1 Division of Cardiology, Cardiovascular Center, Showa Medical University Koto-Toyosu Hospital, 114-0024 Tokyo, Japan
Abstract
Transcatheter cardiac surgery (TCS), which primarily comprises transcatheter aortic valve replacement (TAVR) and mitral transcatheter edge-to-edge repair (M-TEER), has transformed the treatment of valvular heart disease over the past two decades. Moreover, TAVR is now supported by robust randomized trial evidence across the surgical risk spectrum, establishing this technique as a cornerstone therapy for aortic stenosis. Moreover, M-TEER is gaining clinical relevance, with expanding registry and trial data further defining the role of this technique. Meanwhile, advanced imaging has become central to both TAVR and M-TEER, extending beyond diagnosis to patient selection, procedural planning, and risk assessment of complications. Furthermore, advanced imaging enhances procedural safety and improves short- and mid-term clinical outcomes by enabling accurate anatomical characterization, precise device sizing, and early detection of complications such as paravalvular leak or leaflet thrombosis. Echocardiography and computed tomography form the backbone of the preprocedural evaluations, whereas cardiac magnetic resonance and positron emission tomography provide complementary insights into myocardial pathology and prosthetic valve dysfunction. Imaging enables structured surveillance for paravalvular leak, leaflet thrombosis, recurrent regurgitation, and structural valve degeneration, all of which directly affect outcomes. However, despite considerable progress, important challenges persist, including limited evidence on the long-term durability of TAVR, a lack of standardized grading of residual mitral regurgitation after M-TEER, and the need to integrate right heart–pulmonary circulation assessments into decision-making. Recent innovations such as quantitative three-dimensional echocardiography, fusion imaging, and artificial-intelligence-based image analysis are expected to refine procedural planning further, reduce operator variability, and enable more predictive, patient-specific management. Nonetheless, multimodality imaging is slated to remain the cornerstone for lifetime management strategies in TCS.
Keywords
- transcatheter cardiac surgery
- imaging
- transcatheter aortic valve replacement
- mitral transcatheter edge-to-edge repair
Over the past two decades, transcatheter cardiac surgery (TCS) has undergone remarkable evolution, transforming the management of valvular heart disease. Among these procedures, transcatheter aortic valve replacement (TAVR) is the most extensively studied. Multiple landmark randomized controlled trials have demonstrated the noninferiority of TAVR to surgical aortic valve replacement (SAVR) in high- and intermediate-risk populations, as well as its superiority in selected low-risk cohorts [1, 2, 3, 4, 5, 6, 7, 8]. These results have led to broad guideline endorsement of TAVR across the surgical risk spectrum, establishing it as a cornerstone therapy in managing aortic stenosis (AS). In contrast, transcatheter interventions for the mitral valve remain at an earlier stage of evidence development. For treating mitral regurgitation (MR), the EVEREST II trial compared mitral transcatheter edge-to-edge repair (M-TEER) with surgery [9], while The Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy (COAPT) and MITRA-FR trials produced divergent findings regarding the efficacy of M-TEER in secondary MR, underscoring the importance of patient selection [10, 11]. Taken together, these differences highlight that while TAVR has achieved clinical maturity, supported by robust validation, mitral interventions are still in an early phase of clinical adoption, with ongoing trials expected to shape their future role.
In this review, we focus on TAVR and M-TEER, the two TCS interventions with the strongest clinical evidence and broadest adoption. Other emerging techniques such as transcatheter mitral valve replacement (TMVR) are discussed only briefly in the context of future perspectives. By narrowing the scope of our discussion, we provide a focused appraisal of technological advances, clinical trial data, and challenges, with emphasis on the evolving role of multimodality imaging. To ensure a comprehensive and balanced overview, we primarily cited landmark randomized controlled trials published between 2010 and 2024, major international guidelines, and review articles from high-impact journals. Beyond anatomical characterization, imaging now guides patient selection, procedural planning, and complication prediction; imaging therefore shapes both short- and long-term outcomes. Additionally, we highlight the complementary role of cardiac surgery in patient selection, complication management, and hybrid strategies, reinforcing the concept that surgical and transcatheter therapies are not competing but complementary modalities in managing valvular heart disease.
Balloon aortic valvuloplasty (BAV), introduced in the late 1980s, was the first catheter-based therapy for severe AS [12]. Although it provided temporary hemodynamic improvement, restenosis occurred in over 50% of patients within one year, limiting its long-term efficacy [13]. Nevertheless, BAV laid the technical foundation for TAVR, which was first performed in humans in 2002 [1].
Initial randomized controlled trials, including PARTNER 1 and CoreValve High Risk, demonstrated the noninferiority of TAVR compared with SAVR in patients with an inoperable condition and those at high risk [2, 3, 14]. Subsequent trials (PARTNER 2A, SURTAVI) expanded the indications to intermediate-risk populations [4, 5], while PARTNER 3 and Evolut Low Risk confirmed excellent outcomes in selected low-risk cohorts, with one-year mortality and stroke rates as low as 1–2% [6, 7]. These results led to guideline endorsement of TAVR across all surgical risk categories and shifted decision-making toward anatomical suitability, age, and life expectancy.
The currently available devices are broadly categorized into balloon-expandable valves (BEVs) and self-expanding valves (SEVs). BEVs are associated with lower rates of paravalvular leak (PVL) and permanent pacemaker implantation (PPI), while SEVs provide larger effective orifice areas and lower transvalvular gradients [15, 16, 17]. Device selection requires careful assessment of annular dimensions, coronary ostial height, calcium distribution, vascular access, and the feasibility of future coronary re-access, primarily using computed tomography (CT) [18].
The stepwise imaging workflow for TAVR is summarized in Fig. 1 before detailing modality-specific roles.
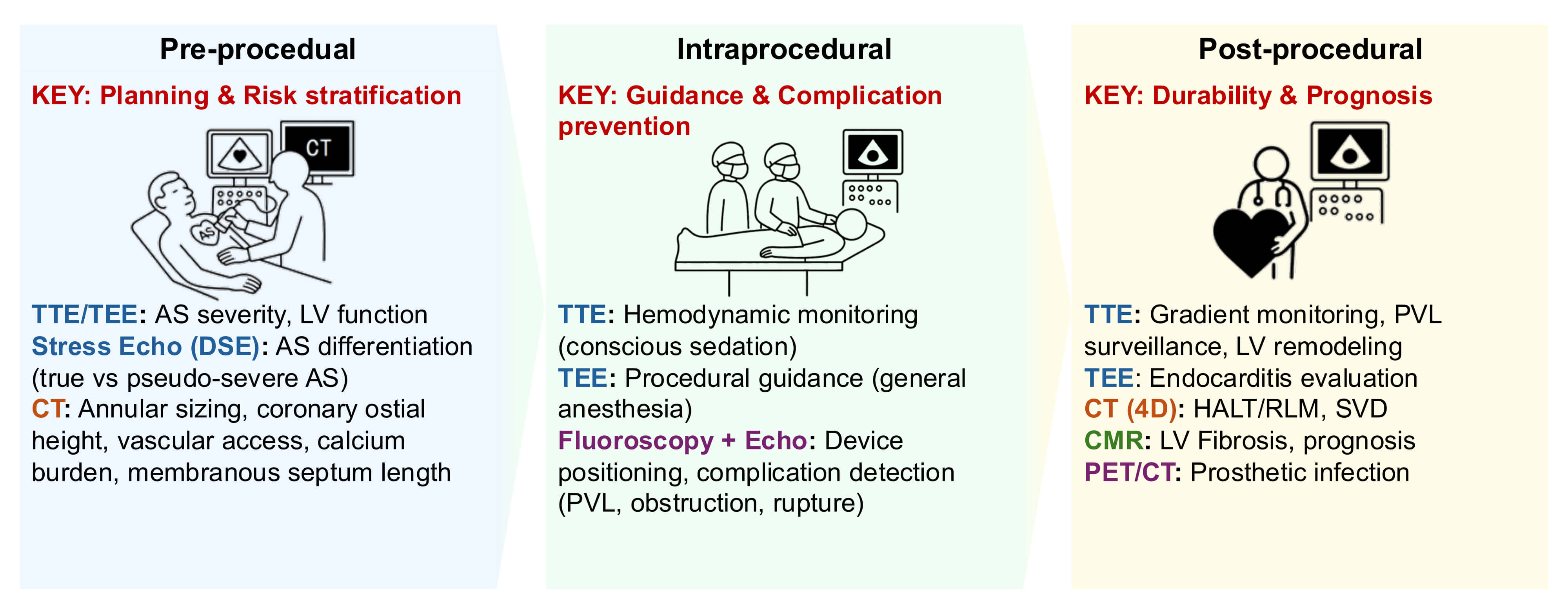 Fig. 1.
Fig. 1.
Imaging workflow for transcatheter aortic valve replacement (TAVR). Multimodality imaging guides every procedural phase, from preprocedural CT-based annular sizing and vascular access assessment, to intraprocedural TEE (under general anesthesia) or TTE (under conscious sedation) guidance, as well as postprocedural surveillance for paravalvular leak (PVL) or leaflet thrombosis. Abbreviations: TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; DSE, dobutamine stress echocardiography; CT, computed tomography; PVL, paravalvular leak; CMR, cardiac magnetic resonance; PET, positron emission tomography. Key takeaway: Accurate preprocedural CT planning and real-time echocardiographic guidance are critical for procedural safety and optimal valve function.
The roles of different imaging modalities across the TAVR continuum are summarized in Table 1.
| Imaging modality | Preprocedural planning | Intraprocedural guidance | Postprocedural follow-up |
| TTE | TAVR | ||
| Screening for AS severity; assessment of LV function and remodeling | Used during conscious sedation or monitored anesthesia care TAVR for immediate hemodynamic and valve function evaluation; however comprehensive PVL grading and complication screening are more accurate with TEE under general anesthesia | First-line modality for PVL surveillance, LV remodeling, prosthetic valve gradients, prosthetic valve thrombosis suspicion | |
| M-TEER | |||
| Screening for MR severity; LV function and remodeling assessment | Not primary | Evaluation of MR recurrence, LV remodeling, and prosthetic valve gradients | |
| STE (DSE/Exercise) | TAVR (DSE) | ||
| To differentiate between true and pseudo-severe AS (assessment of contractile reserve) | Not routinely used | Not routinely used | |
| M-TEER (Exercise) | |||
| Assessing MR dynamic (exercise-induced PH), optimal timing for intervention | Not routinely used | Not routinely used | |
| TEE | TAVR | ||
| Optional for TAVR under general anesthesia | Primarily used during TAVR under general anesthesia; enables real-time valve deployment guidance, PVL assessment, and detection of procedural complications | Evaluation of prosthetic valve endocarditis | |
| M-TEER | |||
| Mechanistic assessment of MR (primary vs. secondary); evaluation of anatomical suitability for M-TEER | Essential for M-TEER — device navigation, leaflet grasping, and residual MR | Quantification of residual MR; evaluation of prosthetic valve endocarditis | |
| ICE | TAVR and M-TEER | ||
| Not routinely used; alternative to TEE if it is contraindicated (e.g., esophageal disease) | Emerging role as an alternative to TEE for intraprocedural guidance | Limited postprocedural use | |
| CT, 3D/4D | TAVR | ||
| Gold standard for annular sizing, coronary ostial height, vascular access route, and calcium quantification; quantification of aortic valve calcium score for differentiation of true vs. pseudo-severe AS | Adjunct to fluoroscopy (fusion imaging, experimental use) | HALT/RLM detection; prosthetic degeneration; evaluation of IE complications and coronary access planning for redo-TAVR | |
| M-TEER | |||
| Comprehensive assessment of mitral annular dimensions and nonplanarity, subvalvular anatomy including chordae and papillary muscles, and neo-LVOT area prediction through 3D reconstruction | Adjunct to fluoroscopy (fusion imaging, experimental use) | Not routinely used | |
| CMR | TAVR and M-TEER | ||
| Assessment of LV/RV function and myocardial fibrosis (LGE, ECV) | Not used intraprocedurally | Prognostic evaluation of fibrosis burden and ventricular remodeling | |
| M-TEER | |||
| MR quantification | |||
| PET/CT | TAVR and M-TEER | ||
| Not routinely used | Not used intraprocedurally | High sensitivity for detection of prosthetic valve endocarditis and perivalvular infection | |
Abbreviations: TAVR, transcatheter aortic valve replacement; M-TEER, mitral transcatheter edge-to-edge repair; TTE, transthoracic echocardiography; STE, stress echocardiography; DSE, dobutamine stress echocardiography; TEE, transesophageal echocardiography; ICE, intracardiac echocardiography; CT computed tomography; 3D, three-dimensional; 4D, four-dimensional; CMR, cardiac magnetic resonance; PET, positron emission tomography; AS, aortic stenosis; LV, left ventricular; PVL, paravalvular leak; MR, mitral regurgitation; PH, pulmonary hypertension; HALT, hypo-attenuated leaflet thickening; RLM, reduced leaflet motion; IE, infective endocarditis; LVOT, left ventricular outflow tract; RV, right ventricular; LGE, late gadolinium enhancement; ECV, extracellular volume.
Multimodality imaging plays a central role in TAVR planning, guiding indications, access strategy, and device selection.
Transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE)
are fundamental for confirming severe AS, defined using an aortic valve area
(AVA)
| Parameter | Thresholds/Criteria | Context | Reference imaging modality |
| AS | AVA |
Definition of severe AS | TTE/TEE |
| Low-flow, low-gradient AS | DSE → ΔSV |
Differentiation between true and pseudo-severe AS | Stress Echo (DSE) |
| CT calcium scoring | Agatston score: |
Supportive criterion for diagnosing true severe AS in low-flow states | CT |
Abbreviations: AS, aortic stenosis; CT, computed tomography; AVA, aortic valve area; PG, pressure gradient; TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; DSE, dobutamine stress echocardiography; SV, stroke volume.
CT is the gold standard for vascular access assessment [18]. A minimum
iliofemoral diameter of 5.5–6.0 mm is generally required, with attention to
calcification and tortuosity [26]. In patients unsuitable for transfemoral
access, CT also guides consideration of alternative routes. For subclavian (or
axillary) access, vessel diameters of at least 5.5–6.0 mm are generally
required, while
Thus, multimodality imaging not only determines the feasibility of transfemoral access but also provides route-specific information essential to the safe performance of alternative access strategies. Intravascular lithotripsy (IVL) has recently emerged as a valuable adjunct in heavily calcified iliofemoral disease. IVL can expand the feasibility of transfemoral TAVR by modifying severely calcified access vessels. Although patients undergoing IVL-assisted TAVR generally present with higher baseline risk, registry and nationwide data demonstrate high procedural success and acceptable short-term outcomes [32].
Device selection relies on CT measurements of annular dimensions, the sinus of Valsalva, and coronary ostial height to determine the appropriate device type and size [18]. Predictable complication risks, such as PVL, conduction disturbances requiring PPI, and coronary obstruction, should be considered when selecting the device, as is discussed in subsequent sections. This section focuses on common imaging-based criteria, including CT-derived annular dimensions and anatomical risk factors; therefore, device-specific comparisons are not included.
Despite technological advances, TAVR remains associated with specific complications. Imaging plays a central role in risk stratification, early detection, and prevention of these events. During TAVR performed under general anesthesia, intraoperative TEE is indispensable for confirming prosthesis positioning, assessing immediate valve function, and detecting early complications such as PVL, pericardial effusion, or annular injury. In contrast, in procedures performed under conscious sedation, TTE is typically used for rapid hemodynamic assessment, although its diagnostic resolution for subtle complications is limited compared with TEE (Table 3).
| Complication | Imaging marker(s) | Modality | Clinical implication |
| PVL | Annular ellipticity, asymmetric LVOT/annular calcium ( |
CT, TTE/TEE | Predictor of malapposition; associated with increased HF hospitalization and mortality |
| Coronary obstruction | Low coronary ostial height ( |
CT | High early mortality ( |
| Annular rupture | Localized annular calcium, ellipticity, prosthesis oversizing | CT | Rare but catastrophic; influences device type (BEV vs SEV) and sizing |
| Conduction disturbances/PPI | Short membranous-septum length ( |
CT | Higher PPI rate with SEV; preprocedural CT can guide implantation depth |
| Stroke | Aortic-arch atheroma, bulky valve calcium, embolic debris | CT, TEE, MRI (DWI) | Occurs in 1–2% of cases; related to atheroma or debris; selective cerebral protection may reduce lesion burden |
Abbreviations: PVL, paravalvular leak; PPI, permanent pacemaker implantation; LVOT, left ventricular outflow tract; CT, computed tomography; TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; HF, heart failure; BEV, balloon-expandable valve; SEV, self-expandable valve; MRI, magnetic resonance imaging; DWI, diffusion-weighted imaging.
Although rare (0.5–1%), coronary obstruction is associated with a high risk of
early mortality (
In TAV-in-TAV, the incidence of coronary obstruction has been lower than
initially anticipated, likely because of rigorous screening and careful patient
selection. A distinctive concern is the formation of a neo-skirt, created by the
displaced host transcatheter heart valve (THV) leaflets against the new frame; if
this skirt extends above the sinotubular junction (STJ), it may cause sinus
sequestration. The height of the neo-skirt depends on host leaflet length, the
type of the index THV, and the implantation depth of the new valve. In addition
to VTC, CT-based measurement of the virtual THV-to-STJ distance has been proposed
for risk stratification, with
Although the incidence of moderate or severe PVL has markedly declined with
newer-generation valves, it remains strongly associated with increased mortality
and heart failure hospitalization [6, 39, 40]. Preprocedural CT plays a
preventive role by identifying anatomic risk factors such as annular ellipticity,
high annular or LV outflow tract (LVOT) calcium burden (
Eccentric calcification and annular ellipticity increase the likelihood of annular rupture, especially in the setting of TAVR with BEVs, where excessive prosthesis oversizing, heavy and asymmetric calcium burden, and elliptical root geometry are major contributors [49, 50, 51, 52]. Preprocedural multidetector CT should evaluate not only global calcium burden but also the regional distribution and protrusion of subannular/LVOT nodules—especially beneath the noncoronary cusp—which, together with excessive BEV oversizing, markedly increases the risk of annular injury [18, 53]. Emerging regional calcium metrics and patient-specific simulations may further refine rupture-risk estimation and inform strategies such as cautious balloon underfilling or device downsizing [54].
The need for PPI remains higher with SEVs (10–15%) than with BEVs (5–8%)
[55, 56]. A short membranous septum length (
Stroke occurs in 1–2% of cases [60]. Predictors include heavy valve calcification and aortic-arch atheroma [61]. Diffusion-weighted magnetic resonance imaging frequently detects silent embolic lesions [62], and the use of cerebral embolic-protection devices can reduce total lesion burden. However, consistent improvements in clinical outcomes have not yet been demonstrated [63].
Summary of imaging-based strategies to minimize procedural complications:
• Coronary obstruction: Perform detailed CT analysis of coronary height, sinus
width, cusp length, and leaflet calcium volume. Calculate both virtual VTC and
virtual THV-to-STJ distances, and use virtual-valve simulation to visualize
potential obstruction. • PVL: Evaluate annular ellipticity and LVOT/annular calcium distribution using
CT. During implantation, confirm valve expansion and sealing with real-time
TEE/TTE, and grade residual PVL to guide post-dilatation or plug placement. • Annular rupture: Assess both global and regional calcium burden and subannular
protrusions on CT, particularly beneath the noncoronary cusp. Avoid • Conduction disturbances: Measure membranous septum length and implantation depth
on CT to predict conduction risk. For short septum ( • Stroke: Evaluate aortic-arch atheroma and bulky valve calcium on CT or TEE and
employ cerebral protection selectively in high-risk anatomy.
Structured imaging follow-up is essential for early detection of complications and monitoring long-term outcomes after TAVR. Each imaging modality has specific strengths, and a multimodality approach provides the most comprehensive assessment of prosthetic valve function and cardiac remodeling.
Follow-up focuses on using structured TTE to quantify PVL severity and its hemodynamic impact; cardiac CT is reserved for delineating mechanisms (e.g., malapposition, calcification bridging) when echocardiography is inconclusive.
PPM is defined as an indexed effective orifice area (EOA) that is too small
relative to the patient’s body surface area, resulting in higher-than-expected
transvalvular gradients despite normally functioning prosthetic leaflets.
According to established criteria, PPM is classified as moderate when the indexed
EOA is
Subclinical leaflet thrombosis after bioprosthetic valve implantation is most often detected using 4D-CT as HALT; it histologically corresponds to fibrin or thrombus deposition on valve leaflets. This structural change may result in RLM, a functional correlation that can lead to increased transvalvular gradients or, less frequently, thromboembolic complications [71]. HALT is observed in 10–40% of patients after TAVR and is often reversible with anticoagulation, although its management remains controversial because of bleeding risks [72]. Similar findings have been described after SAVR, but at lower frequencies and with a less well-defined clinical impact than that seen after TAVR [73].
IE after TAVR occurs at an annual incidence of 0.3–1.2%, similar to that observed after SAVR [74]. However, differences exist in pathogen distribution, therapeutic options, and prognosis [75]. TAVR-associated IE is particularly difficult to treat surgically and is associated with higher mortality [76]. Therefore, early and accurate imaging is essential. Although TTE has limited sensitivity, TEE, contrast-enhanced CT, and PET/CT provide complementary value for detecting prosthetic complications, highlighting the importance of multimodality imaging in this setting.
Long-term durability remains a central concern. SVD manifests as leaflet thickening, calcification, or stent-frame deformation. CT enables early recognition of these changes, facilitating timely decision-making regarding reintervention, including repeat TAVR or surgery. Compared with SAVR, long-term data beyond 10 years for TAVR remain limited. Some studies suggest higher rates of subclinical degeneration in transcatheter valves, whereas surgical bioprostheses benefit from longer follow-up experience [77]. Therefore, ongoing multimodality imaging surveillance is critical to clarify durability differences and guide optimal reintervention strategies.
The reduction in afterload achieved with TAVR directly leads to improvements in LV function [78]. During follow-up, assessing sensitive parameters such as global longitudinal strain (GLS) and LV mass index (LVMI) is important, in addition to assessing conventional LV ejection fraction (LVEF). Echocardiography, owing to its simplicity, noninvasiveness, and feasibility for repeated assessments, is the most practical modality for monitoring longitudinal changes in GLS and LVMI. Patients who demonstrate improvement in these parameters after TAVR generally have better long-term outcomes, whereas those with limited improvement often have residual fibrosis or diastolic dysfunction, which are associated with an increased risk of heart failure and mortality [79, 80]. Moreover, reduced preprocedural GLS and the presence of myocardial fibrosis on CMR, assessed using late gadolinium enhancement (LGE) or elevated extracellular volume, are independent predictors of adverse outcomes. These parameters provide valuable information for risk stratification and the likelihood of functional recovery [81, 82]. In addition, positron emission tomography (PET) has emerged as a complementary modality for assessing myocardial viability and metabolic activity, particularly in patients with coexisting coronary artery disease. These applications may enhance prognostic risk stratification and inform postprocedural management, although evidence in TAVR-specific populations remains limited [83]. Therefore, comprehensive follow-up of LV function is essential for prognostication and risk assessment.
With the expansion of TAVR indications to younger and lower-risk populations, long-term durability has emerged as the foremost challenge. Evidence beyond 10 years remains limited, and surgical explantation after TAVR carries substantial perioperative risk, with reported mortality rates of 10–15% [84, 85, 86]. Beyond durability concerns and subclinical leaflet thrombosis (HALT/RLM), discussed in Sections 2.4.3 and 2.4.5, long-term management requires systematic imaging surveillance and individualized antithrombotic strategies tailored to each patient’s risk profile. Standardized definitions of SVD and consensus on imaging-based follow-up intervals will be essential to ensure consistent assessment of valve durability in the coming decade.
Beyond these durability-related issues, lifetime management has emerged as a central concept, particularly for younger patients with a longer life expectancy [87, 88]. Imaging-guided procedural planning is therefore critical not only for selecting the optimal device and access route but also for anticipating the feasibility of future reinterventions, such as valve-in-valve TAVR or surgical explantation. Furthermore, bicuspid aortic valves remain a distinct challenge because of their heterogeneous anatomy, underscoring the importance of careful patient selection and tailored procedural strategies [89, 90]. Comprehensive lifetime planning, supported by CT-based modeling and risk stratification, will help preserve reintervention options and optimize outcomes throughout the patient’s treatment continuum.
Recent advances in quantitative 3D echocardiography, fusion imaging, and
artificial intelligence (AI) are reshaping procedural planning and surveillance
in TAVR. AI-based segmentation and automated annular measurements achieve
In the coming years, integrating multimodality imaging into standardized lifetime management algorithms encompassing procedural planning, risk stratification, structured follow-up, and neuro-protection strategies will be essential to ensure long-term valve durability and optimize outcomes across decades of patient life expectancy. Nevertheless, major uncertainties persist regarding valve durability beyond 10 years, the optimal antithrombotic strategy for subclinical leaflet thrombosis (HALT/RLM), and the role of cerebral embolic protection during transcatheter valve implantation. Ongoing clinical trials in these domains are anticipated to clarify their clinical impact such as the PROTECTED TAVR study for stroke prevention [83]. Future advancement will depend on integrating emerging imaging tools, clinical data, and multidisciplinary collaboration to achieve truly personalized and durable transcatheter valve therapy.
While TAVR has established the paradigm for transcatheter valve replacement, its success has also accelerated the development of repair-based interventions for the mitral valve. Many of the imaging concepts refined in TAVR—such as CT-based annular sizing and echocardiographic guidance—have been adapted to M-TEER. The following section highlights the distinct imaging challenges and evolving role of multimodality imaging in transcatheter mitral repair.
The mitral valve poses unique anatomical and functional challenges for transcatheter intervention because of its saddle-shaped annulus and subvalvular apparatus. The M-TEER technique, inspired by the surgical Alfieri stitch [94], was first performed in humans in 2003 and commercially introduced in 2008 with the MitraClip system [95, 96]. Initially reserved for patients at high risk or those with an inoperable condition with severe MR, M-TEER rapidly gained acceptance; this progress is supported by registry data demonstrating symptomatic improvement and reduced heart failure hospitalizations [97].
Randomized evidence has further influenced the role of M-TEER. The EVEREST II trial compared M-TEER with surgery in patients with primary MR, showing superior safety, but higher rates of residual MR [9]. More recently, the COAPT and MITRA-FR trials assessed M-TEER in secondary MR, yielding divergent outcomes that underscored the importance of careful patient selection and optimization of medical therapy [10, 11]. Current guidelines now recommend M-TEER for symptomatic patients with severe primary MR at prohibitive surgical risk and for selected patients with secondary MR who remain symptomatic despite guideline-directed medical therapy [19, 20, 21].
Device technology has also evolved. The MitraClip platform has expanded to include multiple clip sizes and configurations, enabling treatment of complex anatomies and multijet regurgitation [98, 99]. In addition, the PASCAL system, incorporating features such as a central spacer and independent leaflet grasping, has emerged as an alternative; early studies have demonstrated comparable safety and efficacy [98, 100]. These advances in device platforms, together with growing clinical evidence, continue to broaden the applicability of M-TEER across diverse populations with MR.
To provide an overview of the imaging workflow in M-TEER, we have summarized the stepwise process in Fig. 2 before detailing modality-specific roles.
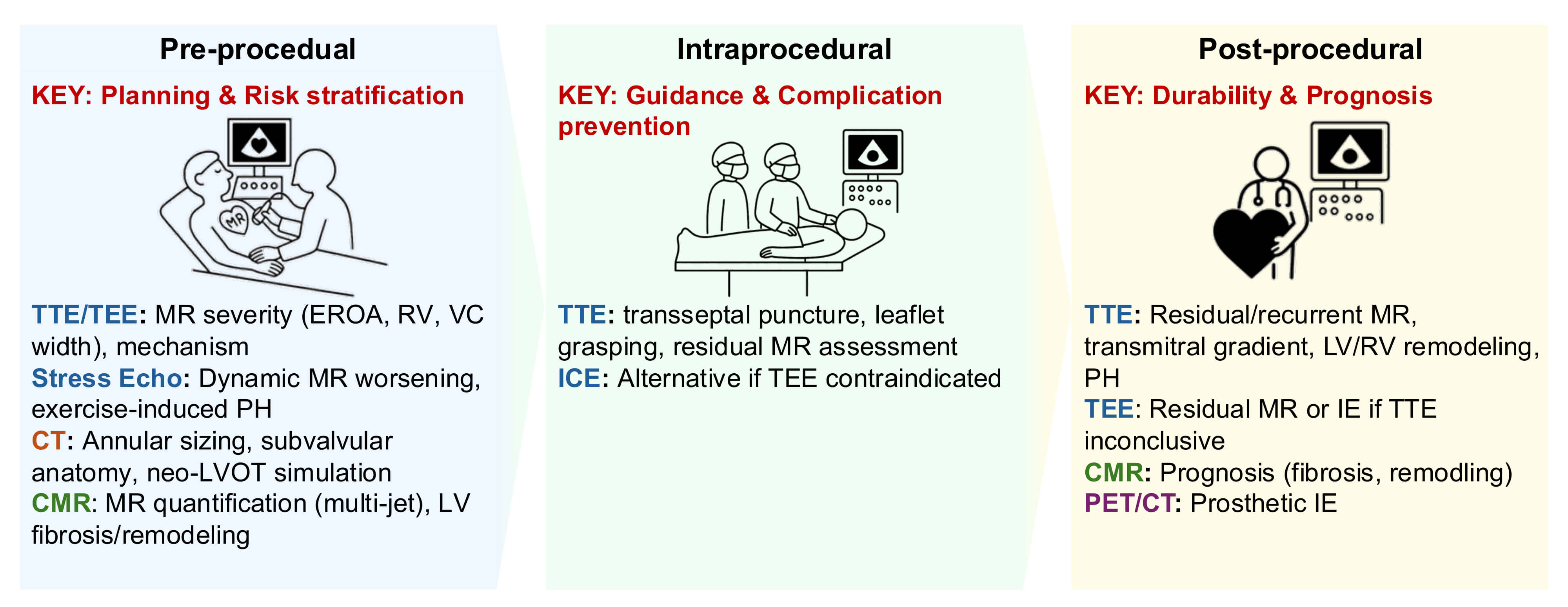 Fig. 2.
Fig. 2.
Imaging workflow for M-TEER. Echocardiographic and CT integration enables patient selection, device orientation, and assessment of leaflet grasping and residual mitral regurgitation. Intraprocedural TEE provides real-time spatial guidance. Abbreviations: TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; MR, mitral regurgitation; CT, computed tomography; CMR, cardiac magnetic resonance; ICE, intracardiac echocardiography; PET, positron emission tomography; LV, left ventricle; RV, right ventricle; IE, infective endocarditis. Key takeaway: Comprehensive multimodality imaging, particularly TEE, enhances clip positioning accuracy and ensures durable MR reduction.
The imaging markers for risk stratification in M-TEER–related complications are summarized in Table 4.
| Complication | Imaging marker(s) | Modality | Clinical implication |
| Residual MR | Multiple eccentric or wall-impinging jets, high transmitral gradient, pulmonary vein flow reversal | TTE/TEE, CMR (if echo inconclusive) | Prediction of adverse outcomes (mortality, HF hospitalization) |
| LAE (SLDA, tear, embolization) | Incomplete leaflet grasp ( |
CT (preprocedural), TTE/TEE | Major cause of recurrent MR and reintervention; early detection enables re-grasping or surgical retrieval |
| Iatrogenic mitral stenosis | Mean gradient |
CT (preprocedural), TTE/TEE | Association with persistent symptoms and HF rehospitalization |
| iASD | Persistent color Doppler shunt; bubble study | TTE/TEE | Worsening of symptoms in PH/RV dysfunction; indication for closure if clinically significant |
| LVOT obstruction | Small LV cavity, basal septal hypertrophy, aortomitral angulation | CT (preprocedural), TTE/TEE | Rare complication ( |
| Cardiac tamponade/hemopericardium | Pericardial effusion during transseptal puncture or catheter manipulation | TTE/TEE | Life-threatening event (0.5–1%); requires immediate pericardiocentesis |
| Stroke/systemic embolism | Pre-existing LA and LAA thrombus, aortic arch atheroma, air, or device debris | TEE (pre/intraprocedural); CT (preprocedural), MRI (DWI, postprocedural) | Embolic event (1–2%); prevention through screening for LA and LAA thrombus and adequate anticoagulation (ACT |
Abbreviations: MR, mitral regurgitation; LAE, leaflet adverse events; SLDA, single-leaflet device attachment; iASD, iatrogenic atrial septal defect; LVOT, left ventricular outflow tract; TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; CMR, cardiac magnetic resonance; HF, heart failure; CT, computed tomography; PH, pulmonary hypertension; RV, right ventricular; LV, left ventricular; LA, left atrial; LAA, left atrial appendage; MRI, magnetic resonance imaging; DWI, diffusion-weighted imaging; ACT, activated clotting time; MVA, mitral valve area.
Multimodality imaging is central to procedural planning for M-TEER, guiding patient selection, anatomical suitability, and device strategy (Table 5).
| Parameter | Thresholds/Criteria | Context | Reference imaging modality |
| MR severity | EROA |
Definition of severe MR | TTE/TEE |
| Vena contracta width | Supportive severity parameter | TTE/TEE | |
| Pulmonary vein flow | Systolic flow reversal | Surrogate marker of severe MR | TEE |
| Exercise-induced MR (STE) | Exercise-induced MR severity (ΔEROA |
Prognostic marker and timing of intervention | STE |
| Anatomical suitability – EVEREST | Coaptation length |
Classic feasibility criteria for M-TEER | TEE |
| Anatomical suitability – posterior leaflet | Additional criterion for M-TEER feasibility | TEE | |
| Annular assessment | Annular dimensions/neo-LVOT simulation | Suitability for M-TEER and future TMVR planning | CT |
Abbreviations: MR, mitral regurgitation; STE, stress echocardiography; EROA, effective regurgitant orifice area; TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; PASP, pulmonary artery systolic pressure; M-TEER, mitral transcatheter edge-to-edge repair; 3D, three-dimensional; LVOT, left ventricular outflow tract; TMVR, transcatheter mitral valve replacement; CT, computed tomography.
TTE and TEE are essential for confirming severe MR and differentiating between
primary and secondary etiologies. Quantitative parameters include effective
regurgitant orifice area (EROA
The mechanism of MR provides critical insights for both prognosis and therapeutic decision-making [101, 102, 103].
Primary (degenerative/organic) MR arises from structural abnormalities of the mitral valve apparatus, such as prolapse, flail, chordal rupture, or leaflet thickening/calcification.
Secondary (functional) MR results from adverse ventricular or atrial remodeling, including papillary muscle displacement, leaflet tethering, annular dilatation, or atrial enlargement, typically in the setting of LV dysfunction or atrial fibrillation.
Mixed MR refers to cases in which organic or structural valve abnormalities (e.g., degenerative changes or prolapse) coexist with functional mechanisms related to ventricular or atrial remodeling. This phenotype is increasingly recognized in clinical practice and may present unique therapeutic challenges, as both components contribute to severity and may respond differently to surgical or transcatheter interventions.
Recognition of these categories is essential for guiding patient selection, anatomical suitability assessment, and device strategy in M-TEER.
Current guideline-based indications for M-TEER are derived from the accumulated trial evidence and registry experience summarized above (Table 4).
For primary (degenerative) MR, surgical repair remains the standard of care. M-TEER is considered appropriate in patients with severe symptomatic MR who are at prohibitive or high surgical risk, provided that their anatomy is suitable. The 2021 ESC/EACTS and 2020 ACC/AHA guidelines assign this a Class IIa recommendation (Level of Evidence B) [19, 20].
For secondary MR, guideline-directed medical therapy (GDMT) and device-based heart-failure management remain first-line treatment options. In patients with severe MR who remain symptomatic despite GDMT, M-TEER may be considered if anatomy is favorable. Current guidelines endorse M-TEER for selected patients as Class IIa, Level of Evidence B [19, 20].
The Japanese Circulation Society (JCS 2020) guidelines similarly acknowledge M-TEER as an option for symptomatic patients at high risk with severe primary MR and for patients with secondary MR whose symptoms persist despite GDMT [21].
Stress echocardiography (STE) refines the timing of intervention in MR by unmasking latent severity and providing prognostic information beyond resting evaluation [104].
Exercise STE increases the venous return, heart rate, and afterload. In primary
MR, this augments regurgitant volume across a fixed orifice, whereas in secondary
MR, LV dilatation and rising systolic pressures exacerbate leaflet tethering,
dynamically enlarging the EROA and regurgitant volume [105, 106].
Exercise-induced pulmonary hypertension is a strong prognostic marker: in
asymptomatic degenerative MR, a pulmonary artery systolic pressure (PASP)
DSE predominantly enhances inotropy and heart rate while reducing LV
end-systolic volume. In secondary MR, regurgitation may decrease if contractile
reserve improves leaflet coaptation, whereas in advanced disease with poor
reserve, MR may worsen because of rising systolic pressures against a tethered
apparatus [105, 109, 110]. A dynamic increase in MR severity during stress,
particularly a
Other approaches, such as passive leg raise, lower-limb compression, or handgrip exercise, have been explored in selected cases but remain less standardized in clinical practice [112, 113, 114].
Long-term studies consistently demonstrate that dynamic worsening of MR during
stress is associated with increased risks of heart failure-related
hospitalization and mortality. Current guidelines recommend STE when the symptoms
and resting severity are discordant. The 2021 ESC/EACTS guidelines note that a
resting PASP
TEE remains the gold standard for assessing anatomical suitability for M-TEER.
The classic “German/EVEREST” thresholds—coaptation length
Device platforms also differ in their adaptation to anatomy. Both current systems (MitraClip G4 and PASCAL) allow independent leaflet grasping; PASCAL additionally incorporates a central spacer that can bridge large coaptation gaps [117], while MitraClip offers multiple arm lengths and widths (e.g., NTR/XTR; NTW/XTW) that help address thick leaflets and wide jets. Selection should therefore be tailored to jet geometry, leaflet quality and length, and the need for single- versus multi-device strategies [96, 98, 100, 116].
Beyond TEE, multimodality imaging refines feasibility assessment and long-term planning. Cardiac CT provides accurate quantification of annular size and shape, as well as its spatial relationship to the LVOT. While echocardiography remains the primary tool for assessing leaflet motion abnormalities such as systolic anterior motion, CT is indispensable for downstream strategy, particularly for simulating the neo-LVOT in the context of TMVR [118]. Importantly, prior M-TEER can complicate subsequent TMVR: clips may immobilize the anterior leaflet, increase the risk of LVOT obstruction, and sometimes preclude leaflet-laceration bailout techniques [119]. Therefore, in contexts where TMVR may be anticipated, the Heart Team should consider “exit-strategy–aware” clip positioning to preserve replacement options.
CMR remains useful when echocardiographic quantification is inconclusive—particularly in multi-jet MR—and for characterizing LV remodeling, complementing echo and CT in an integrative anatomical assessment [120, 121].
Intraprocedural TEE is indispensable for device navigation, leaflet grasping, and residual MR assessment. Real-time 3D imaging allows precise orientation of the device relative to the regurgitant jet, and color Doppler aids in immediate evaluation of procedural success.
In selected cases where TEE is contraindicated or technically limited (e.g., severe esophageal disease, prior surgery, or esophageal stenosis), intracardiac echocardiography (ICE) may serve as an alternative imaging modality. ICE can provide adequate visualization of leaflet grasping and device orientation, while potentially avoiding general anesthesia and complications related to esophageal intubation. Although not yet standard, accumulating clinical experience suggests the feasibility and safety of ICE in challenging scenarios [122, 123, 124, 125].
In rare situations where esophageal anatomy prevents optimal TEE imaging, collaboration with gastroenterologists may be required. Contrast-enhanced TEE has also been reported in a case with severe esophageal achalasia [126].
LAE comprise a spectrum of complications related to leaflet–clip interaction, including single-leaflet device attachment (SLDA), leaflet tear or perforation, leaflet detachment, complete device embolization, and other leaflet injuries [127]. Although the overall incidence is only a few percent, LAE represents a major cause of recurrent MR and the need for reintervention. Careful preprocedural anatomical assessment using CT and TEE, meticulous intraprocedural 3D TEE monitoring of leaflet grasping, and verification before clip release are essential strategies for preventing LAE.
SLDA: SLDA occurs in approximately 1–2% of procedures in contemporary
registries (EVEREST II, COAPT, EXPAND) [127, 128]. Risk factors identifiable
using TEE and CT include inadequate leaflet insertion depth (
Leaflet tear or perforation: This issue is observed in
Leaflet detachment: This represents progression of SLDA, where the leaflet slips out of the clip after initial securement. The device remains attached to the opposite leaflet but becomes functionally ineffective, leading to recurrent MR [131].
Device embolization: This is an extremely rare event (
Other leaflet injury: This category includes perforation, distortion, or laceration of the leaflet not meeting the above definitions. Such injuries are usually related to challenging anatomy, aggressive clip manipulation, or multiple grasping attempts. Although rare, they may lead to recurrent MR or acute procedural failure and highlight the importance of minimizing unnecessary re-grasping and ensuring optimal device–leaflet alignment [135].
Elevated mean transmitral gradients (
An iASD is almost universally present immediately after transseptal access.
However, persistent defects are observed in approximately 10–20% of patients at
1 year after the procedure [138, 139, 140]. Most are hemodynamically insignificant and
managed conservatively. However, follow-up with TEE or TTE including bubble
contrast studies is useful to detect clinically relevant shunts. In patients with
severe pulmonary hypertension or right ventricular (RV) dysfunction, persistent
iASDs may exacerbate right-sided volume overload or, rarely, lead to
right-to-left shunting with hypoxemia. In such cases, percutaneous closure with
ASD occluder devices has been reported to be a safe and effective option [138].
Moreover, morphological assessment by 3D TEE has been shown to be valuable, as an
eccentricity index
LVOT obstruction is extremely rare after M-TEER, with an incidence of
Cardiac tamponade or hemopericardium occurs in approximately 0.5–1% of M-TEER cases and is most commonly a consequence of transseptal puncture or catheter manipulation [85, 142]. Intraprocedural TEE is crucial for early detection of pericardial effusion, allowing immediate intervention with pericardiocentesis when necessary [144]. Although rare, this complication carries significant morbidity; therefore, meticulous puncture technique and real-time imaging guidance are essential preventive measures.
Periprocedural stroke occurs in approximately 1–2% of patients undergoing
M-TEER procedures, according to contemporary registry data [10, 142]. The
underlying mechanisms include embolization of pre-existing thrombus, device- or
catheter-related thrombus formation, and air or atheromatous embolism. The risk
is significantly mitigated by systematic TEE screening to exclude left atrial
appendage thrombus and strict intraprocedural anticoagulation, maintaining
activated clotting time maintained at
Imaging-based strategies to minimize complications in M-TEER:
• Leaflet adverse events (SLDA, tear, embolization): Perform meticulous
preprocedural CT and TEE assessment of leaflet morphology, tissue quality, and
calcification. During the procedure, use 3D TEE to confirm symmetrical leaflet
insertion depth ( • Iatrogenic mitral stenosis: Evaluate baseline MVA and transmitral gradient using
CT and Doppler TEE. Avoid excessive clip number or narrow clip spacing in
patients with small baseline MVA ( • Iatrogenic atrial septal defect (iASD): During and after the procedure, use
color Doppler or bubble contrast studies to detect shunt persistence. In patients
with PH or RV dysfunction, perform follow-up 3D TEE to determine the need for
percutaneous closure. • LVOT obstruction: Identify high-risk features such as small LV cavity, basal
septal hypertrophy, or acute aortomitral angulation on preprocedural CT. During
clip placement, monitor LVOT flow velocity with continuous-wave Doppler to detect
dynamic obstruction early. • Cardiac tamponade/hemopericardium: Use real-time TEE guidance for transseptal
puncture and catheter manipulation; maintain continuous pericardial monitoring to
enable immediate intervention if effusion develops. • Stroke/systemic embolism: Exclude left atrial appendage thrombus by
preprocedural TEE or CT; maintain adequate anticoagulation (activated clotting
time
Structured imaging surveillance after M-TEER is essential not only to confirm procedural success and detect complications but also to provide prognostic insights into long-term durability and patient outcomes.
TTE serves as the cornerstone of longitudinal follow-up after M-TEER [142].
Importantly, even mild residual MR does not necessarily ensure favorable
outcomes. Subanalyses from the EVEREST II High-Risk Registry and the COAPT trial
demonstrated that patients with
In clinical practice, grading residual MR after M-TEER remains particularly challenging. Unlike native MR, where a single central jet is often predominant, post-M-TEER, patients frequently exhibit multiple eccentric or wall-impinging jets that produce complex flow patterns. Conventional integrative parameters such as vena contracta width or PISA are often difficult to apply in this context [129, 150]. Therefore, a multiparametric approach is recommended, including comprehensive color Doppler evaluation of all jets, assessment of pulmonary vein flow, and measurement of transmitral gradients [10]. Advanced imaging modalities, such as 3D echocardiography and quantitative Doppler methods, may provide additional insights. However, interobserver variability remains a significant limitation [86]. These technical challenges should be acknowledged when interpreting residual MR severity, as misclassification can influence both clinical decision-making and long-term outcome assessment. A major unmet need is the standardization of residual MR quantification after M-TEER, since current multiparametric echocardiographic approaches remain highly variable and lack validation against hard clinical endpoints.
Serial assessment of mean transmitral gradients using Doppler echocardiography
is crucial during postprocedural follow-up, particularly in patients treated with
multiple clips [142]. Unlike intraprocedural monitoring, which ensures immediate
procedural feasibility, longitudinal surveillance is needed to detect progressive
increases in gradients that may indicate functional mitral stenosis [151]. A mean
gradient
Reverse LV remodeling is defined as a reduction in LV volumes, particularly end-diastolic and end-systolic volumes, and/or improvement in LVEF, reflecting structural and functional recovery of the ventricle [10, 154]. Effective MR reduction with M-TEER promotes reverse remodeling. In the COAPT trial, LV end-diastolic volume decreased by approximately 25 mL, and LVEF improved by nearly 5%, both strongly correlated with survival benefit [10]. Recent studies have shown that patients who fail to demonstrate significant reverse remodeling despite adequate MR reduction experienced worse outcomes, underscoring the prognostic importance of LV structural response [154]. Furthermore, CMR imaging provides incremental prognostic information, the presence and extent of LGE reflecting myocardial fibrosis, and predict limited reverse remodeling and adverse clinical outcomes beyond echocardiographic parameters [155].
Serial assessment of tricuspid regurgitation (TR) velocity and estimated PASP is
valuable for tracking hemodynamic benefit after M-TEER [156]. Persistent
pulmonary hypertension (PASP
Thus, multimodality imaging follow-up after M-TEER should extend beyond MR quantification to include transmitral gradients, chamber remodeling, pulmonary hemodynamics, and myocardial tissue characterization, all of which contribute to a comprehensive prognostically meaningful assessment of long-term outcomes.
Patients with extensive leaflet calcification, short posterior leaflets, very large regurgitant orifices, or degenerative lesions characteristic of Barlow’s disease continue to pose significant technical challenges for M-TEER [116, 159]. The introduction of newer clip designs, including the fourth-generation MitraClip system, and the development of the PASCAL device have expanded the range of anatomies amenable to transcatheter repair. However, a considerable proportion of patients remain unsuitable for M-TEER and may require alternative strategies such as surgical repair or TMVR [149, 160].
Long-term outcomes after M-TEER depend not only on LV remodeling but also on RV function and pulmonary hemodynamics [158, 161]. Observational studies and COAPT subanalyses have consistently demonstrated that impaired RV function and concomitant severe TR are independently associated with increased mortality and hospitalization for heart failure [162]. Moreover, the concept of RV–pulmonary circulation uncoupling, assessed using indices such as the tricuspid annular plane systolic excursion (TAPSE)/PASP ratio, has emerged as a powerful prognostic marker [163]. These findings highlight that future patient selection and postprocedural follow-up should incorporate comprehensive evaluation of biventricular function and pulmonary hemodynamics, extending beyond MR reduction alone.
Atrial functional MR (AFMR), defined as left atrial enlargement and atrial fibrillation in the absence of significant LV remodeling, has gained increasing attention as a distinct clinical and pathophysiological entity, separate from ventricular functional MR [164, 165]. Mechanistic imaging studies have clarified that AFMR is primarily driven by left atrial dilatation, mitral annular enlargement, and atriogenic leaflet tethering, underscoring its unique substrate and therapeutic implications [165]. Early registry data suggest that M-TEER may provide symptomatic and hemodynamic improvement in patients with preserved LV function [166]. However, in patients with advanced atrial remodeling or persistent atrial fibrillation, recurrence of MR remains frequent [167]. Defining the optimal timing and selection criteria for intervention in AFMR represents an important area for future investigation.
Beyond its role as a standalone intervention, M-TEER is increasingly being positioned within a broad therapeutic continuum for MR. For patients at high surgical risk or with advanced comorbidities, M-TEER may provide interim stabilization, improving symptoms and hemodynamics while preserving candidacy for future therapies, including TMVR [168]. In others cases, it may complement transcatheter approaches targeting additional valves, such as combined treatment of MR and TR, which is frequently encountered in cases of advanced heart failure [169]. Moreover, the growing experience with multimodality imaging and device iterations supports a tailored, stepwise strategy in which M-TEER functions as a component of a long-term disease management plan rather than as an isolated procedure [170]. This integrated perspective highlights the evolution of M-TEER from a palliative tool to a central element of comprehensive transcatheter valve therapy.
Recent advances in quantitative 3D echocardiography, fusion imaging, AI, and 3D
printing are redefining both procedural guidance and preprocedural planning in
M-TEER. Fusion imaging, which integrates 3D TEE and fluoroscopy in real time, has
demonstrated high registration accuracy and can reduce fluoroscopy time by
approximately 20%, facilitating clip alignment, leaflet grasping, and procedural
navigation [171, 172]. AI-based image analysis enables automated quantification
of the regurgitant orifice area and reliable prediction of procedural success
with high reproducibility. Validation studies have demonstrated strong agreement
with expert manual analysis, supporting its potential integration into real-time
guidance and preprocedural planning workflows [173, 174, 175]. Furthermore, 3D-printed
and virtual simulation models have shown
The future of M-TEER will be driven by synergistic advances in device technology, procedural algorithms, and multimodality imaging integration. Building upon recent innovations, the next stage will focus on clinical implementation and validation of advanced imaging tools—ensuring that quantitative 3D echocardiography, fusion imaging, and AI can be seamlessly incorporated into real-world workflows to enhance procedural precision and outcome consistency [172, 178, 179]. Integrating these imaging innovations into routine workflows may reduce operator variability and extend M-TEER applicability to patients with anatomical complexities or those who were previously ineligible. However, many of these technologies, particularly fusion imaging and AI-based tools, still require robust validation in real-world clinical practice before their widespread adoption. In parallel, continued evolution in clip systems, the emergence of alternative repair devices, and evidence-based treatment pathways will further improve procedural safety, durability, and accessibility of M-TEER across diverse populations with MR [96].
Beyond technological innovation, structured education and interprofessional collaboration within the Heart Team remain essential to optimize both medical and surgical decision-making. A shared understanding of imaging is particularly critical, as it provides a common language among cardiologists, cardiac surgeons, anesthesiologists, nurses, technicians, and other staff, thereby facilitating consistent communication and joint problem-solving. Creative approaches to education, such as the recently proposed handgrip model for visualizing tricuspid valve anatomy and leaflet relationships, exemplify how innovative teaching tools can foster deeper anatomical understanding and interdisciplinary dialogue [180]. We hope that the present review contributes to advancing such educational and collaborative initiatives, ultimately fostering a more standardized, efficient, and patient-centered practice in TCS.
Over the past two decades, TCS has evolved from being a last-resort option for patients at high risk to an established therapy across the surgical risk spectrum. Advances in device technology, imaging, and perioperative care have brought outcomes closer to conventional surgery, while the Heart Team approach—anchored using multimodality imaging as a common language—has become fundamental to success.
Despite these advances, unresolved challenges remain. Durability beyond 10 years is uncertain, residual MR grading lacks standardization, and right heart–pulmonary assessment should be incorporated into decision-making. Surgical expertise remains indispensable for patient selection, management of complex anatomies, and hybrid strategies. Future progress will depend on addressing these gaps through continued innovation and collaboration among interventionalists, surgeons, and imaging specialists. Multimodality imaging will be the key to lifetime management strategies in TCS.
AFMR, atrial functional mitral regurgitation; AS, aortic stenosis; AVA, aortic valve area; BAV, balloon aortic valvuloplasty; BEV, balloon-expandable valve; CMR, cardiac magnetic resonance; CT, computed tomography; DSE, dobutamine stress echocardiography; EOA, effective orifice area; EROA, effective regurgitant orifice area; GDMT, guideline-directed medical therapy; GLS, global longitudinal strain; HALT, hypo-attenuated leaflet thickening; ICE, intracardiac echocardiography; IE, infective endocarditis; iASD, iatrogenic atrial septal defect; IVL, intravascular lithotripsy; LGE, late gadolinium enhancement; LV, left ventricle/left ventricular; LVEF, left ventricular ejection fraction; LVMI, left ventricular mass index; LVOT, left ventricular outflow tract; MR, mitral regurgitation; M-TEER, mitral transcatheter edge-to-edge repair; MVA, mitral valve area; PASP, pulmonary artery systolic pressure; PH, pulmonary hypertension; PPI, permanent pacemaker implantation; PPM, patient–prosthesis mismatch; PVL, paravalvular leak; RCT, randomized controlled trial; RLM, reduced leaflet motion; RV, right ventricle/right ventricular; SAVR, surgical aortic valve replacement; SEV, self-expanding valve; STE, stress echocardiography; STJ, sinotubular junction; SVD, structural valve degeneration; TAPSE, tricuspid annular plane systolic excursion; TAVR, transcatheter aortic valve replacement; TCS, transcatheter cardiac surgery; TEE, transesophageal echocardiography; THV, transcatheter heart valve; TMVR, transcatheter mitral valve replacement; TR, tricuspid regurgitation; TTE, transthoracic echocardiography; VTC, valve-to-coronary distance.
KS conceived and designed the review, performed the literature review, wrote the manuscript, and approved the final version of the manuscript. The author read and approved the final manuscript. The author has participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The author declares no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


