1 Department of Cardiothoracic Surgery, The First Affiliated Hospital, Hengyang Medical School, University of South China, 421001 Hengyang, Hunan, China
2 Department of Cardiothoracic Surgery, The Second Affiliated Hospital, Hengyang Medical School, University of South China, 421001 Hengyang, Hunan, China
3 School of Nursing, University of South China, 421001 Hengyang, Hunan, China
Abstract
Heart transplantation (HT) remains the only therapeutic option that significantly improves long-term survival for patients with end-stage cardiomyopathy. However, clinical practice faces challenges including the shortage of donor organs, increased surgical risks, and limitations in long-term survival. Recent advancements in minimally invasive surgery, precision immunosuppression, xenotransplantation, and artificial heart technologies have substantially improved transplant safety and outcomes; however, a systematic integration of these innovations is lacking. This study reviews technological advancements and current trends in HT to optimize clinical decision-making and promote individualized treatment strategies. A systematic review of literature published between 2015 and 2025 was conducted using PubMed and Web of Science databases. The search focused on five key areas of HT: minimally invasive surgery, donor expansion, precision immunosuppression, xenotransplantation, and postoperative management. Literature screening and analysis were performed independently by two researchers to ensure the objectivity and accuracy of the findings. Minimally invasive techniques, including partial Maryland and robot-assisted surgery, have significantly reduced postoperative bleeding and complications in high-risk patients. Normothermic machine perfusion (NMP) has extended donor heart preservation up to 12 hours, improving marginal donor utilization. Genotype-guided immunosuppressive therapy and donor-derived cell-free DNA (dd-cfDNA) monitoring enhance the precision of immunosuppression management. CRISPR-Cas9-mediated xenotransplantation has enabled successful transplantation of genetically modified pig hearts into humans, with reported survival exceeding 60 days. Magnetic Resonance Imaging (MRI) T1/T2 mapping and implantable hemodynamic monitoring technologies also show promise for the non-invasive early detection of rejection. Technological innovations have greatly enhanced clinical outcomes in HT. However, further long-term data and standardized evidence are necessary. Future efforts should focus on standardizing techniques, translating immune tolerance strategies into clinical practice, and establishing safety frameworks for xenotransplantation.
Keywords
- heart transplantation
- normothermic machine perfusion
- marginal donor
- xenotransplantation
- individualized immunosuppression
Cardiomyopathy is a heterogeneous group of cardiac disorders, characterized by myocardial structural and functional abnormalities, often leading to heart failure or sudden cardiac death. Based on phenotype and pathological mechanisms, cardiomyopathy can be classified into several subtypes, including hypertrophic cardiomyopathy (HCM), dilated cardiomyopathy (DCM), arrhythmogenic cardiomyopathy (ACM), restrictive cardiomyopathy (RCM), and left ventricular non-compaction (LVNC) [1]. For patients with end-stage cardiomyopathy, when conventional treatments such as pharmacotherapy, device interventions, and revascularization fail to halt disease progression, heart transplantation (HT) is considered the ultimate life-saving treatment [2].
Although HT has matured, clinical decision-making is increasingly complex,
facing three core challenges requiring systematic optimization. Firstly, the
severe shortage and uneven distribution of donors is a global issue. While the
2018 policy revision by the United Network for Organ Sharing (UNOS) improved
transplantation efficiency [3, 4], traditional ideal donor criteria (e.g., age
Despite ongoing technological progress, a lack of systematic reviews and evidence-based guidance on optimizing clinical decision-making for cardiomyopathy patients remains. This study reviews technological trends in HT over the past decade and identifies key challenges and future directions. By systematically outlining recent advancements in five key areas—minimally invasive techniques, donor expansion, precision immunosuppression, xenotransplantation, and postoperative management—this study aims to analyze their interconnections and synergistic potential. The goal is to provide a theoretical foundation for constructing an evidence-based, multi-dimensional clinical decision-support system, ultimately shifting transplant outcomes from population-based therapy to individualized, precision medicine.
To systematically trace the evolution of technological paradigms in HT, this study begins by outlining key milestones from 1950s animal experiments (see Table 1, Ref. [8, 9, 10, 11, 12, 13, 14, 15, 16, 17]). The table provides a historical reference for the subsequent discussion, highlighting the position, origins, and significance of key technologies in the field’s development.
| Period | Technical milestone | Key figure/Institution | Significance |
| 1950s | Experimental HT in canines | Richard Lower and Norman Shumway (Stanford University, USA) [8] | Laid the foundation for heart transplant surgery; first demonstration of normal blood circulation restoration using a transplanted heart. |
| 1967 | First human HT | Christiaan Barnard (Cape Town, South Africa) [9] | Initiated human HT; recipient died on post-operative day 18, with autopsy confirming pneumonia as the cause of death. |
| 1980s | Cyclosporine clinical application | Borel and Stähelin (Sandoz Laboratory, Switzerland) | Revolutionized HT by significantly reducing post-operative acute rejection, facilitating global adoption [10]. |
| 1990s | Improved heart preservation (UW solution & portable perfusion) | University of Wisconsin (USA) | Extended donor heart cold ischemia time to 4–6 hours, expanding the donor procurement range. |
| 2000s | Artificial hearts (LVAD/TAH) as a bridge | AbioCor (first TAH, 2001) [11] | Provided a “bridge” for patients awaiting transplantation, extending survival in end-stage heart failure. |
| 2010s | Immunoinduction Therapy (Anti-thymocyte globulin) | International multicenter trials | Reduced early rejection episodes, lowered immunosuppressant dosage, and improved long-term outcomes [12]. |
| 2010s | DCD | Royal Papworth Hospital (UK) | Expanded the donor pool by overcoming the limitations of traditional brain death donation [13, 14]. |
| 2020s | Genetically edited pig-to-human heart transplant (Xenotransplantation) | University of Maryland (USA) | First genetically edited pig heart transplant into a human, offering a novel approach to address donor shortage [15, 16]. |
| Future | 3D bioprinted hearts, Immune tolerance induction strategies | Tel Aviv University, Israel, among others [17] | Exploring pathways for personalized transplantation without the need for donor organs, or potentially eliminating lifelong immunosuppression. |
USA, The United States of America; UK, The United Kingdom of Great Britain and Northern Ireland; LVAD, Left Ventricular Assist Device; TAH, Total Artificial Heart; DCD, Donation after Circulatory Death; UW solution, University of Wisconsin solution; HT, Heart transplantation.
This narrative review systematically summarizes technological advancements in HT from 2015 to 2025. Key publications (e.g., those detailing the first human heart transplant and the clinical use of cyclosporine) were included to provide historical context for the technological development. The literature search was conducted in PubMed and Web of Science databases, using a combination of Medical Subject Headings (MeSH) and free-text keywords, including “HT”, “minimally invasive surgery”, “machine perfusion”, “immunosuppression”, “xenotransplantation”, among others. Reference list tracking of retrieved articles was performed to enhance coverage.
The inclusion criteria encompassed prospective or retrospective clinical studies, systematic reviews, and significant guidelines relevant to HT surgical techniques, donor management, immunosuppressive strategies, or alternative therapies. Case reports, conference abstracts, non-English publications, and pure basic research were excluded. The screening process was conducted independently by two investigators, with discrepancies resolved through arbitration by a third researcher to ensure objectivity and consistency.
For data analysis, thematic analysis was used to synthesize the literature into five core dimensions: (1) Minimally Invasive and Precision Surgery, (2) Donor Procurement and Preservation, (3) Immunosuppression Management, (4) Alternative Transplantation Strategies, and (5) Postoperative Management. This framework facilitated a systematic evaluation of the clinical value, limitations, and future trends of each technology, constructing a multi-layered and comprehensive analytical structure for this review.
HT, the only treatment proven to significantly improve long-term survival in patients with end-stage cardiomyopathy, is often performed in high-risk recipients, such as those with a history of previous cardiac surgery or sternal adhesions. While conventional median Maryland increases the risk of postoperative bleeding and infection in these patients, partial sternotomy has been shown in multicenter studies to effectively reduce surgical trauma, complication rates, and recovery time [18]. Although direct data on HT are limited, evidence from valve and coronary surgeries highlights its potential value: a systematic review and meta-analysis demonstrated that partial sternotomy significantly reduces postoperative blood loss by approximately 79 mL (95% CI –135.78 to –22.96 mL) and lowers transfusion requirements compared to full sternotomy [19].
Building on this, laparoscopic and robotic technologies have been widely adopted in valve surgeries. For example, robotic-assisted mitral valve repair with the da Vinci system significantly shortens hospital stays and reduces postoperative pain [20, 21]. Although robotic-assisted HT has been performed in a limited number of human cases [22], its technical feasibility has been confirmed. However, high equipment costs and a steep learning curve still hinder broader adoption. Meanwhile, minimally invasive Coronary Artery Bypass Grafting (CABG) has demonstrated long-term survival rates comparable to traditional methods, indicating its safety and efficacy [23]. Additionally, minimally invasive approaches in LVAD implantation, such as right anterior thoracotomy or subxiphoid access, have significantly reduced postoperative complications and improved quality of life [24]. This suggests broad applicability of minimally invasive techniques in the surgical management of end-stage heart failure (see Table 2).
| Surgical approach | Technical description | Indicated population |
| Partial Sternotomy | Involves splitting only part of the sternum (e.g., upper or lower segment) | Patients undergoing re-transplantation. |
| Lateral Thoracotomy (Left/Right Anterior) | Surgical approach via intercostal incision, entering the thoracic cavity through a smaller incision | Selected donor-recipient matches (e.g., size compatibility). |
| Robotic-/Video-Assisted Transplantation | Utilizes systems such as the da Vinci platform; small incision for donor heart implantation | Applied in individual case reports; still in the early exploratory stage. |
Overall, the adoption of minimally invasive techniques in HT requires a balance of factors such as operative field exposure, hemodynamic management, and capital investment. Future research should focus on evaluating the feasibility of robotic-assisted surgery in complex re-transplantation cases, optimizing the learning curve, and conducting multicenter long-term follow-up studies. These efforts are essential to advancing HT into an era of precision and minimally invasive care.
3D printing technology has emerged as an important adjunct to conventional imaging and ultrasonography, playing a pivotal role in preoperative planning for HT. Using patient-specific computed tomography (CT) and magnetic resonance imaging (MRI) data, this technology enables the reconstruction of highly individualized cardiac models. It serves as an intuitive visual planning tool for complex cases (e.g., congenital heart defects or re-transplantation), helping to reduce intraoperative adjustment time and shorten postoperative recovery [25, 26].
However, several challenges remain. In terms of materials, commonly used resins and silicones are expensive, and unified clinical standardization and validation are lacking. Technically, whole-organ printing still struggles to achieve fine vascular network construction, integration of electrophysiological functions, and long-term functional maintenance of the printed organ [27, 28]. Looking forward, the integration of AI-assisted modeling, stem cell–scaffold printing, and advances in biodegradable biomaterials may enable 3D bioprinting to gradually achieve the construction of functional cardiac tissue. This direction may advance the technology from experimental models to clinical transplantation, providing a potential pathway to address donor shortage and immune rejection [27, 28].
Globally, HT continues to face a marked mismatch between donor supply and recipient demand. Approximately 20% of patients on the waiting list die before a suitable donor heart becomes available, underscoring the critical severity of the organ shortage [29]. This dilemma primarily stems from the stringent traditional donor selection criteria, which typically restrict donors to under 50 years of age, a cold ischemia time within 4 hours, and no prior cardiac disease [30]. While such standards safeguard transplant outcomes, they substantially limit the available donor pool.
To address this shortage, the marginal donor strategy has been increasingly implemented in clinical practice. For a comparison of traditional and marginal donors, see Table 3 (Ref. [31, 32, 33]). This approach cautiously relaxes certain donor criteria—such as upper age limit and left ventricular ejection fraction (LVEF)—to include hearts that do not fully meet conventional standards but are deemed acceptable following comprehensive risk–benefit evaluation. Several retrospective studies have reported that carefully selected marginal donors do not significantly compromise recipient or five-year allograft survival, supporting the clinical feasibility of this strategy [31].
| Assessment parameter | Traditional donor criteria | Marginal donor criteria & Repair strategies |
| Donor Age | Age |
Physiological age-based extension to |
| Cold Ischemia Time | Novel preservation solutions (e.g., Somah) + machine perfusion (normo-/hypothermic) enabling | |
| Cardiac Function | LVEF |
Multimodal assessment: Stress echocardiography (dobutamine challenge); Coronary angiography to exclude obstructive lesions; Biomarker trends (troponin, BNP) |
| Coronary Artery Disease | Moderate-severe stenosis ( |
Ex vivo repair: PCI with drug-eluting stents for focal lesions; CABG performed ex situ pre-transplant |
| Left Ventricular Hypertrophy | Septal thickness |
Functional compensation assessment: Tissue Doppler (E/e′ ratio); Myocardial strain analysis (GLS |
| Infection Risk | HBV/HCV positivity, bacteremia | Antiviral preconditioning (DAAs for HCV) + rapid molecular pathogen detection (NGS) |
| Drug-Induced Injury | Prolonged high-dose vasopressor support | Ex vivo perfusion assessment: Lactate clearance; Coronary flow/resistance indices; Metabolomic profiling |
BNP, B-Type Natriuretic Peptide; PCI, Percutaneous Coronary Intervention; GLS, Global Longitudinal Strain; E/e′, Early Diastolic Mitral Inflow to Mitral Annular Velocity Ratio; HBV, Hepatitis B Virus; HCV, Hepatitis C Virus; DAAs, Direct-Acting Antivirals; NGS, Next-generation Sequencing.
Although the marginal donor strategy has notably expanded the donor pool, its implementation continues to encounter ethical and regulatory challenges. Future efforts should aim to establish standardized donor risk-stratification models and incorporate multimodal imaging with real-time metabolic parameters—such as lactate clearance and coronary perfusion flow—to enable more precise donor heart assessment and evidence-based clinical decision-making.
Traditional static cold storage (SCS), while reducing the risk of post-transplant cardiac dysfunction, imposes strict limitations on donor heart preservation time. In contrast, NMP systems continuously perfuse the donor heart with oxygenated blood and nutrient-enriched solutions, thereby extending the preservation window and enabling real-time functional assessment through electrocardiographic and metabolic monitoring (comparison between the two preservation techniques is provided in Table 4 (Ref. [33, 34, 35, 36, 37, 38])). This dynamic perfusion approach not only maintains the physiological condition of the donor heart but also offers new opportunities to expand the available donor pool [34] (Fig. 1).
| Parameter | SCS | NMP | Clinical Significance |
| Preservation Mechanism | 4 °C cardioplegic arrest (metabolic suppression) [35] | 37 °C oxygenated blood perfusion (maintenance of aerobic metabolism) | NMP fundamentally eliminates cold ischemic injury. |
| Maximum Safe Preservation Time | Extends preservation window by ~200%, enabling interregional donor heart transport [33]. | ||
| Real-Time Functional Assessment | Not feasible | Feasible (continuous monitoring of ECG, coronary flow, and lactate clearance) | Allows intraoperative evaluation and potential repair of marginal donors. |
| Marginal Donor Utilization Rate | Significantly expands the available donor pool [36, 37]. | ||
| Ex Situ Repair Capability | Absent | Present (e.g., pharmacologic perfusion for microthrombus clearance and anti-inflammatory treatment) | Post-repair 3-year survival rate significantly improved [38]. |
| PGF Incidence | 10%–15% | Reduces the risk of PGF by 67% [34]. |
ECG, Electrocardiogram; PGF, primary graft failure; SCS, static cold storage.
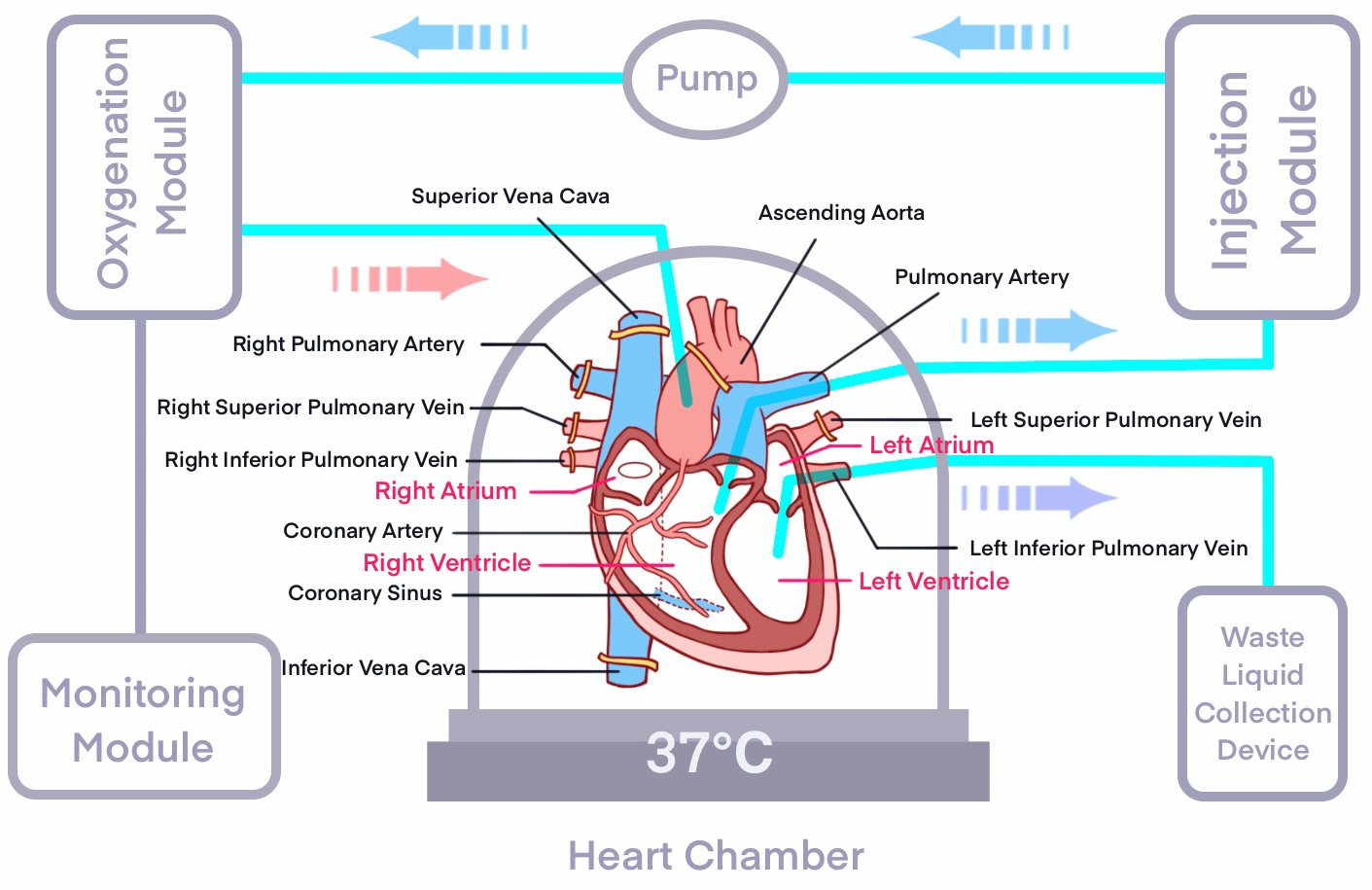 Fig. 1.
Fig. 1.
Schematic Diagram of NMP. NMP, normothermic machine perfusion.
Several studies have demonstrated that NMP effectively preserves myocardial function while improving overall organ quality [39]. During perfusion, the administration of pharmacologic or anticoagulant agents can eliminate microthrombi and mitigate reperfusion injury, thereby reducing transplant-related complications. Ultrastructural analyses have confirmed that hearts preserved under NMP maintain superior integrity of myocardial fibers, endothelial cells, and mitochondria, exhibiting less tissue injury compared to conventional cold storage [40].
Beyond preservation, the application of NMP is progressively expanding toward ex vivo functional assessment and repair prior to transplantation. Under perfusion conditions, quantitative evaluation of contractile performance, metabolic activity, and tissue responsiveness provides an objective basis for assessing donor heart viability—particularly beneficial for the utilization of marginal donors [39, 41, 42].
In summary, NMP offers distinct advantages in extending preservation duration, enhancing donor organ quality, and improving post-transplant outcomes, representing a promising technological advancement in the field of HT.
Despite its demonstrated benefits, broader clinical adoption of NMP remains limited by high equipment costs, procedural complexity, and the lack of large-scale, multicenter, long-term outcome data. Future research should prioritize comprehensive economic analyses and cost-effectiveness evaluations to support the wider implementation of NMP in routine clinical practice.
Modern HT allocation systems—such as the six-tier priority framework implemented by the Organ Procurement and Transplantation Network (OPTN)/UNOS in the United States—have evolved from traditional “first-come, first-served” or proximity-based models to sophisticated stratification schemes guided by medical urgency and donor–recipient matching quality [43]. This algorithm comprehensively integrates multiple parameters, including ABO compatibility, weight ratio, Human Leukocyte Antigen (HLA) sensitization status, donor–recipient age difference, and geographic distance. Its overarching objective is to prioritize life-saving transplants for critically ill patients while optimizing long-term post-transplant survival outcomes.
Empirical evidence demonstrates that this refined allocation strategy has significantly improved the overall efficiency of the transplantation system. Following the implementation of the six-tier priority system, one of the most immediate effects was the enhanced treatment efficiency for critically ill patients. Studies have shown a marked reduction in waiting list mortality, particularly among patients requiring temporary Mechanical Circulatory Support (tMCS), whose likelihood of receiving a transplant has substantially increased [44]. This underscores the system’s success in achieving its primary goal—prioritizing the sickest candidates.
Beyond medical urgency, optimization of donor–recipient matching quality represents another core strength of the new framework. Rather than focusing solely on “faster allocation”, the system emphasizes “better matching”. For instance, the integration of predictive tools such as Predicted Heart Mass has improved donor–recipient size matching, thereby enhancing postoperative hemodynamics and long-term survival [45]. In complex cases such as congenital heart disease and cardiomyopathy, this quality-based stratification has demonstrated clear value, significantly reducing waiting list mortality in these high-risk populations [46].
Importantly, the revised system has also contributed to improved equity in organ allocation, though challenges persist. Policy changes have led to measurable progress in transplant access across demographic groups. Following implementation, survival among Black recipients has improved, with favorable trends also observed in acute rejection rates [47]. However, disparities remain for certain subgroups—for example, patients with blood type O continue to experience relatively longer waiting times, despite partial improvement [48]. This indicates that while efficiency has improved, eliminating inherent inequities in organ allocation remains an ongoing effort.
In summary, modern heart allocation systems—through nuanced stratification models—have achieved substantial advances in allocation efficiency, matching precision, and distribution equity. Future development is expected to be increasingly data-driven, integrating machine learning and predictive analytics to model individual waiting-list mortality risk and post-transplant survival benefit. Such approaches may ultimately enable the optimal balance between maximizing survival benefit and ensuring social equity in the allocation of scarce donor resources.
Post-transplant immunosuppressive therapy in HT is undergoing a paradigm shift—from empirical, broad-spectrum regimens toward precision-based and individualized management, and ultimately toward the induction of immune tolerance [49]. The core of personalized immunosuppression lies in the dynamic assessment and intervention of immune status and drug response through multidimensional biomarkers, thereby minimizing the risk of rejection while reducing drug-related toxicity.
In individualized immunosuppressive strategies, genotype-guided therapy plays a pivotal role. By analyzing pharmacogenomic variants such as single nucleotide polymorphisms (SNPs) in CYP3A5 and ABCB1, clinicians can predict individual metabolic capacity and toxicity thresholds for tacrolimus, enabling dose optimization and precise drug delivery [50, 51]. This approach helps prevent both under-immunosuppression and drug accumulation–related adverse effects, thereby laying the foundation for long-term graft survival.
Simultaneously, advances in rejection monitoring are transforming clinical practice. The dd-cfDNA has emerged as a novel, noninvasive biomarker capable of detecting subclinical allograft injury before histologic rejection becomes apparent. Elevations in dd-cfDNA correlate closely with the severity of both cellular and antibody-mediated rejection [52, 53]. Studies demonstrate that dd-cfDNA testing provides high sensitivity and specificity for early rejection detection, offering a valuable early warning signal to guide timely therapeutic adjustment of immunosuppressive regimens [54].
Regarding toxicity control, prolonged use of immunosuppressants may lead to infections, renal dysfunction, and metabolic disturbances. Therefore, dynamic optimization of drug combinations and dosing—guided by individual pharmacokinetics, immunologic risk stratification, and adverse effect monitoring—is essential to achieve a balance between rejection prevention and toxicity mitigation [55].
Beyond precision suppression, current research is progressively shifting toward immune tolerance induction through regulatory T-cell (Treg) and donor cell–mediated immune reconstruction [56]. Tregs play a critical role in maintaining graft tolerance. Ex vivo expansion and autologous reinfusion of Tregs can suppress excessive immune activation and establish a localized tolerogenic microenvironment, reducing rejection episodes and enabling tapering of immunosuppressive therapy [56]. Similarly, donor bone marrow infusion can induce a state of mixed chimerism in the recipient, promoting Treg expansion and immune reprogramming toward donor-specific tolerance [56, 57].
With the rapid development of bioengineering and nanotechnology, innovative approaches such as Programmed Death Ligand-1 (PD-L1)–modified nanoparticles and immunomodulatory scaffolds are emerging as promising tools for graft-specific immune regulation. These technologies may enable localized immune balance with reduced systemic immunosuppression, offering new opportunities to improve long-term cardiac allograft survival [58, 59].
Nevertheless, achieving stable clinical immune tolerance remains a major challenge. The central dilemma lies in maintaining sufficient immunosuppression to prevent rejection while minimizing drug toxicity. Future studies should focus on rigorously designed multicenter clinical trials exploring combinatorial approaches—integrating cellular therapy, biomaterials, and gene-editing technologies—to ultimately achieve true personalized immune equilibrium.
Gene-editing technologies—particularly the CRISPR-Cas9 system—have revolutionized the field of xenotransplantation. Recent studies have demonstrated that CRISPR-Cas9 enables the generation of multi-gene–edited donor pigs whose organs exhibit markedly prolonged survival in non-human primates [60]. Key genetic modifications include knockout of GGTA1, CMAH, and B4GALNT2, which are responsible for the biosynthesis of major xenoantigens [61]. These edits substantially attenuate human immune recognition and rejection responses against porcine organs [16]. Moreover, CRISPR-Cas9 facilitates the development of novel immunomodulatory strategies, such as co-stimulation blockade using anti-CD40/CD40L monoclonal antibodies, to further extend xenograft survival [62].
Preclinical studies involving the transplantation of 9- and 10-gene–edited pig hearts into baboons, combined with co-stimulation blockade–based immunosuppressive regimens and optimized cold perfusion preservation, have achieved remarkable outcomes—some grafts maintained normal function for several hundred days [63]. These results provide compelling experimental evidence supporting the feasibility of genetically engineered porcine hearts as potential donor sources for end-stage heart failure patients.
Building on these preclinical successes, in 2022, researchers at the University of Maryland performed the world’s first clinical transplantation of a CRISPR-Cas9–engineered, multi-gene–edited pig heart into a human recipient. The transplanted heart maintained adequate function for 60 days, demonstrating the clinical feasibility of overcoming hyperacute rejection through genetic engineering and representing a milestone in cross-species cardiac transplantation [64]. Nevertheless, larger-scale studies and long-term follow-up remain necessary to confirm the reproducibility and safety of this approach.
Despite these breakthroughs, several barriers continue to hinder clinical
translation. These include the need to eliminate hyperacute rejection (e.g., via
Current progress is further constrained by the limited availability of long-term clinical data, the substantial economic costs, and the absence of harmonized regulatory frameworks. Future efforts should prioritize the development of standardized multi-gene editing protocols, comprehensive evaluation of viral safety and cross-species immune compatibility, and establishment of clear recipient selection criteria and post-transplant management guidelines. With continued advances in gene editing, immunoregulation, and bioengineering, xenotransplantation holds considerable promise as a potential solution to the global donor organ shortage. However, its long-term safety, immunological integration, and ethical acceptability will require sustained, multidisciplinary evaluation.
In recent years, the clinical use of TAHs and Ventricular Assist Devices (VADs) as bridge therapies to HT has increased markedly. This trend primarily reflects the persistent shortage of donor hearts, together with continuing advances in extracorporeal circulation and device technologies [68]. Importantly, for certain patients with end-stage heart failure who are ineligible for transplantation due to advanced age, comorbidities, or contraindications, TAH and VAD therapies have transitioned from temporary support measures to feasible destination therapies, providing viable long-term treatment alternatives [2].
Parallel advancements in materials science and engineering have substantially improved the miniaturization, biocompatibility, and durability of both TAH and LVADs. These innovations have reduced surgical trauma, enhanced postoperative mobility and social reintegration, and significantly decreased complication rates and mortality [69].
Currently, LVADs constitute the most widely implemented bridge-to-transplantation strategy. Clinical studies indicate that LVAD implantation stabilizes hemodynamics and improves quality of life during the waiting period for transplantation; however, device-related infections, thromboembolic events, and bleeding remain key complications [70, 71]. In parallel, Extracorporeal Membrane Oxygenation (ECMO) serves as a rapid, short-term bridge for patients in cardiogenic shock. Although associated with a higher risk of postoperative morbidity in prolonged use, ECMO demonstrates favorable outcomes as a direct bridge when transplant waiting times are brief [72, 73].
Emerging surgical innovations are expanding the boundaries of MCS. Partial Heart Transplantation (PHT), primarily explored in pediatric recipients, allows the transplanted cardiac tissue to grow synchronously with the host, substantially reducing the need for repeat surgeries while minimizing the immunologic burden [74]. Moreover, recent studies suggest that combining VAD support with cell-based regenerative therapies may promote reverse myocardial remodeling and functional recovery [75]. In select cases, sufficient myocardial recovery under such combined therapy could obviate the need for transplantation altogether.
Despite these advances, artificial heart technology continues to face major challenges, including limited long-term device durability, the need for further optimization of hemocompatibility, and prevention of thromboembolic complications. Future development should aim to establish a comprehensive “bridge–transplant–integration” continuum, integrating MCS with regenerative medicine approaches. Such a strategy may enable personalized, staged cardiac functional recovery, thereby reshaping the therapeutic paradigm for advanced heart failure.
Postoperative surveillance remains a cornerstone for ensuring graft success and long-term survival following HT. Traditionally, monitoring has relied on invasive methods such as endomyocardial biopsy. However, recent advances in noninvasive imaging and biomarker technologies have transformed postoperative management, offering safer, more dynamic, and more patient-centered approaches.
High-frequency echocardiography is widely utilized as a noninvasive tool for functional assessment. Advanced ultrasound modalities—including strain imaging and tissue Doppler imaging—allow precise quantification of myocardial deformation, facilitating early detection of rejection and other post-transplant complications [76].
In parallel, cardiac magnetic resonance (CMR) with T1 and T2 mapping has demonstrated unique advantages in myocardial tissue characterization. T1 mapping quantitatively reflects interstitial edema and fibrosis, enabling differentiation between acute and chronic rejection, while its high negative predictive value in several studies markedly reduces the need for routine biopsy [77]. T2 mapping, by quantifying myocardial edema, serves as an additional sensitive indicator for acute rejection and offers critical guidance for timely therapeutic intervention [78].
Beyond imaging, circulating biomarkers are gaining increasing attention. Exosomal microRNAs (miRNAs) have emerged as promising candidates, given their regulatory roles in myocardial fibrosis and cardiac remodeling. Although a validated circulating miRNA signature for heart transplant monitoring is not yet established, preliminary studies highlight their potential as sensitive, noninvasive markers of graft injury [79].
Simultaneously, advances in digital health technologies—such as implantable hemodynamic monitoring systems—now enable continuous measurement of pulmonary artery pressure. These systems support real-time clinical decision-making, reduce hospitalizations, and mitigate episodes of acute decompensated heart failure [80].
Looking forward, cardiac regenerative medicine is redefining the frontier of postoperative management. Stem cell–based and gene therapies have demonstrated encouraging preliminary results in promoting myocardial repair and improving ventricular function. Although robust clinical validation remains limited, integration of these regenerative strategies with gene-editing and bioengineering technologies may pave new therapeutic pathways for post-transplant myocardial recovery and long-term graft function optimization [81] (Fig. 2).
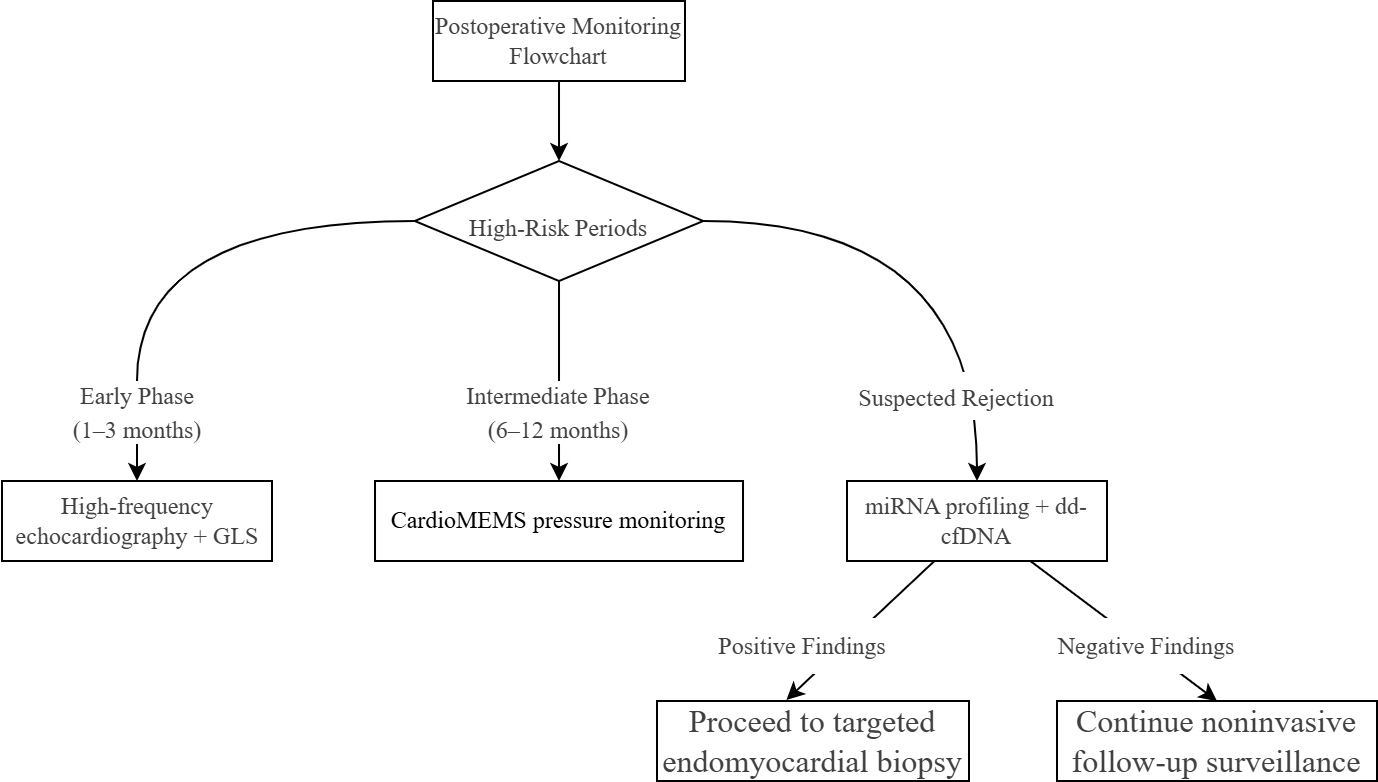 Fig. 2.
Fig. 2.
Non-invasive testing technology application pathway.
In pediatric HT for cardiomyopathy, donor–recipient size matching and immune tolerance induction are critical determinants of clinical outcomes. In recent years, ABO-incompatible (ABO-I) transplantation has been increasingly adopted in infants and young children. This approach takes advantage of the immunologic immaturity of infants, allowing successful acceptance of hearts from donors with different blood groups without precipitating severe rejection. Multiple studies have demonstrated that post-transplant survival following ABO-I transplantation is comparable to that of ABO-compatible procedures, thereby expanding transplant access for pediatric patients with cardiomyopathy [82].
With regard to long-term outcomes, the survival of pediatric heart transplant recipients remains influenced by a constellation of factors. Major barriers to durable graft survival include malignancy, rejection, and CAV—the latter representing a leading cause of late graft failure and mortality [83, 84]. Consequently, preventive and therapeutic strategies targeting these high-risk determinants are essential for improving long-term prognosis.
During the donor waiting period, MCS serves as an established bridge-to-transplantation strategy in pediatric populations. Evidence indicates that MCS significantly enhances waitlist survival, particularly among patients with congenital heart disease [85].
Post-transplant immune management is equally pivotal. For sensitized pediatric patients with high preformed antibody levels, desensitization therapy—including plasmapheresis, intravenous immunoglobulin (IVIG), and rituximab—has demonstrated efficacy in reducing antibody titers and improving transplant success rates [86]. In parallel, immunomodulatory strategies directed at mitigating CAV are evolving. Although no single therapeutic regimen has yet been proven to substantially prolong graft longevity, the combined application of immunosuppressive and anti-inflammatory agents to attenuate immune-mediated vascular injury remains a cornerstone of current management [87]. Furthermore, routine surveillance for donor-specific antibodies (DSA), particularly in patients with a history of recurrent rejection, has been associated with improved graft outcomes [88].
Despite notable progress in surgical techniques and immunologic management, pediatric HT continues to face persistent challenges. Donor shortage remains the primary bottleneck restricting broader clinical implementation. Future research should prioritize refining indications for ABO-I transplantation, improving donor–recipient size-matching algorithms, and developing precision immunomonitoring frameworks tailored to pediatric immune profiles. Additionally, strategies aimed at slowing CAV progression and enhancing long-term survival remain central to ongoing clinical and translational efforts. With the integration of cell-based therapy, gene editing, and regenerative medicine, pediatric HT is poised to achieve transformative advancements in both safety and long-term graft function.
The field of HT is entering a pivotal phase of transformation driven by technological integration and individualized therapeutic approaches. Minimally invasive and robot-assisted techniques have shown promise in reducing surgical trauma and perioperative complications, while the implementation of NMP and systematic marginal donor assessment has substantially expanded the donor pool and prolonged viable preservation time. Concurrently, genotype-guided immunosuppressive therapy combined with emerging biomarker-based monitoring is advancing toward precision and dynamic immune regulation. In parallel, xenotransplantation has achieved encouraging early clinical outcomes, and MCS technologies are becoming increasingly refined. The convergence of non-invasive imaging modalities with regenerative medicine further introduces novel avenues for post-transplant functional recovery.
Despite these remarkable advances, several challenges remain. The widespread clinical adoption of these innovations continues to be limited by the lack of long-term outcome data, incomplete procedural standardization, high economic burden, and lagging ethical and regulatory frameworks. Future efforts should focus on establishing standardized protocols for normothermic perfusion, accelerating the clinical translation of immune tolerance strategies, developing comprehensive ethical and biosafety frameworks for xenotransplantation, and strengthening multicenter collaboration through real-world data integration. Collectively, these initiatives will be essential to drive the paradigm shift in HT—from merely prolonging survival to genuinely enhancing post-transplant quality of life.
Conceptualization was performed by JYC and SXH; methodology was designed by JYC, SXH, HSW, and ZWL; validation was carried out by ZWL and YGF; investigation was conducted by YGF; resources were provided by CZL; the original draft was written by SXH and JYC; review and editing of the manuscript were undertaken by JYC and HSW; visualization was prepared by JYC; supervision was undertaken by YGF; project administration was managed by ZWL; funding acquisition was secured by YGF All authors have reviewed and approved the final version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


