1 Department of Critical Care Medicine, The First Affiliated Hospital of Bengbu Medical University, 233004 Bengbu, Anhui, China
2 Department of Cardiovascular Surgery, The First Affiliated Hospital of Bengbu Medical University, 233004 Bengbu, Anhui, China
Abstract
This study aimed to investigate the incidence, associated factors, and outcomes of nosocomial infections (NIs) among adult patients supported by venoarterial extracorporeal membrane oxygenation (VA-ECMO).
This retrospective study included 97 adult patients who underwent VA-ECMO between July 2020 and January 2025. All patients were treated in a single-center intensive care unit (ICU). The incidence, pathogen characteristics, associated factors, and outcomes of NIs were analyzed.
A total of 61 (62.89%) patients developed NIs. Acinetobacter baumannii was identified as the major pathogen. The hospital mortality rate for patients receiving VA-ECMO with NIs was 49.18%. A long ECMO duration (odds ratio (OR) = 1.26, 95% confidence interval (CI): 1.05–1.51; p = 0.013), blood transfusion (OR = 7.45, 95% CI: 1.89–29.28; p = 0.004), a long central venous catheterization (CVC) duration (OR = 1.13, 95% CI: 1.01–1.27; p = 0.041), and long ICU stay (OR = 1.14, 95% CI: 1.07–1.22; p < 0.001) were factors significantly associated with NIs. The occurrence of adverse events was positively related to that of death (OR = 11.85, 95% CI: 4.52–31.08; p < 0.001). A restricted cubic spline (RCS) revealed that when the ICU stay exceeded 24.13 days, the risk of NIs increased dramatically (p for nonlinearity = 0.036).
NIs are common in ICU patients supported by VA-ECMO. Acinetobacter baumannii was identified as the most common microorganism associated with NI. Longer ECMO and CVC durations, blood transfusions, and a longer ICU stay were associated with NIs. The occurrence of adverse events early in the ICU increased the risk of death in ECMO-supported patients.
Keywords
- venoarterial ECMO
- nosocomial infections
- mortality
Extracorporeal membrane oxygenation (ECMO) is a life-saving medical intervention used for patients with refractory cardiopulmonary failure, providing cardiac and respiratory support by oxygenating the blood outside the body [1]. ECMO was first implemented in the early 1970s by Robert Bartlett. Since then, the application of ECMO has increased dramatically [2, 3]. Recently, the application of venoarterial extracorporeal membrane oxygenation (VA-ECMO) has also increased substantially [4]. Because of underlying critical illness, multiple invasive procedures and devices, and prolonged intensive care unit (ICU) stays, patients supported by VA-ECMO are exposed to a wide range of adverse outcomes [5, 6].
Nosocomial infections (NIs) acquired during VA-ECMO are among the most common adverse outcomes, with an overall incidence ranging from 23% to 64% in patients receiving VA-ECMO support [7, 8, 9, 10, 11]. NIs contribute to longer hospital stays, lower survival rates, and delayed cardiopulmonary recovery. Therefore, the identification of associated factors associated with NIs is critical for effective and targeted intervention to reduce in-hospital mortality. A meta-analysis revealed that continuous renal replacement therapy (CRRT), ECMO support, and ICU stay contributed to the risk of NIs [12]. While NIs is a known serious complication during VA-ECMO, a comprehensive understanding of their associated factors and associations with patient outcomes is still in need. Therefore, the aims of this study were to (1) elucidate the incidence of NIs, (2) identify the characteristics of microorganisms, and (3) explore the associated factors for NIs and in-hospital mortality in patients receiving VA-ECMO support at our center.
This study was performed in the ICU of a single center. Data from patients who received ECMO therapy were retrieved from the electronic medical records system and reviewed by an attending physician. The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Bengbu Medical University (Protocol No. 2025 [039]).
The inclusion criteria were as follows: (1) received VA-ECMO support between
July 2020 and January 1, 2025, and (2) received VA-ECMO support for
Critical care specialists and cardiologists were responsible for the timing of VA-ECMO initiation and weaning. These specialists performed the procedures for implantation, daily management, and weaning according to the recommendations of the Extracorporeal Life Support Organization (ELSO). The femoral vein and femoral artery are the sites of cannulation in our center. For patients undergoing VA-ECMO cannulation, prophylactic antibiotic therapy was administered as a single intravenous injection of a second-generation cephalosporin at the time of ECMO implantation. Subsequently, depending on serum lactate levels and organ function, the ECMO flow rate was adjusted to ensure adequate tissue perfusion. To maintain the activated clotting time within 180–220 seconds, a standardized heparin anticoagulation protocol was used.
The definition of NIs was derived from the criteria established by the U.S.
Centers for Disease Control and Prevention in 1988 [13]. The occurrence of NIs
was evaluated in instances where the initiation of VA-ECMO occurred more than 24
hours prior and within 48 hours subsequent to the discontinuation of VA-ECMO.
Specifically, the following types of infections were considered: respiratory
tract infection (RTI), blood stream infection (BSI), urinary tract infection
(UTI), and surgical site infection (SSI). RTI was suspected if at least one of
the following clinical manifestations was detected: a temperature of
During VA-ECMO application, adverse events included neurological complications such as cerebral haemorrhage, ischaemic stroke, and hypoxic ischaemic encephalopathy; cardiovascular events like malignant arrhythmia, cardiogenic shock, and septic shock; as well as gastrointestinal bleeding, limb ischaemic necrosis, multiple organ dysfunction syndrome, and disseminated intravascular coagulation.
The following information was recorded retrospectively for each patient: demographic data, including age, sex, height, weight, diagnosis, previous medical history, and personal history. We also collected scores such as the Acute Physiology and Chronic Health Evaluation II (APACHE II) and the Sequential Organ Failure Assessment (SOFA). Additionally, data on the duration of ECMO support, IABP support, CRRT support, mechanical ventilation, and the use of tracheotomy and bronchoscopy were documented. The maximal vasoactive-inotropic score (VIS) and laboratory test results within 48 hours of ECMO initiation—such as white blood cell and platelet counts, procalcitonin levels, liver and renal function, N-terminal pro-brain natriuretic peptide (NTproBNP), and troponin (TnI)—were also recorded. Microbiological data, including the type and site of isolated bacteria, presence of adverse events, CVC duration, ICU stay, and in-hospital mortality were noted.
The analyses were performed using SPSS version 26.0 (IBM, Armonk, NY, USA), and
R software, version 4.4.2 (The R Foundation for Statistical Computing, Vienna,
Austria). Continuous variables are presented as the median (interquartile range,
IQR) or the mean (standard deviation, SD), whereas categorical variables are
presented as numbers (percentages). For continuous variables, t-tests
and Mann–Whitney U tests were used for normally distributed and skewed
variables, respectively. The
From July 2020 to January 2025, 166 ICU patients treated with ECMO were screened. We excluded 30 patients who required V-V or mixed ECMO configurations, 2 patients with incomplete records, 3 patients who had infections before ECMO support, 8 patients younger than 18 years, and 26 patients who died within 48 hours. Thus, 97 patients who received VA-ECMO were enrolled in the final analysis (Table 1).
| Variables | Overall population (n = 97) |
| Age, mean (SD), years | 55.01 |
| Gender, male, n (%) | 57 (58.76) |
| BSA, mean (SD), m2 | 1.61 |
| BMI, mean (SD), kg/m2 | 24.75 |
| Smoking, n (%) | 8 (8.25) |
| Alcohol, n (%) | 7 (7.22) |
| Hypertension, n (%) | 40 (41.24) |
| Coronary heart disease, n (%) | 35 (36.08) |
| Diabetes, n (%) | 20 (20.62) |
| Tumor, n (%) | 3 (3.09) |
| Stroke, n (%) | 7 (7.22) |
| APACHE II, mean (SD) | 29.80 |
| SOFA, mean (SD) | 16.07 |
| Cr, median (IQR), µmol/L | 135.00 (107.00, 195.00) |
| eGFR, median (IQR), mL/min/1.73 m2 | 70.50 (56.59, 103.50) |
| TBLI, median (IQR), µmol/L | 20.9 (13.30, 34.00) |
| PLT count, median (IQR), |
140.5 (78.50, 218.25) |
| Highest WBC count, median (IQR), |
16.49 (11.37, 20.99) |
| Highest NTproBNP, median (IQR), ng/L | 6370.00 (1312.50, 15,775.00) |
| Lac |
59 (60.82) |
| Highest TnI, median (IQR), µg/L | 16.9 (5.60, 51.95) |
| Mechanical ventilation support, n (%) | 97 (100) |
VA-ECMO, venoarterial extracorporeal membrane oxygenation; BSA, body surface area; BMI, body mass index; APACHE II, acute physiology and chronic health evaluation II; SOFA, sequential organ failure assessment; Cr, creatinine; eGFR, estimated glomerular filtration rate; TBLI, total bilirubin; PLT, platelet; WBC, white blood cells; NTproBNP, N-terminal pro-brain natriuretic peptide; TnI, troponin; IQR, interquartile range.
62.89% (61/97) of patients experienced NIs after VA-ECMO implantation. As shown in Table 2, 21 pathogens were detected, with the most common gram-negative organisms being Acinetobacter baumannii (n = 21), Klebsiella (n = 17), and Pseudomonas aeruginosa (n = 11). The most common gram-positive organisms were methicillin-resistant Staphylococcus aureus (MRSA, n = 8) and methicillin-sensitive Staphylococcus aureus (MSSA, n = 6). The respiratory tract was the main site of infection.
| Microorganism | RTI | RTI and BSI | RTI and UTI | BSI | SSI | Overall | |
| G-, n (%) | Acinetobacter baumannii | 17 | 3 | 1 | 21 | ||
| Klebsiella | 14 | 3 | 17 | ||||
| Pseudomonas aeruginosa | 8 | 2 | 1 | 11 | |||
| Burkholderia | 7 | 7 | |||||
| Ralstonia solanacearum | 2 | 2 | |||||
| Serratia marcescens | 2 | 2 | |||||
| Stenotrophomonas maltophilia | 1 | 1 | 2 | ||||
| Enterobacter cloacae | 1 | 1 | |||||
| Enterobacter aerogenes | 1 | 1 | |||||
| Escherichia coli | 1 | 1 | |||||
| 65 (69.15) | |||||||
| G+, n (%) | MRSA | 3 | 2 | 3 | 8 | ||
| Staphylococcus aureus | 5 | 1 | 6 | ||||
| Staphylococcus epidermidis | 1 | 1 | 2 | ||||
| Enterococcus faecalis | 1 | 1 | |||||
| Staphylococcus capitis | 1 | 1 | |||||
| 18 (19.15) | |||||||
| Fungi, n (%) | Aspergillus flavus | 2 | 1 | 3 | |||
| Aspergillus fumigatus | 1 | 1 | 2 | ||||
| Candida parapsilosis | 2 | 1 | 3 | ||||
| Candida albicans | 1 | 1 | |||||
| Candida tropicalis | 1 | 1 | 2 | ||||
| 11 (11.7) | |||||||
| Total | 65 | 18 | 2 | 7 | 2 | 94 (100) |
NIs, nosocomial infections; VA-ECMO, venoarterial extracorporeal membrane oxygenation; RTI, respiratory tract infection; BSI, blood stream infection; UTI, urinary tract infection; SSI, surgical site infection; G-, gram negative; G+, gram positive; MRSA, methicillin-resistant staphylococcus aureus.
The incidence of NIs was 117.99 cases per 1000 ECMO-days and 38.61 cases per
1000 ICU-days, respectively. To further investigate the characteristics and
impact of these infections, the cohort was stratified into two groups based on
the occurrence of NIs, as detailed in Table 3. In the NIs group, more patients
were male (67.21% vs. 44.44%, p = 0.028). Longer ECMO duration and
higher incidences of tracheotomy, bronchoscopy, and blood transfusion were more
common in the NIs group (p = 0.003, 0.001, 0.002, and 0.004,
respectively). The CVC duration, ICU stay, and hospital stay were longer in the
NIs group (p = 0.015,
| Variables | Non-NIs (n = 36) | NIs (n = 61) | p value |
| Age, mean (SD), years | 55.14 (14.18) | 54.93 (14.11) | 0.945 |
| Gender, male, n (%) | 16 (44.44) | 41 (67.21) | 0.028* |
| BSA, mean (SD), m2 | 1.55 (0.39) | 1.65 (0.35) | 0.167 |
| BMI, mean (SD), kg/m2 | 24.90 (4.55) | 24.66 (3.82) | 0.787 |
| Smoking, n (%) | 2 (5.56) | 6 (9.84) | 0.706 |
| Alcohol, n (%) | 1 (2.78) | 6 (9.84) | 0.253 |
| Hypertension, n (%) | 13 (36.11) | 27 (44.26) | 0.431 |
| Coronary heart disease, n (%) | 10 (27.78) | 25 (40.98) | 0.191 |
| Diabetes, n (%) | 7 (19.44) | 13 (21.31) | 0.826 |
| Tumor, n (%) | 1 (2.78) | 2 (3.28) | 1.000 |
| Stroke, n (%) | 2 (5.56) | 5 (8.20) | 0.937 |
| APACHE II, mean (SD) | 30.87 (8.20) | 29.21 (7.07) | 0.326 |
| SOFA, mean (SD) | 16.61 (5.53) | 14.84 (4.59) | 0.092 |
| Cr, median (IQR), µmol/L | 134 (108, 196) | 139 (100, 195) | 0.827 |
| eGFR, median (IQR), ml/min/1.73 m2 | 77.15 (61.25, 110.60) | 69.94 (51.74, 103.50) | 0.326 |
| TBLI, median (IQR), µmol/L | 22.75 (11.28, 39.63) | 20.7 (13.55, 33.45) | 0.724 |
| WBC count, median (IQR), |
16.61 (11.14, 21.19) | 16.45 (11.59, 20.91) | 0.662 |
| PLT count, median (IQR), |
121 (63, 201) | 172 (89, 229) | 0.054 |
| PCT, median (IQR), ng/mL | 12.79 (2.59, 28.19) | 7.9 (1.93, 25.52) | 0.395 |
| NT-proBNP, median (IQR), ng/L | 8175 (2154.75, 18,425.00) | 6080 (1057.50, 14,206.50) | 0.544 |
| Lac |
22 (61.11) | 37 (60.61) | 0.965 |
| TnI, median (IQR), µg/L | 12.1 (4.62, 35.90) | 19 (5.29, 71.06) | 0.210 |
| CPR before ECMO, n (%) | 8 (22.22) | 10 (16.39) | 0.476 |
| Maximum VIS, median (IQR) | 160 (85.57, 250.00) | 200 (89.28, 243.04) | 0.635 |
| ECMO duration, median (IQR), days | 4 (2.25, 5.00) | 5 (4.00, 8.00) | 0.003* |
| ECMO flow, mean (SD), L/min | 3.09 (0.70) | 3.12 (0.70) | 0.836 |
| Withdraw ECMO within 30 days, n (%) | 16 (44.44) | 26 (42.62) | 0.861 |
| CRRT support, n (%) | 18 (50.00) | 38 (62.30) | 0.236 |
| IABP support, n (%) | 2 (5.56) | 5 (8.20) | 0.937 |
| Tracheotomy, n (%) | 1 (2.78) | 19 (31.15) | 0.001* |
| Bronchoscopy, n (%) | 2 (5.56) | 20 (32.79) | 0.002* |
| Blood transfusion, n (%) | 26 (72.22) | 58 (95.08) | 0.004* |
| CVC duration, median (IQR), days | 5 (3.00, 8.25) | 8 (5.00, 10.75) | 0.015* |
| Adverse events, n (%) | 16 (44.44) | 35 (57.38) | 0.218 |
| ICU stay, median (IQR), days | 8 (3.0, 14.0) | 17 (11.5, 27.0) | |
| Hospital stay, median (IQR), days | 14 (3.25, 23.75) | 25 (15.00, 37.00) | |
| In-hospital mortality, n (%) | 25 (69.44) | 30 (49.18) | 0.052 |
*p value
Table 4 presents the results of univariate logistic regression analyses
identifying factors associated with NIs. Univariate analysis indicated that
longer durations of ECMO (OR: 1.26, 95% CI: 1.05–1.51, p = 0.013), CVC
duration (OR: 1.13, 95% CI: 1.01–1.27, p = 0.041), ICU stay (OR: 1.14,
95% CI: 1.07–1.22, p
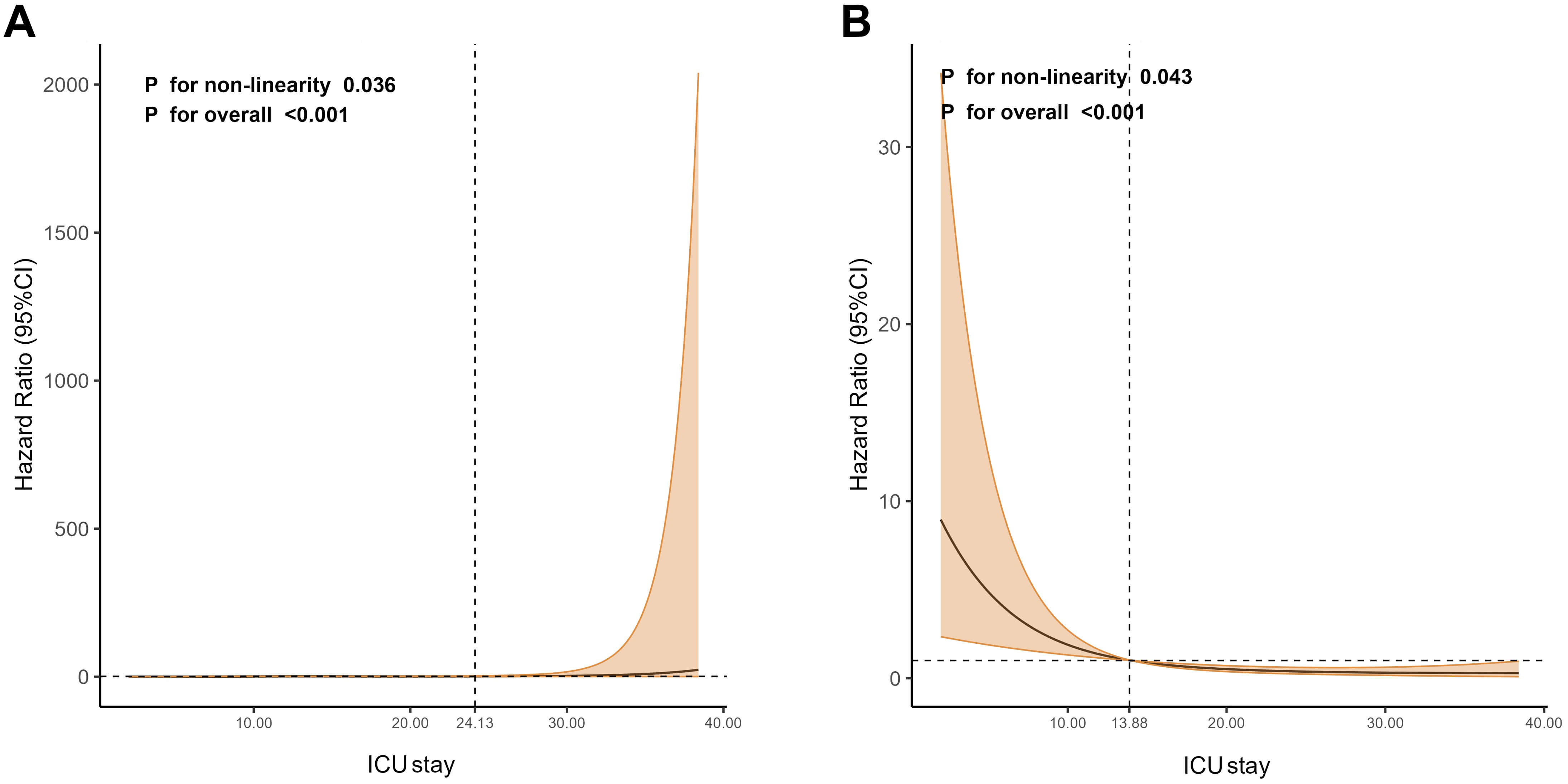 Fig. 1.
Fig. 1.
Restricted cubic spline regression analysis. (A) Nonlinear relationship between ICU stay and NIs. (B) Nonlinear relationship between ICU stay and death. ICU, intensive care unit; NIs, nosocomial infections; CI, confidence interval.
| Univariate analysis | |||
| OR | 95% CI | p value | |
| PLT count | 1.00 | 0.99–1.01 | 0.118 |
| TnI | 1.01 | 0.99–1.03 | 0.179 |
| ECMO duration | 1.26 | 1.05–1.51 | 0.013* |
| CRRT support | 1.65 | 0.72–3.80 | 0.238 |
| Blood transfusion | 7.45 | 1.89–29.28 | 0.004* |
| CVC duration | 1.13 | 1.01–1.27 | 0.041* |
| ICU stay | 1.14 | 1.07–1.22 | |
*p value
Table 5 showed the characteristics of 97 patients stratified into 2 groups based
on survival status. Patients in the survivor group were younger than those in the
nonsurvivor group (51.02
| Variables | Survivor (n = 42) | Non-survivor (n = 55) | p value |
| Age, mean (SD), years | 51.02 (15.20) | 58.05 (12.42) | 0.014* |
| Gender, male, n (%) | 24 (57.14) | 33 (60) | 0.777 |
| BSA, mean (SD), m2 | 1.57 (0.31) | 1.65 (0.41) | 0.311 |
| BMI, mean (SD), kg/m2 | 24.15 (4.12) | 25.22 (4.03) | 0.200 |
| Smoking, n (%) | 4 (9.52) | 4 (7.27) | 0.979 |
| Alcohol, n (%) | 4 (9.52) | 3 (5.45) | 0.710 |
| Hypertension, n (%) | 15 (35.71) | 25 (45.45) | 0.334 |
| Coronary heart disease, n (%) | 13 (30.9) | 22 (40.0) | 0.358 |
| Diabetes, n (%) | 4 (9.52) | 16 (29.09) | 0.018* |
| Tumor, n (%) | 0 (0) | 3 (5.45) | 0.344 |
| Stroke, n (%) | 3 (7.14) | 4 (7.27) | 1.000 |
| APACHE II, mean (SD) | 28.77 (6.59) | 30.65 (8.11) | 0.247 |
| SOFA, mean (SD) | 14.98 (4.97) | 15.89 (5.05) | 0.375 |
| Cr, median (IQR), µmol/L | 125.5 (99.0, 166.5) | 147 (114.5, 217.0) | 0.040* |
| eGFR, median (IQR), mL/min/1.73 m2 | 67.18 (51.59, 102.42) | 84.42 (59.00, 113.87) | 0.127 |
| TBLI, median (IQR), µmol/L | 16.8 (10.30, 33.90) | 35.65 (14.90, 82.18) | 0.017* |
| WBC count, median (IQR), |
11.21 (8.11, 14.61) | 13.69 (10.01, 22.00) | 0.055 |
| PLT count, median (IQR), |
58 (42.5, 115) | 55 (32, 85) | 0.126 |
| PCT, median (IQR), ng/mL | 1.83 (0.71, 11.48) | 4.98 (1.82, 23.25) | 0.060 |
| NT-proBNP, median (IQR), ng/L | 1194.5 (534.18, 4126.50) | 2900 (656.00, 4948.00) | 0.269 |
| Lac |
22 (52.38) | 38 (69.09) | 0.093 |
| TnI, median (IQR), µg/L | 2.84 (0.48, 11.825) | 7.99 (0.13, 20.45) | 0.250 |
| CPR before ECMO, n (%) | 4 (9.52) | 14 (25.45) | 0.046* |
| Maximum VIS, median (IQR) | 200.00 (77.6, 262.5) | 166.42 (97.5, 241.5) | 0.790 |
| ECMO duration, median (IQR), days | 5 (4.0, 5.3) | 5 (3.0, 9.0) | 0.817 |
| Withdraw ECMO within 30days, n (%) | 42 (100) | 25 (45.45) | |
| CRRT support, n (%) | 19 (45.24) | 37 (67.27) | 0.029* |
| IABP support, n (%) | 2 (4.76) | 5 (9.09) | 0.674 |
| Infection, n (%) | 31 (73.81) | 30 (54.55) | 0.052 |
| Tracheotomy, n (%) | 10 (23.81) | 10 (18.18) | 0.497 |
| Bronchoscopy, n (%) | 9 (21.43) | 13 (23.64) | 0.797 |
| Blood transfusion, n (%) | 39 (92.86) | 45 (81.82) | 0.114 |
| CVC duration, median (IQR), days | 7 (5.0, 9.0) | 8 (4.0, 11.3) | 0.884 |
| Adverse events, n (%) | 9 (21.43) | 41 (74.55) | |
| ICU stay, median (IQR), days | 18 (12.75, 27.25) | 11 (4.00, 18.00) |
*p value
Table 6 shows the univariate analyses of factors associated with death.
Univariate logistic regression analysis suggested that age (OR: 1.04, 95% CI:
1.01–1.07, p = 0.017), history of diabetes (OR: 3.90, 95% CI:
1.19–12.73, p = 0.024), total bilirubin (OR: 1.01, 95% CI: 1.00–1.02,
p = 0.020), CRRT support (OR: 2.49, 95% CI: 1.09–5.70, p =
0.031), and occurrence of adverse events (OR: 11.85, 95% CI: 4.52–31.08,
p
| Univariate analysis | |||
| OR | 95% CI | p value | |
| Age | 1.04 | 1.01–1.07 | 0.017* |
| Diabetes | 3.90 | 1.19–12.73 | 0.024* |
| Cr | 1.00 | 0.10–1.01 | 0.283 |
| TBLI | 1.01 | 1.00–1.02 | 0.020* |
| CPR before ECMO | 3.24 | 0.98–10.72 | 0.054 |
| CRRT support | 2.49 | 1.09–5.70 | 0.031* |
| Adverse events | 11.85 | 4.52–31.08 | |
| ICU stay | 0.92 | 0.87–0.96 | |
*p value
In our single-center retrospective cohort study, we found that NIs were common in ICU patients supported by VA-ECMO. The incidence of NIs was 62.89%. The most common microorganism associated with NIs was Acinetobacter baumannii. Longer durations of ECMO and CVC, blood transfusion, and prolonged ICU stay were associated factors for NIs. In addition, after the inflection point of approximately 24 days of ICU stay, the incidence of NIs increased dramatically. Finally, the occurrence of adverse events correlated with increased in-hospital mortality.
In our cohort, gram-negative bacteria were the predominant microorganisms causing NIs in patients supported by VA-ECMO. This finding is consistent with previous studies [14, 15, 16]. Xia et al. [17] have identified Acinetobacter baumannii, Klebsiella, and Pseudomonas aeruginosa as prevalent pathogens responsible for NIs. Patients often require mechanical ventilators, CRRT, and intravenous catheters at the same time during ECMO support. Sites of endotracheal tubes and cannulas are often colonized by microorganisms. Moreover, ECMO patients with compromised immune systems and mucosal barrier damage are prone to infections [18]. However, the effectiveness of prophylactic antibiotic therapy in the prevention of NIs remains inconclusive [19, 20]. Thus, identifying associated factors for NIs and adopting appropriate treatment strategies are critical. In our study, associated factors for NIs included blood transfusion, long ECMO duration, CVC duration, and ICU stay. The need for blood transfusion was an indication that the patient was in a more severe condition. However, a blood transfusion is essential for patients experiencing haemorrhage and disseminated intravascular coagulation. Moreover, the association between blood transfusion and adverse outcomes has been attributed to immunosuppression in the recipient, which can lead to NIs. This finding is consistent with previous studies [21]. This finding has been confirmed by similar studies [22, 23]. A longer duration of ECMO not only increases the risk of prolonged exposure to invasive devices, but also leads to extended ICU and hospital stays. These factors are associated with an increased risk of NIs [24]. However, a prolonged ECMO duration is likely a strong marker of underlying critical illness severity and complexity rather than an independent causal factor for acquiring NIs. In contrast, the persistent association with ICU length of stay, even after the landmark adjustment, strengthens its role as a robust, independent risk factor, highlighting the importance of cumulative exposure to the hospital environment and invasive procedures. These identified associated factors for NIs are likely strong markers of underlying critical illness severity and prolonged exposure to invasive care, rather than independent causal agents of infection. For example, longer ECMO support may reflect more complex primary conditions (e.g., cardiogenic shock or respiratory failure) that inherently increase vulnerability to infections due to immune suppression or compromised barriers. Blood transfusion often signifies complications like hemorrhage or anemia, which are common in critically ill patients and may serve as indicators of disease burden rather than direct causes of infection. Similarly, extended CVC use and ICU stay highlight the intensity of medical interventions and overall patient acuity, which collectively elevate the risk of exposure to pathogens. Therefore, these factors should be interpreted as proxies for the cumulative impact of critical illness and invasive care, underscoring the need for integrated management strategies to mitigate infection risks in this vulnerable population. In contrast to previous studies, we estimated the inflection points of ECMO duration and ICU stay to predict the occurrence of NIs using RCS analysis. The relationship between ECMO duration and NIs incidence was a positive linear correlation. This indicates that prolonged ECMO increases the risk of NIs. The occurrence of NIs increased significantly when ICU stay exceeded approximately 24 days. These findings provide valuable insights for the prevention and management of VA-ECMO-associated NIs.
Previous studies have demonstrated that NIs patients have a lower ECMO weaning survival rate and a higher risk of in-hospital mortality compared with non-NIs patients [25, 26]. Wang J and colleagues reported that the incidence of NIs was associated with a 3.726-fold higher risk of in-hospital death in ECMO-supported patients [27]. In our study, we did not find a statistically significant association between NIs and in-hospital mortality in univariate analysis (p = 0.052). However, this lack of significance may be attributed to limited statistical power due to our small sample size, as well as potential survivor bias, wherein only patients with longer survival times could develop NIs. Additionally, survivor bias could have influenced our results, as patients who died early in the ICU stay (median 13.88 days) may not have been exposed to NIs risks, thereby diluting the observed effect. Instead, we identified that older age, a history of diabetes, and hepatic or renal dysfunction were significantly associated factors for in-hospital mortality. This finding underscores that a patient’s baseline pathophysiological status prior to ECMO initiation is critical for determining outcomes. Furthermore, a shorter ICU stay was also associated with mortality. This is not contradictory, as most deaths in our cohort occurred early during the ICU stay (within a median of 13.88 days), likely due to severe initial clinical deterioration. Therefore, we speculate that patients with a worse clinical status at baseline are at a higher risk of early death despite VA-ECMO support. Conversely, for those who survive this critical early phase, a prolonged ICU stay may subsequently increase the risk of developing NIs.
Our study has several limitations. First, its retrospective, single-center design and small sample size may limit the generalizability of our findings to the broader population of VA-ECMO patients who develop NIs in the ICU. The small sample size may have increased the risk of Type II errors, meaning we might have missed a true association between NIs and mortality. Furthermore, the higher mortality in the non-NIs group could be partly explained by severe initial clinical deterioration, which led to early death before NIs could develop. Second, due to the limited sample size and number of outcome events in our cohort, we were unable to construct stable multivariate models to adjust for potential confounders. Therefore, the identified associated factors are based on univariate analyses and should be interpreted with caution. Future studies with larger sample sizes are warranted to independently validate these associations. Third, the diagnosis of NIs was based on the Centers for Disease Control (CDC) 1988 criteria. While these criteria were rigorously applied, they are less specific than contemporary definitions. However, the consistent application of the same criteria across all patient groups ensures the internal validity of the comparative analyses presented herein. Future studies utilizing current standardized definitions are warranted to confirm our findings. Finally, this study did not analyze the duration of mechanical ventilation, an important potential risk factor for NIs, due to the unavailability of these data in our records. This may limit the comprehensiveness of our risk factor assessment for NIs. Despite these limitations, our findings offer valuable insights, particularly in demonstrating the use of RCS to explore nonlinear correlations between variables and outcomes. We believe that future large-scale, multicenter studies are warranted to validate our findings and provide more robust evidence on the characteristics and impact of NIs in VA-ECMO patients.
Our study found a high incidence of NIs among critically ill patients supported by VA-ECMO, with Acinetobacter baumannii being the most prevalent pathogen. Independent associated factors for NIs included prolonged durations of ECMO and CVC placement, as well as blood transfusion. Furthermore, a prolonged ICU stay is a key modifiable risk factor for NIs in this population. The role of ECMO duration should be de-emphasized as a direct driver of infection risk and instead presented as a proxy indicator for a prolonged and complicated clinical course.
Data are available from the corresponding author upon reasonable request.
ZC, YJ and YL designed the research study. ZC and YJ performed the research. ZC and HW analyzed the data. ZC and YJ drafted the manuscript. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Bengbu Medical University (Protocol No. 2025 [039]). Written informed consent was obtained from all patients for the publication of their data and any accompanying images.
Not applicable.
The study was funded by the Natural Science Research Project of Anhui Educational Committee (2024AH051191, 2024AH052829), Science Foundation for Outstanding Youth of the First Affiliated Hospital of Bengbu Medical College (2021byyfyjq02).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF46896.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

