1 Department of Cardiac Surgery, Harefield Hospital, Royal Brompton and Harefield Hospitals, part of Guy's and St Thomas NHS Trust, UB9 6JH London, UK
2 Department of Cardiothoracic Transplantation Surgery, Harefield Hospital, Royal Brompton and Harefield Hospitals, part of Guy's and St Thomas NHS Trust, UB9 6JH London, UK
3 Department of Anaesthesia and Critical Care, Harefield Hospital, Royal Brompton and Harefield Hospitals, part of Guy's and St Thomas NHS Trust, UB9 6JH London, UK
4 Division of Anaesthesia, Pain Medicine and Intensive Care, Department of Surgery and Cancer, Imperial College London, SW7 2BX London, UK
5 Department of Anaesthesia and Intensive Care, Semmelweis University, 1085 Budapest, Hungary
Abstract
Right ventricular failure (RVF) remains a major determinant of morbidity and mortality following left ventricular assist device (LVAD) implantation. In its most severe form, RVF necessitates right ventricular assist device (RVAD) support, which is associated with markedly worse early and long-term outcomes. While numerous studies have examined predictors of post-LVAD RVF, few have focused specifically on RVAD implantation as a discrete and clinically decisive endpoint. We therefore performed a systematic review and meta-analysis to identify robust preoperative predictors of RVAD requirement following LVAD implantation.
A systematic literature search of MEDLINE, EMBASE, SCOPUS, and PubMed was conducted from inception to August 2025 in accordance with PRISMA guidelines. Studies enrolling adult patients undergoing durable or temporary LVAD implantation and reporting preoperative predictors of postoperative RVAD requirement were included. Pooled odds ratios (ORs) and mean differences (MDs) with 95% confidence intervals (CIs) were calculated using random-effects models. Heterogeneity was assessed using the I2 statistic, with predefined sensitivity and subgroup analyses based on LVAD era and study size.
Twenty studies met inclusion criteria, of which nineteen comprising 31,591 patients were included in the meta-analysis. Several consistent preoperative predictors of RVAD requirement were identified across clinical, haemodynamic, echocardiographic, and laboratory domains. Patients requiring RVAD were younger (MD –3.57 years) and more frequently female, had a higher likelihood of prior cardiac surgery, INTERMACS Profile 1 status, and need for preoperative mechanical support including ventilation and intra-aortic balloon pump. Haemodynamic predictors included elevated central venous pressure, higher CVP/PCWP ratio, lower cardiac index, and reduced right ventricular stroke work index. Echocardiographic predictors included severe tricuspid regurgitation and lower tricuspid annular plane systolic excursion. Laboratory markers of hepatic dysfunction, coagulopathy, malnutrition, anaemia, thrombocytopenia, and elevated natriuretic peptides were also significantly associated with RVAD implantation. Subgroup analyses demonstrated consistent direction of effects across LVAD eras and study sizes.
RVAD requirement after LVAD implantation is driven by a constellation of high clinical acuity, haemodynamic compromise, and end-organ dysfunction rather than any single isolated variable. A comprehensive, multi-parameter preoperative assessment is essential to identify patients at highest risk and to guide patient selection, preoperative optimisation, and consideration of planned biventricular support strategies.
Keywords
- left ventricular assist device
- right ventricular assist device
- right ventricular failure
- mechanical circulatory support
- risk stratification
- preoperative predictors
- biventricular support
Left ventricular assist devices (LVADs) are an established therapy for patients with advanced chronic heart failure (ACHF), significantly improving survival and quality of life as a bridge to transplantation or as a destination therapy [1]. According to recent registry data, the number of LVAD implantations has steadily increased, with more than 15,000 patients supported [2]. However, the success of LVAD therapy is often compromised by the early morbidity and mortality caused by right ventricular failure (RVF) [3]. Severe RVF exhibits an incidence as high as 50% after LVAD implantation, resulting in perioperative mortality rates of 19% to 43% and driving significant complications, including end-organ dysfunction and the need for prolonged intensive care and hospitalization [4, 5].
The most severe form of RVF necessitates the implantation of a right ventricular assist device (RVAD). Meanwhile, the pathophysiology of post-LVAD RVF is multifactorial. The abrupt increase in venous return following LVAD initiation increases RV preload, while the leftward shift of the interventricular septum can alter RV geometry and impair contractility. These changes can overwhelm a ventricle that has already been compromised by chronic heart failure [6]. Thus, recognizing this risk, numerous studies have sought to identify preoperative predictors of RVF to improve the selection of candidates for isolated LVAD therapy and to identify those who might benefit from a planned biventricular ventricular assist device (BiVAD) strategy. These efforts have led to the development of several clinical risk scores, such as the Michigan michigan right ventricular failure risk score (RVFRS), the CRITT score, and the EUROMACS-right sided heart failure (RHF) score [7]. However, these scores have often yielded inconsistent predictors, and external validation studies have demonstrated only modest predictive power, limiting their widespread clinical application [8, 9].
Collectively, this systematic review and meta-analysis were conducted to aggregate the current evidence given the profound impact of post-LVAD and RVAD requirements and the ongoing challenges in risk stratification. Finally, this review aimed to identify the most robust and consistent preoperative predictors of postoperative RVAD requirement in patients undergoing durable, continuous-flow LVAD (CF-LVAD) implantation.
The review was registered at the Prospective Register of Systematic Reviews database (PROSPERO). This systematic review and meta-analysis were performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A comprehensive literature search of OVID MEDLINE, EMBASE, SCOPUS, and PubMed was conducted from database inception until August 2025. The following keywords and MeSH terms were used in various combinations: “left ventricular assist device”, “LVAD”, “right ventricular assist device”, “RVAD”, “right ventricular failure”, and “predictors”. Reference lists of relevant articles were also screened manually to identify additional studies.
Studies were eligible for inclusion if they met the following criteria: (1)
original research articles; (2) included adult patients (
Exclusion criteria included studies focusing solely on paediatric populations, those without clear separation of RVAD from broader right ventricular failure cohorts, and studies without extractable or comparable data. Case reports, editorials, reviews, and conference abstracts were also excluded.
Three reviewers (MC, AD, IZ) independently extracted data, including study characteristics (first author, year, design), patient population (sample size, LVAD type, INTERMACS profile), and preoperative variables. Variables were grouped as: (1) clinical/demographic (age, sex, INTERMACS profile, prior surgery, mechanical circulatory support); (2) haemodynamic central venous pressure (CVP), pulmonary capillary wedge pressure (PCWP), cardiac index, RV stroke work index); (3) echocardiographic tricuspid annular plane systolic excursion (TAPSE), right ventricular dimensions, tricuspid regurgitation); and (4) laboratory creatinine, bilirubin, albumin, international normalised ratio (INR). The primary outcome was the requirement for postoperative RVAD support. For studies that reported continuous variables as medians with interquartile ranges or ranges, we converted these values to means and standard deviations using the validated method described by Wan et al. [10]. This approach provides unbiased estimators for sample means and standard deviations across various sample sizes and is widely recommended for meta-analytic pooling of continuous data.
The Revised Cochrane risk-of-bias 2 (RoB 2) tool was employed to assess bias in randomized studies [11]. For observational studies, the Risk of Bias In Non-randomized Studies of Interventions (ROBINS-I) scale was utilized [12].
Meta-analyses were performed using Review Manager (RevMan) Version 5.4.1 (The
Cochrane Collaboration, Copenhagen, Denmark), developed by The Nordic Cochrane
Centre in Copenhagen, Denmark. For dichotomous variables, pooled ORs with 95%
CIs were calculated; for continuous variables, MDs with 95% CIs were used. A
random-effects model was employed to account for between-study variability.
Statistical significance was set at p
To further investigate the stability of our findings and explore sources of
statistical heterogeneity, a leave-one-out sensitivity analysis was performed.
For any preoperative predictor where heterogeneity was classified as high
(I2
To explore potential sources of heterogeneity, we performed predefined subgroup
analyses based on (1) LVAD type and device era, stratifying studies into
pulsatile-flow and continuous-flow cohorts, and (2) study sample size,
categorising studies as small/medium (
A total of 20 studies met the inclusion criteria for the systematic review (Fig. 1). Of these, 19 studies including 31,591 patients [13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31] provided sufficient quantitative data for inclusion in the meta-analysis. The study by Rivas-Lasarte et al. [8], which focused on validating a risk score rather than providing primary data on individual predictors, was included in the qualitative review and diagnostic accuracy summary but not the quantitative synthesis of individual predictors.
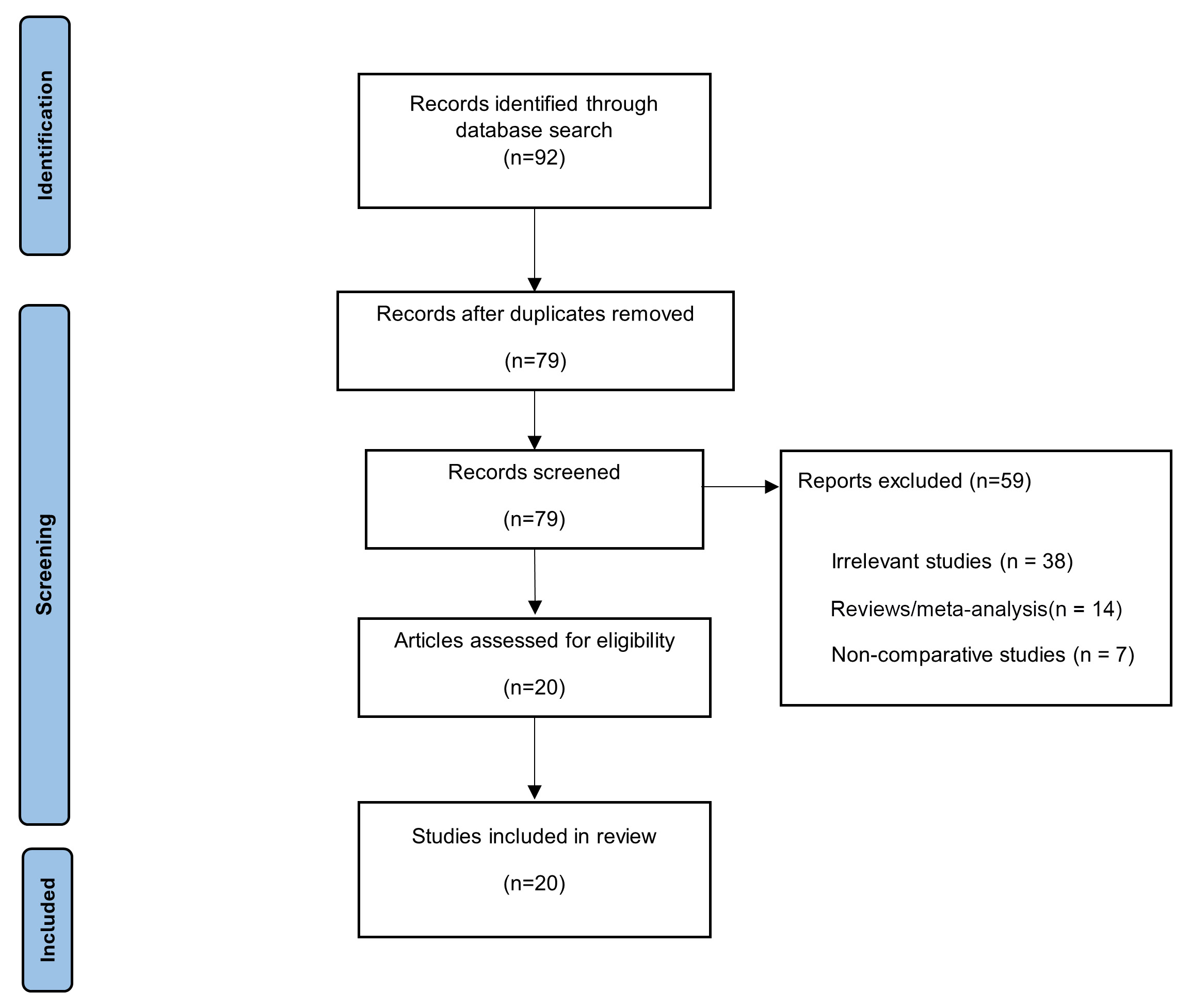 Fig. 1.
Fig. 1.
Flow diagram demonstrating assessment of the available literature using CEBM criteria. CEBM, Center for Evidence-Based Medicine.
The included studies were published between 1999 and 2023 and were geographically diverse, with the majority from the USA, several from Europe (Germany, UK, France, Belgium), and one from Japan (Table 1, Ref. [8, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31]). The studies were predominantly retrospective and varied significantly in size, from single-centre cohorts of fewer than 50 patients to large, multicentre registry analyses encompassing over 28,000 individuals.
| First author | Year | Study design | Country | Time interval of recruitment | No. of patients (total) | No. with LVAD only | No. with RVAD | LVAD types |
| Ahmed et al. [14] | 2023 | Retrospective, Registry | USA | 2010–2020 | 28,971 | 27,325 | 1646 | CF-LVADs (HMII, HVAD, HM3) |
| Aissaoui et al. [31] | 2013 | Retrospective, Single-Centre | Germany | 2001–2011 | 488 | 443 | 45 | Multiple CF & Pulsatile |
| Charisopoulou et al. [17] | 2019 | Retrospective, Single-Centre | UK | 2016–2018 | 70 | 56 | 14 | CF-LVADs |
| Deschka et al. [20] | 2016 | Retrospective, Multi-Centre | Germany | 2011–2014 | 53 | 28 | 25 | CF-LVADs (HMII, HeartWare) |
| Fischer and Kirsch [15] | 2018 | Retrospective, Multi-Centre | France | 2012–2014 | 44 | 22 | 22 | HeartMate II |
| Fitzpatrick et al. [29] | 2008 | Retrospective, Single-Centre | USA | 1995–2007 | 266 | 167 | 99 | Multiple CF & Pulsatile |
| Fukamachi et al. [28] | 1999 | Retrospective, Single-Centre | USA | 1991–1996 | 100 | 89 | 11 | HeartMate (Pulsatile) |
| Kiernan et al. [13] | 2017 | Retrospective, Registry | USA | 2006–2014 | 9976 | 9590 | 386 | CF-LVADs |
| Kormos et al. [18] | 2010 | Prospective, Multi-Centre | USA | 2005–2008 | 484 | 386 | 98 | HeartMate II |
| Lazar et al. [16] | 2013 | Retrospective, Single-Centre | USA | 2006–2011 | 139 | 105 | 34 | HeartMate II |
| Morgan et al. [22] | 2004 | Retrospective, Single-Centre | USA | 1993–2003 | 243 | 226 | 17 | HeartMate (Pulsatile) |
| Ochiai et al. [27] | 2002 | Retrospective, Single-Centre | USA | 1991–2001 | 245 | 222 | 23 | HeartMate & Novacor (Pulsatile) |
| Patil et al. [21] | 2015 | Retrospective, Single-Centre | UK | 2003–2013 | 152 | 117 | 35 | CF-LVADs (HMII, HVAD, Jarvik) |
| Pettinari et al. [26] | 2012 | Retrospective, Single-Centre | Belgium | Not Specified | 59 | 45 | 14 | CF-LVADs (HMII, INCOR) |
| Rivas-Lasarte et al. [8] | 2021 | Retrospective, Multi-Centre | USA | 2007–2017 | 662 | 451 | 211 | CF-LVADs (HMII, HVAD, HM3) |
| Shiga et al. [25] | 2012 | Retrospective, Single-Centre | Japan | 2002–2011 | 79 | 70 | 9 | Multiple CF & Pulsatile |
| Takeda et al. [24] | 2013 | Retrospective, Single-Centre | USA | 2000–2010 | 282 | 242 | 40 | HeartMate I & II |
| Wang et al. [23] | 2012 | Retrospective, Single-Centre | USA | 1996–2009 | 183 | 156 | 27 | Multiple CF & Pulsatile |
| Yost et al. [30] | 2016 | Retrospective, Single-Centre | USA | 2005–2013 | 256 | 200 | 56 | CF-LVADs (HMII, HeartWare) |
| Yoshioka et al. [19] | 2017 | Retrospective, Single-Centre | USA | 2009–2014 | 305 | 278 | 27 | CF-LVADs (HMII, HeartWare) |
CF-LVAD, Continuous-Flow Left Ventricular Assist Device; HMII, HeartMate II; HVAD, HeartWare HVAD; HM3, HeartMate 3; RHF, Right Heart Failure.
The overall risk of bias for the included studies was judged to be moderate. As all 20 studies were non-randomized and predominantly retrospective in nature, they are inherently susceptible to certain biases. The primary concerns across most studies were a moderate risk of bias due to confounding (arising from unmeasured variables like intraoperative events or institutional protocols) and a moderate risk of bias from the selection of reported results (given the large number of potential predictors analysed). Domains such as participant selection, classification of interventions, and measurement of the outcome (RVAD implantation) were generally at low risk of bias due to the use of consecutive patient cohorts and the objective nature of the outcome.
The meta-analysis identified numerous significant preoperative predictors of postoperative RVAD requirement across clinical, hemodynamic, echocardiographic, and laboratory domains. The complete results for all analyzed variables, including both significant and non-significant findings, are detailed in Tables 2,3,4. The individual forest plot for each analysed variable is available in the Supplementary Material.
| Parameter | No. of studies | Participants | Effect estimate (95% CI) | p-value | Heterogeneity (I2) | p-value (Q) | |
| Demographics | |||||||
| Age (years) | 18 | 31,591 | MD: –3.57 [–5.20, –1.93] | 54% | 0.003 | ||
| Male Gender | 19 | 41,243 | OR: 0.56 [0.41, 0.77] | 57% | 0.002 | ||
| Race (African American) | 3 | 28,390 | OR: 1.21 [1.01, 1.44] | 0.04 | 0% | 0.700 | |
| Race (Caucasian) | 3 | 1065 | OR: 0.88 [0.41, 1.87] | 0.74 | 74% | 0.020 | |
| Body Surface Area (m2) | 8 | 1760 | MD: –0.11 [–0.22, 0.00] | 0.05 | 87% | ||
| BMI (kg/m2) | 7 | 29,128 | MD: 0.56 [–0.40, 1.53] | 0.25 | 68% | 0.004 | |
| Weight (kg) | 3 | 483 | MD: –4.65 [–14.12, 4.81] | 0.33 | 87% | ||
| Medical & surgical history | |||||||
| Ischaemic Cardiomyopathy | 15 | 12,831 | OR: 1.04 [0.79, 1.36] | 0.77 | 62% | ||
| Dilated Cardiomyopathy | 6 | 28,932 | OR: 0.89 [0.72, 1.11] | 0.31 | 0% | 0.650 | |
| Prior Cardiac Surgery | 8 | 11,756 | OR: 1.78 [1.18, 2.70] | 0.006 | 80% | ||
| Diabetes Mellitus | 6 | 1320 | OR: 1.07 [0.64, 1.79] | 0.79 | 50% | 0.080 | |
| Hypertension | 2 | 538 | OR: 0.76 [0.43, 1.36] | 0.35 | 0% | 0.690 | |
| Chronic Kidney Disease | 2 | 315 | OR: 0.84 [0.34, 2.05] | 0.71 | 63% | 0.100 | |
| COPD | 5 | 1038 | OR: 0.94 [0.48, 1.82] | 0.85 | 0% | 0.700 | |
| Peripheral Vascular Disease | 2 | 211 | OR: 1.02 [0.33, 3.15] | 0.97 | 0% | 0.870 | |
| Cerebrovascular Accident (CVA) | 3 | 408 | OR: 5.35 [0.64, 44.56] | 0.12 | 0% | 0.440 | |
| Smoking History | 2 | 434 | OR: 0.80 [0.26, 2.53] | 0.71 | 71% | 0.060 | |
| Preoperative status & support | |||||||
| INTERMACS Profile 1 | 6 | 28,285 | OR: 2.27 [1.04, 4.96] | 0.04 | 93% | ||
| Preop. Mech. Ventilation | 12 | 40,335 | OR: 3.47 [2.44, 4.94] | 74% | |||
| Preop. Hemofiltration | 5 | 10,654 | OR: 3.17 [1.64, 6.12] | 68% | 0.010 | ||
| Preop. IABP | 13 | 12,746 | OR: 2.00 [1.54, 2.59] | 48% | 0.030 | ||
| Preop. ECMO | 9 | 38,751 | OR: 2.17 [0.97, 4.85] | 0.06 | 76% | ||
| Inotropic Support Preop. | 4 | 28,302 | OR: 0.92 [0.42, 1.99] | 0.83 | 75% | 0.007 | |
| No. of Inotropes | 2 | 640 | MD: 0.16 [–0.10, 0.41] | 0.23 | 0% | 0.580 | |
| Device strategy & type | |||||||
| Bridge to Transplantation | 7 | 29,318 | OR: 1.14 [0.84, 1.54] | 0.4 | 38% | 0.130 | |
| Destination Therapy | 8 | 39,266 | OR: 1.07 [0.93, 1.23] | 0.35 | 0% | 0.710 | |
| Pulsatile-flow LVAD | 2 | 449 | OR: 2.22 [0.34, 14.62] | 0.4 | 78% | 0.030 | |
| Risk scores | |||||||
| Michigan RV Risk Score | 2 | 532 | MD: 1.33 [0.68, 1.99] | 0% | 0.420 | ||
BMI, Body Mass Index; CI, Confidence Interval; COPD, Chronic Obstructive Pulmonary Disease; CVA, Cerebrovascular Accident; ECMO, Extracorporeal Membrane Oxygenation; IABP, Intra-Aortic Balloon Pump; INTERMACS, Interagency Registry for Mechanically Assisted Circulatory Support; LVAD, Left Ventricular Assist Device; MD, Mean Difference; OR, Odds Ratio; RV, Right Ventricular.
| Parameter | No. of studies | Participants | Effect estimate (95% CI) | p-value | Heterogeneity (I2) | p-value (Q) | |
| Pressures (mmHg) | |||||||
| CVP | 17 | 40,993 | MD: 2.82 [1.35, 4.29] | 93% | |||
| Mean PAP | 14 | 3080 | MD: –3.22 [–5.29, –1.16] | 0.002 | 68% | ||
| Systolic PAP | 8 | 11,400 | MD: –4.22 [–7.46, –0.98] | 0.010 | 72% | ||
| Diastolic PAP | 7 | 11,348 | MD: –1.01 [–3.18, 1.15] | 0.360 | 32% | 0.190 | |
| PCWP | 12 | 12,406 | MD: –0.02 [–1.21, 1.17] | 0.980 | 61% | 0.003 | |
| MAP | 5 | 10,887 | MD: –3.60 [–5.44, –1.76] | 0% | 0.520 | ||
| Systolic Blood Pressure | 6 | 1404 | MD: –3.17 [–8.08, 1.75] | 0.210 | 63% | 0.020 | |
| Diastolic Blood Pressure | 2 | 325 | MD: –3.61 [–6.33, –0.90] | 0.009 | 0% | 0.580 | |
| RVDP | 2 | 253 | MD: 4.91 [–2.92, 12.75] | 0.220 | 90% | 0.001 | |
| RVSP | 2 | 235 | MD: 1.41 [–3.92, 6.74] | 0.600 | 0% | 0.900 | |
| TPG | 5 | 10,551 | MD: –0.86 [–2.57, 0.86] | 0.330 | 59% | 0.040 | |
| Calculated indices | |||||||
| CVP/PCWP Ratio | 5 | 10,855 | MD: 0.15 [0.11, 0.18] | 0% | 0.510 | ||
| Cardiac Index (L/min/m2) | 13 | 12,390 | MD: –0.18 [–0.25, –0.10] | 0% | 0.500 | ||
| Cardiac Output (L/min) | 7 | 1193 | MD: –0.28 [–0.62, 0.05] | 0.100 | 56% | 0.030 | |
| RVSWI (mmHg |
6 | 11,010 | MD: –9.94 [–16.05, –3.82] | 0.001 | 94% | ||
| RVSW (mmHg mL) | 2 | 417 | MD: –95.08 [–316.02, 125.86] | 0.400 | 96% | ||
| PVR (Wood Units) | 10 | 12,122 | MD: 0.15 [–0.38, 0.68] | 0.580 | 83% | ||
| PVRI (WU |
5 | 684 | MD: –0.55 [–0.71, –0.39] | 0% | 0.820 | ||
| SVR (dynes |
3 | 981 | MD: –106.86 [–315.43, 101.72] | 0.320 | 45% | 0.160 | |
| Other | |||||||
| Heart Rate (bpm) | 8 | 11,500 | MD: 4.16 [1.49, 6.83] | 0.002 | 53% | 0.040 | |
| SvO2 (%) | 2 | 418 | MD: –1.13 [–11.31, 9.04] | 0.830 | 90% | 0.002 | |
| Echocardiographic parameters | |||||||
| Severe TR ( |
4 | 37,981 | OR: 1.40 [1.03, 1.89] | 0.030 | 52% | 0.1 | |
| Mitral Regurgitation ( |
4 | 37,981 | OR: 0.90 [0.78, 1.04] | 0.160 | 0% | 0.6 | |
| TAPSE (mm) | 3 | 266 | MD: –2.85 [–5.90, 0.19] | 0.070 | 88% | ||
| LVEF (%) | 11 | 2050 | MD: –0.42 [–1.35, 0.50] | 0.370 | 0% | 0.89 | |
| LVEDD (mm) | 6 | 10,563 | MD: –2.27 [–5.41, 0.88] | 0.160 | 77% | ||
| RVD (mm) | 2 | 222 | MD: –1.93 [–6.34, 2.49] | 0.390 | 94% | ||
CI, Confidence Interval; CVP, Central Venous Pressure; LVEDD, Left Ventricular End-Diastolic Diameter; LVEF, Left Ventricular Ejection Fraction; MAP, Mean Arterial Pressure; MD, Mean Difference; OR, Odds Ratio; PAP, Pulmonary Artery Pressure; PCWP, Pulmonary Capillary Wedge Pressure; PVR, Pulmonary Vascular Resistance; PVRI, Pulmonary Vascular Resistance Index; RVD, Right Ventricular Diameter; RVDP, Right Ventricular Diastolic Pressure; RVSP, Right Ventricular Systolic Pressure; RVSW, Right Ventricular Stroke Work; RVSWI, Right Ventricular Stroke Work Index; SvO2, Mixed Venous Oxygen Saturation; SVR, Systemic Vascular Resistance; TAPSE, Tricuspid Annular Plane Systolic Excursion; TPG, Transpulmonary Gradient; TR, Tricuspid Regurgitation.
| Parameter | No. of studies | Participants | Effect estimate (95% CI) | p-value | Heterogeneity (I2) | p-value (Q) | |
| Hepatic function | |||||||
| Total Bilirubin (mg/dL) | 16 | 40,809 | MD: 0.36 [0.15, 0.57] | 93% | |||
| AST (U/L) | 11 | 12,315 | MD: 35.04 [5.16, 64.91] | 0.020 | 87% | ||
| ALT (U/L) | 7 | 1242 | MD: –11.13 [–39.14, 16.89] | 0.440 | 69% | 0.003 | |
| Renal function | |||||||
| BUN (mg/dL) | 12 | 12,517 | MD: 1.87 [–0.17, 3.92] | 0.070 | 45% | 0.050 | |
| Creatinine (mg/dL) | 16 | 40,809 | MD: 0.09 [–0.00, 0.18] | 0.060 | 45% | 0.030 | |
| Haematologic & coagulation | |||||||
| INR | 7 | 11,438 | MD: 0.17 [0.05, 0.29] | 0.006 | 79% | ||
| Hemoglobin (g/dL) | 7 | 11,121 | MD: –0.53 [–1.01, –0.05] | 0.030 | 66% | 0.007 | |
| Platelets ( |
9 | 11,558 | MD: –13.32 [–22.57, –4.07] | 0.005 | 0% | 0.460 | |
| Hematocrit (%) | 5 | 1230 | MD: –1.46 [–3.09, 0.18] | 0.080 | 75% | 0.002 | |
| APTT (s) | 3 | 525 | MD: 1.83 [–4.20, 7.86] | 0.550 | 77% | 0.010 | |
| Nutritional & inflammatory | |||||||
| Albumin (g/dL) | 10 | 39,338 | MD: –0.20 [–0.30, –0.10] | 63% | 0.004 | ||
| Serum Total Protein (g/dL) | 3 | 663 | MD: –0.29 [–0.66, 0.08] | 0.130 | 49% | 0.140 | |
| WBC ( |
9 | 11,880 | MD: 1.29 [0.70, 1.87] | 58% | 0.010 | ||
| C-Reactive Protein | 4 | 775 | MD: –1.34 [–9.90, 7.22] | 0.760 | 79% | 0.001 | |
| Cardiac biomarkers | |||||||
| BNP (pg/mL) | 4 | 10,346 | MD: 722.79 [26.05, 1419.53] | 0.040 | 93% | ||
| Other | |||||||
| Lactate (mmol/L) | 2 | 103 | MD: 0.24 [–0.19, 0.68] | 0.270 | 0% | 0.730 | |
| Serum Sodium (mmol/L) | 6 | 10,843 | MD: –0.88 [–1.59, –0.17] | 0.020 | 0% | 0.630 | |
CI, Confidence Interval; CVP, Central Venous Pressure; LVEDD, Left Ventricular End-Diastolic Diameter; LVEF, Left Ventricular Ejection Fraction; MAP, Mean Arterial Pressure; MD, Mean Difference; OR, Odds Ratio; PAP, Pulmonary Artery Pressure; PCWP, Pulmonary Capillary Wedge Pressure; PVR, Pulmonary Vascular Resistance; PVRI, Pulmonary Vascular Resistance Index; RVD, Right Ventricular Diameter; RVDP, Right Ventricular Diastolic Pressure; RVSP, Right Ventricular Systolic Pressure; RVSW, Right Ventricular Stroke Work; RVSWI, Right Ventricular Stroke Work Index; SvO2, Mixed Venous Oxygen Saturation; SVR, Systemic Vascular Resistance; TAPSE, Tricuspid Annular Plane Systolic Excursion; TPG, Transpulmonary Gradient; TR, Tricuspid Regurgitation.
Patients requiring RVAD support were significantly younger than those
not requiring RVAD (mean difference [MD] –3.57 years, 95% CI –5.20 to –1.93,
p
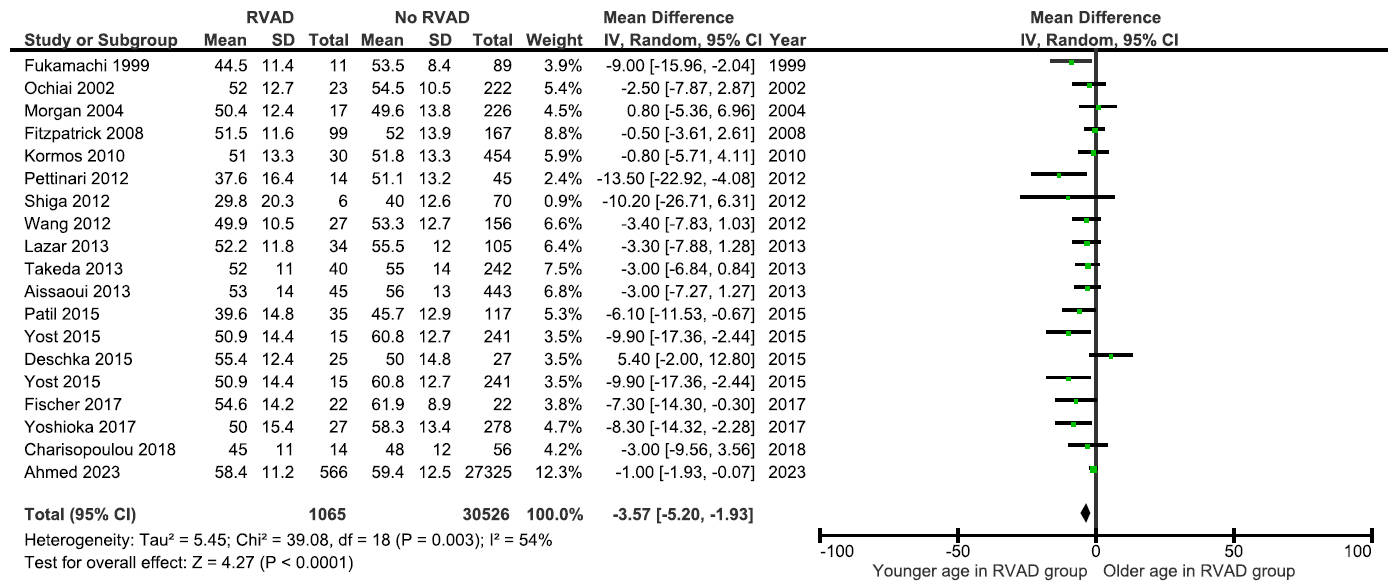 Fig. 2.
Fig. 2.
Forest plot of the mean difference in preoperative age between patients with and without postoperative RVAD requirement.
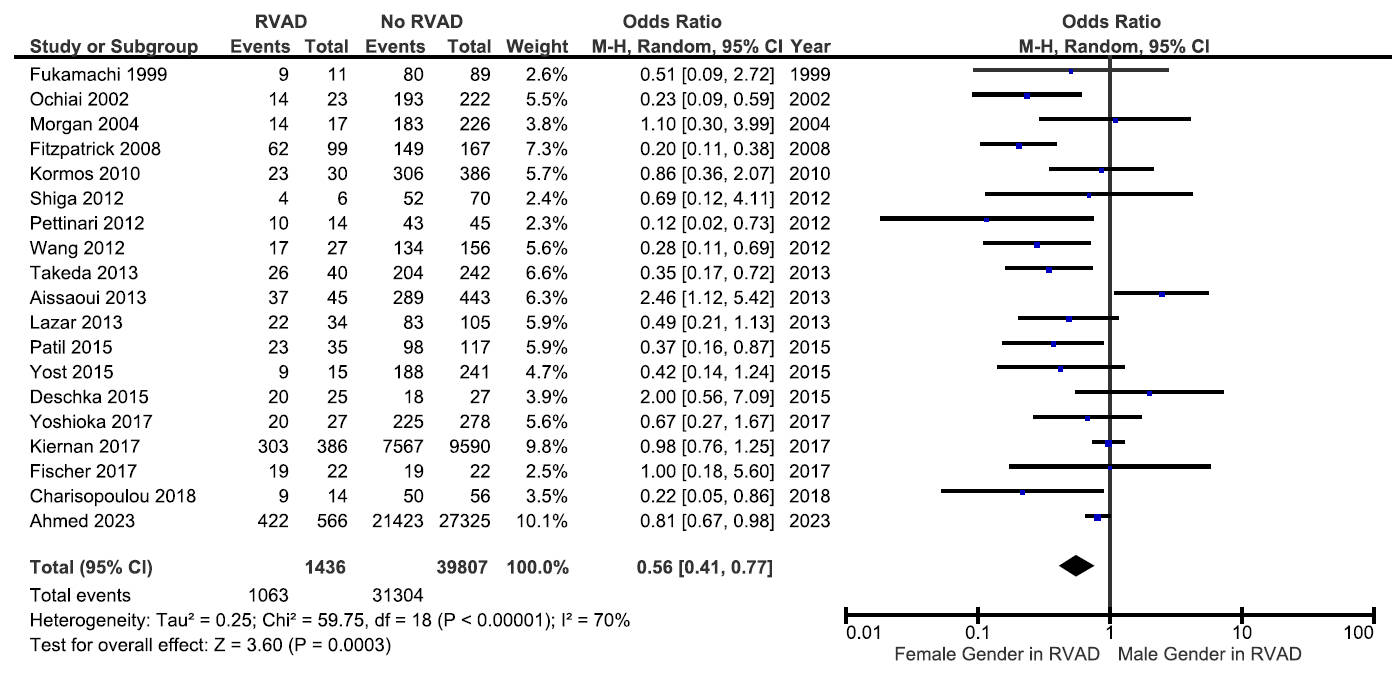 Fig. 3.
Fig. 3.
Forest plot of odds ratio for male gender as a predictor of postoperative RVAD requirement.
A history of previous cardiac surgery was associated with a higher risk of RVAD implantation (OR 1.78, 95% CI 1.18–2.70). Patients with a preoperative INTERMACS Profile 1 were more than twice as likely to need an RVAD (OR 2.27; 95% CI, 1.04 to 4.96). The need for aggressive preoperative support, including mechanical ventilation (OR 3.47; 95% CI, 2.44 to 4.94), hemofiltration (OR 3.17; 95% CI, 1.64 to 6.12), or IABP (OR 2.00; 95% CI, 1.54 to 2.59), were all strong predictors.
A distinct preoperative hemodynamic profile emerged for patients who
subsequently required an RVAD (Table 3). These patients exhibited significantly
higher central venous pressure (CVP) (MD 2.82 mmHg; 95% CI, 1.35 to 4.29; Fig. 4) and a higher CVP/PCWP ratio (MD 0.15; 95% CI, 0.11 to 0.18, Fig. 5). This
evidence of right-sided congestion was coupled with markers of impaired RV
output, including a lower cardiac index (MD –0.18 L/min/m2; 95% CI, –0.25
to –0.10), lower mean pulmonary artery pressure (PAP) (MD –3.22 mmHg; 95% CI,
–5.29 to –1.16), and a markedly lower right ventricular stroke work index
(RVSWI) (MD –9.94 mmHg
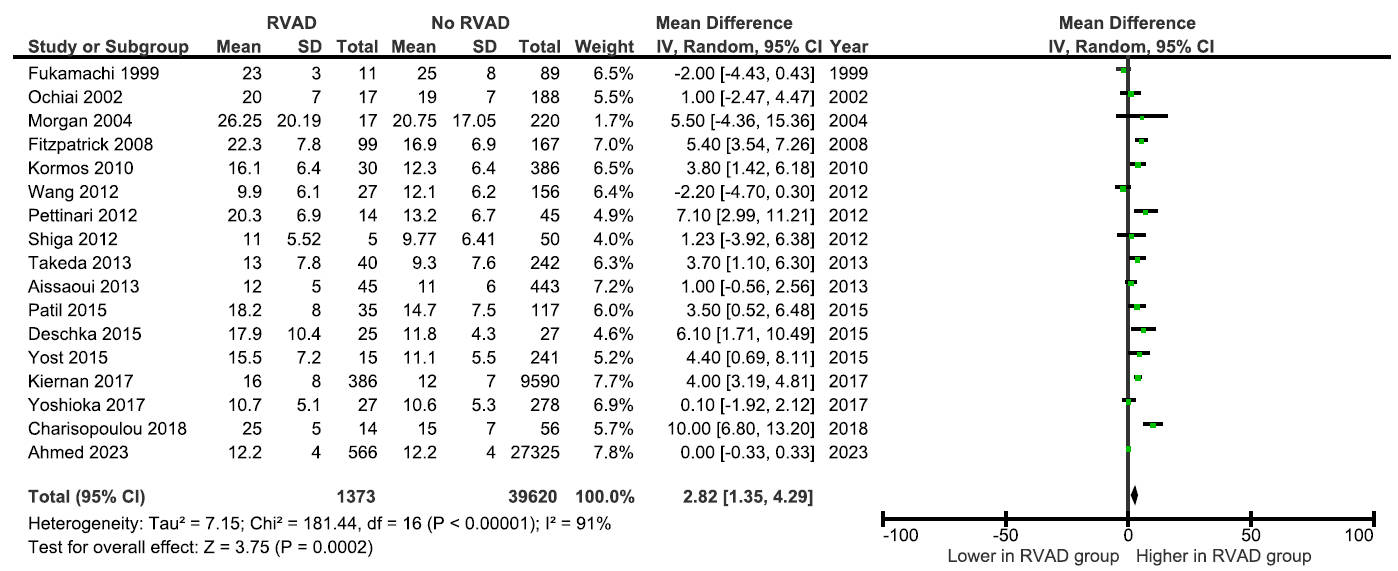 Fig. 4.
Fig. 4.
Forest plot of mean difference in central venous pressure between patients with and without postoperative RVAD requirement.
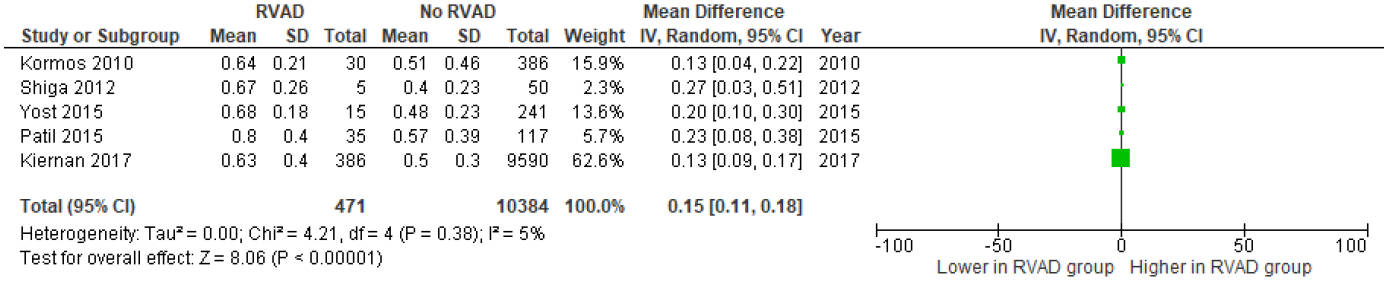 Fig. 5.
Fig. 5.
Forest plot of mean difference in preoperative CVP/PCWP ratio between patients with and without postoperative RVAD requirement.
Preoperative echocardiography provided key structural and functional clues to RV
vulnerability (Table 3). A critical functional marker was lower tricuspid annular
plane systolic excursion (TAPSE), which trended strongly towards predicting RVAD
need (MD –2.85 mm; 95% CI, –5.90 to 0.19). The presence of severe
(
RVAD patients had significantly higher preoperative bilirubin (MD +0.36 mg/dL,
95% CI 0.15–0.57) and aspartate aminotransferase (AST; MD +35.04 U/L, 95% CI
5.16–64.91), suggesting impaired hepatic function (Table 4). Coagulation profile
was abnormal with higher INR (MD +0.17, 95% CI 0.05–0.29). Haematologic
parameters also differed: haemoglobin (MD –0.53 g/dL, 95% CI –1.01 to –0.05),
platelet count (MD –13.32
To investigate the substantial heterogeneity observed for several predictors,
leave-one-out sensitivity analyses were performed (Supplementary Table 2). For
certain variables, the analysis identified influential studies; for example,
removing the study by Patil et al. [21] eliminated heterogeneity for
TAPSE (I2 from 88% to 0%), while removing the study by Deschka et
al. [20] substantially reduced heterogeneity for INTERMACS Profile 1 (I2
from 93% to 37%). In contrast, for other key predictors such as CVP and RVSWI,
the removal of any single study did not meaningfully reduce the high levels of
heterogeneity (I2
In the LVAD-era subgroup analysis (Supplementary Table 3), the direction of
associations between preoperative variables and RVAD requirement was largely
consistent across pulsatile-flow and continuous-flow cohorts. For demographic
predictors, younger age was associated with RVAD use in continuous-flow studies
(MD –3.64, 95% CI –5.41 to –1.86; p
In the sample-size subgroup analysis (Supplementary Table 4), the magnitude and
direction of associations between preoperative variables and RVAD requirement
were broadly consistent across small (
Several studies have attempted to integrate various predictors into risk scores. The performance of these scores has been variable (Table 5, Ref. [8, 13, 17, 21, 23, 29, 30]). For instance, Wang et al. [23] developed a decision tree model achieving a high area under the ROC curve (AUC) of 0.87, and Fitzpatrick et al. [29] created a score with 83% sensitivity and 80% specificity. However, external validation studies by Pettinari et al. [26] and Rivas-Lasarte et al. [8] found that many of these scores have limited discrimination in different patient cohorts, with AUCs often below 0.70. This highlights the challenge of creating a universally applicable predictive tool.
| First author (year) | Model/predictor | Validation status | Cohort characteristics | Area under curve (AUC) | Reported sensitivity | Reported specificity | Cutoff value |
| Wang et al. (2012) [23] | Decision Tree Model | Internal | Single-centre, retrospective (n = 183); mixed pulsatile/CF-LVADs | 0.87 | 85% | 83% | N/A |
| Fitzpatrick et al. (2008) [29] | Risk Score | Internal | Single-centre, retrospective (n = 266); mixed pulsatile/CF-LVADs | N/A | 83% | 80% | Score |
| Yost et al. (2016) [30] | MELD Score | Performance Assessment | Single-centre, retrospective (n = 256); CF-LVADs | 0.636 | N/A | N/A | |
| Patil et al. (2015) [21] | TAPSE | Performance Assessment | Single-centre, retrospective (n = 152); CF-LVADs | 0.85 | 84% | 75% | |
| Charisopoulou et al. (2019) [17] | RA Longitudinal Strain | Performance Assessment | Single-centre, retrospective (n = 70); CF-LVADs | 0.913 | 94% | 65% | |
| Kiernan et al. (2017) [13] | Multivariable Risk Model | Internal | Multi-centre registry (INTERMACS, n = 9976); CF-LVADs | 0.78 | N/A | N/A | N/A |
| Rivas-Lasarte et al. (2021) [8] | EUROMACS-RHF Score | External | Multi-centre, retrospective US cohort (n = 662); CF-LVADs | 0.64 | N/A | N/A | N/A |
AUC, Area Under the Curve; CF-LVAD, Continuous-Flow Left Ventricular Assist Device; EUROMACS-RHF, European Registry for Patients with Mechanical Circulatory Support-Right-Sided Heart Failure; INTERMACS, Interagency Registry for Mechanically Assisted Circulatory Support; MELD, Model for End-Stage Liver Disease; N/A, Not Available; RA, Right Atrial; TAPSE, Tricuspid Annular Plane Systolic Excursion.
Overall, the meta-analysis demonstrated that younger age, female sex, previous cardiac surgery, preoperative mechanical ventilation, IABP support, elevated CVP, reduced cardiac index, abnormal liver and coagulation parameters, hypoalbuminemia, anaemia, thrombocytopenia, elevated BNP, higher Michigan RV risk scores, and severe tricuspid regurgitation were consistent predictors of RVAD requirement following LVAD implantation.
RVF remains the Achilles’ heel of contemporary durable or temporary LVAD therapy and is strongly associated with peri-operative morbidity, prolonged resource use, and excess mortality [32]. In its severest form, RVF mandates RVAD support, a scenario repeatedly linked with worse early and late outcomes compared with isolated LVAD [6]. Patients requiring RVAD after LVAD implantation experience higher peri-operative mortality, more bleeding and neurological complications, and worse long-term survival compared with those managed with isolated LVAD support [33], underscoring the critical importance of early risk recognition and proactive management.
Several prior systematic reviews and meta-analyses have sought to identify predictors of right ventricular dysfunction after LVAD implantation. However, most have considered RVF as a broad and heterogeneous entity, often combining transient inotrope dependence, prolonged nitric oxide therapy, and RVAD implantation into a single outcome. Bellavia et al. [34] synthesized data from 36 studies and identified haemodynamic and echocardiographic predictors of RVF, including elevated central venous pressure, lower right ventricular stroke work index, and increased RV/LV diameter ratio, but RVAD implantation was not assessed separately as a hard endpoint. Frankfurter et al. [7] instead focused on the performance of dedicated RVF risk scores, such as the Michigan, CRITT, and EUROMACS-RHF models, and demonstrated their limited discriminatory ability when externally validated, with most models showing modest predictive value only. Other reviews have concentrated on specific preoperative domains. For example, Chai et al. [35] examined echocardiographic markers, reporting that impaired RV function and significant tricuspid regurgitation were associated with post-LVAD RVF.
Importantly, none of these prior studies have synthesized predictors specifically for RVAD implantation, the most clinically decisive manifestation of RVF. This distinction matters as patients requiring RVAD after LVAD implantation have a three- to fourfold higher early mortality and significantly worse long-term outcomes compared with those supported with isolated LVAD [36]. By treating RVAD implantation as a discrete endpoint, our study moves beyond the heterogeneity of prior RVF definitions and provides a more actionable evidence base for surgical decision-making.
The present meta-analysis therefore builds on the existing literature by systematically evaluating preoperative predictors across all major domains, clinical and demographic factors, haemodynamic, echocardiography, and laboratory parameters, and quantifying their association with the subsequent need for RVAD support. This integrated approach allows for a more holistic risk profile and provides cardiac surgeons with practical, clinically relevant predictors that can guide both preoperative optimization and intraoperative strategy. Synthesizing the evidence from 20 studies, this comprehensive meta-analysis identified a robust profile of preoperative risk factors for RVAD requirement after LVAD implantation. The results clearly indicate that the need for RVAD is driven by a syndrome of global illness severity, characterized by advanced clinical acuity, profound haemodynamic compromise, and significant end-organ dysfunction, rather than any single isolated parameter.
Our analysis reaffirms the central role of haemodynamic assessment in risk stratification. An elevated CVP is a consistent and powerful predictor, reflecting the degree of systemic venous congestion that the RV must overcome. However, isolated CVP can be misleading. Indices that provide broader context, such as the CVP/PCWP ratio, which reflects the balance of right- versus left-sided filling pressures, and the RVSWI, which quantifies the effective work of the RV, were also highly significant in our analysis. This emphasizes that a dynamic assessment of RV function relative to its loading conditions is more informative than static pressure measurements alone.
Recent evidence further supports the prognostic importance of the right atrial
pressure to pulmonary capillary wedge pressure (RAP/PCWP) ratio in the LVAD
population. A 2025 study by Keskin et al. [37] demonstrated that an
elevated RAP/PCWP ratio strongly predicts in-hospital mortality following LVAD
implantation. In that cohort, patients with a high RAP/PCWP ratio (
A key theme emerging from this analysis is the paramount importance of systemic and end-organ function. The strong association with high-acuity INTERMACS profiles and the need for preoperative life support (mechanical ventilation, IABP, ECMO) underscores that patients in cardiogenic shock are at the highest risk. This is further supported by the laboratory findings. Elevated bilirubin and INR, and low albumin are not just markers of hepatic dysfunction; they are powerful surrogates for the systemic effects of chronic right-sided congestion and low cardiac output. These findings validate the components of widely used prognostic scores like the MELD score, which Yost et al. [30] found to be predictive of RVF.
The challenge of risk prediction is highlighted by the inconsistent performance of dedicated RVF risk scores. While scores developed in one cohort may perform well internally, studies by Pettinari et al. [26] and Rivas-Lasarte et al. [8] demonstrate that their predictive power often diminishes upon external validation. Rivas-Lasarte et al. [8] found the EUROMACS-RHF score had an AUC of only 0.64 in a large external cohort, concluding its clinical utility remains to be determined. This suggests that static scoring systems may fail to capture the complex, dynamic pathophysiology of individual patients and underscores the need for a more integrated, clinician-driven assessment.
Previous systematic reviews have largely focused on RVF as a broader construct. Bellavia et al. [34] identified elevated CVP, impaired RVSWI, and RV dilation as predictors of RVF, but did not differentiate between mild, moderate, or severe failure, nor did they isolate RVAD implantation as an endpoint. Frankfurter et al. [7] reviewed existing risk scores (e.g., Michigan, CRITT, EUROMACS-RHF) and demonstrated their limited discriminatory ability when externally validated. More recently, Rodenas-Alesina et al. [38] and Wang et al. [39] provided narrative reviews of RVF incidence, pathophysiology, and prevention strategies, but again encompassed heterogeneous outcomes. Importantly, Reid et al. [36] specifically showed that patients requiring RVAD after LVAD implantation had more than a threefold higher 30-day mortality (31.9% vs 6.7%) compared with isolated LVAD recipients, reinforcing the prognostic weight of this endpoint. Our study advances the field by being the first meta-analysis to quantitatively synthesize preoperative predictors of RVAD implantation as a distinct and clinically decisive outcome.
The findings of this meta-analysis have direct clinical implications. Patient selection for LVAD therapy must involve a comprehensive, multi-parameter risk assessment. A patient presenting with multiple risk factors identified in this analysis, such as an INTERMACS Profile 1, a history of prior cardiac surgery, need for preoperative IABP, an elevated CVP, low TAPSE, and elevated bilirubin, should be considered at very high risk for requiring postoperative RVAD support. In these patients, the clinical team should consider strategies to mitigate risk, such as intensive preoperative medical optimization to decongest the patient and improve end-organ function. Furthermore, there should be a low threshold for a planned biventricular support strategy, either with a temporary RVAD or consideration of a primary BiVAD or total artificial heart. As shown by Ahmed et al. [14], a sequential, rescue RVAD implantation after a period of deterioration is associated with significantly worse survival and higher adverse event rates compared to a concurrent, planned BiVAD implantation. Therefore, anticipating the need for RV support and acting proactively is critical.
This meta-analysis is subject to several limitations inherent to the source literature. The majority of the included studies were retrospective, and significant heterogeneity existed in patient populations, definitions of RVF, and eras of LVAD technology. The decision to implant an RVAD is also subject to institutional practice patterns, which introduces variability. While we focused on the hard endpoint of RVAD implantation, this does not capture the full spectrum of RVF, which includes patients managed with prolonged inotropes who may also have poor outcomes.
Furthermore, a key limitation of this meta-analysis is the probable inclusion of overlapping patient cohorts from large, registry-based studies. Specifically, major studies such as Kiernan et al. [13] and Ahmed et al. [14] both draw from the INTERMACS registry during overlapping time periods. This can potentially skew the summary effect estimates and lead to an overestimation of the precision of our findings. While it was not possible to identify and exclude overlapping patients from the primary data, this inherent limitation should be considered when interpreting the results of this analysis.
Finally, the analysis is limited by the variables reported in the primary studies, and the potential for publication bias cannot be excluded.
In conclusion, this systematic review and meta-analysis identifies a clear and consistent profile of preoperative risk for RVAD requirement after LVAD implantation. This profile is characterized by high clinical acuity, the need for preoperative mechanical support, haemodynamic evidence of severe RV dysfunction, and laboratory signs of systemic congestion and end-organ injury. No single variable is sufficient for risk prediction; rather, a comprehensive, multi-parameter assessment is essential to guide patient selection, facilitate informed decision-making, and plan perioperative strategies to improve the outcomes of this high-risk patient population.
The data are available from the corresponding author after reasonable demand.
MC conceptualised the study and drafted the manuscript. MC, AD, CC, IZ, AS, AP, EK, MO, MMV, NM, and HSA performed data acquisition, data extraction, and data analysis. SGR contributed to the study conceptualisation and provided senior review. AS, AP, EK, MO, MMV, NM, HSA, AD, CC, IZ, and SGR contributed to critical revision of the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work in accordance with the ICMJE authorship criteria.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Shahzad G. Raja is serving as one of the Editorial Board members of this journal. We declare that Shahzad G. Raja had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Tadahisa Sugiura.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF50915.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





