002, respectively).
Intraoperative cerebral oxygen desaturation during CPB, particularly during the initiation, hypothermic, and rewarming phases, was associated with decreased early and late-postoperative cognitive performance after cardiac surgery. Continuous intraoperative cerebral oxygenation monitoring using NIRS may enable earlier identification of cerebral hypoperfusion and facilitate timely interventions in patients at increased risk of POCD.
NCT07361068. https://clinicaltrials.gov/search?term=NCT07361068.
Keywords
- cardiac surgery
- postoperative cognitive dysfunction
- neuropsychological test
- Mini-Mental State Examination
- near-infrared spectroscopy
- cardiopulmonary bypass
- cerebral oxygen saturation
Patients undergoing cardiac surgery are particularly prone to postoperative neurological complications due to aging and the increasing prevalence of comorbidities. In addition to existing cardiovascular diseases, age-related frailty, reduced adaptive mechanisms, intraoperative neuroinflammation, and heightened susceptibility to hypoxemia further contribute to this risk [1]. Postoperative cognitive dysfunction (POCD) has been reported to affect a substantial proportion of cardiac surgery patients and has been associated with prolonged hospitalization, reduced survival, increased healthcare costs, and decreased quality of life [2].
The pathophysiology of POCD in surgeries involving cardiopulmonary bypass (CPB) has not yet been fully elucidated. However, current evidence supports a multifactorial etiology in which surgery-related, anesthesia-related, and patient-related factors interact through interdependent mechanisms. Impaired cerebral autoregulation, neuronal injury secondary to hypoperfusion, and microembolic phenomena have been proposed as key contributors to postoperative cognitive impairment [3]. In addition, a recent study by Glumac et al. [4] demonstrated that the systemic inflammatory response triggered by surgical trauma plays a role in the development of POCD and that preoperative corticosteroid administration was associated with a reduced incidence and severity of POCD. Hemodynamic changes induced by CPB may further adversely affect the microcirculation, thereby contributing to a reduction in cerebral perfusion. Risk factors for POCD include preoperative variables such as age, hypertension, diabetes, and educational level, as well as intraoperative and postoperative factors, including the type of surgery, CPB, and aortic cross-clamp duration, and postoperative atrial fibrillation [5, 6]. In addition, Yan et al. [7] emphasized the critical role of cerebral hypoperfusion, neuroinflammatory processes, and intraoperative neuromonitoring strategies in the multifactorial etiology of POCD.
Near-infrared spectroscopy (NIRS) enables noninvasive measurement of cerebral oxygen saturation, allowing real-time monitoring of intraoperative hemodynamic changes and detection of clinically significant desaturation events. Regional cerebral oxygen saturation (rSO2) values obtained by NIRS reflect the balance between cerebral oxygen supply and demand [8]. Previous studies have demonstrated that intraoperative cerebral oxygen desaturation may be associated with postoperative cognitive impairment [9, 10, 11].
Based on these considerations, we hypothesized that intraoperative cerebral oxygen desaturation during CPB is associated with decreased postoperative cognitive performance in patients undergoing cardiac surgery. Therefore, the present study aims to evaluate the relationship between intraoperative cerebral oxygen desaturation and POCD in order to explore perioperative factors that may be associated with postoperative cognitive outcomes.
This prospective observational study was conducted between March 2023 and March 2024 at the Department of Anesthesiology and Reanimation, University of Health Sciences Bursa Yüksek İhtisas Training and Research Hospital. The study protocol was approved by the institutional ethics committee on February 22, 2023 (approval no: 011-KAEK-25 2023/02-15). Written informed consent was obtained from all participants before enrollment.
A total of 102 patients scheduled for planned elective cardiac surgery with CPB
were screened for eligibility. Inclusion criteria were: age
Patients meeting the inclusion criteria and providing informed consent were
divided into two groups according to their postoperative cognitive outcomes.
Group 1 (POCD group) included patients whose MMSE scores dropped to
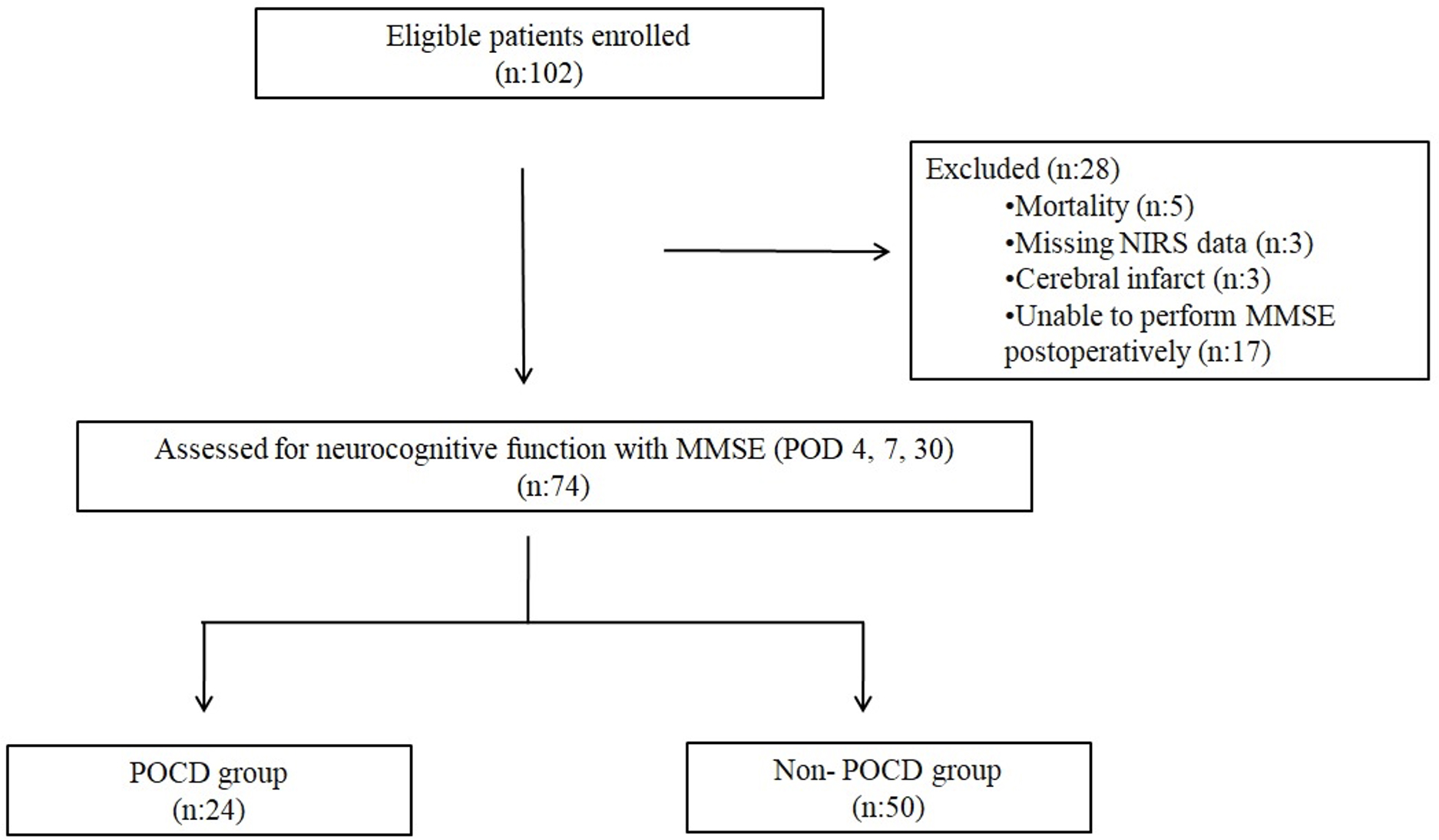 Fig. 1.
Fig. 1.
Flow diagram of patient enrollment, exclusion, and study grouping. n, Number of patients; NIRS, Near-Infrared Spectroscopy; POD, Postoperative day; MMSE, Mini-Mental State Examination; POCD, Postoperative cognitive dysfunction.
Eligible and consenting patients were evaluated during the preoperative anesthesia assessment. Demographic data, comorbidities, EuroSCORE risk evaluation, and current medications were recorded. The MMSE was administered preoperatively. Before the induction of anesthesia, bilateral NIRS probes were placed on the forehead, and baseline rSO2 values were recorded. During surgery, invasive arterial pressure, electrocardiography, pulse oximetry, central venous pressure, arterial blood gas analyses, body temperature, transfused blood and blood products, aortic cross-clamp (ACC) and CPB times, and total operative time were recorded. In the postoperative period, the duration of mechanical ventilation, complications, and length of stay in the intensive care unit (ICU) were monitored.
Cognitive assessments were performed preoperatively and on postoperative days
(POD) 4, 7, and 30. An MMSE score of
All patients underwent standard anesthesia induction with midazolam (Zolamid, Vem Pharmaceuticals, Istanbul, Türkiye) at 0.05–0.1 mg/kg, propofol (Propofol Fresenius, Bad Homburg, Germany) at 2 mg/kg, fentanyl (Talinat, Vem Pharmaceuticals, Istanbul, Türkiye) at 1–2 µg/kg, and rocuronium (Myokron, Vem Pharmaceuticals, Istanbul, Türkiye) at 0.6 mg/kg. An additional dose of fentanyl (3 µg/kg) was administered before sternotomy. Heparin (Koparin, Koçak Farma, Istanbul, Türkiye) was given before cannulation at a dose of 300–400 U/kg, and the activated clotting time was maintained above 400 seconds throughout CPB. Anesthesia was maintained with 2% sevoflurane, fentanyl, midazolam, and rocuronium in a mixture of 50% oxygen and 50% air. During CPB, perfusion flow was adjusted to 2.1–2.6 L/min/m2, maintaining a mean arterial pressure between 50 and 70 mmHg. Arterial blood gas analyses were performed every 30 minutes, and hematocrit levels were kept above 20%. At the end of CPB, heparin was reversed with protamine sulfate. All patients were transferred to the ICU while still intubated.
In the postoperative ICU period, routine sedation was not administered to patients undergoing planned elective cardiac surgery. Once the effects of intraoperative anesthetic agents had resolved, the weaning process from mechanical ventilation was initiated. Sedative agents were administered only to patients who experienced difficulty with weaning or developed clinical indications such as respiratory or hemodynamic instability. These patients were excluded from the study.
Regional cerebral oxygen saturation values were measured using the INVOS™ 5100C monitor (Medtronic, Minneapolis, MN, USA) with two probes placed on the patient’s frontal region. For data analysis, rSO2 values obtained from the right and left frontal sensors were averaged, and a single cerebral oxygen saturation value was analyzed at each measured time point [13]. Measurements were recorded at the following time points: T0 (before induction), T1 (15 minutes after induction), T2 (5 minutes after initiation of CPB), T3 (during the hypothermic phase of CPB), T4 (rewarming phase of CPB, at 34 ℃), T5 (weaning from CPB), and T6 (upon admission to the intensive care unit).
During surgery, standardized interventions were performed if rSO2 values decreased by more than 20% from the baseline to maintain cerebral oxygenation above this threshold [14]. These interventions included eliminating mechanical impediments to cerebral blood flow (such as repositioning of the head or bypass cannulas); improving cerebral oxygen delivery (by increasing FiO2, PaCO2, mean arterial pressure, cardiac output or pump flow, and hematocrit levels); and reducing cerebral oxygen consumption (by deepening anesthesia or lowering temperature). The overall aim was to restore physiological parameters that support adequate microcirculation [15].
Statistical analyses were performed using IBM SPSS Statistics for MacOS, version
30.0 (IBM Corp., Armonk, NY, USA). The distribution of continuous variables was
assessed using the Kolmogorov–Smirnov test; skewness and kurtosis values within
the range of
Between-group comparisons of rSO2 values at individual time points were
conducted for exploratory purposes only. To evaluate time-dependent changes in
intraoperative rSO2 values and differences between groups, repeated measures
analysis of variance (repeated-measures ANOVA) was applied. Repeated-measures
ANOVA was used to evaluate within-subject time effects and group
Repeated-measures ANOVA was also performed on percentage changes in rSO2 relative to baseline (T1–T6) to assess time- and group-dependent differences.
Associations between postoperative MMSE scores (POD4, POD7, and POD30),
intraoperative rSO2 percentage changes, and clinical outcomes (length of
intensive care unit stay and hospital stay) were examined using Spearman’s rank
correlation analysis. All statistical analyses were conducted using two-sided
tests. p
Initially, 102 patients were enrolled in the study. Twenty-eight patients were excluded from the final analysis due to various reasons, including mortality, inability to perform postoperative MMSE assessments, or incomplete intraoperative NIRS data. The final analysis was conducted on 74 patients (Fig. 1). When evaluating the incidence of POCD after cardiac surgery, cognitive decline was identified in 32.4% of patients within the first postoperative week. This rate decreased to 20.2% at the 30-day follow-up.
Among the included patients, 33 (44.6%) were female, and 41 (55.4%) were male.
The demographic and preoperative clinical characteristics of the POCD and
non-POCD groups are presented in Table 1. Patients in the POCD group were
significantly older than those in the non-POCD group (median [IQR]: 63 [58–68]
vs. 59 [52–65] years, p = 0.026). There were no significant differences
between the groups regarding sex distribution or EuroSCORE values (p
| Variables | POCD (n = 24) | Non-POCD (n = 50) | p-value |
| Age (years), Median (IQR) | 63 (58–68) | 59 (52–65) | 0.026 |
| Female, n (%) | 11 (45.8) | 22 (44) | 0.882 |
| Male, n (%) | 13 (54.2) | 28 (56) | |
| EuroSCORE, Median (IQR) | 4 (3–5) | 4 (2–4) | 0.065 |
| Hypertension, n (%) | 24 (100) | 50 (100) | - |
| Diabetes mellitus, n (%) | 14 (58.3) | 28 (56) | 0.850 |
| Chronic pulmonary disease, n (%) | 4 (16.7) | 9 (18) | 0.975 |
| Hyperlipidemia, n (%) | 11 (45.8) | 23 (46) | 0.989 |
| Peripheral artery disease, n (%) | 2 (8.3) | 4 (8) | 1.000 |
| Categories of open-heart surgery | |||
| Isolated CABG, n (%) | 17 (70.9) | 39 (78) | 0.908 |
| CABG + Valve surgery, n (%) | 2 (8.3) | 4 (8) | |
| Valve surgery only, n (%) | 3 (12.5) | 4 (8) | |
| Aortic surgery, n (%) | 2 (8.3) | 3 (6) | |
Abbreviations: p
A comparison of intraoperative and postoperative variables between the POCD and
non-POCD groups is presented in Table 2. The durations of surgery, CPB, and ACC
time were similar between the groups (p
| Variables | POCD (n = 24) | Non-POCD (n = 50) | p-value |
| Operation time (min), Median (IQR) | 262.5 (240–287.5) | 262.5 (240–290) | 0.949 |
| CPB time (min), Median (IQR) | 118 (91.5–152) | 106.5 (84–125) | 0.186 |
| ACC time (min), Median (IQR) | 86 (67.5–107.5) | 71 (55–95) | 0.146 |
| Inotropic support | |||
| No inotrope n (%) | - | 2 (4) | 0.950 |
| 1 inotrope, n (%) | 16 (66.7) | 33 (66) | |
| 2 inotrope, n (%) | 5 (20.8) | 12 (24) | |
| 3 inotrope, n (%) | 2 (8.3) | 3 (6) | |
| 4 inotrope, n (%) | 1 (4.2) | - | |
| IABP n (%) | 1 (4.2) | 1 (2) | 0.546 |
| Pacemaker use, n (%) | 6 (25) | 12 (24) | 0.962 |
| ICU stay (hours), Median (IQR) | 74 (51.3–96) | 50 (46–60) | |
| Hospital stay (days), Median (IQR) | 9 (8–15.5) | 7 (7–9) | |
| Extubation time (hours), Median (IQR) | 8 (6–14) | 7.8 (6–10) | 0.542 |
| CPAP requirement or reintubation n (%) | 5 (20.8) | 5 (10) | 0.277 |
| Arrhythmia, n (%) | 18 (75) | 38 (76) | 0.925 |
| Pneumonia n (%) | 5 (20.8) | 5 (10) | 0.277 |
| Surgical site infection, n (%) | 2 (8.3) | 4 (8) | 1.000 |
Abbreviations: p
Intraoperative rSO2 values, calculated as the average of bilateral frontal
measurements, and their percentage changes relative to baseline are presented in
Table 3. There were no significant differences between the POCD and non-POCD
groups in T0 or T1 rSO2 values (p
| NIRS Values, Median (IQR) | POCD (n = 24) | Non-POCD (n = 50) | p-value |
| T0 rSO2, Median (IQR) | 63.8 (62.3–68.5) | 65.5 (61.5–69) | 0.694 |
| T1 rSO2, Median (IQR) | 69.8 (66–78.5) | 71.5 (67–77) | 0.673 |
| T2 rSO2, Median (IQR) | 51.8 (47.3–55) | 55.5 (50–59.5) | 0.020 |
| T3 rSO2, Median (IQR) | 52.8 (49.3–54.8) | 56.5 (51.5–59) | 0.002 |
| T4 rSO2, Median (IQR) | 52.5 (49.3–56) | 58 (54.5–63.5) | |
| T5 rSO2, Median (IQR) | 61.8 (56.5–63.5) | 63.5 (57.5–68) | 0.155 |
| T6 rSO2, Median (IQR) | 61.8 (56.8–64.8) | 62.8 (59.5–69.5) | 0.212 |
| NIRS percentage change values | |||
| T0–T1 %, Median (IQR) | 10.7 (6.5–15.3) | 9.4 (5.8–15.7) | 0.959 |
| T0–T2 %, Median (IQR) | –21.9 (–25.1 to –15.3) | –14.8 (–19 to –9.6) | 0.005 |
| T0–T3 %, Median (IQR) | –18.4 (–25.5 to –15.7) | –14.9 (–18.8 to –8.7) | 0.003 |
| T0–T4 %, Median (IQR) | –17 (–21.5 to –15) | –10.8 (–15.7 to –3.6) | |
| T0–T5 %, Median (IQR) | –5.2 (–11.3–0) | –0.8 (–10.9–6.7) | 0.179 |
| T0–T6 %, Median (IQR) | –4.7 (–12.2–0.8) | –4.2 (–9.3–5.7) | 0.239 |
Abbreviations: p
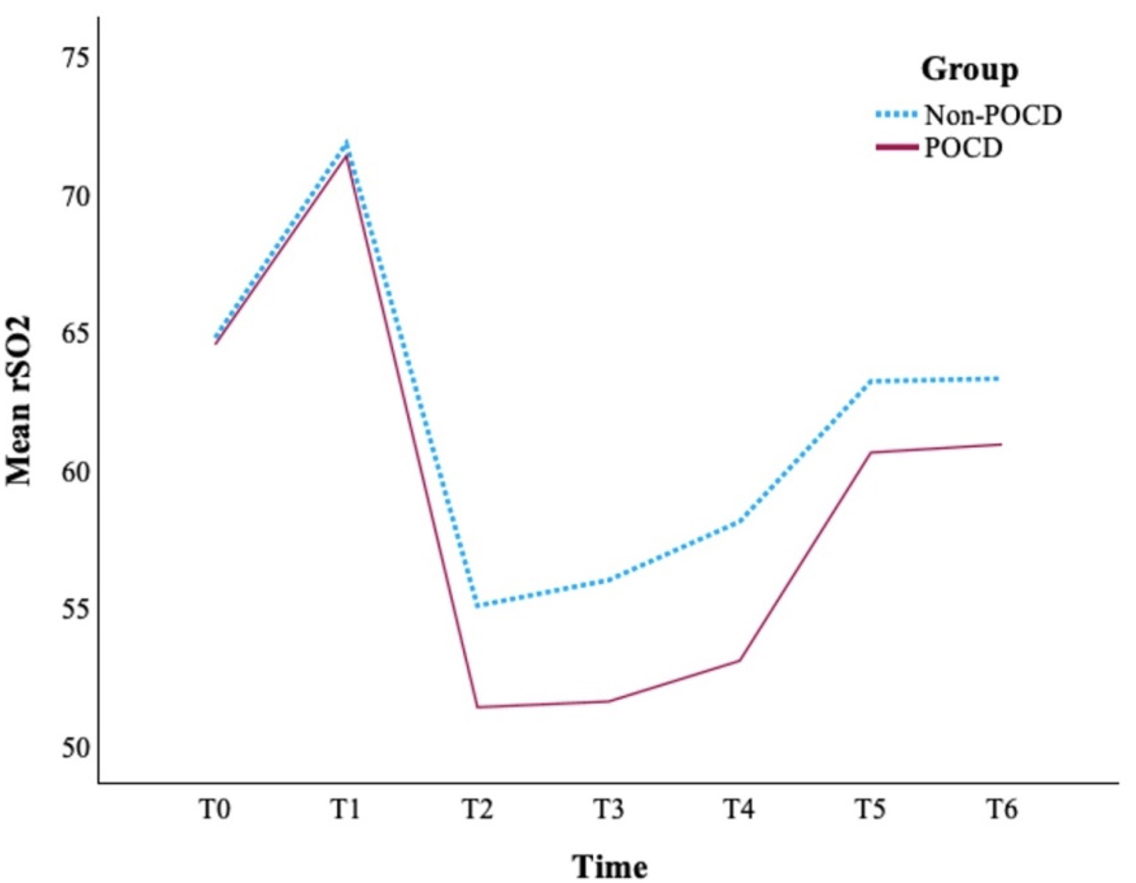 Fig. 2.
Fig. 2.
Time-dependent changes in intraoperative rSO2 values in POCD and non-POCD patients. T0, Preoperative baseline; T1, post-induction (15 min); T2, CPB initiation (5 min); T3, CPB hypothermic phase; T4, Rewarming phase of CPB; T5, Weaning from CPB; T6, ICU admission; rSO2, Regional cerebral oxygen saturation; CPB, Cardiopulmonary Bypass; ICU, Intensive Care Unit.
Repeated measures analysis of variance demonstrated a significant main effect of
time (p
When percentage changes in rSO2 relative to baseline were evaluated, a
significantly greater reduction was observed in the POCD group at T2, T3, and T4
compared with the non-POCD group. At T2, the median percentage change was –21.9%
in the POCD group and –14.8% in the non-POCD group (p = 0.005).
Similarly, greater desaturation was noted at T3 (–18.4% vs. –14.9%, p
= 0.003) and T4 (–17.0% vs. –10.8%, p
For percentage change values, repeated measures analysis revealed a significant
main effect of time (p
The correlations between intraoperative percentage changes in rSO2 relative
to baseline and postoperative cognitive performance are presented in Table 4.
Significant positive correlations were observed between rSO2 percentage
changes at T2, T3, and T4 and MMSE scores on POD4 and POD30 (p
| rSO2 (%) | POD4 | POD7 | POD30 |
| T0–T1 | r = –0.104 | r = 0.026 | r = –0.127 |
| p = 0.377 | p = 0.827 | p = 0.279 | |
| T0–T2 | r = 0.283 | r = 0.206 | r = 0.314 |
| p = 0.014 | p = 0.078 | p = 0.006 | |
| T0–T3 | r = 0.312 | r = 0.144 | r = 0.270 |
| p = 0.007 | p = 0.221 | p = 0.020 | |
| T0–T4 | r = 0.312 | r = 0.216 | r = 0.349 |
| p = 0.007 | p = 0.065 | p = 0.002 | |
| T0–T5 | r = 0.079 | r = –0.025 | r = 0.109 |
| p = 0.506 | p = 0.832 | p = 0.356 | |
| T0–T6 | r = 0.109 | r = 0.001 | r = 0.189 |
| p = 0.353 | p = 0.995 | p = 0.107 |
Abbreviations: p
When factors potentially influencing MMSE scores were analyzed, the duration of
ICU stay (hours) showed a significant negative correlation with MMSE scores on
POD4, POD7, and POD30 (r = –0.435, p
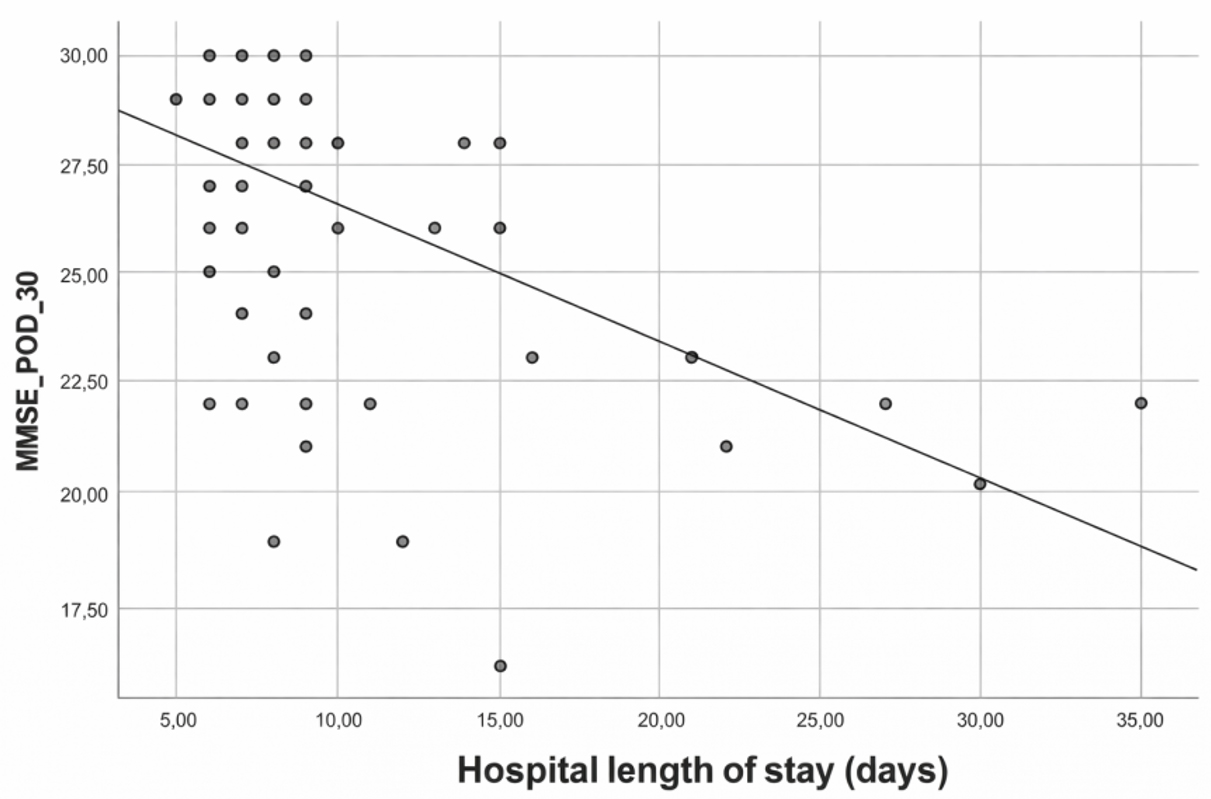 Fig. 3.
Fig. 3.
Relationship between hospital length of stay and MMSE score on postoperative day 30. MMSE, Mini-Mental State Examination; POD, Postoperative day.
| Variables | POD4 | POD7 | POD30 |
| MMSE | MMSE | MMSE | |
| Operation time (min) | r = 0.040 | r = 0.034 | r = 0.110 |
| p = 0.737 | p = 0.773 | p = 0.349 | |
| CPB time (min) | r = –0.096 | r = –0.086 | r = –0.117 |
| p = 0.415 | p = 0.465 | p = 0.322 | |
| ACC time (min) | r = –0.114 | r = –0.135 | r = –0.166 |
| p = 0.333 | p = 0.251 | p = 0.157 | |
| ICU stay (hours) | r = –0.435 | r = –0.270 | r = –0.413 |
| p |
p = 0.020 | p | |
| Hospital stay (days) | r = –0.428 | r = –0.319 | r = –0.489 |
| p |
p = 0.006 | p |
Abbreviations: p
In this prospective observational study, intraoperative cerebral oxygen desaturation during cardiopulmonary bypass was significantly associated with postoperative cognitive dysfunction, with greater intraoperative rSO2 reductions observed in patients who developed cognitive impairment.
Despite significant advances in surgical safety measures and anesthesia management, POCD following cardiac surgery remains a major clinical challenge in terms of both prevention and management. POCD has been associated with adverse outcomes such as prolonged hospital stay, reduced quality of life, increased socioeconomic burden, and higher mortality rates [8]. With the progressive aging of the population, studies have reported that the incidence of POCD may reach up to 56% among individuals older than 55 years after surgery [16].
The variability in the reported incidence across studies may be attributed to heterogeneity in the neurocognitive assessment tools, timing of postoperative testing, the absence of a universally accepted definition of POCD, and differences in the cardiovascular risk profile and surgical techniques of the studied populations [17]. Early postoperative cognitive impairment is a significant predictor of subsequent POCD, and this association persists up to three months postoperatively [18, 19, 20]. Therefore, early identification of cognitive decline and implementation of appropriate follow-up strategies are crucial for reducing the long-term risk of dementia.
A total of 102 patients were initially screened, and 74 were included in the final analysis after applying exclusion criteria (Fig. 1). The incidence of POCD was 32.4% during the first postoperative week and decreased to 20.2% by postoperative day 30, reflecting an early decline followed by partial recovery over time. Baseline characteristics were comparable between the POCD and non-POCD groups regarding sex distribution, comorbidities, and types of surgical procedures; however, patients in the POCD group were significantly older than those without POCD (Table 1). Although EuroSCORE values tended to be higher in the POCD group, this difference did not reach statistical significance and remained at a borderline level. These findings suggest that advanced age represents a key patient-related factor associated with the development of POCD, whereas overall perioperative risk burden, as reflected by EuroSCORE, may contribute but does not independently discriminate between groups. Halacli et al. [21] also reported that increasing age and comorbidity burden adversely influence clinical outcomes in critically ill patients. The comparable distribution of surgical procedures between the two groups further minimizes procedure-related confounding, supporting the interpretation that the observed differences are primarily related to patient-level vulnerability rather than surgical factors. Future studies employing multivariable analytical models are warranted to better delineate the independent contribution of age and other perioperative risk indicators for the development of POCD.
Although the duration of CPB and ACC was longer in the POCD group than in the non-POCD group, these differences were not significant (Table 2). Therefore, no significant association between CPB and ACC durations and POCD was found in this cohort. Nevertheless, previous studies have suggested that prolonged CPB and ACC times may be associated with a tendency toward cognitive decline [22, 23]. Thus, the observed numerical differences, although non-significant, should be interpreted with caution and considered hypothesis-generating rather than confirmatory. De Backer et al. [24] demonstrated that microcirculatory flow is markedly impaired during CPB, which may subsequently compromise postoperative organ function. Such microcirculatory disturbances, in conjunction with an intensified systemic inflammatory response, can lead to tissue hypoperfusion and oxygen deficit—mechanisms recognized as key contributors to the pathogenesis of POCD [25]. Related complications such as atrial fibrillation, acute kidney injury, and prolonged mechanical ventilation may represent clinical manifestations of these shared pathophysiological pathways. Similarly, systemic inflammatory mechanisms leading to multiorgan dysfunction have also been emphasized in critical care studies investigating severe illness [26].
In the present study, extubation time did not differ significantly between the POCD and non-POCD groups (Table 2). Postoperative complication rates were also comparable between groups. However, both ICU length of stay and total hospital stay were significantly prolonged in patients who developed POCD (Table 2). These findings are consistent with the results of Zhuang et al. [27], who reported that prolonged ICU stay after CPB has been associated with an increased risk of postoperative cognitive decline. These findings suggest that POCD is more closely associated with a prolonged postoperative recovery process rather than parameters of intraoperative duration.
Studies in the field of cardiovascular surgery have demonstrated that non-pulsatile blood flow, inflammatory response, hemodilution, and hypothermia during CPB can markedly impair microcirculation, disrupting mitochondrial oxygen balance at the cellular level and contributing to the development of various complications, including POCD [28, 29]. Findings from neuromonitoring research further support the contribution of cerebral hypoperfusion to the pathogenesis of POCD. Studies using NIRS have shown that intraoperative decreases in cerebral oxygen saturation are associated with postoperative cognitive changes, and that early episodes of cerebral desaturation may serve as valuable predictors for the development of POCD [10, 11].
These findings are consistent with the results of the present study. Significant
intraoperative changes in rSO2 values were observed following the initiation
of CPB (Table 3, Fig. 2). In the POCD group, rSO2 levels were significantly
lower than those in the non-POCD group during the intermediate intraoperative
phases, specifically at T2, T3, and T4. In contrast, no significant between-group
differences were observed at later time points (T5 and T6). Furthermore, analyses
of percentage changes relative to preoperative baseline values revealed more
pronounced cerebral desaturation in the POCD group during the same intermediate
intraoperative phases. Although absolute rSO2 levels differed between the
groups, analyses of percentage changes suggested a similar temporal pattern over
time, consistent with the absence of a significant group
Neuropsychological assessments performed in the early postoperative period may be influenced by multiple factors, including pain, residual effects of anesthetic agents, pharmacologic interventions, and sleep disturbances [30]. Therefore, early postoperative declines in cognitive performance cannot be entirely attributed to changes in cerebral oxygenation alone. In the present study, significant positive correlations were identified between intraoperative percentage changes in rSO2 during the CPB (T2, T3, and T4) and MMSE scores assessed on POD4 and POD30 (Table 4). These findings suggest that more pronounced cerebral desaturation during these critical intraoperative periods is associated with lower cognitive performance in both the early and late postoperative phases.
In contrast, no significant correlations were observed between rSO2 percentage changes and MMSE scores on POD7, indicating that the relationship between intraoperative cerebral oxygenation and cognitive performance may vary over time. Furthermore, no significant associations were identified between rSO2 changes at T1, T5, or T6 and MMSE scores at any postoperative assessment. These results suggest that cerebral desaturation occurring during the intermediate phases of CPB (T2–T4) is more closely associated with early and late postoperative cognitive performance than desaturation observed at other intraoperative time points. Nevertheless, given the observational nature of this study, larger prospective investigations are warranted to define clinically meaningful desaturation thresholds and clarify the temporal sensitivity of different intraoperative periods in relation to postoperative cognitive trajectories.
Previous studies have demonstrated that alterations in rSO2 values and impaired cerebral autoregulation—particularly during phases of increased cerebral metabolic demand such as rewarming—may be associated with postoperative cognitive decline [31]. In the present study, significant associations between intraoperative rSO2 percentage changes and postoperative cognitive performance were observed during the initiation and hypothermic phases (T2–T3), particularly during the rewarming phase (T4) of CPB. These findings suggest that periods of increased vulnerability to cerebral desaturation during CPB may be relevant to subsequent cognitive outcomes.
Heterogeneity in desaturation thresholds, intraoperative intervention protocols,
and POCD assessment methods has been identified as a major source of variability
amongst studies. Zhang et al. [32] reported that the relationship
between rSO2 changes and POCD was particularly evident during the rewarming
phase on postoperative day 7; however, their intervention strategy was limited to
adjustments of mean arterial pressure and head positioning. Similarly, Uysal
et al. [33] defined an intervention threshold of rSO2
Future studies with larger sample sizes, standardized intervention thresholds, and comprehensive statistical models are warranted to clarify the specific impact of intraoperative rSO2 fluctuations—particularly on late (30-day) cognitive outcomes. In addition, several studies have shown that prolonged ICU and hospital stays are associated with impaired cognitive function [27, 30]. Consistent with these findings, our study demonstrated a significant negative correlation between both ICU and total hospital stay durations and MMSE scores at POD4 and POD30 (Table 5, Fig. 3).
These findings underscore the importance of implementing strategies such as early mobilization, maintenance of environmental orientation, and delirium prevention protocols, particularly in elderly patients at risk for cognitive impairment. Optimization of ICU and hospital length of stay durations, together with measures aimed at reducing postoperative complications and delirium, may represent an important component of multimodal approaches to support cognitive recovery within the multifactorial framework contributing to the development of POCD. Furthermore, Bendikaite and Vimantaite reported that cognitive impairment following cardiac surgery has not only early postoperative morbidity implications but also lasting effects on patients’ quality of life [34]. Collectively, these observations highlight the necessity of adopting an integrated perioperative approach that simultaneously addresses hemodynamic stability and neurocognitive monitoring to mitigate postoperative cognitive dysfunction.
This study has several limitations. First, cognitive function was assessed only during the early postoperative period (within the first 30 days), and long-term neurocognitive outcomes were not evaluated. This limits the ability to determine whether POCD represents a transient or persistent condition. Second, although patients with clinically evident postoperative delirium were excluded based on routine clinical examination, delirium was not systematically assessed using standardized diagnostic tools, and other potential confounding factors that may influence MMSE scores in the postoperative period—such as pain level, sleep quality, and pharmacologic interventions—were not formally quantified. Therefore, the cognitive changes observed during this period cannot be exclusively attributed to surgical factors or cerebral oxygenation. Third, cognitive evaluation relied solely on the MMSE, and more comprehensive neuropsychological test batteries or alternative tools assessing attention and executive functions were not utilized. Finally, the single-center design and relatively small sample size may limit the generalizability of the findings. Nevertheless, the prospective design, multi-timepoint cognitive assessments, and the use of standardized intraoperative NIRS monitoring enhance the methodological strength of this study.
In this prospective observational study, intraoperative cerebral oxygen desaturation during cardiopulmonary bypass was found to be associated with postoperative cognitive dysfunction in patients undergoing cardiac surgery. More pronounced reductions in rSO2, particularly during the rewarming (T4) phases of cardiopulmonary bypass and the subsequent initiation and periods of hypothermia (T2–T3), were related to poorer cognitive performance in both the early and late postoperative periods. In addition to intraoperative cerebral oxygenation patterns, patient- and postoperative-related factors—most notably advanced age and prolonged intensive care unit and hospital stays—were consistently associated with lower postoperative MMSE scores.
These findings support the multifactorial nature of POCD and highlight intraoperative cerebral oxygenation as an important component within a broader perioperative risk profile rather than as an isolated determinant of cognitive outcome. Continuous intraoperative monitoring of cerebral oxygen saturation using near-infrared spectroscopy may contribute to the early identification of cerebral hypoperfusion and support timely corrective interventions, particularly in patients at increased risk of cognitive impairment following cardiac surgery.
Future large-scale, multicenter studies employing standardized intervention thresholds and comprehensive analytical models are warranted to further clarify the clinical relevance of intraoperative rSO2 changes and better characterize their relationship with short- and long-term postoperative cognitive trajectories.
The data generated and analysed during this study are presented in this article. Additional details or datasets are available from the corresponding author upon reasonable request.
SBU conceived and designed the study, coordinated the data analysis, drafted the manuscript, and served as the corresponding author. BÖ contributed to data interpretation, manuscript writing, and critical revision of the manuscript. KÖ performed the statistical analyses, contributed to data visualization, and critically reviewed the manuscript for important intellectual content. MHU participated in data collection and patient follow-up and critically reviewed the manuscript. GG contributed to the literature review, clinical interpretation of the findings, and critical revision of the manuscript. DBG assisted in data verification, ethical documentation, and critical revision of the manuscript. MG contributed to the study design, supervised the overall project, and critically reviewed the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors agree to be accountable for all aspects of the work.
The study was conducted in accordance with the principles of the Declaration of Helsinki. Ethical approval was obtained from the Ethics Committee of the University of Health Sciences, Bursa Yüksek İhtisas Training and Research Hospital (approval date: February 22, 2023; protocol no: 2011-KAEK-25 2023/02-15). Written informed consent was obtained from all participants prior to inclusion in the study.
The authors sincerely thank all patients who generously agreed to participate in this study.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this manuscript, the authors used ChatGPT-5 (OpenAI) to assist in improving the clarity and fluency of the English language and to check grammar consistency. The authors subsequently reviewed, revised, and approved all contents of the manuscript, and take full responsibility for the integrity and accuracy of the published work.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



