1 Department of Anesthesiology, Intensive Care Medicine, Emergency Medicine, Pain and Palliative Therapy, Asklepios Klinikum Harburg, 21075 Hamburg, Germany
2 Asklepios Campus Hamburg Asklepios Medical School GmbH, 20099 Hamburg, Germany
3 Department of Anesthesiology & Intensive Care Medicine, Sana Herzzentrum Cottbus, 03048 Cottbus, Germany
4 Department of Internal Medicine II – Cardiology and Internal Intensive Care Medicine, Asklepios Klinik Nord – Heidberg, 22417 Hamburg, Germany
5 Department of Cardiac Surgery, Asklepios Klinikum Harburg, 21075 Hamburg, Germany
Abstract
This study aimed to compare the hemodynamic effects of two analgosedation regimens, Remifentanil/Propofol vs. Dexmedetomidine/Propofol, during transfemoral transcatheter aortic valve implantation (tf-TAVI).
In this monocentric, prospective, non-randomized observational study, 143 patients undergoing tf-TAVI between November 2021 and November 2023 were analyzed. Patients received either Remifentanil/Propofol or Dexmedetomidine/Propofol as part of their standard sedation regimen. A sensitivity analysis using propensity score matching was performed to support the primary analysis.
The primary outcome was circulatory stability, assessed by the need for catecholamines to maintain a mean arterial pressure (MAP) of 65 mmHg. Catecholamine use was significantly higher in the Dexmedetomidine group (78% vs. 55%; p = 0.012). No significant differences were observed in major post-procedural complications (Remifentanil: 19.05% vs. Dexmedetomidine: 13.56%; p = 0.389) or postoperative delirium scores. However, intraoperative agitation, defined as observed procedure disruption, was significantly less frequent in the Remifentanil group (33% vs. 69%; p = 0.01). Propensity score matching confirmed the robustness of these findings.
In conclusion, both sedation strategies were associated with comparable overall clinical outcomes in patients undergoing tf-TAVI. However, our findings suggest that a tailored sedation approach may be warranted: Remifentanil–Propofol may be preferable in patients with fragile hemodynamics, whereas dexmedetomidine–propofol may represent a safer option in patients at risk for respiratory compromise. These observations support an individualized, patient-centered sedation strategy rather than a one-size-fits-all approach.
Keywords
- transcatheter aortic valve replacement
- conscious sedation
- procedural sedation and analgesia
- hemodynamics
- high risk patients
- dexmedetomidine
- remifentanil
- treatment outcome
- postoperative complications
Analgosedation has been firmly established as an anesthesia concept for many years. Many procedures or interventions, such as transfemoral transcatheter aortic valve implantation (tf-TAVI), which were initially performed primarily under general anesthesia, are now predominantly carried out under analgosedation [1, 2, 3]. Analgosedation in this study is defined as the use of pharmacological agents to facilitate therapeutic procedures while providing adequate analgesia and anxiolysis, and preserving airway patency, spontaneous respiration, and hemodynamic stability [4]. Compared with general anesthesia, lower medication doses, reduced cardiovascular strain, and preservation of spontaneous respiration appear to contribute to favorable outcomes. Multiple studies have demonstrated advantages of analgosedation over general anesthesia [5]. Including shorter intensive care stays, reduced time of intervention and reduced requirements of vasopressors. Importantly, no inferiority in treatment outcomes has been shown [1, 6, 7]. In patients undergoing tf-TAVI, analgosedation enables early neurological assessment, which may be clinically relevant given the reported peri-interventional stroke risk of approximately 1–3% [8].
The drugs used for analgosedation in published studies included Benzodiazepines, Propofol, Opioids (especially Remifentanil owing to its favorable pharmacokinetic profile and excellent titratability), Dexmedetomidine, Ketamine and Esketamine [9, 10, 11, 12]. These drugs were administered either as monotherapies or in combination to exploit potential synergistic effects across pharmacological classes. While numerous studies have compared analgosedation with general anesthesia, few have focused on specific analgosedation regimens. Existing investigations of explicit sedation protocols primarily focus on the field of intensive care medicine, for instance, studies comparing dexmedetomidine and propofol regarding delirium incidence.
Previously published studies comparing Propofol combined with an opioid (typically Remifentanil) with Dexmedetomidine have demonstrated greater hemodynamic stability in the Dexmedetomidine treatment arm [13]. Furthermore, studies suggest that Dexmedetomidine may confer a protective effect against the development of delirium [14].
The present study aimed to evaluate the hemodynamic effects of two
analgosedation regimens, Propofol-Remifentanil (Remi group) and
Propofol-Dexmedetomidine (Dex group), in a prospective, observational study
during transfemoral transcatheter aortic valve implantation (tf-TAVI). These
combinations were selected to potentially exploit synergistic pharmacological
effects. By combining propofol with either an opioid or an
This investigation was a monocentric, non-randomized, non-blinded, prospective observational study. The two regimens represented standard approaches routinely employed at our institution and were already integrated into clinical practice. The choice of medication regimen was determined at the discretion of the attending anesthesiologists.
The perioperative risk assessment was conducted according to the hospital’s standard procedure, which complies with the mandatory regulations of the German Federal Joint Committee [17]. The resulting group of patients met the criteria for high-risk patients (American Society of Anesthesiology Classification (ASA) 3 or higher) [18].
After verifying the World Health Organization (WHO) checklist, arterial puncture was performed under local anesthesia without sedation in room air. Sedation was then initiated according to study protocol (Remi: Propofol 1 mg/kg/h & Remifentanil 0.05 µg/kg/min; Dex: Propofol 0.5 mg/kg/h & Dexmedetomidine 0.4 µg/kg/h). Further instrumentation via the jugular or femoral route was performed under local anesthesia. The interventionalist administered local anesthesia to the access site. If needed, a propofol rescue bolus was given with infusion adjustments for Propofol, Dexmedetomidine or Remifentanil. Dose modifications followed the study protocol. Post-procedure management was risk-based, with monitoring in the Intensive Care Unit (ICU), Intermediate Care Unit (IMC), or a monitored ward [19]. During the procedure, the aim was to achieve a sedation level of –1 on the Richmond Agitation and Sedation Scale (RASS).
Parameters such as the Score of the Intensive Care Delirium Screening Checklist (ICDSC) were collected by medical personnel. The attending anesthesiologists were specialists with verifiable experience in cardiac anesthesia and had undergone training according to the curriculum of the German Society of Anesthesiology [20]. Funding was provided by internal clinical research funding. Ethical approval was granted on August 31, 2021 (2021-100636-BO-ff).
Patients who met all the following criteria were eligible for inclusion in the study:
• The patient’s capacity to consent.
• The patient’s written consent for tf-TAVI under analgosedation and agreement to participate in the study.
• Age
• Indication for tf-TAVI in accordance with current cardiological guidelines and recommendations.
Patients were excluded if at least of the following conditions were present:
• Obstructive sleep apnea syndrome.
• Severe obstructive (Global Initiative for Chronic Obstructive Lung
Disease (GOLD) III or worse) or restrictive ventilation disorders (forced vital
capacity (FVC)
• Severely impaired left or right ventricular function (ejection
fraction (EF)
• Pulmonary hypertension with high risk (according to the guidelines of the German Society of Cardiology).
• Withdrawal of consent after the intervention.
• Necessity for the procedure to be performed under general anesthesia.
• Conversion of analgosedation into general anesthesia.
The study was conducted exclusively at a tertiary care center with a dedicated cardiac surgery department in Germany. The study period lasted from November 2021 to November 2023.
Our primary endpoint was hemodynamic stability, measured by the requirement for catecholamines. Hemodynamic management followed an algorithm-guided, target-based approach aiming to maintain a mean arterial pressure (MAP) of at least 65 mmHg. In cases of hypotension, a structured, standard operating procedure based sequence was applied: first, assessment of intravascular volume status and correction of hypovolemia if indicated; second, evaluation of cardiac function and rhythm disturbances; third, treatment of vasodilation with norepinephrine as the first-line vasopressor. This standardized approach was consistently applied across both sedation regimens. The type and amount of medication used were documented in a separate protocol.
Secondary endpoints included:
• Patient agitation, which disrupts the intervention, as observed by
the attending anesthesiologist during the procedure (RASS
• Delirium four hours post-intervention and on the first post-intervention day, assessed using the ICDSC score.
• pCO2 in arterial blood gas analysis after arterial cannulation (room air without sedation) and in the second blood gas analysis at the time of valve implantation.
• Occurrence of major post-intervention complications, defined as
major stroke, myocardial infarction, aortic valve insufficiency
• Discharge data (length of stay in IMC, ICU, and general ward).
Other variables for the analysis collected were:
• EuroScore II, calculated using the Royal Papworth Hospital formula (https://www.euroscore.org/index.php?id=17&lang=en).
• Demographic data such as age, sex, medical history and body mass index (BMI).
• Medication doses and administration of all medications used.
• Comorbidities (e.g., diabetes mellitus (DM), Coronary Artery Bypass Graft, transient ischemic attack or stroke).
• Pre-intervention echocardiographic parameters (e.g., right end left ventricular EF, aortic valve area (AVA), peak and mean aortic valve pressure (PmaxAV, PmedAV), systolic pulmonary arterial pressure (sPAP)).
Continuous variables were summarized using means, standard deviations, medians, and interquartile ranges. Categorical data were presented as absolute and relative frequencies, with key variables shown graphically.
Norepinephrine, Propofol, Remifentanil, and Dexmedetomidine doses were standardized to a median body weight of 74 kg and a median procedure time of 140 minutes. The primary outcome, the use of norepinephrine and resulting dose during sedation, was modeled using a gamma model with zero inflation. Factors like sedation group, age, sex, LVEF, Propofol dose and glomerular filtration rate (GFR) were included as potential predictors for norepinephrine use and dose. Model results were reported with parameter estimates, standard errors, statistics, and p-values. Sensitivity analysis was performed using matched samples based on EF, age, sex, DM, and atrioventricular blockage.
The effects of the sedation group on secondary outcomes were assessed using negative binomial models for counting data, ordinal logit models for ordered categories (e.g., patient satisfaction), and linear models for continuous variables. Results were reported with parameter estimates, standard errors, statistics, p-values, and effects (odds ratios (OR), expected values). Propensity score matching was applied to enhance statistical validity in the absence of randomization. A sensitivity analysis of the model results was then performed using matched samples, with matching criteria including ejection fraction, age, sex, diabetes mellitus, and the presence of an atrioventricular (AV) block. A required sample size of 272 was calculated to achieve a statistical power of 0.8.
All p-values were two-sided, with significance set at p
Of the 208 patients included, three underwent open-heart surgery. 38 patients could not be included for structural reasons, like missing ICU capacities. In eight other patients the intervention was performed primarily under general anesthesia for medical reasons. Intubation was required for eight additional patients for medical reasons during the intervention. Another eight patients were excluded according to the study protocol. Further information can be found in Fig. 1. The demographic data of the 143 patients are listed in Table 1. The two sedation regimen groups differed in sex composition; in the Dex group, 59% of participants were female (vs. 40% in the Remi group; p = 0.026). Furthermore, there were more patients with a history of chronic obstructive pulmonary disease (COPD) in the Dex group (15% vs. 8.3%; p = 0.2). The other parameters collected were balanced between the two groups.
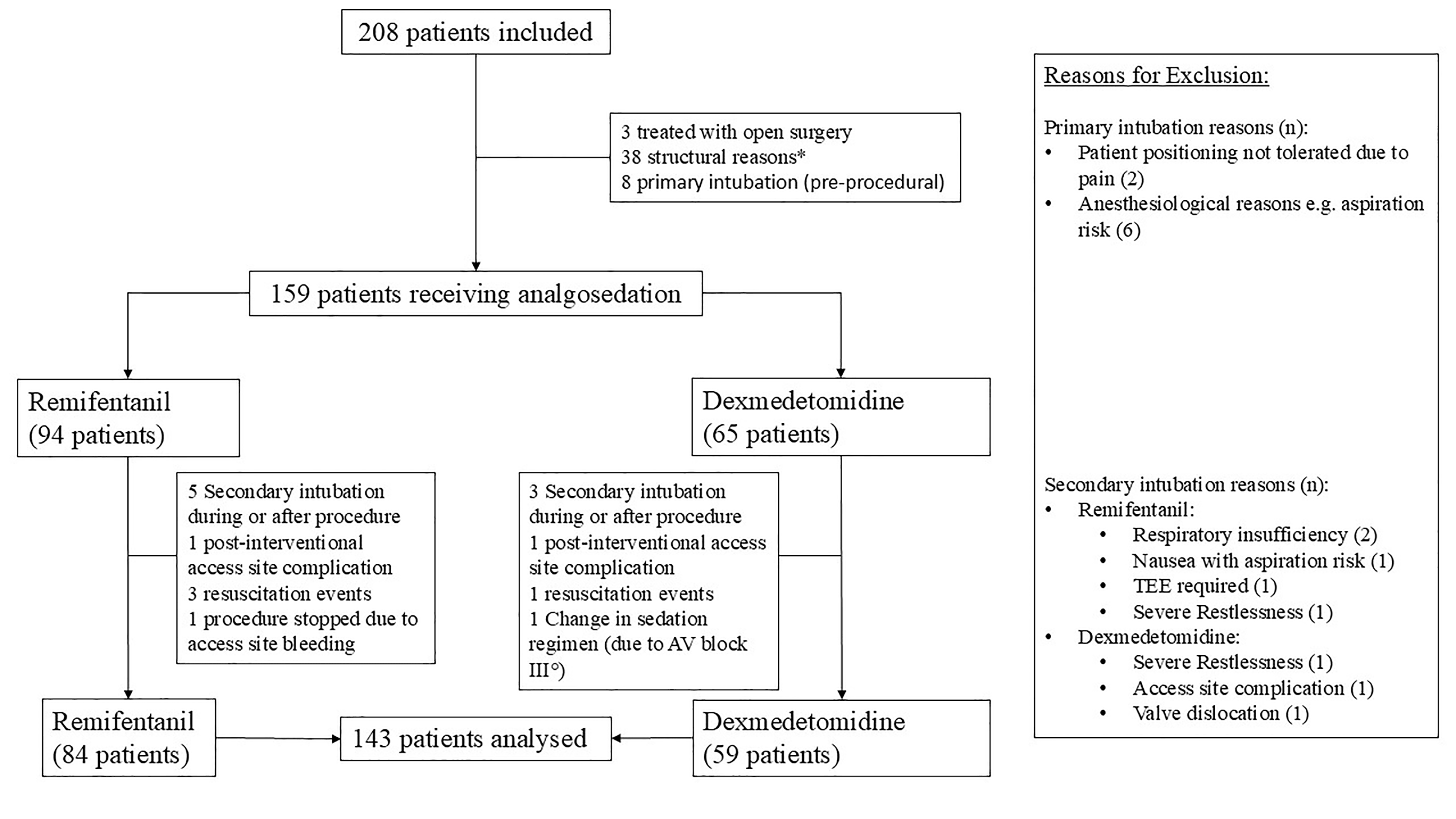 Fig. 1.
Fig. 1.
Flow chart illustrating patient recruitment and reasons for exclusion. Numbers in parentheses indicate the number of patients excluded for each criterion. The exclusion criteria applied in the study are listed on the right-hand side of the figure. *Structural reasons were not systematically recorded. The most common reason was limited post-interventional monitoring capacity. Abbreviations: TEE, transesophageal echocardiography; AV, atrioventricular.
| Population | Dex | Remi | ||
| N = 143 | N = 59 | N = 84 | ||
| Sex [female, % (n)] | 48% (69) | 59% (35) | 40% (34) | |
| Age [Mean, years (SD)] | 82.7 (4.8) | 82.9 (4.7) | 82.5 (4.8) | |
| BMI [Mean, kg/m2 (SD)] | 26.1 (3.7) | 25.7 (3.7) | 26.4 (3.7) | |
| Smokers [% (n)] | 13% (18) | 15% (9) | 11% (9) | |
| Diabetes mellitus [% (n)] | 22% (31) | 15% (9) | 26% (22) | |
| AV Block [% (n)] | 17% (24) | 6.8% (4) | 24% (20) | |
| CIED [% (n)] | 8.4% (12) | 3.4% (2) | 12% (10) | |
| Myocardial infarction [% (n)] | 11% (16) | 14% (8) | 9.5% (8) | |
| CABG [% (n)] | 5.6% (8) | 6.8% (4) | 4.8% (4) | |
| COPD [% (n)] | 11% (16) | 15% (9) | 8.3% (7) | |
| TIA [% (n)] | 20% (28) | 19% (11) | 20% (17) | |
| Aortic valve opening area [Median, cm2 (Q1, Q3)] | 0.80 (0.70, 0.90) | 0.80 (0.70, 0.90) | 0.80 (0.70, 0.90) | |
| Left Ventricular EF [categories, % (n)] | ||||
| Normal | 81% (116) | 81% (48) | 81% (68) | |
| Slightly reduced | 13 (18) | 12 (7) | 13 (11) | |
| Moderately reduced | 6.3 (9) | 6.8 (4) | 6.0 (5) | |
| Coronary heart disease [% (n)] | 71% (102) | 68% (40) | 74% (62) | |
| GFR [Mean, mL/min (SD)] | 58 (19) | 60 (20) | 57 (19) | |
| EuroSCORE II [Median (Q1, Q3)] | 3.3 (2.2, 4.5) | 3.1 (2.1, 4.9) | 3.3 (2.4, 4.4) | |
AV, atrioventricular; BMI, body mass index; CABG, coronary artery bypass graft; CIED, cardiac implantable electric device; COPD, chronic obstructive pulmonary disease; Dex, Dexmedetomidine group; EF, Ejection fraction; GFR, glomerular filtration rate; Remi, Remifentanil group; TIA, transient ischemic attack.
Regarding the primary endpoint, norepinephrine was used as the sole catecholamine in all analyzed patients. Patients receiving adrenaline due to resuscitation events were excluded from the analysis. The use of norepinephrine was three times less frequent in the Remi group compared to the Dex group. This difference was statistically significant (OR 2.89; 95% CI 1.26–6.59; p = 0.012; see Fig. 2). Among patients who required norepinephrine, the required dose in the Remi group was 1.8 times higher than in the Dex group. In standardized terms, the median norepinephrine dose used was approximately 0.02 mcg/kg/min in the Dex group and 0.03 mcg/kg/min in the Remi group. This difference was also statistically significant (RR 1.81; 95% CI 1.21–2.7; p = 0.004).
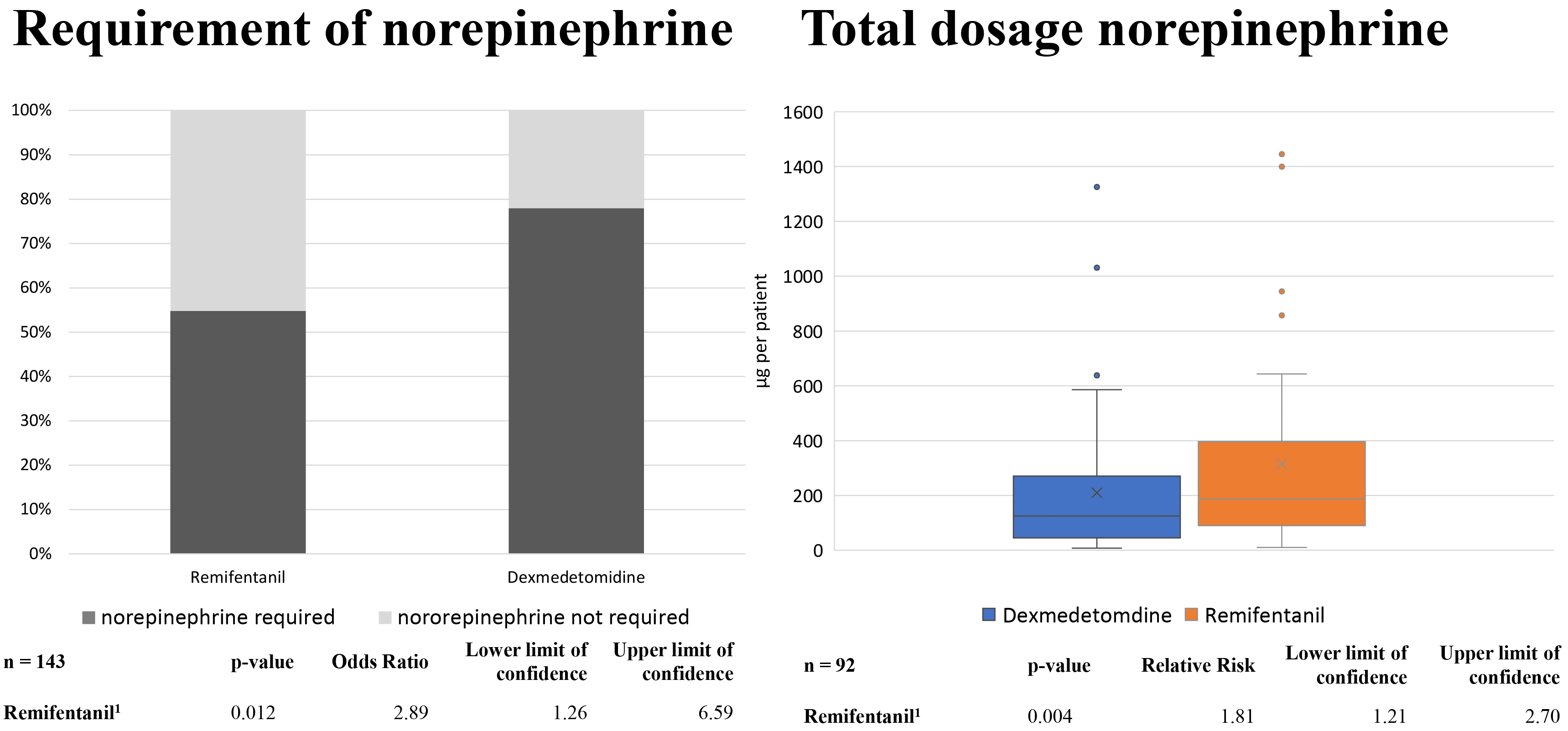 Fig. 2.
Fig. 2.
Requirement of norepinephrine (left panel) and total norepinephrine dose (right panel) during analgosedation. The total dose was only analyzed in patients requiring norepinephrine. 1 adjusted for age, sex, left ventricular ejection fraction, propofol dose, and glomerular filtration rate.
Regarding the delirium assessment, an ICDSC score of 0 was observed in 82% of
patients four hours after the intervention and in 87% of patients on the first
post-intervention day. No statistically significant difference was detected
between the two sedation regimens (RR 0.85; 95% CI 0.33–2.19; p = 0.74
and respectively RR 0.76; 95% CI 0.25–2.27; p = 0.626). Agitation occurred
significantly less frequently in the Remi group (RR 0.51; 95% CI 0.3–0.85;
p = 0.01). In the Remi group, 33% of patients experienced at least one
episode of agitation that disrupted the intervention process. In the Dex group,
this occurred in 69% of patients (see Fig. 3). Significantly more Propofol was
consumed in the Dex group (1.74 mg/kg/h vs. 1.14 mg/kg/h; p
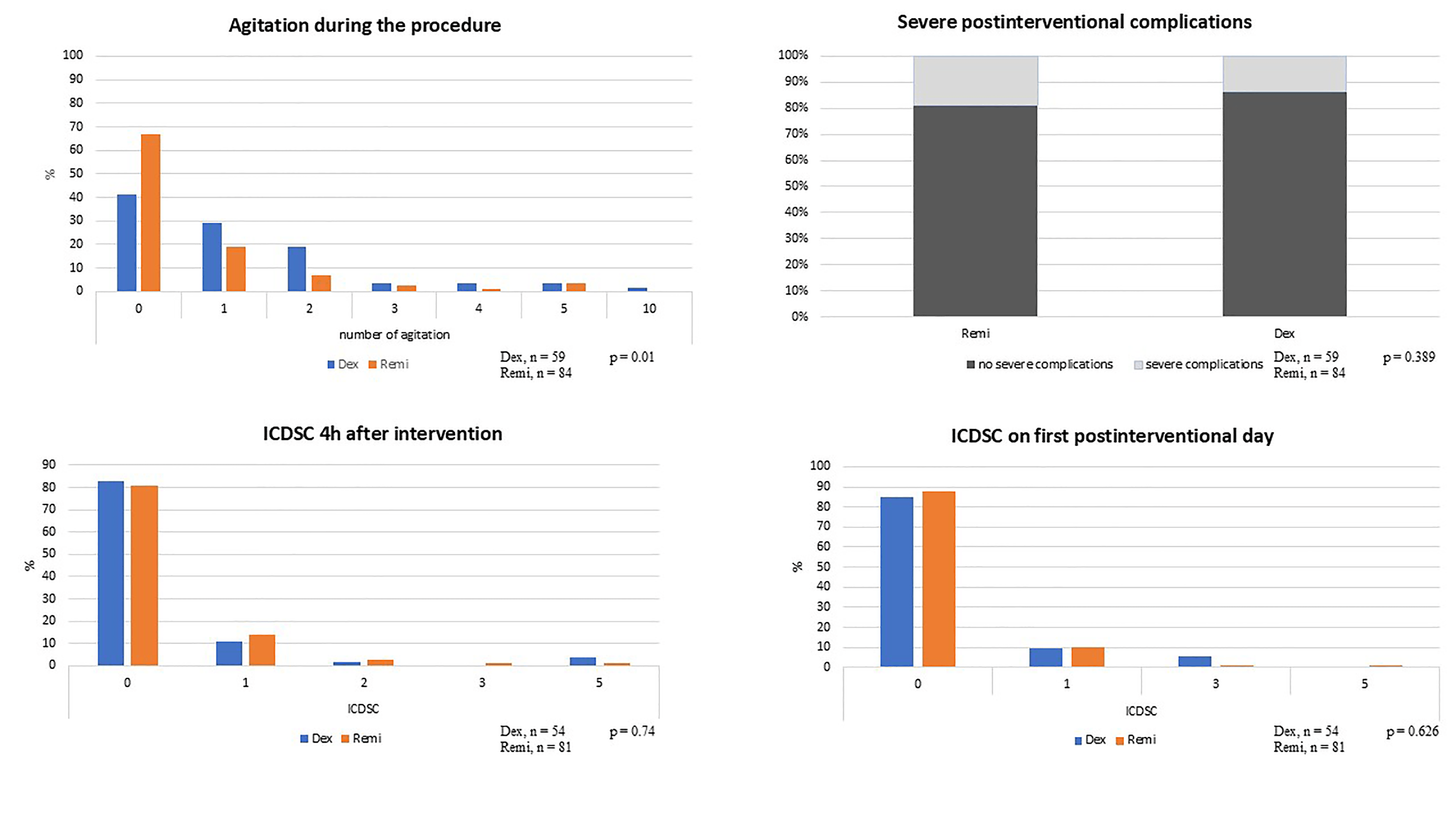 Fig. 3.
Fig. 3.
Secondary endpoints. Shown are the relative proportions of each parameter across the two groups. ICDSC scores with a value of zero were excluded from graphical representation. Abbreviations: Dex, Dexmedetomidine group; Remi, Remifentanil group; h, hours; ICDSC, Intensive Care Delirium Screening Checklist.
No statistically significant differences were detected in the incidence of major post-intervention complications between the Remi group (19.05%) and the Dex group (13.56%) (RR 0.67; 95% CI 0.25–1.64; p = 0.39). Severe adverse events included twelve cases of pacemaker implantation or upgrade, seven strokes, and four vascular complications. In addition, two instances of valve embolization were observed and two patients required blood transfusion of two or more units of red blood cells. One case of cardiopulmonary resuscitation occurred on the first postoperative day. Overall, 28 complications were observed in 24 patients. The two regimens did not differ with respect to length of hospital stay (Remi group vs. Dex group, RR 1.04; 95% CI 0.6–1.67; p = 0.68). Due to changes in perioperative monitoring, analysis of the length of stay in ICU was not feasible.
The overall increase in arterial CO2 partial pressure between the first and second blood gas analyses was 12.16 mmHg. The CO2 increase in the Remi group was 2.35 mmHg (SD 1.39) higher than in the Dex group. This difference was not statistically significant (p = 0.092; see Fig. 4).
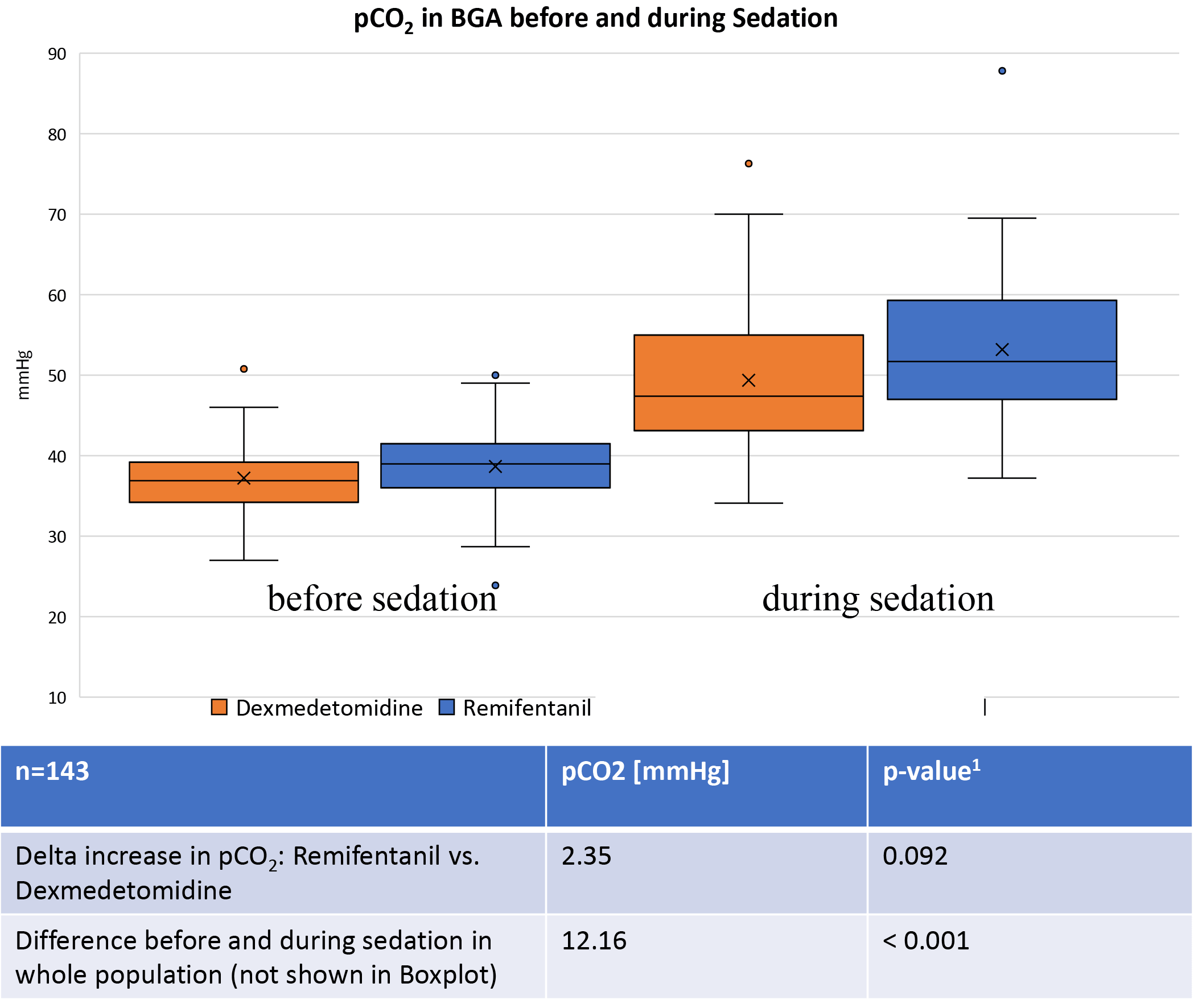 Fig. 4.
Fig. 4.
Comparison of mmHg pCO2 before procedural sedation with the moment of valve placement. Both sedation regimens show a significant rise in the pCO2 during sedation. The delta increase was 2.35 mmHg higher in the Remifentanil group. Abbreviations: BGA, blood gas analysis. 1 adjusted for age, sex, left ventricular ejection fraction, propofol dose and glomerular filtration rate.
This study suggests that both sedation regimens, Propofol-Remifentanil and Propofol-Dexmedetomidine, are safe and effective for use during tf-TAVI, with distinct characteristics that may support individualized anesthetic strategies according to patient-specific risk factors.
The Remifentanil-based sedation regimen was associated with a lower requirement for norepinephrine. However, when norepinephrine was required, the Remifentanil group required higher doses than the Dexmedetomidine group. This difference was of uncertain clinical relevance if expressed as absolute catecholamine dose. Moreover, given the study design, where the primary objective was to maintain a MAP of at least 65 mmHg, the requirement for norepinephrine rather than the administered dose was the more relevant parameter. The use of vasopressors by experienced anesthesiologists in tf-TAVI procedures, which usually involve dynamic blood pressure fluctuations, appears to better reflect impaired circulatory conditions than isolated blood pressure measurements [21]. Nonetheless, evidence suggests that, beyond hypotension, the use of norepinephrine is also associated with an increased risk of post-anesthetic acute kidney injury, making a sedation regimen that minimizes or eliminates its necessity beneficial [22, 23, 24]. In terms of respiratory safety, the Dexmedetomidine-based regimen, demonstrated a more favorable respiratory profile. Patients in the Dex group experienced less CO2 accumulation, and none required intubation due to respiratory insufficiency during the intervention (see Figs. 1,4). These findings did not achieve statistical significance, owing to the low event rate and the limited sample size [25]. Despite the more favorable respiratory outcomes, there was a higher proportion of COPD patients in the Dex group, suggesting potential selection bias by the attending anesthesiologists. Conversely, baseline CO2 levels were higher in the Remi group. COPD was assessed anamnestically without stratification by disease severity, resulting in a heterogeneous subgroup. Chronic hypercapnia, which is typically associated with more severe forms of COPD, may therefore not have been uniformly present [26]. The average increase in CO2 was similar to another publication [27]. Patients with severe respiratory impairment were primarily managed with general anesthesia. Further studies are needed to confirm or refute these results.
Given the well-established benefits of analgosedation over general anesthesia, sedation-related conversion rates play a crucial role in individualized anesthesiological care [7]. The conversion rate in our study was 5% for the whole population with no differences between the two groups (Remi 5.3%; Dex 5.1%). When examining the conversion rates reported in other publications, a comparison between Propofol-Opioid and Dexmedetomidine reveals that conversion to general anesthesia was lower in the Dexmedetomidine group [13]. The primary reason for conversion was hypoventilation. Furthermore, in this analysis, the need for mask-bag ventilation was observed exclusively in the Propofol-Opioid group. When comparing Remifentanil and Propofol as monotherapies, overall conversion rates were similar. However, also in this study, intubation due to hypoventilation was required only in the Remifentanil group [21]. These findings suggest that the combination of Dexmedetomidine and Propofol may represent a safer alternative for patients at risk of respiratory complications, particularly in cases where the preservation of spontaneous breathing is essential.
Nevertheless, it should also be noted that patient induced disruptions during the intervention may necessitate conversion to general anesthesia. The Remifentanil group exhibited less intraoperative agitation, which may be attributable to improved procedural control. However, agitation did not substantially affect major complication rates or procedural duration (mean time: Dex group 142 min (SD 27) Remi group 147 min (SD 36)). Although increased agitation in the Dex group might be expected to confer a disadvantage compared with the Remi group, this was not reflected in conversion rates, neither in the present study nor in previously published reports [13, 21]. Regarding the management of patient-induced disruptions, it is notable that, in contrast to the other studies mentioned, rescue therapy was exclusively performed with propofol. In previous studies on sedation during tf-TAVI, rescue therapy was required in up to 16% of interventions, with various medications being used. Conversion to a surgical valve replacement in the investigated interventions was not necessary in any included case.
Both sedation regimens were associated with comparable post-interventional outcomes, with no significant differences in complication rates, length of hospital stay, or overall delirium incidence. Accordingly, a protective effect of dexmedetomidine on postoperative delirium, as reported in several randomized trials, could not be confirmed in the present tf-TAVI cohort [14]. This contrasts with recent high-quality randomized controlled data demonstrating a significant reduction in postoperative delirium with dexmedetomidine compared with propofol in older patients. Low ICDSC scores aligned with previous findings, but assessment timing prevented conclusions on late-onset delirium. However, meta-analyses have highlighted substantial heterogeneity across studies, suggesting that the delirium-modulating effects of dexmedetomidine are highly context-dependent and influenced by patient population, procedural characteristics, and perioperative management [14, 28]. Importantly, delirium after tf-TAVI often manifests as hypoactive delirium, which may be underrecognized. In the present study, low ICDSC scores were observed in both groups. However, the timing of delirium assessments precluded conclusions regarding late-onset delirium. Given that delirium in tf-TAVI patients is driven by multiple interacting factors—including advanced age, frailty, cerebrovascular vulnerability, hemodynamic fluctuations, and procedural stress—the absence of a measurable protective effect of dexmedetomidine in this setting likely reflects the multifactorial pathophysiology of delirium rather than a lack of pharmacological efficacy [29, 30, 31].
A major strength of this study is the prospective collection of data within routine clinical practice. However, this approach also introduced a limitation, as a number of patients had to be excluded because of organizational constraints. Consequently, the patient recruitment period had to be extended to achieve the prespecified statistical power. Although group sizes varied since the attending anesthesiologist selected sedation methods based on their clinical experience with each regimen, a sensitivity analysis using Propensity Score Matching was conducted, yielding consistent results. However, this non-randomized allocation based on individual clinical judgment may have introduced a selection bias. A randomized study design was intentionally avoided for feasibility reasons.
The study was a monocentric investigation owing to hospital-specific procedural workflows. However, precise procedural data, such as the duration of rapid pacing, were not recorded. Additionally, changes in anesthetic management occurred during the study period. These were limited to modifications in monitoring protocols and central venous catheter insertion sites. While the shift in puncture approach from the internal jugular to the femoral vein is unlikely to impact procedural duration, no catheter-associated complications were reported [32]. As a result of changes in peri-interventional monitoring practices, it was not possible to include the length of ICU stay in this study.
Most therapeutic decisions were based on institution-specific standard operating procedures and the study protocol. Arterial blood gas analyses and ICDSC assessments were performed at predefined timepoints and sedation depth was monitored using the RASS which demonstrates a good interrater reliability [33]. Continuous MAP monitoring was not feasible due to the introduction of an electronic data management system during the study period. As outlined above, vasopressor use has been considered a marker of circulatory impairment in tf-TAVI [21].
This monocentric, non-randomized study is subject to limited internal and external validity. Sedation strategy was selected by the attending anesthesiologist, introducing potential selection bias despite consistent results in the propensity score–matched sensitivity analysis. Organizational constraints led to patient exclusions and prolonged recruitment, with possible temporal effects. Relevant procedural variables (e.g., rapid pacing duration, continuous MAP monitoring) were not consistently recorded, and changes in anesthetic management during the study period limited data completeness, including the inability to assess ICU length of stay. Although methodological measures were implemented to mitigate bias, residual confounding cannot be fully excluded.
In this study, both Remifentanil–Propofol and Dexmedetomidine–Propofol provided comparable overall clinical outcomes in patients undergoing tf-TAVI. However, the observed differences in hemodynamic and respiratory profiles highlight the importance of an individualized sedation strategy. Remifentanil–propofol may be advantageous in patients with hemodynamic vulnerability, whereas dexmedetomidine–propofol appears to offer a safer alternative for patients at increased risk of respiratory compromise. These findings underscore that sedation during tf-TAVI should be tailored to patient-specific risk factors rather than applying a uniform approach. Further prospective studies in broader and more heterogeneous populations are warranted to optimize analgosedation protocols and improve the anticipation and management of sedation-related side effects.
95% CI, 95% confidence interval; ASA, American Society of Anesthesiologists; AV, atriventricular; AVB, atrioventricular block; AVA, aortic valve opening area; BMI, body mass index; COPD, chronic obstructive pulmonary disease; CVC, central venous catheter; DM, diabetes mellitus; EF, ejection fraction; FVC, functional vital capacity; GFR, glomerular filtration rate; GOLD, Global Initiative for Chronic Obstructive Lung Disease; ICDSC, Intensive Care Delirium Screening Checklist; ICU, intensive care unit; IMC, intermediate care unit; MAP, mean arterial pressure; No, number; OR, odds ratio; SD, standard deviation; RR, relative risk; sPAP, systolic pulmonal arterial pressure; tf-TAVI, transfemoral transcatheter aortic valve implantation.
All data reported in this paper will also be shared by the lead contact upon request.
TM-J and TK designed the research study. ETM, TM-J, CW and MvdH performed the research. AFP provided cardiosurgical insight for conception and design of the work and significantly contributed to the analysis and interpretation of clinically relevant data in this domain. ETM, MvdH and TM-J analyzed the data. ETM wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Declaration of Helsinki. The research protocol was approved by the Ethics Committee of the ‘Ärztekammer Hamburg’ (Ethic Approval Number: 2021-100636-BO-ff), and all of the participants provided signed informed consent. Ethical approval was granted on August 31, 2021.
The authors would like to express their sincere gratitude to all cardiac anesthesiologists at Asklepios Klinikum Harburg for their invaluable support and collaboration during the study period.
This research received no external funding, but was supported by Asklepios proresearch.
The authors declare no conflict of interest. ETM, MvdH, TM-J, CW, AFP, and TK are employees of Asklepios Medical School GmbH or have a financial relationship with the company. However, the company had no role in the handling or conduct of the study. The authors had full access to all data in the study and take full responsibility for the integrity of the data and the accuracy of the data analysis.
During the preparation of this work the authors used ChatGpt-3.5 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




