1 Department of Obstetrics and Gynecology, The First Hospital of Anhui University of Science & Technology, 232001 Huainan, Anhui, China
2 Department of Obstetrics and Gynecology, Maternal and Child Health Hospital Affiliated to Anhui Medical University, 230001 Hefei, Anhui, China
3 Department of Obstetrics and Gynecology, The Fifth Clinical College of Anhui Medical University, 230032 Hefei, Anhui, China
4 School of Computer Science, Huainan Normal University, 232038 Huainan, Anhui, China
Abstract
Women with hyperglycemia in pregnancy (HIP) have an increased risk of delivering large-for-gestational-age (LGA) infants, a condition associated with both short- and long-term adverse outcomes for mothers and offspring. The identification of women with HIP at high risk of LGA using routinely available clinical indicators could help optimize antenatal management.
This retrospective cohort study included 675 women diagnosed with HIP who delivered at Anhui Maternal and Child Health Hospital in Hefei, China, between January 2017 and December 2019. Maternal demographic characteristics and biochemical parameters measured at 25–29+6 weeks of gestation were extracted from medical records. LGA was defined as birthweight above the 90th percentile for gestational age and sex, according to the INTERGROWTH-21st international newborn standards. Candidate predictors were selected based on clinical relevance and correlation analysis. A multivariable logistic regression model with stepwise selection was used to develop a prediction model for LGA, followed by the construction of a nomogram. Internal validation was performed using 1000 bootstrap resamples. Discrimination, calibration, and clinical utility were assessed using the area under the receiver operating characteristic curve (AUC), Brier score, Hosmer-Lemeshow goodness-of-fit test, calibration plots, and decision curve analysis (DCA).
Among the 675 women with HIP, 113 (16.7%) delivered LGA infants. The final multivariable model retained 8 predictors: maternal height, serum albumin, lactate dehydrogenase, triglycerides, total cholesterol, blood urea nitrogen, homocysteine, and fasting plasma glucose. The model showed acceptable discrimination, with an AUC of 0.7333 (95% confidence interval [CI]: 0.6856–0.7810) in the original dataset and an optimism-corrected AUC of 0.7132 after 1000 bootstrap resamples. Calibration was acceptable (Brier score 0.1260; Hosmer-Lemeshow p = 0.0644), and DCA indicated a net clinical benefit of using the model across a wide range of threshold probabilities.
We developed and internally validated a simple mid-pregnancy prediction model to estimate the risk of LGA in infants born to women with HIP. This nomogram, based on routinely available clinical and biochemical indicators, may help clinicians identify high-risk women in mid-pregnancy, thus facilitating individualized antenatal management. However, external validation in independent populations is required before broader clinical implementation.
Keywords
- hyperglycemia in pregnancy
- large-for-gestational-age infants
- risk factor
- prediction model
- nomogram
Hyperglycemia in pregnancy (HIP), also known as pregnancy with diabetes, includes pregestational diabetes (PGDM) and gestational diabetes mellitus (GDM) [1]. According to the International Diabetes Federation (IDF) Global Diabetes Map 2021, 21.1 million (16.7%) live births worldwide are affected by HIP [2]. In China, approximately 17.5% of pregnant women will experience hyperglycemia. Several specific risks are associated with uncontrolled diabetes during pregnancy, including an increased likelihood of having large for gestational age (LGA) infants [3]. LGA infants have a higher risk of adverse perinatal outcomes, such as birth asphyxia, shoulder dystocia, and neonatal morbidity. LGA infants also face increased risks of infant death, future obesity, hypertension, and type 2 diabetes [4]. Therefore, identification of the risk of LGA infants is important, as this may provide opportunities for early intervention. Several studies have focused on predicting the occurrence of LGA before delivery. Late pregnancy ultrasound is currently an important tool for estimating LGA [5, 6]. However, prenatal ultrasound prediction allows only a short period of time for intervention before delivery. Moreover, its clinical value is limited due to racial disparities in different countries. Therefore, the aim of this study was to identify routine biochemical indicators during the mid-pregnancy period that are associated with LGA in women with HIP. These indicators were then used to develop and internally validate a nomogram to predict LGA risk.
This retrospective cohort study was conducted at Anhui Maternal and Child Health Hospital, a tertiary maternity hospital in Anhui Province, China. Medical records of pregnant women with HIP who delivered at the hospital between January 2017 and December 2019 were reviewed.
HIP in this study referred to GDM diagnosed during the current pregnancy. Women with known pregestational type 1 or type 2 diabetes were managed in a different clinical pathway and were not included in this cohort. GDM was diagnosed between 24 and 28 weeks of gestation based on a one-step, 75-g oral glucose tolerance test (OGTT) in accordance with the American Diabetes Association Standards of Medical Care in Diabetes, 2022 [1, 3].
For the present analysis, blood biochemistry indices measured at 25–29+6
weeks of gestation were extracted from the hospital electronic database. A total
of 675 women with HIP who delivered singleton live births at
The inclusion criteria were: (1) singleton live birth; and (2) gestational age
at delivery
Women with HIP were managed at our institution according to national and
international guidelines. Initial management consisted of individualized medical
nutrition therapy and advice on moderate physical activity. Women were instructed
to monitor capillary blood glucose at home. When fasting or postprandial glucose
values repeatedly exceeded the target levels (fasting
General characteristics and biochemical indexes were obtained from medical
records. The variables included maternal age, height, gravidity, parity, and the
following laboratory parameters: total protein (TP), albumin (ALB), globulin
(GLO), albumin-to-globulin ratio (A/G), alanine aminotransferase (ALT), aspartate
aminotransferase (AST), total bilirubin (TBIL), total bile acid (TBA), indirect
bilirubin (IBIL), direct bilirubin (DBIL), alkaline phosphatase (ALP),
All biochemical measurements were performed on fasting venous blood samples in
the same hospital laboratory following standard operating procedures. Continuous
variables were summarized as the median (P25–P75) and compared between groups
using the Wilcoxon rank-sum test. Categorical variables were expressed as n (%)
and compared using the chi-square test or Fisher’s exact test, as appropriate. A
two-sided p-value
Information on pre-pregnancy weight, body mass index (BMI), gestational weight gain, and glycated hemoglobin (HbA1c) was not systematically recorded in the electronic database and therefore could not be included as candidate predictors in this analysis.
Pearson correlation analyses were performed to explore collinearity among
candidate predictors. When two variables were highly correlated (absolute
correlation coefficient
Subsequently, univariable logistic regression was used to examine the
association between each candidate predictor and LGA. Predictors with p
Model discrimination was evaluated by calculating the area under the receiver operating characteristic curve (AUC). Calibration was assessed using the Brier score, the Hosmer-Lemeshow goodness-of-fit test, and calibration plots that compared predicted and observed risks across deciles of predicted probability. For internal validation, we ran bootstrap resampling with 1000 repetitions and reported optimism-corrected performance estimates. Clinical usefulness was examined by decision curve analysis (DCA), which quantifies the net benefit of applying the model across a range of threshold probabilities relative to treating all patients or none. All analyses were conducted in R (version 4.1.3; R Foundation for Statistical Computing, Vienna, Austria). Model development and validation were reported in accordance with the Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD) statement.
The correlation coefficients between A/G and GLO, TP and GLO, AST and ALT, IBIL
and TBIL, HBDH and LDH, and CHOL and LDL were all
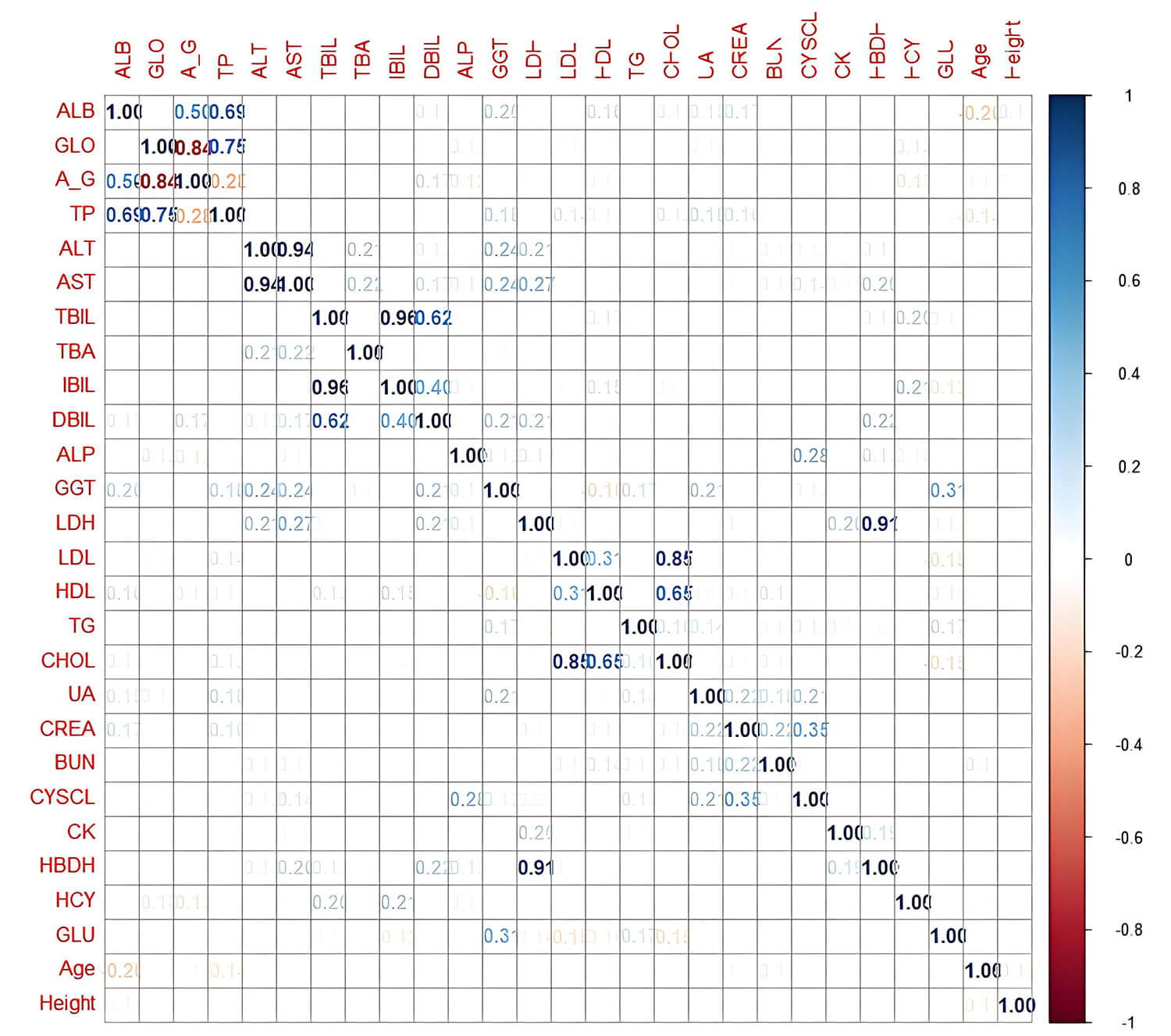 Fig. 1.
Fig. 1.
Pairwise correlation matrix of candidate predictors included in
the model development. Darker colors indicate stronger positive or negative
correlations. ALB, albumin; GLO, globulin; A_G, albumin-to-globulin ratio; TP,
total protein; ALT, alanine aminotransferase; AST, aspartate aminotransferase;
TBIL, total bilirubin; TBA, total bile acid; IBIL, indirect bilirubin; DBIL,
direct bilirubin; ALP, alkaline phosphatase; GGT,
The results of the univariable analysis comparing the two groups are shown in
Table 1. Among the general characteristics, only maternal height was
significantly different between the LGA and non-LGA groups (p
| Indicator | non-LGA group (n = 562) | LGA group (n = 113) | p | |
| ALB, g/L | 39.70 (38.10, 41.50) | 39.00 (37.30, 40.50) | 0.003 | |
| GLO, g/L | 26.50 (24.70, 28.40) | 25.80 (24.80, 27.40) | 0.038 | |
| ALT, IU/L | 13.40 (10.60, 18.70) | 13.10 (9.50, 18.10) | 0.200 | |
| TBA, µmol/L | 2.24 (1.50, 3.26) | 2.38 (1.72, 3.71) | 0.130 | |
| IBIL, µmol/L | 6.14 (5.04, 7.79) | 5.87 (4.66, 7.30) | 0.064 | |
| DBIL, µmol/L | 1.94 (1.62, 2.33) | 1.94 (1.55, 2.33) | 0.700 | |
| ALP, IU/L | 77.25 (63.30, 95.00) | 77.70 (58.60, 93.00) | 0.500 | |
| GGT, IU/L | 14.00 (10.70, 19.60) | 13.40 (10.10, 21.20) | 0.600 | |
| LDH, IU/L | 160.75 (143.30, 181.05) | 168.50 (145.20, 188.90) | 0.130 | |
| HDL, mmol/L | 2.19 (1.90, 2.48) | 2.05 (1.81, 2.29) | ||
| TG, mmol/L | 2.72 (2.21, 3.42) | 3.09 (2.42, 4.04) | ||
| CHOL, mmol/L | 6.42 (5.62, 7.15) | 6.02 (5.49, 6.59) | ||
| UA, µmol/L | 230.85 (199.62, 270.00) | 230.50 (204.40, 263.30) | 0.800 | |
| CREA, µmol/L | 39.70 (36.30, 43.77) | 38.30 (35.40, 42.50) | 0.130 | |
| BUN, mmol/L | 2.88 (2.44, 3.44) | 2.64 (2.34, 3.01) | 0.001 | |
| CysC, mg/L | 5.80 (5.20, 6.40) | 6.00 (5.40, 6.60) | 0.058 | |
| CK, IU/L | 39.70 (30.90, 53.68) | 39.70 (30.70, 58.60) | 0.400 | |
| HCY, µmol/L | 4.00 (2.70, 4.90) | 3.70 (2.60, 4.30) | 0.010 | |
| GLU, mmol/L | 5.01 (4.60, 5.28) | 5.07 (4.69, 5.39) | 0.022 | |
| Age, years | 31.58 (29.42, 35.50) | 32.25 (29.33, 35.58) | 0.500 | |
| Gravidity | 0.600 | |||
| Primigravid | 207 (36.8%) | 41 (36.3%) | ||
| Repregnant | 355 (63.2%) | 72 (63.7%) | ||
| Parity | 0.800 | |||
| Multiparity | 263 (46.8%) | 53 (46.9%) | ||
| Primiparity | 299 (53.2%) | 60 (53.1%) | ||
| Height, cm | 161.00 (158.00, 165.00) | 163.00 (160.00, 165.00) | 0.004 | |
The Pearson correlation test was conducted on 29 independent variables. The
‘rms’ package was employed on the R 4.1.3 platform to fit a regression model via
stepwise regression. The final mathematical equation derived from the
multivariable logistic regression model to predict LGA infants was: logit(p) =
–9.5305 – 0.1564
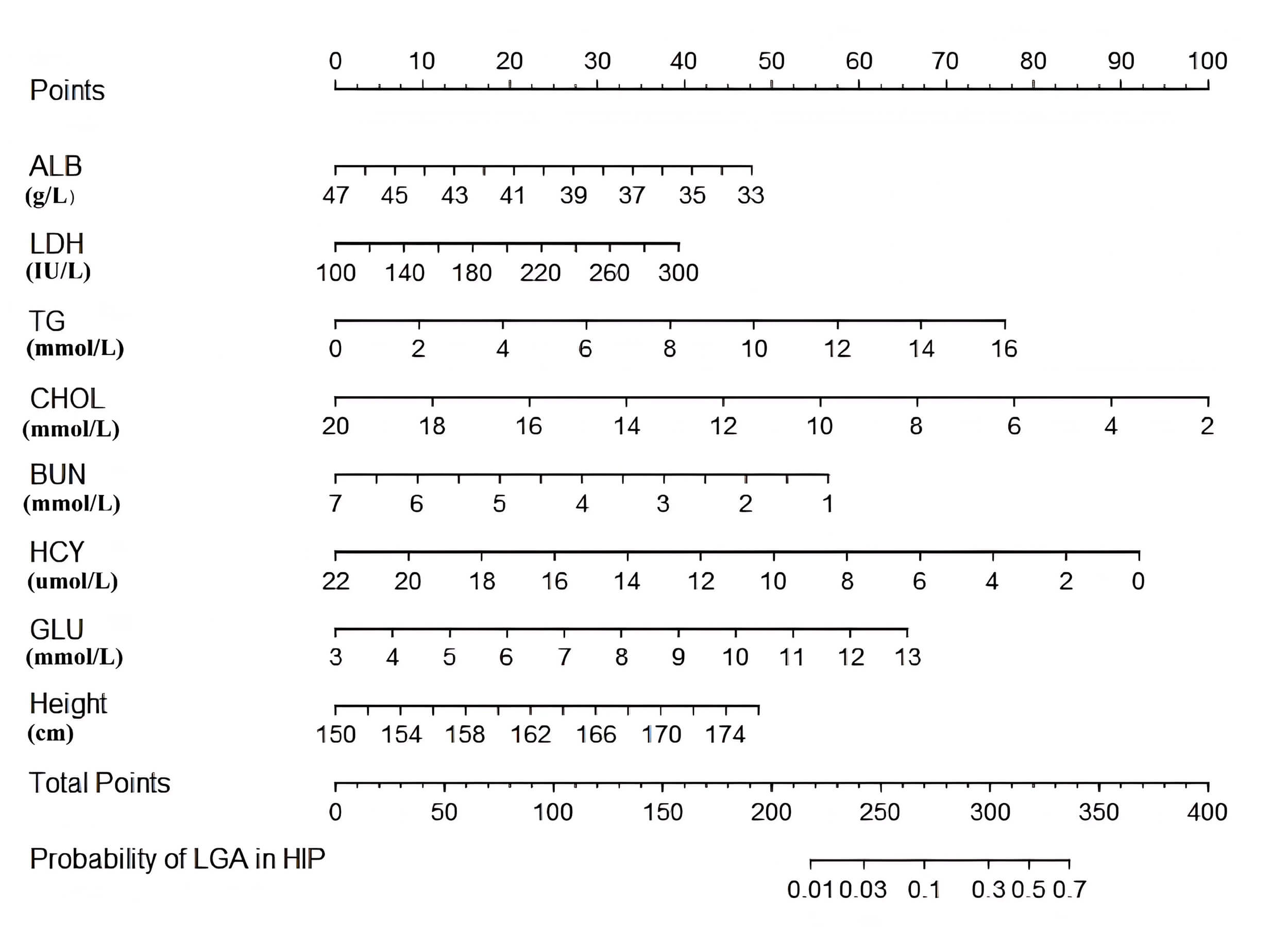 Fig. 2.
Fig. 2.
Nomogram for predicting the risk of delivering a large-for-gestational-age infant in women with hyperglycemia in pregnancy based on maternal height and mid-pregnancy biochemical indicators.
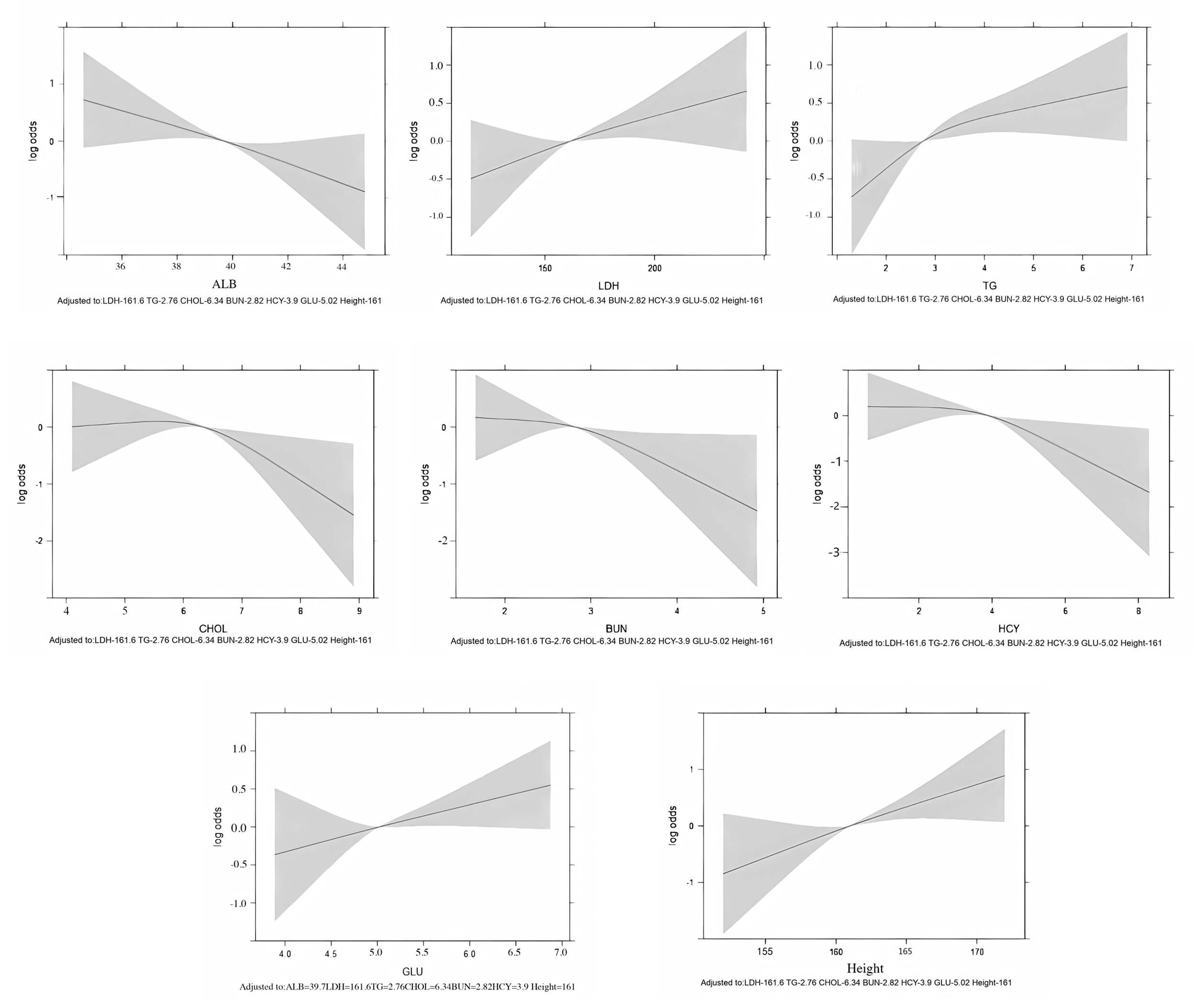 Fig. 3.
Fig. 3.
Restricted cubic spline plots showing the relationship between each continuous predictor and the log odds of large-for-gestational-age birth. No strong evidence of non-linearity was observed.
 Fig. 4.
Fig. 4.
Cook’s distance plots for the final logistic regression model, indicating the influence of individual observations.
| Characteristic | OR | 95% CI | p-value |
| ALB | 0.86 | 0.78, 0.94 | 0.002 |
| LDH | 1.01 | 1.00, 1.02 | 0.010 |
| TG | 1.25 | 1.07, 1.46 | 0.005 |
| CHOL | 0.77 | 0.63, 0.94 | 0.011 |
| BUN | 0.65 | 0.47, 0.89 | 0.009 |
| HCY | 0.83 | 0.72, 0.94 | 0.004 |
| GLU | 1.35 | 1.01, 1.77 | 0.032 |
| Height | 1.09 | 1.04, 1.14 |
Abbreviations: OR, odds ratio; CI, confidence interval.
We assessed discrimination using the area under the receiver operating
characteristic (ROC) curve. The nomogram achieved an AUC of 0.7333 (95% CI:
0.6856–0.7810), indicating moderate discriminatory ability (Fig. 5). Model
calibration was primarily evaluated using the calibration plot, which showed
close agreement between the predicted and observed probabilities of LGA across
deciles of predicted risk (Fig. 6). The Brier score was 0.1260, indicating
acceptable overall accuracy. The Hosmer Lemeshow goodness-of-fit test yielded
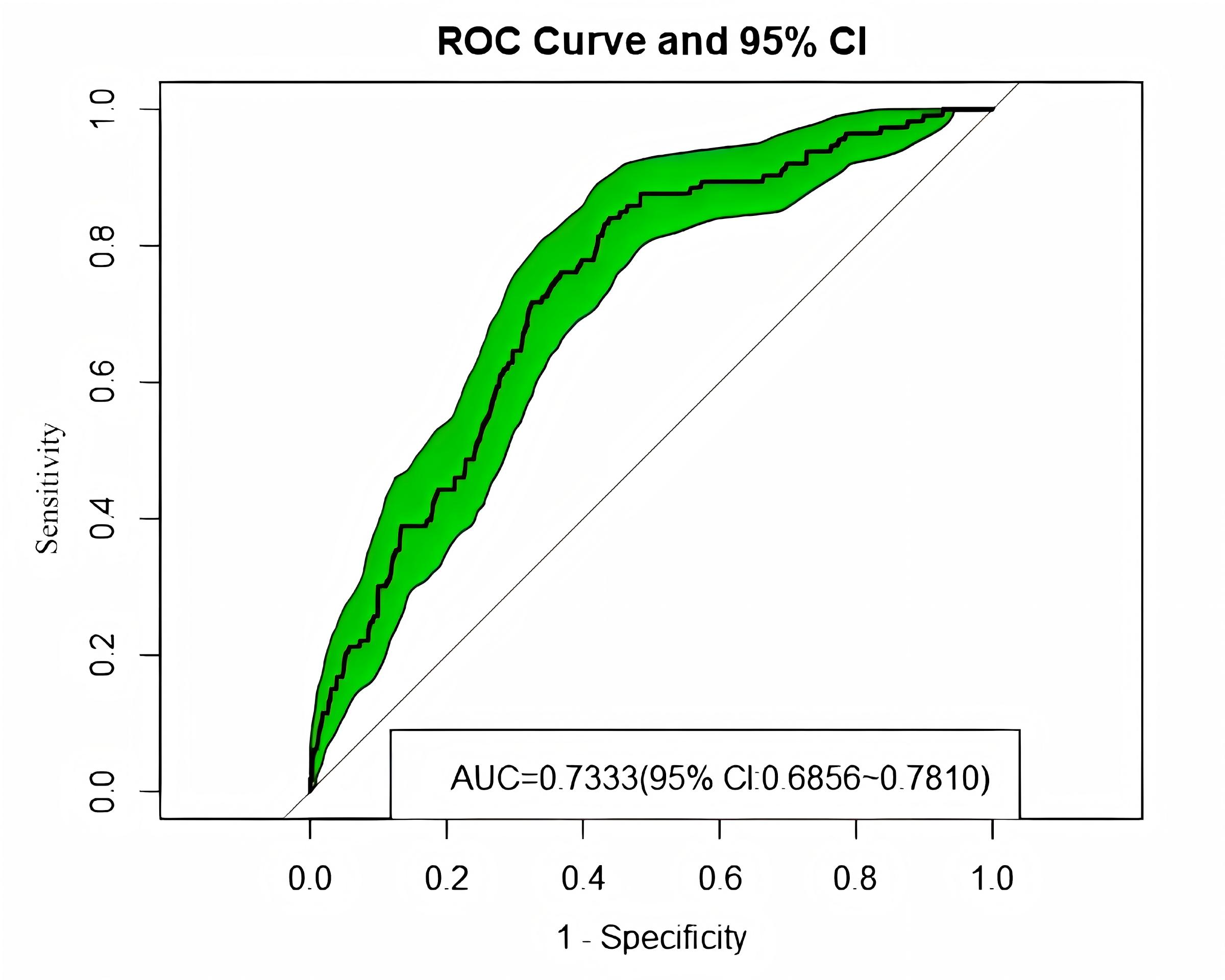 Fig. 5.
Fig. 5.
Receiver operating characteristic (ROC) curve of the prediction model in the original dataset. The shaded area represents the 95% confidence interval for the AUC. AUC, area under the receiver operating characteristic curve.
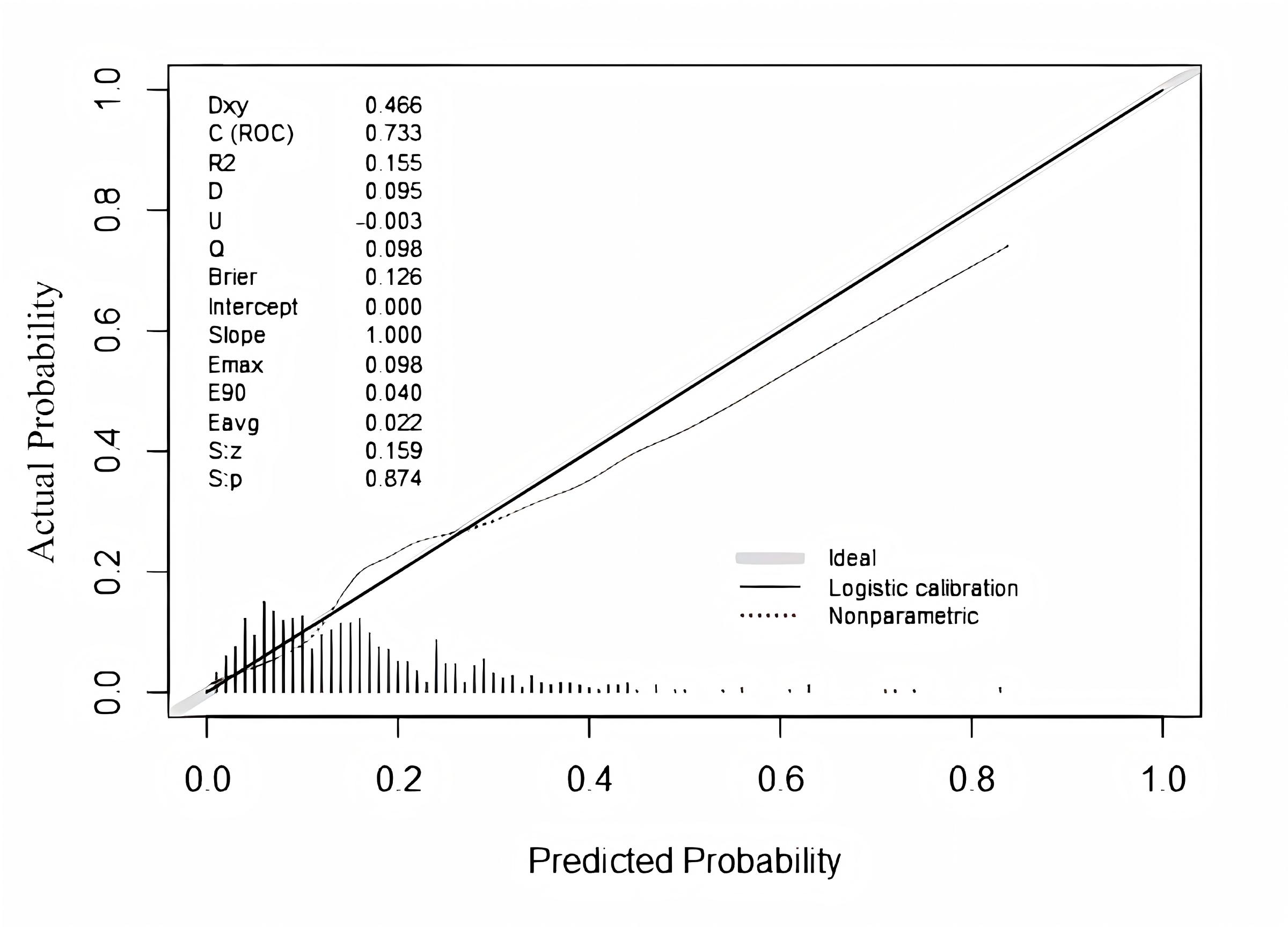 Fig. 6.
Fig. 6.
Calibration plot comparing predicted and observed risks of large-for-gestational-age birth across deciles of predicted probability in the original dataset.
DCA was used to assess the model’s clinical effectiveness, with the results shown in Fig. 7. Application of the nomogram prediction model yields a higher net benefit for women with HIP when the predicted probability ranges from 0.0 to 0.65.
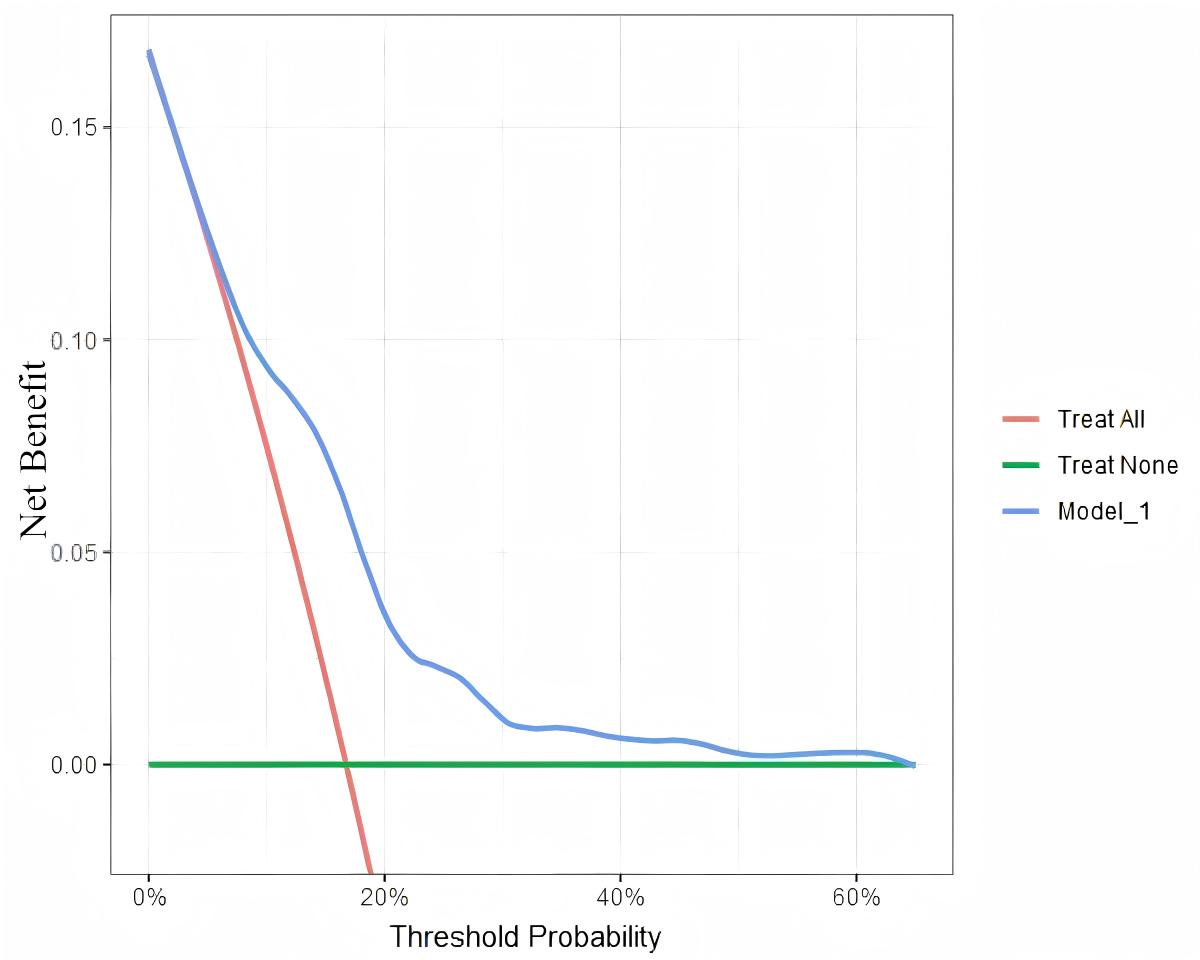 Fig. 7.
Fig. 7.
Decision curve analysis of the prediction model, showing net benefit across a range of threshold probabilities compared with treat-all and treat-none strategies.
Bootstrap resampling was performed 1000 times to validate the model. The ROC curve after resampling (Fig. 8) shows an AUC of 0.7132 (95% CI: 0.7052–0.7329). The Brier score after resampling was calculated as 0.1302. The calibration chart after resampling is depicted in Fig. 9. Hence, the model demonstrates acceptable predictive capability.
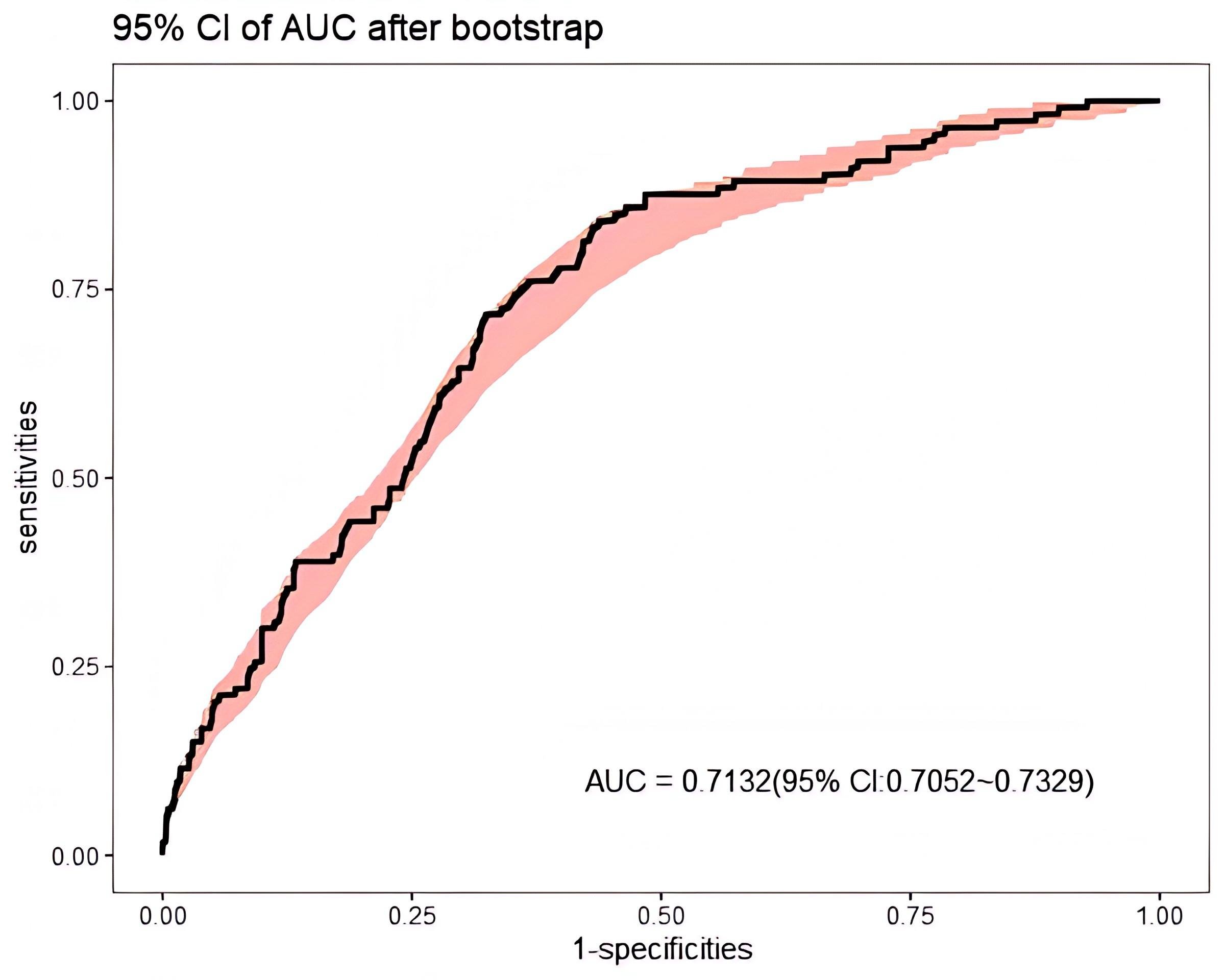 Fig. 8.
Fig. 8.
ROC curve of the model after 1000 bootstrap resamples for internal validation.
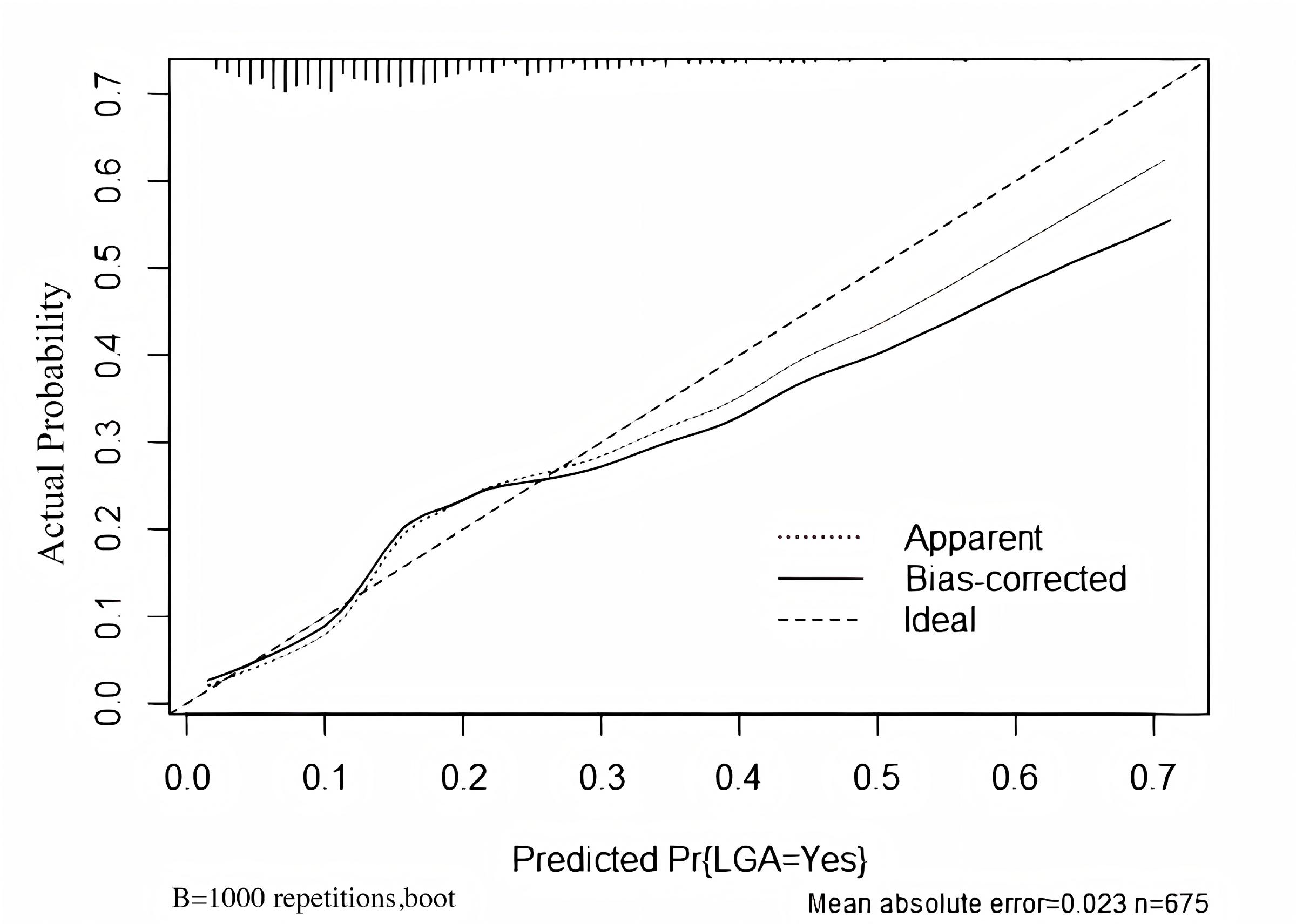 Fig. 9.
Fig. 9.
Calibration plot of the model after 1000 bootstrap resamples for internal validation.
LGA infants are traditionally predicted using late-pregnancy ultrasound, most commonly through estimated fetal weight equations such as the Hadlock formula, or by specific sonographic parameters such as fetal anterior abdominal wall thickness in women with GDM [5, 6]. Because these assessments are usually performed in the third trimester, the window for effective intervention is short, and their performance may vary across ethnic populations. More recently, several studies have explored the predictive value of maternal lipid profiles or other biochemical markers for LGA in general obstetric populations [8, 9]. Although a study by Oben demonstrated that glycosylated hemoglobin collected at 24–28 weeks gestation can predict the risk of adverse perinatal outcomes in HIP pregnant women, it did not specifically predict the occurrence of LGA alone [10]. However, data that is specific to women with HIP is still scarce. In this context, our study extends the existing literature by developing a nomogram based solely on routinely measured mid-pregnancy biochemical indicators and height in women with HIP. By focusing on a high-risk group and using indicators that are widely available in clinical practice, our model offers a pragmatic tool for earlier risk stratification and individualized antenatal management.
Multiple logistic regression (MLR) is widely used in health outcomes research, with applications in medicine, statistics, and machine learning (computer science). MLR involves fitting multiple parameters in a predictive model by assuming a linear or additive relationship between predictors and outcomes [11]. In the current study, we built a predictive model for LGA using general information and biochemical indicators for HIP patients extracted from hospital electronic medical records. The AUC of this model was 0.7333 (95% CI: 0.6856–0.7810), indicating moderate discriminative ability. To validate the model’s accuracy, we performed bootstrap validation 1000 times. The AUC value of our model after bootstrapping was 0.7132 (95% CI: 0.7052–0.7329), demonstrating acceptable predictive performance and reasonable stability. Univariate analysis results revealed statistically significant differences in height, ALB, GLO, HDL, TG, CHOL, BUN, HCY, and GLU between the LGA and non-LGA groups. Furthermore, multivariate logistic regression analysis indicated that height, ALB, LDH, TG, CHOL, BUN, HCY, and GLU are significant predictors of LGA risk in HIP women.
Some authors have proposed that LGA may be linked to factors such as
pre-pregnancy overweight and obesity, excessive weight gain during pregnancy,
abnormal glucose metabolism, abnormal lipid metabolism, and genetic factors
[12, 13, 14]. The results of multivariate regression analysis indicated that maternal
height (OR = 1.09, p
Several studies have emphasized the importance of fasting blood glucose (FBG) as a screening tool for GDM and as a predictor of adverse neonatal outcomes [20]. A meta-analysis revealed a positive association between elevated fasting glucose levels in OGTT trials and adverse pregnancy outcomes, including LGA [21]. Fetal glucose exposure and subsequent insulin secretion can lead to excessive conversion of glucose to fat, resulting in fetal weight gain [22]. This finding is consistent with results from the present study.
Substantial literature exists on the relationship between blood lipid levels and GDM. However, there is still some uncertainty regarding the reported results on abnormal blood lipid status in diabetic pregnant women [23]. Some studies have found that women with GDM have elevated triglycerides, LDL, and total cholesterol (CHOL), while HDL levels are decreased [24, 25]. A meta-analysis of 4168 women with GDM and 9718 healthy pregnant women showed that compared to women with standard glucose tolerance, those with GDM had higher triglyceride levels and lower HDL levels. However, CHOL and LDL levels were similar between the two groups [26]. Our study also found that high triglyceride levels are a risk factor for LGA in women with HIP (OR = 1.25, p = 0.005), in line with the findings of Shi et al. [27, 28, 29]. A possible reason could be that excess triglycerides in the mother are absorbed, metabolized by the placenta, and transported to the fetus in various forms, leading to excessive fetal growth. However, it remains unclear whether abnormal lipid levels in the blood directly affect fetal body mass.
Additionally, our study found that CHOL was a protective factor against LGA in women with HIP (OR = 0.77, p = 0.011). In contrast, Kaneko et al. [30] found that among Japanese mothers without GDM who delivered full-term babies, lower and higher maternal CHOL levels in the second trimester were associated with small-for-gestational-age (SGA) and LGA infants, respectively. In contrast, Shi et al. [27] found no significant correlations between CHOL or LDL concentrations and perinatal outcomes, including LGA, in either GDM or non-GDM women. These varying research findings may be attributed to pre-pregnancy differences in BMI, age, diet, region, and race. Our study also identified LDH as a risk factor for LGA in women with HIP (OR = 1.01, p = 0.010). There is still only limited domestic and international research on the relationship between LDH and LGA. Increased LDH activity indicates metabolic disturbances in glucose utilization and lactate production, which can lead to diabetic complications [31]. This may explain why elevated LDH levels contribute to LGA in pregnant women with HIP.
Human serum albumin (HSA) plays a crucial role in various physiological functions and is vital for maintaining good health. Hypoalbuminemia occurs due to a combination of inflammation and insufficient nutritional intake, and is commonly observed in patients with chronic diseases. Inflammation is closely associated with vascular diseases and can result in damage to the vascular endothelium [32]. Clinical and biomedical research has indicated a positive regulatory cycle between hyperglycemia and interstitial inflammation. Hyperglycemia triggers the synthesis of proinflammatory cytokines, chemokines, and adipokines in both the placenta and peripheral blood, ultimately affecting fetal growth [33]. In the present study, higher serum albumin levels were inversely associated with the risk of LGA in women with HIP (OR = 0.86, p = 0.002). To our knowledge, this specific association has not been reported previously, although study has suggested that albumin may modulate systemic inflammation and oxidative stress [34]. It is therefore plausible that better-preserved albumin levels reflect a more favorable metabolic and inflammatory profile, which might contribute to a lower risk of LGA. However, this finding should be interpreted with caution and confirmed in future studies.
Several authors have investigated the association between GDM and homocysteine (HCY), but the findings have so far been inconsistent. Liu et al. [35] found that HCY levels in women with GDM were significantly higher than those in women with standard glucose tolerance. On the other hand, Radzicka et al. [36] found no significant correlation between HCY levels and glucose tolerance. In a study of 7587 participants from the Ottawa and Kingston birth cohorts, maternal HCY concentration was found to be associated with a higher incidence of placental-mediated complications, including SGA, preeclampsia, and placental abruption [37]. Our research found similar results, suggesting that HCY acts as a protective factor against LGA (OR = 0.83, p = 0.004). This may be attributed to the involvement of HCY in inflammatory reactions, leading to dysfunction of maternal and placental vascular endothelial cells [38], which in turn affects fetal growth and development.
Urea nitrogen is a byproduct of protein metabolism and is excreted by the kidneys. Previous studies have shown that urea nitrogen is an independent risk factor for GDM [39]. However, it remains unclear whether BUN (blood urea nitrogen) can cause LGA. The current study found that BUN was a protective factor for LGA in pregnant women with HIP (OR = 0.65, p = 0.009). A possible explanation is that the hyperglycemic state leads to glycosylation of plasma and tissue proteins, impairing their normal function and reducing the excretion of urea nitrogen by the kidneys. Consequently, the level of BUN increases, and excessive amounts enter the placenta, thereby affecting the growth and development of the fetus.
In our model, FBG measured at 25–29+6 weeks was retained as an independent predictor of LGA. Although fasting glucose is part of the diagnostic workup for HIP, the measurement used in this study reflects glycemic status at mid-pregnancy, following the diagnosis and start of lifestyle management or insulin therapy in many women, rather than the diagnostic OGTT value per se. We were unable to include HbA1c as an additional marker of longer-term glycemic control because it was not routinely measured in all pregnant women with HIP during the study period. Future studies with more complete information on HbA1c and longitudinal glucose profiles may help to clarify the relative contributions of chronic versus short-term glycemic control to the risk of LGA.
HCY emerged as a significant predictor in our final model. HCY is measured as part of routine biochemical testing during mid-pregnancy at our hospital, thus facilitating its inclusion in the present analysis. However, testing for HCY is not universally available in all antenatal care settings. This may limit the direct transferability of our nomogram to some institutions, and also highlights the need to evaluate simplified models that rely only on universally available predictors.
Our study has several limitations. First, because of the challenges in obtaining
comprehensive biochemical data during pregnancy, the sample size was limited to
675 women, and the number of LGA events was modest. We therefore used bootstrap
resampling rather than a split-sample approach for internal validation, and
external validation in independent cohorts is still required. Second, the
analysis was restricted to women who delivered at
Third, important clinical factors such as pre-pregnancy BMI, gestational weight gain, HbA1c, and detailed longitudinal information on self-monitored blood glucose and insulin use were not consistently available in the electronic database and could not be incorporated. Consequently, we were unable to formally quantify overall glycemic control during pregnancy or evaluate its interaction with the biochemical predictors, which may have led to residual confounding. Fourth, although several predictors in the final model (albumin, BUN, HCY) are biologically linked to endothelial dysfunction and hypertensive disorders of pregnancy, we could not reliably ascertain the occurrence of preeclampsia or other hypertensive disorders in this retrospective cohort. The observed associations should therefore be interpreted with caution, as they may partially reflect underlying placental pathology.
Fifth, we focused exclusively on LGA and did not analyze SGA or fetal growth restriction (FGR) as competing fetal growth outcomes. Some of the predictors associated with a lower risk of LGA in our model may in fact be markers of placental dysfunction and increased risk of SGA/FGR. Future studies with larger sample sizes are warranted to simultaneously evaluate predictors of both excessive and restricted fetal growth in women with HIP. Finally, to handle multicollinearity, we used a simple, correlation-based strategy to choose between pairs of highly correlated predictors. Although this approach is transparent and easy to implement, it may not be optimal from a predictive modeling perspective. Instead, penalized regression methods such as LASSO or elastic net could be explored in future work to further refine variable selection.
Based on analysis of a retrospective cohort of 675 women with HIP, we developed and internally validated a nomogram that estimates the risk of delivering an LGA infant using only maternal height and routinely measured biochemical indicators at 25–29+6 weeks of gestation. The model demonstrated acceptable discrimination and calibration, as well as favorable clinical utility in decision curve analysis. Because the predictors are easily available during routine care, this tool may facilitate the early identification of high-risk women and support individualized antenatal management. External validation in larger, multicenter cohorts is warranted to confirm the generalizability of our model and refine its clinical application.
The datasets used and analyzed in the current study are available from the corresponding author upon reasonable request.
YCH, YL, XJ, XJK, FL, FLS, PZ, RSW contributed to the study concept and design. YL and XJ collected the data. RSW participated in data processing and analysis. YCH wrote the first draft of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This retrospective study was conducted in accordance with the principles of the Declaration of Helsinki. This study protocol was approved by the ethical committee of Maternal and Child Health Hospital Affiliated to Anhui Medical University, with ethical approval numbers YYLL2022-2108085MH260-03-01. The requirement for written informed consent was waived for this retrospective study due to the complete anonymization of all patient clinical data.
The authors wish to thank all the staff members of the Department of Obstetrics and Gynecology at Anhui Maternal and Child Health Care Hospital for their strong support of this study.
This work was supported by the Anhui Province Key Research and Development Project (202204295107020050), Anhui Province Key Research and Development Project (No.201904a07020032), and the Higher Education Science Research Project of Anhui Province (NO. 2023AH053400).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









