1 Department of Gynecology, Hospital of Chengdu University of Traditional Chinese Medicine, 610072 Chengdu, Sichuan, China
Abstract
Natural polyphenols are plant secondary metabolites with a broad spectrum of biological activities. Extensive preclinical studies have demonstrated the significant therapeutic efficacy of polyphenolic compounds in animal models of endometriosis (EMs).
A comprehensive literature search was conducted in PubMed, Embase, Web of Science, the Cochrane Library, China National Knowledge Infrastructure (CNKI), Chongqing VIP Information Co., Ltd. (or VIP Database) (VIP) Database, and the Wanfang Database from their inception to December 2024. After screening titles, abstracts, and full texts, 43 studies were included. The risk of bias in the included studies was assessed with the Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE) risk of bias tool for animal studies. Meta-analysis of outcome measures was performed using STATA 17.0 software.
Polyphenolic compounds reduced lesion size, histopathological scores, and adhesion scores in ectopic lesions in animal models of EMs. They also decreased peripheral levels of estradiol (E2), tumor necrosis factor-alpha (TNF-α), interleukin-8 (IL-8), interleukin-1β (IL-1β), interleukin-6 (IL-6), monocyte chemoattractant protein-1 (MCP-1), and prostaglandin E2 (PGE2). Furthermore, they inhibited the expression of vascular endothelial growth factor (VEGF), proliferating cell nuclear antigen (PCNA), B-cell lymphoma 2 (BCL-2), and hypoxia-inducible factor-1α (HIF-1α). The underlying mechanisms include anti-inflammatory effects, immune regulation, anti-angiogenesis, antioxidant stress, and alleviation of tissue hypoxia.
The therapeutic effects of polyphenolic compounds in animal models of EMs involve multiple mechanisms, including anti-inflammatory, immunomodulatory, anti-angiogenic, antioxidant stress, and hypoxia-alleviating effects. These findings provide preclinical evidence that supports the potential use of polyphenolic compounds for the treatment of EMs.
The study has been registered on https://www.crd.york.ac.uk/prospero/ (registration number: CRD42024570708; registration link: https://www.crd.york.ac.uk/PROSPERO/view/CRD42024570708).
Keywords
- endometriosis
- polyphenol
- animal model
- meta-analysis
Endometriosis (EMs) is a hormone dependent disease in which the endometrial stroma and glands grow, infiltrate, and repeatedly bleed outside the uterus, leading to chronic pain, masses, and infertility [1]. This condition is highly prevalent among women of reproductive age. A meta-analysis indicates that its overall prevalence may reach approximately 18% [2], which constitutes a significant challenge to women’s health and public health.
The pathogenesis of EMs is not fully understood. Current leading theories include retrograde implantation of menstrual blood [3], embryonic remnants, coelomic metaplasia [4], and in situ endometrial determinism [5]; however, no single theory fully explains its pathogenesis. Based on existing research, the onset and progression of EMs are closely associated with factors such as hormonal, immune, inflammatory, angiogenesis, oxidative stress (OS), abnormal cell proliferation and apoptosis, epithelial-mesenchymal transition, epigenetic alterations, and microbial imbalance [6, 7, 8]. Although considered a benign disease, EMs exhibit biological characteristics similar to those of cancer, manifested as invasiveness to local and distant tissues and resistance to apoptosis [9]. Research has shown that most deep EMs lesions harbor somatic mutations and known cancer-related mutations (CAMs), and in very rare cases, they may progress into cancer [10]. A large amount of epidemiological evidence indicates that women with EMs have a higher risk of developing gynecological-related cancers, autoimmune diseases, and cardiovascular diseases. The mechanism may relate to common risk factors, treatments for EMs, and the potential impact of the disease itself [11]. In addition, chronic pain and infertility caused by EMs not only seriously impair physical and mental health, as well as quality of life, but also impose a heavy economic burden on individuals, families, and society [12, 13].
Treatment options for EMs include pharmacological therapy and surgical treatment. Pharmacological therapy is typically used for long-term management to control the disease progression and prevent postoperative recurrence. Its mechanism involves regulating the hypothalamic-pituitary-ovarian (HPO) axis to inhibit ovulation and maintain a low estrogen state [14]. However, medication-associated adverse effects, such as gastrointestinal reactions, mood changes, menopausal symptoms, and effects on fertility, reduce patient compliance and limit long-term use. Surgery can remove ovarian ectopic cysts, destroy superficial lesions, relieve pain, and improve pregnancy rates, but the postoperative recurrence rates are high, and there is a potential risk of surgical complications. The current treatment plan has certain limitations, and the development of new nonhormonal therapies has been slow. Therefore, there is an urgent need to explore new therapies that are effective, have low adverse effects, and are affordable.
Over the past few decades, natural products and their derivatives have become an important source of new drug development. Many drugs widely used in clinical practice are derived from active plant ingredients, such as acetylsalicylic acid (aspirin), coumarin drugs (warfarin), quinine, opioid drugs, digitalis, and atropine [15, 16]. Compared with chemically synthesized drugs, those derived from natural products exhibit unique structural novelty, biological compatibility, and diverse pharmacological properties, which are attributed to natural selection during their evolutionary process [17]. Natural polyphenols are plant secondary metabolites that are widely present in plants, vegetables, fruits, grains, nuts, tea, red wine, and coffee. To date, more than 8000 polyphenolic compounds have been identified from plants [18], which can be further classified based on the number of benzene rings and the structural elements that connect these rings. Based on the Phenol-Explorer database (http://phenol-explorer.eu/compounds; accessed July 2024), polyphenolic compounds can be broadly classified into five major categories, namely flavonoids, phenolic acids, stilbenes, lignans, and other polyphenols. Polyphenolic compounds exhibit a wide range of biological activities. Numerous in vivo and in vitro studies have shown that they exert significant intervention effects on chronic diseases and malignant tumors, including inhibition of lipid peroxidation, reduction of fibrinogen expression, suppression of vascular cell proliferation, and reversal of tumor suppressor gene methylation [19, 20]. It is worth noting that EMs are characterized by chronic inflammation and share pathological features with malignant tumors, indicating that polyphenolic compounds have potential as alternative therapeutic strategies for this condition [21]. Currently, multiple studies have confirmed that polyphenolic compounds have the potential to treat EMs through several pathways, such as anti-inflammatory, antioxidant, anti-angiogenic, and regulation of cell proliferation and apoptosis [22, 23, 24].
Natural compounds are often considered low risk. However, some components may pose potential toxicity risks due to complex factors such as insufficient purity, unknown adulteration, excessive dosage, and interactions with other drugs [25, 26]. Therefore, clarifying the biological activity and safety of natural products through preclinical evaluation, including in vitro and in vivo studies, is an important prerequisite for their scientific development and effective use. Animal studies are an important approach to evaluate the efficacy and safety of natural products and their metabolites. They can reveal pharmacokinetics and pharmacological mechanisms by simulating the complex internal environment of living organisms. However, results from individual animal studies can be misleading to clinical research due to various bias factors, leading to wasted resources in subsequent clinical trials. Systematic evaluation of animal studies is considered an effective method to explore and strengthen their relevance to clinical research, reduce the risk of misleading results, and avoid unnecessary clinical studies [27]. At present, various animal models of EMs have been established, including primates, rabbits, and rodents. Among them, primates are considered the most suitable model due to their close physiological similarity to humans, but their scarcity and high cost limit large-scale applications. In contrast, rodents are the most widely used animal models in EMs research due to their strong reproductive capacity and ease of handling. At present, three main approaches are used to establish rodent models of EMs, namely spontaneous models based on kirsten rat sarcoma viral oncogene homolog (KRAS) activation, homograft models, and xenograft models [28]. Among these, homograft and xenograft methods are well established.
To date, several systematic reviews have summarized the potential mechanisms of polyphenols in EMs intervention [21, 29]. However, meta-analyses based on evidence from preclinical animal studies remain limited, and few have quantitatively synthesized and evaluated the overall efficacy of polyphenols across multiple pathological indicators, such as lesion volume, inflammatory markers, angiogenesis, and hormone levels. Therefore, to integrate existing preclinical evidence and clarify its potential value, this study addresses the following research question: Do polyphenolic compounds exert significant ameliorative effects on key pathological indicators, such as lesion growth, inflammatory response, and hormone levels, in animal models of EMs? Based on the broad biological activities of polyphenols, we hypothesize that their intervention will positively impact these indicators. To test this hypothesis, a systematic review and meta-analysis of relevant studies will be conducted, aiming to provide a robust evidence base for subsequent translational research on polyphenolic compounds in the treatment of EMs.
This systematic review was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and was registered in the PROSPERO database (CRD42024570708). Two investigators (Shanshan Lu and Peixuan Fan) independently developed the search strategy using a combination of controlled vocabulary (MeSH terms) and free-text terms. Boolean operators (AND/OR) were employed to construct the search strings. Key search terms included “polyphenols”, “flavonoids”, “lignans”, “stilbenes”, “resveratrol”, “quercetin”, “curcumin”, “apigenin”, “baicalein”, “genistein”, and “epigallocatechin gallate”. A comprehensive literature search was performed in the following databases from their inception through December 2024: PubMed, Embase, Web of Science, the Cochrane Library, China National Knowledge Infrastructure (CNKI), the VIP Database, and the Wanfang Database. The detailed search strategy and specific terms are provided in Supplementary Material 1. The search was limited to publications in Chinese and English. The final search was conducted on December 28, 2024.
Inclusion Criteria: (1) The experimental subjects were animal models of EMs,
with no restrictions on species or modeling methods. (2) The intervention in the
experimental group was a single polyphenolic compound; the control group received
saline, vehicle, or no intervention. (3) Outcome measures reflected the impact of
the polyphenolic compound on disease progression in the EMs animal model. Outcome
measures included one or more of the following: lesion volume, histopathological
score, adhesion score, vascular endothelial growth factor (VEGF), interleukin-6
(IL-6), interleukin-8 (IL-8), interleukin-1
Exclusion Criteria: (1) Clinical trials, ex vivo studies, case reports, conference abstracts, reviews, studies unrelated to the topic, and articles without full text. (2) Interventions that involved nonpolyphenolic compounds or combinations of polyphenolic compounds with other therapeutic agents. (3) Studies that did not report the dosage or duration of polyphenolic compound administration. (4) Duplicate publications. (5) Studies with outcome data formats that precluded meta-analysis. (6) Publications in languages other than Chinese or English.
Two researchers (Lingling Yuan and Yijun Hao) independently performed the
literature screening and data extraction. Discrepancies were resolved through
consultation with the corresponding author, Yi Wen. The extracted data included:
the author, publication year, animal species, animal weight or age (weeks),
sample size of the experimental and model groups, modeling method used in the
model group, intervention in the model group, anesthetic agents, intervention in
the experimental group (type of polyphenol, dosage, duration, administration
route), and the type of polyphenol. Outcome data included the mean, standard
deviation (SD) or standard error (SEM), median, interquartile range, or maximum
and minimum values. For data presented as SEM, SD was calculated using the
formula SD = SEM
Two investigators (Wenjing Jiang and Xialin Zhang) independently assessed the quality of the included studies. Any disagreements were resolved through consultation with the corresponding author (Yi Wen). The Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE) risk of bias tool for animal studies [30] was used to evaluate bias. This tool covers bias across six domains: selection bias, performance bias, detection bias, attrition bias, reporting bias, and other biases, comprising a total of 10 items. The results for each item were categorized as “Yes”, “No”, or “Unclear”.
A meta-analysis was performed using STATA 17.0 software (version 17.0; StataCorp
LLC, College Station, TX, USA), and the standardized mean difference (SMD) was
used as the effect size. Given the anticipated biological and methodological
heterogeneity among the included studies, including factors such as animal
strain, age, and experimental protocols, a random-effects model was applied for
data synthesis. Statistical heterogeneity among studies was assessed using
Cochrane’s Q test and quantified with the I2 statistic. To
investigate the sources of heterogeneity, the following approaches were employed.
First, meta-regression analysis was conducted with polyphenol type, dosage,
duration of intervention, route of administration, and animal species as
potential covariates (only for outcomes with more than 10 studies). If a
significant factor was identified, subgroup analysis was further performed.
Second, to evaluate the influence of individual studies on the overall pooled
estimate, a leave-one-out sensitivity analysis was conducted by sequentially
removing one study and performing a meta-analysis on the remaining studies. This
approach assessed whether the original pooled estimate changed significantly due
to the influence of any single study. Publication bias was evaluated with Egger’s
test for outcomes with a sufficient number of studies. If publication bias was
present (p
The initial search yielded 1413 records. After duplicate removal, 1000 articles remained. Two reviewers independently screened the titles, abstracts, and full texts against predefined inclusion and exclusion criteria. Ultimately, 43 studies published between 2007 and 2024 met the criteria, comprising 36 English-language and 7 Chinese-language publications. The detailed selection process is illustrated in Fig. 1.
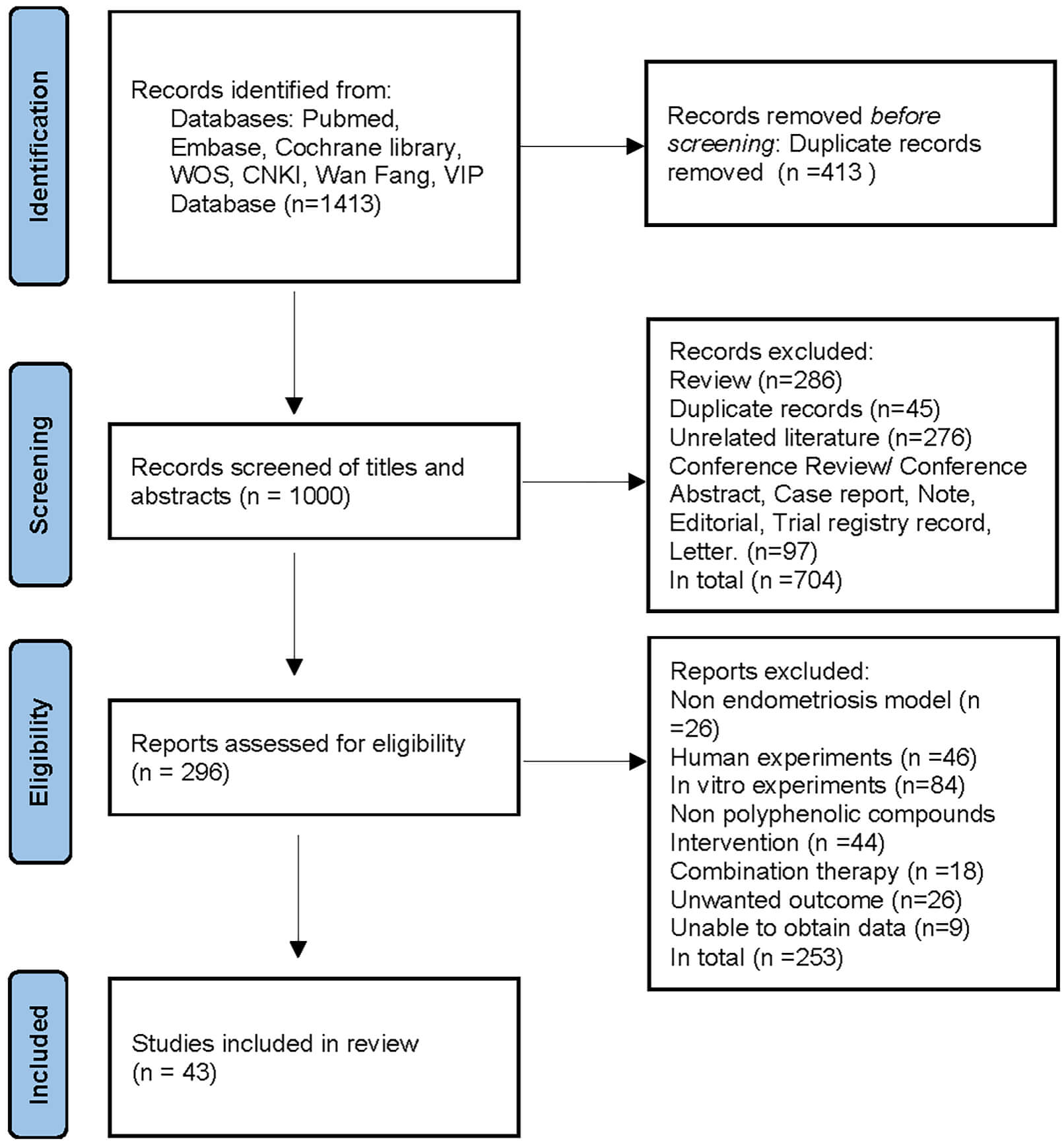 Fig. 1.
Fig. 1.
Flowchart of the literature screening process.
A total of 843 female animal models of EMs were included from the 43 selected studies, with 421 animals assigned to the experimental groups and 422 to the model groups. For animal selection, 16 studies used mice, including strains such as Mus musculus (3 studies), NCr strain (1 study), BALB/c (7 studies), NOD-SCID (3 studies), and C57BL/6 (2 studies). 27 studies used rats, including Sprague Dawley rats (17 studies) and Wistar rats (10 studies). The body weight of mice ranged from 18–30 g, and their age ranged from 6–8 weeks. The body weight of rats ranged from 200–250 g. Only 1 study did not report the body weight or age of the animals.
With respect to modeling methods, two primary approaches were identified: autologous endometrial tissue transplantation and heterologous endometrial tissue transplantation (from human donors or donors of the same species). Regarding anesthetic agents, 31 studies reported at least one type of anesthetic. Among these, ketamine was used in 14 studies, xylazine in 12, sodium pentobarbital in 9, chloral hydrate in 4, rompun in 2, isoflurane in 2, zoletil in 1, and urethane in 1. 12 studies did not specify the type of anesthetic used.
For interventions, the experimental groups received polyphenol compounds, while the model groups received normal saline, vehicle, or no intervention. The polyphenols used were diverse and included resveratrol, curcumin, epigallocatechin gallate, hesperidin, nobiletin, puerarin, wogonin, quercetin, xanthohumol, scutellarin, genistein, fisetin, apigenin, silymarin, isoliquiritigenin, and naringenin.
The outcome measures included ectopic lesion volume, histopathological score,
adhesion score, estradiol (E2), progesterone, monocyte chemoattractant protein-1
(MCP-1), hypoxia-inducible factor-1
The quality assessment of the included studies is detailed in Supplementary Material 4. All studies employed at least one method to control for bias. 33 studies reported random allocation, whereas only 1 explicitly described the method of random sequence generation and allocation concealment. Animal housing randomization was mentioned in 35 studies. 2 studies implemented investigator blinding. 1 study reported random selection of animals for outcome assessment. 11 studies implemented blinding of outcome assessors. In summary, all included studies had an unclear risk of bias in one or more domains, and no study was rated as low risk across all domains. This assessment suggests that the results of this meta-analysis should be interpreted with caution.
A total of 25 articles comprising 28 studies, reported the effects of polyphenolic compounds on the growth of endometriotic lesions. The results demonstrated that, compared with the model group, polyphenolic compounds significantly inhibited the growth of ectopic lesions in animal models of EMs, with a statistically significant difference (SMD = –3.06, 95% confidence interval [CI]: –3.83, –2.29; heterogeneity: I2 = 86.3%, p = 0.000) (Fig. 2).
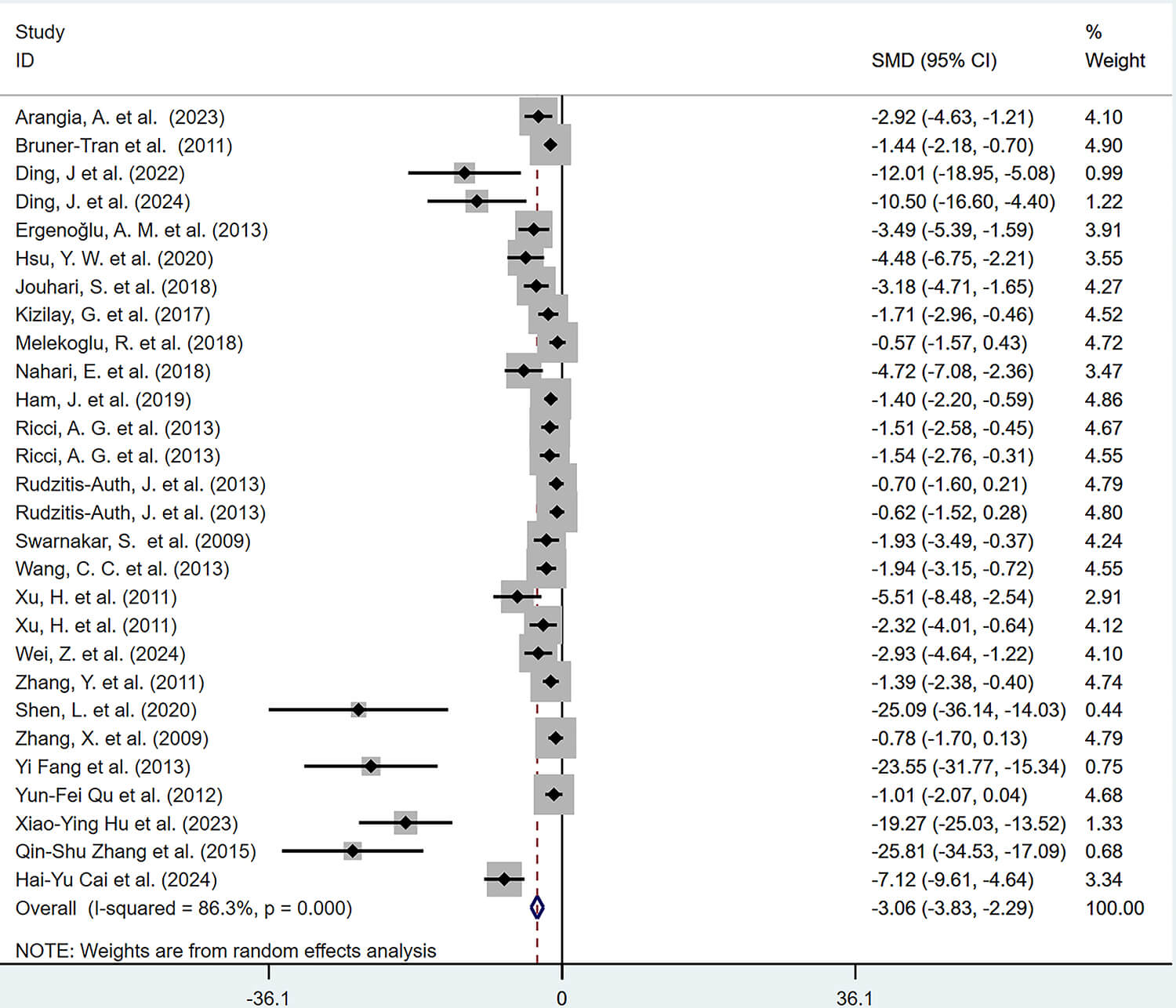 Fig. 2.
Fig. 2.
Forest plot of the effects of polyphenolic compounds on endometriotic lesions. CI, confidence interval; SMD, standardized mean difference.
6 studies, comprising 7 reports, investigated the effects of polyphenolic compounds on the histopathological scores of lesion tissues. The results demonstrated that, compared with the model group, polyphenolic compounds significantly reduced the histopathological scores of ectopic lesions in animal models of EMs, with a statistically significant difference (SMD = –1.31, 95% CI: –1.81, –0.81; heterogeneity: I2 = 1.3%, p = 0.399) (Fig. 3).
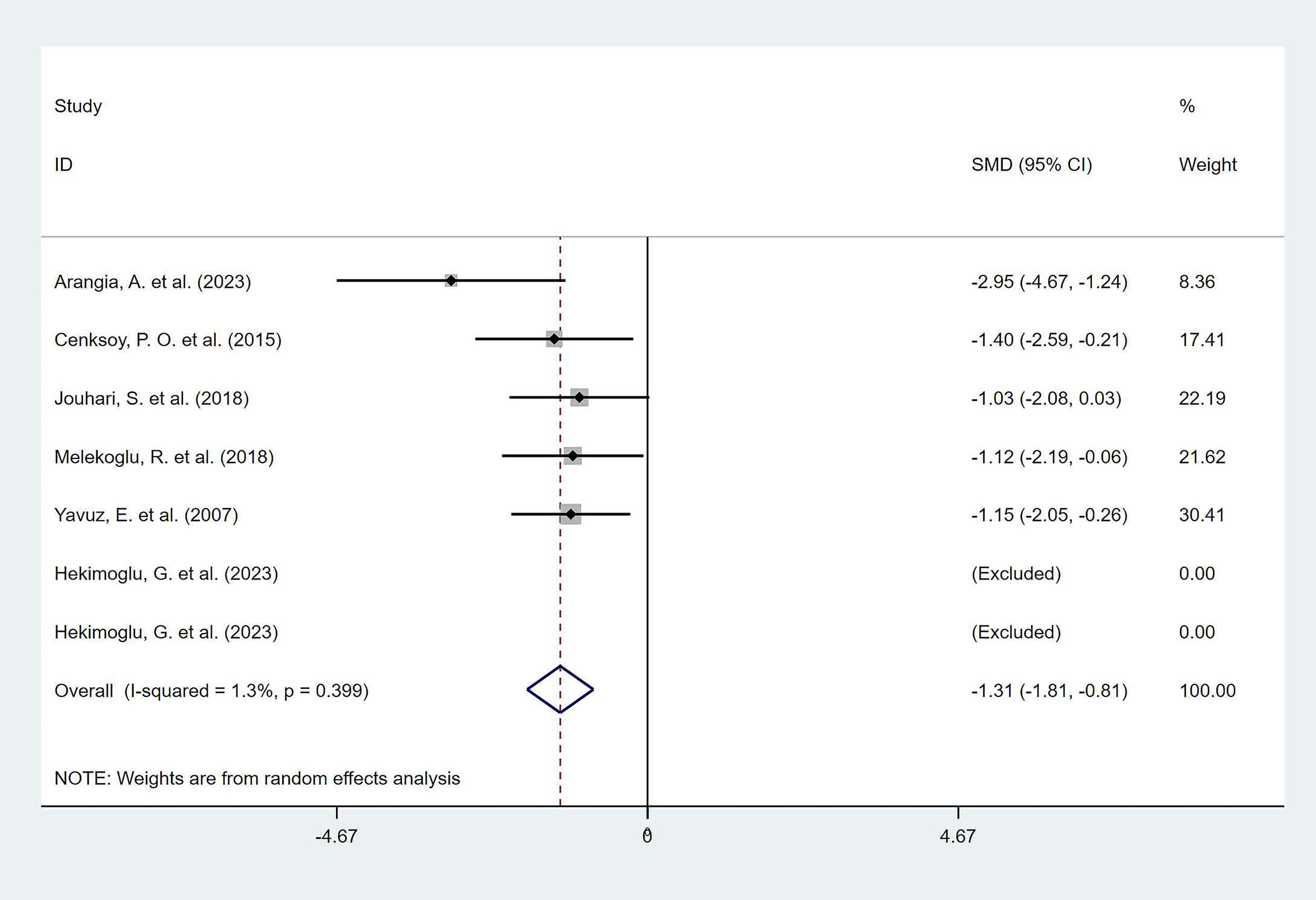 Fig. 3.
Fig. 3.
Forest plot of the effect of polyphenols on the histopathological score of ectopic lesions.
6 studies, comprising 6 reports, examined the effects of polyphenolic compounds on adhesion scores. The results indicated that, compared with the model group, polyphenolic compounds significantly reduced the severity of intra-abdominal adhesions, with a statistically significant difference (SMD = –3.14, 95% CI: –4.71, –1.57; heterogeneity: I2 = 82.0%, p = 0.000) (Fig. 4).
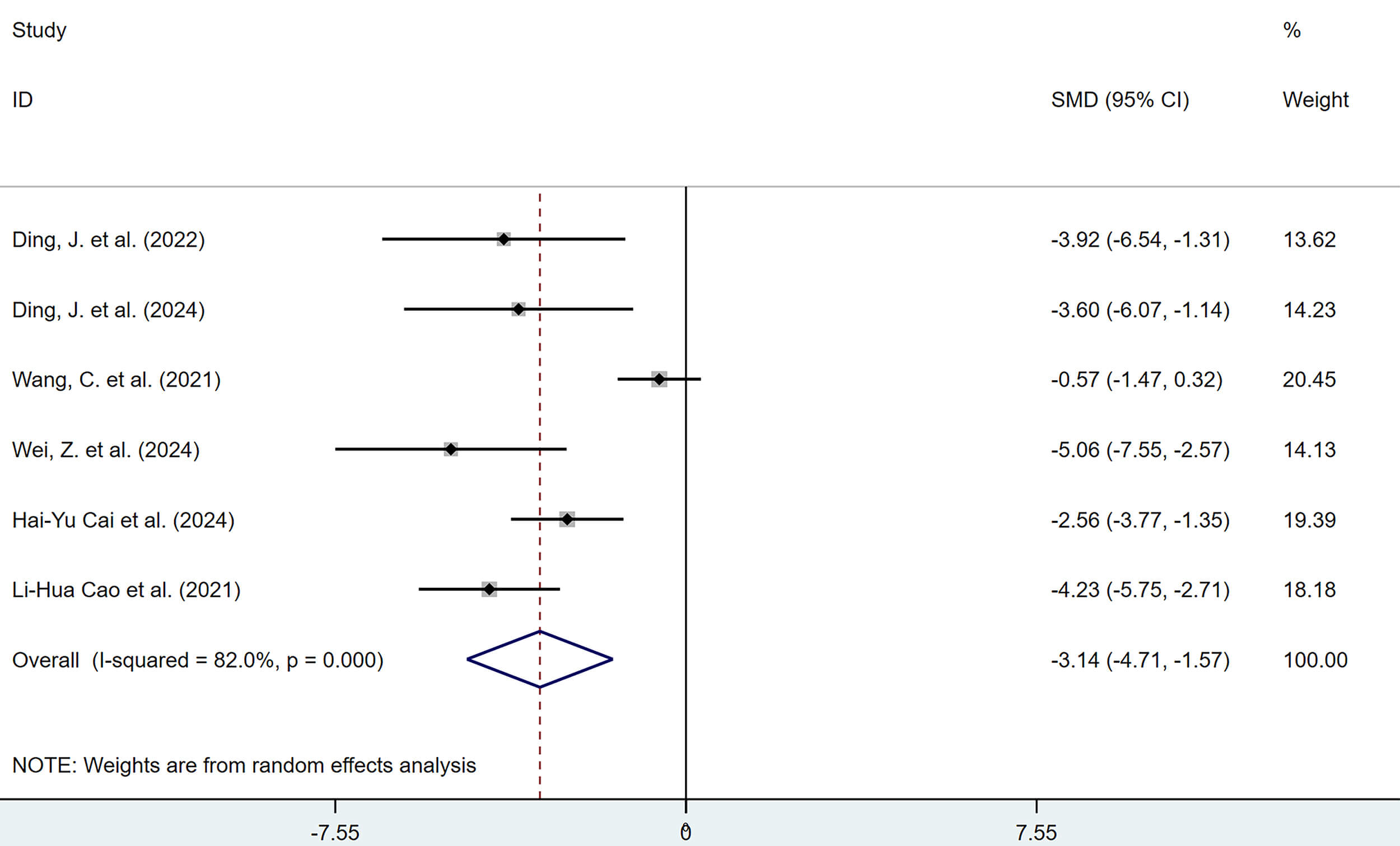 Fig. 4.
Fig. 4.
Forest plot of the effects of polyphenols on peritoneal adhesion scores.
3.3.4.1 Serum Estrogen Levels
6 studies reported the effects of polyphenolic compounds on serum E2 levels in animal models of EMs. The results demonstrated that, compared with the model group, polyphenolic compounds significantly reduced serum E2 levels in EMs animal models, with a statistically significant difference (SMD = –7.24, 95% CI: –10.88, –3.60; heterogeneity: I2 = 95.6%, p = 0.000) (Fig. 5).
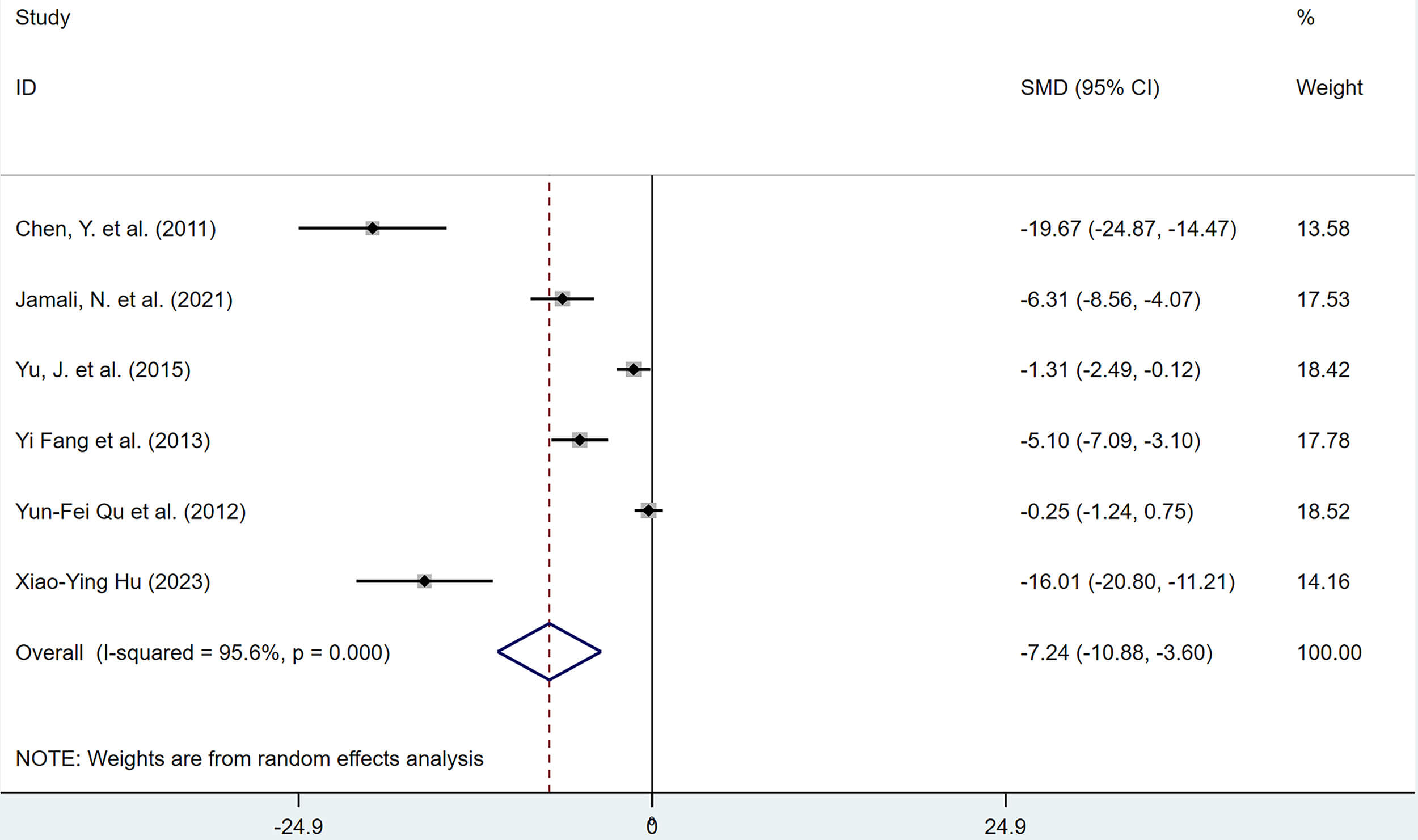 Fig. 5.
Fig. 5.
Forest plot of the effects of polyphenols on serum estradiol (E2) levels.
3.3.4.2 Serum Progesterone Level
3 studies reported the effects of polyphenolic compounds on serum progesterone levels in animal models of EMs. The results showed no significant difference in serum progesterone levels between the polyphenol-treated and the model group (Fig. 6).
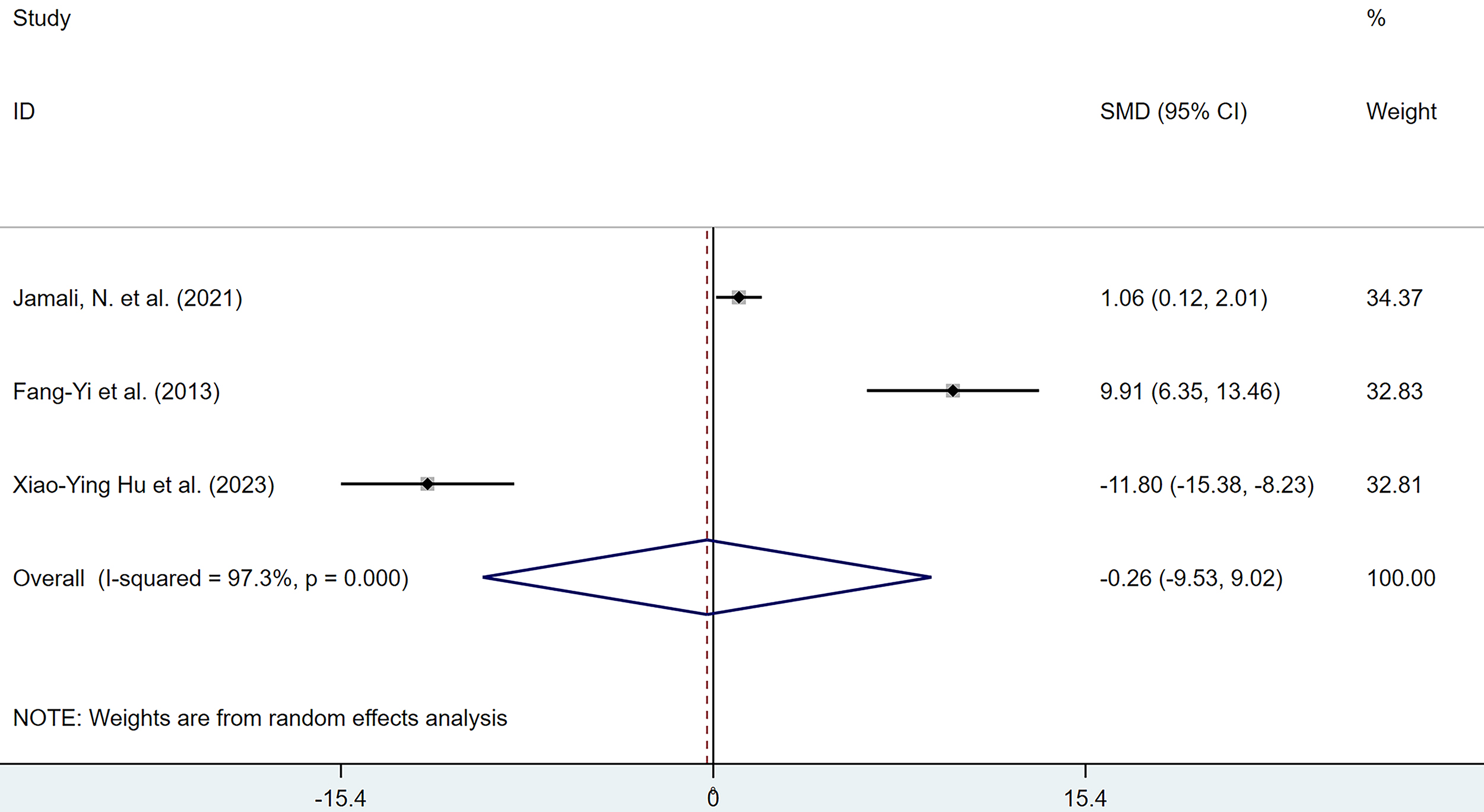 Fig. 6.
Fig. 6.
Forest plot of the effects of polyphenols on serum progesterone levels.
3.3.5.1 TNF-
6 studies reported the effects of polyphenolic compounds on serum TNF-
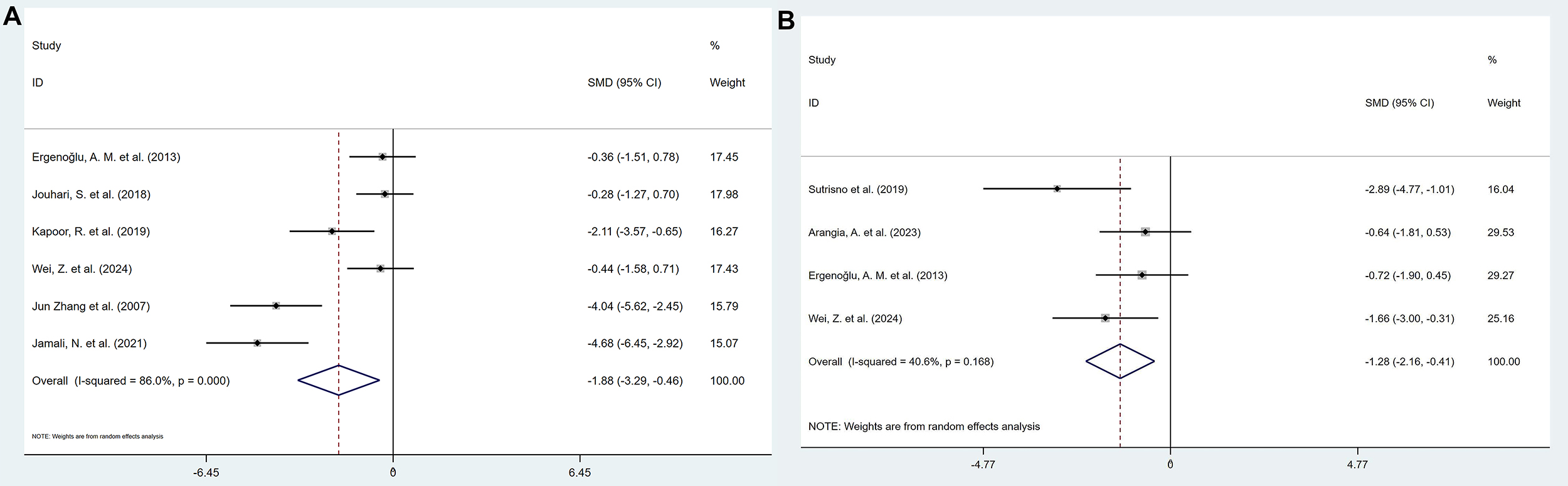 Fig. 7.
Fig. 7.
Forest plot of the effect of polyphenols on tumor necrosis
factor-
3.3.5.2 IL-8 Levels in Serum and Peritoneal Fluid
2 studies reported the effects of polyphenolic compounds on IL-8 levels in serum and peritoneal fluid. Compared with the model group, polyphenolic compounds reduced the levels of IL-8 in both serum (SMD = –1.61, 95% CI: –2.42, –0.80, heterogeneity: I2 = 0.0%, p = 0.927) and peritoneal fluid (SMD = –1.70, 95% CI: –2.72, –0.69, heterogeneity: I2 = 0.0%, p = 0.423), with statistically significant differences (Fig. 8).
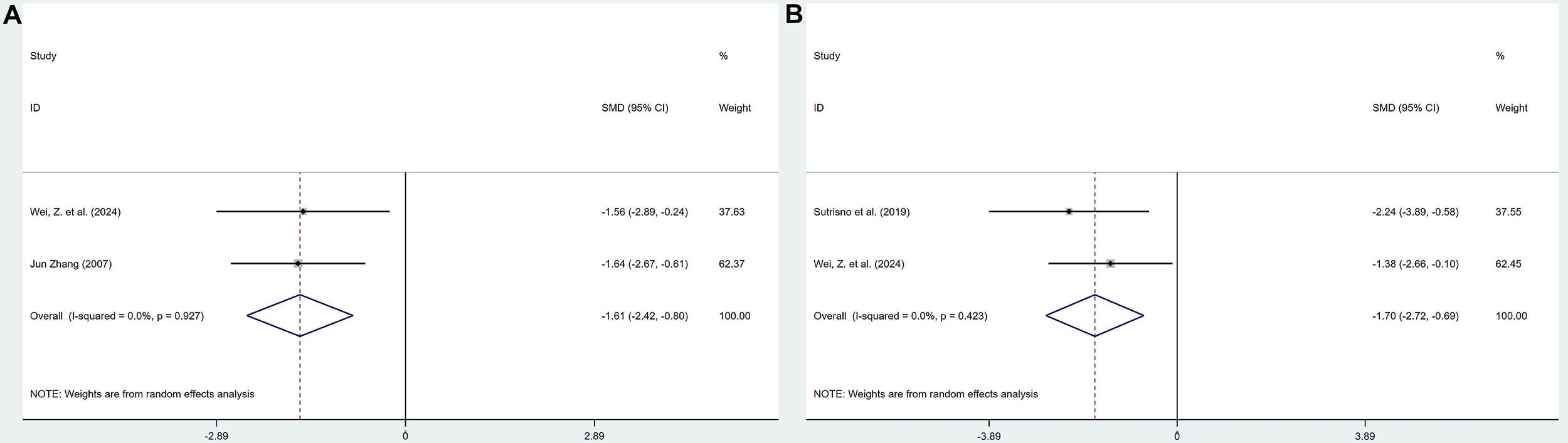 Fig. 8.
Fig. 8.
Forest plot for polyphenols on interleukin-8 (IL-8) levels in serum (A) and peritoneal fluid (B).
3.3.5.3 IL-1
10 studies reported the effects of polyphenolic compounds on IL-1
 Fig. 9.
Fig. 9.
Forest plot of the effect of polyphenols on interleukin-1
3.3.5.4 IL-6 Levels in Serum and Peritoneal Fluid
4 studies reported the effects of polyphenolic compounds on IL-6 levels in serum and peritoneal fluid. The results showed that, compared with the model group, polyphenolic compounds reduced the levels of IL-6 in both serum (SMD = –4.61, 95% CI: –7.69, –1.53, heterogeneity: I2 = 92.3%, p = 0.000) and peritoneal fluid (SMD = –3.37, 95% CI: –5.22, –1.51, heterogeneity: I2 = 74.0%, p = 0.009), with statistically significant differences (Fig. 10).
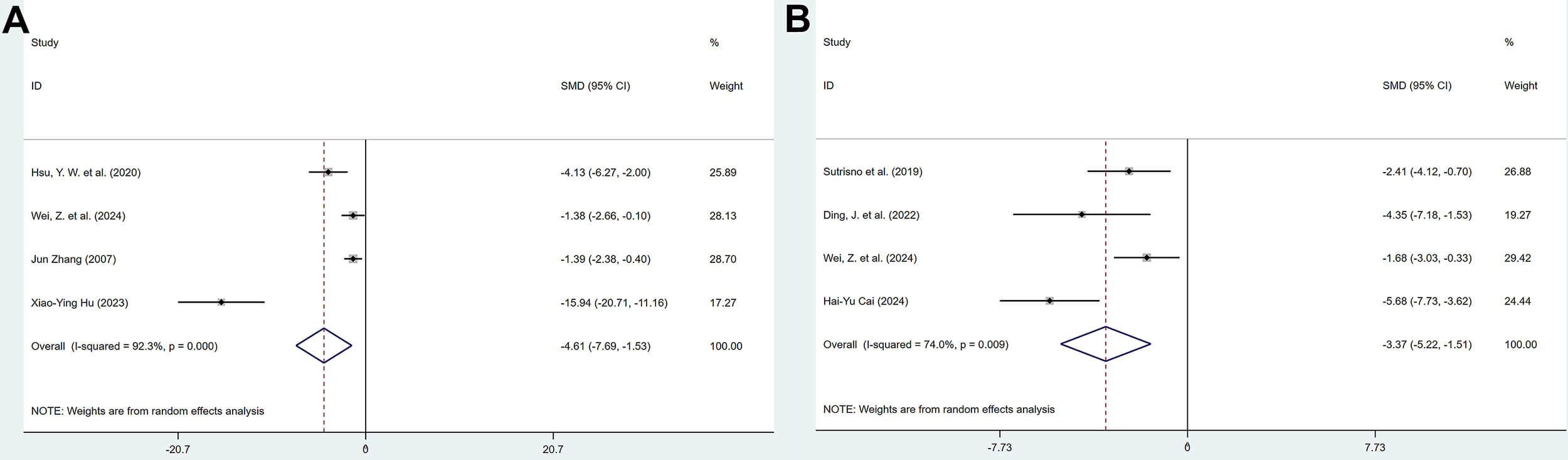 Fig. 10.
Fig. 10.
Forest plot of the effect of polyphenols on interleukin-6 (IL-6) levels in serum (A) and peritoneal fluid (B).
3.3.5.5 MCP-1 Levels in Serum and Peritoneal Fluid
3 studies reported the effects of polyphenolic compounds on MCP-1 levels in serum and peritoneal fluid. The results showed that, compared with the model group, polyphenolic compounds reduced MCP-1 levels in both serum (SMD = –1.24, 95% CI: –2.23, –0.26, heterogeneity: I2 = 45.5%, p = 0.160) and peritoneal fluid (SMD = –1.19, 95% CI: –1.99, –0.39, heterogeneity: I2 = 19.8%, p = 0.287), with statistically significant differences (Fig. 11).
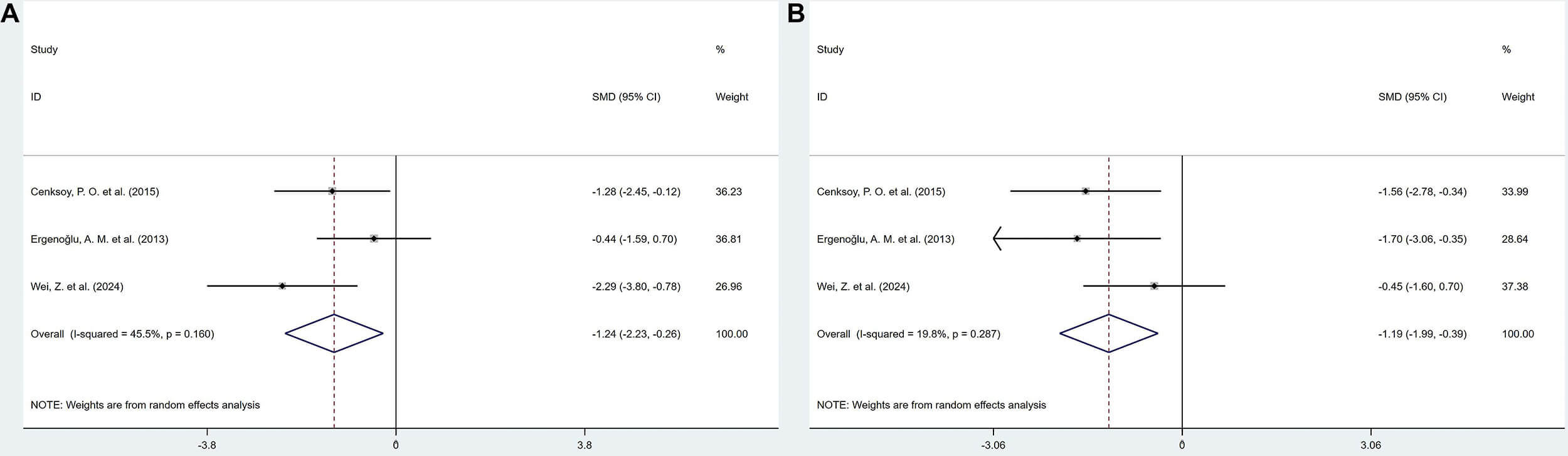 Fig. 11.
Fig. 11.
Forest plot of the effect of polyphenols on monocyte chemoattractant protein-1 (MCP-1) levels in serum (A) and peritoneal fluid (B).
3.3.6.1 Microvascular Density
6 studies, comprising 8 reports, investigated the effects of polyphenolic compounds on MVD in lesions. The results demonstrated that, compared with the model group, polyphenolic compounds significantly reduced MVD in lesions, with a statistically significant difference (SMD = –2.38, 95% CI: –3.39, –1.37; heterogeneity: I2 = 74.7%, p = 0.000) (Fig. 12).
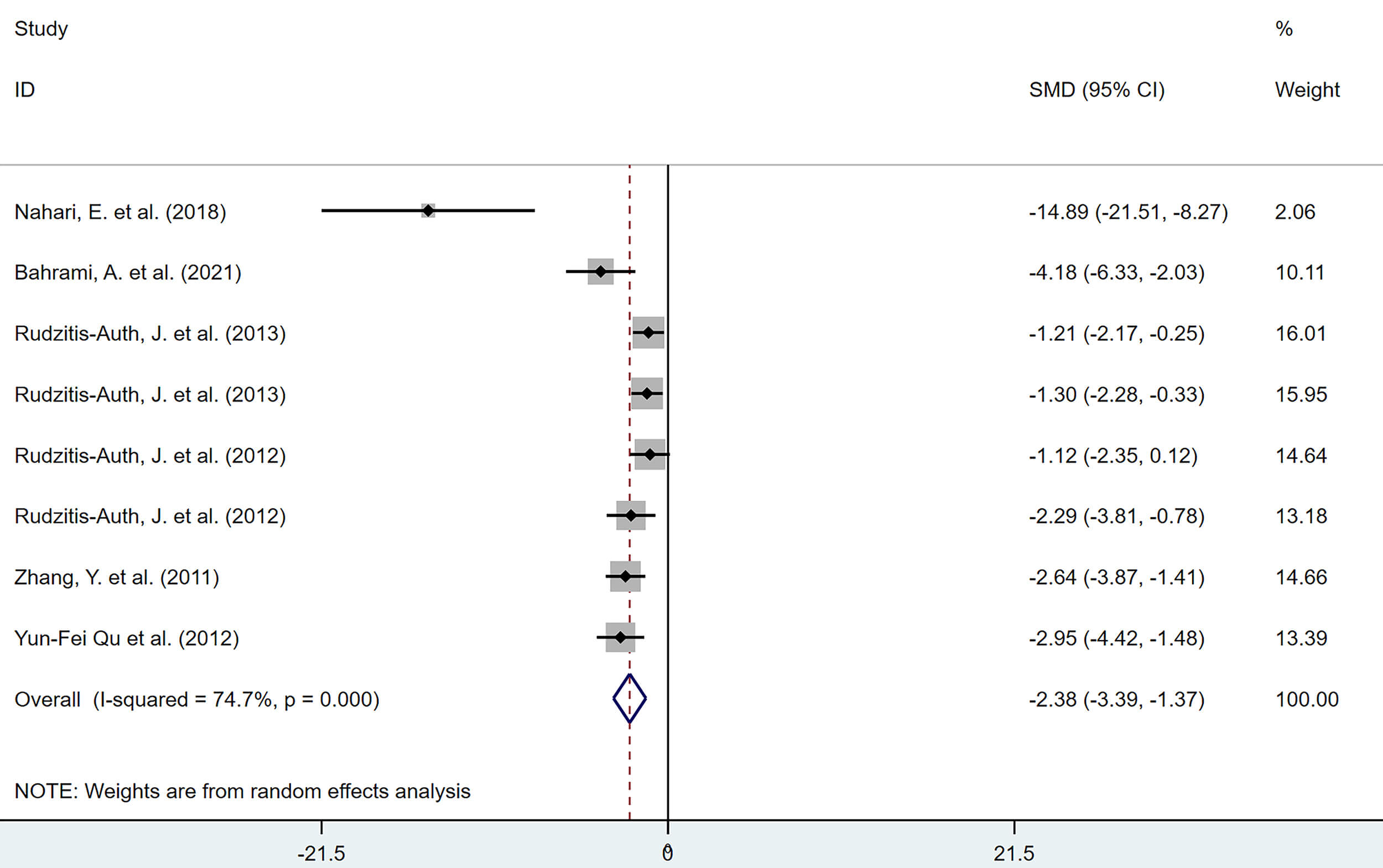 Fig. 12.
Fig. 12.
Forest plot of the effect of polyphenols on microvessel density (MVD) in lesions.
3.3.6.2 Vascular Endothelial Growth Factor (VEGF)
Among the included studies, VEGF was assessed in several forms, including serum VEGF levels (4 studies), peritoneal fluid VEGF levels (5 studies), VEGF protein expression in lesions (3 studies), and VEGF mRNA levels (4 studies). The results showed that polyphenolic compounds significantly reduced VEGF levels in serum (SMD = –1.68, 95% CI: –2.92, –0.45, heterogeneity: I2 = 68.8%, p = 0.022), peritoneal fluid (SMD = –3.12, 95% CI: –4.81, –1.43, heterogeneity: I2 = 82.5%, p = 0.000), VEGF protein expression in lesions (SMD = –1.98, 95% CI: –2.97, –0.99, heterogeneity: I2 = 22.5%, p = 0.275), and VEGF mRNA levels (SMD = –3.03, 95% CI: –5.02, –1.05, heterogeneity: I2 = 82.3%, p = 0.001), with all differences being statistically significant (Fig. 13).
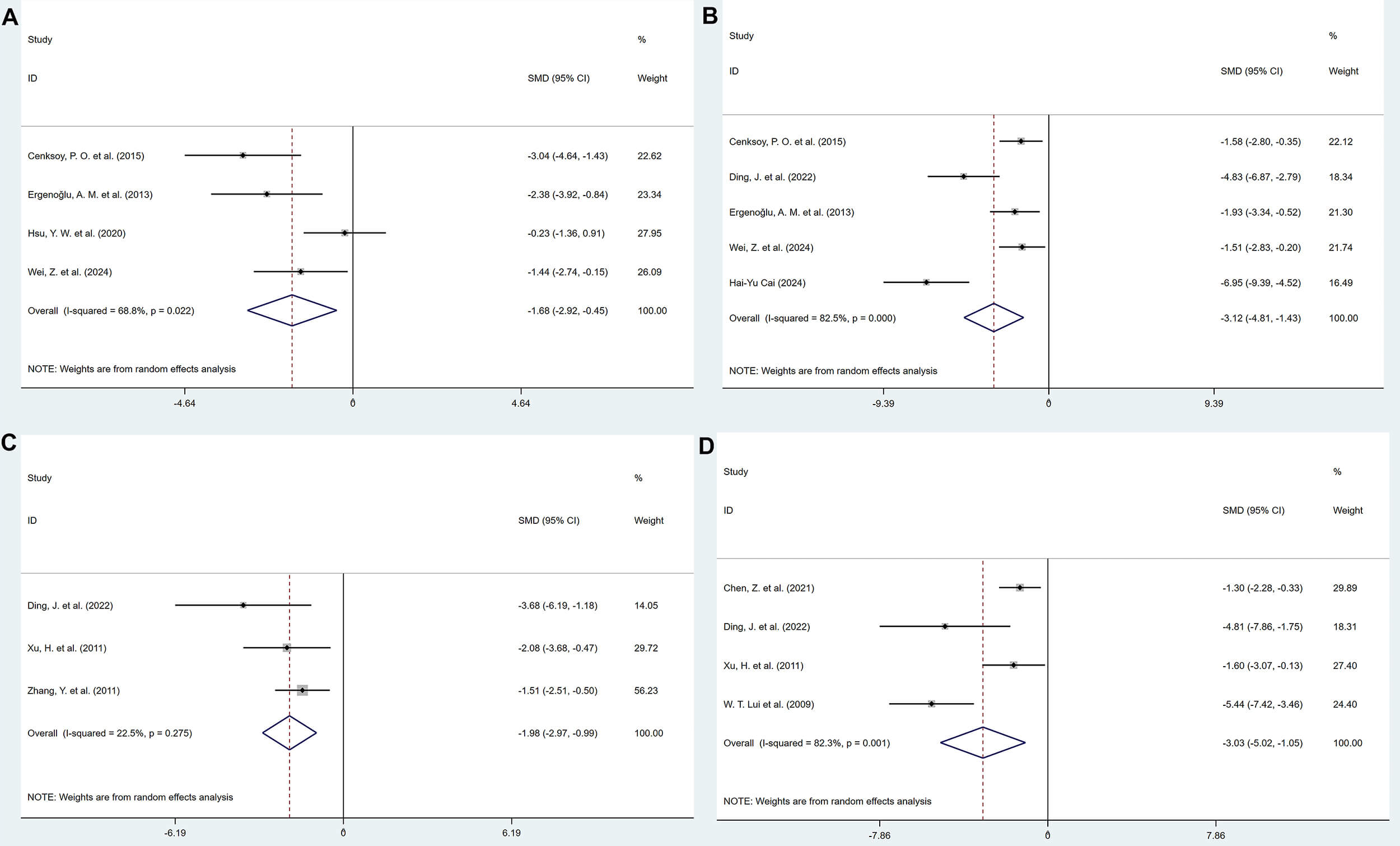 Fig. 13.
Fig. 13.
Forest plots of the effect of polyphenols on vascular endothelial growth factor (VEGF) levels. (A) Serum. (B) Peritoneal fluid. (C) Protein Expression in lesion tissue. (D) mRNA expression in lesion tissue.
3.3.7.1 Proliferating Cell Nuclear Antigen (PCNA)
3 articles, comprising 5 studies, reported the inhibitory effects of polyphenolic compounds on the proliferation of lesion cells. The results demonstrated that, compared with the model group, polyphenolic compounds significantly reduced the percentage of PCNA-positive cells in the lesions (SMD = –2.34, 95% CI: –3.35, –1.33; heterogeneity: I2 = 57.9%, p = 0.050) (Fig. 14).
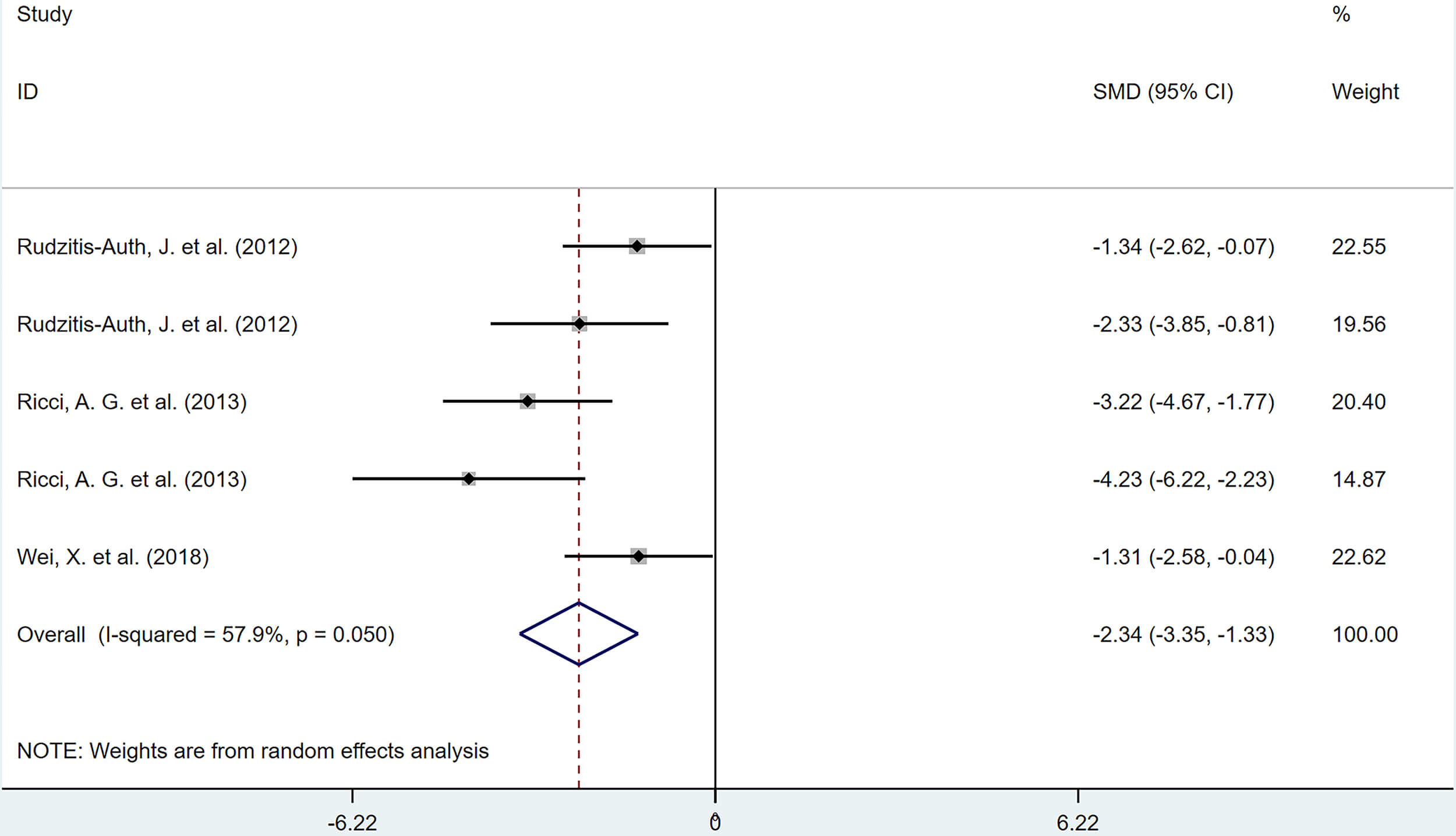 Fig. 14.
Fig. 14.
Forest plot of the effects of polyphenols on the proliferation of lesional cells (percentage of proliferating cell nuclear antigen [PCNA]-positive cells).
3.3.7.2 B-cell Lymphoma 2 (BCL-2)
3 studies reported the promoting effect of polyphenolic compounds on apoptosis of lesion cells. The results showed that, compared with the model group, polyphenolic compounds reduced the mRNA expression of BCL-2 in lesions (SMD = –1.95, 95% CI: –2.81, –1.08, heterogeneity: I2 = 0.0%, p = 0.579). The difference was statistically significant (Fig. 15).
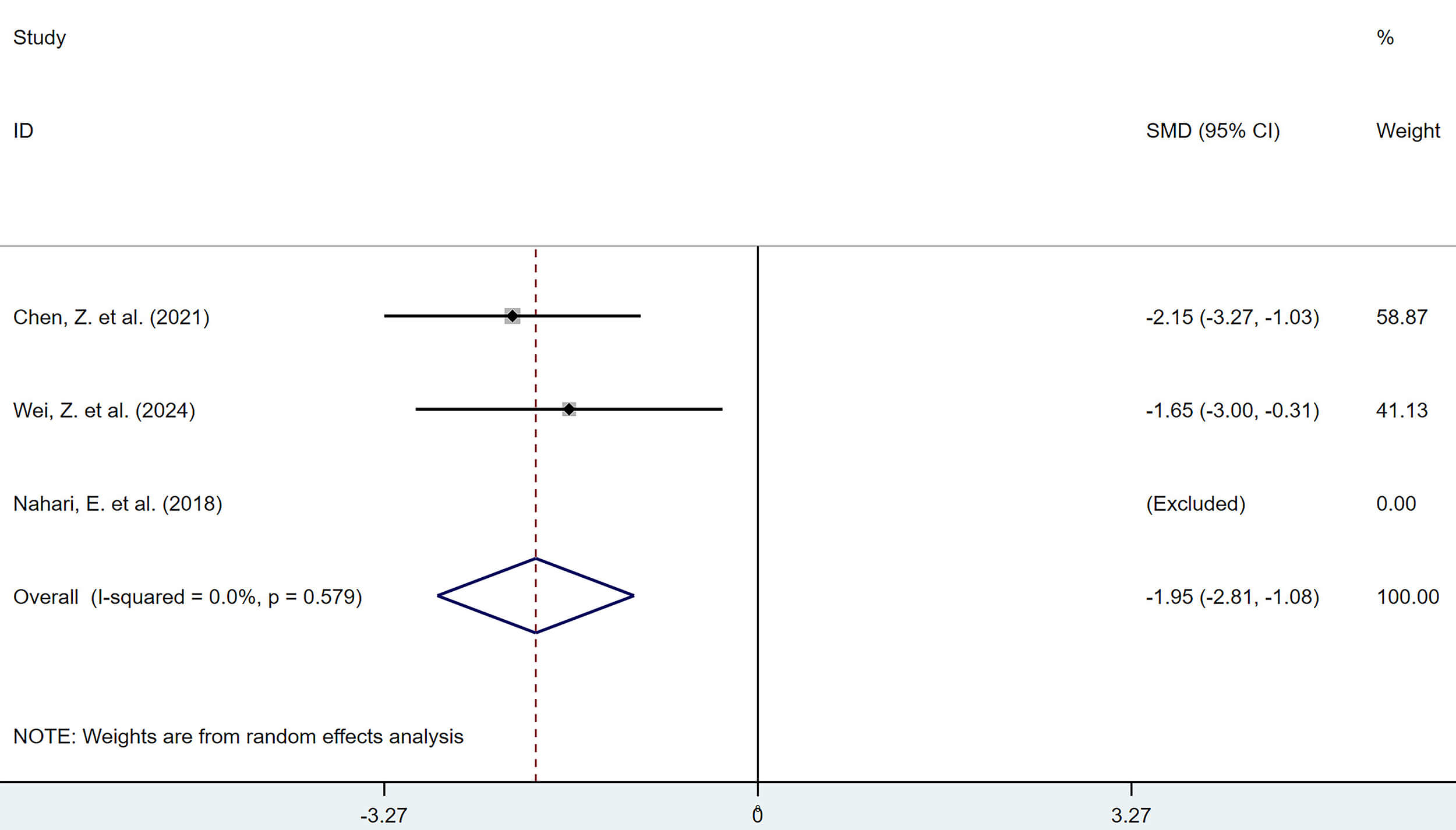 Fig. 15.
Fig. 15.
Forest plot of the effect of polyphenols on apoptosis (B-cell lymphoma 2 [BCL-2] mRNA expression).
3.3.8.1 Hypoxia-inducible Factor-1
The combined results from 2 studies indicated that, compared with the model
group, polyphenolic compounds significantly reduced the percentage of
HIF-1
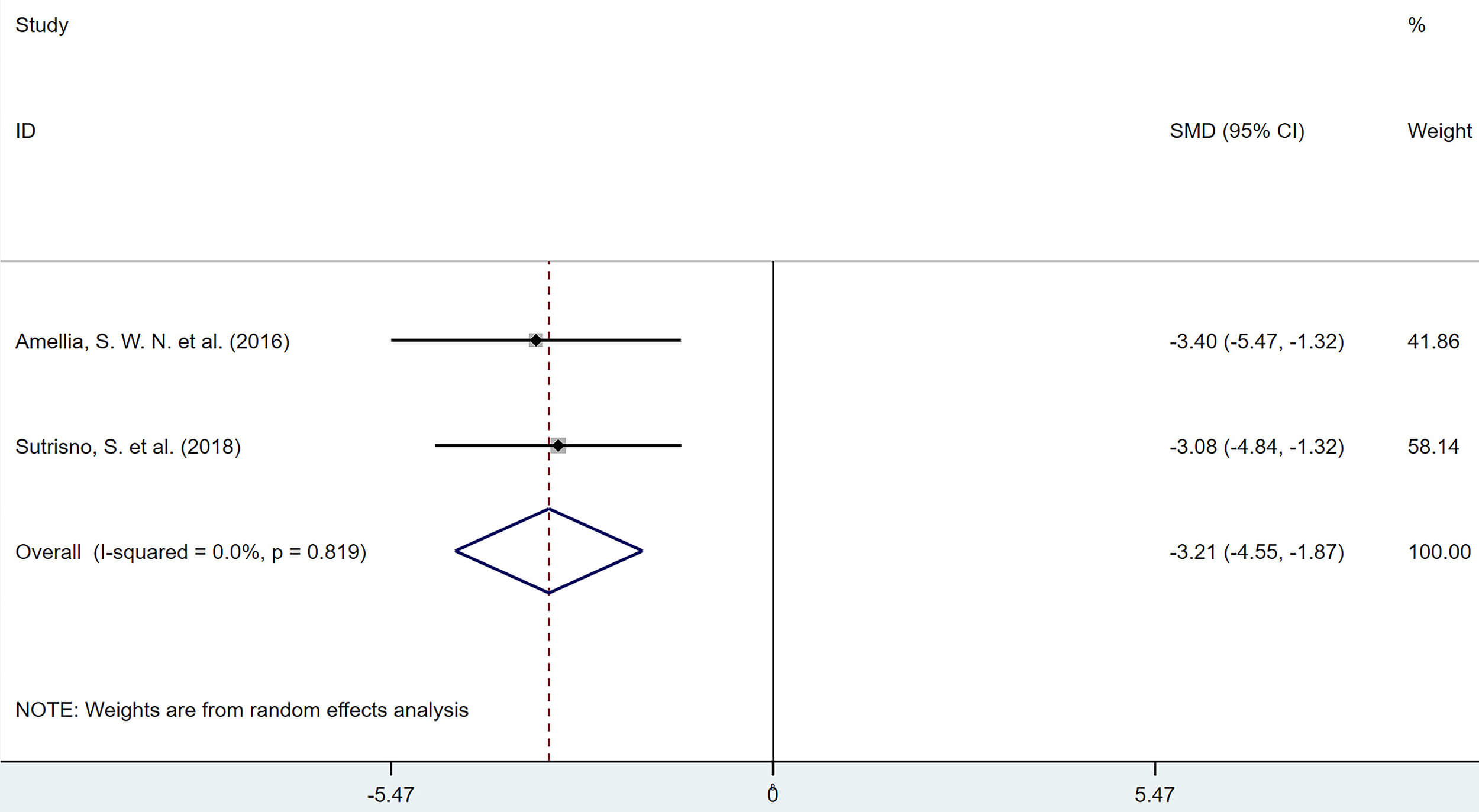 Fig. 16.
Fig. 16.
Forest plot of the effect of polyphenols on the percentage of
hypoxia-inducible factor-1
3.3.8.2 Prostaglandin E2 (PGE2)
The combined results of 3 studies showed that, compared with the model group, polyphenolic compounds reduced serum PGE2 levels (SMD = –3.44, 95% CI: –5.47, –1.41, heterogeneity: I2 = 77.2%, p = 0.012). The difference was statistically significant (Fig. 17).
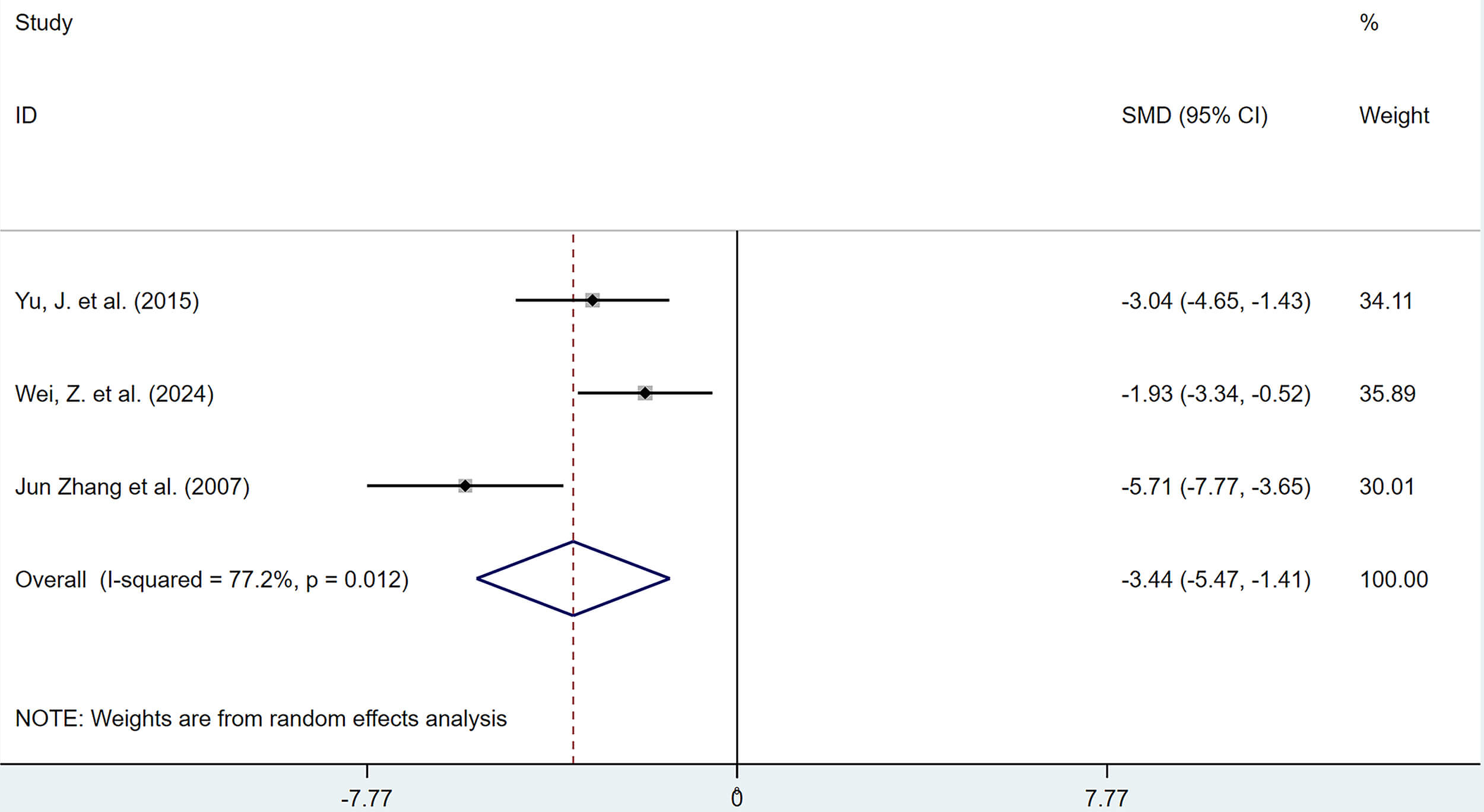 Fig. 17.
Fig. 17.
Forest plot of the effects of polyphenols on serum prostaglandin E2 (PGE2) levels.
To investigate the sources of substantial heterogeneity observed in some outcome
measures, the following analyses were conducted: Meta-regression and subgroup
analyses were performed for lesion volume, which included a larger number of
studies; for adhesion score and serum E2 level, which included fewer studies (n
Meta-regression analysis for lesion volume revealed that only publication
language significantly influenced the effect size (SMD) (
 Fig. 18.
Fig. 18.
Meta-regression analysis of lesion volume.
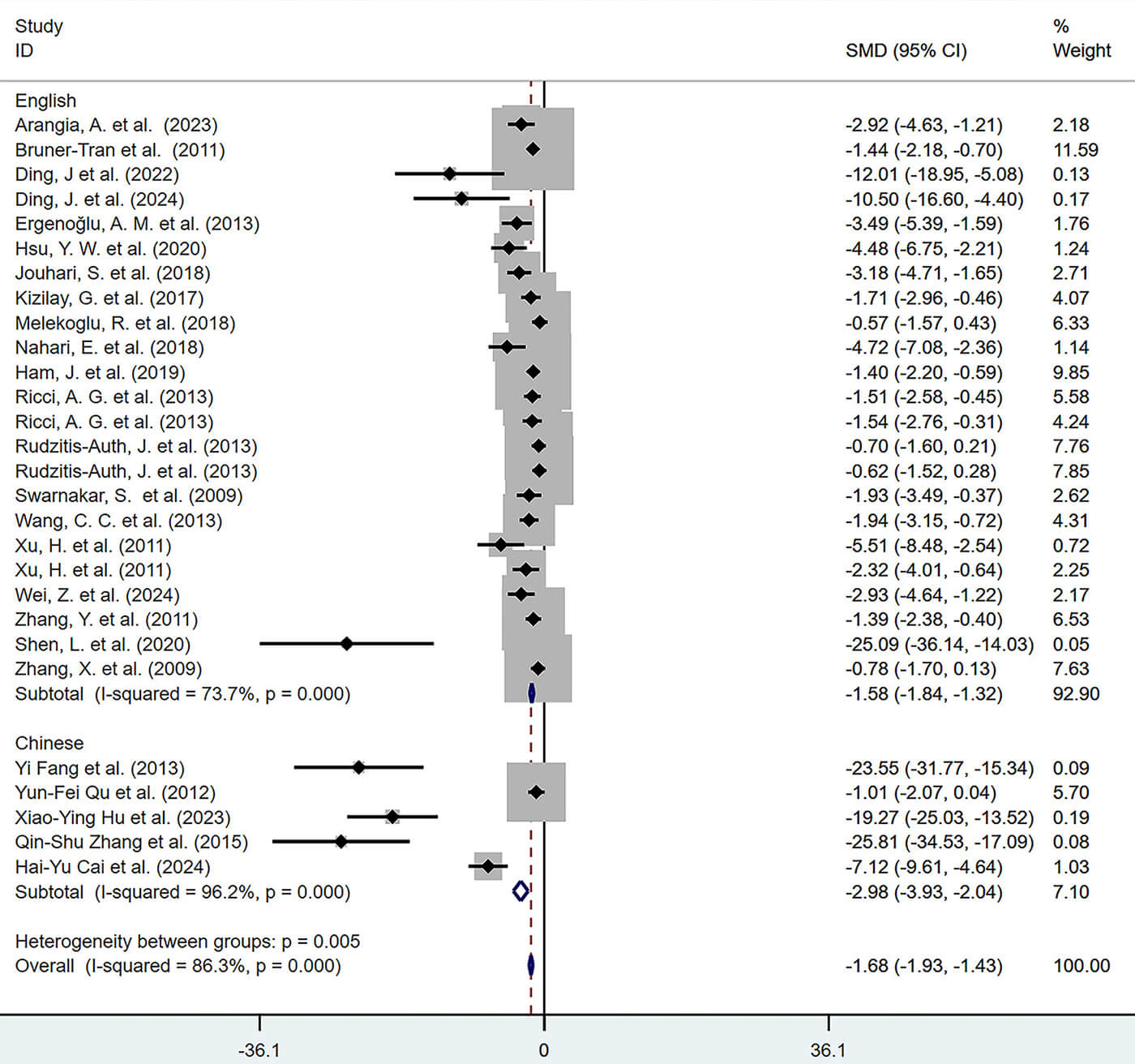 Fig. 19.
Fig. 19.
Subgroup analysis of lesion volume by publication language (English and Chinese).
For adhesion score, subgroup analyses suggested that polyphenol type and route of administration may be associated with differences in effect sizes (Figs. 20,21), whereas other variables (e.g., dosage, treatment duration, animal species) did not show clear associations.
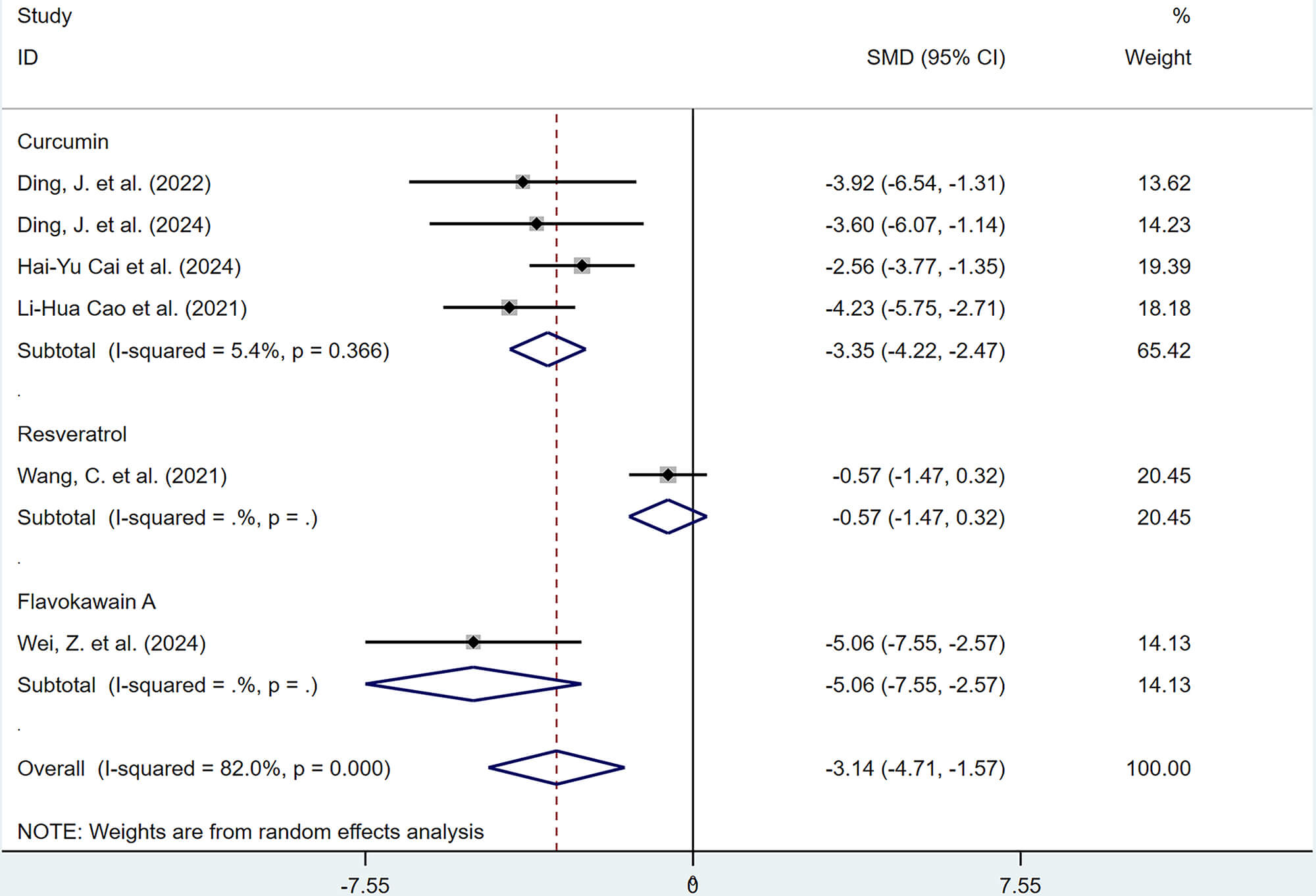 Fig. 20.
Fig. 20.
Subgroup analysis of adhesion scores by type of polyphenol.
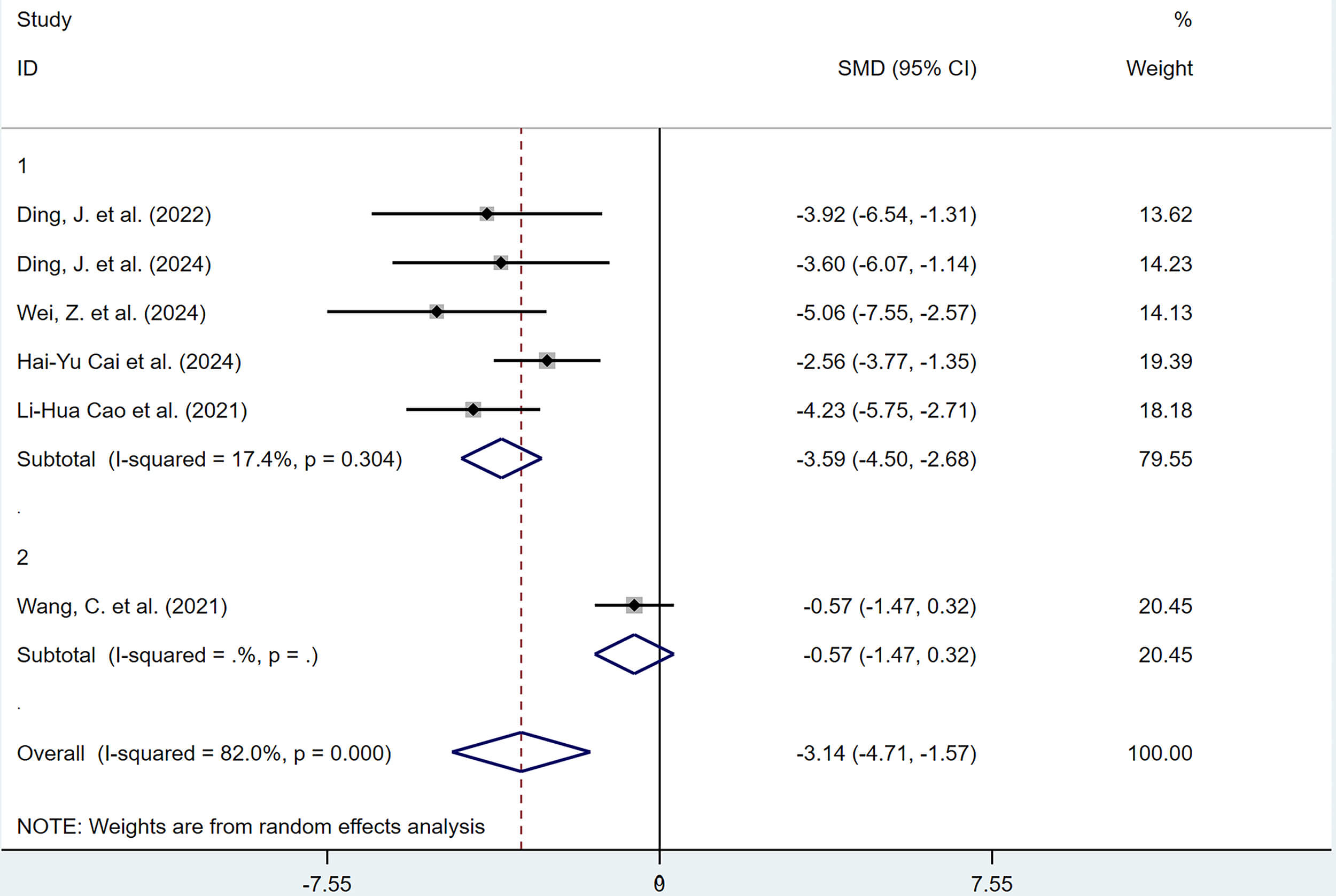 Fig. 21.
Fig. 21.
Subgroup analysis of adhesion scores by route of administration.
For serum E2 levels, subgroup analyses did not identify polyphenol type, dosage, treatment duration, route of administration, or animal species as significant sources of heterogeneity.
To investigate the influence of individual studies on the pooled results,
sensitivity analysis was performed for outcomes with substantial heterogeneity,
including lesion volume, adhesion score, serum estrogen level, plasma
TNF-
For outcome measures with more than 5 studies (lesion volume, microvessel
density, serum E2, adhesion score, peritoneal fluid IL-1
Estrogen receptors (ERs) are members of the nuclear receptor superfamily and are
classified into two subtypes, ER
Our study observed considerable heterogeneity in the effect sizes for estrogen across the included studies, which may be attributed to the following factors. First, a primary source of variation stems from inconsistencies in detection methodologies. The 6 studies employed enzyme-linked immunosorbent assay (ELISA), radioimmunoassay, and chemiluminescence immunoassay. Different methods exhibit poor correlation when detecting low concentrations of E2 [41], which directly affects the comparability of results. Second, polyphenolic compounds exert bidirectional regulation of serum E2 levels, and the direction and magnitude of their effects depend on the disease model and the administered dose [42, 43]. Furthermore, serum E2 levels vary among individual animals of the same strain, and differences in the timing of blood collection, such as sampling at a specific stage of the estrous cycle or at a fixed time point post-intervention, can also lead to discrepancies in the measured results.
Due to insufficient data, we did not pool results on the effects of polyphenolic compounds on estrogen and progesterone receptors. However, existing studies suggest that polyphenols have the potential to regulate ERs, and additional data is needed for comprehensive analysis in the future.
A defined population of immune cells resides in normal endometrial tissue. These
cells are located in the epithelial compartment of the functional layer and in
lymphoid aggregates of the basal layer, supporting maternal-fetal immune
tolerance, clearing apoptotic endometrial cells, promoting endometrial
regeneration, promoting angiogenesis, and wound healing [44]. EMs is considered a
chronic inflammatory disease. During the formation of ectopic lesions, a large
number of immune cells are recruited to the lesion sites and secrete various
proinflammatory cytokines, forming a local inflammatory microenvironment that
further exacerbates EMs progression [45]. For example, early studies have shown
that the number of macrophages significantly increases in the peritoneal fluid
and ectopic lesions of patients with EMs [46], and these cells secrete various
inflammatory factors, such as IL-1
Early epidemiological studies have shown that consumption of foods rich in
polyphenols reduces the risk of chronic inflammatory diseases in the population
[60], but the anti-inflammatory mechanisms of dietary polyphenols remain unclear.
As research has progressed, evidence indicates that the anti-inflammatory
mechanism of polyphenolic compounds may involve regulation of immune cell
numbers, inhibition of inflammatory factors, regulation of gene expression, and
activation of relevant signaling pathways [61, 62, 63, 64]. Many in vitro and
in vivo studies have shown that polyphenolic compounds exhibit excellent
anti-inflammatory and immunomodulatory effects in the treatment of EMs. For
example, in vitro experiments by Kolahdouz-Mohammadi R et al.
[65] showed that resveratrol significantly reduces the gene and protein
expression of MCP-1, IL-6, and IL-8 in both eutopic and ectopic endometrial
stromal cells from EMs. In the study by Arangia A et al. [66], the
natural polyphenol fisetin inhibits the recruitment and activation of mast cells
in EMs lesions, suppresses activation of the inflammation-related
NLRP3/NF-
Our results show that polyphenolic compounds significantly reduce the levels of
TNF-
Apoptosis is a form of programmed cell death regulated by genes, aimed at clearing excess or abnormal cells and playing an important role in maintaining homeostasis. In the late secretory phase of the normal endometrium, the expression of apoptosis-related proteins increases [6], whereas in women with EMs, decreased expression of proapoptotic factors and increased expression of antiapoptotic factors are observed during menstruation EMs [71], which promotes the survival and proliferation of cells in ectopic lesions. Previous studies have reported that polyphenolic compounds exert cytotoxic effects by inhibiting the cell cycle, promoting autophagy, and directly inducing tumor cell apoptosis [72, 73, 74].
Our research indicates that polyphenolic compounds significantly inhibit the
proliferation of ectopic lesion cells in EMs rats, promote cell apoptosis, and
reduce the volume of ectopic lesions. The underlying mechanisms may involve the
following aspects: (1) Inhibition of EMs cell proliferation by suppression of the
transcriptional activation of PI3K/Akt and NF-
OS refers to a state in which the balance between oxidative and antioxidant systems is disrupted due to the excessive production of ROS, leading to cellular stress and damage [81]. It is closely related to chronic inflammatory diseases. In addition to chronic inflammatory stimuli, lipid accumulation and obesity are also risk factors for excessive intracellular ROS production and imbalance of redox homeostasis [82]. As early as 1987, Zeller JM et al. [83] found that ROS levels in the peritoneal fluid of women with EMs was significantly increased, and suggested that these ROS were released by macrophages after chronic stimulation. However, this study did not explain the imbalance between oxidation and antioxidant systems in EMs [84]. Subsequently, increasing evidence has shown that OS markers in the serum, peritoneal fluid, and follicular fluid of patients with EMs are significantly elevated compared with those of normal individuals, suggesting that OS may be involved in the development of EMs. For example, in a study of differences in follicular fluid composition among different populations, Collodel G et al. [85] reported higher levels of propylene glycol (malondialdehyde [MDA]) in the follicular fluid of women with EMs. The study by Nasiri N et al. [86] showed that, compared with the control group, the levels of the OS marker lipid peroxide in the serum and follicular fluid of women with EMs were significantly increased, while the levels of total antioxidant capacity were decreased. Research has shown that an antioxidant diet can effectively reduce OS markers (lipid hydroperoxides [LHP] and malondialdehyde MDA) in the peripheral blood and peritoneal fluid of women with EMs, and enhance the activity of antioxidant enzymes (superoxide dismutase [SOD] and glutathione peroxidase [GPx]) [87]. Polyphenolic compounds are antioxidants that have been widely studied in the treatment of various chronic diseases, including EMs, inflammatory bowel disease, neurodegenerative disorders, and malignant tumors [88, 89, 90].
Due to limited data on OS markers in the included studies, we did not statistically analyze OS indicators such as SOD, MDA, GSH, and CAT. Based on the available evidence, we infer that the antioxidant effects of polyphenolic compounds on EMs may be achieved by inhibiting the synthesis of proteases involved in OS (such as cyclooxygenase [COX] and inducible nitric oxide synthase [iNOS]) [78], reducing peripheral lipid levels [91], and alleviating cell apoptosis and inflammatory response in EMs lesions [92, 93].
Menstrual reflux is a common physiological phenomenon in women during the
menstrual period [94]. When viable endometrial tissue fragments reflux back into
the pelvic cavity through the fallopian tubes, most cells die due to a lack of
oxygen and nutrient supply. However, due to some unclear epigenetic regulation
mechanisms, some endometrial cells exhibit strong survival and invasion abilities
under hypoxic conditions [95]. Hypoxia is widely involved in various biological
processes involved in the development of EMs, with the most common being the
promotion of angiogenesis [96]. Hypoxia induces upregulation of local
HIF-1
Numerous studies have confirmed abnormal expression of HIF-1
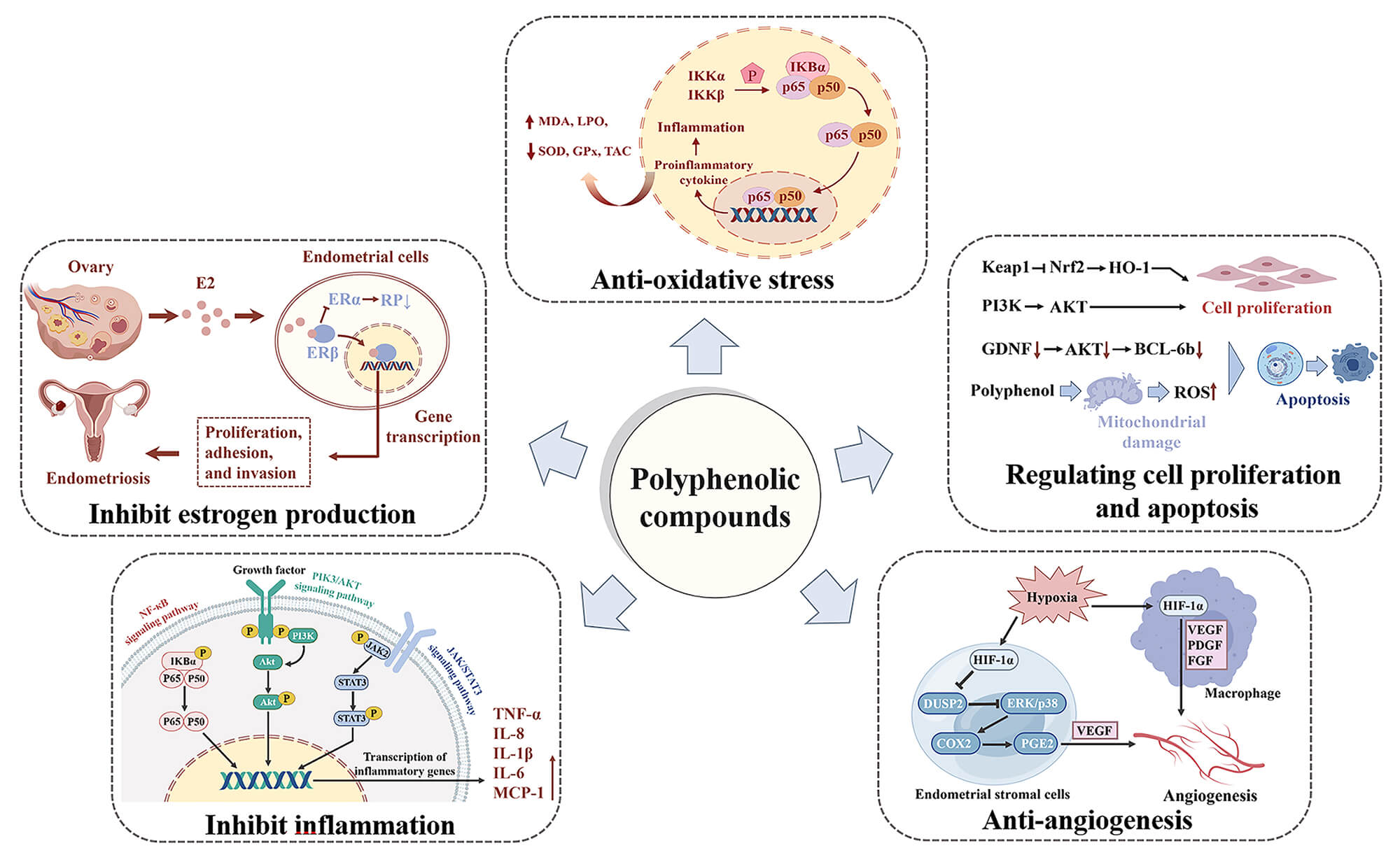 Fig. 22.
Fig. 22.
Mechanisms of polyphenolic compounds in the treatment of
endometriosis (EMs). Polyphenolic compounds exert therapeutic effects on EMs by
inhibiting estrogen production, resisting oxidative stress (OS), regulating cell proliferation and
apoptosis, anti-inflammatory effects, improving hypoxia, and anti-angiogenesis.
Anti-OS: Polyphenols inhibit OS levels by downregulating the transcriptional
activation of the nuclear factor kappaB (NF-
Our research indicates that polyphenolic compounds have the potential to treat EMs through multiple mechanisms of action and may represent a clinically valuable alternative treatment method. Notably, some polyphenolic compounds, such as resveratrol, have been applied in clinical trials of EMs and have shown preliminary efficacy [103, 104]. However, the development of new drugs from natural products is a lengthy and costly process, and is mainly limited by their physicochemical properties and pharmacokinetic profiles, including low bioavailability, poor water solubility, and potential adverse reactions [105]. In recent years, advances in nanotechnology have greatly promoted innovation of drug delivery systems. Currently, new nano drug delivery systems (NDDS) loaded with natural products have been developed, including nanoliposomes, nanocapsules, and polymer micelles. Compared with traditional drug delivery methods, NDDS offer advantages such as enhanced drug activity, good biocompatibility, precise targeting, and improved bioavailability [106]. In clinical applications of polyphenolic compounds, NDDS can significantly improve the bioavailability of polyphenols, reduce their degradation, preserve their antioxidant activity, enhance therapeutic efficacy, and reduce adverse drug reactions. Moreover, nanoparticles can undergo biodegradation without significant toxicity [107], and these advantages may help overcome the limitations of polyphenolic compounds in clinical applications. At present, research on NDDS based on natural products mainly focuses on single compounds, and studies on systems that incorporate multiple natural products into NDDS remain limited. In the future, greater attention should focus on NDDS based on combination therapies, achieving synergistic effects through rational multi-component design, thereby improving therapeutic outcomes [108].
This systematic review has several limitations. First, during the exploration of heterogeneity, we found that publication language was significantly associated with effect sizes. This association may reflect systematic differences in publication preferences, experimental methods, or study quality across regions, thus indicating a need for caution regarding potential publication bias. However, this factor did not explain most of the observed heterogeneity. The high heterogeneity in lesion volume and serum estrogen levels may stem from other underlying biological or methodological factors, as discussed earlier. However, due to the limited number of included studies and the diversity of potential variables, the current analysis was unable to fully elucidate these influences. Second, exclusion of studies in languages other than Chinese and English may have introduced language bias. Third, the absence of randomization, investigator blinding, and outcome assessor blinding in some studies may have introduced bias into the results. Fourth, data for certain outcomes were extracted through tool-based conversion, which may have resulted in minor discrepancies from the original data, potentially affecting the precision of the synthesis. Finally, due to the limited number of studies for some outcome measures, their statistical significance could be assessed only through descriptive analysis. Although subgroup and sensitivity analyses were conducted, the high heterogeneity observed for some outcomes could not be fully explained. Future research should comprehensively consider the interactive effects of various factors, such as polyphenol type, administration route, dosage, treatment duration, and animal models. Therefore, some results of this study should be interpreted with caution.
Our systematic review and meta-analysis indicate that polyphenolic compounds demonstrate therapeutic potential for endometrioma in preclinical animal models. These effects involve multiple pathways, including reduced lesion volume, improved histopathology, decreased levels of key inflammatory factors, and modulation of angiogenesis-related protein expression. The underlying mechanisms may include anti-inflammatory, antioxidant, and anti-angiogenic effects.
However, all the above conclusions derive solely from animal experiments. This study has several important limitations: the included original studies exhibited considerable methodological heterogeneity, and reports of bias control measures such as randomization and blinding were incomplete. Consequently, the current strength of evidence remains primarily at the preclinical stage.
In summary, this study provides a robust synthesis of preclinical evidence and a mechanistic overview of the use of polyphenolic compounds in treating endometrioma, which clarifies the scientific rationale for their translational potential. These findings offer direction in the design of future rigorous clinical studies. However, direct extrapolation of these results to humans and their application in clinical practice remains premature. Future research should prioritize high-quality clinical trials to verify the beneficial effects observed in these animal models.
All data supporting the findings of this systematic review and meta-analysis are derived from previously published studies, which are cited in the reference list. The extracted data used for analysis are available from the corresponding author upon reasonable request.
SSL designed the research study. PXF, SSL and YW contributed to the conceptualization and methodology. LLY and YJH performed the research (data extraction). WJJ, XLZ and LY conducted the formal analysis. YW provided help and advice on supervision, validation, and funding acquisition. SSL wrote the original draft. SSL, LY and YW wrote and revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We gratefully acknowledge the funding support that made this study possible.
This research was funded by two projects, namely (1) Central Guidance for Local Science and Technology Development Fund Proiects, grant number 2024ZYD0014. (2) Molecular Mechanism Study of Clearing Dampness and Removing Blood Stasis Therapy for Endometriosis Based on Single-cell Transcriptomics, grant number WXLH202403019.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG48794.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






















