1 Department of Cardiac Surgical Intensive Care Unit, Guangdong Provincial People's Hospital (Guangdong Academy of Medical Sciences), Southern Medical University, 510080 Guangzhou, Guangdong, China
Abstract
Malnutrition frequently occurs in older adults receiving cardiac surgery and is often linked to poor long-term prognosis. In geriatric care, the Geriatric Nutritional Risk Index (GNRI) offers a practical and efficient assessment tool to evaluate malnutrition risk, yet its relationship with cardiovascular mortality following cardiac valve surgery remains inadequately explored.
This research employed a retrospective cohort design with a sample of 170 patients of ≥60 years or older who underwent valve surgery at a tertiary hospital in 2013. To evaluate nutritional status, we employed using GNRI. The principal outcome was cardiovascular death with a median 7.2 years follow-up. Our methodological framework for investigating the GNRI-cardiovascular mortality link integrated three key components: multivariable Cox regression (to estimate hazard ratios), restricted cubic spline modeling (to explore non-linear trends), and Kaplan–Meier analysis (to visualize survival curves).
The results indicated a significant inverse linear the correlation of GNRI with the risk of cardiovascular mortality (p for nonlinearity = 0.274). After multivariable adjustment, per unit increment in GNRI showed a significant association 41% decline in the risk of death from cardiovascular causes (hazard ratios (HR): 0.59, 95% CI: 0.38–0.90, p = 0.015). Individuals with GNRI ≤92 had significantly worse long-term survival than those with GNRI >92 (p = 0.0057).
Preoperative GNRI is independently related to cardiovascular deaths in elderly postoperative cardiac valve surgery patients. A reduced GNRI signifies a significant marker for late cardiovascular death over the long term in cardiac valve surgery.
Keywords
- cardiac valve surgery
- older adults
- geriatric nutritional risk index
- cardiovascular mortality
- nutritional risk
- cardiovascular outcomes
Surgery performed on the heart has become increasingly prevalent worldwide, driven by the growing burden of cardiovascular diseases such as valvular disorders, especially in elderly populations [1]. Despite advances in surgical techniques and perioperative care, these procedures continue to carry substantial risks of postoperative complications and mortality [2]. Malnutrition has been increasingly acknowledged as an essential and adjustable determinant of postoperative recovery and long-term survival [3].
Based on serum albumin and the actual-to-ideal body weight ratio, the Geriatric Nutritional Risk Index (GNRI) serves as a valuable instrument for assessing nutritional risk [4]. It reflects both visceral protein reserves (through albumin, a key marker of nutritional status and systemic inflammation levels) and somatic protein mass (through body weight), providing a composite measure of nutritional health [5]. Beyond its initial purpose of evaluating nutritional risk in elderly inpatients, the GNRI demonstrates utility in forecasting clinical outcomes in heart failure, coronary disease, and cardiac surgery [6, 7, 8].
In cardiac surgery, accumulating evidence has demonstrated the prognostic value of GNRI, though inconsistencies persist across studies. Investigations by Gürbak et al. [6] and Naganuma et al. [9] identified clear ties between low GNRI and elevated mortality in valve replacement surgical cohorts, with median follow-up durations of 4.2 and 2.7 years, respectively. Pavone et al. [10] also confirmed the impact of poor nutritional state on valve surgery results. Conversely, Luo et al. [11], analyzing studies with median follow-ups 4.5 years, pointed to ongoing discrepancies. These conflicting findings may arise from methodological variations in structural characteristics of the studies and cohort sizes, GNRI thresholds, and especially insufficient, follow-up duration, as relatively short observation periods likely obscure the true long-term prognostic significance of preoperative nutritional status.
To overcome these research limitations, we implemented a retrospective cohort study with a median 7.2 years follow-up-substantially longer than previous reports. This research purposes to examine the independent connection preoperative GNRI and cardiovascular death among older adults receiving cardiac valve surgery, to provide robust evidence to guide the management of this clinically susceptible group over the long term.
This research employed a retrospective cohort design utilizing data from all
consecutive elderly patients who received valvular heart surgery at a tertiary
hospital during 2013. After applying the exclusion criteria, 170 out of 213
adults who had surgery with cardiopulmonary bypass were included in the analysis.
Eligible patients were aged
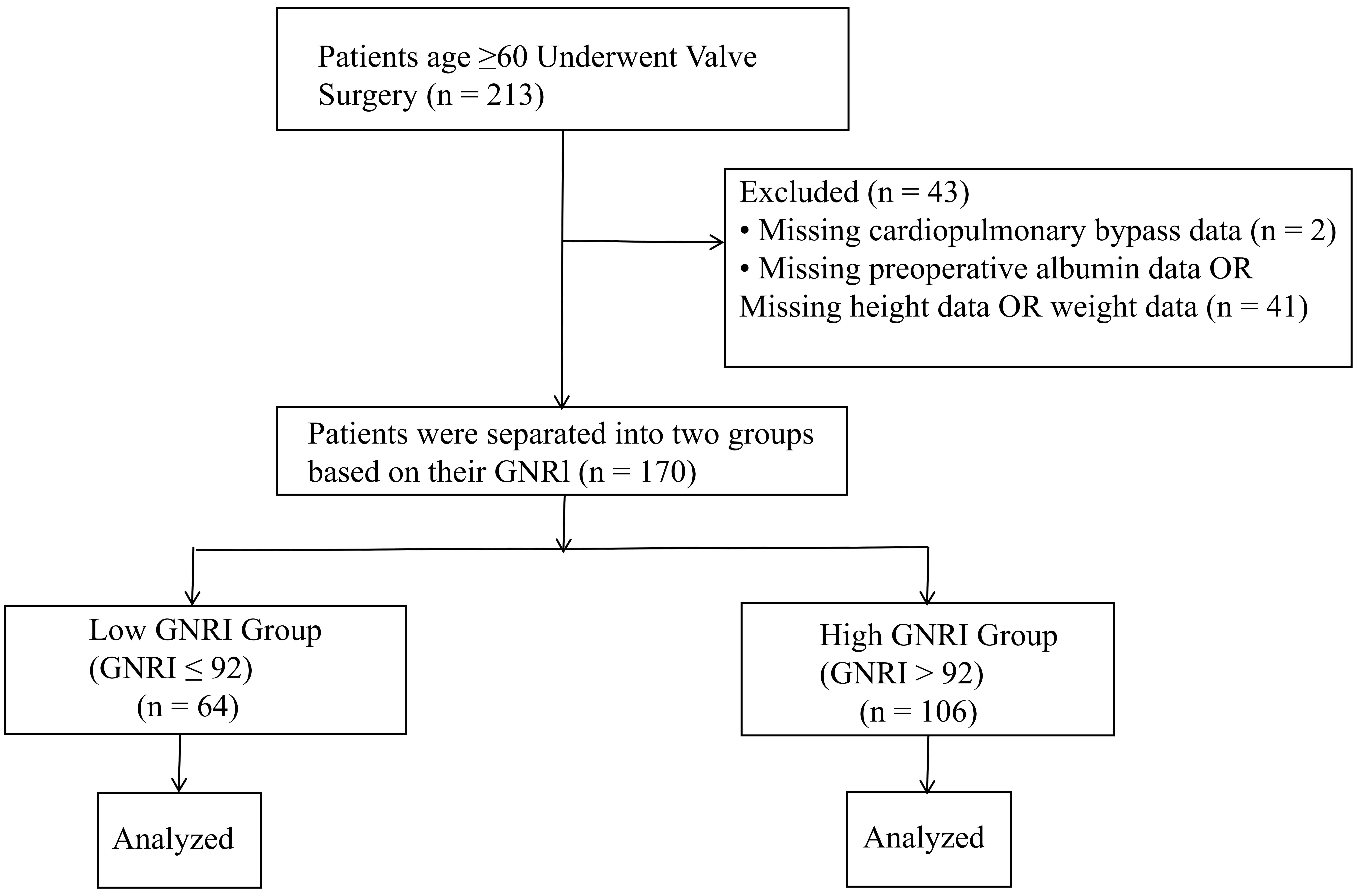 Fig. 1.
Fig. 1.
Patient flow diagram. GNRI, Geriatric Nutritional Risk Index.
As a tool for nutritional assessment, the standard GNRI equation, scores were
computed based on serum albumin (g/L) and the ratio of actual to ideal body
weight (kg), specifically: 1.489
Patient data, comprising baseline demographics and clinical characteristics were systematically obtained from medical records, including male, female, height, weight, age, comorbidities (hypertension, diabetes, atrial fibrillation), preoperative laboratory values (albumin, hemoglobin, platelet count), ejection fraction, and operative parameters (cardiopulmonary bypass and cross-clamp time). Follow-up extended for a median of 7.2 years, combining electronic medical record reviews and structured telephone interviews to evaluate long-term cardiovascular mortality. Cardiovascular mortality was rigorously defined according to contemporary standards [14, 15, 16], including deaths attributable to: (1) acute myocardial infarction; (2) progressive heart failure; (3) sudden cardiac death; and (4) fatal malignant arrhythmias. An independent endpoint adjudication committee comprising two board-certified cardiologists reviewed all available clinical documentation, including hospital records, laboratory results, and death certificates. In cases of disagreement, a third senior cardiologist provided final arbitration. Survival duration was computed from surgery until cardiovascular death or the study’s conclusion (July 1, 2021). Patient survival status was diligently ascertained via telephone by a team of well-trained researchers. In cases where patients had expired, the date and circumstances of death were meticulously documented. Duration of survival was calculated using a time-to-event analysis, spanning from the index cardiac procedure to the first occurrence of either death (all-cause) or the study termination date (July 1, 2021).
For normally distributed continuous variables, data were presented as the mean
For analytical purposes, we stratified patients into two nutritional risk
categories using established GNRI score thresholds:
| Characteristic | Overall (n = 170) | Low group (n = 64) | High group (n = 106) | p | |
| Gender | 0.177 | ||||
| Female | 75 (44.12) | 24 (37.50) | 51 (48.11) | ||
| Male | 95 (55.88) | 40 (62.50) | 55 (51.89) | ||
| Age (years) | 67.00 (66.00–70.00) | 68.50 (66.75–71.25) | 67.00 (65.00–69.00) | ||
| Height (cm) | 160.00 (155.00–168.00) | 163.00 (156.00–170.00) | 160.00 (155.00–168.00) | 0.621 | |
| Weight (kg) | 57.97 |
53.69 |
60.56 |
||
| Hypertension | 0.033 | ||||
| No | 125 (73.53) | 53 (82.81) | 72 (67.92) | ||
| Yes | 45 (26.47) | 11 (17.19) | 34 (32.08) | ||
| Diabetes | 0.749 | ||||
| No | 158 (92.94) | 60 (93.75) | 98 (92.45) | ||
| Yes | 12 (7.06) | 4 (6.25) | 8 (7.55) | ||
| Atrial fibrillation | 0.313 | ||||
| No | 117 (68.82) | 47 (73.44) | 70 (66.04) | ||
| Yes | 53 (31.18) | 17 (26.56) | 36 (33.96) | ||
| Preoperative ALB (g/L) | 36.24 |
32.61 |
38.44 |
||
| Preoperative HGB (g/L) | 130.00 (118.00–138.00) | 123.50 (108.75–134.50) | 132.00 (123.00–139.75) | 0.003 | |
| Preoperative PLT (109/L) | 164.00 (136.25–199.75) | 167.00 (133.50–207.50) | 162.00 (138.00–191.00) | 0.723 | |
| LVEF | 63.50 (57.00–69.00) | 60.50 (48.75–68.00) | 65.00 (60.00–69.00) | 0.006 | |
| GNRI | 93.74 (89.20–97.76) | 87.72 (80.53–90.02) | 96.84 (94.06–100.37) | ||
| CPB time (min) | 127.00 (97.50–164.00) | 134.50 (97.00–164.25) | 126.00 (104.50–159.50) | 0.988 | |
| Aortic cross-clamp | 87.50 (64.25–113.75) | 86.50 (59.25–114.25) | 88.00 (65.00–112.50) | 0.903 | |
| Time (min) | |||||
| MV time (hours) | 21.00 (14.00–43.75) | 23.00 (16.25–66.25) | 21.00 (13.25–34.00) | 0.082 | |
| ICU stay (hours) | 65.00 (40.25–137.00) | 89.50 (45.00–165.00) | 50.00 (27.25–111.50) | 0.022 | |
| Postoperative hospital stay (days) | 16.00 (9.25–25.00) | 20.00 (9.75–31.25) | 15.00 (9.25–21.00) | 0.011 | |
| In-hospital death | 0.154 | ||||
| No | 162 (95.29) | 59 (92.19) | 103 (97.17) | ||
| Yes | 8 (4.71) | 5 (7.81) | 3 (2.83) | ||
| Days of follow-up | 2831.00 (2725.50–2938.25) | 2843.50 (2071.00–2934.75) | 2810.50 (2733.25–2939.75) | 0.962 | |
| Cardiovascular mortality in long-term | 0.003 | ||||
| follow-up | |||||
| No | 130 (76.47) | 41 (64.06) | 89 (83.96) | ||
| Yes | 40 (23.53) | 23 (35.94) | 17 (16.04) | ||
ALB, albumin; HGB, hemoglobin; PLT, blood platelet; LVEF, left ventricular ejection fraction; GNRI, Geriatric Nutritional Risk Index; CPB, cardiopulmonary bypass; MV, mechanical ventilation; ICU, intensive care unit.
Univariate Cox regression revealed that a high GNRI (HR: 0.42, 95% CI:
0.23–0.79, p = 0.007), older age (HR: 1.09, 95% CI: 1.01–1.17,
p = 0.023), lower left ventricular ejection fraction (HR: 0.97, 95% CI:
0.95–0.99, p = 0.005), and longer cardiopulmonary bypass time (HR:
1.01, 95% CI: 1.00–1.01, p
| Variables | HR | 95% CI | p-value | |
| GNRI | ||||
| Low | ref | ref | ref | |
| High | 0.42 | 0.23–0.79 | 0.007 | |
| Male | 1.53 | 0.80–2.93 | 0.199 | |
| Age | 1.09 | 1.01–1.17 | 0.023 | |
| Height | 1.01 | 0.97–1.05 | 0.713 | |
| Weight | 1.01 | 0.98–1.04 | 0.511 | |
| Hypertension | 1.21 | 0.62–2.39 | 0.574 | |
| Diabetes | 0.67 | 0.16–2.79 | 0.585 | |
| Atrial fibrillation | 1.10 | 0.57–2.13 | 0.779 | |
| Preoperative HGB | 0.99 | 0.97–1.01 | 0.258 | |
| Preoperative PLT | 1.00 | 1.00–1.01 | 0.670 | |
| LVEF | 0.97 | 0.95–0.99 | 0.005 | |
| CPB time (min) | 1.01 | 1.00–1.01 | ||
HR, hazard ratio; 95% CI, 95% confidence interval; GNRI, Geriatric Nutritional Risk Index; HGB, hemoglobin; PLT, blood platelet; LVEF, left ventricular ejection fraction; CPB, cardiopulmonary bypass.
A quantitative link between the GNRI, measured on a continuous scale, and long-term cardiovascular mortality was established (change per 1 standard deviation (SD), HR: 0.59, 95% CI: 0.38–0.90, p = 0.015) and (change per 10, HR: 0.49, 95% CI: 0.28–0.87, p = 0.015). Patients categorized in the higher GNRI group exhibited a significantly reduced risk of long-term cardiovascular mortality compared to those in the lower group (HR: 0.38, 95% CI: 0.18–0.81, p = 0.012) in Model 2 (Table 3).
| Variables | Crude model | Model 1 | Model 2 | ||||
| HR (95% CI) | p-value | HR (95% CI) | p-value | HR (95% CI) | p-value | ||
| GNRI (Per 1 SD) | 0.67 (0.50–0.90) | 0.007 | 0.64 (0.42–0.96) | 0.32 | 0.59 (0.38–0.90) | 0.015 | |
| GNRI (Per 10 SD) | 0.59 (0.40–0.86) | 0.007 | 0.55 (0.32–0.95) | 0.32 | 0.49 (0.28–0.87) | 0.015 | |
| GNRI | |||||||
| Low group | ref | ref | ref | ||||
| High group | 0.42 (0.23–0.79) | 0.007 | 0.38 (0.18–0.81) | 0.012 | 0.38 (0.18–0.81) | 0.012 | |
Crude model: Without any adjustment.
Model 1: With adjustment for male, age, hypertension, diabetes, atrial fibrillation, preoperative HGB, preoperative PLT and LVEF.
Model 2: Adjusted for Model I+CPB time and aortic cross-clamp time.
HR, hazard ratio; 95% CI, 95% confidence interval; SD, standard deviation; GNRI, Geriatric Nutritional Risk Index; HGB, hemoglobin; PLT, blood platelet; LVEF, left ventricular ejection fraction; CPB, cardiopulmonary bypass.
An approximately linear relationship between GNRI and long-term cardiovascular
mortality was observed, as assessed by RCS modeling (p for nonlinearity
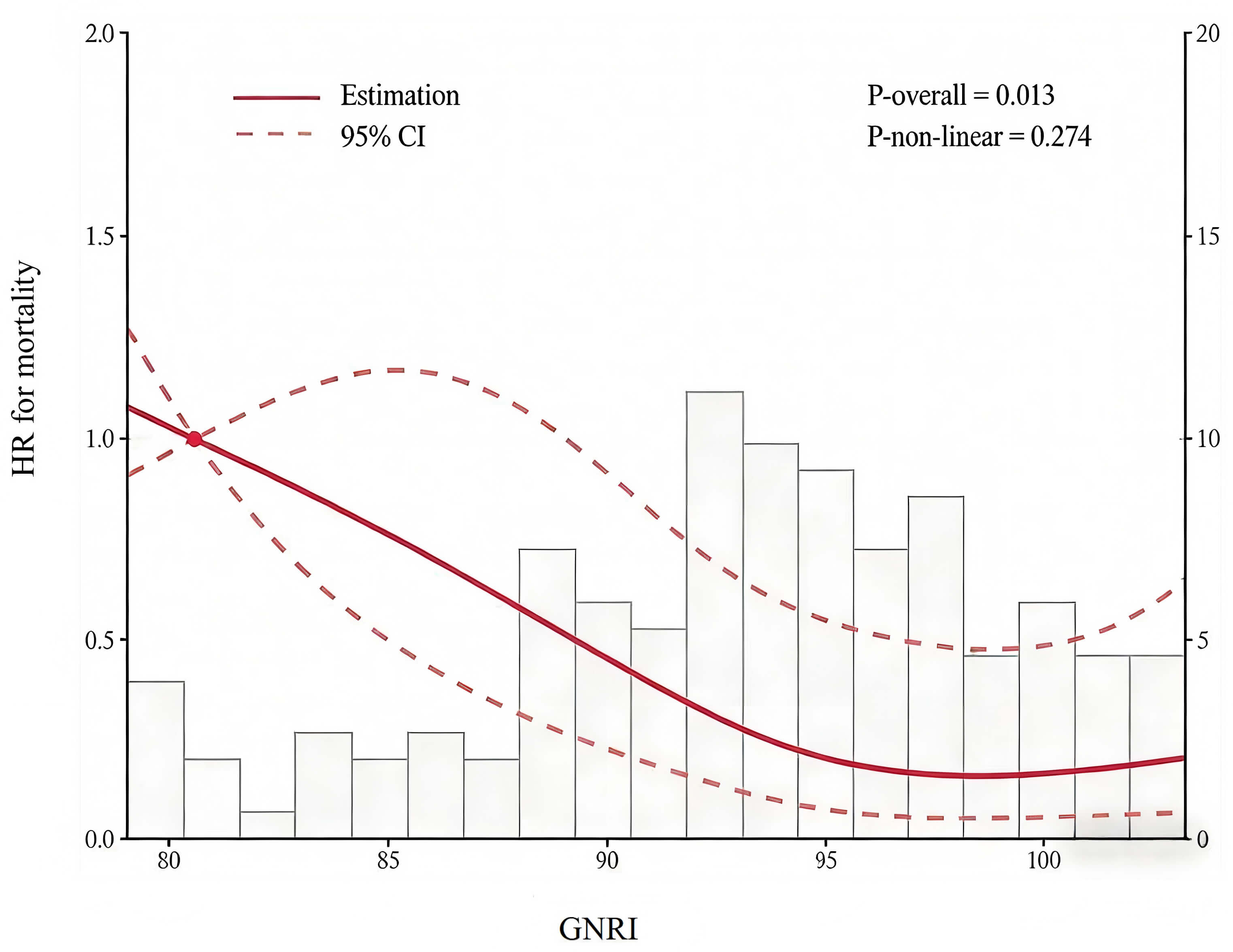 Fig. 2.
Fig. 2.
RCS curve of GNRI for long-term follow-up cardiovascular mortality. GNRI, Geriatric Nutritional Risk Index; RCS, restricted cubic splines; CI, confidence interval.
We performed subgroup analyses to confirm the stability of our results from the
fully adjusted Cox multivariate regression Model 3. These analyses stratified the
cohort by several prespecified factors, including age, gender, hypertension,
atrial fibrillation as a comorbidity, combined with valvular operations (mitral
or aortic). These analyses attempted to investigate potential interactions among
GNRI and long-term cardiovascular deaths and to identify any biases in different
population groups. GNRI had a more significant effect on long-term mortality in
the population without hypertension, with no significant interaction detected in
other subgroups (Fig. 3), confirming the consistent and reliable results of the
other subgroup analyses (p
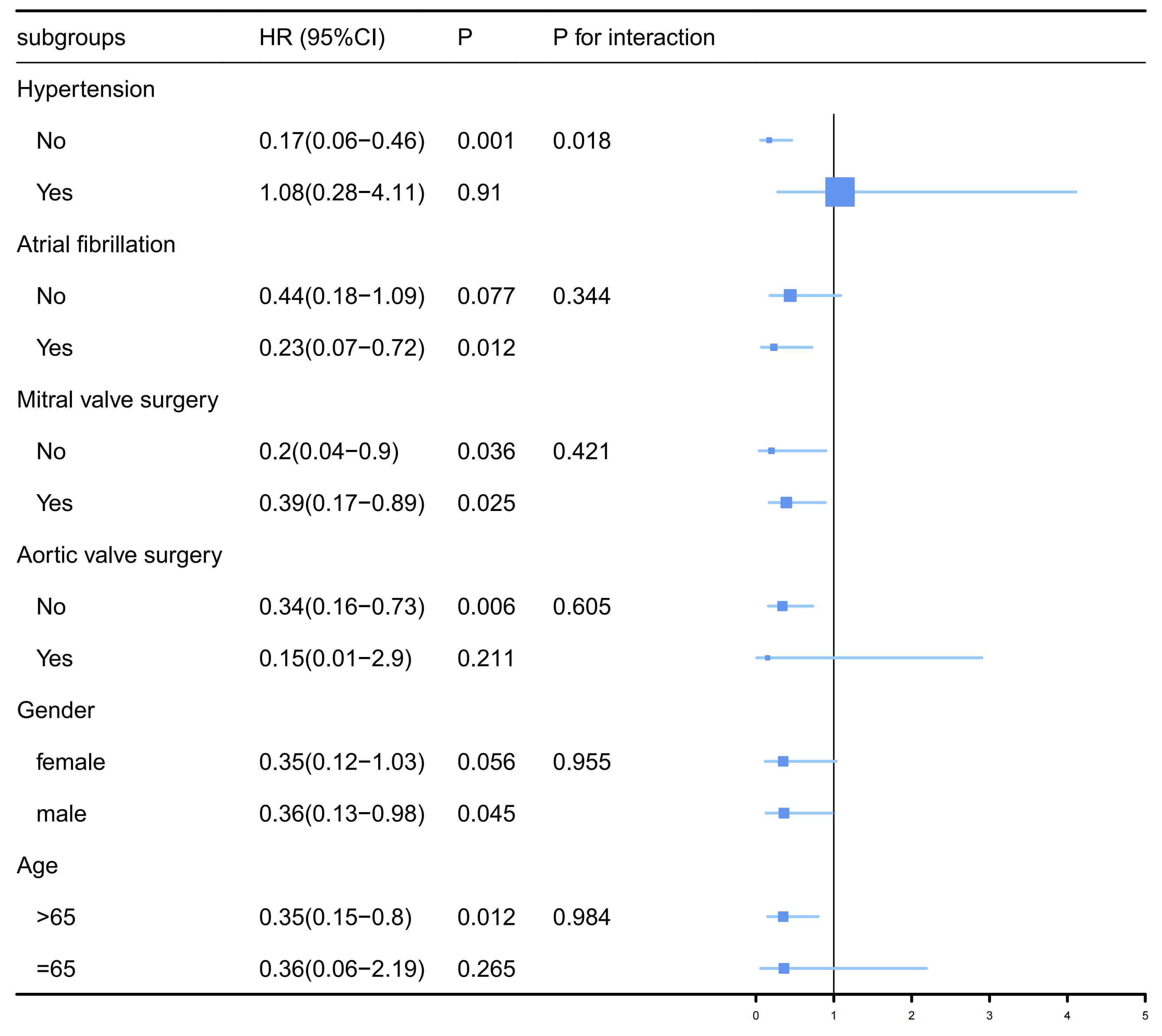 Fig. 3.
Fig. 3.
Subgroup assessment of the connection between GNRI and cardiovascular deaths.
A significantly reduction in survival probability was observed in patients with
GNRI
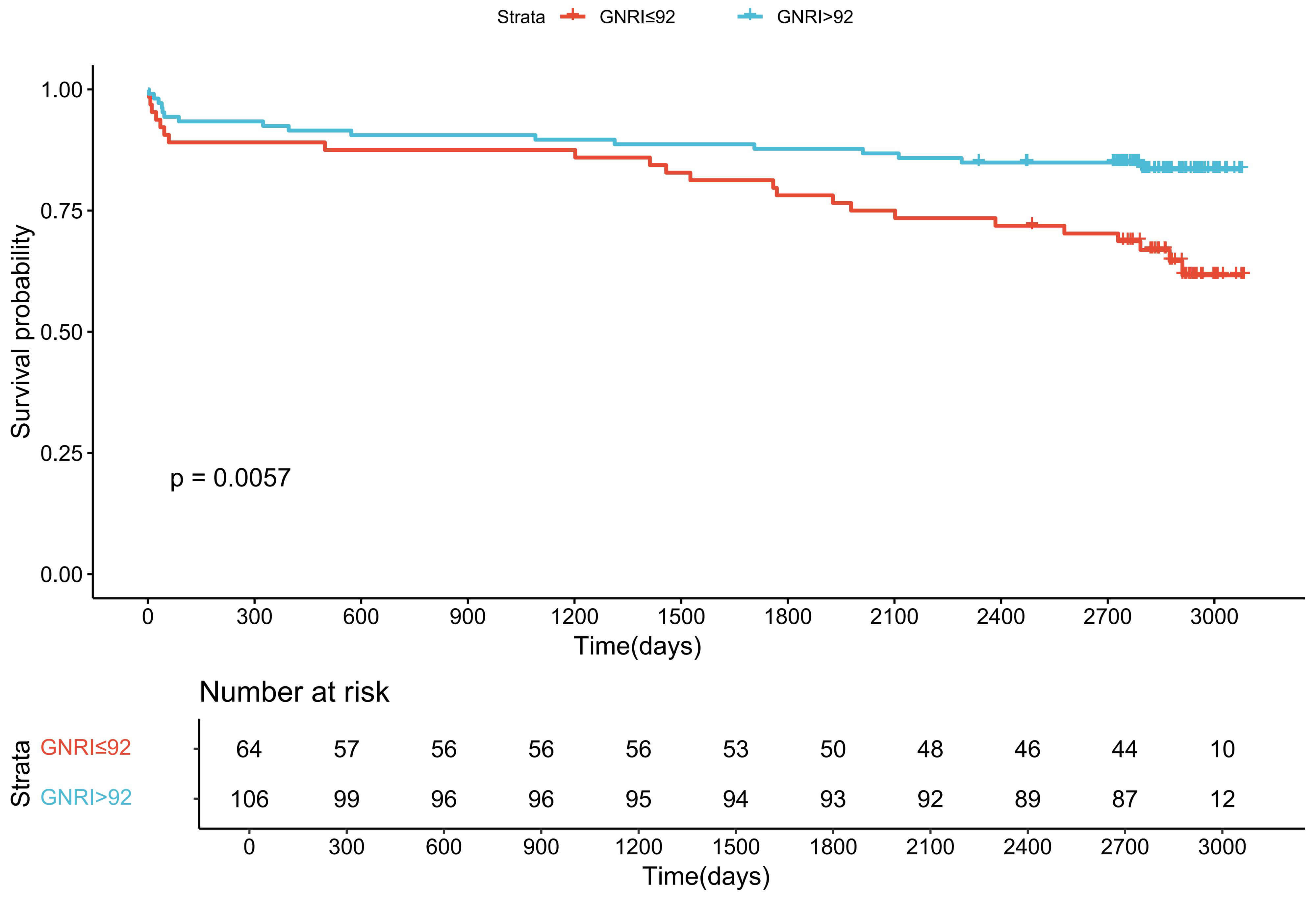 Fig. 4.
Fig. 4.
Long-term cardiovascular survival stratified by GNRI category
(Kaplan–Meier analysis). The solid blue line represents patients with GNRI
This study of elderly patients following valve surgery demonstrated that a lower preoperative GNRI was independently and significantly linked to a higher long-term risk of cardiovascular death. With a median 7.2 years follow-up, our findings provide compelling evidence that impaired nutritional status is substantially associated with adverse late cardiovascular outcomes in this vulnerable patient population.
Our results are consistent with, but substantially extend, previous reports investigating GNRI in cardiac surgery populations. Prior studies in transcatheter and surgical aortic valve replacement (transcatheter aortic valve implantation (TAVI) and surgical aortic valve replacement (SAVR), respectively) cohorts have demonstrated associations between GNRI and mid-term outcomes [6, 9, 17, 18]. However, these investigations were characterized by considerably shorter follow-up periods, typically ranging from 2.7 to 4.5 years [6, 9, 11]. The consistent relationship observed across various cardiac procedures, including TAVI, SAVR, and coronary artery bypass grafting, suggests that association between nutritional status and cardiovascular mortality represents a fundamental biological relationship independent of the specific type of intervention. Importantly, our study demonstrates that this association remains robust beyond seven years of follow-up, representing the longest observation period reported in this clinical context.
The pathophysiological pathway underlying the link between malnutrition and increased long-term cardiovascular mortality likely involves a cascade of interconnected biological processes. Malnutrition initiates a state of chronic low-grade inflammation, which subsequently promotes muscle protein catabolism and cardiac cachexia, ultimately leading to progressive myocardial dysfunction [13, 19]. Concurrently, hypoalbuminemia compromises the capacity for tissue repair, immune competence, and antioxidant defense mechanisms [6, 11]. This multifaceted physiological deterioration significantly diminishes patients’ stress tolerance and functional reserve, creating a vulnerable clinical phenotype that is less capable of withstanding the long-term challenges of cardiovascular disease progression and age-related physiological decline.
The robust relationship between GNRI and cardiovascular mortality, maintained throughout 7.2-year follow-up period, highlights the essential role of systematic nutritional evaluation during the preoperative assessment for elderly patients undergoing heart surgery. GNRI provides an efficient method for stratifying patients at elevated risk for late cardiovascular mortality, who may benefit from optimized long-term management strategies. These should include protocol-driven nutritional support, vigilant monitoring, and sustained cardiac rehabilitation programs that extend well beyond the conventional postoperative care stage.
Several constraints in our study design warrant consideration during results interpretation. The retrospective, single-institution nature combined with a limited cohort size (n = 170) could lead to selection bias and constrain the external validity of our conclusions. Additionally, determination of mortality relied on electronic medical records and telephone follow-up, and only four confirmed non-cardiovascular deaths were identified. Given the limited number of competing events, we utilized standard Cox proportional hazards models rather than competing risk analyses, as the latter approach would have generated hazard ratios with excessively wide confidence intervals, potentially compromising the statistical reliability of our conclusions. Future prospective studies with larger multicenter cohorts and more rigorous cause-specific mortality adjudication would help strengthen the evidence base for these observations.
This investigation establishes GNRI as an independent factor associated with long-term cardiovascular mortality in elderly valve surgery recipients, with 7.2-year, follow-up data representing the longest observation period reported in this population. Preoperative nutritional status emerges as a persistent determinant of late cardiovascular outcomes, supporting the value of nutritional assessment as an essential element in the multidisciplinary management of elderly surgical populations.
All datasets generated and analyzed in this study are contained in this published article.
CJZ: designed the study, data analysis, revised the manuscript; HLH: Conceptualization and Writing original draft; QL: data analysis, Writing & editing; RXB and SML: data curation, Writing & editing. XLG and QFL: contributed to follow-up data collection, patient tracking, outcome verification, and manuscript editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript to be submitted. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the guidelines of the Helsinki Declaration, and the study protocol was approved by the Research Ethics Committee of the Guangdong Provincial People’s Hospital in September 2024 with code No. KY2024-043-01. The institutional review board waived the requirement for informed consent for this retrospective, anonymized study.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




