1 Department of Pediatric Cardiac Surgery, Cho Ray Hospital, 72723 Ho Chi Minh City, Vietnam
2 Department of Surgery, College of Medicine and Pharmacy, Tra Vinh University, 87118 Vinh Long Province, Vietnam
3 Department of Thoracic and Vascular, University Medical Center Ho Chi Minh City, University of Medicine and Pharmacy at Ho Chi Minh City, 72723 Ho Chi Minh City, Vietnam
4 Department of Health Management, Faculty of Public Health, University of Medicine and Pharmacy at Ho Chi Minh City, 72723 Ho Chi Minh City, Vietnam
Abstract
Fenestration is a well-established modification of the Fontan procedure intended to reduce systemic venous pressure and limit early postoperative complications. However, the optimal fenestration diameter that balances clinical benefits with systemic oxygenation remains uncertain. This study aimed to evaluate the association between fenestration size and early outcomes following extracardiac Fontan surgery.
We retrospectively analyzed pediatric patients who underwent fenestrated extracardiac Fontan surgery at Cho Ray Hospital between January 2020 and March 2025. Fenestration diameters ranged from 4 to 8 mm. Postoperative complications were categorized as pulmonary, cardiovascular, infectious, renal, or mortality events. Continuous outcomes included postoperative pleural drainage duration, intensive care unit stay, hospital stay, and perioperative change in peripheral arterial oxygen saturation (SpO2). Correlation analyses were employed to assess the associations between fenestration size and outcomes.
Twenty patients (median age: 5 years; 70% male) were included. Pulmonary complications were most common (85%), with pneumonia and pleural effusion observed in 65% of cases. Fenestration size was not associated with overall complication incidence; however, cardiovascular complications differed between groups (p = 0.045), without a linear trend (p for trend = 0.271). Oxygenation improved significantly perioperatively (median SpO2 increased from 76% to 86%, mean change: 10.6%; p = 0.001). Fenestration size correlated inversely with perioperative SpO2 change (ρ = –0.75; p < 0.001), while larger fenestrations correlated with shorter pleural drainage (ρ = –0.71; p = 0.001) and reduced hospital stay (ρ = –0.59, p = 0.012).
Fenestration significantly impacts early outcomes after a Fontan operation. Larger fenestrations promoted faster recovery, with shorter pleural drainage and hospital stays, but conferred a smaller oxygenation benefit. Individualized fenestration sizing may help optimize early outcomes in pediatric Fontan patients.
Keywords
- Fontan surgery
- fenestration size
- pleural drainage
- hospital stay
- oxygen saturation
Managing early postoperative morbidity following a Fontan operation remains a significant clinical challenge. This surgery is the definitive palliation for single-ventricle physiology, yet its unique circulatory arrangement inherently leads to elevated systemic venous pressures [1, 2], predisposing patients to severe complications such as prolonged pleural effusions and protein-losing enteropathy [2, 3, 4]. To mitigate these adverse outcomes, surgical fenestration, a controlled communication between the Fontan circuit and the atrium, is widely employed as a decompression strategy [1]. While surgical fenestration has become standard practice, the optimal fenestration diameter remains poorly defined, and its net clinical benefit is a subject of ongoing controversy [5, 6, 7].
The existing literature presents inconsistent findings [7, 8]. While some evidence supports routine fenestration to reduce postoperative morbidity [5, 6, 9], other reports suggest that its advantages are confined to high-risk cohorts, prompting some centres to move away from its routine use [9, 10]. This clinical discrepancy is further highlighted by computational and translational models [11, 12, 13], which indicate that only a narrow range of fenestration sizes may optimise systemic oxygen delivery without causing excessive arterial desaturation, a critical haemodynamic trade-off [11]. In addition, recent evidence has suggested that preoperative NT-proBNP may serve as a useful biomarker for risk stratification when deciding on fenestration strategy [14].
For example, some computational studies suggest that while fenestration can relieve venous pressure, this comes at the expense of reduced oxygen saturation, even with commonly used fenestration sizes around 4 mm [1, 11]. These analyses suggest that the physiological trade-off becomes increasingly pronounced as fenestration size enlarges, with the greatest haemodynamic impact likely occurring at diameters exceeding 4 mm. Nevertheless, this size-dependent relationship remains insufficiently defined in clinical settings, emphasising the need for empirical studies to clarify the optimal balance between decompression and oxygenation.
Given this uncertainty, a significant knowledge gap persists in defining the precise relationship between fenestration dimensions and key clinical outcomes. The aim of this study was to rigorously evaluate the association between fenestration size and early postoperative outcomes following Fontan surgery, including major complications, changes in peripheral oxygen saturation and time-related recovery metrics.
This retrospective study was conducted at Cho Ray Hospital and included patients who underwent a Fontan operation between January 2020 and March 2025. Data were comprehensively extracted from the institution’s electronic health records and operative logs. This report has been prepared in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement [15].
The study cohort comprised all patients diagnosed with functionally single-ventricle physiology who underwent an extracardiac fenestrated Fontan surgery within the aforementioned study timeframe. Inclusion criteria strictly mandated a complete preoperative evaluation, encompassing transthoracic echocardiography and cardiac catheterisation data. Patients were systematically excluded from the analysis if their clinical records were incomplete, if they presented with substantial missing data or if they had undergone alternative Fontan modifications.
Patient management during the perioperative period followed a standardised institutional protocol. Preoperatively, all patients underwent comprehensive transthoracic echocardiography to assess ventricular function and atrioventricular valve competence, as well as cardiac catheterisation to measure key haemodynamic parameters, including mean pulmonary artery pressure and pulmonary vascular resistance.
Intraoperatively, all patients underwent a standardised approach that included a balanced anaesthetic technique to preserve cardiac function and uniform cardiopulmonary bypass protocols with an emphasis on minimising bypass and cross-clamp times. The Fontan operation was performed using an extracardiac conduit in all cases, with the fenestration surgically created through a side-to-side anastomosis between the conduit and the right atrial free wall. Two techniques were employed: a direct window for 4-mm fenestrations and a polytetrafluoroethylene tube graft for fenestrations measuring 5–8 mm. Final fenestration size was determined intraoperatively by the surgical team, based on individualised haemodynamic assessments that included pulmonary artery pressure, pulmonary vascular resistance, body weight and overall clinical risk. Surgical and imaging features of fenestrated extracardiac Fontan procedures are illustrated in Fig. 1.
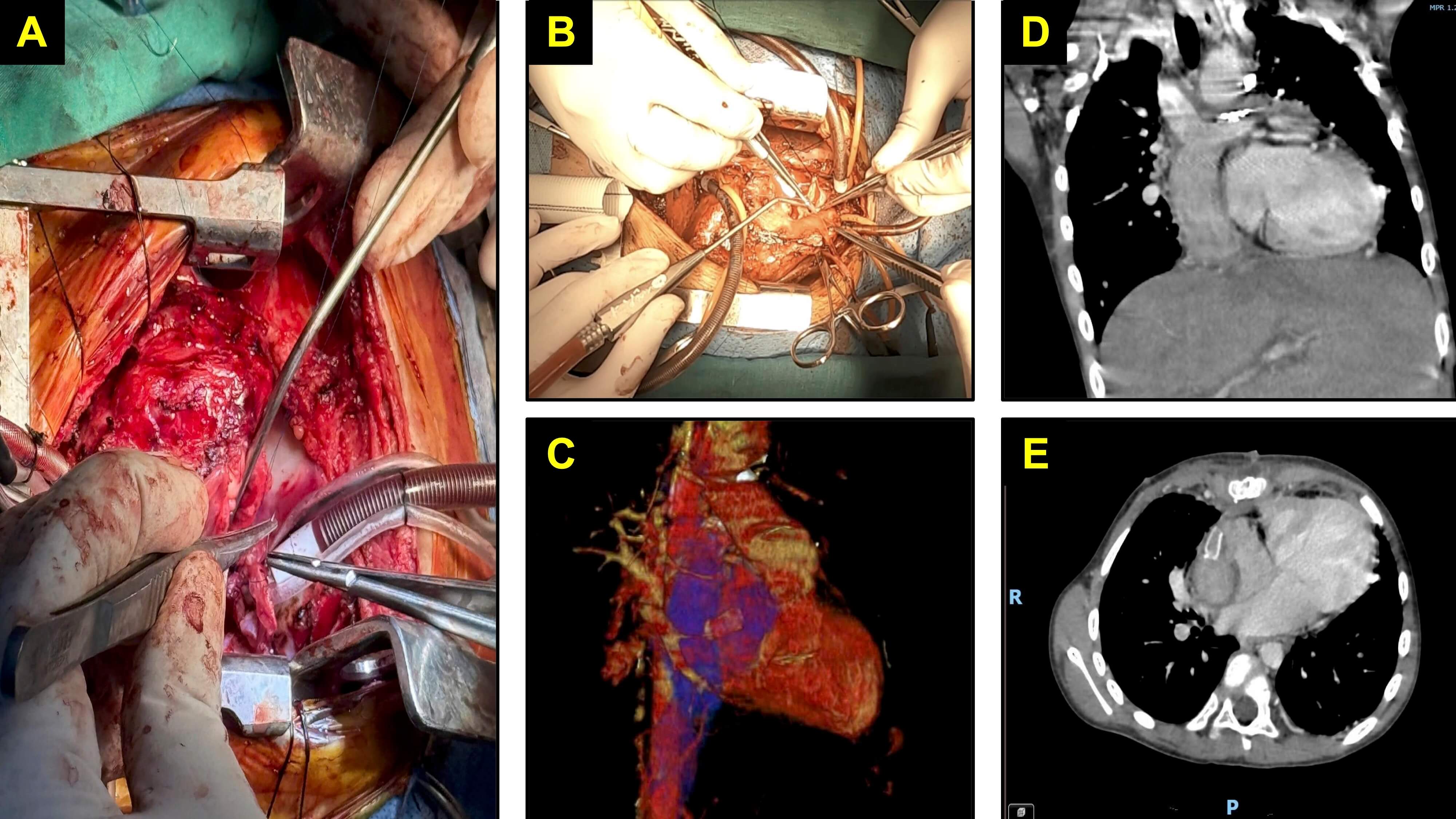 Fig. 1.
Fig. 1.
Surgical and imaging features of fenestrated extracardiac Fontan procedures in paediatric patients. (A) Intraoperative view of the inferior vena cava anastomosis opening. (B) Intraoperative view of the superior vena cava anastomosis opening. (C) Three-dimensional computed tomography (CT) reconstruction of the extracardiac Fontan conduit. (D) Coronal CT image demonstrating the Fontan conduit in situ. (E) Axial CT image of the Fontan conduit.
Postoperatively, patients were managed in the intensive care unit (ICU) with continuous tracking of haemodynamics and oxygen saturation. Intraoperatively placed chest tubes were used to manage pleural effusions, with output recorded hourly. Chest tubes were removed when drainage was less than 2 mL/kg/day, and prolonged pleural effusion was defined as the need for chest drainage for more than 7 days. Standard care also included prophylactic anticoagulation and close observation for signs of low cardiac output syndrome and other early postoperative morbidities.
The incidence of all early postoperative complications was systematically
recorded. For clarity, complications were grouped into composite categories.
Pulmonary complications included pneumonia, pleural effusion, prolonged
mechanical ventilation, pneumothorax and chylothorax. Chylothorax was defined as
pleural fluid triglyceride
Peripheral arterial oxygen saturation (SpO2) was measured via pulse oximetry at two time points: preoperatively (baseline) and at the time of hospital discharge, when the patient was stable and breathing room air without respiratory or haemodynamic compromise. The correlation between fenestration size and both the change in saturation and the final postoperative saturation level was also assessed. Recovery duration was quantified by three variables: the total duration of pleural drainage, the ICU stay and the postoperative hospital length of stay (LOS).
All analyses were conducted using R software (version 4.3.2; R Foundation for
Statistical Computing, Vienna, Austria). Given the small sample size and the
non-normal distribution of several variables, nonparametric methods were applied.
Statistical significance was defined as a two-tailed p-value
Categorical variables were summarised as frequencies and percentages. Postoperative complications were evaluated as both individual outcomes and grouped or composite events. Comparisons between fenestration size groups were performed using Fisher’s exact test, while trends in complication rates across ordered fenestration sizes were assessed with the Cochran–Armitage test for trend.
Continuous variables were expressed as means with standard deviations or medians
with interquartile ranges (IQR). Intra-group changes were examined using
one-sample tests and paired analyses with the Wilcoxon signed-rank test.
Associations between fenestration size and clinical outcomes were assessed using
Spearman’s rank correlation coefficients (
A total of 20 patients were included in this study, and their demographics and baseline data are presented in Table 1. The median age and weight at the time of surgery were 5 years [IQR 3–6] and 14.5 kg [IQR 13.5–18.25]. The cohort consisted of 6 (30%) female patients and 14 (70%) male patients. The median preoperative oxygen saturation was 76 [IQR 75–78]. The majority of patients had undergone at least one previous surgery (90%), and 12 patients (60%) were receiving treatment for pulmonary hypertension. The most common type of single ventricle was tricuspid atresia (35%), followed by unbalanced atrioventricular septal defect (20%) and heterotaxy syndrome (20%). Most patients (80%) presented with trivial to mild systemic atrioventricular valve regurgitation, and the median systemic ventricular ejection fraction was 70 [IQR 64–74].
| Characteristics | Values | ||
| Demographics | |||
| Age, years | 5 [3–6] | ||
| Female, n (%) | 6 (30%) | ||
| Male, n (%) | 14 (70%) | ||
| Weight, kilograms | 14.5 [13.5–18.3] | ||
| Number of previous surgeries, n (%) | |||
| 0 surgeries | 2 (10%) | ||
| 1 surgery | 11 (55%) | ||
| 2 surgeries | 7 (35%) | ||
| Pulmonary hypertension treatment, n (%) | 12 (60%) | ||
| Glenn with additional pulmonary blood flow, n (%) | 4 (20%) | ||
| Cardiac morphology | |||
| Type of single ventricle, n (%) | |||
| Tricuspid atresia | 7 (35%) | ||
| Unbalanced atrioventricular septal defect | 4 (20%) | ||
| Heterotaxy syndrome | 4 (20%) | ||
| Double inlet left ventricle | 2 (10%) | ||
| Other types | 3 (15%) | ||
| Systemic atrioventricular valve function, n (%) | |||
| Trivial or mild regurgitation | 16 (80%) | ||
| Moderate to severe regurgitation | 4 (20%) | ||
| Systemic ventricular ejection fraction, % | 70 [64–74] | ||
| Haemodynamic status | |||
| Superior vena cava pressure, mmHg | 14.0 [13.0–15.5] | ||
| Systolic pulmonary artery pressure, mmHg | 15.5 [14.0–19.8] | ||
| Diastolic pulmonary artery pressure, mmHg | 12.5 [10.0–13.8] | ||
| Mean pulmonary artery pressure, mmHg | 14.0 [12.0–14.3] | ||
| Indexed pulmonary vascular resistance, WU.m2 | 1.6 [1.1–2.0] | ||
| Systemic ventricular end-diastolic pressure, mmHg | 12.0 [9.8–15.0] | ||
| Oxygenation status | |||
| Preoperative peripheral oxygen saturation, % | 76 [75–78] | ||
Annotation: Data are presented as mean
Intervention characteristics are shown in Table 2. The median cardiopulmonary bypass time was 198 minutes [IQR 180–248]. Conduit sizes of 18 and 20 mm were used in 65% and 35% of patients, respectively. Pulmonary artery angioplasty was performed in 55% and tricuspid valve repair in 10% of patients. Fenestration diameters were 4 mm in 35%, 5 mm in 25%, 6 mm in 25% and 7 mm in 15% of patients.
| Characteristics | Values | |
| Cardiopulmonary bypass duration, minutes | 198 [180–248] | |
| Fontan conduit size, n (%) | ||
| 18 mm | 13 (65%) | |
| 20 mm | 7 (35%) | |
| Pulmonary artery angioplasty, n (%) | 11 (55%) | |
| Tricuspid valve repair, n (%) | 2 (10%) | |
| Type of conduit, n (%) | ||
| Atriopulmonary window | 7 (35%) | |
| Extracardiac tube | 13 (65%) | |
| Fenestration size, n (%) | ||
| 4 mm | 7 (35%) | |
| 5 mm | 5 (25%) | |
| 6 mm | 5 (25%) | |
| 8 mm | 3 (15%) | |
Fig. 2 illustrates the distribution of early postoperative complications in this cohort. Pulmonary (85%) and cardiovascular (40%) events were the most common composite complication. The leading specific complications included pneumonia (45%), pleural effusion (45%), heart failure (30%) and sepsis (25%). Co-occurrence analysis revealed that pneumonia and pleural effusion frequently appeared together (15%). There were three deaths: one due to refractory heart failure progressing to multiorgan failure despite ECMO support, one from septic shock and one from intracranial haemorrhage.
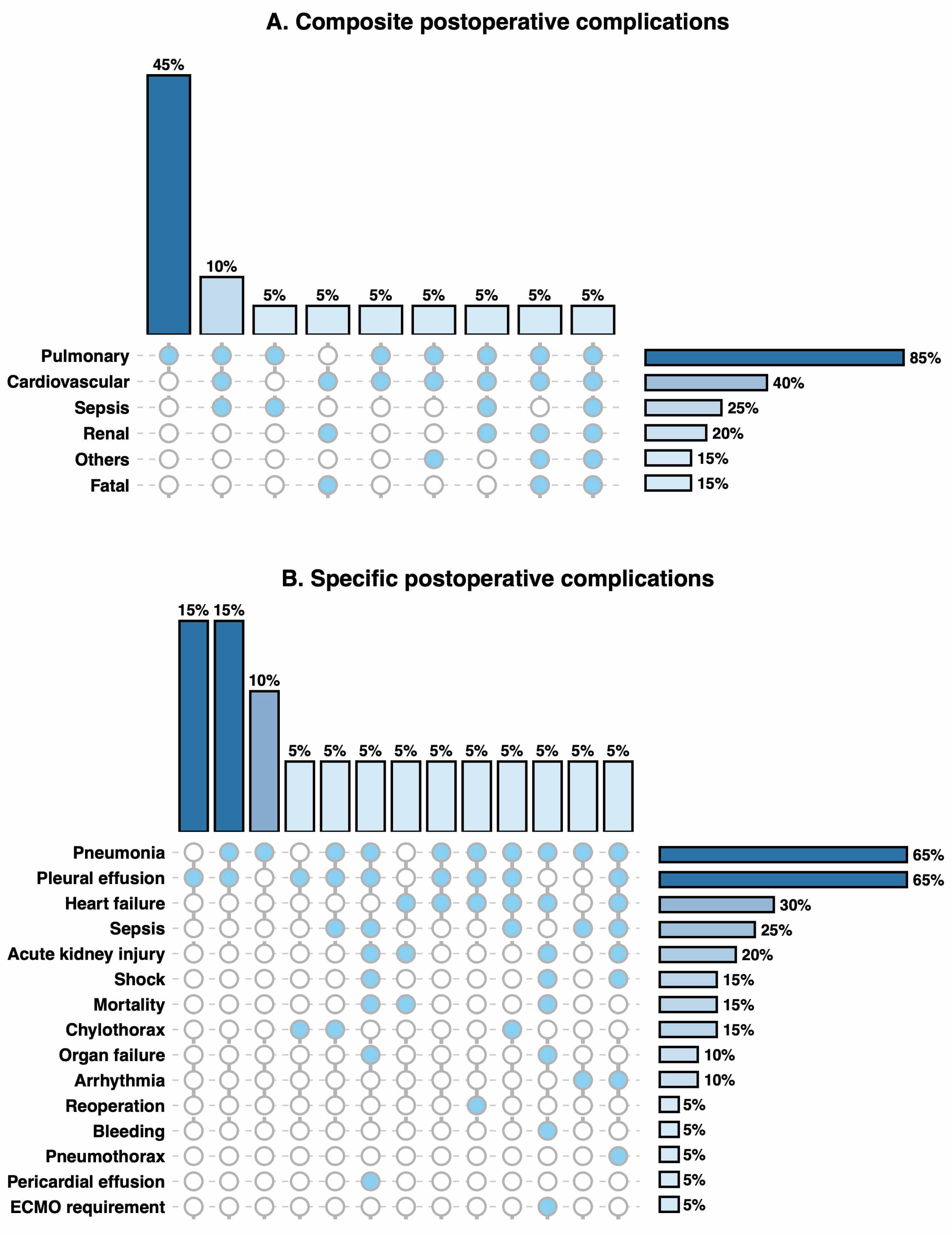 Fig. 2.
Fig. 2.
Postoperative complications and co-occurrence patterns. (A) Composite postoperative complications. (B) Specific postoperative complications. Filled circles indicate the presence of a complication within a given co-occurrence pattern; empty circles indicate absence. Each vertical column represents a distinct combination of complications, with bars above showing the proportion of patients with that combination. Horizontal bars show the overall incidence of each complication in the study population. Percentages are calculated using the total study population as the denominator (n = 20). Annotation: Three deaths occurred: severe heart failure with multiorgan failure requiring ECMO support, septic shock, and intracranial haemorrhage. ECMO, extracorporeal membrane oxygenation.
As shown in Fig. 3, fenestration size was not significantly associated with most categories of postoperative complications. The only exception was a statistical association with cardiovascular complications (p = 0.045); however, this did not demonstrate a consistent linear trend across fenestration sizes (p for trend = 0.271).
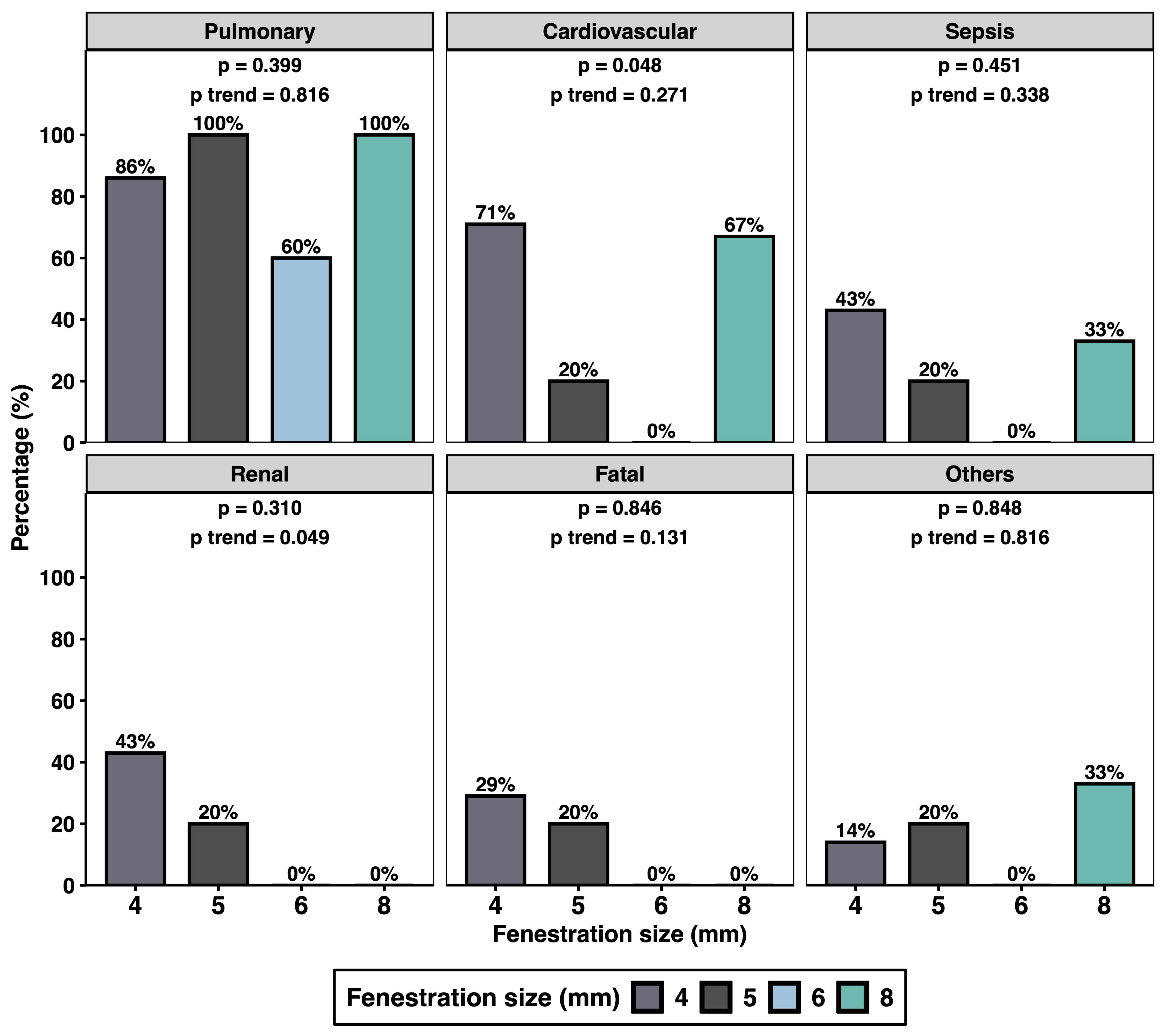 Fig. 3.
Fig. 3.
Early postoperative complications associated with fenestration size. Annotations: Other complications include bleeding, extracorporeal membrane oxygenation or re-operation requirements. p-values were derived from Fisher’s exact test for comparing complication frequencies across the fenestration size groups. The Cochran–Armitage test was applied to evaluate the presence of a linear trend in complication rates across increasing fenestration sizes.
A significant improvement in oxygenation was observed postoperatively, as shown
in Fig. 4A. The mean increase was 10.6% (p
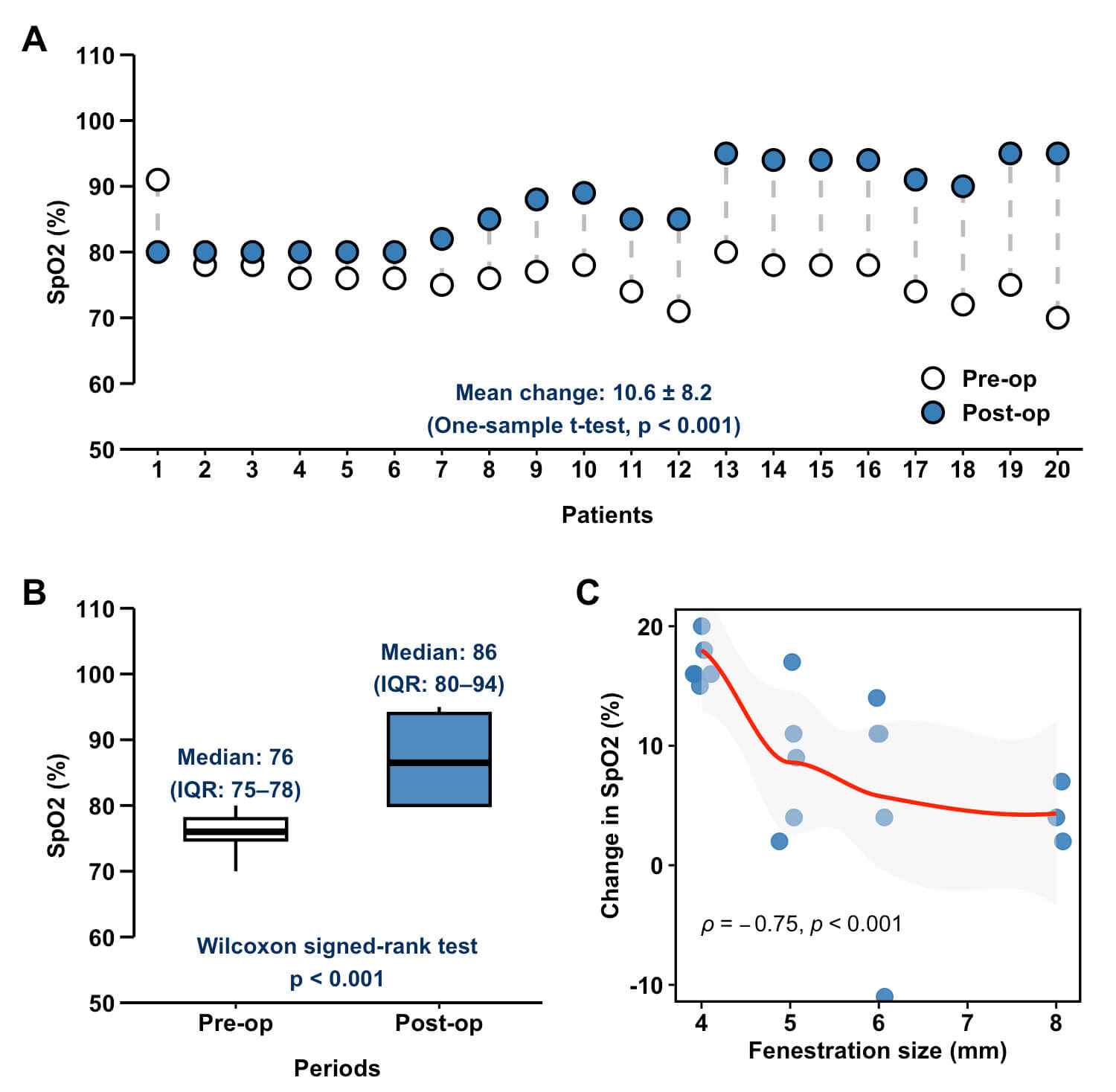 Fig. 4.
Fig. 4.
Relationship between fenestration size and postoperative oxygen saturation outcomes. (A) Individual changes in peripheral arterial oxygen saturation (SpO2) for each patient. (B) Comparison of preoperative and postoperative SpO2 values. (C) Correlation between fenestration size and perioperative SpO2. Pre-op, preoperative; Post-op, postoperative.
Early postoperative outcomes are summarised in Table 3, and correlations are
illustrated in Fig. 5. The median duration of mechanical ventilation was 16 hours
(IQR 12–73), pleural drainage 11 days (IQR 6–24), ICU stay 5 days (IQR 3–6)
and postoperative LOS 21 days (IQR 15–30). Larger fenestration size was
significantly associated with more favorable recovery outcomes, showing a strong
inverse correlation with pleural drainage duration (
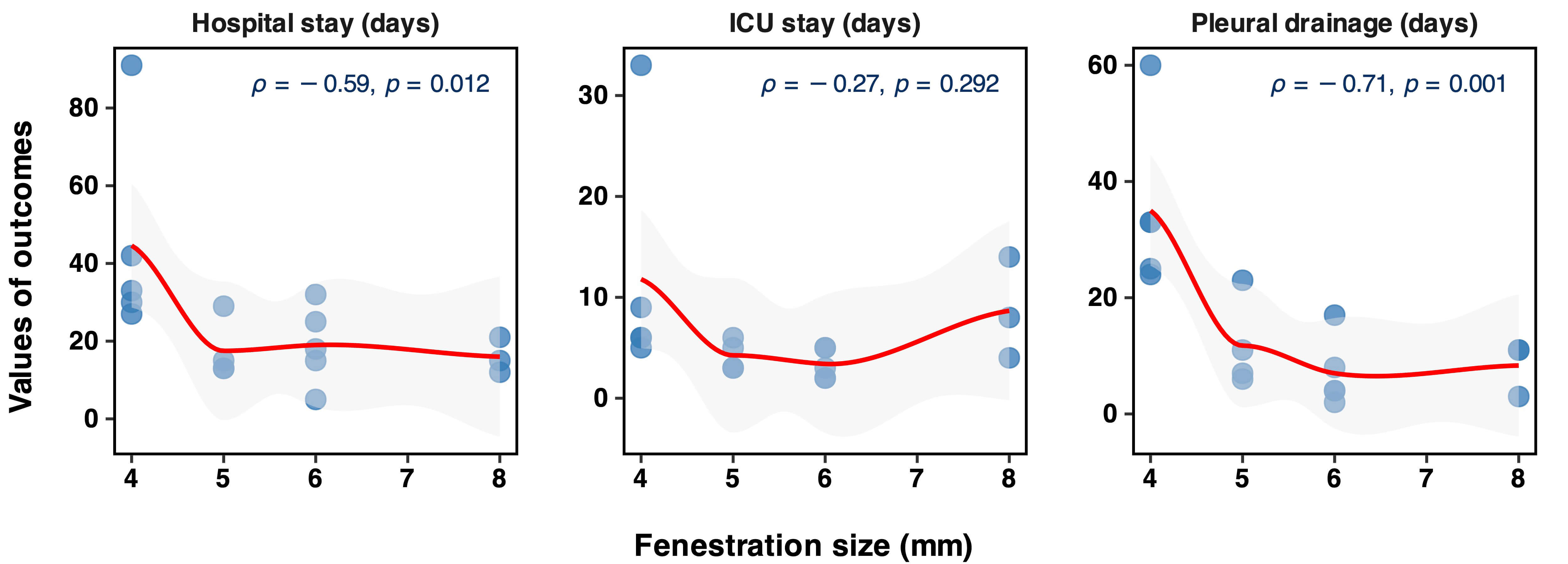 Fig. 5.
Fig. 5.
Correlation between fenestration size and recovery durations in patients undergoing Fontan surgeries. ICU, intensive care unit.
| Outcomes | Values |
| Peripheral oxygen saturation, % | 87 [80–94] |
| Central venous pressure, mmHg | 16.0 [15.5–18.0] |
| Mechanical ventilation time, hours | 16 [12–73] |
| Pleural drainage duration, days | 11 [6–24] |
| Intensive care unit stay, days | 5 [3–6] |
| Hospital stay, days | 21 [15–30] |
The objective of this study was to evaluate the relationship between fenestration size and early postoperative outcomes in paediatric patients undergoing Fontan surgery, a relatively uncommon procedure, over a 5-year period at a specialised cardiac centre in southern Vietnam. In this cohort, fenestration diameters ranged from 4 to 8 mm and were not clearly associated with the overall incidence of postoperative complications. Smaller fenestrations were linked to higher postoperative oxygen saturation, whereas larger fenestrations were associated with more favourable recovery, including shorter pleural drainage and reduced hospital stay. These findings present the double-edged nature of fenestration strategy in children, where the gain in oxygenation must be weighed against the advantage of a smoother postoperative recovery. Our findings, while consistent with established physiology, provide clinical evidence in an underrepresented Southeast Asian population. Importantly, they underscore the need for a risk-stratified, individualised approach to fenestration sizing. In patients with borderline oxygen saturation and low risk of prolonged effusions, smaller fenestrations may be preferable to avoid severe hypoxemia. Conversely, in those with elevated systemic venous pressures or high risk of effusions, larger fenestrations may facilitate smoother early recovery, with a subsequent plan for transcatheter closure once haemodynamic stability is achieved. This framework may help guide practical decision-making beyond the traditional ‘one-size-fits-all’ strategy.
Our analysis did not demonstrate a dose-dependent relationship between fenestration diameter and the overall incidence of postoperative complications. This result is consistent with previous meta-analyses by Li et al. [7] and Bouhout et al. [8], which also found no significant difference in Fontan failure or mortality between fenestrated and non-fenestrated patients. The lack of association may reflect the multifactorial nature of major complications following Fontan surgery, where factors such as ventricular function, pulmonary vascular resistance and underlying cardiac anatomy likely play a greater role than fenestration size [16]. Although fenestration is a critical element of the modern Fontan operation, its diameter alone does not appear to be the key determinant of postoperative morbidity.
This study demonstrated a strong inverse correlation between fenestration diameter and perioperative oxygen saturation, an observation consistent with established physiology. Larger fenestrations permit greater right-to-left shunting, resulting in lower systemic SpO2 [11], a relationship supported by both clinical and computational studies [12, 13]. This is consistent with transcatheter closure studies, where Greenleaf et al. [17] reported a 7.9% increase in mean SpO2, compared with a 10.6% increase in our cohort. Likewise, Devanagondi and Leonard [18] observed an improvement from 87% to 95%, whereas we noted a rise from a median of 76%–86%. Although persistent cyanosis is associated with long-term risks such as collateral formation and thromboembolic events [19, 20, 21], studies indicate that patients with oxygen saturation levels of 85%–90%, similar to those observed in our cohort, may still achieve comparable exercise capacity to non-fenestrated patients [17, 22]. Collectively, these findings confirm that fenestration size strongly influences systemic oxygenation.
Our data demonstrated a significant inverse correlation between fenestration diameter and the durations of pleural drainage and hospitalisation. This observation is in strong agreement with prior literature that consistently highlights the decompressive benefits of fenestration. Multiple meta-analyses and comparative studies have shown that fenestrated patients experience reduced ICU stay, overall LOS and chest tube drainage duration [7, 9, 23]. Corno et al. [5] further reported that a prospective randomised trial confirmed shorter ICU stay and LOS in fenestrated patients, while Fu et al. [3] identified non-fenestration as an independent predictor of prolonged pleural drainage, with their cohort showing a median postoperative LOS of 14 days. The underlying mechanism is likely related to the role of the fenestration as a functional ‘pop-off valve’ that relieves systemic venous pressure and reduces effusion formation [6]. Larger fenestrations provide more effective decompression, thereby accelerating recovery and shortening both drainage duration and hospitalisation. Therefore, these findings also suggest that a larger fenestration may be a rational strategy for patients at increased risk of persistent effusions.
The clinical implications of our findings are clear: a ‘one-size-fits-all’ approach to fenestration sizing is suboptimal [5, 11]. The decision requires a careful, individualised balance between the decompressive benefits, recovery outcomes and hypoxemia risks. We recommend a risk-stratified approach to be incorporated into clinical protocols. For patients with borderline preoperative oxygenation or at low risk of effusions, a smaller fenestration may be prudent to avoid severe hypoxemia [10]. Conversely, in patients with elevated systemic venous pressures or other risk factors for prolonged effusions, a larger fenestration confers a net advantage by facilitating a smoother early recovery [5, 7]. We further recommend that clinicians view the fenestration as a temporary, two-stage intervention by leveraging the early benefits of a larger fenestration and having a clear plan for subsequent transcatheter closure to durably improve oxygenation once the patient is haemodynamically stable [24].
This study examines a rare clinical population, with only 20 fenestrated Fontan procedures performed over a 5-year period at a tertiary centre in Vietnam. The limited number of patients reflects both the uncommon nature of the Fontan operation in our country and the challenges of conducting research in a resource-constrained setting. Consequently, the small sample size reduces statistical power and limits the generalisability of our findings. These results should therefore be interpreted with caution. To validate and extend our observations, larger multicentre prospective studies are warranted. The median age at Fontan completion was 5 years, later than the international standard of 1.5–3 years. This reflects delayed referral and limited access to specialised surgery in Vietnam, which may reduce external validity and influence outcomes.
This study provides quantitative evidence of the dual effects of fenestration size in the Fontan procedure. Fenestration diameters of 4–8 mm were associated with significant perioperative improvement in oxygenation, whereas larger fenestrations facilitated better early recovery metrics but at the expense of lower oxygen saturation. These findings suggest that a uniform sizing strategy is inadequate and highlight the importance of an individualised approach that balances systemic venous decompression with acceptable arterial desaturation. Future multicentre studies with larger cohorts are needed to establish evidence-based guidelines for optimal fenestration sizing.
The datasets used in this study can be obtained from the corresponding author upon a reasonable request.
Conceptualization: LTKV. Data curation: LTKV, HNV. Investigation: LTKV, HNV. Methodology: HTB. Formal analysis: LTKV, HTB. Statistical analysis: HTB. Visualization: HTB. Validation: LTKV, HNV. Writing — original draft: LTKV, HNV. Writing — review & editing: HNV, HTB. Supervision: LTKV, HNV. Final approval of the manuscript: LTKV, HNV, HTB. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and was approved by the Institutional Review Board of Cho Ray Hospital (approval number: 67-27/CN HDDD on June 3rd 2025). In view of its retrospective design, the requirement for individual informed consent was waived by the board. All patient data were anonymised, and confidentiality was strictly maintained throughout the study.
The authors thank the nurses, physicians, and participants at Cho Ray Hospital, where the study was conducted.
This research received no external funding.
The authors declare no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





