1 Department of Management, Health Systems Management Program, Bar Ilan University, 5290002 Ramat Gan, Israel
2 The Faculty of Medicine, Tel Aviv University, 6997801 Tel Aviv, Israel
3 Lis Hospital for Women’s Health, Tel Aviv Sourasky Medical Center, 6423906 Tel Aviv, Israel
4 Psychiatric Division, Tel Aviv Sourasky Medical Center, 6423906 Tel Aviv, Israel
5 Be’eri Community Psychiatry Clinic, Clalit Health Services, 5239530 Ramat Gan, Israel
6 Executive Management, Clalit Health Services, 6209804 Tel Aviv, Israel
Abstract
Armed conflict-related evacuation may expose pregnant women to acute stress and disrupted care, and the specific obstetric consequences of evacuation remain poorly defined. This study evaluated outcomes among evacuated women.
A retrospective cohort study was conducted using data from nine university-affiliated hospitals in Israel. Deliveries between October 7, 2023, and April 7, 2024, were included. Women who resided in government-designated evacuation zones formed the study group, whereas women from non-evacuated regions served as controls. Electronic medical records (EMRs) were reviewed and included maternal demographics, obstetric history, comorbidities, and obstetric and neonatal outcomes. Propensity score matching (1:3) was performed based on maternal age, socioeconomic status (SES), parity, and chronic comorbidities.
The total cohort included 14,290 deliveries, of which 321 (2.2%) occurred in evacuated zones. Evacuated women had higher rates of pregestational diabetes mellitus (PGDM), malignancy, lower SES, and were more often Jewish. In unadjusted analyses, late preterm birth (PTB) (34 to 36+6 weeks) was significantly more common among evacuated women (5.6% vs. 3.4%; odds ratio [OR]: 1.67, 95% confidence interval [CI]: 1.03–2.71; p = 0.038). In contrast, overall PTB and earlier PTB did not differ significantly between groups. Postpartum hemorrhage (PPH) was significantly lower in the evacuated group (1.6% vs. 4.4%; OR: 0.35, 95% CI: 0.14–0.84; p = 0.013). Other obstetric and neonatal outcomes did not differ significantly between groups. Following matching (287 evacuated vs. 861 controls), baseline characteristics were well balanced. The incidence of PPH remained significantly lower in the evacuated group (1.4% vs. 4.3%; OR: 0.31, 95% CI: 0.11–0.86; p = 0.027). Other obstetric and neonatal outcomes, including PTB, did not differ significantly between the two groups.
Among women from evacuation zones, obstetric and neonatal outcomes did not differ significantly from those of non-evacuated women, except for a lower rate of PPH. These findings suggest that, despite acute stress and potential disruptions in care, evacuation itself was not associated with adverse obstetric outcomes, underscoring the resilience of maternity care systems during periods of conflict.
Keywords
- stress
- war
- obstetrical outcomes
- residents of evacuation zones
Widespread armed conflicts often lead to the evacuation of civilian populations from high-risk areas due to ongoing security threats. Beyond displacement, armed conflict itself, especially when prolonged or unplanned, is associated with substantial psychological stress, disruption of daily routines, and limited access to healthcare services [1, 2]. Pregnant women represent a particularly vulnerable population in these contexts, as war-related stress may negatively affect maternal well-being and lead to adverse obstetric outcomes [3, 4].
Several studies have examined the effects of stress during pregnancy [5, 6, 7, 8, 9]. A large systematic review assessed the impact of armed conflict on obstetric outcomes and analyzed over 1.17 million pregnancies from conflict-affected regions [10]. The most consistent finding across studies was an increased risk of low birth weight (LBW), defined as birth weight less than 2500 grams, among exposed women. In contrast, results regarding preterm birth (PTB), defined as delivery before 37 weeks of gestation, were mixed. Some studies reported increased rates [11, 12], whereas others found no significant association [9, 13]. Additional adverse outcomes included increased rates of miscarriage, stillbirth, congenital anomalies, and premature rupture of membranes (PROM), although the strength of evidence varied across studies. Israeli studies have similarly identified associations between maternal exposure to military-related stress and heightened risks of both PTB and LBW [14, 15, 16]. One study further demonstrated that the timing and nature of stress exposure may influence specific outcomes: in hospitals exposed to acute stress, exposure during the first- and second-trimesters was primarily associated with increased PTB rates; in contrast, in populations exposed to prolonged, conflict-related stress, exposure during the third-trimester was associated with higher rates of macrosomia compared with exposure during the first- and second-trimesters [17].
The potential mechanisms underlying these associations have been explored in the literature, suggesting that maternal stress affects pregnancy outcomes through neuroendocrine and immunologic pathways. Specifically, stress activates the hypothalamic-pituitary-adrenal (HPA) axis, resulting in elevated cortisol levels, inflammatory responses, hormonal fluctuations, and altered immune function [18, 19, 20].
Despite the breadth of existing research, most studies have examined general exposure to conflict or war-related stress at the population level without focusing specifically on women who were actively evacuated from their homes. Evacuation represents a unique and acute form of displacement and often involves abrupt separation from support systems, disruption of prenatal care, and heightened uncertainty about personal and family safety. These circumstances may amplify the physiological and psychological stress experienced during pregnancy, potentially influencing maternal and neonatal outcomes differently from generalized conflict exposure. To date, no large-scale, multicenter study has investigated the specific obstetric effects of evacuation during armed conflict using a matched cohort design.
This study focused on obstetric outcomes among women who experienced evacuation due to widespread armed conflict. By analyzing a large national cohort over a defined six-month post-evacuation period, the research highlights the potential physiological and clinical effects of acute displacement and conflict-related stress during pregnancy. The findings may provide valuable insights to guide healthcare responses and preparedness strategies in settings of civilian evacuation, with the goal of safeguarding maternal and neonatal health in times of crisis.
We conducted a retrospective cohort analysis to assess the impact of direct war-related exposure, specifically evacuation due to missile attacks, on obstetric and neonatal outcomes. Data were extracted from electronic medical records (EMRs) of deliveries that occurred between October 7, 2023, and April 7, 2024, at nine university-affiliated hospitals in Israel. Women were categorized into two groups: the evacuated group, which comprised those who resided in officially designated evacuation zones and were displaced due to the conflict, and the non-evacuated group, consisting of women who lived in other regions affected by the war but were not subject to evacuation orders [21]. Deliveries were excluded if they occurred before 24+0 weeks of gestation (below the viability threshold defined by Israeli obstetrics and neonatology guidelines) [22, 23], after 43+0 weeks (as routine induction is recommended at 42 weeks and later gestations are likely documentation errors [24]), or if the case involved perinatal mortality.
Maternal characteristics comprised age, categorized as
Obstetric and neonatal outcomes comprised PTB (defined as delivery before 37
weeks of gestation) and gestational age at birth, categorized as 24 to 33+6, 34
to 36+6, and
No formal a priori sample size calculation was performed. Post hoc
power analyses were therefore conducted using the full cohort with PROC POWER
in SAS Enterprise Guide version 8.3 (SAS Institute Inc., Cary, NC, USA),
assuming a two-sided
Baseline maternal characteristics were compared between groups using descriptive univariate tests (chi-square or Fisher exact tests for categorical variables and t-tests for continuous variables), with p-values reported to describe group differences. Continuous variables such as parity were presented as median (interquartile range [IQR]) due to their distributional characteristics.
Associations between evacuation status and obstetric and neonatal outcomes in the unmatched cohort were evaluated using regression-based models to estimate effect sizes with 95% confidence intervals (CIs). Binary outcomes were analyzed using logistic regression; for rare outcomes with sparse data, Firth penalized logistic regression was applied. Continuous outcomes were analyzed using generalized linear models (PROC GLM in SAS). Nominal outcomes with more than two categories were analyzed using multinomial logistic regression with a generalized logit link. Effect estimates were reported as ORs or mean differences with 95% CIs.
Due to demographic differences between the evacuated and non-evacuated groups, a
matching procedure was performed using the PROC PSMATCH (SAS). The PROC PSMATCH
procedure implements propensity score matching (PSM) to reduce bias due to
confounding variables in observational studies by matching subjects from
different groups based on their probability for exposure [26]. Propensity scores
were estimated using logistic regression. The matching model incorporated age,
categorized into
Because PSM induces correlation within matched sets, all post-matching inferential analyses accounted for the matched design. Binary outcomes were analyzed using conditional logistic regression; for rare outcomes or sparse data within matched sets, exact conditional logistic regression was applied when feasible. Continuous outcomes were analyzed using linear mixed effects models with a random effect for the matched set. Nominal categorical outcomes with more than two levels were analyzed using multinomial logistic regression, implemented via PROC SURVEYLOGISTIC in SAS with a generalized logit link and cluster robust standard errors at the matched set level. Effect estimates were reported as ORs or mean differences with 95% CIs. No formal adjustment for multiple comparisons was applied; however, findings from exploratory analyses were interpreted with caution. Analyses were conducted using available-case data, and variable-specific denominators are reported in all tables.
All statistical analyses were performed using SAS Enterprise Guide version 8.3 (SAS Institute Inc., Cary, NC, USA). The study was approved by the institutional review board of Clalit Health Services (IRB No. 0117-23-COM2). Due to the retrospective design and the use of deidentified EMRs, the requirement for informed consent was waived.
Overall, 14,600 births were initially identified for the study. After exclusion of cases due to missing data or other predefined criteria, 14,290 deliveries were included in the analysis. Among these, 321 women (2.2%) resided in regions officially evacuated due to security threats following the October 7, 2023, conflict, while 13,969 deliveries (97.8%) occurred among women who remained in non-evacuated areas (Fig. 1).
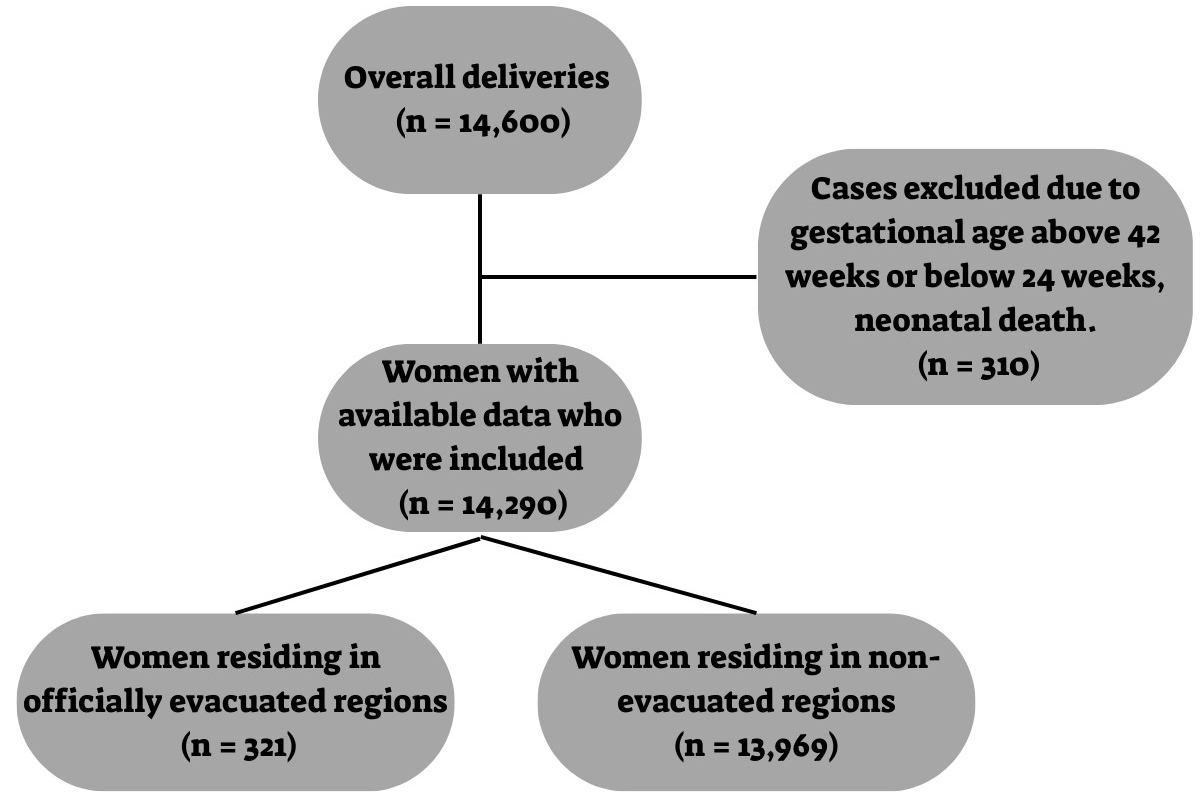 Fig. 1.
Fig. 1.
Flowchart of study participants.
Maternal demographic and clinical characteristics are detailed in Table 1. No
significant differences were observed across maternal age groups. Regarding
comorbidities, evacuated women exhibited higher rates of PGDM (2.8% vs. 1.4%,
p = 0.042) and malignancy (4.1% vs. 2.4%, p = 0.049).
Additionally, evacuated women were more likely to belong to a lower SES
(p
| All (n = 14,290) | Residents of evacuation zones (n = 321) | Control group (n = 13,969) | p-value | |||||
| Total | Value | Total | Value | Total | Value | |||
| Age, mean |
14,290 | 30.1 |
321 | 30.3 |
13,969 | 30.1 |
0.436 | |
| Age, n (%) | 14,290 | 3185 (22.3) | 321 | 60 (18.7) | 13,969 | 3125 (22.4) | 0.291 | |
| 25 to 40 years | 10,663 (74.6) | 251 (78.2) | 10,412 (74.5) | |||||
| 442 (3.1) | 10 (3.1) | 432 (3.1) | ||||||
| Parity, median (IQR) | 14,290 | 1 (0–2) | 321 | 1 (0–3) | 13,969 | 1 (0–2) | 0.021 | |
| SES, n (%) | Low | 12,370 | 5340 (43.2) | 288 | 153 (53.1) | 12,082 | 5187 (42.9) | |
| Medium | 3416 (27.6) | 113 (39.2) | 3303 (27.3) | |||||
| High | 3614 (29.2) | 22 (7.6) | 3592 (29.7) | |||||
| BMI, kg/m2 | 10,686 | 739 (6.9) | 220 | 15 (6.8) | 10,466 | 724 (6.9) | 0.919 | |
| 18.5–24.9 | 5765 (53.9) | 118 (53.6) | 5647 (54.0) | |||||
| 25–29.9 | 2591 (24.2) | 57 (25.9) | 2534 (24.2) | |||||
| 1591 (14.9) | 30 (13.6) | 1561 (14.9) | ||||||
| Ethnicity, n (%) | Arab | 8756 | 4686 (53.5) | 147 | 5 (3.4) | 8609 | 4681 (54.4) | |
| Jews | 4070 (46.5) | 142 (96.6) | 3928 (45.6) | |||||
| Fibromyalgia, n (%) | 14,290 | 1091 (7.6) | 321 | 28 (8.72) | 13,969 | 1063 (7.6) | 0.457 | |
| Hypothyroidism, n (%) | 14,290 | 1117 (7.8) | 321 | 21 (6.5) | 13,969 | 1096 (7.8) | 0.389 | |
| Malignancy, n (%) | 14,290 | 342 (2.4) | 321 | 13 (4.1) | 13,969 | 329 (2.4) | 0.049 | |
| Lipid disorders, n (%) | 14,290 | 475 (3.3) | 321 | 8 (2.5) | 13,969 | 467(3.3) | 0.400 | |
| Chronic hypertension, n (%) | 14,290 | 177 (1.2) | 321 | 3 (0.9) | 13,969 | 174 (1.2) | 0.618 | |
| PGDM, n (%) | 14,290 | 209 (1.4) | 321 | 9 (2.8) | 13,969 | 200 (1.4) | 0.042 | |
SD, standard deviation; IQR, interquartile range; SES, socioeconomic status; BMI, body mass index; PGDM, pregestational diabetes mellitus.
Bold indicates statistically significant results (p
Obstetric and neonatal outcomes are presented in Table 2. Among women who
delivered between 34 and 36+6 weeks of gestation, the proportion was higher in
the evacuated group compared with the non-evacuated group (5.6% vs. 3.4%). This
difference was statistically significant, with an OR of 1.67 (95% CI:
1.03–2.71; p = 0.038). In contrast, no significant difference was
observed for earlier PTBs (24 to 33+6 weeks). When overall PTB (
| All (n = 14,290) | Residents of evacuation zones (n = 321) | Control group (n = 13,969) | Effect estimate (OR/beta) | 95% CI | p-value | |||||
| Total | Value | Total | Value | Total | Value | |||||
| Gestational age at delivery, mean |
14,285 | 39.0 |
321 | 39.0 |
13,964 | 39.0 |
–0.04 | (–0.12)–(0.20) | 0.635 | |
| PTB, n (%) | 14,285 | 701 (4.9) | 321 | 23 (7.2) | 13,964 | 678 (4.9) | 1.51 | 0.98–2.32 | 0.058 | |
| PTB groups, n (%) | 37 weeks and above | 14,285 | 13,584 (95.0) | 321 | 298 (92.8) | 13,964 | 13,286 (95.1) | REF | REF | REF |
| 34 to 36+6 weeks | 499 (3.4) | 18 (5.6) | 481 (3.4) | 1.67 | 1.03–2.71 | 0.038 | ||||
| 24 to 33+6 weeks | 202 (1.4) | 5 (1.5) | 197 (1.4) | 1.13 | 0.46–2.77 | 0.786 | ||||
| Birth type, n (%) | SVB | 12,160 | 9565 (78.6) | 310 | 242 (78.1) | 11,850 | 9323 (78.7) | REF | REF | REF |
| Assist vaginal delivery | 560 (4.6) | 14 (4.5) | 546 (4.6) | 0.99 | 0.57–1.71 | 0.945 | ||||
| CD | 2035 (16.7) | 54 (17.4) | 1981 (16.7) | 1.05 | 0.78–1.42 | 0.744 | ||||
| PPROM, n (%) | 14,285 | 482 (3.4) | 321 | 9 (2.8) | 13,964 | 473 (3.4) | 0.93 | 0.68–1.27 | 0.567 | |
| GDM, n (%) | 14,290 | 1511 (10.6) | 321 | 32 (10.0) | 13,969 | 1479 (10.6) | 0.94 | 0.65–1.35 | 0.721 | |
| HDP, n (%) | 14,290 | 581 (4.1) | 321 | 17 (5.3) | 13,969 | 564 (4.0) | 1.33 | 0.81–2.18 | 0.259 | |
| Placental abruption, n (%) | 14,290 | 118 (0.8) | 321 | 2 (0.6) | 13,969 | 116 (0.8) | 0.93 | 0.26–3.28 | 0.684 | |
| PPH, n (%) | 14,290 | 618 (4.3) | 321 | 5 (1.6) | 13,969 | 613 (4.4) | 0.35 | 0.14–0.84 | 0.013 | |
| Birth weight, mean |
14,290 | 3246.5 |
321 | 3215.1 |
13,969 | 3247.3 |
–32.11 | (–83.17)–18.94 | 0.217 | |
| LBW, n (%) | 14,290 | 729 (5.1) | 321 | 20 (6.2) | 13,969 | 709 (5.1) | 1.24 | 0.79–1.97 | 0.352 | |
| VLBW, n (%) | 14,290 | 135 (0.9) | 321 | 5 (1.6) | 13,969 | 130 (0.9) | 1.84 | 0.78–4.36 | 0.251 | |
| Apgar 1 minute, mean |
13,962 | 8.9 |
309 | 8.9 |
13,653 | 8.9 |
0 | (–0.08)–0.08 | 0.980 | |
| Apgar 5 minutes, mean |
13,961 | 10.0 |
309 | 10.0 |
13,652 | 10.0 |
0.02 | (–0.02)–0.07 | 0.131 | |
| Apgar 5 minutes |
13,961 | 33 (0.2) | 309 | 0 | 13,652 | 33 (0.2) | 0.66 | 0.04–10.80 | 0.387 | |
OR, odds ratio; CI, confidence interval; PTB, preterm birth; SVB, spontaneous vaginal birth; REF, reference; CD, cesarean delivery; PPROM, preterm premature rupture of membranes; GDM, gestational diabetes mellitus; HDP, hypertensive disorders of pregnancy; PPH, postpartum hemorrhage; LBW, low birth weight; VLBW, very low birth weight.
Bold indicates statistically significant results (p
Table 3 compares obstetric outcomes between evacuated and non-evacuated women
after PSM. The matched cohort included 287 women in the evacuated group and 861
controls. Postmatching comparisons demonstrated similar maternal characteristics
between groups (Supplementary Table 1); all postmatching SMDs were
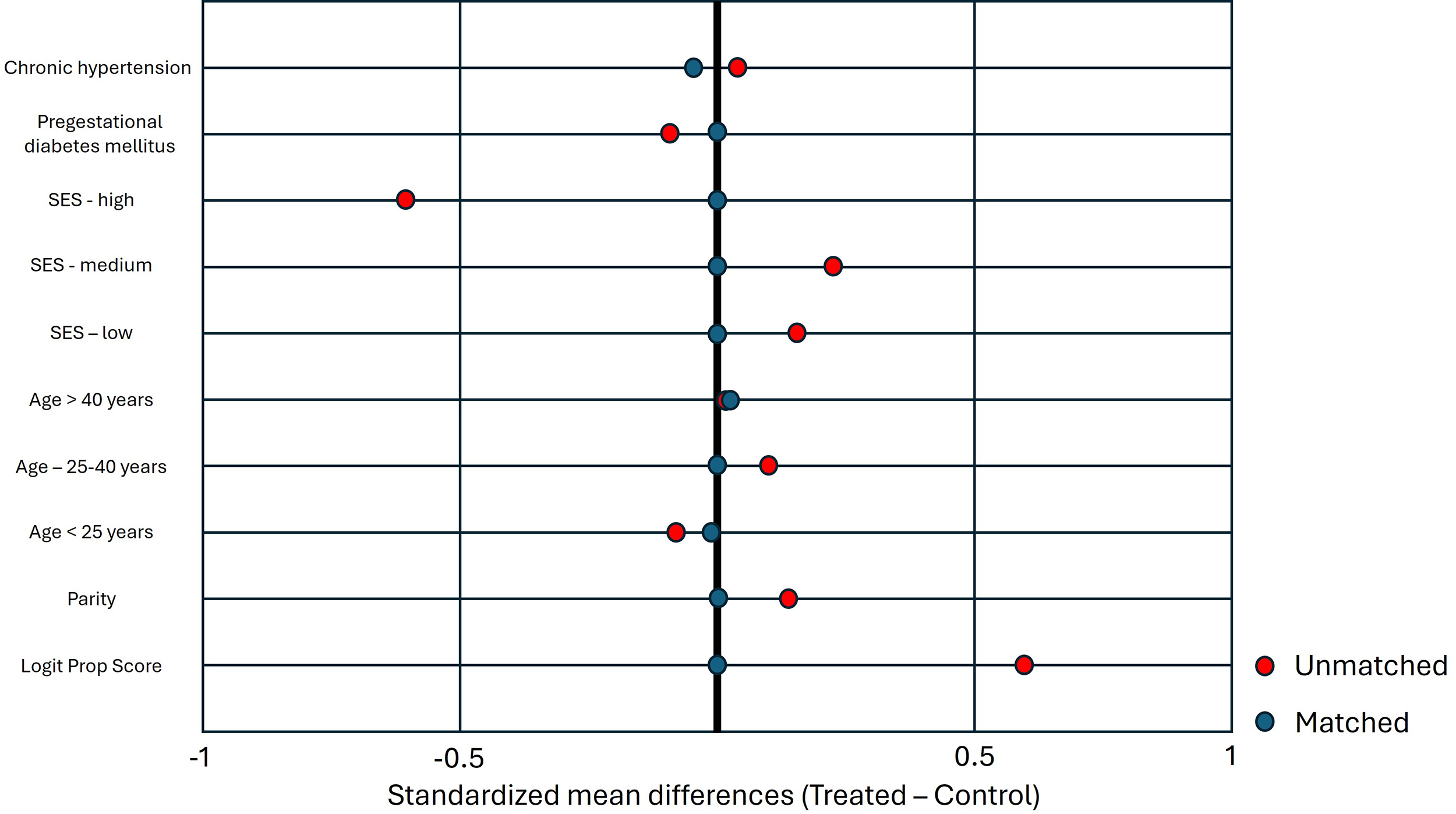 Fig. 2.
Fig. 2.
Love plot showing standardized mean differences for baseline
covariates before and after propensity score matching. After matching, all
covariates demonstrated good balance (
| All (n = 1148) | Residents of evacuation zones (n = 287) | Control group (n = 861) | Effect estimate (OR/beta) | 95% CI | p-value | |||||
| Total | Value | Total | Value | Total | Value | |||||
| Gestational age at delivery, mean |
1147 | 39.0 |
287 | 39.0 |
860 | 38.9 |
0.06 | (–0.13)–0.25 | 0.725 | |
| PTB, n (%) | 1147 | 62 (5.4) | 287 | 21 (7.3) | 860 | 41 (4.8) | 1.59 | 0.92–2.75 | 0.098 | |
| PTB groups, n (%) | 37 weeks and above | 1147 | 1085 (94.6) | 287 | 266 (92.7) | 860 | 819 (95.2) | REF | REF | REF |
| 34 to 36+6 weeks | 52 (4.5) | 17 (5.9) | 35 (4.1) | 1.50 | 0.85–2.62 | 0.159 | ||||
| 24 to 33+6 weeks | 10 (0.9) | 4 (1.4) | 6 (0.7) | 2.05 | 0.57–7.46 | 0.273 | ||||
| Birth type, n (%) | SVB | 1148 | 796 (79.1) | 277 | 220 (79.4) | 730 | 576 (78.9) | REF | REF | REF |
| Assist vaginal delivery | 44 (4.3) | 10 (3.6) | 34 (4.7) | 0.77 | 0.37–1.62 | 0.489 | ||||
| CD | 167 (16.6) | 47 (17.0) | 120 (16.4) | 1.03 | 0.71–1.48 | 0.893 | ||||
| PPROM, n (%) | 1147 | 33 (2.9) | 287 | 9 (3.1) | 860 | 24 (2.8) | 1.13 | 0.52–2.45 | 0.761 | |
| GDM, n (%) | 1148 | 118 (10.3) | 287 | 28 (9.8) | 861 | 90 (10.5) | 0.92 | 0.57–1.47 | 0.722 | |
| HDP, n (%) | 1148 | 43 (3.8) | 287 | 14 (4.9) | 861 | 29 (3.4) | 1.48 | 0.77–2.85 | 0.244 | |
| Placental abruption, n (%) | 1148 | 9 (0.8) | 287 | 2 (0.7) | 861 | 7 (0.8) | 0.86 | 0.09–4.50 | 1.000 | |
| PPH, n (%) | 1148 | 41 (3.6) | 287 | 4 (1.4) | 861 | 37 (4.3) | 0.31 | 0.11–0.86 | 0.027 | |
| Birth weight, mean |
1148 | 3241.2 |
287 | 3210.2 |
861 | 3251.5 |
–41.35 | (–102.47)–19.77 | 0.108 | |
| LBW, n (%) | 1148 | 61 (5.3) | 287 | 18 (6.3) | 861 | 43 (5.0) | 1.27 | 0.72–2.23 | 0.358 | |
| VLBW, n (%) | 1148 | 11 (1.0) | 287 | 5 (1.7) | 861 | 6 (0.7) | 2.50 | 0.60–9.83 | 0.351 | |
| Apgar 1 minute, mean |
1123 | 8.9 |
277 | 8.9 |
846 | 8.9 |
–0.06 | (–0.14)–0.02 | 0.627 | |
| Apgar 5 minutes, mean |
1123 | 10.0 |
277 | 10.0 |
846 | 10.0 |
0 | 0.04–0.04 | 0.178 | |
| Apgar 5 minutes |
1123 | 1 (0.1) | 277 | 0 | 846 | 1 (0.1) | 0.33 | 0.02–0.99 | 0.414 | |
Bold indicates statistically significant results (p
Obstetric outcomes after matching based on maternal age group, SES, presence of
Sensitivity analyses were conducted among women with available prepregnancy BMI, with BMI incorporated into the matching model. Results were consistent with the primary analyses (Supplementary Table 2).
Post hoc power analyses indicated that the study had adequate statistical power
(
The objective of this study was to evaluate the effects of evacuation from conflict zones on obstetric and neonatal outcomes during a large-scale armed conflict. The main findings were as follows: (1) PPH was significantly less common among evacuated women, and (2) other obstetric and neonatal outcomes, including PTB and LBW, did not differ significantly between evacuated and non-evacuated women.
Unexpectedly, our findings demonstrated a significantly lower incidence of PPH among evacuated women compared with non-evacuated controls, both before and after PSM. Although psychological stress has been linked to adverse obstetric outcomes such as PTB and LBW [10, 14, 15, 16], there is currently insufficient evidence to support a direct association between stress and altered risk of PPH. From a physiological perspective, acute stress may influence hemostatic pathways or uterine contractility, thereby affecting bleeding risk [27]. However, the lower PPH rates observed among evacuated women are more plausibly explained by systemic factors, such as heightened clinical vigilance, timely obstetric intervention, and more proactive intrapartum care provided to women perceived as high-risk due to their evacuation status. This finding may also reflect the strength and adaptability of the Israeli healthcare system, which maintained high standards of maternal care even under the strain of conflict and mass civilian displacement. Another important consideration is the potential influence of unmeasured confounders. Our study did not account for several clinically relevant factors known to affect PPH risk, including prophylactic or therapeutic use of low molecular weight heparin (LMWH), a prior history of PPH, multiple gestation, labor induction or augmentation, underlying coagulopathies, uterine overdistension (e.g., due to polyhydramnios or macrosomia), differences in obstetric management strategies such as rates of labor induction or third-stage management, and other obstetric complications known to be associated with PPH incidence [27, 28]. Differences in the distribution of these factors between the evacuated and control groups may have contributed to the observed association and introduced bias. We performed a quantitative bias analysis using the E-value. After refining the PSM to include clinically relevant comorbidities (preexisting diabetes mellitus [DM] and chronic hypertension entered separately), the association between evacuation and PPH remained robust. The E-value for the observed OR was 5.98, indicating that an unmeasured confounder would need to be associated with both evacuation status and PPH by an OR of nearly six, beyond the measured covariates, to fully explain the observed association. This finding suggests substantial robustness to unmeasured confounding. Nevertheless, given the relatively small sample size and retrospective study design, further research is required to determine whether evacuation is associated with a true reduction in PPH risk or whether this finding reflects residual confounding or random variation.
In our analyses, no statistically significant difference in overall PTB rates was observed between evacuated and non-evacuated women after PSM. Although unadjusted analyses showed a higher proportion of late PTB (34 to 36+6 weeks) among evacuated women, this association did not remain statistically significant after adjustment and should therefore be interpreted with caution. Previous studies have suggested that exposure to prenatal stress may be associated with increased risk of PTB [5, 7, 8, 12, 14, 15, 29]. A large meta-analysis encompassing over 5 million pregnancies reported that stressful life events were associated with a 20% increase in PTB risk [5]. Similarly, maternal psychological distress, including depression, anxiety, and perceived stress, has been associated with an increased risk of PTB, particularly when internal and social coping resources are insufficient, potentially due to physiological disruptions that affect pregnancy continuity [7, 8]. The association between conflict-related stress and PTB has been observed during military operations; Israeli studies reported significantly higher rates of PTB among women exposed to repeated missile attacks [14, 15]. These findings are consistent with results from international studies, which also support an association between war exposure and increased rates of PTB [12, 29]. In contrast, studies from Bosnia did not demonstrate the same effect [13, 30], highlighting variability likely influenced by context, population characteristics, and study design. In the present study, we did not observe a statistically significant increase in PTB rates among evacuated women after adjustment. This finding may suggest that evacuation, as a protective public health measure, mitigated some of the harmful effects of conflict exposure, such as direct physical danger or severe psychological stress, and thereby attenuated potential risks. In addition, the adaptability of the Israeli healthcare system and its capacity to rapidly prioritize and deliver high-quality obstetric care to evacuated women may have contributed to maintaining overall favorable pregnancy outcomes. If confirmed in larger studies, this finding would have important implications for public health responses in conflict settings, highlighting evacuation as a potentially neutral or even protective intervention with respect to certain obstetric outcomes. Conversely, the absence of a statistically significant difference may reflect limited statistical power rather than a true lack of association, particularly given the relatively small number of evacuated women included in the cohort. In unadjusted analyses, higher rates of PTB were observed among evacuated women, which may be biologically and clinically plausible in light of prior evidence that links conflict-related stress to adverse pregnancy outcomes. Potential mechanisms include psychosocial stress and disruptions in access to medical care during evacuation, such as loss of continuity with a primary obstetric provider and reduced access to familiar healthcare facilities. These disruptions may delay routine antenatal monitoring, early identification of complications, and timely intervention, all of which are critical for the prevention of PTB [3, 4].
Other adverse obstetric outcomes examined in this study, such as birth type, LBW, PPROM, GDM, HDP, and placental abruption, did not differ significantly between evacuated and non-evacuated women. While a large systematic review reported a consistent association between armed conflict and increased risk of LBW [10], this association has been challenged by other studies [15, 31]. Moreover, evidence regarding other obstetric outcomes remains limited and inconclusive, partly because these outcomes have not been adequately studied in the context of armed conflict, particularly among evacuated populations, where existing findings remain inconsistent [10, 13, 14, 16, 32]. The absence of significant differences in these outcomes may reflect the effective clinical response and the adaptability of the healthcare system, including proactive care for this vulnerable population. These findings should be interpreted in the context of a well-resourced healthcare system with nationwide coverage, advanced obstetric and neonatal services, and a high level of emergency preparedness, such as that present in Israel. These features may distinguish this setting from other conflict-affected regions with more limited healthcare infrastructure [33]. Nonetheless, the relatively small sample size may have limited the statistical power to detect more subtle associations.
Several limitations warrant consideration. First, the retrospective design inherently limits causal inference and is subject to biases due to missing or incomplete data in EMRs. Second, the relatively small number of evacuated women may have reduced the study’s statistical power, particularly for the detection of differences in less common outcomes (Supplementary Tables 3,4). Third, psychological stress was inferred from geographic exposure and evacuation status rather than directly measured at the individual level, which may have led to exposure misclassification or underestimation of stress severity. Although the data were derived from nine hospitals, hospital identifiers were not available in the analytic dataset due to anonymization. Therefore, hospital-level clustering could not be modeled directly. Potential within-center correlation and center-level variation in practice patterns are acknowledged as limitations. Moreover, evacuation was treated as a binary exposure, although the actual exposure experience was likely heterogeneous with respect to timing during pregnancy, duration of evacuation, living conditions, and gestational age at exposure. Specifically, while some women delivered shortly after the onset of the conflict, others remained pregnant throughout the six-month study period. This resulted in variable exposure duration and timing, which may differentially influence obstetric and neonatal outcomes and could not be fully accounted for in the present analysis. Finally, pregnancies complicated by perinatal mortality, including intrauterine fetal demise, were excluded because definitions varied across participating centers and individual case validation was not feasible in the context of anonymized data.
Despite these limitations, this study has several methodological strengths. It utilizes a large, multicenter cohort drawn from nine hospitals across diverse regions, enhancing the generalizability of the findings to the broader Israeli population. The use of data from EMRs allowed for the comprehensive evaluation of a wide range of obstetric outcomes. Importantly, the application of PSM helped minimize baseline differences and the effects of confounders, thereby improving the validity of the comparisons. Moreover, by focusing on a clearly defined exposure, evacuation due to armed conflict, the study provides specific insights into the effects of acute war-related stress on maternal and neonatal health.
In conclusion, no significant differences were observed in the obstetric and neonatal outcomes examined between evacuated and non-evacuated women, except for a lower rate of PPH among evacuated women. Although these findings require further validation, they highlight the complex mechanisms through which acute stress and displacement may influence obstetric outcomes. Further research is required to better understand these associations and to inform clinical preparedness for the management of pregnancies in populations affected by armed conflict.
The datasets generated and analyzed during the current study are not publicly available due to institutional data protection policies. De-identified data may be made available from the corresponding author upon reasonable request and pending approval by Clalit Health Services.
RB, UA, OW, and RM designed and planned the study, performed data analysis, and wrote the manuscript. IR and AT contributed to the study design and manuscript writing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Ethics Committee of Clalit Health Services (IRB number: 0117-23-COM2). Due to the retrospective nature of the study and use of de-identified data, informed consent was waived. The study was carried out in accordance with the guidelines of the Declaration of Helsinki.
We thank Arnona Ziv and Carmit Rubin from the Data and Analytics Division at Sheba Medical Center for their contribution to the statistical analysis. This publication is part of the doctoral research conducted at Bar-Ilan University in partial fulfillment of the requirements for the Doctor of Philosophy (PhD).
This work was supported by the President of Israel’s Excellence and Innovation Grant in the field of Medical Science and Technology and by a doctoral research grant from the National Insurance Institute of Israel, awarded to Dr. Roy Bitan.
The authors declare no conflict of interest.
The authors used ChatGPT (OpenAI, GPT-5.3) to assist in language editing, organization, and phrasing of portions of the manuscript. All content generated using AI was thoroughly reviewed, verified, and fully approved by the authors, who assume full responsibility for the accuracy and integrity of the work.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG48423.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


