1 Department of Cardiovascular Surgery, The First Medical Center of Chinese PLA General Hospital, 100853 Beijing, China
2 Department of Thoracic Surgery, The Third Medical Center of Chinese PLA General Hospital, 100039 Beijing, China
†These authors contributed equally.
Abstract
This study aimed to evaluate the safety and efficacy of total endoscopic removal of patent foramen ovale (PFO) or atrial septal defect (ASD) occluder devices in managing nickel hypersensitivity complications.
A retrospective analysis of 95 patients (2020–2025) undergoing total endoscopic occluder device removal via femoral cardiopulmonary bypass was performed using preoperative nickel allergy screening via patch testing. Outcomes included procedural success, symptom resolution, quality-of-life (QoL) trends, and complications.
All devices were removed successfully without thoracotomy. The median bypass time was 71.2 min; 96.8% of residual defects were directly sutured. Nickel hypersensitivity was confirmed in 89.5% of cases. QoL “good” ratings increased from 7.4% preoperatively to 95.8% at 6 months (p < 0.001). No major complications were observed; however, there were two cases of transient atrial fibrillation (2.1%). The median blood loss was 36.8 mL; no reoperations/mortality were noted.
Total endoscopic removal is safe and effective for nickel allergy-related complications, with high symptom resolution and improvement in QoL. Preoperative nickel screening optimizes outcomes, while this minimally invasive approach reduces morbidity, thereby supporting the adoption of this approach for device explantation.
Keywords
- patent foramen ovale
- atrial septal defect
- nickel allergy
- endoscopic device removal
- cardiac surgery
- residual septal defects
Patent foramen ovale (PFO) occluder devices are commonly used in clinical practice to prevent recurrent cryptogenic strokes and treat migraines [1, 2]. These devices, which are often composed of nickel-containing alloys, have demonstrated long-term efficacy in reducing the risk of thromboembolic events [3]. However, complications related to device implantation, such as device migration, incomplete endothelialization, and nickel hypersensitivity, have been increasingly reported [4, 5]. Nickel allergy, in particular, is a significant concern due to the widespread use of nickel in medical devices. The prevalence of nickel allergy in the general population is high, yet its incidence following PFO or atrial septal defect (ASD) occluder is not well documented [6]. Despite these challenges, the exact pathophysiology behind nickel hypersensitivity in intracardiac devices remains unclear, prompting further investigation into the role of nickel allergy in device-related complications [7, 8].
Nickel hypersensitivity reactions can manifest with a broad range of symptoms,
including chest pain, migraines, palpitations, and dyspnea, often appearing weeks
to years after device implantation. In patients with pre-existing nickel
sensitivity, the body’s immune response can lead to chronic inflammation, which
can result in persistent symptoms that do not resolve with conventional medical
management [9]. Nickel ions (Ni2+) leached from occluders act as haptens
that bind to host proteins (e.g., toll-like receptor 4/TLR4), triggering innate
immune activation via the NF-
Despite the growing awareness of nickel hypersensitivity in patients with implanted PFO/ASD occluder devices, there are no standardized guidelines for preoperative allergy screening or the management of nickel-related complications. Current diagnostic approaches rely on skin patch testing, which can identify patients at risk of developing hypersensitivity reactions before or after device implantation [15]. Additionally, the decision to remove the device often depends on the severity of symptoms and the patient’s overall quality of life (QoL) post-implantation. The present study aims to retrospectively analyze the outcomes of patients who underwent totally endoscopic removal of PFO/ASD occluder devices due to nickel hypersensitivity, focusing on symptom resolution and the safety of the minimally invasive surgical approach.
This retrospective study included 95 patients who underwent totally endoscopic
removal of PFO/ASD occluder devices at our center between 2020 and 2025.
Consecutive patients referred for endoscopic device explantation were evaluated.
Device removal was considered only after documented failure of a standardized
medical optimization protocol [16, 17, 18]. Inclusion required: (1)
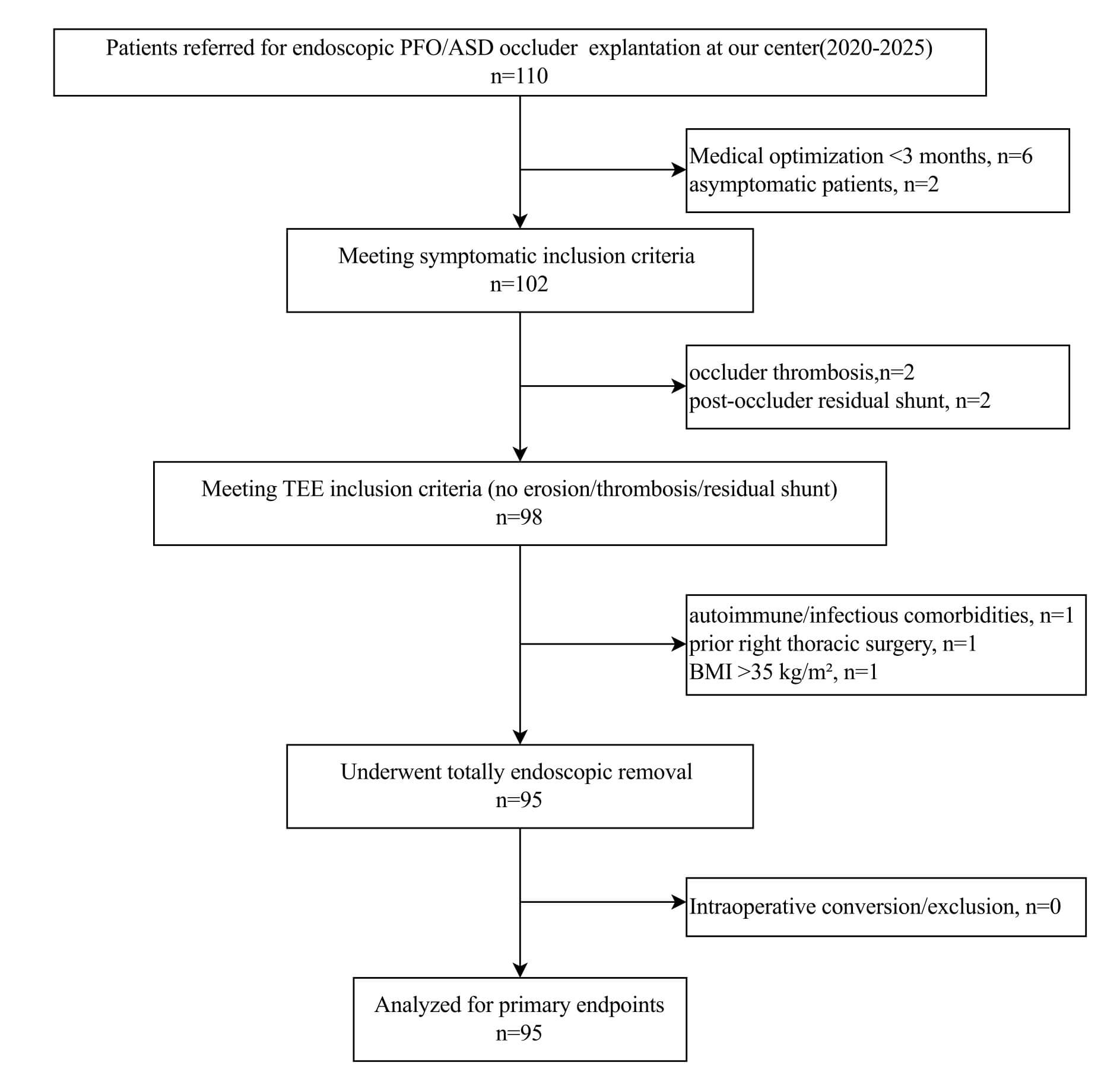 Fig. 1.
Fig. 1.
Patient enrollment flow chart. This diagram illustrates the screening process for patients undergoing totally endoscopic PFO/ASD closure device removal (2020–2025). Of 110 initially assessed patients, 15 were excluded due to: insufficient medical optimization (n = 5), asymptomatic status (n = 3), device thrombosis (n = 2), significant residual shunt (n = 2), or contraindications to endoscopic surgery (n = 3). The final cohort consisted of 95 patients who underwent successful endoscopic explantation. TEE, transesophageal echocardiography.
Comprehensive transthoracic echocardiography (TTE) and transesophageal
echocardiography (TEE) were performed preoperatively and 48 h postoperatively
following the guidelines of the American Society of Echocardiography (ASE). Key
parameters included: (1) Septal morphology: Defect size, rim thickness
(aortic/atrial/caval), septal aneurysm (excursion
The patient was placed in a supine position with the right side elevated.
Following the induction of general anesthesia and endotracheal intubation,
invasive monitoring was established via left radial arterial and venous
cannulation. Systemic heparinization was achieved. Cardiopulmonary bypass (CPB)
was established through cannulation of the right femoral artery and vein. The
femoral venous cannula was advanced to the orifice of the inferior vena cava
(IVC), and the superior vena cava (SVC) was percutaneously cannulated via the
right supraclavicular approach. The 3 cm main operating port was created in the
4th intercostal space just lateral to the right midclavicular line. The
thoracoscope was introduced through a port in the 4th intercostal space at the
right anterior axillary line. A transthoracic Chitwood clamp was introduced
through 5th intercostal space at the midclavicular line to cross-clamp the
ascending aorta and cardioplegic arrest was successfully induced. The SVC was
temporarily occluded with an occlusion clamp and ensure satisfactory drainage
with central venous pressure below 15 mmHg. The right atrium was incised and the
previously implanted occluder was identified and meticulously dissected and
completely removed. The iatrogenic atrial septal defect created by the occluder
was repaired with no residual shunt confirmed by testing. After thorough
de-airing, the aortic cross-clamp was released. The patient was weaned from CPB,
heparin reversed, hemostasis achieved. A right thoracic drainage tube was placed
and the thoracic incisions were closed in layers. Based on preoperative contrast
echocardiography, intraoperative re-evaluation was performed to confirm the
device condition, followed by complete removal and assessment of
endothelialization status using a standardized visual scale: Complete (smooth
tissue coverage, no visible metal), Partial (patchy coverage with
Intraoperative video of totally endoscopic intracardiac patent foramen ovale (PFO)/atrial septal defect (ASD) occluder device removal. Video associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF49911.
Data on patient demographics, clinical symptoms, nickel allergy testing results,
surgical details, and postoperative outcomes were collected from medical records.
QoL was assessed preoperatively and at follow-up intervals (3 days, 1 month, and
6 months postoperatively). Preoperative nickel allergy screening via patch
testing (TRUE Test™, 5% nickel sulfate) and the patch test
contraindications included active dermatitis at test site, immunosuppressant use
Patients assessments were performed at preoperative baseline and postoperatively
(3-day, 1-month, 6-month). During each follow-up visit, clinical symptoms, QoL,
and any potential complications were recorded. The primary endpoint was the
complete resolution or significant improvement of symptoms, such as migraines,
chest pain, and palpitations, as assessed through patient-reported outcomes and
clinical evaluations. Secondary endpoints included the absence of major
complications such as atrial fibrillation (AF), stroke, or the need for
reoperation. QoL Scores were transformed to a 0–100 scale according to RAND
Corporation protocols, with higher scores indicating better health status. QoL
was categorized as: Poor: Score
Descriptive statistics were used to summarize patient demographics and clinical
characteristics. Continuous variables were expressed as mean (SD) or median with
interquartile range (IQR), as appropriate. Categorical variables were presented
as counts and percentages. Patients’ QoL was assessed using the 36-Item Short
Form Survey (SF-36), administered at 3-day, 1-month, and 6-month postoperative
intervals. Missing domain items were imputed using the domain mean if
Of 110 patients initially referred for endoscopic explantation evaluation, 95
(86.4%) comprised the final study cohort after application of exclusion criteria
(Fig. 1). The screening process excluded 15 patients: 8 (7.3%) for insufficient
medical optimization or asymptomatic status, 2 (1.8%) for device thrombosis, 2
(1.8%) for significant residual shunt, and 3 (2.7%) for contraindications to
endoscopic approach (1 with autoimmune disease, 1 with prior right thoracic
surgery, 1 with BMI
All 95 patients in the final cohort successfully underwent totally endoscopic device removal without conversion to open procedure (100% technical success rate). The cohort of 95 patients had a median age of 32.6 years, with a notable female predominance (89.5%). Among the patients, the most commonly used devices were from LifeTech Scientific Corporation accounting for 34.7%, while AGA Amplatzer devices were used in 10.5% of cases. Cardiovascular comorbidities, such as coronary artery disease (2.1%), hyperlipidemia (5.3%), and hypertension (5.3%), were relatively uncommon. Psychiatric conditions were also infrequent, with 7.4% of patients diagnosed with anxiety and 5.3% with depression. Autoimmune and thyroid conditions were rare. Notably, metal allergies were highly prevalent, with 47.4% of patients reporting allergies to common items like belt buckles or earring studs, indicating a potential link between metal sensitivity and device-related complications.
The primary indication for device placement was a history of stroke or TIA (47.4%), followed by refractory headaches or migraines (36.8%). ASD accounted for 10.5% of cases, and 5.3% of patients were asymptomatic. The high prevalence of neurological conditions, particularly migraines and stroke, reflects their established association with PFO. Importantly, patients with documented allergy histories, particularly metal allergies, may have experienced complications that led to the need for device removal, suggesting a possible influence of allergies on long-term device success (Table 1).
| Characteristic | Result (n = 95) | |
| Age (y) | 32.6 (12–52) | |
| Female sex | 85 (89.5) | |
| Device type | ||
| AGA Amplatzer | 10 (10.5) | |
| Lepu Medical | 32 (33.7) | |
| LifeTech Scientific Corporation | 33 (34.7) | |
| Starway Medical company | 20 (21.1) | |
| Cardiovascular | ||
| Coronary artery disease | 2 (2.1) | |
| Hyperlipidemia | 5 (5.3) | |
| Hypertension | 5 (5.3) | |
| Psychiatric | ||
| Anxiety | 7 (7.4) | |
| Depression | 5 (5.3) | |
| Autoimmune | ||
| Rheumatoid arthritis | 2 (2.1) | |
| Hashimoto thyroiditis | 2 (2.1) | |
| Thyroid | ||
| Hypothyroidism | 2 (2.1) | |
| Hyperthyroidism | 2 (2.1) | |
| Other | ||
| Chronic kidney disease | 0 (0) | |
| Type 2 diabetes | 2 (2.1) | |
| Documented allergy histories | 65 (68.4) | |
| Metal allergy (Belt buckles/Earring stud/Intrauterine device) | 45 (47.4) | |
| Allergic rhinitis | 5 (5.3) | |
| Urticaria | 5 (5.3) | |
| Antibiotic allergy | 5 (5.3) | |
| Aspirin | 5 (5.3) | |
| Indications for PFO/ASD occluder device placement | ||
| History of stroke/TIA | 45 (47.4) | |
| Refractory headaches/migraines | 35 (36.8) | |
| ASD | 10 (10.5) | |
| Asymptomatic | 5 (5.3) | |
Values are presented as median (IQR) or n (%). PFO, Patent Foramen Ovale; ASD, Atrial Septal Defect; TIA, Transient Ischemic Attack.
After device placement, 84.2% of the patients reported experiencing pain, with specific symptoms varying widely. Headaches or migraines were prevalent in 47.4% of patients, while chest pain affected 21.1%. Neurological symptoms were reported in 42.1% of the cohort, such as dizziness or vertigo (21.1%) and blurred vision (21.1%). Cardiovascular issues were common, such as palpitations (15.8%), breathlessness on exertion (21.1%), and arrhythmias or tachycardia (15.8%). Fatigue affected 31.6% of patients, and gastrointestinal symptoms, skin rash, and depression were reported by 10.5% of patients each. Notably, the majority of symptoms (84.2%) occurred immediately post-operatively, while 10.5% experienced symptoms within the first month, and 5.3% had symptoms that appeared more than one month after surgery (Table 2).
| Symptoms | n = 95 | |
| Time of symptom onset | ||
| Post-operative immediate | 80 (84.2) | |
| Within 1 month post-operatively | 10 (10.5) | |
| Over 1 month post-operatively | 5 (5.3) | |
| Pain | ||
| Chest pain | 20 (21.1) | |
| Headaches/migraines | 45 (47.4) | |
| Muscle/body aches | 15 (15.8) | |
| Neurologic | ||
| Dizziness/vertigo | 20 (21.1) | |
| Blurred vision | 20 (21.1) | |
| Cardiovascular | ||
| Palpitations | 15 (15.8) | |
| Breathlessness on exertion | 20 (21.1) | |
| Arrhythmia/Tachycardia | 15 (15.8) | |
| Fainting | 10 (10.5) | |
| Fatigue | 30 (31.6) | |
| Depression | 10 (10.5) | |
| Gastrointestinal | 10 (10.5) | |
| Cough | 10 (10.5) | |
| Skin rash/Itching | 10 (10.5) | |
Values are presented as median (IQR) or n (%).
Fig. 2 demonstrates the outcomes of nickel allergy patch testing in patients with implanted PFO/ASD occluder devices. In the left image, a clear positive reaction (indicated by the red arrow) is visible, showing localized inflammation at the nickel test site. The right image presents the testing process, where various allergens are applied on the patient’s back using patches. Positive reactions to nickel were commonly observed in patients who experienced postoperative complications, reinforcing the connection between nickel hypersensitivity and the need for device explantation.
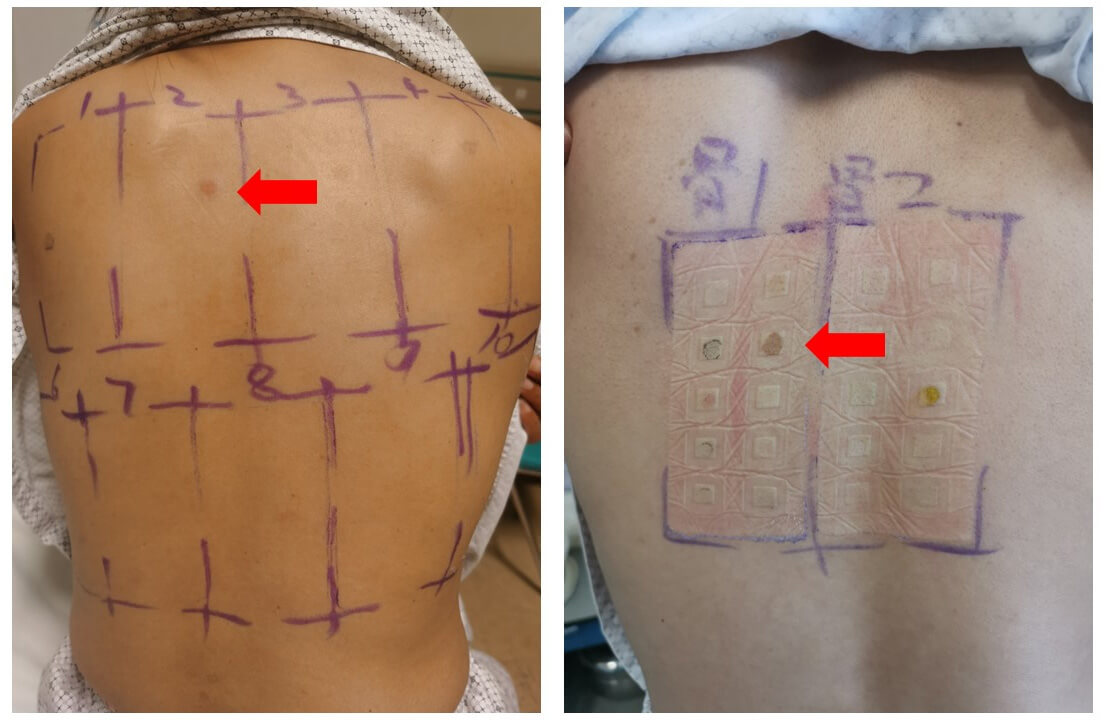 Fig. 2.
Fig. 2.
Patch testing for nickel allergy in patients with PFO/ASD occluder devices. The images depict the results of patch testing, with positive reactions indicated by the red arrows. The left image shows a skin reaction with inflammation at the test site, while the right image displays the test setup, including various allergens applied via patches. The positive reaction suggests nickel hypersensitivity, a key factor in postoperative complications and device explantation decisions.
Fig. 3 illustrates the critical steps in the totally endoscopic removal of a PFO occluder device, highlighting both the technical aspects and intraoperative findings. Upon exposure of the right atrium, the device is delicately separated from the septal tissue, after that, the residual septal defect is inspected for any structural abnormalities or incomplete healing. The procedure is completed with direct suturing of the septal defect or using tissue patch. The totally endoscopic approach, as demonstrated in these images, was efficient and resulted in a clear operative field, allowing for a successful and complication-free surgery.
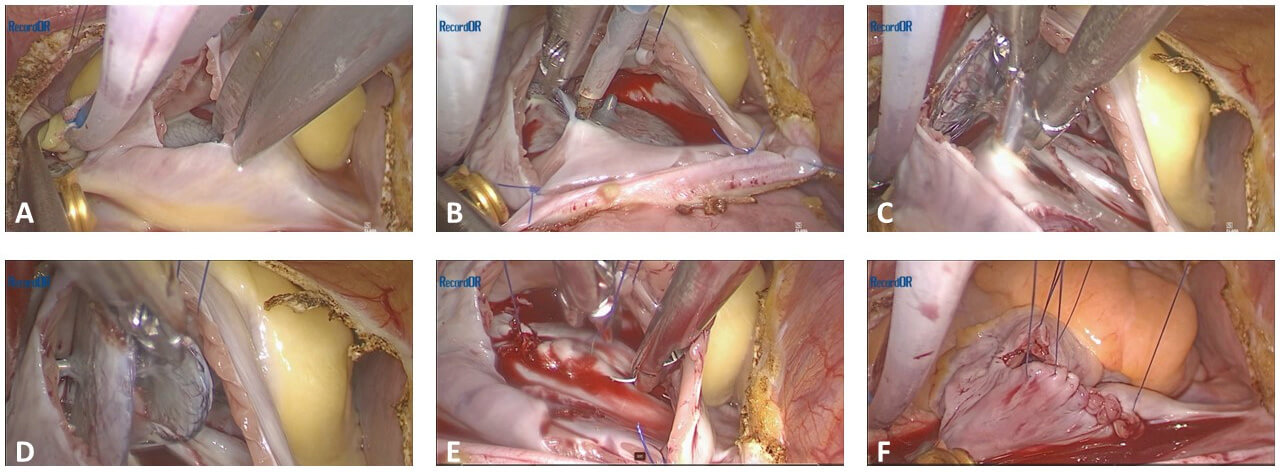 Fig. 3.
Fig. 3.
Intraoperative images of totally endoscopic removal of a PFO occluder device. The sequence illustrates (A) initial exposure of the heart and surrounding structures, (B) identification and isolation of the PFO occluder device, (C) device removal and (D) inspection of the residual defect, (E) preparation for septal repair, and (F) completion of the repair with direct suturing.
All 95 patients undergoing PFO or ASD occluder removal were treated with a totally endoscopic surgical approach, without the need for thoracotomy or sternotomy. Residual septal defects after devices removal were sutured directly in 96.8% patients, and only 3 patients (3.2%) requiring a tissue patch. Nickel allergy testing was performed in all patients, with a majority (89.5%) testing positive, suggesting a strong correlation between nickel sensitivity and device-related complications. The mean CPB time was 71.2 minutes, while cross-clamp time averaged 34.6 minutes. Intraoperative blood loss was minimal, averaging 36.8 mL, and ventilation time postoperatively averaged 4.7 hours. There were no cases of reoperation for bleeding or in-hospital deaths, reflecting a favorable safety profile. The tricuspid valve was inspected intraoperatively and found to be functionally normal, thus requiring no repair. There was no iatrogenic damage to the valve. Chest drainage volume was low, averaging 17.5 mL, with an average chest drainage weaning time of 10.9 hours. Only 2 patients (2.1%) developed postoperative AF, further indicating a low complication rate (Table 3). Fig. 4 depicts the explanted PFO occluder device in multiple orientations. The images clearly show the device’s structural integrity post-explantation, with visible tissue remnants and signs of complete endothelialization.
 Fig. 4.
Fig. 4.
Explanted PFO occluder device, displaying post-removal condition. The figure shows multiple views of the device, including top-down and side perspectives.
| Characteristic | Result | |
| Surgical approach | ||
| Totally endoscopic | 95 (100) | |
| Thoracotomy/sternotomy | 0 | |
| Residual septal defects | ||
| Directly repair | 92 (96.8) | |
| Tissue Patch | 3 (3.2) | |
| Nickel allergy testing | ||
| Patch positive | 85 (89.5) | |
| Patch negative | 10 (10.5) | |
| CPB time, median (IQR), min | 71.2 (50–128) | |
| Cross Clamp time, median (IQR), min | 34.6 (14–76) | |
| Blood loss, mean (SD), mL | 36.8 (21.6) | |
| Ventilation time, mean (SD), h | 4.7 (1.8) | |
| Reoperation for bleeding, n (%) | 0 | |
| In hospital death (%) | 0 | |
| Chest drainage volume, mean (SD), mL | 17.5 (19.3) | |
| Chest drainage weaning time, mean (SD), h | 10.9 (1.8) | |
| Postoperative AF, n (%) | 2 (2.1) | |
Values are presented as median (range) or n (%). PFO, Patent Foramen Ovale; ASD, Atrial Septal Defect; CPB, Cardiopulmonary Bypass; SD, Standard Deviation; AF, Atrial Fibrillation.
Fig. 5 shows the postoperative incisions following a totally endoscopic PFO occluder device removal. The red arrows indicate small thoracoscopic entry sites used during the surgery, while the white arrow highlights the primary incision through which the occluder device was removed. The image reveals minimal scarring and localized bruising, demonstrating the effectiveness of the endoscopic approach.
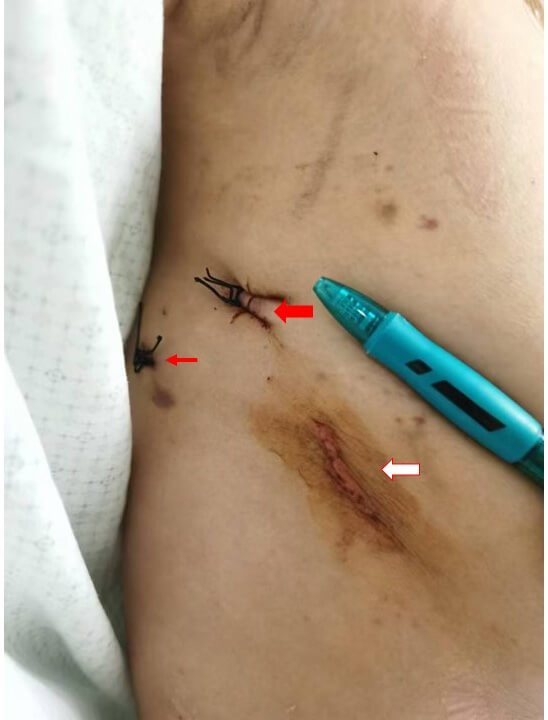 Fig. 5.
Fig. 5.
Postoperative thoracoscopic incisions following totally endoscopic removal of a PFO occluder device. The red arrows highlight small thoracoscopic entry points, while the white arrow indicates the primary incision for device removal. Minimal scarring is visible, demonstrating the less invasive nature of the procedure. The image illustrates the favorable cosmetic outcome and limited surgical trauma associated with the endoscopic approach.
QoL ratings among the 95 patients markedly improved following PFO/ASD occluder
removal (Table 4). Preoperatively, most patients reported poor QoL (55.8%, n =
53), with only 7.4% (n = 7) rating it as good. By 3 days post-surgery, a
substantial improvement was observed: 52.6% (n = 50) reported good QoL, and poor
ratings decreased sharply to 5.3% (n = 5). At 1-month follow-up, good QoL
further increased to 76.8% (n = 73), while poor ratings persisted in only 2
patients (2.1%). The most dramatic improvement occurred at 6 months
post-surgery, with 95.8% (n = 91) reporting good QoL, and only 2 patients each
categorized as fair (2.1%) or poor (2.1%) (Fig. 6). Sensitivity analyses
confirmed the robustness of these findings. Linear mixed models of continuous
SF-36 scores showed a significant time effect (p
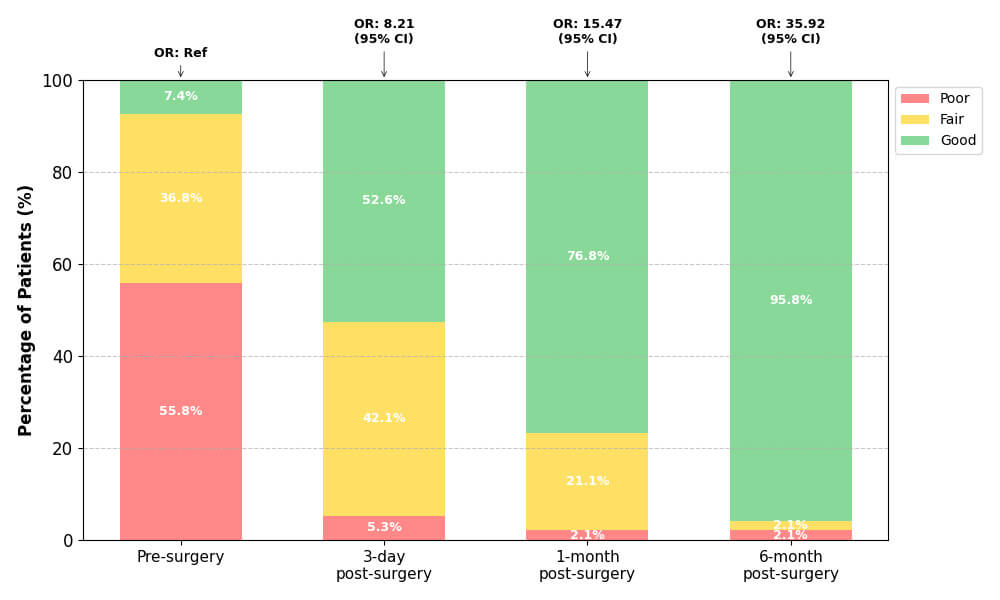 Fig. 6.
Fig. 6.
Longitudinal quality of life improvement following endoscopic
PFO/ASD occluder removal. Stacked percentage bar chart showing the
distribution of Quality of Life ratings (Poor, Fair, Good) at four time points
among 95 patients. Percentages are labeled within each segment. Adjusted odds
ratios (OR) from GEE ordinal regression model are displayed above each bar,
demonstrating significant improvement over time (all p
| Follow-up timepoint | Quality of life rating, n (%) | Adjusted OR* (95% CI) | p-value† | ||
| Poor | Fair | Good | |||
| Pre-surgery | 53 (55.8) | 35 (36.8) | 7 (7.4) | 1.00 (Ref) | |
| 3-day post-surgery | 5 (5.3) | 40 (42.1) | 50 (52.6) | 8.21 (4.12–16.35) | |
| 1-month post-surgery | 2 (2.1) | 20 (21.1) | 73 (76.8) | 15.47 (7.89–30.35) | |
| 6-month post-surgery | 2 (2.1) | 2 (2.1) | 91 (95.8) | 35.92 (16.84–76.61) | |
GEE, generalized estimating equations; OR, Odds Ratio; CI, Confidence Interval,
*Adjusted for age, sex, indication, device brand, implant-to-explant interval,
and nickel allergy status. †p-values adjusted for
multiple comparisons (Bonferroni correction;
The present study demonstrates that totally endoscopic removal of PFO/ASD occluder devices is a safe and effective treatment for patients with severe, device-related complications, particularly those associated with nickel hypersensitivity. The primary findings indicate that device explantation significantly improves clinical symptoms, with 95.8% of patients reporting good QoL at six months postoperatively. Additionally, the totally endoscopic approach was associated with minimal perioperative complications, no conversions to thoracotomy, and excellent cosmetic outcomes, reinforcing its safety and efficacy. The findings underscore the need for careful patient selection and monitoring for nickel hypersensitivity in individuals undergoing PFO/ASD device implantation.
Our findings are consistent with previous studies that have highlighted the positive outcomes of device removal in patients experiencing complications associated with nickel hypersensitivity. Similar to the results reported by Sharma et al. [9] who found significant symptom relief in 58 patients following the removal of atrial septal occluders, our study showed a high rate of symptom resolution. Likewise, Fernandes et al. [22] reported complete migraine resolution in a 16-year-old patient following device explantation due to nickel allergy, a result that aligns with the improvements observed in our cohort. However, our study differs from prior research in terms of the surgical approach. While previous studies have primarily focused on open thoracotomy for device removal, such as in the case series by Verma and Tobis [23], we employed a totally endoscopic approach, which resulted in minimal scarring and faster recovery times. This minimally invasive method demonstrated similar efficacy in symptom resolution without the need for more invasive surgical techniques. Furthermore, our study highlights the importance of preoperative nickel allergy testing, as 89.5% of patients with positive tests showed substantial improvement post-explantation, which reinforces the findings of Apostolos et al. [6] on the role of nickel allergy in device-related complications. Besides, the 10.5% nickel-negative patients also exhibited improvement post-explantation, which may attributed to removal of endothelial dysfunction profiles, confirming device removal as a universal solution for refractory complications. The combination of our surgical approach and focus on allergy testing makes our study unique compared to prior literature.
The finding that a substantial majority of patients in this cohort presenting for occluder removal had a documented allergy—most commonly metal hypersensitivity—is striking but consistent with a growing body of evidence. This high prevalence is not representative of the general population receiving cardiac occluders but is instead a profound example of selection bias. This cohort does not consist of random post-operative patients; it is a highly specific group whose devices have failed, often due to intolerable adverse symptoms that prompted surgical intervention. Therefore, the 80%+ figure likely reflects the key etiological role of hypersensitivity reactions in driving device failure and explantation, rather than the incidence of allergy in all implanted patients.
Supplementary Table 1 highlight various cases and studies concerning patients who underwent device explantation due to complications related to PFO or ASD occlusion devices. A recurrent theme across several studies is the resolution of symptoms, such as migraines, chest pain, and systemic hypersensitivity, following the removal of the devices. In the case of Fernandes et al. [22], a 16-year-old male experienced complete relief from migraines post-explantation after six years, with incomplete endothelialization identified as a contributing factor [22]. Other studies, such as Spina et al. [24] and Wertman et al. [4], also underscore the link between nickel hypersensitivity and adverse outcomes, recommending preoperative allergy screening. Studies like Rodés-Cabau et al. [1] showed that dual antiplatelet therapy could reduce migraines in patients with ASD occlusion devices. Finally, Sadasivan Nair et al. [14] and Verma and Tobis [23] highlight that nickel allergies are a significant reason for explantation, with complete symptom resolution post-device removal in most cases. These findings underscore the importance of preoperative assessments and monitoring for nickel allergies to avoid long-term complications.
The endoscopic method used in our cohort resulted in shorter recovery times, minimal postoperative complications, and excellent cosmetic outcomes. Unlike traditional open thoracotomy, which has been the standard in cases requiring PFO or ASD device explantation [23]. Furthermore, the high success rate of direct septal repair (96.8%) without the need for extensive patch repair emphasizes the effectiveness of the endoscopic technique in managing residual septal defects, a finding that has not been as prominently addressed in prior studies. The minimal blood loss (36.8 mL) and low incidence of postoperative AF (2.1%) in our study further highlight the safety profile of this technique compared to previous reports where complications such as arrhythmia were more prevalent following device explantation using traditional methods [9]. Additionally, the emphasis on preoperative nickel allergy testing, with 89.5% of patients showing positive results, supports the growing recognition of the importance of allergy screening in device-related procedures [6]. This comprehensive combination of a minimally invasive approach and targeted preoperative assessments makes our study unique and highlights the potential for improving patient outcomes in future cases.
One limitation of our study is its retrospective design, which may introduce
selection bias and limit the generalizability of the results. While our
retrospective design precludes definitive causal attribution, the temporal
association between device implantation and symptom onset provides compelling
evidence: 84.2% (80/95) of patients developed symptoms within 24 hours
post-procedure (Table 2), corresponding precisely to the 12–72 hour activation
window of Type IV nickel hypersensitivity reactions. This acute manifestation
pattern—distinct from endothelialization failure (typically
In conclusion, the totally endoscopic removal of PFO/ASD occluder devices offers a promising, minimally invasive option for patients experiencing complications related to nickel hypersensitivity or device intolerance. This approach is not only associated with favorable cosmetic outcomes and reduced surgical trauma but also provides significant symptom relief. However, the current understanding of nickel hypersensitivity and its long-term impact on intracardiac devices remains limited. Future studies should focus on developing standardized screening protocols for nickel allergy prior to device implantation and exploring the long-term safety and effectiveness of endoscopic explantation procedures. Additionally, more extensive research is needed to elucidate the pathophysiological mechanisms linking nickel exposure to device-related complications, which could inform both preoperative assessments and post-implantation monitoring strategies. Multicenter, prospective trials with larger, more diverse populations will be crucial to improving the management and treatment outcomes for patients requiring PFO/ASD occluder.
The datasets generated and analyzed during the current study are not publicly available due to privacy and confidentiality concerns but are available from the corresponding author on reasonable request. Access to the data may be granted for research purposes following approval by the institutional review board and adherence to applicable data protection regulations.
HS contributed to the conception and design of the work; acquired, analyzed, and interpreted the patient data; and drafted the manuscript. MR Contributed to the design of the work; acquired data (surgical procedures); analyzed and interpreted the intraoperative and outcomes data; and critically revised the manuscript for important intellectual content. DL contributed to the acquisition and analysis of the data, including statistical analysis; and critically revised the manuscript. SJ contributed to the design of the work, supervised the surgical procedures and data acquisition, and critically revised the manuscript. NC contributed to the conception of the work, the analysis and interpretation of follow-up data, and the revision of the manuscript. LZ contributed to the conception and design of the work; coordinated the study and supervised data acquisition; interpreted the results; critically revised the manuscript for important intellectual content; served as the corresponding author and finalized the manuscript; and ensured adherence to ethical standards. All authors provided final approval of the version to be published and agree to be accountable for all aspects of the work.
This study was conducted in accordance with the Declaration of Helsinki and was approved by the institutional review board of The First Medical Center of Chinese PLA General Hospital (No. S2025-492-01). Given the retrospective nature of the study, the requirement for informed consent was waived. All patient data were anonymized to ensure privacy and confidentiality. The ethical guidelines of the hospital were strictly followed throughout the research process.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/HSF49911.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






