1 Philadelphia College of Osteopathic Medicine, Philadelphia, PA 19131, USA
2 Department of Cardiothoracic Surgery, Ascension Saint Thomas, Nashville, TN 37205, USA
3 Cardiothoracic Surgery, St Alphonsus Regional Medical Center, Boise, ID 83706, USA
4 WakeMed Clinical Research Institute, WakeMed, Raleigh, NC 27610, USA
5 Department of Cardiovascular and Thoracic Surgery, WakeMed Heart and Vascular, Raleigh, NC 27610, USA
Abstract
Postoperative atrial fibrillation (POAF) is a common complication following cardiac surgery and is associated with high rates of morbidity. Oral or intravenous administration of amiodarone is commonly used for treatment and prophylaxis; however, systemic delivery of amiodarone is associated with significant, well-known extracardiac side effects. Therefore, this study aimed to evaluate the effectiveness of combining a pericardial reconstruction matrix (CardiaMend) with amiodarone to prevent POAF in patients undergoing isolated coronary artery bypass grafting (CABG) or valve surgery.
This was a single-center, prospective, pilot study that enrolled patients undergoing CABG or valve surgery via complete median sternotomy. The amiodarone-soaked CardiaMend patch was applied intraoperatively. The primary outcome was the incidence of POAF up to patient discharge. Secondary outcomes were time until discharge and burden of atrial fibrillation.
A total of 29 patients were included, all undergoing CABG, of whom 10% (3/29) had concomitant valve replacements. POAF was experienced by 10 patients (34%). All patients were discharged in sinus rhythm, and none required a wearable rhythm monitor.
The use of a pericardial reconstruction matrix for local amiodarone delivery during cardiac surgery is a safe, potential prophylactic treatment for POAF. The clinical results of this pilot study showed a trend toward reduced POAF following cardiac surgery. These results, in combination with prior research, suggest that a more targeted application of amiodarone-eluting patches could be used to treat the atria and further improve outcomes.
NCT05681182, https://clinicaltrials.gov/study/NCT05681182.
Keywords
- amiodarone
- pericardial reconstruction
- postoperative atrial fibrillation
- CABG
- atrial fibrillation prophylaxis
- reconstruction matrix
Open-heart cardiovascular procedures, such as coronary artery bypass grafting (CABG), aortic or mitral valve replacement/repair, or implant procedures with ventricular assist devices (VADs), are associated with a high rate of postoperative atrial fibrillation (POAF). Indeed, 20–50% of these patients develop POAF, depending on the exact procedure(s) and patient comorbidities, which can potentially lead to further complications, including an increased risk of perioperative strokes and hemodynamic instability requiring cardioversion [1].
The etiology of POAF is multifactorial and involves atrial inflammation from retained blood products following sternotomy, as well as atrial stretch from edema and heart manipulation [2]. Vagal afferent denervation from cutting the aortic fat pad has also been implicated [3]. The initial onset of POAF occurs in a nonlinear fashion, with a peak incidence 2 to 3 days after surgery, followed by a subsequent decrease over time [1, 4]. POAF is associated with an increased risk of stroke, increased length of stay, and a 10-year increased risk of cardiovascular and all-cause mortality [1, 5].
Beta blocker use is the only class I medication treatment recommendation for preventing POAF [3]. Amiodarone, a class III antiarrhythmic drug, is currently approved for oral or IV administration to treat arrhythmias and is commonly considered the most effective drug for the prophylaxis and treatment of AF. However, due to the significant extracardiac side effects associated with systemic administration, amiodarone remains a second-line agent for POAF. Indeed, systemic administration of amiodarone can result in significant side effects, including bradycardia, skin reactions, visual disturbance, pulmonary toxicity, thyroid dysfunction, and liver dysfunction [6], and carries a risk for drug interactions [7]. Serious lung problems and liver problems that may result from using amiodarone are specifically listed as black box warnings in the food and drug administration (FDA)-approved drug labeling.
The American Association for Thoracic Surgery 2014 guidelines state that administering postoperative amiodarone is reasonable to reduce the incidence of POAF in patients undergoing general thoracic surgery [8]. The 2006 American College of Cardiology/American Heart Association Task Force on practice guidelines includes a class IIa recommendation, due to conflicting evidence or divergence of opinion, for preoperative amiodarone in patients at high risk of POAF [9].
Amiodarone has been investigated for local topical administration in numerous preclinical and clinical studies, in which the drug was delivered via various mechanisms, including direct infusion and a patch [10]. These efforts were undertaken to minimize systemic side effects and reduce the need for a drug loading period. These studies have demonstrated that local administration of amiodarone is safe and that the pharmacological mode of action is the same [10].
CardiaMend, an FDA-cleared pericardial reconstruction matrix, has been used as a physical patch to repair and reconstruct the pericardium following cardiac surgery; such repair with a soft-tissue device has been shown to reduce postoperative complications. A recent multicenter, prospective clinical study by Rego et al. [2] of 1420 patients undergoing open cardiac surgery with device-mediated pericardial closure found statistically significant decreases in pleural and pericardial effusions, bleeding, and all-cause hospital readmissions compared with control data from a national registry. In this large study, POAF was 14.4%, compared with historic rates reported to range from 20% to 50% [1, 2, 5].
While primarily used for pericardial repair, CardiaMend has also demonstrated the ability to absorb fluid, serving as a short-term reservoir for local delivery of amiodarone hydrochloride to help reduce the incidence of POAF. This result is supported by current preclinical data from Helios Cardio Inc., which show therapeutic levels of amiodarone in bovine heart tissue 3 days after surgery following pericardial repair with amiodarone-soaked CardiaMend, with very low levels in the liver and blood.
Therefore, this study aimed to evaluate the combination of CardiaMend and amiodarone for the prevention of POAF in patients undergoing isolated CABG with or without concomitant valve surgery.
This is a single-center, prospective, investigator-initiated pilot study that enrolled patients undergoing CABG or valve surgery via complete median sternotomy between January 2023 and January 2024 (NCT05681182, 20 January 2023, WakeMed Health and Hospitals).
Patients aged 20 to 85 years were eligible to participate. Participants had to
be scheduled to undergo open-chest coronary surgery via complete sternotomy,
including CABG with or without concomitant aortic valve repair or replacement, or
mitral valve repair or replacement. Patients had to be in sinus rhythm before
surgery. Patients who were pregnant, had a history of chronic wounds or wound
healing disorders, had known connective tissue diseases, were immunosuppressed,
had a history of atrial fibrillation (treated or untreated), had implantable
cardiac devices, had end-stage chronic renal disease, had heart failure, or had
an society of thoracic surgeons (STS) risk
Posterior pericardiotomy was not routinely performed, although all patients had
a left lateral pericardial opening for placement of the left internal mammary artery (LIMA) graft. Postoperative
care was provided through a standardized enhanced recovery after surgery (ERAS)
cardiac pathway [11]. This included goal-directed therapy for postoperative fluid
management, magnesium and potassium replacement protocols (
The CardiaMend patch was soaked in an amiodarone solution before use, instead of in saline, and was applied to the patient to deliver an amiodarone dose of 450 mg into the pericardial cavity (Fig. 1). The patch was then fixed in place using a continuous stitch with a non-absorbable suture to approximate the edge of the pericardial defect (Fig. 2). A continuous electrocardiogram (ECG) was performed for each patient until discharge. If clinically indicated, patients received a home-monitoring device in accordance with the standard of care.
 Fig. 1.
Fig. 1.
The CardiaMend patch prior to being soaked in amiodarone solution.
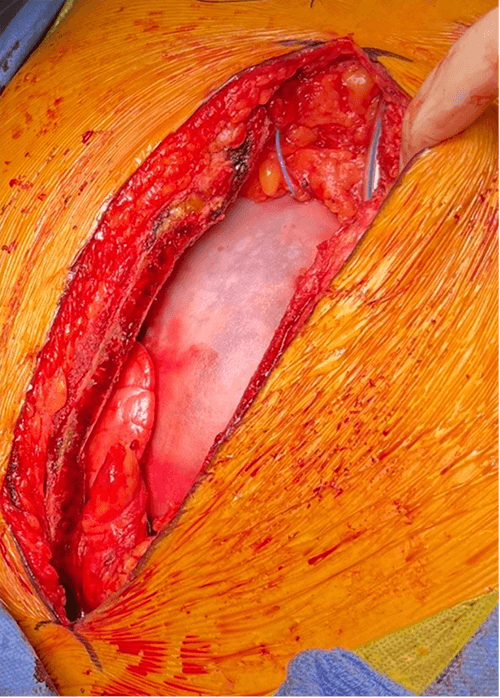 Fig. 2.
Fig. 2.
The pericardial patch soaked in amiodarone and sutured in place.
The primary outcome was the incidence of POAF up to patient discharge, defined as the patient experiencing atrial fibrillation or atrial flutter lasting longer than 1 hour or less than 1 hour that required medical or procedural intervention after exiting the operating room. Secondary outcomes were STS metrics of 30-day hospital readmission, death, stroke, deep sternal wound infection, renal failure, prolonged ventilation, and reoperation for bleeding.
Continuous variables are presented as the mean (mean
A total of 30 patients were enrolled in the study, and 29 met the inclusion criteria. One consented and enrolled patient experienced atrial fibrillation in the operating room before incision, meaning this patient was excluded from the study. While this patient received the amiodarone-soaked patch, as discussed in the preoperative consultation for the operation and study participation, and did not experience POAF, the patient was excluded from the analysis because the exclusion criteria were met prior to surgical treatment (Fig. 3).
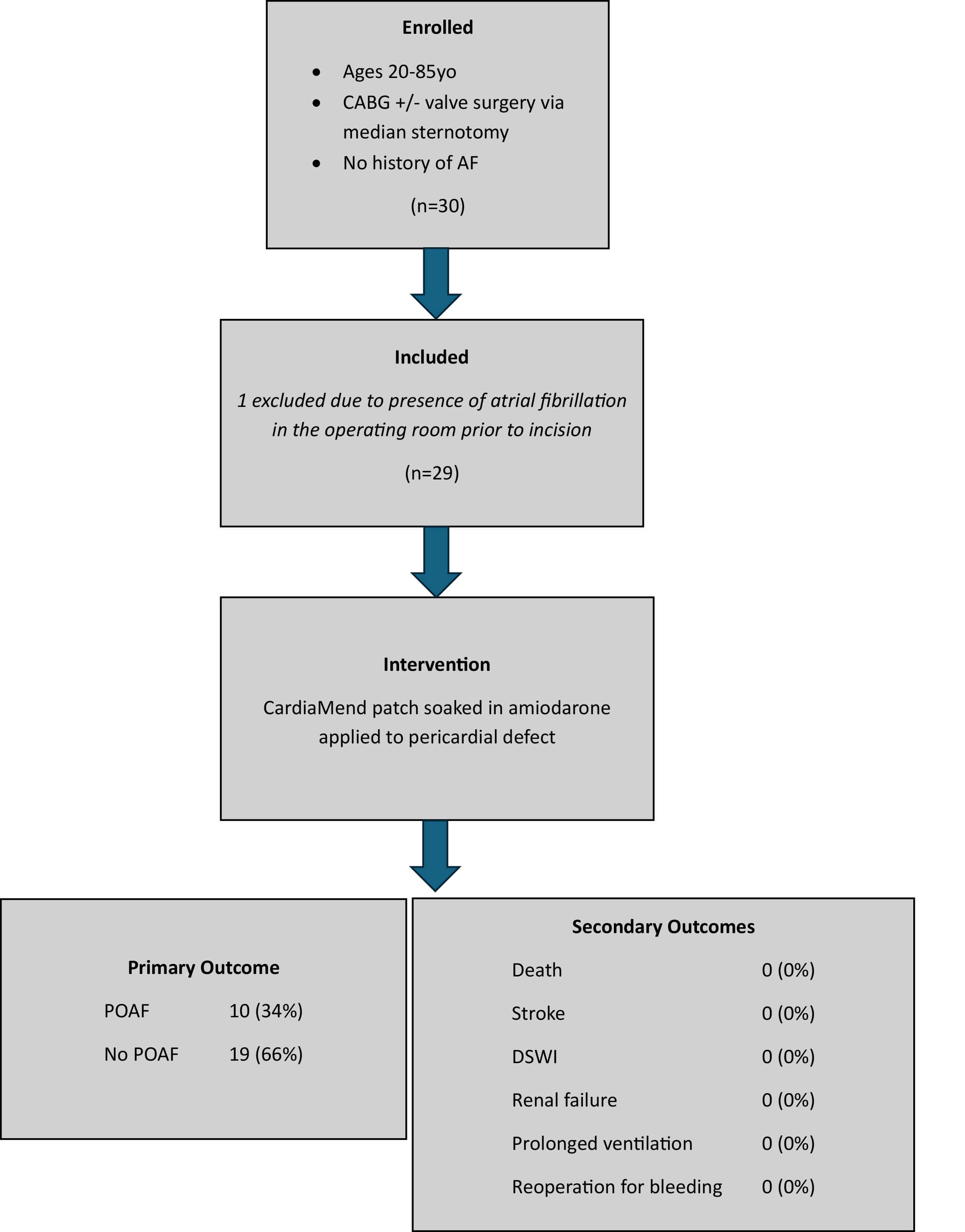 Fig. 3.
Fig. 3.
Diagram of trial enrollment and inclusion with outcomes. CABG, coronary artery bypass grafting; AF, atrial fibrillation; POAF, postoperative atrial fibrillation; DSWI, deep sternal wound infection.
The median age of the included patients was 69 years (range, 45 to 81; IQR, 17.5). The study cohort comprised 14% females (4/29) and 86% males (25/29). Races included Black (7%, 2/29), White (86%, 25/29), Asian (3%, 1/29), and American Indian (3%, 1/29); Hispanics comprised 7% (2/29). Operative intervention included CABG in all patients, with 10% (3/29) undergoing concomitant aortic valve replacement and 3% (1/29) undergoing concomitant mitral valve replacement. The median number of bypass grafts was 3 (range, 2 to 6; Table 1).
| Characteristic | n | % | |
| Total patients | 29 | ||
| Gender | |||
| Male | 25 | 86.21 | |
| Female | 4 | 13.79 | |
| Race | |||
| Black | 2 | 6.90 | |
| White | 25 | 86.21 | |
| Asian | 1 | 3.45 | |
| American Indian | 1 | 3.45 | |
| Ethnicity | |||
| Hispanic | 2 | 6.90 | |
| Non-Hispanic | 27 | 93.10 | |
| Age | |||
| Range | 41–81 | ||
| Median (IQR) | 69 (17.5) | ||
| Procedure | |||
| CABG | 26 | 89.66 | |
| CABG + AV | 2 | 6.90 | |
| CABG + AV + MV | 1 | 3.45 | |
CABG, coronary artery bypass graft; AV, aortic valve; MV, mitral valve.
The median hospital length of stay was 7 days (range, 4 to 20 days). POAF occurred in 10 patients (34%). All patients were in sinus rhythm at hospital discharge; thus, no patients were discharged home with a wearable rhythm monitor or with anticoagulation medication. All patients were enrolled in a TeleHeartCare monitoring program that included blood pressure, scales, and pulse oximeter monitors [12]. Moreover, no patient was found to have atrial fibrillation, new or recurrent from hospital discharge through the time of postoperative surgical follow-up (between 4 and 6 weeks postoperatively).
Of the 10 patients who experienced POAF, the average duration of atrial fibrillation was 3.5 hours, ranging from 1 to 13 hours. All received a medication intervention, with nine having additional oral (400 mg) or intravenous (IV) (150 mg) amiodarone doses, and one having increased metoprolol dosing. The total postoperative chest tube output through 48 hours was 886 mL (range, 490–2092 mL).
Local administration, by providing short-term local delivery, can potentially improve safety and efficacy compared with systemic administration of amiodarone, achieving an effective localized dose in heart tissue while reducing the drug dosage delivered to the patient. The observed POAF rate of 31% in a comorbid CABG/valve cohort compares favorably with contemporaneous national database results in similar patients with POAF rates of 30–50% in high-quality reporting centers [13]. Among the 29 included patients, there was zero incidence of hospital readmission, death, stroke, deep sternal would incision (DSWI), renal failure, prolonged ventilation, reoperation for bleeding, or presence of atrial fibrillation at the 30-day follow-up.
A small number of patients (n = 2) had pericardial patches trimmed to achieve an optimal anatomic fit for pericardial closure, which would have slightly reduced the delivered amiodarone dose. The amount trimmed was small in size; however, this was not precisely quantified, which remains a limitation. Notably, the patch only touches the right atrium when used for pericardial closure, and atrial fibrillation mostly originates from the left atrium. Furthermore, this is a relatively small study cohort, largely male and lacking diversity. However, the included participants spanned a wide age range. Every patient included in this study underwent surgery by the same surgeon, further homogenizing the intraoperative methods and strengthening the study. The same patch was used for fixation in every patient using the suture technique.
It has been suggested that a more targeted application of amiodarone-eluting patches to the atria could further improve outcomes [14]. The CardiaMend patch, or any other pericardial reconstruction matrix, could be studied in combination with other medications, such as anti-inflammatories, analgesics, or other antiarrhythmics; meanwhile, longer-term follow-up periods should be assessed. The liquid-absorption capacity of the CardiaMend patch enabled it to serve as a short-term reservoir for amiodarone. While we did not achieve a large reduction in POAF, this pilot study showed the amiodarone-soaked patch to be clinically safe and demonstrated a trend toward reduction, which we aim to study further.
The use of a pericardial reconstruction matrix for local amiodarone delivery during cardiac surgery is a safe, potential prophylactic treatment for POAF. The clinical results of this pilot study showed a trend toward reduced POAF following cardiac surgery.
Data to support the findings of this study are available on reasonable requests from the corresponding author.
JBW, RN, CK, and RSF designed the research trial. JBW and RN performed the clinical research. CK and RSF assisted in the clinical research conducted. All authors analyzed and interpreted the data. MD drafted the manuscript. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of WakeMed under protocol name CAMP POAF (package 1937514). Patients who met the inclusion criteria were given an opportunity to consider participating in the study. Patients provided written informed consent after being informed of the study and given an opportunity to ask questions. The study was registered with clinicaltrials.gov as CAMP POAF. The NCT identifier is NCT05681182.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Clayton Kaiser has a financial relationship with Helios Medical. However, the company had no role in the handling or conduct of the study. The authors had full access to all data in the study and take full responsibility for the integrity of the data and the accuracy of the data analysis.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF50004.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



