1 Department of Cardiovascular Surgery, Shandong Provincial Hospital Affiliated to Shandong First Medical University, 250021 Jinan, Shandong, China
2 Department of Cardiovascular Surgery, The Affiliated Hospital, Southwest Medical University, Metabolic Vascular Diseases Key Laboratory of Sichuan Province, Key Laboratory of Cardiovascular Remodeling and Dysfunction, 646000 Luzhou, Sichuan, China
3 Key Laboratory of Medical Electrophysiology, Ministry of Education & Medical Electrophysiological Key Laboratory of Sichuan Province, (Collaborative Innovation Center for Prevention of Cardiovascular Diseases), Institute of Cardiovascular Research, Southwest Medical University, 646000 Luzhou, Sichuan, China
†These authors contributed equally.
Abstract
This study aimed to investigate the incidence and risk factors of postoperative bloodstream infections (BSIs) in patients with Stanford type A aortic dissection (SAAD) and to develop a reliable predictive model to provide a more comprehensive understanding of the characteristics of this complication.
Clinical data from 257 patients who underwent surgical repair for SAAD at the Shandong Provincial Hospital Affiliated to Shandong First Medical University between January 2017 and July 2023 were retrospectively analyzed. Risk factors for postoperative BSIs were identified using univariate and multivariate logistic regression. A predictive model was constructed and validated based on the receiver operating characteristic (ROC) curve.
Based on a comprehensive analysis of 257 patients who underwent surgical repair for type A aortic dissection, this study identified an incidence of postoperative BSIs of 10.5%. Patients with BSIs experienced significantly worse outcomes, including prolonged intensive care unit (ICU) and overall hospital stays, and a higher incidence of complications such as liver failure, acute kidney injury, and cerebral infarction. In addition, postoperative BSI was associated with an increase in in-hospital mortality. Blood culture analysis revealed Gram-negative bacilli as the primary pathogens, with Acinetobacter baumannii and Enterobacter cloacae being the most prevalent, collectively accounting for 22.22% of all BSI cases. Multivariable analysis identified the following independent risk factors for postoperative BSI: preoperative C-reactive protein (odds ratios (OR) = 1.010, 95% confidence interval (CI) 1.002–1.019, p = 0.020), tracheostomy (OR = 9.186, 95% CI 2.463–34.266, p = 0.001), infectious pneumonia (OR = 32.872, 95% CI 4.186–258.174, p = 0.001), circulatory arrest time (OR = 1.048, 95% CI 1.004–1.093, p = 0.033), and age (OR = 1.055, 95% CI 1.010–1.103, p = 0.016). A predictive model constructed from these factors demonstrated strong discriminatory power, with an area under the ROC curve of 0.897. The model exhibited a sensitivity of 85.0% and a specificity of 90.0%, indicating good predictive accuracy within the study cohort.
Postoperative bloodstream infection is a significant complication after surgical repair of Stanford type A aortic dissection, and is associated with worse clinical outcomes. A predictive model incorporating the independent risk factors of advanced age, elevated preoperative C-reactive protein, prolonged circulatory arrest time, tracheostomy, and infectious pneumonia aids in the early identification of high-risk patients. Future large-scale, multi-center studies are warranted to further validate and future refine these findings.
Keywords
- Stanford type A aortic dissection
- bloodstream infection
- risk factors
- predictive model
Aortic dissection is a critical condition, with Stanford type A aortic dissection (SAAD) representing 60–70% of cases and carrying a particularly high mortality risk. International data show that without surgery, mortality exceeds 50% within 48 hours and reaches 70% within two weeks of onset [1, 2]. Although surgical advances have substantially reduced postoperative mortality, early complications remain a major cause of in-hospital death [3, 4]. Among these, bloodstream infection (BSI) is a serious postoperative complication characterized by an insidious onset and a diagnosis largely dependent on time-consuming blood cultures, which complicates early detection [5]. Without timely intervention, BSI may progress to shock and multiorgan failure, resulting in increased morbidity and mortality.
Given these challenges, identifying risk factors for BSI after SAAD surgery is clinically important for guiding treatment and improving outcomes. This study systematically examined the incidence and risk factors for BSI following SAAD surgery and developed a predictive model to identify high-risk patients. We analyzed clinical data from 257 SAAD patients who underwent conventional open surgery at the Shandong Provincial Hospital Affiliated to Shandong First Medical University between January 2017 and July 2023. Based on comprehensive statistical analyses, a predictive model was established to assess the individual risk of infection and to facilitate targeted preventive strategies. The findings provide valuable scientific insights for postoperative management and prevention of complications in SAAD patients.
This study retrospectively identified patients with SAAD who underwent conventional open-heart surgery with cardiopulmonary bypass between January 2017 and July 2023 through the electronic medical record system of Shandong Provincial Hospital. SAAD diagnosis was based on the 2022 ACC/AHA Guideline for the Diagnosis and Management of Aortic Disease [6]. All data—including medical history, surgical, anesthetic, and bypass records, nursing charts, imaging, and laboratory results—were derived from original sources to ensure authenticity and traceability. The requirement for informed consent was waived due to the retrospective nature of the study, which was conducted in compliance with Good Clinical Practice (GCP) and the Declaration of Helsinki.
(1) Patients with a definitive diagnosis of Stanford Type A aortic dissection, confirmed by preoperative imaging (echocardiography or CTA) and subsequent intraoperative findings.
(2) Patients who were assessed as surgical candidates by cardiovascular surgeons and subsequently underwent open aortic surgery with cardiopulmonary bypass.
(3) Patients with complete demographic and essential perioperative clinical data.
(1) Patients scheduled for aortic surgery who did not undergo cardiopulmonary bypass, including those who did not receive surgery or underwent only endovascular aortic repair.
(2) Those diagnosed with Stanford type B aortic dissection.
(3) Patients who died before surgery, during surgery, or within 72 hours postoperatively.
(4) Cases with missing essential research data.
The following is a flow chart of patient selection criteria (Fig. 1).
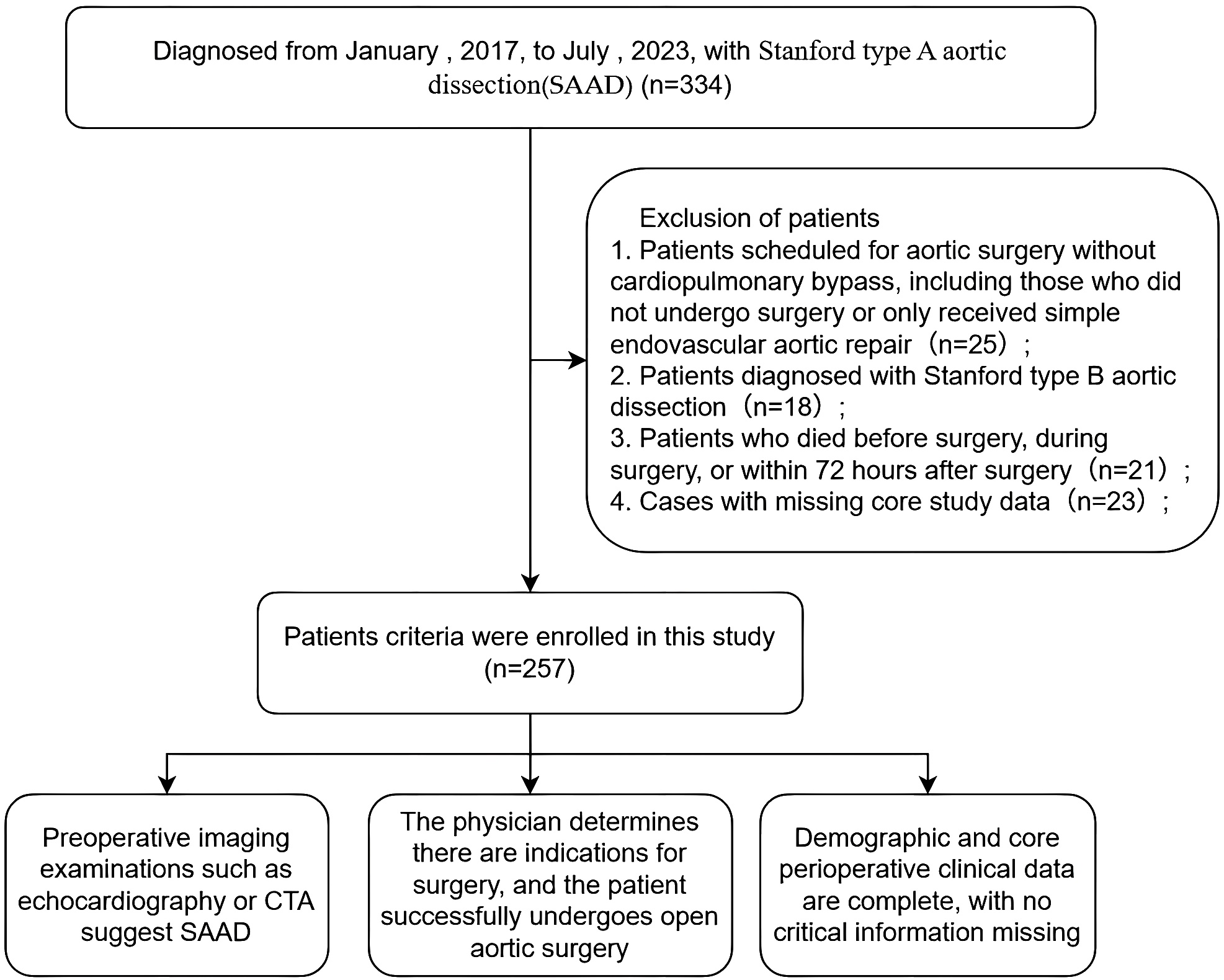 Fig. 1.
Fig. 1.
Flowchart of study patient selection and decision-making.
The diagnosis of bloodstream infection was established in accordance with the Diagnostic Criteria for Hospital Infections (Trial) issued by the Ministry of Health of China in 2001, and aligns with relevant guidelines from international societies such as the Infectious Diseases Society of America (IDSA) [7]. Patients were required to meet one of the following sets of criteria [8]:
(1) Positive blood culture for a recognized pathogen, or repeated positivity for common skin commensals, with clinical symptoms not attributable to other infections.
(2) Positive blood culture with an identified portal of entry or metastatic infectious focus.
(3) Clinical signs of systemic infection (e.g., fever
Acute renal insufficiency is defined as a postoperative serum creatinine level increased by more than 50% from the preoperative baseline level or an absolute increase exceeding 26.5 µmol/L [9].
Following surgery for aortic dissection, patients were closely monitored for level of consciousness and promptly assessed for focal neurological signs. The occurrence of neurological complications was comprehensively evaluated by integrating various imaging modalities—including transcranial Doppler, cranial CT, MRI, and electromyography—with consultations from neurology and neurosurgery specialists [10].
Liver failure is defined as serum total bilirubin concentration exceeding 205 µmol/L, or the concurrent presence of bilirubin-enzyme dissociation [11].
Postoperative pulmonary infection was defined by the presence of cough with purulent sputum and moist rales on auscultation, plus at least one of the following criteria: fever; elevated white blood cell count or neutrophil percentage; inflammatory infiltrates on chest radiography; with confirmation requiring the identification of the same pathogen in two separate sputum cultures [12].
Clinical data for all enrolled patients were systematically collected from the electronic medical record system of Shandong Provincial Hospital Affiliated to Shandong First Medical University The collected variables encompassed baseline characteristics, preoperative status, intraoperative details, and postoperative course.
Baseline information included patient demographics (age, sex), health habits (smoking history, alcohol use), and relevant medical history (hypertension, diabetes, atherosclerosis, aortic aneurysm, aortic valve regurgitation, prior cardiac surgery, Marfan syndrome). Presenting symptoms such as chest pain, back pain, abdominal pain, or syncope were also documented.
Preoperative assessment included left ventricular ejection fraction (LVEF), preoperative malperfusion (including intestinal ischemia, acute kidney injury, coronary artery ischemia, and lower extremity ischemia), left ventricular hypertrophy, heart failure, pleural or pericardial effusion, and a panel of laboratory tests including hemoglobin, white blood cell count, neutrophil, lymphocyte, monocyte, and platelet counts, International Normalized Ratio (INR), C-reactive protein (CRP), serum creatinine, liver function markers (aspartate aminotransferase, alanine aminotransferase, albumin, prealbumin, albumin-to-globulin ratio, bilirubin fractions, alkaline phosphatase, gamma-glutamyl transferase), and cardiac injury biomarkers (high-sensitivity troponin T, myoglobin, creatine kinase-MB mass, D-dimer).
Intraoperative variables recorded were the emergency status of the surgery, key procedural times (total operative time, cardiopulmonary bypass time, aortic cross-clamp time, and circulatory arrest time), and the types and quantities of blood products transfused, including red blood cells, plasma, platelets, and cryoprecipitate.
Postoperative data collection focused on serial laboratory values (hemoglobin, platelet count, white blood cell and differential counts, CRP), the occurrence of specific complications (acute renal insufficiency, cerebral infarction, liver failure, pulmonary infection, pleural effusion requiring thoracentesis), and details of postoperative support. This included durations of various catheter placements (arterial, central venous, pericardial/mediastinal drainage, nasogastric), tracheal intubation and tracheostomy events, need for continuous renal replacement therapy (CRRT), re-operation details (exploratory thoracotomy, wound debridement, secondary cardiopulmonary bypass), transfusion volumes post-surgery, antibiotic administration (classes and duration), maximum body temperature, and ICU length of stay.
Data analysis was performed using R software (version 4.5.1, Vienna, Vienna, Austria) and R Studio
(version 2025.05.1+513, Boston, Massachusetts, United States), with a two-sided p-value
To identify risk factors and develop a prediction model for postoperative BSI, patients were categorized into BSI-positive and BSI-negative groups. Binary logistic regression with Forward Likelihood Ratio (Forward LR) stepwise selection was used to identify independent risk factors. Model performance was evaluated using the Wald test, the Hosmer-Lemeshow goodness-of-fit test, and receiver operating characteristic (ROC) curve analysis, with results reported as the area under the curve (AUC). Effect estimates are presented as odds ratios (ORs) with 95% confidence intervals (CIs).
To internally validate the prediction model and estimate its optimism, we performed bootstrap resampling with 1000 repetitions. The optimism-corrected performance metrics, including the area under the ROC curve, were calculated.
All statistical test results in this article have been cross-validated by Stata software.
Of the 257 patients included in the final analysis, 27 (10.5%) developed postoperative bloodstream infection (BSI). A comprehensive analysis was performed to characterize postoperative BSI, evaluating baseline admission characteristics, preoperative status, and intraoperative and postoperative indicators. Key findings are summarized below.
The cohort was predominantly male (172 patients, 66.9%), with a mean age of
50.81
Comparative analysis revealed that patients who developed bloodstream infection
were more likely to have higher preoperative systolic blood pressure (p
= 0.012), a history of alcohol consumption (p = 0.033), and an age of 70
years or older (p = 0.001). Back pain was significantly more common in
this group (70.37% vs. 43.91%, p
| Whole cohort | Positive group | Negative group | p-value | |
| N = 257 | N = 27 | N = 230 | ||
| Aged |
14 (5.45) | 5 (18.52) | 9 (3.91) | 0.001 |
| Aged |
243 (94.55) | 23 (85.19) | 220 (95.65) | |
| Male | 172 (66.93) | 22 (81.48) | 150 (65.22) | 0.138 |
| SBP (mmHg) | 137.02 |
149.89 |
135.51 |
0.012 |
| DBP (mmHg) | 76.18 |
79.04 |
75.85 |
0.368 |
| Smoking history | 98 (38.12) | 9 (33.33) | 89 (38.70) | 0.739 |
| History of alcohol use | 82 (31.90) | 14 (51.85) | 68 (29.56) | 0.033 |
| Hypertension | 188 (73.15) | 22 (81.48) | 166 (72.17) | 0.422 |
| Diabetes mellitus | 14 (5.45) | 0 (0) | 14 (6.09) | 0.384 |
| History of cardiac surgery | 11 (4.28) | 0 (0) | 11 (4.78) | 0.510 |
| Marfan syndrome | 13 (5.06) | 0 (0) | 13 (5.65) | 0.422 |
| Aortic aneurysm | 56 (21.79) | 3 (11.11) | 53 (23.04) | 0.240 |
| Chest pain | 197 (76.65) | 24 (88.89) | 173 (75.22) | 0.178 |
| Back pain | 120 (46.69) | 19 (70.37) | 101 (43.91) | 0.016 |
| Abdominal pain | 82 (31.90) | 6 (22.22) | 76 (33.04) | 0.356 |
| Syncope | 14 (5.44) | 0 (0) | 14 (6.08) | 0.384 |
| Preoperative malperfusion state | 41 (15.95) | 10 (37.04) | 31 (13.48) | 0.004 |
| Intestinal ischemia | 12 (4.67) | 4 (14.81) | 8 (3.48) | 0.080 |
| Acute kidney injury | 23 (8.95) | 3 (11.11) | 20 (8.70) | 0.270 |
| Coronary artery ischemia | 15 (5.84) | 4 (14.81) | 11 (4.78) | 0.310 |
| Lower extremity ischemia | 31 (12.06) | 5 (18.52) | 26 (11.30) | 0.067 |
Footnotes:
The comparative analysis of baseline characteristics of the two groups of patients.
SBP, systolic blood pressure; DBP, diastolic blood pressure.
Laboratory findings indicated a proinflammatory and prothrombotic state in the
BSI group, with significantly elevated levels of C-reactive protein, white blood
cell counts, absolute monocyte counts, absolute neutrophil counts, and D-dimer
(all p
| Whole cohort | Positive group | Negative group | p-value | |
| N = 257 | N = 27 | N = 230 | ||
| WBC (109/L) | 10.54 |
12.85 |
10.27 |
0.019 |
| RBC (1012/L) | 4.19 |
4.15 |
4.19 |
0.763 |
| Hb (g/L) | 128.34 |
128.89 |
128.28 |
0.893 |
| Plt (109/L) | 191.49 |
186.85 |
192.03 |
0.689 |
| LYM (109/L) | 1.22 |
1.31 |
1.21 |
0.490 |
| MONO (109/L) | 0.73 |
0.95 |
0.70 |
0.007 |
| NE (109/L) | 8.42 |
10.58 |
7.485 |
0.015 |
| CRP (mg/L) | 45.35 |
64.53 |
43.09 |
0.032 |
| AST (U/L) | 24.0 (18.0–49.0) | 27.0 (22.5–53.0) | 23.5 (22.5–53.0) | 0.160 |
| ALT (U/L) | 22.0 (13.0–42.0) | 26.0 (17.0–47.5) | 21.0 (13.0–40.0) | 0.130 |
| GGT (U/L) | 28.0 (17.0–51.0) | 37.0 (17.0–74.5) | 28.0 (17.0–48.0) | 0.314 |
| ALP (U/L) | 71.0 (59.0–91.0) | 73.0 (56.5–83.0) | 71.0 (59.0–91.0) | 0.930 |
| Prealbumin | 198.16 |
220.84 |
195.50 |
0.070 |
| Albumin | 3.67 |
37.07 |
36.62 |
0.665 |
| A/G Ratio | 1.45 |
1.51 |
1.44 |
0.315 |
| INR | 1.13 (1.07–1.22) | 1.11 (1.06–1.165) | 1.13 (1.07–1.22) | 0.198 |
| Creatinine | 82.4 (66.71–111.5) | 107.1 (86.5–137.14) | 80.15(66.05–107.83) | 0.004 |
| hs-cTnl | 20.94 (9.83–140.9) | 29.46 (10.93–194.65) | 20.74 (9.54–137.35) | 0.366 |
| CK-MB | 2.26 (1.12–7.2) | 3.21 (1.37–19.42) | 2.15 (1.11–6.61) | 0.106 |
| Myoglobin | 51.21 (24.1–195.0) | 124.0 (36.94–1128.0) | 47.04 (23.57–141.88) | 0.009 |
| T-Bil | 5.7 (3.3–10.8) | 7.15 (4.27–14.11) | 5.52 (3.20–10.63) | 0.108 |
| Direct bilirubin | 14.6 (9.08–21.5) | 15.1 (9.57–22.72) | 14.57 (9.11–21.42) | 0.761 |
| Indirect bilirubin | 20.1 (13.33–29.72) | 20.88 (13.79–29.13) | 20.1 (13.33–29.85) | 0.924 |
Footnote:
Based on the preoperative characteristic data in Table 2, the laboratory test results of the positive group (N = 27) and the negative group (N = 230) were compared and analyzed.
WBC, White blood cell; RBC, Red blood cell; Hb, Hemoglobin; Plt, Platelet; LYM, Lymphocytic lymphoid cell; MONO, Monocytes; NE, Neutrophil granulocyte; CRP, C-reactive protein; AST, Aspartate aminotransferase; ALT, Alanine aminotransferase; GGT, Gamma-glutamyl transferase; ALP, Alkaline Phosphatase; A/G Ratio, Albumin-to-Globulin Ratio; INR, International Normalized Ratio; hs-cTnl, High-sensitivity cardiactroponin T; CK-MB, creatine kinase-myocardial band; T-Bil, Total bilirubin.
In this study, several cardiac surgical procedures were identified and defined. They are: S stands for Sun’s procedure; B is for Bentall procedure; C is for Cabrol procedure; D is for David procedure; W is for Wheat surgery; E is for total arch or half arch prosthesis replacement of the ascending aorta; F is for ascending aorta prosthesis replacement; G is for aortoplasty. In addition, we also defined hybrid procedures, such as B+S, representing Bentall + Sun’s procedure. “Emergency surgery” is defined in this study as follows: after a patient is diagnosed with Stanford type A (or DeBakey type I, II) aortic dissection via imaging examination, the cardiovascular surgery team evaluates the case as the highest priority with an immediate life-threatening risk, which requires scheduling and performing the surgery within a few hours after admission.
When exploring the relationship between each surgical procedure and bloodstream
infection, we found that the association between surgical procedure S and
bloodstream infection was statistically significant (p-value was 0.009),
which suggested that there was a certain correlation between Sun’s procedure and
bloodstream infection. However, for the other procedures, the p-values
were relatively high, meaning that the association with bloodstream infection was
not statistically significant, and we cannot conclude that they have a clear
association with bloodstream infection. Ultimately, we found that prolonged
procedure times were strongly associated with BSI. The BSI-positive group had
significantly longer total operation time and circulatory arrest time (all
p
| Whole cohort | Positive group | Negative group | p-value | ||
| N = 257 | N = 27 | N = 230 | |||
| Emergency surgery | 149 (57.98) | 19 (70.37) | 130 (56.52) | 0.241 | |
| Aortic cross-clamp time (min) | 113.98 |
117.19 |
113.60 |
0.606 | |
| CPB time (min) | 204.21 |
217.70 |
202.63 |
0.160 | |
| Circulatory arrest time (min) | 18.04 |
23.78 |
13.37 |
0.004 | |
| Total operation time (h) | 6.5 (6.0–7.5) | 7.5 (6.8–9.0) | 6.5 (5.5–7.5) | 0.002 | |
| Intraoperative valve implantation | 51 (19.84) | 3 (11.11) | 48 (20.87) | 0.343 | |
| Concomitant CABG | 30 (11.67) | 3 (11.11) | 27 (11.74) | 1.000 | |
| Surgical methods | |||||
| Surgical methods S | 186 (72.66) | 25 (96.15) | 161 (70.00) | 0.009 | |
| Surgical methods B | 11 (4.2) | 0 (0) | 11 (4.30) | 0.529 | |
| Surgical methods C | 7 (2.73) | 0 (0) | 7 (3.04) | 0.789 | |
| Surgical methods D | 3 (1.17) | 0 (0) | 3 (1.30) | 1.000 | |
| Surgical methods W | 1 (0.39) | 0 (0) | 1 (0.43) | 1.000 | |
| Surgical methods E | 11 (4.30) | 0 (0) | 11 (4.78) | 0.529 | |
| Surgical methods F | 4 (1.56) | 0 (0) | 4 (1.74) | 1.000 | |
| Surgical methods G | 1 (0.39) | 0 (0) | 1 (0.43) | 1.000 | |
| Surgical methods B+S | 20 (7.81) | 0 (0) | 20 (8.70) | 0.238 | |
| Surgical methods C+S | 9 (3.12) | 2 (3.85) | 7 (3.04) | 1.000 | |
| Surgical methods D+S | 4 (1.56) | 0 (0) | 4 (1.74) | 1.000 | |
Footnote:
Based on the intraoperative factor data in Table 3, a comparative analysis was conducted on the intraoperative conditions of the positive group (N = 27) and the negative group (N = 230).
CPB time, Cardiopulmonary Bypass time; Concomitant CABG, Concomitant coronary artery bypass surgery.
BSI was associated with greater levels of postoperative care, including: longer
duration of pericardial mediastinal drainage, tracheal intubation, arterial and
central venous catheterization, and nasogastric intubation, higher reintubation
rate, increased use of CRRT, and more frequent invasive procedures such as
reintubation, thoracentesis, tracheostomy, exploratory sternotomy, and chest
wound debridement (all p
| Whole cohort | Positive group | Negative group | p-value | ||
| N = 257 | N = 27 | N = 230 | |||
| Duration of pericardial mediastinal drainage | 240 (144.0–312.0) | 360 (192.0–360.0) | 237.0 (69.0–237.0) | ||
| Duration of tracheal intubation (h) | 43.0 (19.0–110.0) | 216.0 (110.5) | 38.0 (18.0–95) | ||
| Duration of arterial catheterization | 6.0 (4.0–10.0) | 21.0 (13.5–36.0) | 6.0 (4.0–9.0) | ||
| Duration of central venous catheterization | 10.0 (6.0–15.0) | 27.0 (18.5–36.0) | 9.0 (6.0–13.0) | ||
| Duration of nasogastric tube placement | 2 (0–6.0) | 14 (7.0–26.5) | 2 (0–6.0) | ||
| Postoperative invasive procedures | |||||
| Thoracentesis | 65 (25.29) | 12 (44.44) | 53 (23.04) | ||
| Tracheostomy | 15 (5.84) | 8 (29.63) | 7 (3.04) | ||
| Reintubation | 27 (10.51) | 12 (44.44) | 15 (6.52) | ||
| Exploratory sternotomy | 16 (6.23) | 6 (44.44) | 10 (6.52) | ||
| Chest wound debridement | 5 (1.95) | 3 (11.11) | 2 (0.87) | ||
| Postoperative transfusion volume | |||||
| RBC (U) | 2 (0–4) | 21 (8–30) | 2 (0–4) | ||
| Plt (Unit) | 0 (0–1) | 2 (0–19) | 0 (0–1) | ||
| Postoperative complications | |||||
| Pleural effusion | 116 (414) | 16 (59.26) | 100 (43.48) | 0.120 | |
| Liver failure | 29 (11.28) | 15 (55.56) | 14 (6.09) | ||
| Acute renal insufficiency | 32 (12.45) | 15 (55.56) | 17 (7.39) | ||
| Cerebral infarction | 35 (13.62) | 11 (40.74) | 24 (10.43) | ||
| Infectious pneumonia | 111 (43.19) | 26 (96.30) | 85 (39.96) | ||
| Outcome | |||||
| Hospital stay |
16 (6.22) | 12 (44.44) | 4 (1.74) | ||
| Postoperative maximum body temperature | 38.32 |
39.03 |
38.24 |
||
| Postoperative ICU length of stay | 6 (4–11) | 27 (13.5–38) | 6 (4–9) | ||
| In-hospital mortality | 13 (5.06) | 8 (29.63) | 5 (2.17) | ||
Footnote:
Significant differences between the two groups of patients in terms of postoperative course and outcomes of the included subjects.
RBC, Red blood cell; Plt, Platelet; ICU, Intensive care unit.
Variables with significant univariate associations were subsequently entered into a multivariate binary logistic regression analysis using the forward likelihood ratio method. This identified five independent predictors for postoperative bloodstream infection: preoperative C-reactive protein (OR = 1.010, 95% CI: 1.002–1.019, p = 0.020), tracheostomy (OR = 9.186, 95% CI: 2.463–34.266, p = 0.001), infectious pneumonia (OR = 32.872, 95% CI: 4.186–258.174, p = 0.001), circulatory arrest time (min) (OR = 1.048, 95% CI: 1.004–1.093, p = 0.033), and age (OR = 1.055, 95% CI: 1.010–1.103, p = 0.016). These factors were incorporated to establish the final prediction model.
Based on the multivariate logistic regression analysis, a prediction model for
postoperative bloodstream infection was established using five significant
independent factors: preoperative C-reactive protein, tracheostomy, infectious
pneumonia, circulatory arrest time, and age. The Wald test confirmed the
statistical significance of all included variables. The model’s goodness-of-fit
was evaluated using the Hosmer-Lemeshow test, which showed good calibration with
a chi-square of 9.873 (p = 0.274). The likelihood ratio test further
supported model significance with p
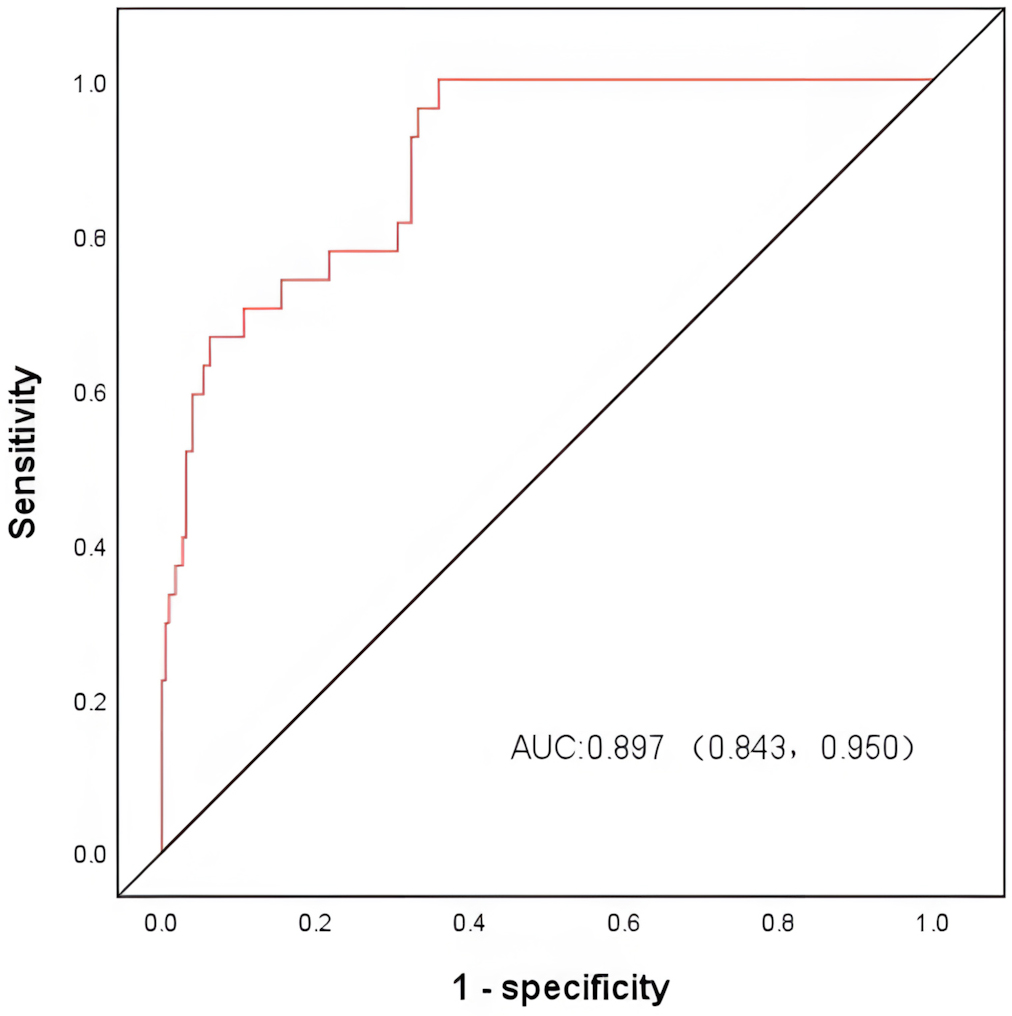 Fig. 2.
Fig. 2.
ROC curves for the model to predict the occurrence of
bloodstream infection after admission in the SAAD. After plotting the ROC curve,
the area under the (AUC) was 0.897 (95% CI: 0.843–0.950, p
| Odds ratio | 95% confidence interval | p-value | |
| Age | 1.055 | 1.010, 1.013 | 0.016 |
| Infectious Pneumonia | 32.872 | 4.186, 258.174 | 0.001 |
| Tracheostomy | 9.186 | 2.463, 34.266 | 0.001 |
| Preoperative C-Reactive Protein | 1.010 | 1.004, 1.093 | 0.020 |
| Circulatory Arrest Time | 1.048 | 1.018, 1.095 | 0.003 |
Footnote:
Multivariate analysis showed that age, preoperative C-reactive protein (CRP), time of circulatory arrest, tracheotomy, and infectious pneumonia were independent predictors of BSI.
The above variables were then assigned appropriate scores based on their relative importance in the nomogram (Fig. 3). Finally, we constructed the decision curve analysis (DCA) curve (Fig. 4). These analyses demonstrated that the developed nomogram performed well across all threshold probabilities, confirming that it would provide the highest clinical net benefit across a wide range of clinical scenarios.
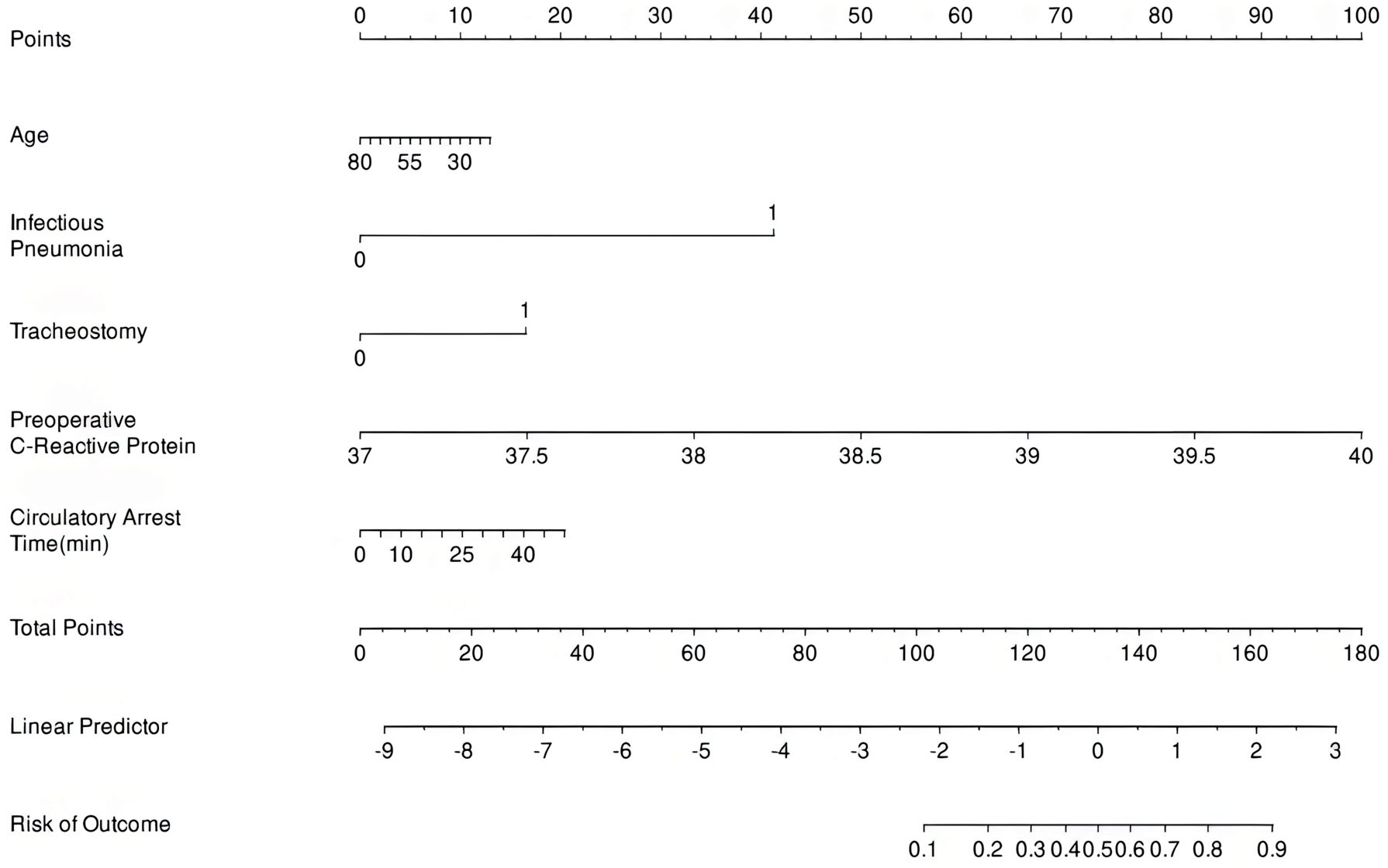 Fig. 3.
Fig. 3.
Prediction chart for all-cause in-hospital mortality of TAAAD. A bar plot was drawn to visualize the results of the prediction model. A total of five prediction variables were involved in the composition of the bar plot. According to the extent of influence of each prediction variable in the model, i.e., the magnitude of the regression coefficient, a score was assigned to each value level of the variable, resulting in five separate scores, and then these scores were added together to obtain the total score. TAAAD, Type A acute aortic dissection.
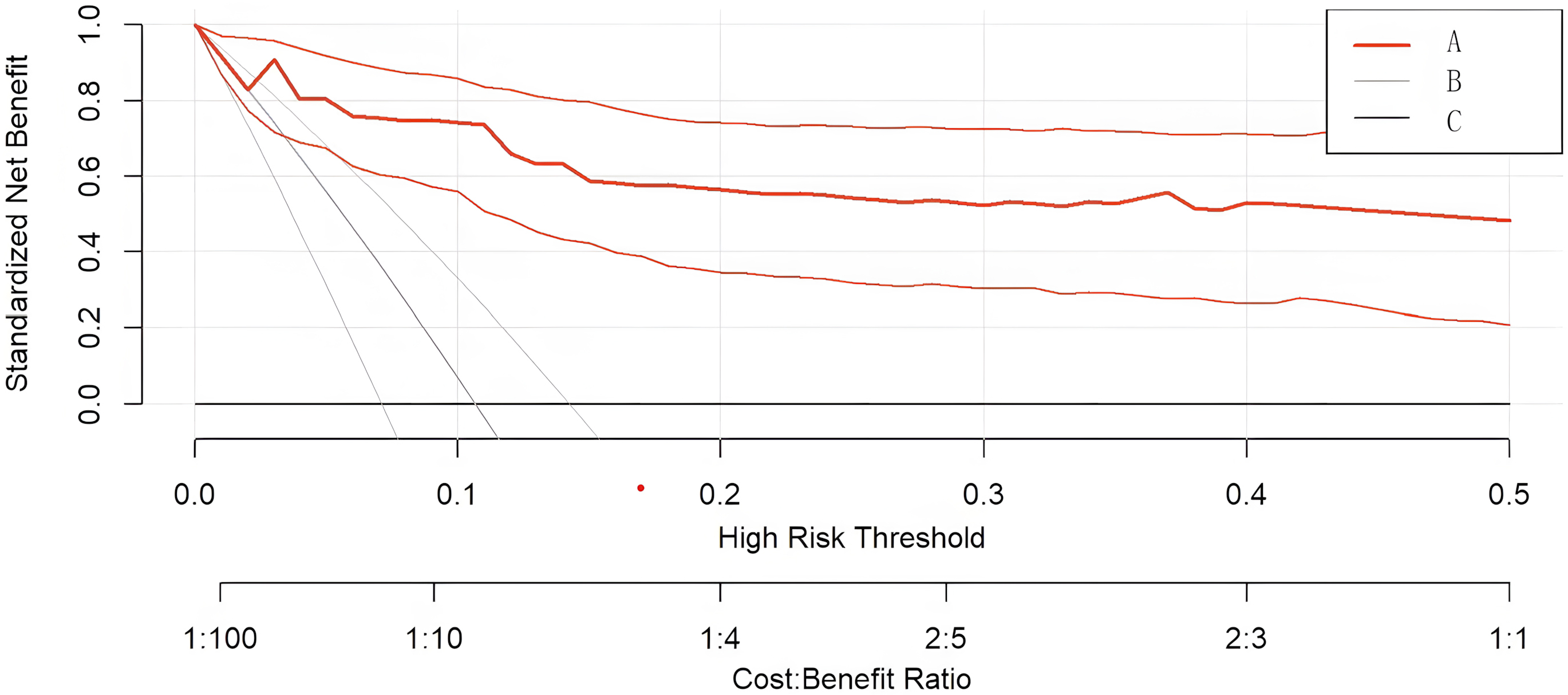 Fig. 4.
Fig. 4.
Curve A represents model construction, Curve B represents full intervention, and Curve C represents no intervention. The clinical decision curve for predicting all-cause in-hospital mortality in TAAAD. The DCA curve was plotted based on the constructed CPM. The horizontal axis indicates the risk threshold, and the vertical axis indicates the net benefit (NB) after considering benefits and harms. The DCA results show that the risk assessment model brings net benefit to patients over a risk threshold range of 0.01–0.7.
This retrospective study of 257 patients undergoing surgical repair for Stanford type A aortic dissection provides a comprehensive analysis of the incidence, risk factors, and clinical impact of postoperative bloodstream infection. Our key findings indicate that BSI is a frequent (10.5%) and devastating complication, associated with a significantly prolonged hospital course, a higher incidence of multi-organ failure, and a significant increase in in-hospital mortality (29.63% vs. 2.17%). Furthermore, we developed and internally validated a predictive model incorporating five independent risk factors—age, preoperative C-reactive protein, circulatory arrest time, tracheostomy, and infectious pneumonia—which demonstrated strong discriminatory power (AUC 0.897) for identifying high-risk patients.
The observed BSI incidence of 10.5% aligns with the higher end of rates reported for major cardiac surgery, a finding likely attributable to the extreme acuity and complexity of SAAD surgery [13]. The profound clinical consequences of BSI in our cohort underscore its role as a critical determinant for poor outcomes. Patients with BSI had a more invasive postoperative course, characterized by prolonged dependence on mechanical ventilation and invasive catheters, a greater need for renal replacement therapy, and higher rates of re-operation. The cascade of complications, including cerebral infarction, acute renal insufficiency, and liver failure, suggests that BSI acts as a potent trigger for exacerbation of the systemic inflammatory response syndrome and subsequent multi-organ dysfunction syndrome [14]. The dramatic 13-fold increase in mortality in the BSI group illustrates that BSI is not merely a comorbid condition but a pivotal event that can tip the precarious balance of a post-dissection patient towards a fatal outcome. Therefore, strategies aimed at preventing and aggressively managing BSI are paramount to improving overall survival in SAAD repair.
The five independent risk factors identified in our multivariate analysis provide crucial insights into the pathophysiology of BSI. Elevated preoperative CRP emerged as a significant predictor, highlighting the importance of the pre-existing inflammatory state [15]. SAAD itself is now recognized as a highly inflammatory condition [16], with recent studies elucidating the role of damage-associated molecular patterns released from the dissected aorta in triggering a fulminant innate immune response [17]. A high CRP level upon admission is a biomarker of this intense systemic inflammation, which is often coupled with a state of compensatory immunoparalysis [18]. This early immunodysfunctional state, characterized by impaired neutrophil function and monocyte suppression, renders the patient highly susceptible to secondary postoperative infections [19]. Thus, preoperative CRP serves as a quantifiable measure of a patient’s baseline vulnerability to infection even before the surgical insult [20].
The highly significant associations of tracheostomy and infectious pneumonia with BSI underscore the central role of the respiratory tract as a primary source for bloodstream invasion [21]. Tracheostomy, while sometimes necessary for prolonged ventilation, disrupts the anatomical barrier of the airway, facilitating colonization with hospital-acquired pathogens [22]. Infectious pneumonia, particularly ventilator-associated pneumonia, then establishes a persistent nidus of infection from which bacteria can readily translocate into the pulmonary circulation and cause BSI [23, 24]. The predominance of Gram-negative bacilli in our blood culture analysis is consistent with this pathway and strongly suggests that rigorous adherence to VAP prevention bundles is not only a pulmonary protective strategy but a critical defense against life-threatening BSI [25].
Prolonged circulatory arrest time, a marker of surgical complexity, implicates gut mucosal injury and bacterial translocation as another key pathophysiological pathway [26]. Deep hypothermic circulatory arrest induces splanchnic hypoperfusion and ischemia-reperfusion injury, which can compromise the integrity of the intestinal barrier [27]. This allows commensal Gram-negative bacteria and their endotoxins to leak into the systemic circulation. The role of gut-derived infection in critical illness is a topic of renewed interest, and our data provide strong clinical evidence supporting this pathway in SAAD surgery, linking the technical demands of the procedure directly to the risk of a subsequent septic complication. Furthermore, advanced age, a non-modifiable risk factor, contributes through the well-documented decline in immune competence known as immunosenescence, which reduces the patient’s ability to withstand the cumulative insults of dissection, surgery, and infection [28].
The predictive model developed from these factors offers a practical tool for early risk stratification. Its strength lies in the integration of readily available preoperative and immediate postoperative variables, allowing clinicians to identify high-risk patients at the conclusion of the operation or upon ICU admission. This risk stratification enables a proactive, targeted management approach. For high-risk patients, clinicians can institute intensified surveillance, such as enhanced microbiological monitoring, have a lower threshold for initiating prudent empiric antibiotic therapy guided by local antibiograms, aggressively pursue source control, and consider adjunct supportive measures such as immunonutrition. By focusing resources on the patients who need them most, this model facilitates a paradigm shift from reactive treatment to proactive prevention, potentially reducing the incidence and mortality of BSI.
Several limitations of our study must be acknowledged. Its single-center,
retrospective design allows for the potential for selection and information bias.
The number of BSI events, while substantial for this patient cohort, is
relatively small for a multivariate model with five predictors, raising a
potential risk of overfitting. Most critically, the model requires external
validation in independent, multi-center cohorts to confirm its generalizability
before widespread clinical adoption. Moreover, the number of BSI events, while
substantial for this patient cohort, results in a low events-per-variable ratio
in our multivariate model, which remains a limitation. Although bootstrap
internal validation suggested acceptable optimism, external validation in
independent, multi-center cohorts is crucial to confirm generalizability before
any clinical application. Furthermore, the predictors identified in our
model—advanced age, elevated preoperative inflammation, prolonged circulatory
arrest, tracheostomy, and infectious pneumonia—are based on strong biological
plausibility regarding immune function, procedural invasiveness, and infection
pathways. As such, they are likely to remain relevant risk factors in other
settings. However, the magnitude of their association (i.e., their specific odds
ratios) may vary across institutions employing different surgical techniques
(e.g., strategies to shorten circulatory arrest) or postoperative care bundles
(e.g., rigorous ventilator-associated pneumonia prevention protocols that reduce
the need for tracheostomy). Consequently, while the core predictors are expected
to be generalizable, the model’s absolute risk estimates may require
recalibration during external validation in centers with distinct clinical
protocols before it can be applied for local risk stratification. Finally, the
inclusion of “infectious pneumonia” as a predictor means the model is most
accurately applied in the early postoperative period for dynamic risk assessment,
rather than as a pure preoperative prediction tool. While the sample size
provided sufficient statistical power to identify the strong independent
predictors included in our final model, it may have been underpowered to detect
weaker associations (e.g., odds ratios
In conclusion, this study confirms the critical impact of postoperative BSI following type A aortic dissection surgery. We established a predictive model incorporating five independent risk factors: age, preoperative CRP, circulatory arrest time, tracheostomy, and infectious pneumonia. The model shows strong predictive ability, offering a practical tool for early risk stratification. This enables timely intervention for high-risk patients, potentially improving outcomes. External validation in future studies is recommended to generalize its application.
Data will be made available from the corresponding author upon reasonable request.
Conception and design of the study: ZFX, FYL, FHS, LYW, HZZ and XCM. Acquisition of data: FHS, ZFX, FYL, LYW, MQW, and XZL. Data analysis and interpretation: ZFX, FYL, FHS, LYW, and MQW. Drafting of manuscript: ZFX, FYL, FHS, LYW, MQW, and XZL. Critical revision: ZFX, FYL, FHS, LYW, MQW, XZL, HZZ, and XCM. Final approval and supervision: HZZ and XCM. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This retrospective study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University (Number: NSFC2018-002). Informed consent was exempted due to the retrospective nature of the study.
Not applicable.
This study was supported by the fund with an additional project number. Add content as follows: This work was supported by the Young Experts of Taishan Scholar Program of Shandong Province (grant no. tsqn202408359), the Nature Science Foundation of Shandong Province (ZR2023MH124), and Jinan Science and Technology Plan Project (202225050).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




