1 Center of Training & Direction of Healthcare Activities, Hanoi Obstetrics and Gynecology Hospital, 100000 Hanoi, Vietnam
2 Department of Obstetrics & Gynaecology, University of Medicine and Pharmacy, Vietnam National University, 100000 Hanoi, Vietnam
3 Department of Gynecological Oncology, Hanoi Obstetrics and Gynecology Hospital, 100000 Hanoi, Vietnam
4 Department of Global Public Health, Karolinska Institute, S-17177 Stockholm, Sweden
5 Department of Obstetrics & Gynaecology, Hanoi Medical University, 100000 Hanoi, Vietnam
6 Now with: Center of Regenerative Medicine and Cell Therapy, Vinmec Healthcare System, 100000 Hanoi, Vietnam
Abstract
Hydatidiform mole (HM) is a benign trophoblastic disease in which early diagnosis, timely treatment, and strict adherence to posttreatment follow-up are essential to achieve remission and ensure early detection of gestational trophoblastic neoplasia (GTN). Reports on HM in Vietnam remain limited and inconsistent. This study aimed to describe the incidence and clinical features of HM and to analyze factors associated with mole subtype, treatment, and follow-up compliance at a tertiary obstetrics and gynecology hospital in Hanoi.
A retrospective case study was conducted among patients diagnosed and treated at Hanoi Obstetrics and Gynecology Hospital between January 1, 2020, and December 31, 2021. Demographic, clinical, and diagnostic data were collected from medical records. Descriptive analyses characterized the study population, and univariate and multivariate logistic regression models identified factors associated with treatment decisions and follow-up compliance.
A total of 205 patients were diagnosed and treated for HM, corresponding to an incidence of 3.6 per 1000 deliveries. In 50 cases, histopathology confirmed HM without subclassification; therefore, only the remaining 155 patients were included in subgroup and association analyses. Complete hydatidiform moles (CHM) and partial hydatidiform moles (PHM) accounted for 71.6% and 28.4%, respectively. Maternal age showed a strong association with mole type: CHM predominated in women aged ≥40 years, whereas PHM occurred more frequently in younger patients. Invasive moles occurred in 16.1% of cases. Hysterectomy was performed in 29.0% of patients, and 34.2% received methotrexate (MTX) chemotherapy. Factors associated with hysterectomy included maternal age ≥40 years, CHM, and a pre-evacuation β-human chorionic gonadotropin (β-hCG) level ≥100,000 IU/L. Prophylactic chemotherapy was also correlated with older age, CHM, and invasive mole. Only 54% of patients completed follow-up with at least three consecutive negative β-hCG measurements, and chemotherapy administration was a significant factor associated with improved compliance.
Posttreatment follow-up completion among patients with HM remains suboptimal, which may explain the relatively high rates of surgical and chemotherapeutic interventions observed. Strengthening patient education, surveillance, and adherence to follow-up protocols is crucial for improving treatment outcomes for molar pregnancy in Vietnam.
Keywords
- hydatidiform moles
- β-hCG
- hysterectomy
- chemotherapy
- follow-up completion
Hydatidiform moles (HM), also known as molar pregnancy, encompass both complete and partial forms and represent a benign subset of gestational trophoblastic diseases (GTDs) that arise from abnormal fertilization and aberrant proliferation of placental tissue, predominantly of androgenetic origin. These conditions are characterized by villous hydrops and trophoblastic hyperplasia, with or without embryonic development [1, 2]. HM can be divided into two morphologically and genetically distinct groups: complete hydatidiform mole (CHM) and partial hydatidiform mole (PHM). The risk of progression to gestational trophoblastic neoplasia (GTN) after HM depends on the histological type, with rates of approximately 15–20% in CHM and only 1–5% in PHM [3].
The global incidence of HM shows marked geographic variation, with rates generally higher in Asia than in Europe or North America. The lowest incidence is reported in Europe, North America, Australia, and New Zealand, with rates ranging from 0.57 to 1.1 per 1000 pregnancies. In contrast, studies from Asian countries demonstrate greater variability, with reported incidences of 1.5–3 per 1000 in Japan, 1.6–4.4 in South Korea, 0.81 in China, 2.8 in Malaysia, 1.2 in Singapore, 2.4 in the Philippines, and 1.7 in Thailand [3, 4]. In Vietnam, available data indicate a notably higher incidence, reaching up to 11 per 1000 births, suggesting potential regional or methodological differences [5].
Suction evacuation under ultrasound guidance remains the standard treatment for molar pregnancy, whereas hysteroscopic removal of residual tissue serves as an adjunct option. Hysterectomy with ovarian preservation may be considered for women who have completed childbearing, whereas medical induction and hysterotomy are discouraged due to increased morbidity and risk of GTN. Prophylactic chemotherapy can reduce the incidence of GTN but should be limited to high-risk cases [4].
Beta-human chorionic gonadotropin (
Hormonal contraception is considered safe during follow-up, and a structured program incorporating nursing care, psychosocial support, and telemedicine-based monitoring can improve patients’ compliance, thereby optimizing patient care and preventing dropouts and delays in the diagnosis and treatment of complications [9].
This study aimed to describe the incidence and clinical features of HM and to analyze factors associated with mole subtype, treatment, and follow-up compliance at a tertiary obstetrics and gynecology hospital in Hanoi, northern Vietnam. The findings of this study provide updated epidemiological and clinical data from a tertiary referral center in Northern Vietnam and offer region-specific insights into the diagnosis, management, and long-term follow-up of patients with HM.
This retrospective case study was conducted at Hanoi Obstetrics and Gynecology Hospital, a tertiary referral center in northern Vietnam, from January 1, 2020, to December 31, 2021. Inclusion criteria were as follows: (1) confirmed diagnosis and management of HM at the institution; (2) availability of complete clinical and pathological records; and (3) absence of metastasis at diagnosis. Exclusion criteria were as follows: (1) prior management of HM at a referring facility; and (2) coexisting malignancy or autoimmune disease.
The study was conducted at Hanoi Obstetrics and Gynecology Hospital, one of the
largest tertiary referral centers for maternal health in northern Vietnam. The
hospital manages a high volume of GTD cases annually, with most patients being
referred from primary and secondary care facilities. Management of HM at this
center follows a standardized protocol that includes uterine evacuation under
ultrasound guidance, histopathological confirmation, and serial postevacuation
Serum
Data extracted from medical records included demographic characteristics,
clinical and paraclinical findings, pre- and post-treatment
Study data were collected and managed using the Research Electronic Data Capture (REDCap) software (version 10.8.3; Vanderbilt University, Nashville, TN, USA), hosted at Hanoi Obstetrics and Gynecology Hospital, and were analyzed using SPSS software (version 20.0; International Business Machines Corporation, Armonk, NY, USA).
Continuous variables were presented as mean
Univariate and multivariate logistic regression models were used to evaluate
factors associated with pathological HM, treatment methods, and complete
compliance with
Multivariate analysis was performed to account for potential confounders and
imbalanced covariates. Variables with p
For multivariate logistic regression analyses, variables with p
Statistical analyses were performed using SPSS version 20.0. A two-sided
p
During the study period, 223 cases with an initial diagnosis of HM were
identified among 57,203 deliveries at the institution. After exclusion of cases
not confirmed and not treated as HM, 205 cases met the inclusion criteria and
were used to calculate the incidence rate, corresponding to 3.6 per 1000
deliveries over the two-year study period. Among these, 50 cases lacked
histopathological subclassification and were therefore excluded from subgroup
analyses. The remaining 155 women were eligible for classification-based
correlation analysis (Fig. 1). Of these, 17 patients had missing anemia data at
T0 and 12 patients had missing first
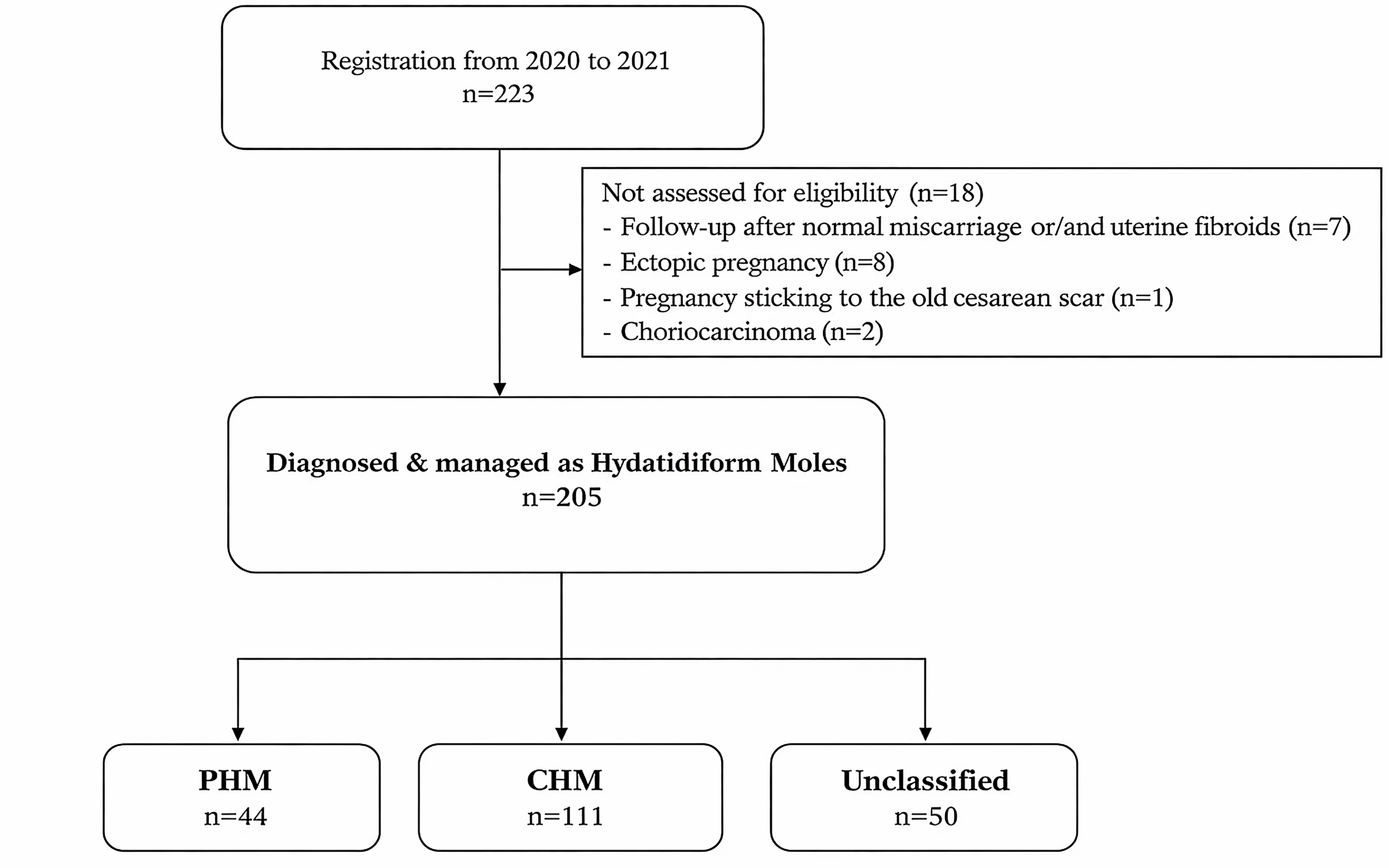 Fig. 1.
Fig. 1.
Flowchart of patient selection and inclusion in the study. CHM, complete hydatidiform mole; PHM, partial hydatidiform mole.
| Variables | HM (n = 155) | PHM (n = 44) | CHM (n = 111) | p-value | |
| Mean age (SD), years | 34.5 (10.2) | 29.8 (8.1) | 36.3 (10.4) | ||
| Age groups, n (%) | |||||
| Under 30 years | 65 (42.0) | 28 (63.6) | 37 (33.3) | ||
| 30–39 years | 36 (23.2) | 10 (22.7) | 26 (23.4) | ||
| Over 39 years | 54 (34.8) | 6 (13.6) | 48 (43.2) | ||
| Job, n (%) | |||||
| General labor | 53 (34.0) | 14 (31.8) | 39 (35.1) | 0.995 | |
| Educated jobs | 63 (40.4) | 20 (45.5) | 43 (38.7) | ||
| Others | 39 (25.6) | 10 (22.7) | 29 (26.1) | ||
| Symptom, n (%) | |||||
| Vaginal bleeding | 67 (42.9) | 11 (25.0) | 56 (50.5) | 0.004 | |
| Delayed menstruation | 75 (48.1) | 20 (45.5) | 55 (49.5) | 0.646 | |
| Abdominal pain | 27 (17.3) | 4 (9.1) | 23 (20.7) | 0.103* | |
| Menorrhagia | 17 (10.9) | 2 (4.5) | 15 (13.5) | 0.154* | |
| Preclinical examination, n (%) | |||||
| Typical HM images (Ultrasound) | 92 (59.0) | 25 (56.8) | 67 (60.4) | 0.686 | |
| Larger uterus (Ultrasound) | 50 (32.1) | 12 (27.3) | 38 (34.2) | 0.403 | |
| A corpus luteum cyst (Ultrasound) | 10 (6.4) | 2 (4.5) | 8 (7.2) | 0.726* | |
| Anemia (blood test)⁑ | 39 (28.3) | 9 (24.3) | 30 (29.7) | 0.534 | |
| Median |
|||||
| Pretherapeutic (T0) | 136,530 (73,309–295,031) | 124,466 (54,806–202,673) | 155,405 (77,886–359,438) | 0.037† | |
| First |
2887 (969–8760) | 3321 (689–9885) | 2841 (1143–8072) | 0.838† | |
| Invasive HM, n (%) | |||||
| Yes | 25 (16.1) | 1 (2.3) | 24 (21.6) | 0.003* | |
| No | 130 (83.9) | 43 (97.7) | 87 (78.4) | ||
| Treatment, n (%) | |||||
| Evacuation | 110 (71.0) | 42 (95.5) | 68 (61.3) | ||
| Hysterectomy | 45 (29.0) | 2 (4.5) | 43 (38.7) | ||
| Chemotherapy, n (%) | |||||
| No | 102 (65.8) | 40 (90.9) | 62 (55.9) | ||
| Yes | 53 (34.2) | 4 (9.1) | 49 (44.1) | ||
*Fisher’s Exact Test; †Mann-Whitney U Test;
⁑Missing data at T0 for 17 patients (7 PHM and 10 CHM); valid cases (n = 138);
ºMissing data at first
| Variables | Univariate analysis | Multivariate analysis | |||
| OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| Age (years) | |||||
| 1 | 1 | ||||
| 114.3 (30.7–426.2) | 499.5 (42.0–5941.6) | ||||
| Pathological report | 0.001 | 0.010 | |||
| PHM | 1 | 1 | |||
| CHM | 13.3 (3.1–57.7) | 25.1 (1.9–327.3) | |||
| Vaginal bleeding | 0.110 | ||||
| No | 1 | 1 | |||
| Yes | 5.3 (2.5–11.3) | 4.0 (0.7–21.6) | |||
| Delayed menstruation | 0.007 | 0.200 | |||
| No | 1 | 1 | |||
| Yes | 0.4 (0.2–0.8) | 0.3 (0.1–1.7) | |||
| Abdominal pain | 0.940 | / | |||
| No | 1 | / | |||
| Yes | 1.0 (0.4–2.6) | / | |||
| Menorrhagia | 0.007 | 0.770 | |||
| No | 1 | 1 | |||
| Yes | 4.2 (1.5–11.9) | 1.4 (0.1–16.1) | |||
| Typical HM images | 0.124 | / | |||
| No | 1 | / | |||
| Yes | 1.8 (0.9–3.7) | / | |||
| Enlarged uterus | 0.090 | / | |||
| No | 1 | / | |||
| Yes | 1.9 (0.9–3.8) | / | |||
| Corpus luteum cyst | 0.201 | / | |||
| No | 1 | / | |||
| Yes | 0.3 (0.1–2.1) | / | |||
| Anemia (Blood test)⁑ | 0.006 | 0.160 | |||
| No | 1 | 1 | |||
| Yes | 3.0 (1.4–6.5) | 4.7 (0.5–40.4) | |||
| 0.015 | 0.030 | ||||
| 1 | 1 | ||||
| 2.8 (1.2–6.3) | 7.5 (1.2–47.2) | ||||
⁑Missing data at T0 for 17 patients; valid cases (n = 138);
OR, odds ratio; CI, confidence interval.
| Variables | Univariate analysis | Multivariate analysis | ||||
| OR (95% CI) | p-value | OR (95% CI) | p-value | |||
| Age (years) | 0.013 | |||||
| 1 | 1 | |||||
| 7.3 (3.5–15.4) | 3.3 (1.3–8.5) | |||||
| Pathological report | 0.011 | |||||
| PHM | 1 | 1 | ||||
| CHM | 7.9 (2.6–23.6) | 6.6 (1.6–28.4) | ||||
| Invasive mole | 0.003 | |||||
| No | 1 | 1 | ||||
| Yes | 23.4 (6.6–83.6) | 15.9 (2.6–97.2) | ||||
| Vaginal bleeding | 0.001 | 0.508 | ||||
| No | 1 | 1 | ||||
| Yes | 3.3 (1.7–6.6) | 1.4 (0.5–3.5) | ||||
| Delayed menstruation | 0.026 | 0.956 | ||||
| No | 1 | 1 | ||||
| Yes | 0.5 (0.2–0.9) | 0.9 (0.3–2.5) | ||||
| Abdominal pain | 0.732 | / | ||||
| No | 1 | / | ||||
| Yes | 1.2 (0.5–2.8) | / | ||||
| Menorrhagia | 0.331 | / | ||||
| No | 1 | / | ||||
| Yes | 0.6 (0.2–1.8) | / | ||||
| Preclinical examination | ||||||
| Typical HM images (Ultrasound) | 0.119 | / | ||||
| No | 1 | / | ||||
| Yes | 1.7 (0.9–3.5) | / | ||||
| Larger uterus (Ultrasound) | 0.078 | / | ||||
| No | 1 | / | ||||
| Yes | 1.9 (0.9–3.8) | / | ||||
| Corpus luteum cyst (Ultrasound) | 0.773 | / | ||||
| No | 1 | / | ||||
| Yes | 0.8 (0.2–3.3) | / | ||||
| Anemia (blood test)⁑ | 0.008 | 0.061 | ||||
| No | 1 | 1 | ||||
| Yes | 2.8 (1.3–6.0) | 3.1 (0.9–9.8) | ||||
| 0.585 | / | |||||
| 1 | / | |||||
| 0.8 (0.4–1.6) | / | |||||
| 0.002 | 0.453 | |||||
| 1 | 1 | |||||
| 3.0 (1.5–5.9) | 0.6 (0.2–2.0) | |||||
⁑Missing data at T0 for 17 patients; valid cases (n = 138).
Among 155 patients with HM, CHM accounted for 111 (71.6%) cases and PHM for 44
(28.4%), while invasive moles occurred in 16.1% of these patients. Comparison
between the two groups revealed distinct demographic and clinical patterns. The
mean age of patients with CHM was significantly higher than that of patients with
PHM (36.3
Univariate logistic regression identified significant predictors of
hysterectomy, including age
Chemotherapy was administered in 34.2% of patients. Univariate analysis
identified significant associations with age
During the follow-up period, 54% of patients achieved complete monitoring,
defined as at least three consecutive negative
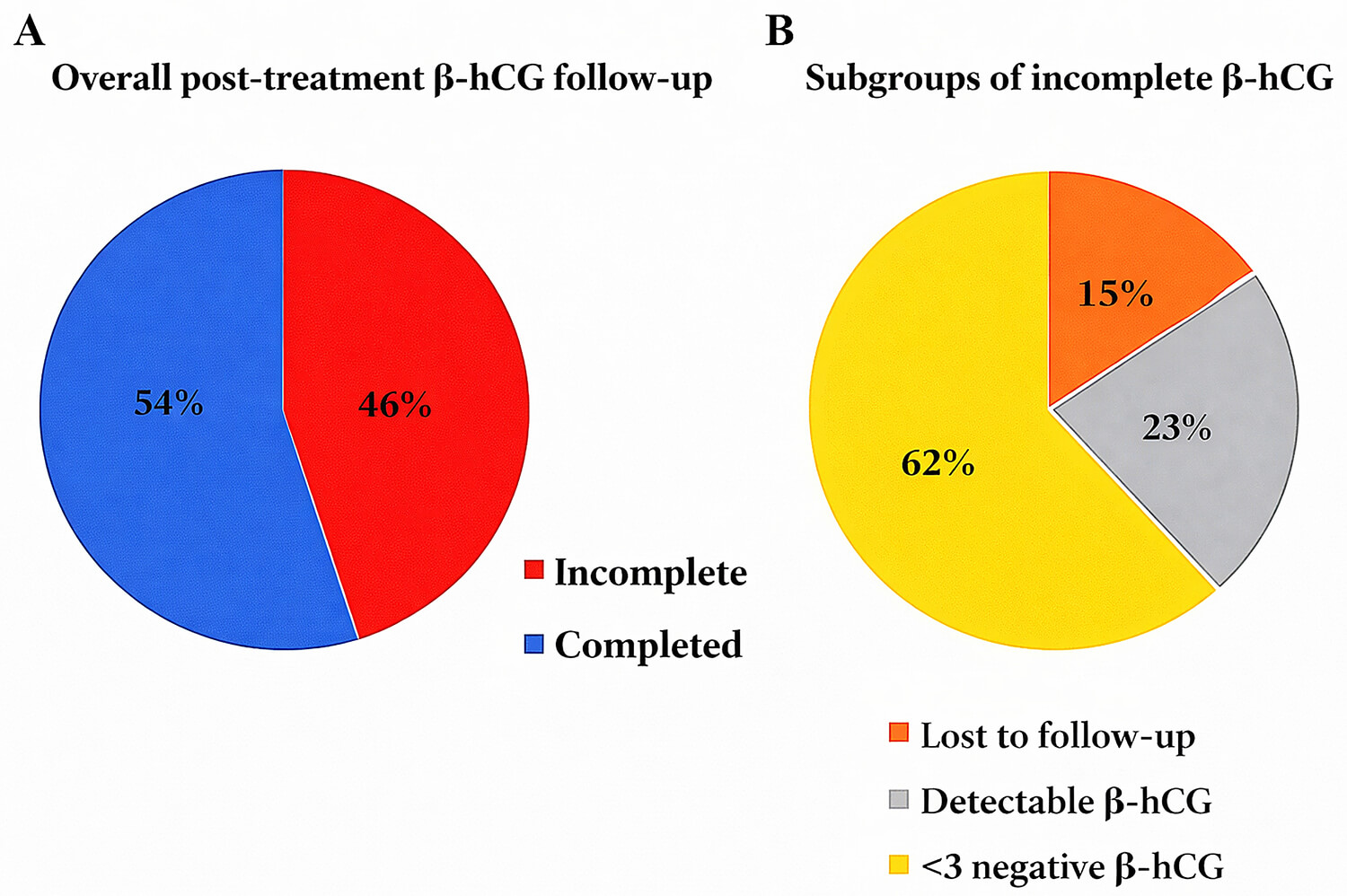 Fig. 2.
Fig. 2.
Posttreatment
Univariate analysis showed that age
| Variables | Univariate analysis | Multivariate analysis | |||
| OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| Age (years) | 0.010 | 0.462 | |||
| 1 | 1 | ||||
| 2.5 (1.2–5.0) | 1.6 (0.5–5.2) | ||||
| Type of HM | 0.016 | 0.357 | |||
| PHM | 1 | 1 | |||
| CHM | 2.4 (1.2–5.0) | 1.5 (0.7–3.2) | |||
| Invasive HM | 0.056 | / | |||
| No | 1 | / | |||
| Yes | 2.5 (0.98–6.4) | / | |||
| Hysterectomy | 0.020 | 0.543 | |||
| No | 1 | 1 | |||
| Yes | 2.4 (1.1–5.0) | 0.7 (0.2–2.5) | |||
| Chemotherapy | |||||
| No | 1 | 1 | |||
| Yes | 6.4 (2.9–14.2) | 5.7 (2.3–14.0) | |||
This study provides an updated overview of the epidemiology, clinical features, treatment patterns, and follow-up outcomes of HM at a tertiary obstetrics and gynecology hospital in northern Vietnam. The findings contribute new evidence on the contemporary management of HM in a middle-income country setting, highlighting key determinants of treatment choice and adherence to postmolar surveillance.
In this study, the incidence of HM at Hanoi Obstetrics and Gynecology Hospital was 3.6 per 1000 deliveries, which is lower than earlier reports from Vietnam but consistent with findings from other Asian countries. The incidence of HM varies globally, with rates of 0.66 to 1.21 per 1000 pregnancies in Europe and North America and 1.5–4.4 per 1000 in East and Southeast Asia [3, 4]. Differences across studies likely reflect variations in genetic background, nutritional status, and diagnostic methods. Previous Vietnamese data from a national referral hospital in southern Vietnam reported a higher incidence, up to 11 per 1000 births [5], compared with the rate observed in the present study. However, the lower rate observed here may be partly explained by differences in delivery volume, disease patterns, and regional context, as this institution, one of the largest obstetric and gynecologic centers in northern Vietnam, records more than 40,000 births annually in the pre-Coronavirus Disease 2019 (COVID-19) period. Our study also showed that CHM accounted for 72% of all HM cases, similar to domestic studies [5], but higher than rates reported in Europe, North America, and other developed Asian regions, where CHMs occur in approximately 1 to 3 per 1000 pregnancies and PHMs in about 3 per 1000 [11]. Across developed countries, variations in CHM and PHM proportions have been reported. In Japan, CHM accounted for 46.5% of cases between 1992 and 2018, rising to 58.8% in recent years [12]. In the United States, CHM and PHM were nearly equal (53% vs. 47%) [13]. In Sweden, improved diagnostic techniques have increased PHM detection [14]. Taken together, these findings suggest that although the overall incidence of HM in Vietnam remains higher than in developed countries, the apparent predominance of CHM may reflect underdiagnosis of PHM, as many partial moles are misclassified as spontaneous or missed abortions without histopathologic confirmation.
The widespread use of ultrasonography has facilitated earlier detection of molar
pregnancy, often before overt clinical manifestations appear. Consequently, the
frequency of clinical symptoms such as vaginal bleeding or abdominal pain was
lower in this study than in earlier reports. CHM occurred more frequently in
older women, whereas PHM predominated in younger patients, a distribution pattern
also observed in Japan, Korea, and Thailand [3, 4]. The strong association between
maternal age and CHM may be related to age-dependent oocyte abnormalities and
defects in genomic imprinting, which can lead to androgenetic conceptions [7]. In
our cohort, patients with CHM were generally older, presented more frequently
with vaginal bleeding, exhibited higher pre-evacuation
Most molar pregnancies regress after uterine evacuation, yet a small proportion invade the myometrium, which may lead to uterine perforation or extension to adjacent structures. An invasive mole is typically defined histologically as trophoblastic invasion of the myometrium and uterine vessels, most often confirmed after hysterectomy [17, 18, 19]. In recent years, the preference for uterine preservation has increased, which has led to a lower reported incidence of invasive mole relative to overall molar pregnancies. In our study, invasive mole was identified in 25 cases (0.45 per 1000 deliveries per year). Of these, 23 were confirmed histopathologically following hysterectomy, and two were diagnosed based on postevacuation histology combined with ultrasonographic evidence of myometrial invasion. The relatively higher incidence observed in our cohort likely reflects a higher hysterectomy rate, which allows more histopathologic confirmation, as well as improved detection practices. By contrast, a large population-based study from Sweden (1994–2013) reported an incidence of only 0.01 case per 1000 deliveries [20], which reflects the rarity of invasive mole in settings with comprehensive follow-up and widespread uterine preservation. Notably, nearly all invasive moles in our cohort arose from CHM, consistent with previous studies [21, 22].
Vacuum aspiration remains the first-line treatment for HM, whereas hysterectomy
is reserved for women aged
Regarding prophylactic chemotherapy, 34.2% of patients received methotrexate
(MTX) monotherapy. Independent risk factors associated with chemotherapy
administration included maternal age
Follow-up compliance remains a key determinant of patient outcomes in HM. In
this cohort, only 54% of patients completed posttreatment monitoring with three
consecutive undetectable
The present study provides updated evidence on the clinical management and follow-up of patients with HM at a tertiary referral center in Northern Vietnam. Several important findings merit emphasis. First, the incidence of HM in our cohort (3.6 per 1000 deliveries) was substantially lower than that reported in earlier Vietnamese and regional studies. Second, posttreatment follow-up compliance was suboptimal, which likely reflects the absence of a centralized and structured follow-up management system. Third, this limitation in follow-up care may have influenced clinical decision-making, particularly in women aged 40 years and older, and may have contributed to the relatively high reliance on definitive surgical treatment, such as hysterectomy, in this age group.
Nevertheless, several limitations should be acknowledged. As a single-center
retrospective study, the findings may not be fully generalizable. Data
completeness and follow-up adherence may have been affected by disruptions during
the COVID-19 pandemic. In addition,
In this single-center study, the incidence of HM was lower than that reported in
previous Vietnamese studies. Age
The raw data of this study are deposited to REDCap (https://redcap.benhvienphusanhanoi.vn/) and will be publicly available as of the date of publication. All data reported in this paper will also be shared by the lead contact upon request.
MTH and TTVT designed the research study. MTH performed the research. LTKL collected the data. NMT, NDP, NCC, NKT, VTTH, NCD, NDL, and VTV contributed to study design, clinical data interpretation, and critical revision of the manuscript. MTH analyzed the data. MTH and TTVT developed the manuscript. All authors contributed to editorial revisions of the manuscripts. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki. Ethical approval was obtained from the Ethics Committee of Hanoi Obstetrics and Gynecology Hospital (IRB code: IRB-VN02.030; approval No. CS/PSHN/DC/23/21). As this study was a retrospective review of anonymized medical records, the Ethics Committee waived the requirement for written informed consent.
We gratefully acknowledge Ms. Ha Nguyen Thi and Mr. Anh Nguyen Xuan for their support in data collection at Hanoi Obstetrics and Gynecology Hospital.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT-5.1 to support English language editing. After using this tool, the authors reviewed and edited the content as needed and took full responsibility for the final version of the manuscript.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG48315.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


