1 Department of Cardiac and Vascular Surgery, Beijing Tiantan Hospital, Capital Medical University, 100070 Beijing, China
Abstract
Atrial fibrillation (AF) is associated with an increased risk of thromboembolism, primarily due to thrombus formation in the left atrial appendage (LAA). While anticoagulation represents the standard therapy, patients with a high risk of bleeding require alternative strategies such as LAA clipping (LAAC); hypotension has been observed in some patients post-LAAC. This study aimed to investigate the hypotensive effect after LAAC in patients with AF, and to explore the potential mediating role of B-type natriuretic peptide (BNP).
A retrospective single-center analysis was conducted on 99 patients who underwent a standalone totally thoracoscopic LAAC. Plasma BNP levels, aldosterone, electrolyte levels, and blood pressure were measured preoperatively and at defined intervals postoperatively. Echocardiographic parameters and medication use were also analyzed.
A significant increase in BNP level was observed following LAAC (150.9 pg/mL [interquartile range (IQR) 77.8–260.7] to 316.2 pg/mL [IQR 197.5–466.8]; p < 0.001), accompanied by a significant decrease in aldosterone (5.5 ng/dL [IQR 3.0–8.7] to 2.5 ng/dL [IQR 1.4–3.4]; p < 0.001). Moreover, LAAC was associated with a marked reduction in systolic (127.3 ± 12.3 mmHg to 116.9 ± 12.4 mmHg; p < 0.001) and diastolic (80.7 ± 7.7 mmHg to 67.4 ± 9.3 mmHg; p < 0.001) blood pressure. Significant decreases in serum sodium and increases in serum potassium were also noted. A significant reduction in the use of angiotensin converting enzyme inhibitor (ACEI)/angiotensin receptor blocker (ARB) and calcium channel blocker (CCB) antihypertensive agents was observed postoperatively. No significant changes in cardiac structure or function were detected. The decrease in blood pressure persisted at 6-month follow-up.
LAAC-induced hypotension may be partly mediated by BNP-driven suppression of aldosterone, thereby promoting natriuresis and reduced blood volume. These findings highlight the need for postoperative hemodynamic monitoring and antihypertensive medication adjustment in AF patients who undergo LAAC.
Keywords
- atrial fibrillation
- left atrial appendage clipping
- blood pressure
- B-type natriuretic peptide
- aldosterone
- surgery
Atrial fibrillation (AF) is a common cardiac arrhythmia with an overall
prevalence of 1–2%. The incidence of AF increases by 1% annually with advancing
age, reaching 15% in individuals aged
A retrospective analysis was conducted on patients who underwent totally thoracoscopic LAAC at Beijing Tiantan Hospital, Capital Medical University, between January 2020 and May 2025. A total of 99 patients were included in this study, comprising 71 males (71.7%) and 28 females (28.3%). Among them, 69 patients (69.7%) had a history of cerebral infarction, 12 (12.1%) had cerebral hemorrhage, and 75 (75.8%) had hypertension. Detailed demographic and clinical characteristics of the study cohort are presented in Table 1.
| Variable | Value | |
| Age (years) | 66.1 | |
| Gender (male) | 71 (71.7%) | |
| Height (cm) | 170 (162, 175) | |
| Weight (kg) | 72.1 | |
| CHA2DS2-VASc score | 5 (4, 6) | |
| HAS-BLED score | 4 (2, 5) | |
| Cerebral infarction | 69 (69.7%) | |
| Cerebral hemorrhage | 12 (12.1%) | |
| Type of atrial fibrillation | ||
| Paroxysmal | 11 (11.1%) | |
| Persistent | 56 (56.6%) | |
| Permanent | 32 (32.3%) | |
| Coronary artery disease | 30 (30.3%) | |
| Myocardial infarction | 4 (4.0%) | |
| Hypertension | 75 (75.8%) | |
| Peripheral vascular disease | 82 (82.8%) | |
| Type 2 diabetes mellitus | 31 (31.3%) | |
Note: Data is presented as the mean
The study enrolled patients aged
All enrolled patients were deemed unsuitable for rhythm control therapy based on
three primary clinical considerations: (1) the predominance of long-standing,
persistent, and permanent AF; (2) documented failure of prior anti-arrhythmic
drug therapy; and (3) patients with permanent AF (
The patient was placed in the right lateral decubitus position with the left
upper limb suspended. General anesthesia was induced with double-lumen
endotracheal intubation, and invasive monitoring was established via radial
arterial line and central venous catheterization. Three thoracic ports were
created: first, a 5-mm trocar at the 4th intercostal space (ICS), anterior
axillary line (operative port); second, a 5-mm trocar at the 5th ICS, posterior
axillary line (operative port); and finally, a 10-mm trocar at the 6th ICS,
anterior axillary line (thoracoscopic port). An additional 2-cm incision was made
at the 7th ICS, posterior axillary line for LAA clip delivery. Upon entering the
thoracic cavity, the lung was retracted posteriorly using a peanut dissector to
expose the left pericardium and phrenic nerve. The pericardium was incised 1 cm
posterior to the phrenic nerve using electrocautery, extending from the inferior
border of the left pulmonary artery to the base of the LAA. The pericardium was
then suspended to fully expose the LAA. The root diameter of the LAA was
measured, and an appropriately sized C-Clip® device (Med-zenith,
Inc., Beijing, China) was selected. The clip was introduced through the 2-cm
incision, looped over the LAA apex, and positioned at the LAA base. It was
deployed gradually under continuous hemodynamic and electrocardiographic
monitoring. Final release was performed only after meeting the following criteria
under transesophageal echocardiographic (TEE) guidance: (1) absence of
significant ECG changes, (2) complete cessation of LAA flow on color Doppler
imaging, and (3) confirmation of clip placement at the LAA base with a residual
stump length of
The patient’s blood pressure was measured each morning while they were quiet in the ward. Three measurements were taken to record the mean blood pressure value. Bilateral upper limb blood pressure was measured, and the blood pressure of the higher limb was also recorded. Arterial pressure was systematically assessed at the following predefined peri- and post-operative intervals: preoperatively, on the day of surgery, and on the first, second, and third postoperative days, concluding with a final measurement prior to discharge. As part of the long-term follow-up, blood pressure was again recorded during the outpatient clinic visit at 6 months post-discharge.
Transthoracic echocardiography was performed using a GE Vivid E95 ultrasound system (GE Healthcare, Chicago, IL, USA) to obtain standardized measurements of left atrial anteroposterior diameter (LAAPD), ejection fraction (EF), and left ventricular end-diastolic diameter (LVEDD) at baseline (preoperative) and 1-week postoperative time points. All examinations were conducted with patients in the resting supine position following standard protocols. The following measurements were obtained from the parasternal long-axis view: left atrial diameter was measured at end-systole as the maximal vertical distance between the anterior and posterior walls of the left atrium, perpendicular to the long axis of the ventricle. Left ventricular internal diameter was measured at end-diastole, defined as the frame immediately preceding mitral valve closure.
For each parameter, measurements were performed across three consecutive cardiac cycles that demonstrated optimal visualization of cardiac structures. The mean value of these measurements was calculated and used for subsequent analysis to minimize beat-to-beat variability. All measurements were performed offline by an experienced sonographer using dedicated analysis software (EchoPAC, GE Healthcare, Chicago, IL, USA).
Fasting venous blood samples were collected preoperatively, on the operative day, postoperative days 1–3, and at 1 week after surgery. After centrifugation, plasma samples were sent to the central laboratory for analysis. Plasma BNP and aldosterone levels were measured by chemiluminescence immunoassay using an automated biochemical analyzer, while serum sodium and potassium concentrations were determined by spectrophotometry.
The use of medications was captured through the extraction of electronic health records and direct patient interviews. Medication profiles documenting antihypertensives, anticoagulants, and antiplatelet agents were systematically recorded at preoperative baseline, as well as 1-week and 6-month post-LAAC.
All statistical analyses were performed using SPSS 22.0 (IBM Inc., Chicago, IL,
USA). Normally distributed continuous variables were presented as the mean
This study enrolled 99 patients comprising 71 males (71.7%) and 28 females
(28.3%), with a mean age of 66.1
Comparative analysis revealed significant postoperative changes across multiple
physiological parameters. BNP levels showed a marked elevation from preoperative
levels of 150.9 pg/mL (IQR 77.8–260.7) to 316.2 pg/mL (IQR 197.5–466.8)
(p
| Variable | Pre-operative | Post-operative | t/Z | p |
| BNP (pg/mL) | 150.9 (77.8, 260.7) | 316.2 (197.5, 466.8) | –5.937 | |
| Aldosterone (ng/dL) | 5.5 (3.0, 8.7) | 2.5 (1.4, 3.4) | 5.155 | |
| Systolic BP (mmHg) | 127.3 |
116.9 |
5.949 | |
| Diastolic BP (mmHg) | 80.7 |
67.4 |
12.497 | |
| Serum sodium (mmol/L) | 142.0 |
137.6 |
12.992 | |
| Serum potassium (mmol/L) | 3.9 |
4.1 |
–4.207 |
Note: Data, mean
Pre-discharge echocardiographic evaluation revealed no significant changes in
cardiac structure or function compared to preoperative measurements. The left
ventricular end-diastolic diameter (LVEDD) remained stable (46.3
| Transthoracic echocardiography parameters | Pre-operative | Discharge | t | p |
| LVEDD (mm) | 46.3 |
46.2 |
0.484 | 0.629 |
| LAAPD (mm) | 40.7 |
40.0 |
1.864 | 0.066 |
| EF (%) | 61.1 |
60.8 |
0.135 | 0.893 |
Note: Data, mean
Pharmacological analysis demonstrated significant postoperative reductions in
the utilization of ACEI/ARB (33.3% to 13.1%, p = 0.001) and CCB
(29.2% to 16.1%, p = 0.027) after 7 days (Table 4). The use of
anticoagulants (e.g., warfarin and DOACs), antiplatelets (e.g., aspirin and
aspirin+clopidogrel), and amiodarone regimens remained largely unchanged (all
p
| Medication Class | Pre-op (n = 99) | Post-op day 7 (n = 99) | p | ||
| 6 months (n = 97) | |||||
| Anticoagulants | |||||
| Warfarin | 4 (4.0%) | 0 (0%) | 4.082 | 0.121 | |
| 0 (0%) | 4.001 | 0.120 | |||
| DOACs | 50 (50.5%) | 49 (49.5%) | 0.020 | 0.887 | |
| 49 (50.5%) | 0.000 | 0.999 | |||
| Antiplatelets | |||||
| Aspirin | 49 (49.4%) | 44 (44.4%) | 0.507 | 0.476 | |
| 43 (44.3%) | 0.525 | 0.478 | |||
| Aspirin+Clopidogrel | 6 (6.1%) | 1 (1.0%) | 3.702 | 0.118 | |
| 1 (1.0%) | 3.599 | 0.116 | |||
| Antihypertensives | |||||
| ACEI/ARB | 33 (33.3%) | 13 (13.1%) | 11.327 | 0.001 | |
| 15 (15.5%) | 8.460 | 0.005 | |||
| Beta-blockers | 64 (64.6%) | 63 (63.6%) | 0.022 | 0.882 | |
| 61 (62.8%) | 0.066 | 0.880 | |||
| CCB | 29 (29.2%) | 16 (16.1%) | 4.860 | 0.027 | |
| 20 (20.6%) | 1.966 | 0.161 | |||
| Others | |||||
| Amiodarone | 18 (18.1%) | 19 (19.1%) | 0.033 | 0.855 | |
| 17 (17.5%) | 0.014 | 0.905 | |||
Note: Data presented as n (%); DOACs, new oral anticoagulants; ACEI, Angiotensin Converting Enzyme Inhibitor; ARB, Angiotensin Receptor Blocker; CCB, Calcium Channel Blocker.
Binary logistic regression analysis was performed to identify independent
factors associated with changes in blood pressure. The Hosmer-Lemeshow test
revealed the model had a good fit to the data (
The dynamic changes in blood pressure following the LAAC procedure are
summarized in Fig. 1. A clear postoperative reduction in both systolic and
diastolic blood pressure was observed. The trend analysis for systolic blood
pressure (SBP) revealed a nadir on postoperative day 3 (POD3), while the peak
decline in diastolic blood pressure (DBP) occurred earlier on postoperative day 1
(POD1). Statistical analysis using repeated measures ANOVA further confirmed
these trends, as illustrated by the plots of the estimated marginal means.
Critically, the follow-up data at 6 months demonstrated a persistent and
statistically significant reduction in both SBP and DBP compared to preoperative
baseline levels (p
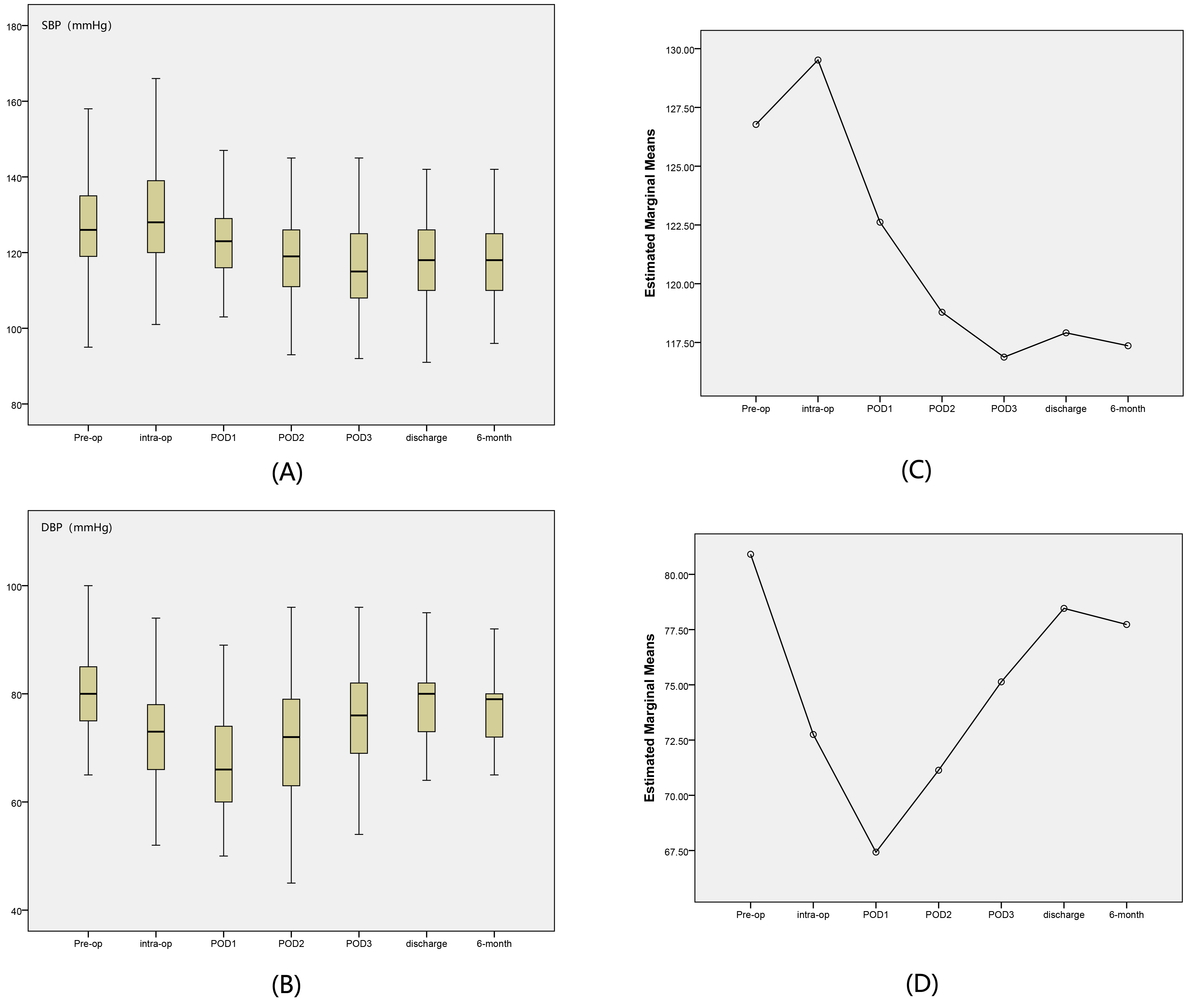 Fig. 1.
Fig. 1.
Blood pressure dynamics before and after LAAC. (A) Trend for
SBP, showing peak SBP reduction at POD3. (B) Trend for DBP, showing peak decline
in DBP at POD1. (C) Estimated marginal means for SBP from repeated measures
ANOVA. (D) Estimated marginal means for DBP from repeated measures ANOVA.
Follow-up to 6 months showed that both SBP and DBP were significantly lower than
preoperative levels (p
The principal thromboembolic complications of AF stem from left atrial thrombus
formation and subsequent systemic embolization, manifesting as cerebral
infarction, peripheral arterial embolism, or visceral organ ischemia [13].
Current pharmacological prophylaxis relies on anticoagulant therapy, including
warfarin and DOACs such as rivaroxaban and edoxaban [14, 15]. However, patients
with an elevated risk of bleeding (HAS-BLED score
Hemodynamic studies have shown that approximately 90% of left atrial thrombi originate in the LAA due to blood stasis during AF. This pathophysiological mechanism supports LAA closure as a targeted intervention, where physical isolation of the appendage from the circulatory system achieves thromboprophylaxis [17]. Totally thoracoscopic LAAC represents a minimally invasive surgical technique that results in complete anatomical exclusion of the LAA, while avoiding the morbidity associated with open cardiac procedures [18].
Our findings align with previous research by Turagam et al. [12], who compared 247 patients undergoing LAAC with 124 patients receiving transcatheter left atrial appendage occlusion (LAAO). These authors found a significant postoperative decrease in blood pressure in the LAAC group compared to the LAAO group, which persisted for more than a year [12]. Similarly, Maybrook et al. [19] observed a postoperative decrease in blood pressure in a cohort of 76 patients undergoing percutaneous LAA ligation. Both authors postulated that BNP may be a key regulatory mediator in the reduction of blood pressure, although neither investigated the specific underlying mechanisms. The LAA possesses endocrine function, with its primary secretory product being BNP. In humans, the majority of BNP is derived from atrial and ventricular tissues. When atrial pressure increases, the appendage releases BNP into the bloodstream to regulate water and sodium metabolism. Elevated BNP levels inhibit aldosterone production, leading to reduced levels of systemic aldosterone [20]. Decreased aldosterone levels subsequently promote sodium retention, water reabsorption, and potassium excretion, leading to enhanced sodium excretion, increased urine output, and reduced blood volume, potentially contributing to lower blood pressure [21]. In addition, the vasodilatory effects of BNP can further reduce blood pressure [22]. The proposed mechanism for LAAC-induced blood pressure reduction is shown in Fig. 2.
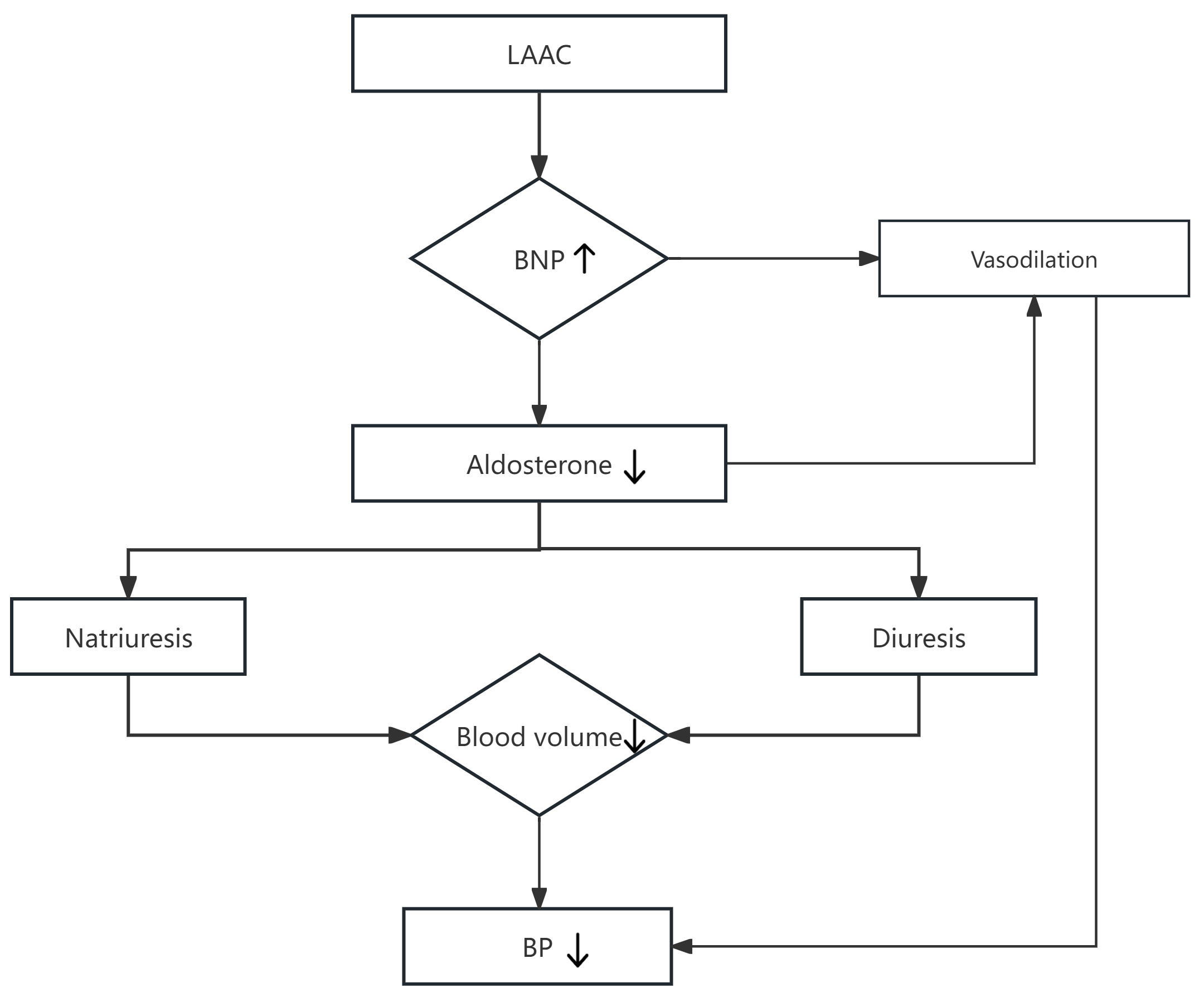 Fig. 2.
Fig. 2.
Potential mechanistic pathway for LAAC-induced reduction of blood pressure. Down arrows: After LAAC, LAA releases BNP into the bloodstream. Elevated BNP promotes vasodilation and inhibits aldosterone production. The decrease in aldosterone levels subsequently promotes natriuresis and diuresis, leading to reduced blood volume and ultimately contributing to lower blood pressure.
The current study found that BNP levels increased significantly after LAA clip
placement (150.9 [IQR 77.8, 260.7] vs. 316.2 [IQR 197.5, 466.8] pg/mL, p
BNP has been shown to decrease the expression of aldosterone synthase mRNA, thus
reducing the production of aldosterone [26]. In the present study, the increase
in BNP led to a reduction in aldosterone (5.5 [IQR 3.0, 8.7] vs. 2.5 [IQR 1.4,
3.4] ng/dL, p
The significant postoperative reductions in the use of ACEI/ARB (33.3% to 13.1%, p = 0.001) and CCB (29.2% to 16.1%, p = 0.027) likely reflect the proactive management of LAAC-induced hemodynamic alterations by clinicians. However, our binary logistic regression analysis indicated that adjustments in these antihypertensive medications were not independent predictors of reduced blood pressure. Given the association of the LAAC procedure with BNP-mediated volume depletion and vasodilation, continued administration of antihypertensive agents could risk potentiating severe hypotension. Conversely, the continued use of beta-blockers (64.6% to 63.6%, p = 0.882) prioritizes ventricular rate control without exacerbating a reduction in vascular tone. These coordinated adjustments in medication represent a meticulous clinical response to LAAC-induced hypotension effects. At the 6-month follow-up, the use of ACEI/ARB remained lower than the preoperative level, while the use of CCB approached preoperative rates. Collectively, this demonstrates a reduced intensity of antihypertensive medication compared with the preoperative period.
Furthermore, LAAC did not significantly alter cardiac structure [27], as evidenced by unchanged left ventricular end-diastolic diameter, left atrial anteroposterior diameter, and left ventricular ejection fraction compared to preoperative measurements. These findings suggest that LAAC does not impair cardiac systolic function or left atrial architecture, indicating the observed decrease in blood pressure is unlikely to be related to cardiac functional changes.
The hemodynamic alterations following LAAC were characterized by a distinct
pattern of blood pressure changes. The most pronounced reduction in SBP was
observed on postoperative day 3. Although a partial recovery was noted by the
time of discharge, SBP levels remained lower than preoperative baseline values.
In contrast, the peak decline in DBP occurred earlier, on postoperative day 1,
with DBP returning to near-baseline levels upon discharge. Severe hypotension,
defined as SBP
The male predominance of our cohort aligns with the epidemiology of AF in older patients undergoing invasive interventions, but limits the generalizability of the results to women [28]. Moreover, our cohort had a high proportion of patients with a history of stroke, which is characteristic of the elevated thromboembolic and bleeding risk profile typical of candidates for LAAC.
We acknowledge that the hemodynamic changes observed in this study are likely to be multifactorial. The significant postoperative reduction in ACEI/ARB and CCB use is undoubtedly due to the decline in blood pressure. However, the rapid and substantial rise in BNP and fall in aldosterone occurred in the immediate postoperative period, temporally preceding or coinciding with adjustments in medication. This sequence suggests that hormonal changes may be the primary instigator, with drug de-escalation representing a prudent clinical response to the observed hemodynamic shifts. Furthermore, other perioperative factors, including anesthesia, fluid balance, and the physiological stress response to surgery, may have contributed to the initial changes. While our study design cannot definitively isolate the effect of LAAC from all potential confounders, the strong and consistent hormonal and electrolyte pattern strongly supports a specific role for LAA compression.
Our study has several limitations that should be considered. Its retrospective, single-center design and modest sample size may limit the generalizability of the findings and introduce potential for selection bias. The lack of long-term follow-up data prevents assessment of whether the hypotensive effect was sustained. Although we have proposed a BNP-aldosterone mediated mechanism, the absence of data on other RAAS components (e.g., renin, angiotensin II) means the hormonal cascade cannot be fully characterized. Finally, the potential influence of unmeasured confounders cannot be entirely ruled out, despite the compelling hormonal data. Taken together, current evidence suggests that LAAC may influence blood pressure through BNP-mediated RAAS suppression, however its precise mechanism requires validation through multicenter controlled studies.
Our findings indicate that LAAC is associated with a significant postoperative decline in blood pressure. The observed increase in BNP level and concurrent decrease in aldosterone suggest a potential mechanism involving BNP-mediated natriuresis and vasodilation. While BNP elevation may contribute to this hypotensive effect, other perioperative factors could also play a role. Consequently, proactive management is often required, including de-escalation of antihypertensive medication and vigilance for severe hypotensive events in the postoperative period. Nevertheless, the increase in BNP level remains a potential contributing factor to the postoperative decrease in blood pressure after LAAC.
AF, atrial fibrillation; LAA, left atrial appendage; LAAC, left atrial appendage clipping; BNP, B-type natriuretic peptide; LAAPD, left atrial anteroposterior diameter; LVEDD, left ventricular end-diastolic diameter; EF, ejection fraction; SBP, systolic blood pressure; DBP, diastolic blood pressure.
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
QY designed the research study and wrote the manuscript. FL contributed to the study design, data analysis, and manuscript writing and revision. HM was involved in data acquisition, data analysis, and manuscript writing and revision. DX contributed to data analysis and manuscript writing, and revision. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of Beijing Tiantan Hospital, Capital Medical University (No. KY2022-013-02). Written informed consent was obtained from all participating patients prior to data collection and analysis.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


