1 Department of Obstetric Nursing, West China Second University Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
2 Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, 610041 Chengdu, Sichuan, China
Abstract
Oxytocin is the first-line uterotonic for the prevention of postpartum hemorrhage (PPH), yet the optimal dosage remains uncertain. We conducted a systemic review to assess the preventive effects of high- and low-dose oxytocin on PPH and its impact on clinical outcomes.
Randomized controlled trials (RCTs) comparing high- versus low-dose oxytocin for PPH prevention were systematically searched in PubMed, China National Knowledge Infrastructure (CNKI), Embase, Web of Science, the Cochrane Library, Ovid Technologies (OVID), and Wanfang databases up to April 2025. The data were pooled using Review Manager (RevMan), version 5.3. The primary outcome was the incidence of PPH, and secondary outcomes included adverse outcomes.
6 RCTs involving 3615 women (1705 receiving high-dose oxytocin; 1910 receiving low-dose oxytocin) were included. High-dose oxytocin significantly reduced the incidence of PPH compared with the low-dose regimens (odds ratio [OR] = 0.44, 95% confidence interval [CI]: 0.32–0.62, p < 0.00001). There was no significant difference in adverse outcomes between groups (p > 0.05).
High-dose oxytocin is more effective than low-dose regimens in preventing PPH without increasing adverse events, supporting dose optimization in clinical protocols.
Keywords
- oxytocin
- postpartum hemorrhage
- different doses
Postpartum hemorrhage (PPH) is a common and serious complication in high-risk parturients and one of the leading causes of parturient death [1]. A relevant study has shown that PPH accounts for 80% of maternal deaths in China [2]. There are many causes of PPH, including uterine atony, soft birth canal injury, abnormal coagulation function and placental factors, among which uterine atony is the most common [3, 4]. In recent years, advancements in obstetric technology have reduced the maternal mortality rate directly caused by PPH; however, it remains a main cause of maternal death [5]. In view of this, clinicians need to develop scientific prevention strategies and take timely drug prevention measures based on the specific conditions of the mother to reduce bleeding and the risk of death. Oxytocin is a first-line drug during the perinatal period. It can effectively promote uterine contractions and exert hemostatic effects through intravenous or intramuscular injection [6]. Intramascular injection of oxytocin is recommended by the World Health Organization (WHO) as the standard treatment for preventing PPH [6]. However, due to the certain saturation of its receptor, uterine contractions may also produce a capping effect. High-dose use not only fails to achieve the expected uterine contraction effect but also increases the risk of complications such as arrhythmia and hypotension [7, 8]. Although there are domestic and international systematic reviews on the preventive effect of oxytocin on PPH, there are currently no evaluations of the efficacy and clinical outcomes of different administration doses. In recent years, studies on high- and low-dose oxytocin have gradually increased, but the results are inconsistent. International guidelines consistently recommend oxytocin as the first-line uterotonic for preventing PPH, but the recommended doses vary by country [8, 9, 10]. The American College of Obstetricians and Gynecologists (ACOG) advises administration of 10 IU of oxytocin intramuscularly after delivery of the anterior shoulder or placenta for prophylaxis. The Royal College of Obstetricians and Gynaecologists (RCOG) in the UK recommend 10 IU intramuscular oxytocin for active management of the third stage of labor and notes that higher intravenous infusion regimens may be considered for cesarean births. The Society of Obstetricians and Gynaecologists (SOGC) of Canada endorses 10 IU intramuscular oxytocin or 20–40 IU in 1000 mL crystalloid solution as the standard to prevent PPH [9, 10, 11, 12, 13]. However, despite its widespread use, there remains no consensus on the optimal oxytocin dosing regimen for prevention of postpartum hemorrhage.
These variations underline the lack of consensus on optimal dosing, providing the rationale for meta-analytic comparisons of high- and low-dose oxytocin regimens.
The present study systematically retrieved relevant studies and estimated the effects of different doses of oxytocin on prevention of PPH and clinical outcomes. The aim of this study was to provide a systematic review and meta-analysis of PPH prevention in clinical practices.
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
This systematic review contains studies that met the following inclusion
criteria: (1) Research type: This research included randomized controlled trials
(RCTs) published both domestically and internationally, regardless of whether
blinding was used. (2) Research subjects: All the parturients selected within
this study were those who took oxytocin to prevent PPH. (3) Intervention
measures: RCTs evaluating different doses of oxytocin. Oxytocin administration
was initiated either after delivery of the anterior shoulder, following placental
expulsion, or immediately after cesarean closure, depending on individual trial
protocols. (4) Primary outcome measure: Incidence of PPH. Secondary outcome
measure: Adverse reactions. For consistency, “low-dose” oxytocin was defined as
standard prophylactic regimens commonly recommended in international guidelines
(
Literature retrieval was conducted through PubMed, China National Knowledge
Infrastructure (CNKI), Embase, Web of Science, Cochrane Library, Ovid
Technologies (OVID) and Wanfang databases. The literature search time frame
spanned from the database’s establishment to April 2025. The search strategy
combined subject terms and free terms. The English search strategy used the
keywords, “oxytocin”, “dosage”, “postpartum hemorrhage”, “random”,
“randomized controlled trials (RCTs)”. The Chinese search strategy used the
keywords “oxytocin”, “postpartum hemorrhage”, “dose” and “randomized
control”. Thresholds were defined following WHO (2022) and ACOG (2023)
recommendations, where a high dose exceeds 10 IU and a low dose is
Two researchers independently screened the literature through title and determine the eligibility from the extracted data. Disagreements regarding the inclusion of extracted data from the literature were resolved by consensus with the third researcher. The EndNote software 2025.3.1 (Clarivate, Philadelphia, PA, USA) was used for literature screening and deduplication. Secondary screening of the selected literature meeting the inclusion criteria was conducted by reviewing abstract, followed by full-text reading, to determine the final literature to be included. The content extracted from the literature mainly includes the author’s name, publication dates, sample sizes and other relevant details. The literature search was completed in March 2025, with inclusion of one early online publication from April 2025. All data were obtained directly from published RCTs; no additional author’s contact was required.
The quality of the included studies was assessed by applying the Cochrane Collaboration’s risk of bias assessment tool. This included (1) random sequence generation methods (computer random number generator, random number table); (2) hidden distribution (central distribution, opaque sealed envelopes); (3) blinding of researchers and participants (single-blind or double-blind); (4) blinded evaluation of the research outcomes (whether blinding was adopted or not); (5) integrity of outcome data and selective reporting bias in study results; (6) other biases.
Data were analyzed using Review Manager (RevMan), version 5.3 (The Cochrane
Collaboration, London, UK). For measurement outcomes, effect sizes were expressed
as mean difference (MD) with 95% confidence intervals (CI). For categorical
outcomes, odds ratios (OR) with 95% CI was calculated. Heterogeneity was
assessed using I2 statistic and Chi-square test. Results were considered
homogeneous when p-value was
A total of 746 records were identified through database searches (PubMed, CNKI, Embase, Web of Science, Cochrane Library, OVID and Wanfang). After removing 158 duplicates, 588 unique records were screened. Following screening, 546 records were excluded for not fulfilling the eligibility criteria. After full text screening of 42 records, 36 records were further excluded from the study. Therefore, 6 RCTs met the inclusion criteria and were included in the meta-analysis (Fig. 1).
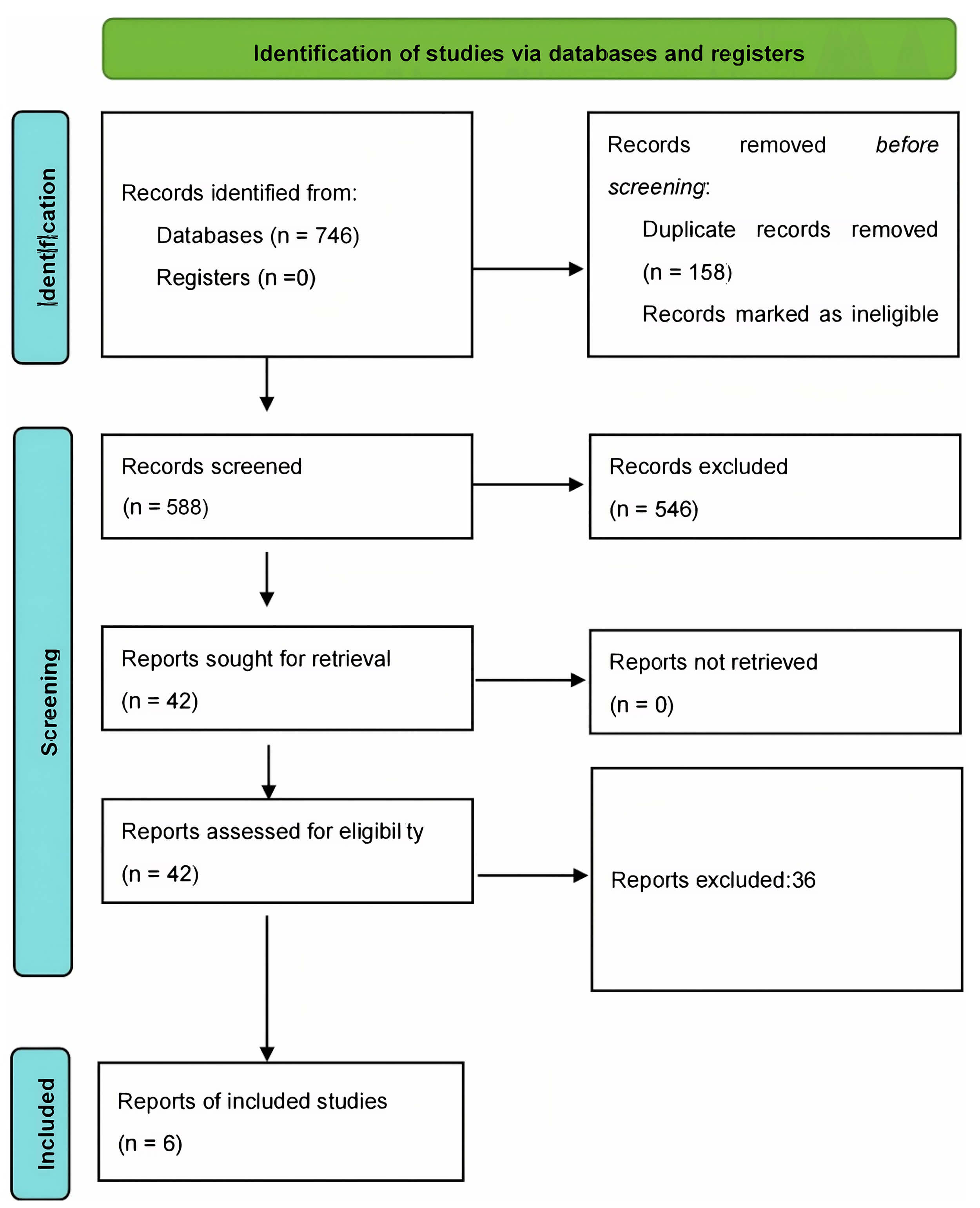 Fig. 1.
Fig. 1.
PRISMA Flowchart. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-analyses.
Among the 6 records included in the analysis, 2 were Chinese articles and 4 were foreign language articles, all of which were RCTs. There were 3615 subjects among them (1705 receiving high-dose oxytocin group and 1910 receiving low-dose oxytocin group). The studies included were published between 2012 and 2025 (Table 1, Ref. [9, 10, 11, 12, 13, 14]). The literature was evaluated using the Cochrane quality evaluation standard (Fig. 2).
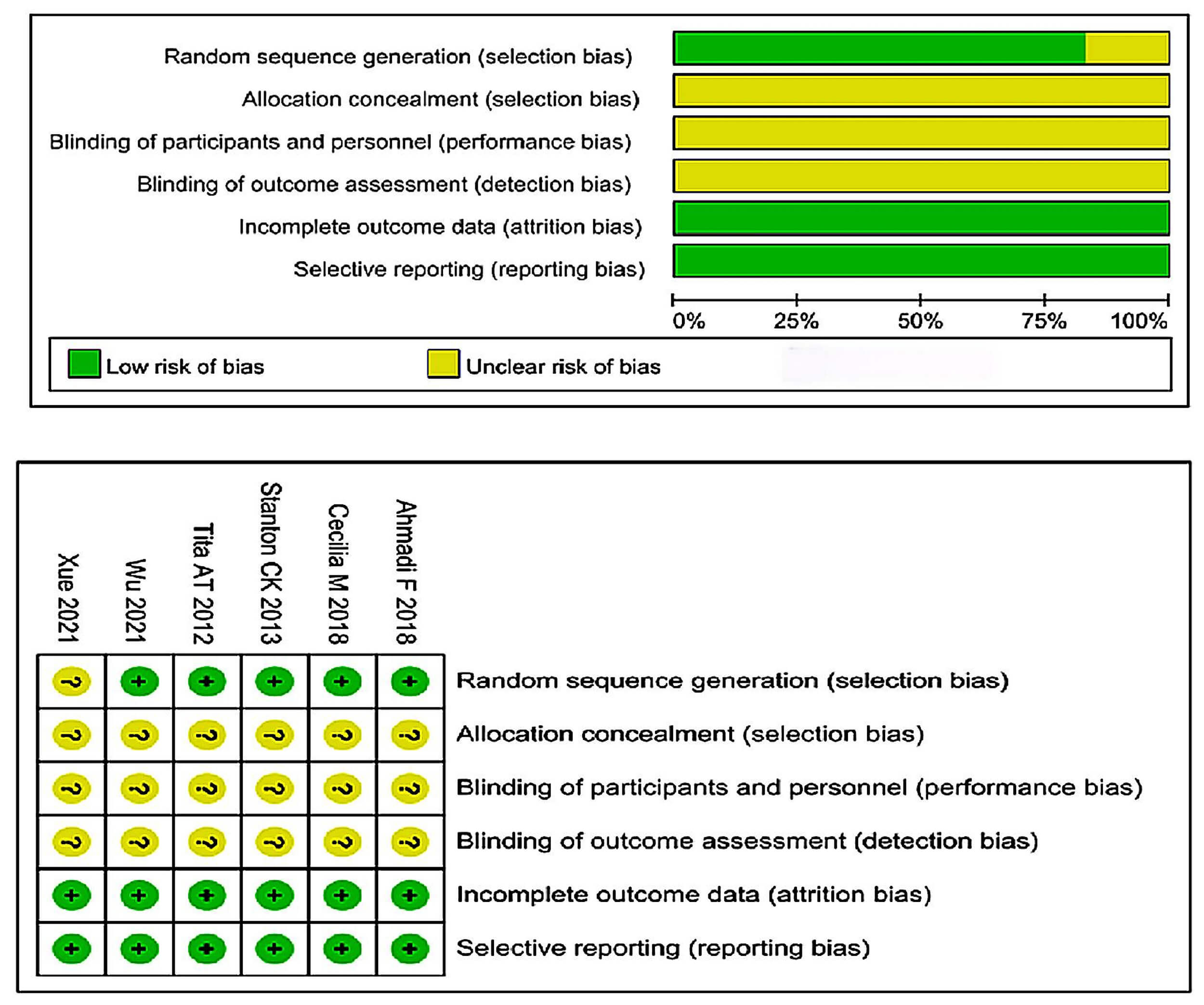 Fig. 2.
Fig. 2.
Risk of bias assessment of included RCTs. RCT, randomized controlled trial. For each bias domain, the symbol “+” denotes low risk of bias, whereas “?” denotes unclear risk of bias, based on the Cochrane Collaboration risk-of-bias tool.
| Study | Year | Design | Intervention (n) | Control (n) | Observation index |
| Stanton CK et al. [9] | 2013 | RCT | high dose (682) | low dose (887) | PPH incidence |
| Cecilia M et al. [10] | 2018 | RCT | high dose (136) | low dose (135) | PPH incidence, Incidence of adverse reactions |
| Ahmadi F [11] | 2018 | RCT | high dose (75) | low dose (75) | PPH incidence |
| Tita ATN et al. [12] | 2012 | RCT | high dose (658) | low dose (659) | PPH incidence, Incidence of adverse reactions |
| Xue [13] | 2021 | RCT | high dose (51) | low dose (51) | PPH incidence |
| Wu and Yang [14] | 2020 | RCT | high dose (103) | low dose (103) | PPH incidence, Incidence of adverse reactions |
RCT, randomized controlled trial; PPH, prevention of postpartum hemorrhage.
Table 2 (Ref. [9, 10, 11, 12, 13, 14]) summarizes how each trial defined high-dose and low-dose oxytocin including the route and timing of administration and whether participants delivered vaginally or via cesarean section (C-section). The details allow for a transparent comparison of dose categorizations across studies and reduce interpretive heterogeneity.
| Study (Author, Year) | High-dose definition | Low-dose definition | Mode of delivery | Justification |
| Stanton et al., 2013 [9] | 10 IU IM by community officers | Standard facility-based regimen (5 IU IM) | Vaginal | Classified as high due to higher prophylactic dose |
| Cecilia et al., 2018 [10] | 30 IU IV over 8–12 h | 10 IU IV over 2–4 h | Cesarean | Dose clearly categorized |
| Ahmadi, 2018 [11] | 80 IU IV infusion | 10 IU IV infusion | Cesarean | Large dose difference justified |
| Tita et al., 2012 [12] | 40 IU IV infusion | 10 IU IM | Vaginal | Clear cut-off used |
| Xue, 2021 [13] | 30 IU IV infusion | 10 IU IV infusion | Cesarean | Standard vs. high-dose |
| Wu and Yang, 2020 [14] | 20 IU IV infusion | 10 IU IV infusion | Cesarean | Double dose classification |
IU, International Unit; IV, Intravenous; IM, Intramuscular.
Six studies [9, 10, 11, 12, 13, 14] reported the incidence of PPH. The analysis of the 6 studies
showed no significant heterogeneity in statistics (p = 0.17, I2 =
36). After treatment, the results indicated that high-dose oxytocin was
associated with a lower incidence of PPH (OR = 0.44, 95% CI: 0.32–0.62,
p
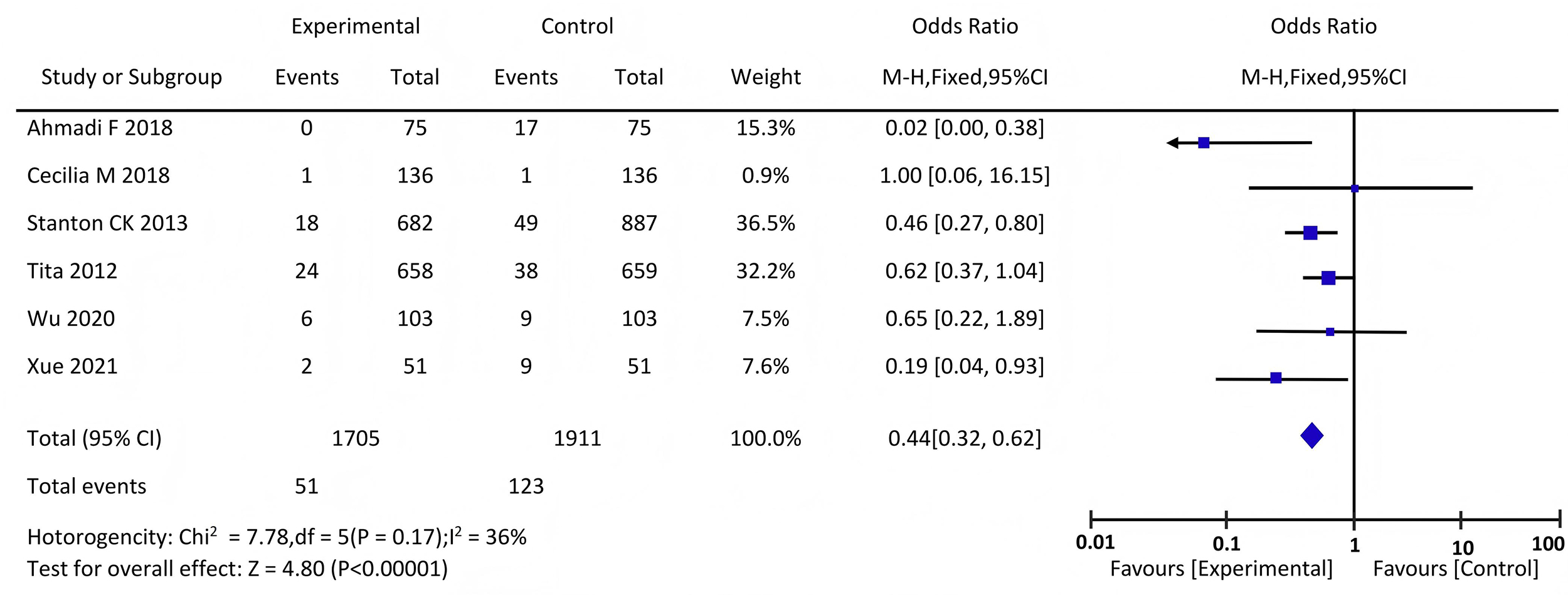 Fig. 3.
Fig. 3.
Forest plot comparing the incidence of PPH in the two groups of participants. PPH, prevention of postpartum hemorrhage.
Three literature sources [10, 12, 14] reported the incidence of adverse
reactions. Analyzing the statistics of the 3 studies revealed no heterogeneity
(p = 0.88, I2 = 0%), and a fixed-effect model was applied. After
treatment, there was no statistically significant difference in the incidence of
adverse reactions between the two groups (p = 0.33
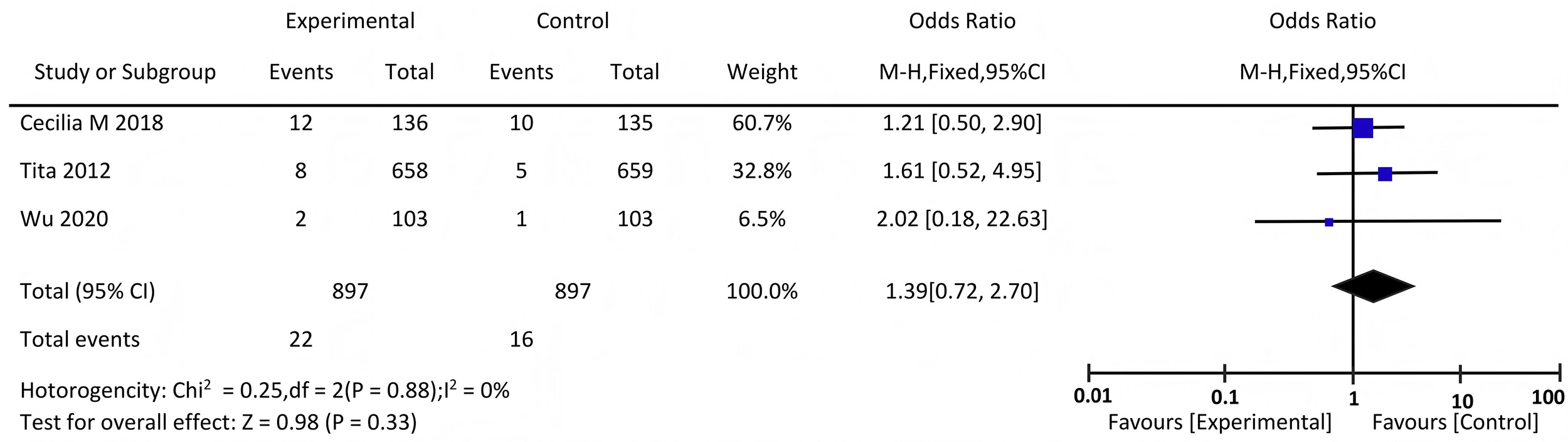 Fig. 4.
Fig. 4.
Forest plot comparing the incidence of adverse reactions.
Subgroup analysis stratified by mode of delivery showed that the preventive
effect of high-dose oxytocin was consistent for both vaginal and cesarean births,
although the magnitude of effect was greater in cesarean deliveries. Sensitivity
analysis excluding smaller studies (n
In addition to pooled estimates, we qualitatively summarized adverse reactions reported in the included trials. The most common adverse events were hypotension, nausea/vomiting, and tachycardia. No trial reported severe arrhythmias or maternal deaths attributable to oxytocin. A supplementary forest plot (Fig. 4) illustrates the distribution of adverse outcomes across studies, showing no significant imbalance between high- and low-dose groups.
To investigate the publication bias in this study, 6 records comparing the incidence of PPH with different doses of oxytocin were selected to draw funnel plots. The results of the funnel plots are shown in Fig. 5. The funnel plot was visually symmetrical, and Egger’s test confirmed no evidence of publication bias (p = 0.256), suggesting that the pooled effect is unlikely to be driven by selective reporting. We therefore consider publication bias to be unlikely for the primary outcome. However, as most studies originated from Asia and Africa, caution is required when generalizing results to populations in high-income countries.
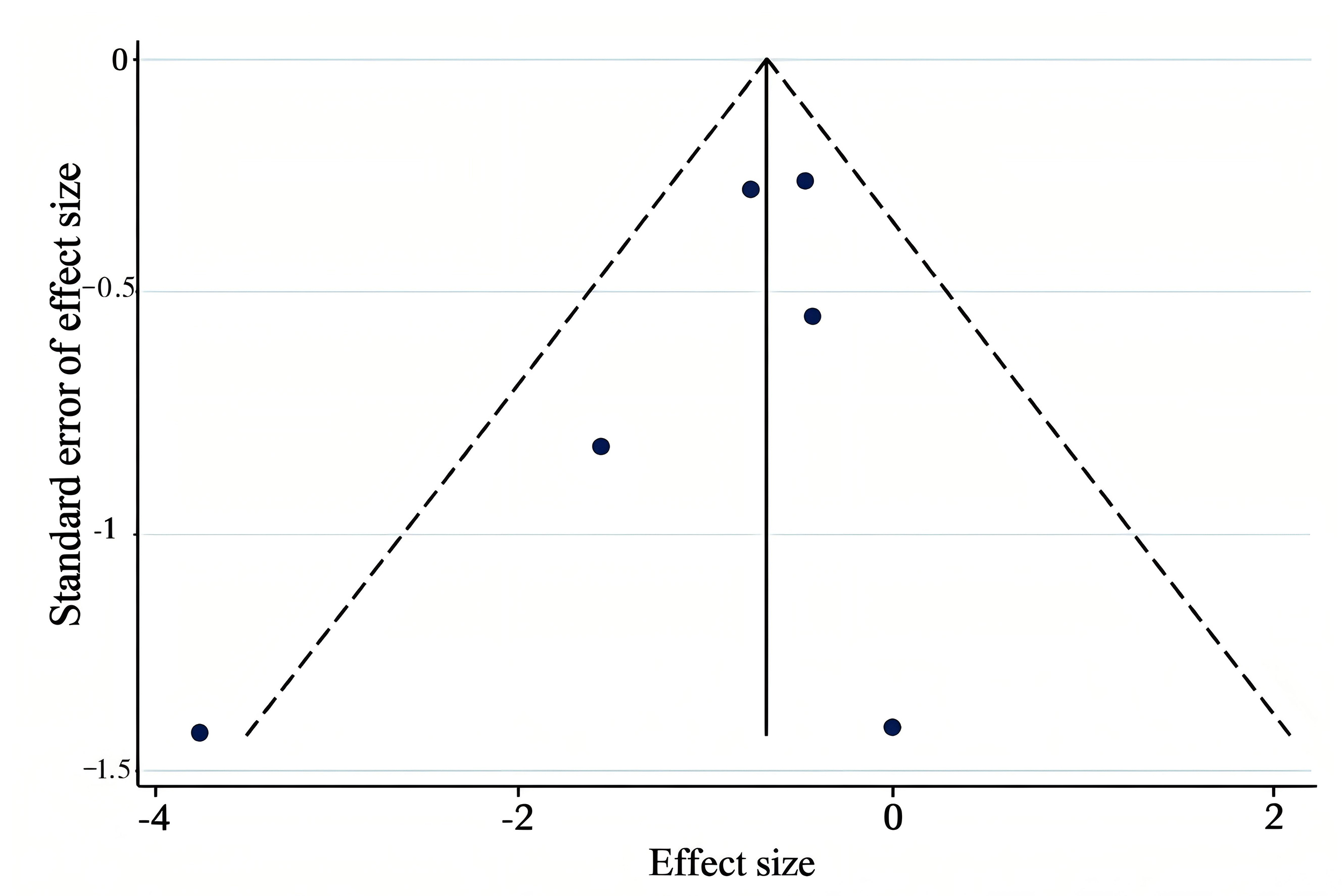 Fig. 5.
Fig. 5.
Funnel plot of publication bias in the incidence.
A prespecified subgroup analysis was performed by both dose range (10–20 IU vs.
This meta-analysis demonstrated that high-dose oxytocin was significantly more
effective than low-dose regimens in reducing the incidence of PPH, without
increasing adverse events. Across six RCTs including 3615 participants, high-dose
oxytocin consistently lowered the risk of PPH (OR = 0.44, 95% CI: 0.32–0.62,
p
Because the majority of included trials were conducted in Asia and Africa, regional variations in obstetric protocols, resource availability, and patient comorbidities may influence the external validity of our findings. In high-income settings where continuous electronic monitoring and standardized uterotonic protocols are routine, the absolute benefit of high-dose regimens might be smaller. Nonetheless, the physiological response to oxytocin is universal, supporting cautious extrapolation.
The reason for consideration might be that a high dose of oxytocin can more effectively promote uterine contractions. Uterine contractions are a key link in preventing PPH. High doses of oxytocin can stimulate uterine smooth muscle to produce stronger and more persistent contractions, thereby more forcefully pressing the blood vessels within the uterine muscle layer and reducing bleeding [19].
Because only three trials systematically reported adverse outcomes and none captured severe arrhythmias or maternal deaths, the safety analysis is underpowered. High-dose oxytocin may still confer risk in women with cardiovascular disease or impaired uterine tone, and future large-scale studies should address this limitation. From the perspective of its mechanism of action, oxytocin binds to receptors on uterine smooth muscle cells to exert its effects. At high doses, it can occupy more receptors and activate a more intense contraction response [20].
While major guidelines advocate standard low-dose oxytocin for routine prophylaxis, they also permit higher doses within recommended therapeutic ranges for cesarean births or refractory bleeding. Our pooled findings indicate that such higher-range doses, when properly supervised, may enhance uterine contraction without elevating risk. Thus, our results complement existing guidelines by refining the evidence base for dose optimization rather than contradicting them.
In addition, high-dose oxytocin can reach the effective blood drug concentration more quickly. After a drug enters the body, it must reach a certain concentration to achieve its optimal effect. Therefore, high-dose oxytocin plays a significant role in the early stage of PPH by activating the uterine contraction mechanism and reducing the risk of bleeding [21]. In contrast, low-dose oxytocin takes a longer time to reach its effective concentration, which may result in increased bleeding during this period. Within this study, the incidence of adverse reactions in high- and low-dose groups was not statistically significant. This indicates that increasing the oxytocin dose within a reasonable range is safe. This result suggests that in clinical practice, an appropriate increase in the dose of oxytocin can be prioritized for preventing PPH without excessive concern about the risk of adverse reactions. High dose of oxytocin not only has a better effect, but also has comparable safety. It is beneficial to optimize PPH prevention plan and improve the clinical treatment effect. This provides a basis for clinicians to choose more effective high-dose oxytocin when preventing PPH, however, excessive doses should still not be used in clinical applications. Relevant studies have shown that excessive oxytocin doses may lead to adverse reactions [22, 23]. High doses of oxytocin may cause spastic contractions of the uterus, which not only bring more intense pain to the parturient but may also affect the blood supply to the uterus, having an adverse effect on uterine recovery [23]. Therefore, when using high-dose oxytocin within a reasonable range in clinical medication, it can effectively prevent PPH without significantly increasing the risk of adverse reactions.
Based on the synthesized evidence, clinicians should consider using high-dose oxytocin regimens within the ranges supported by international guidelines (10–40 IU depending on mode of delivery and administration route). This strategy reduces the risk of PPH without increasing adverse events. Standardization of dose definitions in clinical protocols and future trials will further strengthen the quality of evidence and improve maternal outcomes.
The main strength of this meta-analysis is that it included only RCTs, which represent the highest level of evidence for clinical interventions. A robust methodology was employed, including comprehensive database searches, standardized quality assessment using the Cochrane risk-of-bias tool, and a pooled analysis of more than 3600 participants. Moreover, the consistency of findings across multiple geographic regions and delivery modes is also a major strength of this meta-analysis.
However, several limitations of this study must be acknowledged. Firstly, the number of included trials was relatively small (n = 6), which may limit generalizability. Secondly, there was variation in how individual studies defined “high-dose” and “low-dose” oxytocin, potentially contributing to heterogeneity. Thirdly, most studies were conducted in limited regional contexts, reducing applicability to all health-care settings. Finally, subgroup analysis (e.g., vaginal vs. cesarean delivery) were limited by available data, and future studies should explore these differences more comprehensively.
The results of this meta-analysis provide clinically meaningful evidence that high-dose oxytocin is more effective in preventing PPH than low-dose regimens, without additional safety concerns. This supports the adoption of standardized high-dose protocols within national and international guidelines to reduce maternal morbidity and mortality. Clinicians should evaluate dose optimization with individual patient risk factors, but these findings suggest that concerns over adverse events may be less evident within recommended dose ranges.
For future research, harmonizing dose definitions across trials will be essential to reduce heterogeneity and improve comparability. Large, multicentre RCTs conducted in diverse populations could strengthen the evidence base, particularly for cesarean versus vaginal deliveries. Additional work is needed to report adverse outcomes more systematically, enabling clearer risk–benefit assessments.
This systematic review and meta-analysis described that high-dose oxytocin significantly reduces the incidence of PPH compared with low-dose regimens, without increasing adverse events. These findings support the adoption of standardized high-dose oxytocin protocols in clinical practice to optimize maternal outcomes. In addition, this evidence provides a solid basis for clinicians to prioritize high-dose oxytocin regimens, which achieve stronger uterine contractions and faster therapeutic blood concentrations, thereby offering superior prevention of PPH while maintaining safety.
All data analyzed during the current meta-analysis study were extracted from previously published scientific articles which are cited and listed in the reference section of this manuscript. Therefore, no new primary data were generated, and the underlying data necessary to reproduce our findings are fully available within the cited references.
YQ: Conception and design, patient protocols, supervisions, drafting the manuscript. SZ: Acquisition of data, lab reports, analysis and interpretation of data, revising the manuscript critically for important intellectual content. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGpt-3.5 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG42696.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





