1 Department of Thoracic and Cardiovascular Surgery, Miller Family Heart, Vascular & Thoracic Institute, Cleveland Clinic, Cleveland, OH 44195, USA
2 Division of Surgical Critical Care, Integrated Hospital Care Institute, Cleveland Clinic, Cleveland, OH 44195, USA
3 Division of Cardiovascular Anesthesia, Integrated Hospital Care Institute, Cleveland Clinic, Cleveland, OH 44195, USA
Abstract
Coronary artery bypass grafting (CABG) is the most frequently performed cardiac surgery worldwide. Improvements in operative technique and perioperative care have led to a significant reduction in associated morbidity and mortality. Achieving optimal outcomes requires meticulous surgical technique that is complemented by comprehensive postoperative care. This review aims to summarize the principles of postoperative care following CABG based on latest evidence and our extensive institutional experience. Immediate postoperative care in the intensive care unit focuses on management of acute cardiorespiratory issues, bleeding, and pain management. Ward care focuses of ensuring a smooth transition from inpatient treatment to outpatient recovery. Protocolized postoperative interventions, including Enhanced Recovery after Surgery-cardiac and emerging applications of automation and artificial intelligence, are transforming postoperative CABG care by promoting faster recovery, reducing complications, and enabling more personalized, data-driven decision-making.
Keywords
- critical care
- mechanical circulatory support
- postoperative care
- vasoplegia
Coronary artery bypass grafting (CABG) is the recommended treatment for complex triple vessel coronary artery disease [1, 2]. According to the Society of Thoracic Surgeons (STS) database, 161,907 CABG operations were performed in the United States in 2023 [3]. Isolated CABG mortality is less than 3% but may increase to 11% in combined CABG and valve surgery [3]. The success of CABG surgery relies heavily on postoperative care, which represents a critical determinant of short- and long-term outcomes. This review article explores key management principles after CABG surgery in a system-based format throughout every phase of care (Fig. 1).
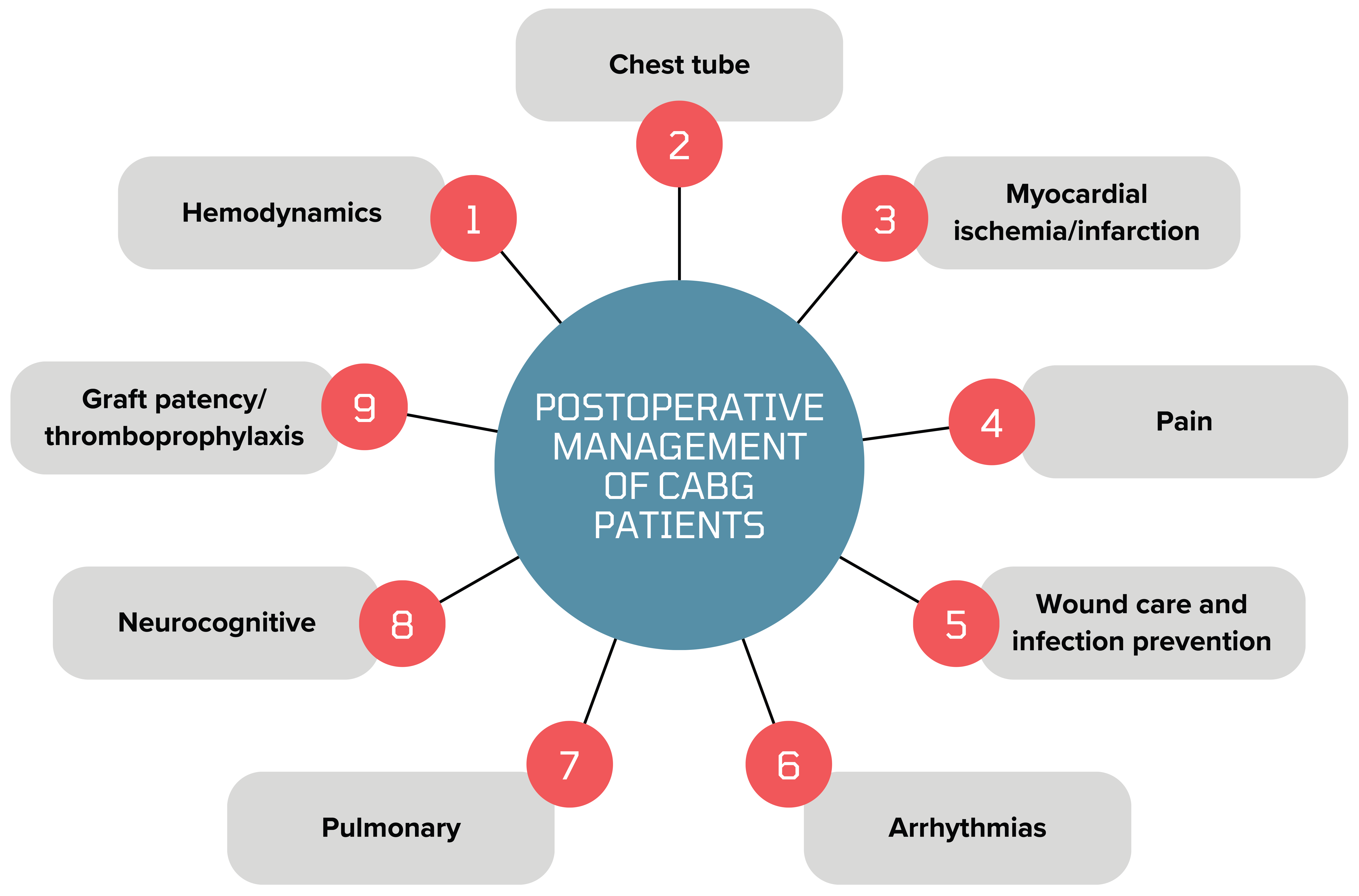 Fig. 1.
Fig. 1.
Key aspects of postoperative care after CABG (coronary artery bypass grafting). Created by Canva. https://www.canva.com/.
After CABG, patients need multiorgan system management that requires effective communication with protocolized hand-off among surgical, anesthesia, and cardiac surgical intensive care teams. This hand-off needs to provide detailed communication about surgical indication and procedure, comorbidities, intraoperative valve and myocardial function, vasoactive medication use, blood products administered in the operating room, and a subjective impression of hemostasis. Assuming an uncomplicated postoperative course, early recovery after surgery (ERAS®) principles have been adapted to cardiac surgery (ERAS®-Cardiac) and provide reliable pathways and targets to guide and expedite transfer out of the intensive care unit (ICU) and discharge from the hospital.
The key components of ERAS-Cardiac protocols [4] for CABG surgery, specifically in the context of postoperative patient management, include several checkpoints:
1. Multimodal opioid-sparing analgesia: reduces opioid consumption and associated side effects.
2. Early extubation: enables early mobilization and lowers risk of ventilator-associated pneumonia.
3. Early mobilization: reduces thromboembolic risk and accelerates overall recovery.
4. Postoperative nausea and vomiting prophylaxis: facilitates early oral intake and improves gastrointestinal function and nutrition recovery.
5. Removal of invasive lines and catheters: decreases infection risk and enhances patient mobility.
6. Optimized fluid management (goal-directed therapy): ensures adequate preload and prevents fluid overload complications.
7. Normothermia maintenance: reduces surgical site infections and supports normal metabolic function.
Extubation before 6 hours is a STS benchmark for quality of care. Early
extubation (
Once a patient demonstrates cardiorespiratory stability and stable end organ function, a transfer to an inpatient floor or stepdown unit is considered. Ward care focuses on incrementally advancing mobilization, and identifying adequate doses of medications at discharge with ongoing surveillance for complications. Hospital discharge preparation includes education on wound care, medications, and complication recognition. Post-CABG depression (occurring up to 1 year later) may require psychological support [8, 9]. Disposition depends on preoperative function. Postoperatively, most patients discharge home, while others transition to rehab or skilled nursing facilities [10].
Proper management of sedation and opioid management is critical to minimize the
risk of delirium. In a large meta-analysis, 24% patients were noted to have
delirium after CABG [11]. Known risk factors for delirium after CABG are presence
of cognitive impairment, history of stroke, depression, arrhythmias (including
atrial fibrillation [AF]), peripheral vascular disease, kidney disease, body mass
index (BMI)
While the patient is on mechanical ventilation, sedation at its lowest effective dose is administered to provide patient comfort. Propofol and dexmedetomidine are commonly used sedative hypnotics. Dexmedetomidine is a short active alpha-2 receptor agonist that has sedative and analgesic effects. Systematic review and meta-analyses have noted that dexmedetomidine may be associated with decreased duration of mechanical ventilation, and lowered incidence of delirium and ventricular arrhythmias as compared to propofol, but at the possible expense of increased risks of bradycardia and hypotension [16, 17]. As soon as cardiorespiratory stability and hemostasis are established, sedation is weaned off to prepare for extubation.
Opioid-based analgesia is a mainstay of postoperative pain management due to its established effectiveness [18]. Opioid-related adverse effects, including respiratory depression, constipation, nausea, vomiting, and analgesic tolerance and dependence, may however impede recovery and impact patient experience [19]. Multimodal analgesia protocols, including acetaminophen, gamma-aminobutyric acid analogues, muscle relaxants, topical anesthetic patches, and nerve blocks, have been increasingly used. Nonsteroidal anti-inflammatory use in CABG patients is controversial due to a “black-box warning” from the U.S. Food and Drug Administration for all cyclooxygenase (COX)-1 and COX-2 inhibitors, on the basis of two randomized, controlled trials on select COX-2 inhibitors [20]. Nevertheless, retrospective studies have shown the potential safety of nonselective COX inhibitors and their association with improved outcomes, including for graft patency, but these inhibitors are still classified as off-label use [20]. Studies have shown superiority of multimodal analgesia compared to opioids in lowering pain scores and decreasing reducing opioid-related adverse effects [21]. A recent systematic review have shown that regional blocks may help in reducing pain and postoperative opioid usage when compared to systemic analgesia alone [22]. Nonetheless, patients undergoing minimally invasive CABG usually require more analgesia due to initially higher postoperative pain due to lateral thoracotomy.
Postoperative cerebrovascular accidents have an incidence of 0.7% to 1.2%, and carry a significantly increased risk of mortality (11.7% for early stroke vs 3.4% without stroke) [23, 24, 25]. Ischemic mechanisms, such as embolism from aortic plaque, atrial fibrillation, or cerebral hypoperfusion, predominate over hemorrhagic causes. Major risk factors include advanced age, peripheral vascular disease, prior stroke, aortic atherosclerosis, and intraoperative hypotension [26]. Diagnosis requires maintaining a high level of suspicion while following serial neurologic assessments upon arrival to the ICU. Endovascular thrombectomy is pursued for large-vessel occlusion, since parenteral thrombolytics are contraindicated during the immediate postoperative period [27].
Systemic inflammatory response syndrome, which is induced by cardiopulmonary bypass and ischemic reperfusion injury with its associated cytokine release (interleukin-1 and -6, and tumor necrosis factor-alpha), causes multiple downstream effects resulting in vasoplegia [28]. This vasoplegia is associated with an increased risk of mortality, prolonged length of stay, and varying re-admission rate (9–44%, depending on the definition used for reporting) [29, 30]. Pre-existing renal failure, long aortic cross-clamp time, and cardiopulmonary bypass time, combined CABG with valve surgery, and intraoperative red blood cell transfusions are associated with a higher risk of vasoplegia [31]. Goal-directed volume resuscitation with crystalloids, colloids, or blood combined with conventional vasopressors is first-line therapy. A mean arterial pressure (MAP) of 70 to 80 mmHg is a practical target in patients with atherosclerotic disease to ensure adequate cerebral perfusion in the absence of evidence-based targets. Multiple randomized controlled trials have demonstrated no clinical advantage for colloids versus crystalloids, although colloids may result in a reduction in administered volume [32, 33]. Restrictive versus liberal blood transfusion strategies have shown equivalent clinical outcomes in randomized controlled trials, but studies on patients with critical hemodynamics are lacking [34]. Transfusion triggers and thresholds should target adequate tissue oxygen delivery and account for the rate of bleeding. Systemic corticosteroids, methylene blue, hydroxocobalamin, and angiotensin II agonists can be used for refractory vasoplegia [28]. Methylene blue should be avoided in patients with pulmonary hypertension, right ventricular failure, or glucose 6 phosphate dehydrogenase deficiency.
Low cardiac output syndrome (LCOS) is defined by insufficient cardiac output (CO) for metabolic demand despite adequate preload in the absence of cardiac tamponade, pneumothorax, or significant arrhythmias [35]. LCOS or postcardiotomy shock can result from myocardial stunning, ischemia, ischemic-reperfusion injury, and pre-existing myocardial dysfunction [36]. Its incidence (1.5–91%) varies due to the lack of a standardized definition [37]. Duncan and colleagues [38] noted a 12-fold increased risk of mortality after LCOS, which was defined as the need for mechanical circulatory support (MCS) within the first 5 days after the procedure or 2 inotropes on postoperative day 1. Advanced age, female sex, pre-existing myocardial dysfunction, atrial fibrillation, chronic kidney disease, combined CABG with valve surgery, prolonged cardiopulmonary bypass time, and number of intraoperative blood transfusions appear to be associated with increased risk of LCOS [38, 39, 40, 41]. Right ventricular failure as a case of LCOS is characterized by adequate or elevated right-sided filling pressures along with echocardiographic features of right ventricular dysfunction, although definitions vary [42]. Risk factors include mitral valve surgery, tricuspid valve surgery, baseline-elevated pulmonary artery pressure, pre-existing atrial fibrillation, and low ejection fraction (EF) [43, 44]. Echocardiographic findings include decreased systolic function by global (visual, 3D-derived right ventricle (RV) ejection fraction, fractional area change, right ventricle/left ventricle (RV/LV) size ratio) or segmental markers (tricuspid anular plane systolic excursion, and systolic velocity).
If LCOS is suspected, pulmonary artery catheter-derived parameters, such as thermodilution cardiac index (TDCI), estimated Fick’s cardiac index (eFCI), central venous pressure, pulmonary artery diastolic pressure or pulmonary capillary wedge pressure, and pulmonary artery pulse pressure index, are essential to integrate into patient management. Although correlation between TDCI and eFCI was modest (r = 0.65), a large retrospective study of more than 14,000 patients showed higher mortality at 90 days and 1 year in those with low TDCI and normal eFCI when compared to patients with low eFCI and normal TDCI (2.2–4 L/min/m2) [39]. Elderly patients with normal to high BMI, kidney disease, male sex, atrial fibrillation, and lower hemoglobin were at higher risk of discordantly low TDCI and normal eFCI. Epinephrine, dobutamine, and milrinone are inotropes that can be used in isolation or combination for the treatment of LCOS. Due to milrinone’s vasodilation effect, long half-life, and delayed clearance in the setting of renal dysfunction, alternate agents should be used in the patient with profound vasoplegia.
Patients with RV failure may benefit from a higher than usual goal mean arterial pressure of 65–70 mmHg, as elevated central venous pressure (CVP) may impair myocardial tissue perfusion. A small retrospective study noted improved renal function in patients with pre-capillary pulmonary hypertension using a MAP goal of 60 mmHg with CVP [45]. Providing inhalation of pulmonary vasodilators (nitric oxide, epoprostenol), along with decreasing pulmonary vascular resistance and increasing cardiac output in patients with RV failure may serve as additive therapy [46, 47]. Inhaled milrinone decreases pulmonary vascular resistance but has not been shown to increase cardiac output [48, 49]. Vasopressin may produce less pulmonary vasculature constriction compared to adrenergic agents and therefore may be beneficial [50]. Epicardial pacing via atrial or ventricular wires can be used even in the absence of bradycardia to support cardiac output. Acidemia, hypoxemia, and electrolyte abnormalities should be aggressively corrected.
Key points about hemodynamic abnormalities:
1. Vasoplegia risk factors include renal failure, long CPB time, combined CABG/valve surgery, and transfusions. First-line therapy is fluids and vasopressors.
2. LCOS risk factors include age, AF, chronic kidney disease (CKD), long bypass time, combined surgery, and transfusions.
3. RV failure as a cause of LCOS requires higher MAP targets (65–70 mmHg), pulmonary vasodilators (NO, epoprostenol), appropriate inotropes, and correction of hypoxemia, acidosis, and electrolyte abnormalities.
Refractory LCOS necessitates early MCS initiation to prevent end-organ damage.
While severe LCOS requiring MCS carries high mortality, initiation of
preoperative or intraoperative MCS improves outcomes as compared to postoperative
use [51, 52]. The decision to escalate to MCS can be challenging due to limited
guideline recommendations. The consensus statement by the Society for
Cardiovascular Angiography and Interventions, and endorsed by the STS [53],
classifies cardiogenic shock into 5 stages. Stage D is characterized by
deteriorating hemodynamics and organ perfusion while on inopressors, which
triggers escalation to MCS. Stage E is condition in extremis and cardiac arrest;
MCS may be the only option to save the patient (Table 1, Ref. [54]). At our
institution, more than moderate doses of inotropes (
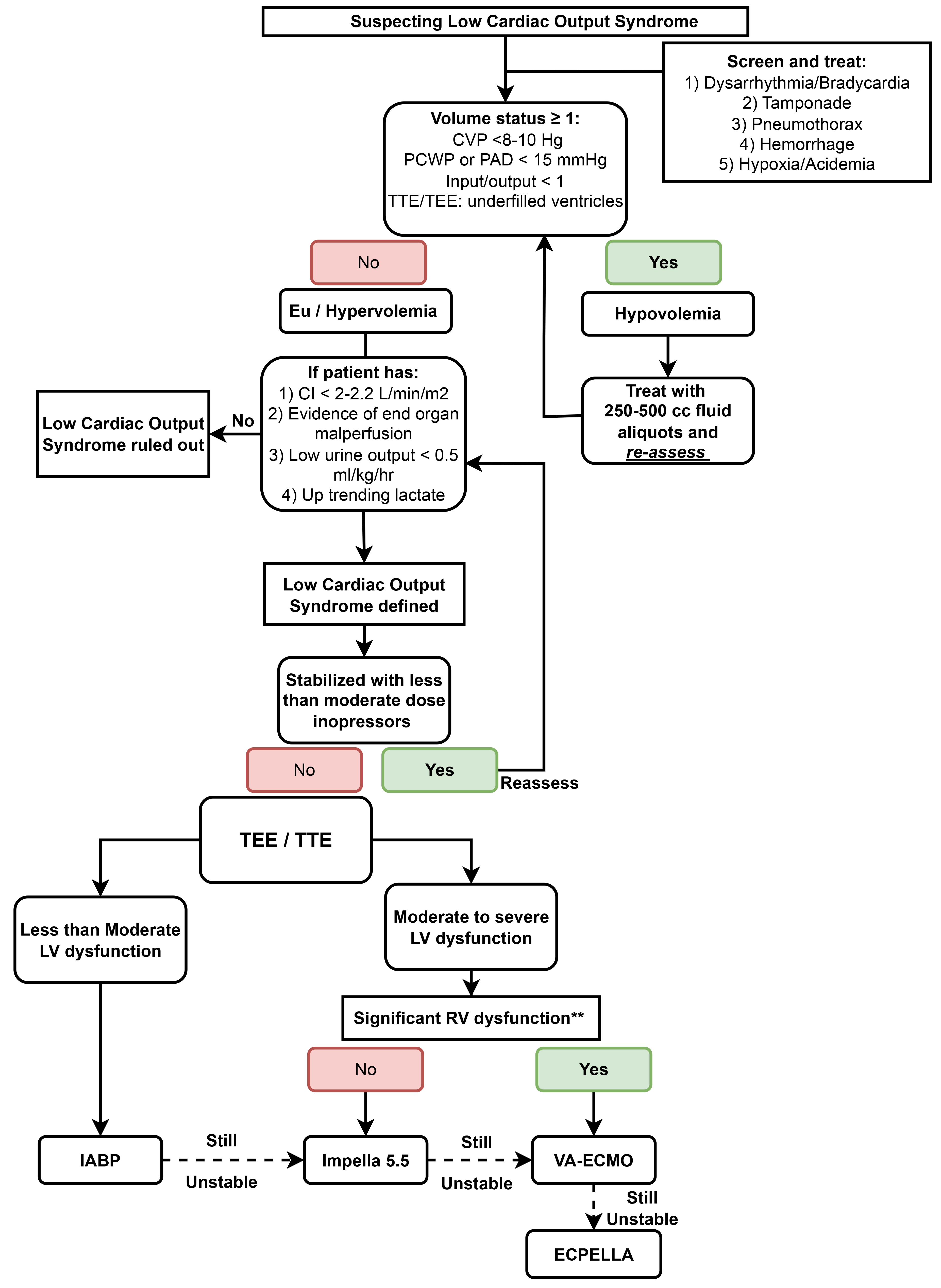 Fig. 2.
Fig. 2.
Proposed algorithm for managing low cardiac output syndrome after CABG. CVP, central venous pressure; PAD, pulmonary artery diastolic pressure; PCWP, pulmonary capillary wedge pressure; CI, cardiac index; TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; LV, left ventricle; RV, right ventricle; IABP, intra-aortic balloon pump; VA-ECMO, venoarterial-extracorporeal membrane oxygenation; LVAD, left ventricular assist device; RVAD, right ventricular assist device.
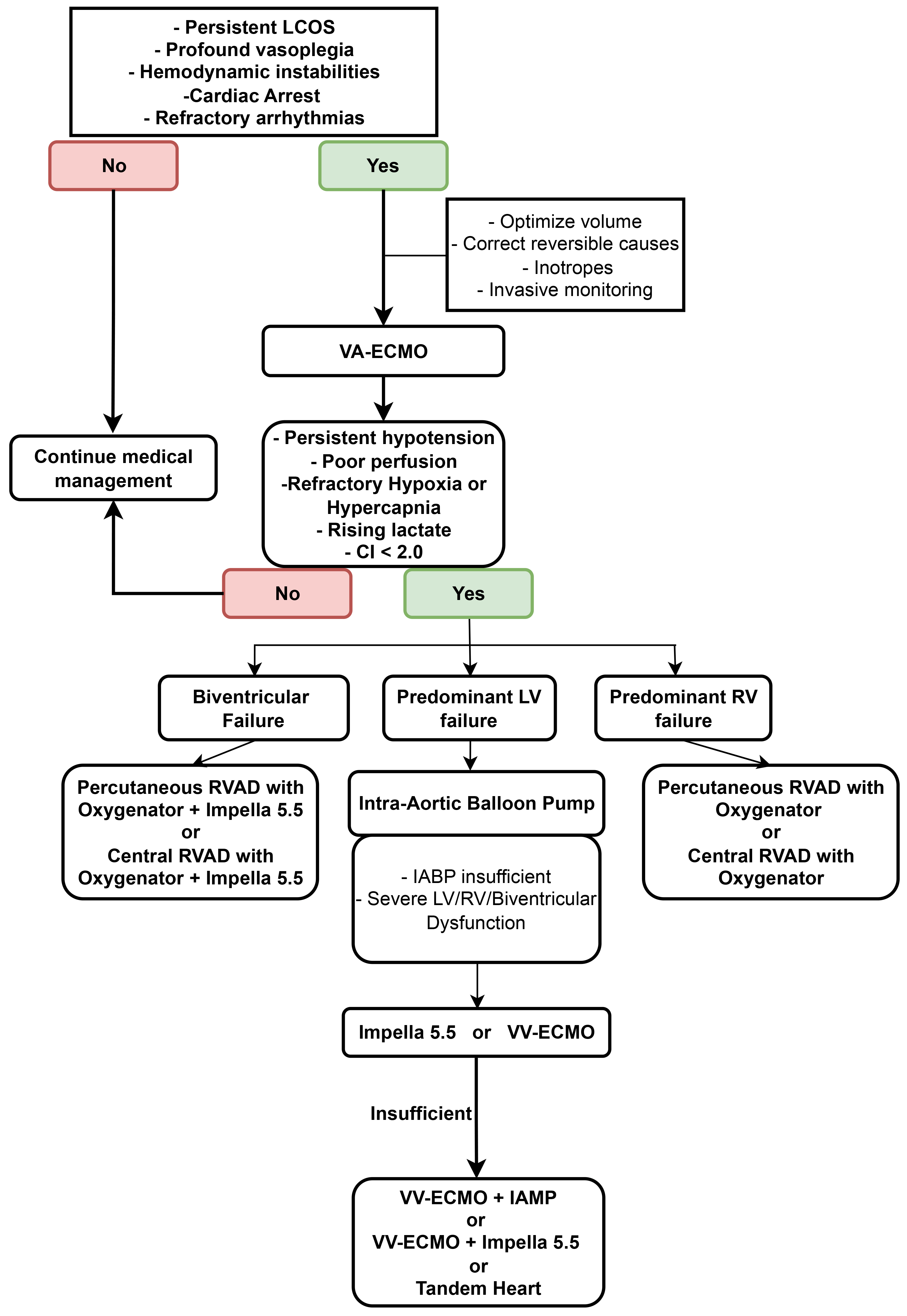 Fig. 3.
Fig. 3.
Proposed algorithm for mechanical circulatory support. LCOS, low cardiac output syndrome; VA-ECMO, venoarterial-extracorporeal membrane oxygenation; CI, cardiac index; LV, left ventricle; RV, right ventricle; RVAD, right ventricular assisted device; IABP, intra-aortic balloon pump; VV-ECMO, veno-venous extracorporeal membrane oxygenation.
| Stage | Description | Laboratory Data | Hemodynamics | Role of MCS |
| A (at risk) | No current signs or symptoms of CS, but at risk due to myocardial injury such as MI, acute heart failure or recent surgical procedure. | Normal labs (i.e., Cr, lactate, LFTs) | Normotensive; CI |
Not indicated |
| B (beginning CS) | Altered hemodynamics (i.e., hypotension or tachycardia) without evidence of hypoperfusion. | Mild Cr elevation (stage 1 AKI) Elevated [BNP] Normal lactate | SBP |
Early planning |
| C (classic CS) | Evidence of hypoperfusion that requires hemodynamic support beyond fluid resuscitation (i.e., inotrope, vasopressor). | Lactate |
SBP |
Strongly consider based on clinical response |
| D (deteriorating) | Classic CS with worsening condition and failure to respond to initial therapies. | Similar to stage C | Similar to stage C + deteriorating | Indicated |
| E (extremis) | Cardiac arrest with ongoing CPR, supported by multiple interventions. | Labs consistent with organ failure; pH 5 mmol/L | Pulselessness | Indicated |
This document was endorsed by the American College of Cardiology (ACC), the American Heart Association (AHA), the Society of Critical Care Medicine (SCCM), and the Society of Thoracic Surgeons (STS) in April 2019. CS, cardiogenic shock; MI, myocardial infarction; Cr, creatinine; LFTs, liver function tests; CI, cardiac index; CVP, central venous pressure; PaO2, arterial partial oxygen pressure; AKI, acute kidney injury; SBP, systolic blood pressure; MAP, mean arterial pressure; HR, heart rate; PCWP, pulmonary capillary wedge pressure; CPR, cardiopulmonary resuscitation.
Bleeding, tamponade, arrhythmias, ischemia, and LCOS are leading causes of post-CABG cardiac arrest. The treatment algorithm differs from advanced cardiac life support protocols [57]. Due to potential disruption of surgical anastomoses suture lines, external cardiopulmonary resuscitation can be very briefly delayed in order to initiate early defibrillation and epicardial pacing for unstable bradyarrhythmia or asystole. Intravenous lidocaine and amiodarone can be used in isolation or in combination for unstable ventricular tachyarrhythmias. Intravenous lidocaine may be associated with higher rates of return of spontaneous circulation, survival to hospital discharge, and favorable neurologic outcomes when compared to amiodarone [53]. Ventricular tachyarrhythmias should prompt suspicion for graft failure. In case of apparent pulseless electrical activity, epicardial wire pacing should be paused briefly to rule out underlying ventricular fibrillation [58]. In case of true pulseless electrical activity, external cardiopulmonary resuscitation can be started within 5 minutes while preparing for early re-sternotomy. The arterial line pressure readings are used as a guide to avoid over-aggressive compressions which can result in iatrogenic cardiovascular injury. Early re-sternotomy facilitates an effective internal cardiac massage, provides exposure for central VA-ECMO cannulation, relieves tamponade effect from mediastinal collections, and aids in prompt identification of the source of bleeding. Epinephrine bolus dosing should be administered with consideration of the lowest effective dosage under the supervision of an experienced provider since severe hypertension may precipitate bleeding and surgical suture line disruption. After the return of spontaneous circulation and once tamponade and bleeding have been ruled out, multidisciplinary evaluation for myocardial infarction and need for re-vascularization is essential.
The rate of surgical re-exploration is approximately 1.5% after isolated CABG and 3.5% after combined CABG surgery [59]. High chest-tube output even after correction of coagulopathy is an indication for re-exploration. Hemodynamic instability with or without elevated filling pressures can be an indication of tamponade. Prompt bedside echocardiographic assessment with transthoracic or transesophageal echocardiography can be helpful in making the diagnosis but cannot replace surgical judgment. Tamponade or significant hemothorax requires surgical re-exploration in the operating room or occasionally at the bedside for emergent hemodynamic stabilization.
Chest tubes should be assessed frequently for signs of obstruction, through visual inspection and functional assessment, since obstruction may lead to hemothorax and tamponade [60]. Active clearance technologies, such as the PleuraFlow® system, may help maintain patency and reduce the incidence of retained blood and the need for re-exploration in some studies [60]. The timing of chest tube removal is based on clinical criteria, including the amount and nature of drainage, absence of residual pleuro-pericardial effusion or air leak, or pneumothorax. Early chest tube removal within the first 24 hours of surgery is associated with residual pleuro-pericardial effusions requiring invasive interventions [61, 62]. Our typical practice is to remove the chest tubes on postoperative day 1 once the output is serosanguinous and less than 200 mL/day.
The incidence of post-CABG myocardial infarction is variable, ranging from 0.3% to 9.8% after isolated CABG and from 0.7% to 11.8% after combined CABG and valve surgery [63]. Differentiating myocardial injury due to cardiotomy, ischemia-reperfusion injury, or insufficient myocardial protection from bypass grafts or compromise of native coronary arteries can be challenging, and therefore requires a high level of suspicion and multiple diagnostic modalities. Ischemic chest pain can be masked by sternotomy pain and sedatives or analgesia. The fourth universal definition of myocardial infarction following CABG arbitrarily suggests troponin elevation 10 times the 99th percentile upper reference limit, with a further 20% increase if baseline troponin is elevated [64]. This definition also requires evidence of loss of viable myocardium in the form of abnormalities seen on electrocardiography, angiography, or myocardial imaging [64]. Biomarkers are often elevated immediately after cardiac surgery with lack of discrete threshold values predictive of graft failure or native coronary occlusion [64, 65, 66, 67]. Electrocardiography findings of new pathological Q waves, ST-T segment changes (especially ST elevation along with reciprocal ST depression), and new left bundle branch block pattern can be suggestive of post-CABG myocardial infarction, but in isolation have modest correlation with acute graft dysfunction [65, 66, 67]. New regional wall motion abnormalities detected by transesophageal echocardiography only identify 15.8% to 20.5% of acute graft failure [65, 66]. Our algorithm for detecting post-CABG myocardial infarction is described in Fig. 4.
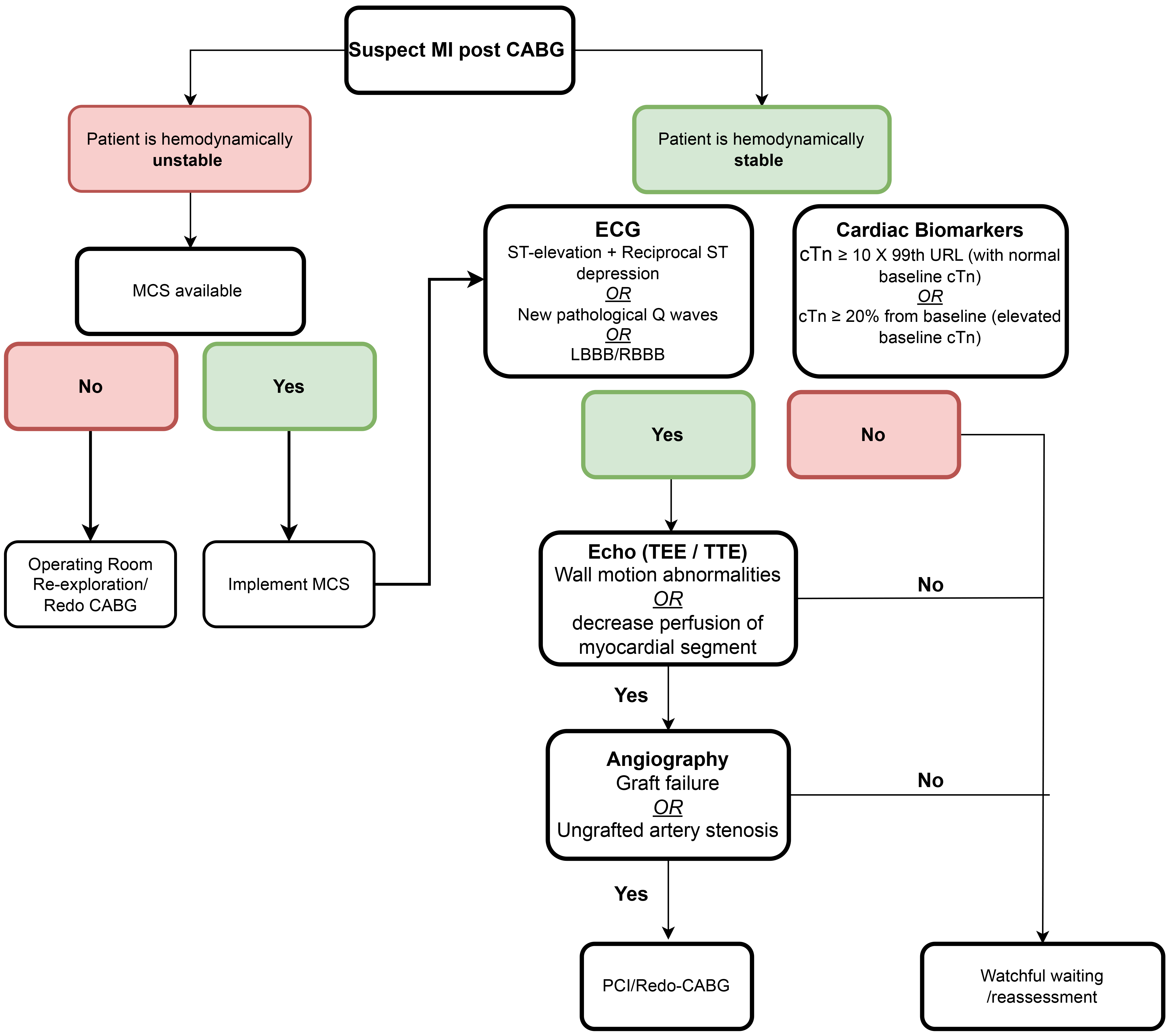 Fig. 4.
Fig. 4.
Proposed algorithm for management of postoperative myocardial ischemia. CABG, coronary artery bypass grafting; EKG, electrocardiography; MCS, mechanical circulatory support; LBBB, left bundle branch block.
The American Heart Association recommends life-long use of aspirin (81–325 mg daily) for patients post-CABG regardless of conduit used to reduce graft occlusion and adverse cardiac events [8]. For off-pump CABG or those with recent acute coronary syndrome, dual antiplatelet therapy with aspirin and clopidogrel for one year is advised to enhance graft patency [8]. CABG with endarterectomy or poor targets may benefit from dual antiplatelet therapy [68]. Arterial grafts are more prone to spasm due to their muscular nature when compared to vein grafts. Radial artery grafts have lower intrinsic nitric oxide synthase enzyme activity and may be more prone to spasm than internal thoracic artery grafts. Low-dose intravenous nitroglycerine or diltiazem is used postoperatively and is transitioned to oral long-acting dihydropyridine calcium channel blockers. These are continued for at least 3 months to maintain radial artery graft patency [8]. Statins improve long-term graft patency, lower risk of atherosclerosis, and improve survival, and are continued for life after CABG [8].
Postoperative atrial fibrillation (POAF) has an incidence of 20% to 30% after isolated CABG, and up to 60% with combined CABG and valve surgery [69]. POAF increases the risk of stroke (early and late) and mortality [70]. Risk factors for POAF include chronic obstructive pulmonary disease, advanced age, male sex, congestive heart failure, prolonged cross-clamp time, duration of mechanical ventilation, and inotrope use [71]. Prophylactic beta blockers or amiodarone are both associated with decreased incidence of POAF [72]. Current evidence does not support superiority of rhythm over rate control for POAF [73], but in our practice we favor rhythm control when possible (Fig. 5). POAF with rapid ventricular rate is treated with beta blockers, amiodarone, calcium channel blockers, digoxin, and cardioversion. POAF of more than 48 hours without systemic anticoagulation requires transesophageal echocardiography to rule out left atrial appendage clot prior to cardioversion [74]. The decision for subsequent systemic anticoagulation is nuanced as a large meta-analysis showed a decreased thromboembolism risk but a significantly increased risk of bleeding [75]. The PACeS trial is an ongoing Cardiothoracic Surgical Trial Network study designed to identify the optimal antithrombotic management in this population [76].
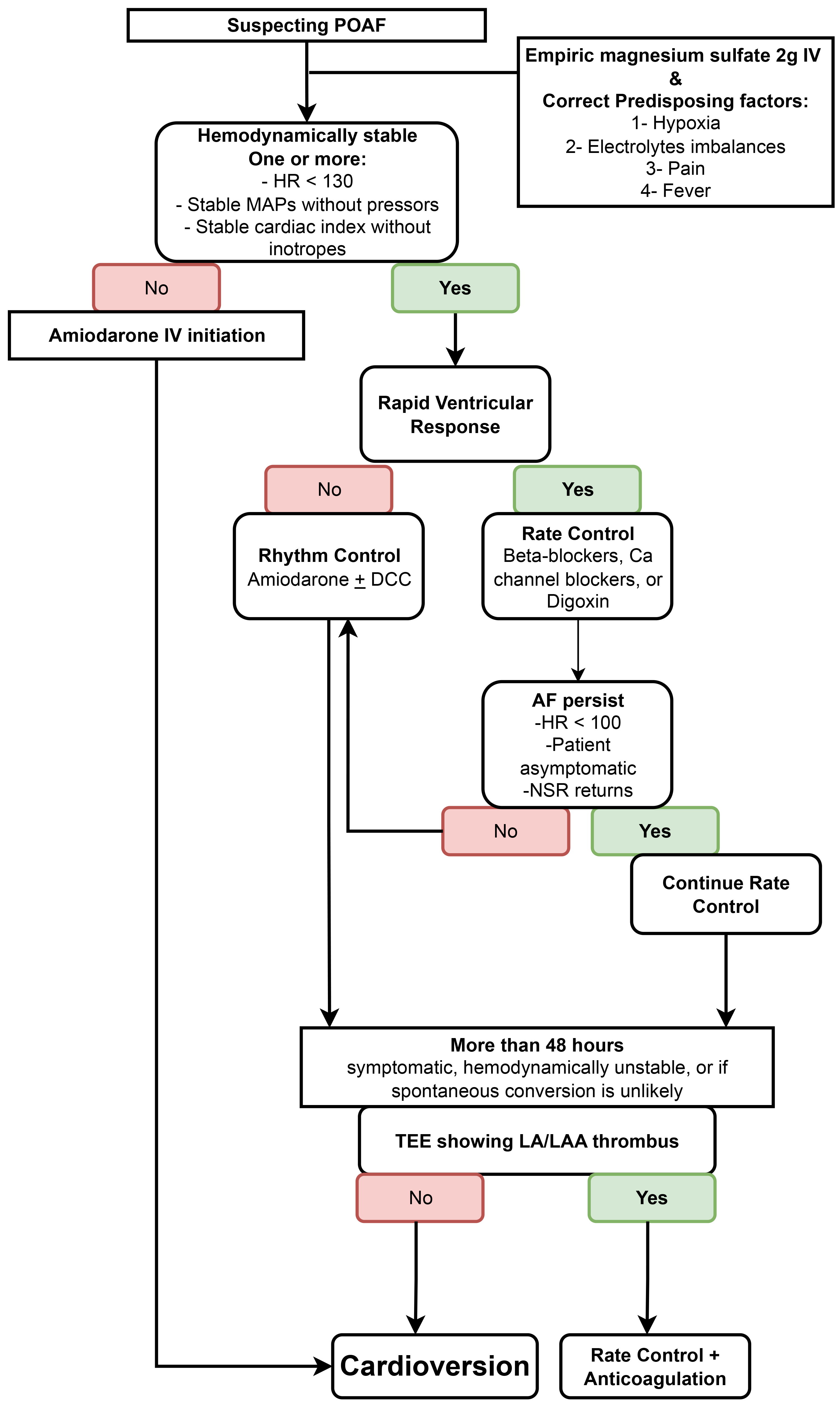 Fig. 5.
Fig. 5.
Proposed algorithm for postoperative atrial fibrillation arrythmia. POAF, postoperative atrial fibrillation; IV, intravenous; HR, heart rate; MAP, mean arterial pressure; DCC, direct current cardioversion; AF, atrial fibrillation; NSR, non-spontaneous rhythm; TEE, transesophageal echocardiogram; LA, left atrial; LAA, left atrial appendage.
Isolated premature ventricular contractions (PVCs) in patients with normal EF
typically are inconsequential. However, malperfused PVCs can impair CO and should
be treated aggressively in patients with LCOS. Short-coupled PVCs (distance
between preceding QRS and PVC peak
Temporary epicardial pacing wires (EPW) are routinely placed in on-pump CABG to manage transient bradyarrhythmias, particularly in patients with preexisting conduction abnormalities [22]. Persistent symptomatic bradycardia may necessitate permanent pacemaker (PPM) implantation [85]. Isolated CABG has the lowest PPM requirement (~1%) while combined valve/CABG procedures increase this risk [22]. PPM implantation is typically delayed 5 to 7 days postoperatively to allow for AV node conduction recovery [86]. Once stable rhythm has been noted for days without pacing requirements, EPW are removed despite the small risk of cardiac tamponade (incidence of 0.1%) [87]. The timing of wire removal should align with anticoagulation decisions to mitigate bleeding complications. If EPW cannot be pulled safely or there is high risk of bleeding such as when patient is on anticoagulation, they can be cut flush to the skin, with a small risk of including infection and lead migration [87].
Key points about arrythmias:
1. POAF increases stroke and mortality risk, and is prevented with beta-blockers or amiodarone.
2. Rhythm and rate control have similar outcomes, but rhythm control is often preferred in practice; anticoagulation decisions remain nuanced.
3. Ventricular arrhythmias (PVCs, VT/VF) are uncommon and management includes beta-blockers, antiarrhythmics (amiodarone, lidocaine), defibrillation for pulseless VT/VF, and consideration of ablation or ECMO for refractory cases.
4. Bradyarrhythmias are managed with temporary epicardial pacing wires, with permanent pacemaker required in ~1% after isolated CABG.
Multiple mechanisms may cause a decline in respiratory function postoperatively, including ischemic reperfusion injury, ventilator-associated lung injury, transfusion-associated lung injury, cardiogenic pulmonary edema, diaphragmatic dysfunction from phrenic nerve injury, atelectasis, pneumonia, pleural effusions, pneumothorax, and pulmonary embolism [88]. Treatment includes optimizing pain control, minimizing duration of mechanical ventilation, promoting physical activity and bronchopulmonary hygiene, and treating specific pulmonary pathology. Our routine practice is to get patients out of bed and into the chair by the morning of postoperative day one with initiation of physical and occupational therapy in the ICU. Moreover, patients are expected to perform hourly positive pressure airway exercises while in bed. Regarding patients with chronic obstructive pulmonary disease, preoperative intensive inspiratory muscle training is proven to improve pulmonary mechanics thus reducing incidence of pulmonary complications and the need for reintubation as well as noninvasive positive pressure ventilation.
Cardiac surgery associated acute kidney injury affects 5% to 10% of patients after cardiac surgery [89]. In a large, retrospective study of patients undergoing cardiac surgery, postoperative dialysis initiation ranged from 1.1% after isolated CABG to 5.1% for CABG plus mitral valve surgery [90]. Advanced age, preexisting renal disease, peripheral arterial disease, diabetes mellitus, increase body weight, prolonged cardiopulmonary bypass, re-operation, nature and complexity of cardiac surgery, perioperative transfusion requirements, perioperative hypotension, low cardiac index, and high CVP are known risk factors for development of cardiac surgery associated acute kidney injury [89, 91]. Based on these risk factors, maintaining optimum cardiac index, high MAP, and low CVP is recommended for adequate renal perfusion. This may require active diuresis despite renal injury as it may relieve renal congestion and improve renal perfusion.
Intraoperative hyperglycemia is an independent predictor of mortality, both for
diabetic and non-diabetic patients [92]. Postoperative serum glucose levels
The American Heart Association [8] recommends a controlled dietary intake post-CABG, emphasizing a balanced diet with 30 kcal/kg, comprising 15% to 20% protein, 30% fat, and 50% to 55% carbohydrates (rich in fruits and vegetables). Postoperative gastrointestinal complications after cardiac surgery include ileus, nausea, vomiting, gastrointestinal bleeding, pancreatitis, bowel obstruction, Clostridium difficile colitis, acute cholecystitis, postcardiotomy liver dysfunction, and acute mesenteric ischemia, with ileus being the most common postoperative gastrointestinal complication. Postoperative ileus management relies on minimizing opioids, early mobilization, and laxatives. Enteral naloxone gel or subcutaneous methylnaltrexone may be beneficial for opioid-induced constipation. For massive colon distension, we follow an aggressive strategy to avoid ischemic complications and resort to decompressive colonoscopy along with neostigmine drip as needed.
Despite occurring in only 1% of post-CABG patients, acute mesenteric ischemia is one of the devastating post-cardiac surgery complications with mortality reaching 67% to 94% [97] and is usually non-vaso-occlusive. Risk factors for acute mesenteric ischemia include advanced age, cardiogenic shock, atrial fibrillation, need for intra-aortic balloon pump, reoperation, emergency surgery, blood transfusions, and prolonged mechanical ventilation [97]. Abdominal pain, tenderness, distention, hematochezia, radiographic ileus on X-ray, and unexplained lactic acid elevation can point towards the diagnosis. In hemodynamically tenuous patients who cannot obtain a computed tomography scan, bedside sigmoidoscopy or colonoscopy can aid in diagnosis. Patients with acute mesenteric ischemia with bowel infarction require bowel resection and prolonged parenteral nutrition and appropriate antibiotic coverage to target peritonitis and gut translocation (Fig. 6).
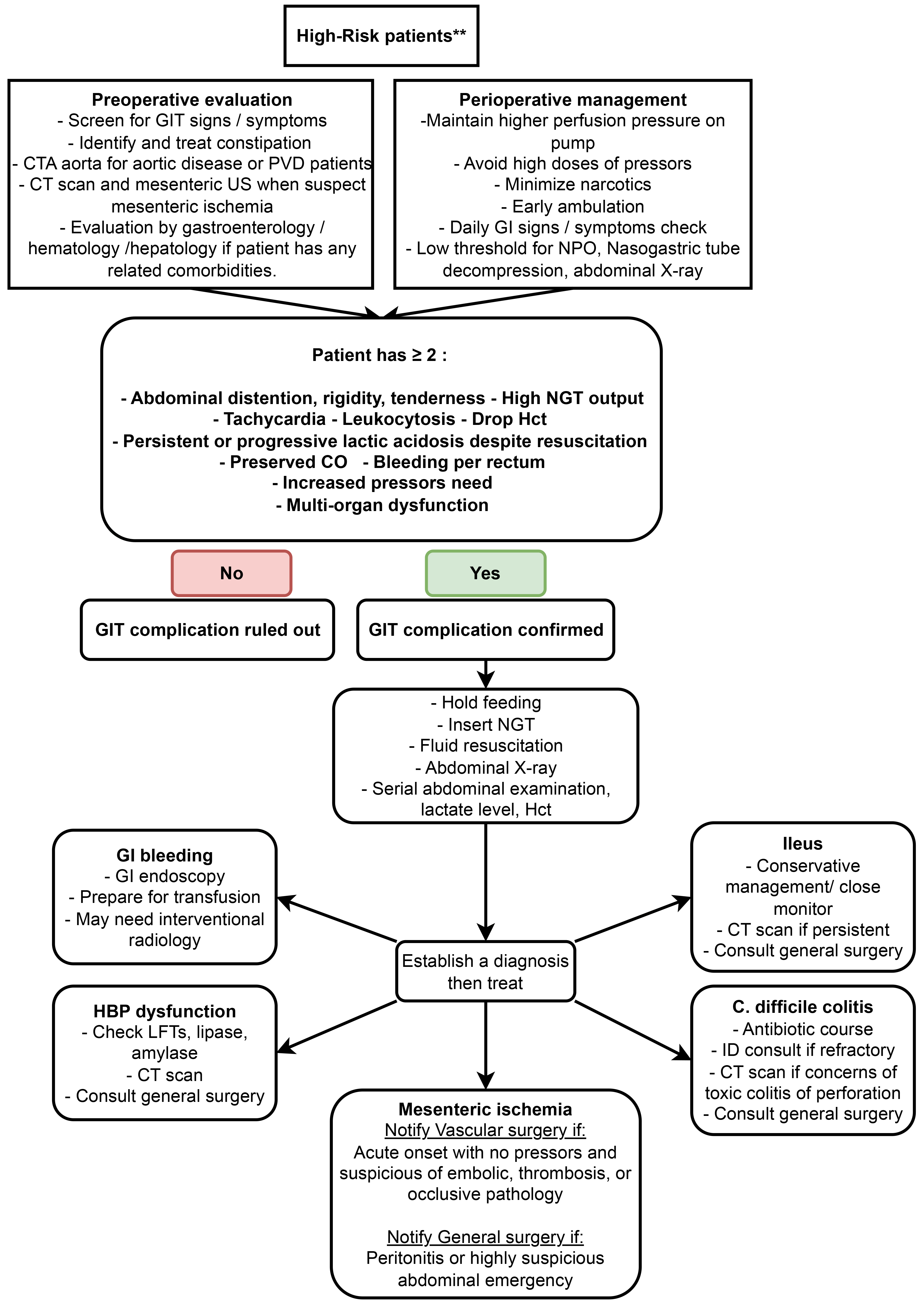 Fig. 6.
Fig. 6.
Proposed algorithm for suspecting and handling gastrointestinal complications. GI, gastrointestinal; GIT, gastrointestinal tract; CT, computed tomography; CTA, CT-angiography; PVD, peripheral vascular disease; US, ultrasound; NPO, nothing per os; NGT, nasogastric tube; CO, cardiac output; Hct, hematocrit; HBP, high blood pressure; LFT, liver function tests; ID, infectious diseases. **High-risk patients: advanced age, coronary artery disease, peripheral vascular disease, aortic disease, heart failure with low ejection fraction, history of abdominal surgery, liver disease, coagulation disorder, emergency surgery, preoperative pressors, or mechanical circulatory support.
Surgical site infections of the sternal wound and mediastinum affect
approximately 0.7% of isolated CABG procedures (range 0.4–4% for all cardiac
surgeries) [98]. These deep sternal infections are associated with
Despite the global burden of coronary artery disease, access to CABG remains highly inequitable, with low-to-medium income countries facing significant shortages in cardiac surgical capacity and resources [105]. In many of those countries, optimized medical therapy remains the mainstay for managing LCOS postoperatively [106]. When advanced MCS is not available, intra-aortic balloon pump is the simplest and most widely accessible device for temporary support in high-risk or hemodynamically unstable patients, as supported by the American Association for Thoracic Surgery [106]. Off-pump CABG is another resource-conserving strategy with comparable outcomes with experienced teams [107]. The ERAS® Society protocols have been shown to be adaptable to low-to-medium income countries, improving improve outcomes and reducing costs [107].
Global resource concerns and high rates of unplanned re-admissions after CABG has carved a niche for digital health revolution, including in low-to-medium income countries. Digital health holds promise of improving patient outcomes, engagement, and reducing healthcare resource utilization through [108, 109]:
1. Enhanced communication and education: Digital health tools, such as mobile applications and chatbots, facilitate continuous communication between patients and their care teams, empowering patients. These platforms can provide medication reminders and cardiac health education, making patients more active in their care. Video-based patient education and consultations have also been shown to reduce unplanned healthcare utilization and aid in faster recovery.
2. Remote monitoring and early detection of complications: Telehealth visits and digital health tools are effective for postoperative screening and early identification of complications. Chatbots can collect care assessment data related to common causes of readmission after CABG, allowing proactive intervention by the care team.
3. Support for cardiac rehabilitation: Digital health tools can promote cardiac rehabilitation, even serving as an alternative or adjunct to traditional center-based rehab. Studies have shown that patients using digital health tools have better cardiovascular fitness and exercise habits, and combining digital health with center-based rehab can lead to lower healthcare utilization and better patient outcomes.
4. Improved medication adherence: Digital health tools can significantly improve medication adherence, which is vital for transplant patients and equally important for CABG patients to prevent complications.
5. Reduced healthcare costs and readmissions: By improving patient engagement, facilitating remote monitoring, and supporting rehabilitation, Digital health tools can reduce healthcare utilization, including emergency department visits and hospitalizations with associated lower healthcare costs.
6. Increased accessibility and patient satisfaction: Given that much of the world’s population now owns a Smartphone. Digital health tools are easily accessible and often cost-negligible for consumers. They can also improve patient satisfaction and provide a more seamless transition from hospital to home.
Artificial intelligence and machine-learning algorithms, such as Extreme Gradient Boosting (XGBoost) and neural networks, have been developed to predict postoperative outcomes, such as mortality, major morbidity, complications, and costs, and exhibit a high degree of accuracy [110]. Artificial intelligence is increasingly integrated into post-CABG care, with real-world examples including the Epic Sepsis Model for early detection of sepsis in the ICU and machine-learning-enhanced CABG risk calculators, such as the STS and EuroSCORE systems, which support perioperative risk stratification and outcome prediction [111]. The American Heart Association emphasizes that while predictive analytics (e.g., risk calculators and early warning systems) are already in clinical use, prescriptive analytics—such as reinforcement learning models for dynamic anesthetic or hemodynamic management—remain investigational and are not yet standard of care [110]. Key challenges include data bias (which can perpetuate health disparities if training data are not representative) and lack of algorithm transparency (which limits clinical interpretability).
1. Delirium prevention: Implement non-pharmacologic measures (mobilization, sleep hygiene) and avoid benzodiazepines; consider dexmedetomidine for sedation.
2. Pain management: Use multimodal analgesia, including regional blocks, to reduce pain and opioid side effects.
3. Maintain high suspicion for stroke with serial neurologic assessments due to high mortality risk.
4. Pursue endovascular thrombectomy for large-vessel occlusion; parenteral thrombolytics are contraindicated postoperatively.
5. Treat vasoplegia with goal-directed volume resuscitation and conventional vasopressors; consider advanced agents for refractory cases.
6. MAP targets: Aim for MAP 70–80 mmHg in atherosclerotic patients; consider 65–70 mmHg for RV failure, adjusting for CVP.
7. Integrate pulmonary artery catheter parameters (TDCI, eFCI, CVP, PCWP) for LCOS management.
8. Inotrope choice: Use epinephrine, dobutamine, or milrinone; avoid milrinone in profound vasoplegia or renal dysfunction.
9. Initiate MCS early for refractory LCOS to prevent end-organ damage, ideally pre- or intraoperatively.
10. Choose MCS (IABP, Impella, VA-ECMO) based on specific hemodynamic needs and LV/RV support requirements.
11. Modify advanced cardiovascular life support: Delay external CPR briefly for early defibrillation/pacing; avoid over-aggressive compressions.
12. Consider surgical re-exploration for high chest-tube output.
13. The roles of innovations such as ERAS-cardiac, digital health, and artificial intelligence are promising.
Postoperative care after CABG is an intricate and interdisciplinary process that involves meticulous coordination across specialties to attain the best long-term outcome for the patient. This review highlights the essential elements of postoperative management, ranging from early ICU management to later follow-up by the heart team. Advancements in perioperative care, ERAS guidelines, digital health solutions, and artificial intelligence hold great potential in streamlining the delivery of care, enhancing patient engagement in the recovery process, and optimizing outcomes. The integration of these emerging tools into routine practice holds the potential to revolutionize the care of CABG patients, making recovery faster, safer, and more personalized.
AKA, SA, AT, MA, MJK, HE, MZYT, CD, SU, EGS, AMA, MAG, FGB, and JD contributed to the design of the work. MJK, HE, MZYT, SU, EGS, AMA, MAG, CD, JD, and FGB contributed to writing the first draft. AKA, SA, MA, SU, EGS, AMA, MAG, FGB, and AT finalized the last draft. FGB supervised the whole process. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors thank Ingrid Schaefer Sprague, MBA, Miller Family Heart, Vascular & Thoracic Institute, Cleveland Clinic, for her editorial assistance.
This study was funded in part by the Sheikh Hamdan bin Rashid Al Maktoum Distinguished Chair in Thoracic and Cardiovascular Surgery (FGB).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






