1 Department of Anesthesia, First Affiliated Hospital, Zhejiang University School of Medicine, 310003 Hangzhou, Zhejiang, China
2 Department of Cardiovascular Surgery, First Affiliated Hospital, Zhejiang University School of Medicine, 310003 Hangzhou, Zhejiang, China
3 Department of Intensive Care Unit, First Affiliated Hospital, Zhejiang University School of Medicine, 310003 Hangzhou, Zhejiang, China
Abstract
Left ventricular assist device (LVAD) implantation is a common intervention for patients with end-stage heart failure. However, comorbid chronic obstructive pulmonary disease (COPD) poses significant challenges in perioperative management. In such cases, ultrafast track anesthesia (UFTA) has been proposed to minimize cardiopulmonary disturbances and promote rapid recovery.
This report describes the case of a 53-year-old man diagnosed with dilated cardiomyopathy and COPD. Despite extensive medical treatment, the patient's condition did not improve, and his activities were strictly limited (New York Heart Association Class IV). Echocardiography revealed an enlarged heart with an ejection fraction of 24%. The patient was scheduled to undergo LVAD implantation via minimally invasive surgery. An UFTA protocol based on propofol and remifentanil was consequently designed and implemented. Postoperatively, the patient regained consciousness and exhibited stable respiratory function and hemodynamic parameters. On-table extubation was performed, and the patient was transferred to the intensive care unit. However, he received a butorphanol infusion after surgery and had to be re-intubated 23 h later.
This case reveals that if a multidisciplinary team effort and tailored treatment protocols had not been executed, reintubation may not have been avoided, even though the patient had been extubated in the operating room shortly after surgery.
Keywords
- ultra-fast-track anesthesia
- left ventricular assist device implantation
- minimally invasive surgery
- chronic obstructive pulmonary disease
End-stage heart failure, which affects millions of individuals worldwide, is associated with a mortality rate exceeding 50% within five years [1]. For patients ineligible for heart transplantation, left ventricular assist devices (LVADs) have emerged as life-sustaining therapies that enhance both survival and quality of life [2, 3]. However, management of patients with heart failure remains challenging, particularly in patients with comorbid cardiogenic shock or pulmonary hypertension. The coexistence of chronic obstructive pulmonary disease (COPD) in patients requiring LVAD implantation complicates perioperative management. Consequently, patients undergoing LVAD implantation often require customized anesthetic approaches. Conventional anesthesia protocols for LVAD surgery involve additional opioid administration and delayed extubation, which may increase the risk of postoperative delirium, ventilator-associated pneumonia, and prolonged intensive care unit (ICU) hospitalization [4]. In contrast, fast-track anesthesia (FTA) emphasizes early extubation, implements multimodal analgesia, and utilizes goal-directed fluid therapy to improve recovery outcomes [5]. Ultrafast track anesthesia (UFTA) further shortens intubation time, avoids postoperative ventilation, and has been applied in cardiac procedures in recent years [6]. In the present study, we present a case involving the application of UFTA in conjunction with minimally invasive surgery for LVAD implantation in a patient with end-stage heart failure combined with COPD. However, the patient received a butorphanol infusion postoperatively, and was reintubated the following day.
A 53-year-old man (height: 168 cm; weight: 42 kg) was diagnosed with dilated cardiomyopathy, heart failure, type II respiratory failure, and COPD. He was admitted to the emergency department because of an acute cough with sputum production that had persisted for 2 months, and was accompanied by fatigue for the past 1 month. Upon admission, the patient received cardiotonic, diuretic, and anti-infective therapies. Physical examination revealed a distended abdomen and mild lower limb edema. His heart sounds were distant and no significant murmurs were detected. Patients’ overall physical status and cardiac function were evaluated and classified as follows: American Society of Anesthesiologists Physical Status Classification System IV, New York Heart Association Class IV, Interagency Registry for Mechanically Assisted Circulatory Support Profile IV, and American Heart Association Stage D.
Electrocardiography revealed sinus tachycardia (115 beats/minute). Brain magnetic resonance imaging revealed no apparent abnormalities. Computed tomography (CT) of the lungs revealed signs of inflammation and emphysema (Fig. 1A). Pulmonary function tests revealed moderate to severe restrictive ventilatory impairment with moderate diffusion impairment. Echocardiography revealed mild mitral and tricuspid regurgitation, elevated pulmonary systolic pressure, significant enlargement of the left ventricle, diffuse hypokinesis of the left ventricular wall, and reduced left ventricular systolic function, with a left ventricular end-diastolic dimension of 63 mm and left ventricular ejection fraction (LVEF) of approximately 24% (Fig. 2A,B).
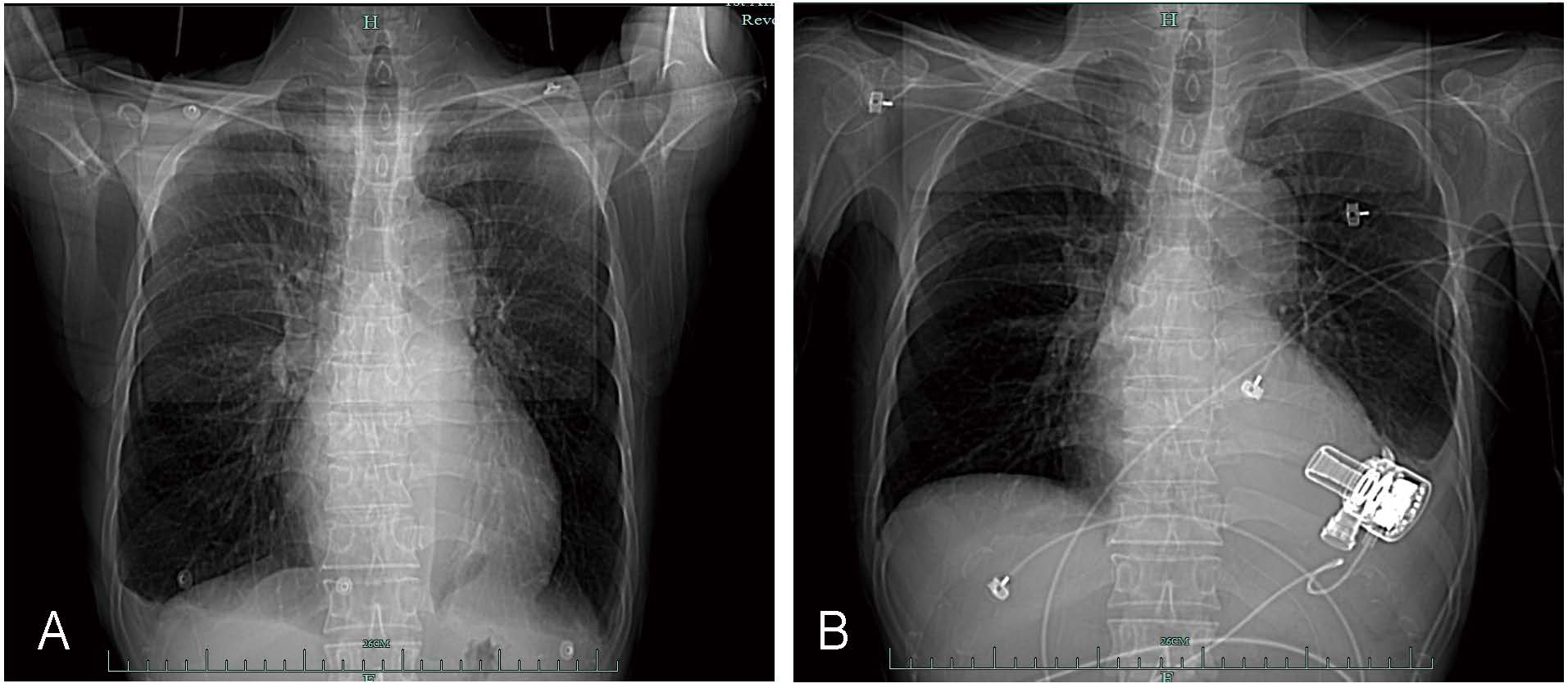 Fig. 1.
Fig. 1.
Chest computed tomography scans taken before and after surgery. (A) Before the operation. (B) After left ventricular assist device implantation.
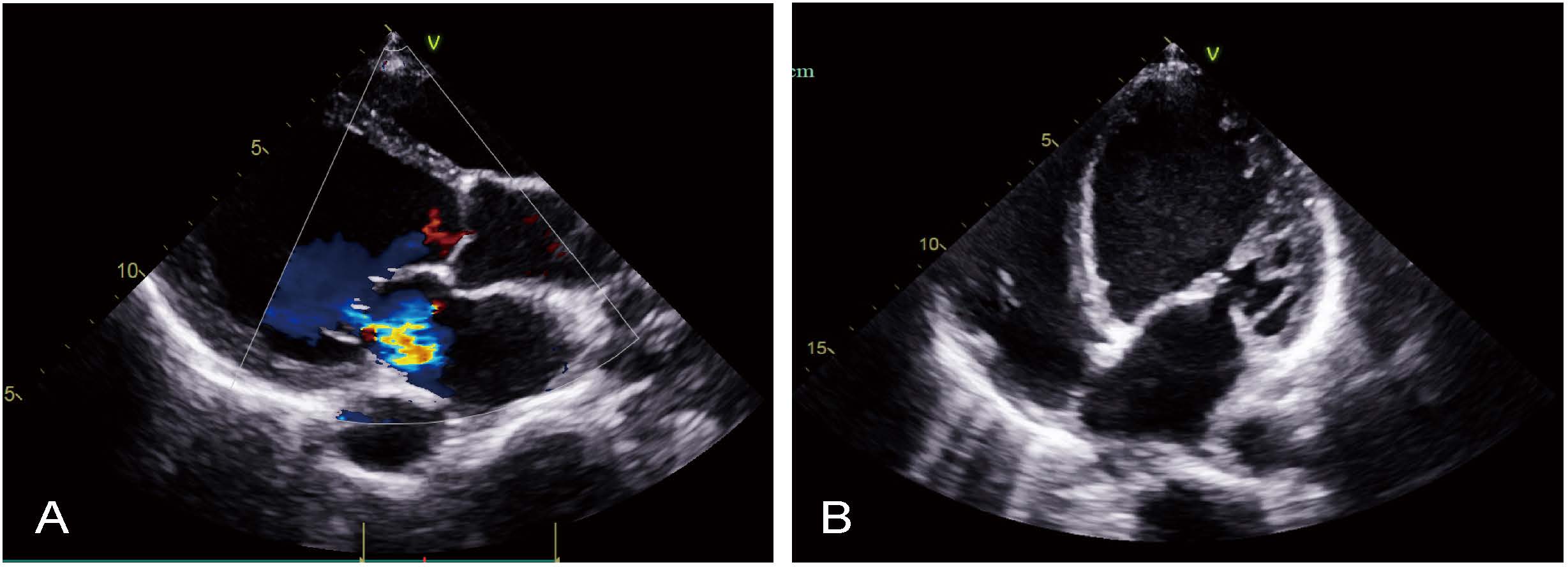 Fig. 2.
Fig. 2.
Preoperative transthoracic echocardiography. (A) Parasternal long-axis view. (B) Apical four-chamber view.
Laboratory analyses revealed the following results: hemoglobin concentration,
12.3 g/dL; platelet count, 90
Upon arrival to the operating room, the patient’s blood pressure was 95/67 mmHg, heart rate was 102 beats/min, and respiratory rate was 23 breaths/min (Fig. 3A). The patient’s preoperative blood analysis are presented in Table 1. Standard monitoring was performed using electrocardiography and arterial blood pressure and peripheral oxygen saturation measurements. Prior to anesthesia induction, epinephrine was infused at 0.025 µg/kg/min. Anesthesia was induced with etomidate at a dosage of 0.2 mg/kg and fentanyl at 5 µg/kg. Rocuronium at 0.6 mg/kg was administered to facilitate endobronchial intubation using a double-lumen tube (left 35 Fr, Covidien, Mansfield, MA, USA). The tidal volume was set at 6 mL/kg of ideal body weight, with a positive end-expiratory pressure (PEEP) of 5 cmH2O, and the peak airway pressure was maintained below 30 cmH2O. Controlled ventilation was implemented using a 50% oxygen/air mixture at a rate of 15 breaths per minute to maintain the partial pressure of end-tidal carbon dioxide (PETCO2) between 35 and 40 mmHg, with intraoperative alveolar re-expansion occurring every 30 min. Anesthesia was maintained with propofol at a rate of 200–300 mg/h, remifentanil at 0.2 µg/kg/min, and rocuronium at 20 mg/h, with adjustments made based on bispectral index monitoring. An infusion of epinephrine at 0.1 µg/kg/min was administered to stabilize hemodynamics.
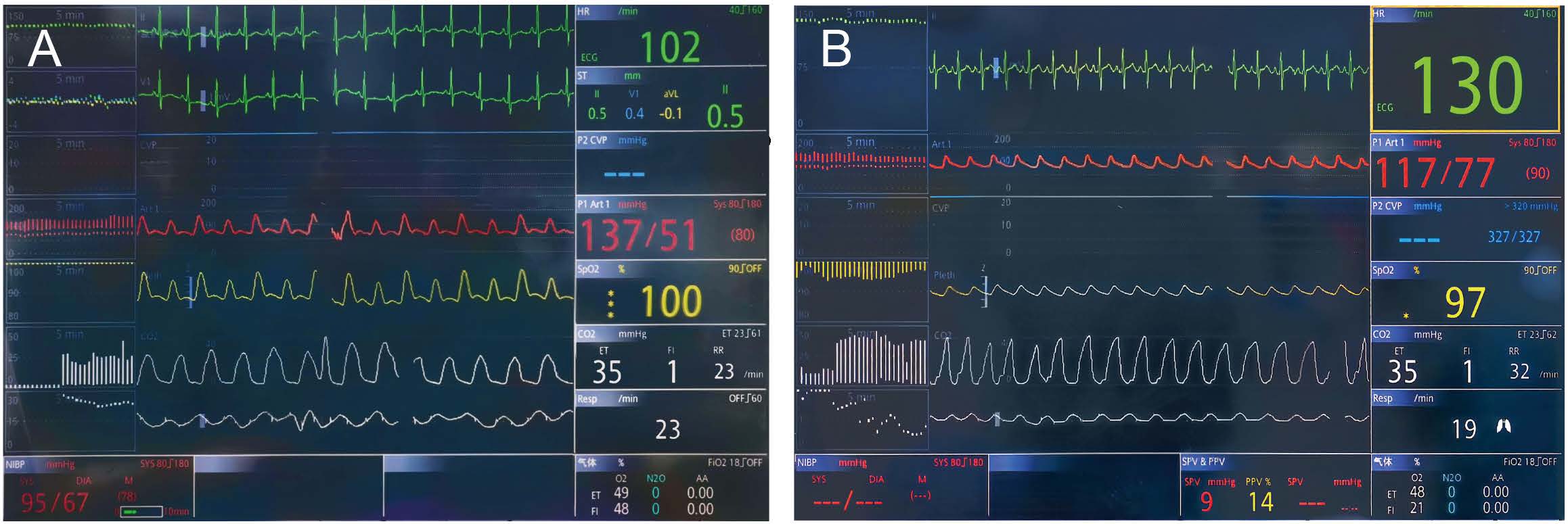 Fig. 3.
Fig. 3.
Screenshots of monitoring data. (A) Monitoring data under oxygen before anesthesia. (B) Monitoring data during oxygen inhalation via a nasal catheter after extubation.
| A | B | C | D | E | F | |
| pH | 7.451 | 7.442 | 7.502 | 7.323 | 7.378 | 7.341 |
| PCO2 (mmHg) | 56.0 | 47.7 | 38.7 | 59.2 | 54.6 | 55.1 |
| PO2 (mmHg) | 64.1 | 261 | 270 | 131 | 271 | 87.3 |
| SO2 (%) | 94.5 | 100 | 100 | 99.1 | 100 | 97.1 |
| Na+ (mmol/L) | 139 | 143 | 143 | 146 | 146 | 147 |
| K+ (mmol/L) | 4.0 | 4.1 | 4.3 | 4.2 | 4.2 | 4.1 |
| Ca2+ (mmol/L) | 1.11 | 0.91 | 0.94 | 1.25 | 1.22 | 1.08 |
| Glu (mmol/L) | 6.2 | 8.0 | 7.7 | 5.0 | 7.3 | 7.3 |
| Lac (mmol/L) | 1.7 | 2.1 | 3.2 | 4.7 | 3.8 | 1.8 |
| Hct (%) | 36 | 26.6 | 27 | 27.6 | 31.3 | 28.5 |
| BE (mmol/L) | 15.0 | 8.4 | 7.1 | 4.6 | 7.0 | 4.0 |
| HCO3– (mmol/L) | 39.0 | 32.5 | 30.3 | 30.7 | 32.2 | 29.8 |
| Hb (g/dL) | 11.8 | 8.7 | 8.8 | 9.0 | 10.2 | 9.3 |
(A) Pre-anesthesia; (B,C) cardiopulmonary bypass; (D) non-extracorporeal circulation and one-lung ventilation; (E) End of operation; (F) After extubation.
PCO2, partial pressure of carbon dioxide; PO2, partial pressure of oxygen; SO2, oxygen saturation; Glu, glucose; Lac, lactate; Hct, hematocrit; BE, base excess; Hb, hemoglobin.
Following intubation, a 3-lumen central venous catheter (Certofix Trio V720, Braun, Melsungen, Germany) was placed in the internal jugular vein along with a hemostasis introducer (8.5 Fr, Fast-CathTM, Abbott Medical, Plymouth, MN, USA). Subsequently, a Swan-Ganz pulmonary catheter (141F7, Edwards Lifesciences LLC, Puerto Rico, USA) was advanced through the introducer, and the pulmonary artery pressure (24/17 mmHg) and pulmonary capillary wedge pressure (12 mmHg) were recorded. Additionally, a pulse index continuous cardiac output (PiCCO) catheter (PV2015L20-A and PV8215, Getinge, Feldkirchen, Germany) was inserted into the left femoral artery to monitor hemodynamic parameters. Cardiac function was assessed by transesophageal echocardiography.
The procedure was performed using minimally invasive techniques on an arrested heart under cardiopulmonary bypass (CPB), achieved through cannulation of the right femoral artery and vein. This minimally invasive approach involves exposing the apex of the heart via the sixth intercostal space on the left side of the chest and accessing the aorta through the right side of the second and third parasternal intercostal spaces. After clamping the ascending aorta and inducing cardiac arrest with cardioplegic fluid, the surgeon positioned the LVAD (Corheart® 6, Core Medical Technology Co. Ltd., Shenzhen, Guangdong, China) inflow cannula at the left ventricular apex, created a tunnel for the driveline, and performed anastomosis between the outflow graft and aorta. The procedure was uneventful. Upon release of the ascending aorta clamp, the heart resumed spontaneous rhythm. The LVAD was subsequently initiated, and the flow rate was gradually increased to 4.11 L/min. The mean arterial pressure was maintained at 70–80 mmHg using dobutamine (3 µg/kg/min), epinephrine (0.1 µg/kg/min), and nitroglycerin (0.15 µg/kg/min), with adjustments made based on the PiCCO parameters during the post-CPB period. The CPB flow was gradually reduced and the patient was weaned; at this stage, the pulmonary artery pressure and pulmonary capillary wedge pressure were 29/11 and 11 mmHg, respectively.
Rocuronium infusion was discontinued following chest closure. Propofol infusion was stopped 10 min before procedure completion. At the end of the procedure, fentanyl (1 µg/kg) and tramadol (1 mg/kg) were administered, and an intercostal nerve block was performed using 0.375% ropivacaine (20 mL). After surgery, the patient awoke under remifentanil infusion (0.05 µg/kg/min). Subsequently, sugammadex sodium (200 mg) was administered. Pressure support ventilation (with an oxygen fraction of 60%, pressure of 15 cmH2O, and PEEP of 5 cmH2O) was utilized to maintain a stable oxygen saturation of 100%. Fifteen minutes later, the patient regained consciousness and followed the instructions well. Tracheal extubation was performed under the supervision of both the attending surgeon and anesthesiologist. Oxygen inhalation via a nasal catheter was provided for 30 min, and repeat blood analysis showed a PCO2 level of 55 mmHg (Table 1 and Fig. 3B). The patient was transferred to the ICU.
The total operation time was 220 min, which included 98 min of cardiopulmonary bypass. Throughout the procedure, 350 mL of plasma, 1500 mL of crystalloid, and 100 mL of albumin were infused. Additionally, fibrinogen (0.5 g), prothrombin complex (300 units), and tranexamic acid (0.75 g) were administered. The estimated total blood loss was 500 mL, and the urine volume was 1300 mL. Echocardiography revealed that the left ventricular end-diastolic dimension was 48 mm, with an LVEF of 45%. The postoperative chest radiographs are shown in Fig. 1B.
Around extubation and in the early stages of ICU stay, the patient’s condition was stable, and he remained awake and alert. His pain intensity was 2 on the numerical rating scale. However, the patient was administered butorphanol infusion for sedation. During the subsequent 23 h postoperatively, the patient’s coughing ability weakened, resulting in sputum accumulation, and his blood PCO2 level increased to 82 mmHg, necessitating endotracheal reintubation and mechanical ventilation. Propofol was administered under sedation. Four days later, the tracheal catheter was removed. The patient was subsequently managed with high-flow nasal cannula therapy set at a fraction of inspired oxygen of 25% and a flow rate of 40 L/min, achieving a peripheral oxygen saturation of 92%–97%, PCO2 of 50–55 mmHg, and a respiratory rate of approximately 20 breaths per minute. The patient was transferred to the general ward and discharged after a total hospital stay of 33 days. At the 3-month follow-up, the patient demonstrated good functional recovery.
This case highlights the fact that if a multidisciplinary team effort and tailored treatment protocols had not been executed rigorously, reintubation may not have been avoided, even though the patient had been extubated successfully in the operating room shortly after surgery. However, this case demonstrates the possibility of conducting early extubation in a patient undergoing LVAD implantation with marginal lung function. Although Kim et al. [5] documented a patient who underwent LVAD implantation and was extubated 4 h after surgery, our protocol revealed an on-table extubation after the procedure.
The UFTA was first introduced by Djaiani et al. [7] in off-pump coronary surgery in 2001, and has been widely used in cardiac anesthesia [8]. The UFTA offers significant advantages over conventional anesthesia protocols. A particular benefit of UFTA for COPD patients with COPD is the immediate resumption of spontaneous coughing. Effective coughing requires closure of the glottis, and endotracheal extubation makes this closure possible. Under conditions of full consciousness and alertness combined with full recovery of muscle relaxation, spontaneous coughing is the best alternative for patients with marginal lung function. However, UFTA imposes challenges on anesthesiologists regarding the selection of anesthetic agents, surgical procedures, and patient conditions. Ahmad et al. [6] previously explored the combined use of UFTA and minimally invasive techniques in three patients with HeartMate, showing that this approach was safe and effective. The UFTA protocol is even more favorable in patients with compromised lung function as this population is susceptible to ventilator-associated infections. In our previous reports, we have successfully applied UFTA during pediatric heart and liver transplantations, and achieved favorable postoperative outcomes [9, 10].
In the present case, the patient’s PCO2 level remained at 50–60 mmHg before the operation, despite maximum treatment. His lung function indicated that extubation was delayed under routine anesthesia management. To promote early extubation and resuming immediate spontaneous coughing, we designed and implemented a UFTA approach with on-table extubation, in the hope of avoiding delayed extubation and minimizing ventilator-associated complications [11]. During the surgery, low tidal volumes, low levels of PEEP, permissive hypercapnia, and intermittent alveolar re-expansion [12, 13, 14, 15, 16] were applied to minimize barotrauma and postoperative atelectasis. Towards the end of the operation, a multimodal analgesic regimen of small doses of opioids combined with nerve block was selected. Low-dose remifentanil infusion was maintained until full recovery of consciousness, while muscle relaxation was antagonized by administration of sufficient sugammadex [17, 18].
As expected, the patient regained consciousness and responded well shortly after the procedures. After his PCO2 stabilized to a level similar to that before surgery, the patient was extubated. Around the extubation period and in the early hours in the ICU, the patient’s respiratory rate and PCO2 remained stable. Unfortunately, the patient received butorphanol, an opioid with significant cough-suppressing effects, which suppressed consciousness and cough reflex, necessitating reintubation the next day.
Coughing reflex and sputum excretion are both fundamental factors in maintaining airway patency; therefore, airway sensitivity and alertness are crucial for maintaining effective coughing and airway patency in these patients [19]. Consequently, coughing depression agents, such as opioids, are extraordinarily sensitive and should be considered contraindicated after extubation. The key lesson from the present case is that if a multidisciplinary team effort and tailored treatment protocols have not been executed throughout the perioperative unit, UFTA only requires reintubation. Taken together, although UFTA presents a possibility for patients with marginal lung function undergoing LVAD, challenges remain in detailing protocols across different procedures and institutions.
This case demonstrates the possibility of implementing UFTA with LVAD implantation in patients with COPD. However, teamwork and coordination are critical for achieving favorable outcomes.
Data sharing is not applicable to this article, as no datasets were generated or analyzed during the current case report.
WBT, FH, PT, XZ, DSS, YMN, and YXY participated in the study’s conception, design, data collection, analysis, and manuscript discussion and finalization. All authors actively contributed to editorial revisions, read, and approved the final manuscript. All authors agree to be accountable for the work’s accuracy and integrity.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of the First Affiliated Hospital of Zhejiang University (Protocol No. 2025B-0392). Written informed consent for the publication of this case report was obtained from the patient.
The authors would like to thank the patient for his cooperation in this study and Medjaden Inc. for English language editing.
This work was supported by the First Affiliated Hospital, Zhejiang University School of Medicine (BL202234).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF51959.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



